Abstract
Objective
Our objectives were to evaluate trends in percutaneous endoscopic gastrostomy tube placement rate and timing in acute stroke patients. We hypothesized that non-compliance with clinical practice guidelines for timing of tube placement and an increase in placement has occurred in concordance with a decreased length of hospital stay.
Methods
We conducted a retrospective observational study of archival hospital billing data from the Florida state inpatient healthcare cost and utilization project database from 2001 to 2012 for patients with the primary diagnosis of stroke. Outcome measures: timing of percutaneous endoscopic gastrostomy tube placements by year (2006 to 2012), rate of placements by year (2001 to 2012), length of hospital stay. Univariate analyses, simple and multivariable logistic regression analyses were conducted.
Results
The timing of gastrostomy tube placement remained stable with a median of 7 days post admission from 2006 through 2012. The proportion of tubes that were placed at or after 14 days and, thereby met the guideline recommendations, varied from 14.09% in 2006 to 13.41% in 2012. The rate of tube placement in stroke patients during the acute hospital stay decreased significantly by 25% from 6.94% in 2001 to 5.22% in 2012 (p<0.0001). Length of hospital stay for all stroke patients decreased over the study period (p<0.0001).
Conclusions
The vast majority of percutaneous endoscopic gastrostomy tube placements happen earlier than clinical practice guidelines recommend. Over the study period, the rate of tubes placed in stroke patients decreased during the acute hospital stay despite an overall reduced length of stay.
Keywords: Stroke, Gastrostomy, Clinical Practice Pattern, Guideline Adherence
Introduction
Percutaneous Endoscopic Gastrostomy (PEG) tubes are used to support nutrition and hydration in acute stroke patients. Commonly considered a low risk procedure, PEG tubes, however, are associated with short- and long-term complications [1-6]. Evidence exists linking PEG tube placement in the acute stage with worse outcomes, in terms of mortality, functional status and potential for tube removal [7]. Clinical practice guidelines, therefore, provide recommendations for the timing and indication of PEG tube placements in acute stroke patients. The American Heart Association/American Stroke Association (USA), the National Collaborating Centre for Acute Care (UK), the German Society for Clinical Nutrition (Germany) and the German Society for Neurology (Germany) all recommend PEG tube placement in stroke patients with a given medical indication after 14-28 days [8-11]. Further, the Scottish Intercollegiate Guidelines Network (Scotland), the German Society for Clinical Nutrition (Germany) and the German Society for Neurology (Germany) and the European Society for Clinical Nutrition and Metabolism (Europe) recommend placement of PEG tubes for patients with an anticipated prolonged need of enteral nutrition for more than 28 days [6, 10-12].
Compliance with these guidelines seems to be especially challenging with the last decade’s increased tendency for shorter hospitalizations due in part to widespread changes in the payment for healthcare. Previous research supports the hypothesis that a decrease in the average length of hospital stay (LOS) can lead to a changes in practice patterns [13]. In terms of PEG tube placements, one can speculate that a shorter LOS for stroke patients might increase the pressure on clinical decision making for PEG tube placement. In 2010, the average LOS for stroke patients was 6.1 days in the US [14] and, therefore, notably below the recommended timing for PEG tube placements of 14-28 days.
The aim of our study was to evaluate current practice and trends in PEG tube placement rate and timing in order to identify discrepancies with guideline recommendations and to identify the potential to improve health care for acute stroke patients. Considering that stroke patients are usually in the hospital less than a week, we hypothesized: 1) that the timing of PEG tube placement happens earlier than clinical practice guidelines recommend and 2) that placement occurs near the end of the hospital stay. We further hypothesized that an increase in PEG tube placement in acute stroke patients has occurred.
Methods
We conducted a retrospective analysis of hospital discharge billing data from the Healthcare Cost and Utilization Project (HCUP) Florida State Inpatient Database (SID) from 2001 to 2012. This dataset includes all inpatient discharge records from acute care community hospitals. Discharge records were included for patients with a primary diagnosis of stroke (ICD-9-CM codes of 434.xx for ischemic or 431.xx for hemorrhagic stroke). These codes and code 436.xx were proven to be most accurate and highly specific [15]. Since 2004, the code 436.xx was removed and re-indexed to code 434.91. Thus, we excluded 436.xx for all years for consistency, while accepting that stroke patients coded with 436.xx before 2004 may be underrepresented in our sample. PEG tube placement during a hospital stay should be coded through an ICD-9-CM procedure code, however in rare occasions a current procedure terminology (CPT) code might be used instead. Since CPT codes were not available in the analyzed database, we calculated the estimated amount of missed PEG tube placements by comparing CPT and ICD-9-CM procedure codes for PEG tube placement in Medicare data from 2012, which included both CPT and ICD-9-CM procedure codes. We found that in Medicare 2012, 2.5% of all PEG tube placements were missed by using ICD-9-CM procedure codes only. Acknowledging this as a limitation of our study, we felt comfortable identifying PEG tube placement in the HCUP databases through the ICD-9-CM procedure code 43.11 only, because we anticipated that we will be able to identify the vast majority, 97.5%, of all PEG tube placements.
In addition to the comparison of CPT and ICD-9 procedure codes, we assessed whether differences in the number of diagnosis and procedure codes between the years may have caused an ascertainment bias. The years 2001-2005 only included 10 diagnosis and 10 procedure codes, whereas 2006-2012 included 31 diagnosis and procedure codes. We compared the frequency of PEG tube placements by taking all 31 codes and by taking only the first 10 codes. The difference for the overall PEG placement rate when comparing these two approaches was between 0.04%-0.08% and, therefore, considered negligible.
Discharge records were analyzed for demographic characteristics (age, sex, race), stroke type (ischemic or hemorrhagic stroke), comorbidities (Charlson Comorbidity Index), death during hospital stay, and acute hospital length of stay (LOS) information. The Charlson Comorbidity Index is a validated method to predict 1-year mortality based on the presence and weighting of comorbid conditions as retrieved from medical chart reviews [16]. We used the enhanced, most recently updated, ICD-9-CM coding algorithms for the Charlson Comorbidity Index for which good-to-excellent discrimination in the prediction of in-hospital mortality has been shown [17, 18]. Patients who died during the hospital stay remained in the analyses. Outcome measures were (a) timing of PEG tube placement (number of days after admission, number of days before discharge, and timing of placement in relation to total LOS), and (b) frequency of PEG tube placement. As a limitation, the timing of PEG tube placement could only be assessed from 2006 to 2012, because information on the day of procedures has only been included since 2006.
Univariate analyses for demographic and clinical information and timing of PEG tube placement were conducted. We determined differences in variables with bivariate comparisons of the first (2001 or 2006) and last year (2012) with the Chi-Square Test or Wilcoxon Rank-Sum Test, where appropriate. Simple and multivariable logistic regression analyses determined the influence of the year as the main independent variable on the rate of PEG tube placement. To determine the final multivariable regression model, we used manual backward selection regression with increased Akaike Information Criterion and p-values of the covariates greater than 0.05 to determine final variable inclusion. To determine the impact of the covariates on the main predictor, year of placement, we examined the influence of the covariate by calculating the amount that the regression coefficient changed after adding the covariate. We defined each covariate that changed the regression coefficient of the variable year of placement by more than 20% as a confounding variable and defined variables that remained significant but changed the primary predictor estimate by less than 20% as a control variable. Analyses were conducted using SAS statistical software (version 9.3; SAS Institute, Inc., Cary, NC). Statistical significance was defined at 0.05. The study was reviewed by our institutional review board as not-human research.
Results
Patient Characteristics
From a total of 30,458,243 discharge records available in the database, we analyzed the 334,273 discharge records from stroke patients amounting to approximately 30,000 records each year. The total number of stroke patients increased steadily from 2001 to 2012. Table 1 shows the demographic and clinical characteristics for all stroke patients. Overall, patients had small, but statistically significant differences in demographic characteristics across the years in regard to age, sex, race, and Charlson Comorbidity Index. The number of ischemic stroke patients increased, while the number of hemorrhagic stroke patients decreased from 2001 to 2012 (p<0.0001). Overall, the proportion of stroke patients that died in hospital showed a significant decrease of approximately 50% (p<0.0001). Further, the LOS for all stroke patients decreased from a median of 5 days in 2001 to 4 days in 2012 (p<0.0001) in contrast to an increase from 12 to 13 days in the LOS for stroke patients with PEG tube placement (p=0.0036).
Table 1.
Demographic and clinical characteristics for stroke patients with discharge records for each year of the analyzed Florida HCUP database
| 2001 | 2002 | 2003 | 2004 | 2005 | 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | Statistical difference between 2001 and 2012 |
|
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Total N stroke patients | 26,398 | 26,963 | 27,160 | 28,454 | 32,457 | 31,225 | 31,074 | 31,144 | 31,564 | 32,560 | 33,182 | 34,552 | NA |
| PEG tube placements N (%) | 1,833 (6.94) | 1,974 (7.33) | 1,803 (6.64) | 1,820 (6.4) | 1,927 (5.94) | 1,835 (5.93) | 1,853 (5.96) | 1,777 (5.71) | 1,782 (5.65) | 1,749 (5.37) | 1,808 (5.45) | 1,804 (5.22) | p<0.0001 |
| Age Mean (SD) | 72.71 (13.72) | 72.5 (13.9) | 72.08 (14.02) | 71.99 (14.38) | 71.96 (14.38) | 71.61 (14.55) | 71.33 (14.78) | 71.5 (14.66) | 71.34 (14.65) | 71.23 (14.76) | 71.26 (14.7) | 71.36 (14.74) | p<0.0001 |
| Female N (%) | 14,226 (53.89) | 14,370 (53.35) | 14,253 (52.48) | 15,035 (52.84) | 16,874 (51.99) | 16,238 (52) | 16,079 (51.74) | 16,072 (51.61) | 16,181 (51.26) | 16,578 (50.92) | 16,859 (50.81) | 17,740 (51.34) | p<0.0001 |
| White N (%) | 19,483 (73.8) | 19,582 (72.7) | 19,467 (71.68) | 20,160 (70.85) | 22,884 (70.51) | 22,116 (70.83) | 21,729 (69.93) | 21,503 (69.04) | 21,416 (67.85) | 21,706 (66.66) | 22,005 (66.32) | 22,678 (65.63) | p<0.0001 |
| Charlson Comorbidity Index (max. 32) Mean (SD) | 2.6 (1.6) | 2.63 (1.61) | 2.61 (1.61) | 2.6 (1.61) | 2.61 (1.63) | 2.84 (1.81) | 2.89 (1.85) | 2.97 (1.88) | 3.02 (1.9) | 3.02 (1.91) | 3.03 (1.91) | 3.04 (1.9) | p<0.0001 |
| Ischemic stroke N (%) | 21,655 (82.03) | 22,137 (82.18) | 22,317 (82.17) | 23,624 (83.03) | 27,462 (84.61) | 26,432 (84.65) | 26,342 (84.77) | 26,518 (85.15) | 26,977 (85.47) | 27,982 (85.94) | 28,838 (86.91) | 30,137 (87.22) | p<0.0001 |
| Hemorrhagic stroke N (%) | 4,743 (17.97) | 4,799 (17.82) | 4,843 (17.83) | 4,830 (16.97) | 4,995 (15.39) | 4,793 (15.35) | 4,732 (15.32) | 4,626 (14.85) | 4,587 (14.53) | 4,578 (14.06) | 4,344 (13.09) | 4,415 (12.78) | |
| LOS for all stroke patients Mean (SD); Median | 6.23 (6.32); 5 | 6.32 (7); 5 | 6.31 (6.89); 5 | 6.35 (7.96); 4 | 6.12 (7.5); 4 | 6.15 (7.44); 4 | 6.05 (7.79); 4 | 5.95 (8.35); 4 | 5.76 (6.92); 4 | 5.69 (7.25); 4 | 5.75 (8.32); 4 | 5.62 (8.39); 4 | p<0.0001 |
| LOS for stroke patients with PEG tube placement Mean (SD); Median | 15.56 (12.48); 12 | 15.8 (14.21); 12 | 16.65 (14.66); 13 | 17.44 (19.36); 13 | 17.55 (16.51); 13 | 17.98 (16.79); 13 | 18.18 (19.45); 13 | 18.26 (22.63); 13 | 17.23 (15.32); 13 | 18.33 (18.02); 14 | 18.42 (23.14); 13 | 19.24 (24.3); 13 | p=0.0036 |
| Patients died in hospital of all stroke patients N(%) | 2,932 (11.11) | 2,931 (10.88) | 2,823 (10.39) | 2,700 (9.49) | 2,782 (8.57) | 2,426 (7.77) | 2,300 (7.4) | 2,245 (7.21) | 2,169 (6.87) | 2,112 (6.49) | 1,926 (5.8) | 1,995 (5.77) | p<0.0001 |
N = Number, SD = Standard Deviation, LOS = Length of Stay, p = p-value
Timing of PEG tube placement
For all stroke patients, the day of PEG tube placement remained stable with a median of 7 days after admission each year (mean: day 8.83 in 2006 and day 9.12 in 2012; p=0.5007) (Figure 1). The proportion of PEG tubes that were placed at or after 14 days and, thereby, met guideline recommendations was on average 14.06% across the years (14.09% in 2006, 13.41% in 2012). Ischemic stroke patients received a PEG tube on day 7, median for all years (mean: day 7.9 in 2006 and day 8.3 in 2012; p=0.2024). On average 10.34% (9.99% in 2006, 10.14% in 2012) of PEG tube placements were conducted at or after 14 days. The LOS did not change significantly for ischemic stroke patients with PEG tube placement from an average of 14.99 days (median: 11) in 2006 to 16.19 days (median: 11) in 2012 (p=0.7228). On average, ischemic stroke patients remained in the hospital 7 to 8 (median: 4) days after PEG tube placement, as the PEG placement was conducted on average at 59% of the total hospital stay. Hemorrhagic stroke patients received a PEG tube on day 9 or 10, median for all years (mean: day 11.7 in 2006 and day 12.5 in 2012; p=0.6946). On average 25.31% (26.83% in 2006, 24.46% in 2012) of PEG tube placements were conducted at or after 14 days. The LOS for hemorrhagic stroke patients did not change significantly from a mean of 27.26 days (median: 20) in 2006 to a mean of 29.52 days (median: 19) in 2012 (p=0.2226). PEG tube placement was conducted in hemorrhagic patients at 54% of their hospital stay and they remained in the hospital approximately 15 days (median: 10) after the PEG tube placement.
Figure 1.
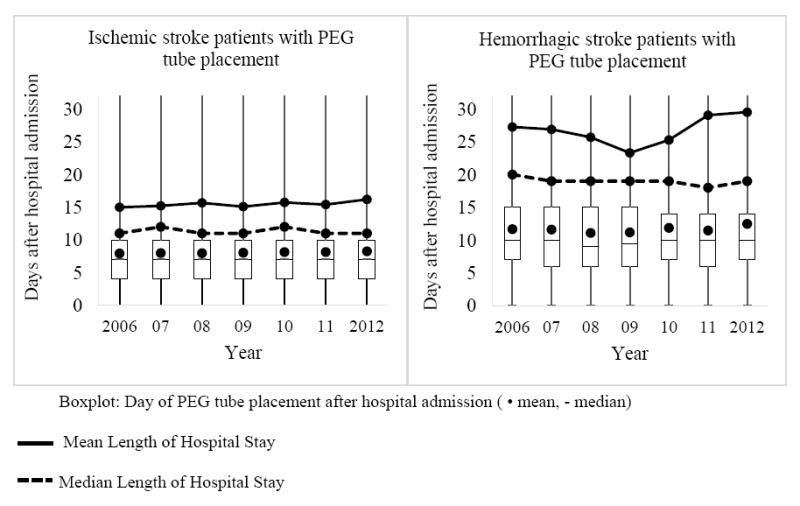
Day of PEG tube placement and length of stay for ischemic and hemorrhagic stroke patients with PEG tube placement.
PEG tube placement rate
The frequency of PEG tube placements in all stroke patients decreased significantly by 25% from 6.94% in 2001 to 5.22% in 2012 (p<0.0001). From 2001 to 2012, ischemic stroke patients showed a decrease of 28.8% from 6.49% to 4.62% (p<0.0001) and hemorrhagic stroke patients showed a non-significant slight increase of 3.8% from 9% to 9.35% (p=0.2923), respectively (Figure 2). After controlling for differences in demographic and clinical characteristics by multivariable logistic regression, both ischemic and hemorrhagic stroke patients showed a significant or near significant decrease, on average, in the odds that a PEG tube would be placed over the study time period (Odds Ratio (OR)=0.963, 95% confidence interval (CI) 0.958-0.968, p-value<0.0001; OR=0.991, CI=0.981-1.000, p-value=0.0586, respectively). Analyses for odds of PEG tube placement over the study period for all stroke patients combined (ischemic and hemorrhagic) were similar to those for ischemic stroke patients only (Table 2). The year that a patient was treated in the hospital had a significant influence on the likelihood that a PEG tube was placed. Variables such as demographics (age and race), comorbidities (Charlson Comorbidity Index), stroke type and died during the hospital stay significantly predicted odds of PEG placement. Of all control variables, only the variable Charlson Comorbidity Index had a confounding impact on our main predictor “year of placement” by changing its regression coefficient by more than 20% (29.7%).
Figure 2.
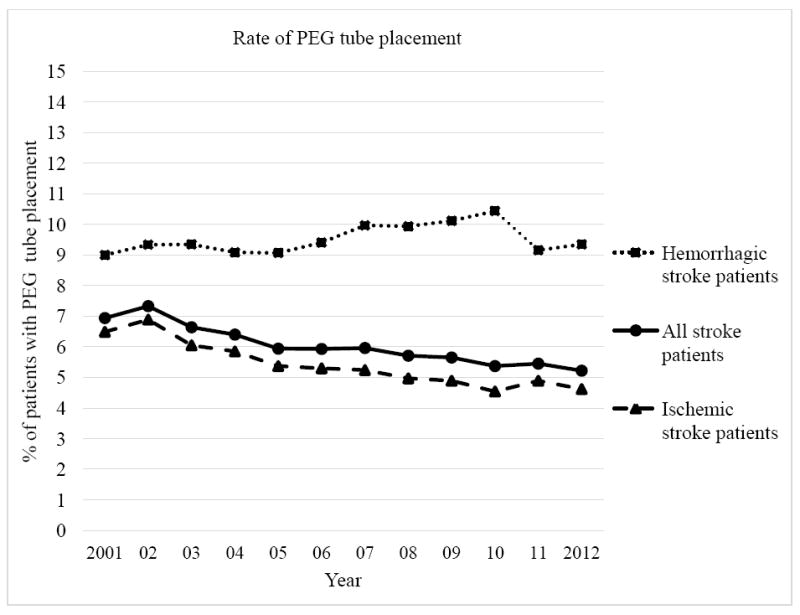
Trend of PEG tube placement rate for all, ischemic and hemorrhagic stroke patients.
Table 2.
Multivariable logistic regression model for the influence of the year on the event of PEG tube placement in stroke patients
| Variable | β | SE β | OR (95% CI) |
|---|---|---|---|
| Intercept | -4.1300 | 0.0433 | - |
| Year (2001 - 2012) | -0.0437 | 0.00208 | 0.957 (0.953-0.961) |
| Age | 0.0180 | 0.000544 | 1.018 (1.017-1.019) |
| White | -0.5116 | 0.0150 | 0.600 (0.582-0.617) |
| Charlson Comorbidity Index | 0.1882 | 0.00326 | 1.207 (1.199-1.215) |
| Hemorrhagic stroke | 0.7680 | 0.0172 | 2.155 (2.084-2.229) |
| Died during hospital stay | -0.5571 | 0.0286 | 0.573 (0.542-0.606) |
β =regression coefficient; SE=standard error; OR=odds ration; CI=confidence interval
Discussion
The aim of our study was to understand the timing and rate of PEG tube placement and if the timing and rate has changed over the last decade in relation to LOS. We found in Florida from 2001 to 2012 that 1) the timing of PEG tube placement was earlier than clinical practice guidelines recommend, 2) PEG tubes were not placed immediately before discharge and 3) fewer PEG tubes were placed in stroke patients during the hospital stay while the median LOS for patients with PEG placement remained stable. When conservatively taking 15 days as the recommended time post-stroke of PEG tube placement, ischemic stroke patients received a PEG tube about 8 days earlier and hemorrhagic stroke patients about 4-5 days earlier than recommended even though the average length of stay was 15.5 days (median 11) and 26.7 days (median 19), respectively. Across all years only 14.06% of PEG tubes were placed after 14 days and, thereby, met guideline recommendations.
Reasons for the early PEG tube placement remain speculative. For example, patients who do not tolerate NG tubes are in the need of an alternative feeding route and may receive PEG tube placements earlier. Further, an admittance preference for patients with PEG tubes over patients with NG tubes has commonly been reported [8, 19], which may result in an earlier decision to place PEG tubes in acute facilities to promote discharge. Also, in specific cases PEG placement needs to precede other medical interventions (such as Anticoagulation [20]). Last but not least, a simple lack of practitioners’ knowledge of guidelines or disagreement with the guidelines can be a reason for an early PEG tube placement. While PEG placement needs to be determined on an individual basis, our results suggest that there might be opportunities for a change in current practice patterns towards a better alignment with clinical practice guidelines by postponing PEG tube placement within the hospital stay. In order to achieve a better alignment with guidelines, we suggest to improving practitioner/clinician education regarding recommended PEG tube placement timing, considering changes in the admission policies for rehabilitation or skilled nursing facilities, and reconsidering reimbursement metrics. Further, a re-evaluation of the guidelines should be conducted regularly to assure a steady alignment with recommendations and the best available evidence.
Against our hypothesis, we found a decrease in the number of PEG tube placements over the last decade. The year a stroke patient was admitted to a hospital in Florida had a significant impact on the odds that a PEG tube was placed. This change may be due to differences in demographic and medical characteristics of stroke patients over the studied decade. We found that the number and severity of comorbidities had increased from 2001 to 2012 to such a degree that it was a confounding factor on the impact of the year on rate of PEG tube placement. An increase in comorbidities, such as diabetes mellitus, hypertension and cardiac diseases, has been previously described in patients hospitalized for a stroke [21, 22]. Whether the increased number of comorbidities is a reflection of a general trend in the population, of advances in diagnostic quality, or sequels of the stroke and its care cannot be distinguished in our data and remains to be determined [21].
The proportions of race and stroke type also changed over the studied decade with an increase in the proportion of minority race compared to white stroke patients and an increase in the proportion of hemorrhagic compared to ischemic stroke patients. Changes in race distribution have been described (a) for stroke patients with a decrease in stroke incidence in whites but not in blacks [23] and for the general population of Florida with a slight increase of minority race [24]. Changes in the proportional frequency and incidence in stroke type in the last decade have also been previously described. Reasons for the observed proportional increase of hemorrhagic stroke patients remain speculative [25, 26]. Both, race and stroke type, were important predictive factors for rate of PEG tube placement. PEG tubes were more likely to be placed in minority race patients and patients with hemorrhagic stroke, both of which have been noted in previous research [27-29]. Nevertheless, the changes in the proportional frequency of race and stroke type did not have a confounding influence on the impact of the year on the rate of PEG tube placement in our study.
Besides changes in the demographic and medical characteristics of stroke patients, the observed changes in practice patterns might be due to changes in health care reimbursement, better stroke care in general and better awareness of dysphagia management and PEG tube placement in specific. Stroke care underwent fundamental changes during the last 15 years, which was also reflected by the observed 50% decrease in mortality in our study. For example, stroke units have become widespread, tissue Plasminogen Activator has been implemented, and the diagnosis and treatment of dysphagia following stroke have received increased attention. Thus, we speculate that the decrease in PEG tube placement may, in part, be explained by better stroke care including better dysphagia management.
In contrast to the decrease in LOS for the total stroke cohort, we have observed an increase in LOS for those stroke patients with a PEG tube. This might reflect that PEG tubes are more typically placed in stroke patients with higher severity levels necessitating longer hospitalizations. Unfortunately, clinical indications of PEG tube placement, like stroke severity and presence of dysphagia, or Speech and Language Pathology service utilization were not available in our study data due to the nature of the database and secondary data analyses. In addition, we did not examine the impact of hospital-level factors such as hospital type and volume of stroke patients [28]. Variations in practice pattern and, therefore, the possibilities to align with practice guidelines will be influenced by these and other factors and warrant further, prospective investigation.
Conclusions
In conclusion, the main finding of our study was that current PEG tube placement patterns in Florida in acute stroke patients are not in line with recommendations from clinical practice guidelines. The FOOD trial has suggested that early PEG tube placement in comparison to sustained NG tube feeding is associated with higher risks of death, a worse functional outcome, and reduced chances of being weaned from a feeding tube [7]. Therefore, current practice patterns might expose patients to the afore mentioned risks what should be addressed in future studies. Nevertheless, our results suggest that there is the potential to delay PEG tube placement, in some patients, without prolonging the hospital length of stay. Therefore, future, prospective research is needed to: investigate the generalizability of our results into other states and countries; understand the barriers related to delayed PEG placement; study the impact of delayed PEG tube placement on patient outcomes, hospital length of stay and costs; and to help identify those patients who would benefit from a delayed placement.
Acknowledgments
Grant support / acknowledgement of financial support
None / nothing to disclose
Footnotes
Institution where the study was performed
Medical University of South Carolina, Department of Health Sciences and Research, College of Health Professions
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Janes SEJ, Price CSG, Khan S. Percutaneous endoscopic gastrostomy: 30-Day mortality trends and risk factors. Journal of Postgraduate Medicine. 2005;51:23–9. [PubMed] [Google Scholar]
- 2.Ha L, Hauge T. Percutaneous endoscopic gastrostomy (PEG) for enteral nutrition in patients with stroke. Scand J Gastroenterol. 2003;38:962–6. doi: 10.1080/00365520310005190. [DOI] [PubMed] [Google Scholar]
- 3.Wijdicks EF, McMahon MM. Percutaneous endoscopic gastrostomy after acute stroke: complications and outcome. Cerebrovascular diseases (Basel, Switzerland) 1999;9:109–11. doi: 10.1159/000015907. [DOI] [PubMed] [Google Scholar]
- 4.Allison MC, Morris AJ, Park RH, et al. Percutaneous endoscopic gastrostomy tube feeding may improve outcome of late rehabilitation following stroke. Journal of the Royal Society of Medicine. 1992;85:147–9. [PMC free article] [PubMed] [Google Scholar]
- 5.Powell-Tuck J, van Someren N. Enterostomy feeding for patients with stroke and bulbar palsy. Journal of the Royal Society of Medicine. 1992;85:717–9. doi: 10.1177/014107689208501202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Scottish Intercollegiate Guidelines Network. A national clinical guideline. Edinburgh (Scotland): Scottish Intercollegiate Guidelines Network (SIGN); 2010. Management of patients with stroke: identification and management of dysphagia. [Google Scholar]
- 7.Dennis M, Lewis S, Cranswick G, et al. FOOD: a multicentre randomised trial evaluating feeding policies in patients admitted to hospital with a recent stroke. Health technology assessment (Winchester, England) 2006;10:iii–iv. ix–x, 1–120. doi: 10.3310/hta10020. [DOI] [PubMed] [Google Scholar]
- 8.Jauch EC, Saver JL, Adams HP, Jr, et al. Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke; a journal of cerebral circulation. 2013;44:870–947. doi: 10.1161/STR.0b013e318284056a. [DOI] [PubMed] [Google Scholar]
- 9.National Collaborating Centre for Acute Care (UK) Nutrition Support for Adults: Oral Nutrition Support, Enteral Tube Feeding and Parenteral Nutrition. London (UK): 2006. [PubMed] [Google Scholar]
- 10.Wirth R, Smoliner C, Jäger M, et al. Guideline clinical nutrition in patients with stroke. Experimental and Translational Stroke Medicine. 2013;5 doi: 10.1186/2040-7378-5-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Prosiegel M. Neurogene Dysphagien. Leitlinien für Diagnostik und Therapie in der Neurologie 2012 der DGN (Deutsche Gesellschaft für Neurologie) Kapitel Rehabilitation. 2012 [Google Scholar]
- 12.Volkert D, Berner YN, Berry E, et al. ESPEN Guidelines on Enteral Nutrition: Geriatrics. Clinical nutrition. 2006;25:330–60. doi: 10.1016/j.clnu.2006.01.012. [DOI] [PubMed] [Google Scholar]
- 13.Bueno H, Ross JS, Wang Y, et al. Trends in length of stay and short-term outcomes among Medicare patients hospitalized for heart failure 1993-2006. JAMA : the journal of the American Medical Association. 2010;303:2141–7. doi: 10.1001/jama.2010.748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Centers for Disease Control and Prevention (CDC) Cerebrovascular Disease or Stroke. 2015 Retrieved February 24, 2015, from http://www.cdc.gov/nchs/fastats/stroke.htm.
- 15.Reker DM, Hamilton BB, Duncan PW, et al. Stroke: who’s counting what? J Rehabil Res Dev. 2001;38:281–9. [PubMed] [Google Scholar]
- 16.Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373–83. doi: 10.1016/0021-9681(87)90171-8. [DOI] [PubMed] [Google Scholar]
- 17.Quan H, Li B, Couris CM, et al. Updating and validating the Charlson comorbidity index and score for risk adjustment in hospital discharge abstracts using data from 6 countries. American journal of epidemiology. 2011;173:676–82. doi: 10.1093/aje/kwq433. [DOI] [PubMed] [Google Scholar]
- 18.Quan H, Sundararajan V, Halfon P, et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Medical care. 2005;43:1130–9. doi: 10.1097/01.mlr.0000182534.19832.83. [DOI] [PubMed] [Google Scholar]
- 19.Plonk WM., Jr To PEG or not to PEG. Practical Gastroenterology. 2005;29:9–31. [Google Scholar]
- 20.Lee C, Im JP, Kim JW, et al. Risk factors for complications and mortality of percutaneous endoscopic gastrostomy: a multicenter, retrospective study. Surgical endoscopy. 2013;27:3806–15. doi: 10.1007/s00464-013-2979-3. [DOI] [PubMed] [Google Scholar]
- 21.Teuschl Y, Brainin M, Matz K, et al. Time trends in patient characteristics treated on acute stroke-units: results from the Austrian Stroke Unit Registry 2003-2011. Stroke; a journal of cerebral circulation. 2013;44:1070–4. doi: 10.1161/STROKEAHA.111.676114. [DOI] [PubMed] [Google Scholar]
- 22.Arboix A, Cendros V, Besa M, et al. Trends in risk factors, stroke subtypes and outcome. Nineteen-year data from the Sagrat Cor Hospital of Barcelona stroke registry. Cerebrovascular diseases (Basel, Switzerland) 2008;26:509–16. doi: 10.1159/000155989. [DOI] [PubMed] [Google Scholar]
- 23.Kleindorfer DO, Khoury J, Moomaw CJ, et al. Stroke incidence is decreasing in whites but not in blacks: a population-based estimate of temporal trends in stroke incidence from the Greater Cincinnati/Northern Kentucky Stroke Study. Stroke; a journal of cerebral circulation. 2010;41:1326–31. doi: 10.1161/STROKEAHA.109.575043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.U.S. Census Bureau. Census 2000 and 2010 Census. Florida: Profile of General Population and Housing Characteristics. Prepared by the Florida Legislative Office of Economic and Demographic Research. 2011 Retrieved June 7, 2016, from http://edr.state.fl.us/Content/population-demographics/2010-census/data/Florida_Comparison_Profile.pdf.
- 25.Feigin VL, Lawes CM, Bennett DA, et al. Worldwide stroke incidence and early case fatality reported in 56 population-based studies: a systematic review. Lancet neurology. 2009;8:355–69. doi: 10.1016/S1474-4422(09)70025-0. [DOI] [PubMed] [Google Scholar]
- 26.Fang MC, Coca Perraillon M, Ghosh K, et al. Trends in stroke rates, risk, and outcomes in the United States, 1988 to 2008. The American journal of medicine. 2014;127:608–15. doi: 10.1016/j.amjmed.2014.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Faigle R, Bahouth MN, Urrutia VC, et al. Racial and Socioeconomic Disparities in Gastrostomy Tube Placement After Intracerebral Hemorrhage in the United States. Stroke; a journal of cerebral circulation. 2016;47:964–70. doi: 10.1161/STROKEAHA.115.011712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.George BP, Kelly AG, Schneider EB, et al. Current practices in feeding tube placement for US acute ischemic stroke inpatients. Neurology. 2014;83:874–82. doi: 10.1212/WNL.0000000000000764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Dubin PH, Boehme AK, Siegler JE, et al. New model for predicting surgical feeding tube placement in patients with an acute stroke event. Stroke; a journal of cerebral circulation. 2013;44:3232–4. doi: 10.1161/STROKEAHA.113.002402. [DOI] [PMC free article] [PubMed] [Google Scholar]


