Abstract
Background/Aim
The renal arterial resistance index (RRI) is a Doppler measure, which reflects abnormalities in the renal blood flow. The aim of this study was to verify the value of RRI as a predictor of worsening renal function (WRF) in a group of chronic heart failure (CHF) outpatients.
Methods
We enrolled 266 patients in stable clinical conditions and on conventional therapy. Peak systolic velocity and end diastolic velocity of a segmental renal artery were obtained by pulsed Doppler flow, and RRI was calculated. Creatinine serum levels were evaluated at baseline and at 1 year, and the changes were used to assess WRF occurrence.
Results
During follow-up, 34 (13%) patients showed WRF. RRI was associated with WRF at univariate (OR: 1.13; 95% CI: 1.07–1.20) as well as at a forward stepwise multivariate logistic regression analysis (OR: 1.09; 95% CI: 1.03–1.16; p = 0.005) including the other univariate predictors.
Conclusions
Quantification of arterial renal perfusion provides a new parameter that independently predicts the WRF in CHF outpatients. Its possible role in current clinical practice to better define the risk of cardiorenal syndrome progression is strengthened.
Key Words: Chronic kidney disease, Systemic disease and the kidney, Glomerular hemodynamics
Introduction
In the last few years, there has been a growing interest in the close link between the disorders of the kidney and the heart, two organs that share many common pathophysiological mechanisms and that can negatively affect each other. Recently, this condition has been recognized as a clinical entity called cardiorenal syndrome [1]. Its clinical relevance has been widely demonstrated in chronic heart failure (CHF) patients, in whom an accurate assessment of renal function and its worsening play a crucial role in order to better stratify the outcome. [2]
In this context, the renal resistance index (RRI), a parameter obtained by renal pulsed Doppler that reflects renal resistances, could be a useful tool to better characterize renal disease. It has been demonstrated that it can integrate information obtained from the estimation of the glomerular filtration rate (GFR), thus improving patients’ prognosis [3,4,5,6,7]. Moreover, RRI could detect early renal function abnormalities because it has been demonstrated to be associated with both vascular lesions and histological abnormalities [6]. Finally, in different clinical settings, RRI values have been associated with a greater probability of renal disease progression [3,4,5].
The aim of this study was to verify the role of RRI as a predictor of worsening renal function (WRF) in a group of CHF outpatients in stable clinical conditions.
Methods
The study involved outpatients with CHF of any origin who had been referred to the Heart Failure Unit of the University of Bari between January 2010 and June 2014. The diagnosis of CHF was based on the criteria of the European Society of Cardiology [8]. According to the left ventricular ejection fraction (LVEF) at the time of diagnosis, patients were classified as affected by CHF with reduced or preserved LVEF [8]. At the time of enrolment, they had been clinically stable for at least 30 days and had been receiving conventional medical and electrical therapy for at least 3 months. Patients with acute decompensated HF (ADHF), acute worsening of kidney function or severe renal failure (GFR <15 ml/min) were excluded from the study. The study was approved by the local ethics committee, and all patients provided written informed consent.
Baseline Evaluations
Medical Examination and Electrocardiogram
The presence of ischemic heart disease, arterial hypertension and diabetes mellitus was accurately recorded, as well as the cardiovascular drugs taken, New York Heart Association (NYHA) class, weight, height, systolic and diastolic arterial pressure. Heart rhythm and heart rate were assessed by a 12-lead echocardiogram.
Echocardiographic Evaluation
Echocardiographic images were obtained using an echocardiograph (Vivid 7; GE Vingmed Ultrasound, General Electric, Milwaukee, Wis., USA) equipped with a 4-MHz probe. Left and right systolic function was assessed by measuring LVEF (Simpson rule) and the systolic peak of the tricuspid annular plane excursion [9]. Mitral and tricuspid regurgitation were semiquantitatively evaluated by means of color Doppler, with arbitrary units ranging from 0 to 4. Dilatation of the inferior vena cava and its collapsibility during inspiration were evaluated to establish central venous pressure (CVP) >5 mm Hg. [9]. Finally, pulmonary arterial systolic pressure was evaluated on the basis of the peak velocity of tricuspid regurgitation and estimation of the CVP [9].
Chemical Evaluations
Blood samples were taken in order to evaluate amino-terminal brain natriuretic peptide (NT-proBNP) (immunoassay; Dade Behring, Eschborn, Germany), serum creatinine (mg/dl) and hemoglobin (g/dl) levels. The baseline GFR was calculated using the Chronic Kidney Disease Epidemiology Collaboration equation (GFR-EPI) [10].
Renal Arterial Echo Color Doppler
Renal arterial Doppler was performed after echocardiographic examination by using the same echograph (Vivid 7; GE Vingmed Ultrasound, General Electric) and the same 4-MHz probe as previously described [7]. Briefly, the course of the right or left kidney segmental arteries were visualized by color flow Doppler, and pulsed Doppler was performed at the middle tract level of the best one visualized. An average of 2–3 measurements of the peak systolic velocity and the minimum end-diastolic velocity were used to calculate the RRI according to Peurcelot's formula. Patients showing a Doppler pattern suggesting renal artery stenosis were excluded [7].
Follow-Up
At 1 year of follow-up, WRF was defined as an increase in serum creatinine of >0.3 mg/dl, associated with a change of >25% or a decrease in GFR-EPI of >20% [2]. Within the first year of follow-up, hospitalizations due to ADHF were also registered. At 1 year, also NYHA class and LVEF were evaluated. Finally, after 1 year, all-cause mortality was evaluated.
Statistical Analysis
The continuous variables are expressed as mean values ± SD, and the categorical variables are given as percentages.
Univariate logistic regression analyses were used to assess the association of candidate variables with the occurrence of WRF. ORs with 95% CIs were calculated per unit change of the analyzed variables. A forward stepwise logistic regression analysis (p < 0.05 for removal) was used to assess the univariate predictors independently associated with WRF. Variable selection in multivariable modelling was based on strong statistical significance of the association with the events at univariate analysis (p value < 0.01).
Analysis of variance (ANOVA) was used to compare the mean creatinine change after 1 year according to the number of risk factors. A χ2 analysis was used to determine whether the proportion of WRF increased with the number of risk factors.
Receiver-operating characteristic (ROC) curves and the area under the curve of the ROC curves were calculated to determine the associations of RRI with events. The best cutoff for analyzed events was defined on the basis of the greatest sum of sensitivity and specificity. The effect of RRI on the risk classification was evaluated by adding the variable to a referring model using integrated discrimination improvement and category-free net reclassification improvement.
Cox univariate and multivariate analyses were used to evaluate the association between WRF endpoints and all-cause mortality.
The analyses were made using STATA software, Version 12 (StataCorp, College Station, Tex., USA). p values <0.05 were considered statistically significant.
Results
From a total of 341 patients, 266 were enrolled. Twenty-seven (8%) who had died were excluded, as were 48 subjects (12%) who missed the 1-year evaluation. The clinical characteristics of those enrolled are shown in table 1. At baseline, according to the GFR-EPI, 24.8% of the patients were in KDOQI stage I, 39.1% in stage II, 20.3% in stage IIIa, 12.4% in stage IIIb and 3.4% in stage IV. Eleven patients (4%) were affected by CHF with preserved LVEF.
Table 1.
Baseline clinical characteristics of the patients
| Number | 266 |
| Age, years | 64 ± 14 |
| Males | 79 |
| Ischemic cardiomyopathy | 42 |
| Diabetes mellitus | 29 |
| Arterial hypertension | 59 |
| Body mass index | 28 ± 4 |
| Systolic arterial pressure, mm Hg | 120 ± 16 |
| NYHA class | 2.2 ± 0.7 |
| Atrial fibrillation | 17 |
| LVEF | 33 ± 9 |
| TAPSE, mm | 19 ± 4 |
| CVP >5 | 12 |
| PASP | 35 ± 10 |
| GFR-EPI, ml/min/1.73 m2 | 71 ± 25 |
| NT-proBNP, pg/ml | 1,404 ± 1,678 |
| Concomitant therapy | |
| ACE inhibitors and/or ARBs | 87 |
| Beta-blockers | 97 |
| Aldosteron antagonists | 63 |
| Loop diuretics | 92 |
| Mean furosemide equivalent dose, mg/day | 77 ± 83 |
Values are expressed as mean ± SD or percentages, unless otherwise indicated. ACE = Angiotensin-converting enzyme; ARBs = angiotensin II receptor blockers; PASP = pulmonary arterial systolic pressure; TAPSE = tricuspid annular plane systolic excursion.
Association between RRI and WRF at Logistic Regression Analysis
During follow-up, 34 (13%) patients showed WRF. Table 2 shows the clinical characteristics of patients with and without WRF. At univariate logistic regression analysis, age, NYHA class, furosemide dose, high diuretic dose, GFR-EPI, log NT-proBNP, CVP >5 mm Hg, ADHF within 1 year and RRI were significantly associated with WRF occurrence.
Table 2.
Clinical characteristics of the patients with and without WRF and univariate logistic regression analysis
| WRF |
Univariate regression analysis |
|||
|---|---|---|---|---|
| yes (n = 34) | no (n = 232) | OR (95% CI) | p | |
| Age, years | 70 ± 9 | 63 ± 14 | 1.05 (1.01–1.08) | <0.001 |
| Males | 88 | 78 | 2.17 (0.73–6.43) | 0.164 |
| HFPEF | 3 | 4 | 0.67 (0.08–5.42) | 0.710 |
| Systolic arterial pressure, mm Hg | 120 ± 14 | 120 ± 16 | 0.99 (0.98–1.02) | 0.996 |
| Diabetes | 38 | 28 | 1.59 (0.75–3.36) | 0.224 |
| Hypertension | 65 | 58 | 0.93 (0.45–1.91) | 0.846 |
| NYHA class | 2.4 ± 0.5 | 2.2 ± 0.6 | 2.28 (1.18–4.41) | 0.014 |
| ACE inhibitors and/or ARBs | 76 | 88 | 0.47 (0.19–1.12) | 0.089 |
| Furosemide equivalent dose | 124 ± 96 | 71 ± 79 | 1.01 (1.00–1.01) | 0.002 |
| High diuretic dose | 56 | 20 | 4.98 (2.36–10.4) | <0.001 |
| GFR-EPI, ml/min/1.73 m2 | 60 ± 18 | 73 ± 25 | 0.98 (0.96–0.99) | 0.009 |
| NT-proBNP, pg/mla | 2,252 ± 1,994 | 1,278 ± 1,597 | 1.80 (1.27–2.56) | 0.001 |
| LVEF | 31 ± 9 | 34 ± 9 | 0.96 (0.92–1.00) | 0.075 |
| TAPSE, mm | 18 ± 4 | 20 ± 4 | 0.48 (0.19–1.15) | 0.099 |
| CVP >5 | 35 | 9 | 5.48 (2.38–12.6) | <0.001 |
| PASP, mm Hg | 39 ± 13 | 34 ± 10 | 1.04 (1.01–1.08) | 0.005 |
| ADHF within 1 year of follow-up | 29 | 7 | 5.63 (2.29–13.77) | <0.001 |
| RRI | 77 ± 5 | 70 ± 8 | 1.13 (1.07–1.20) | <0.001 |
Values are expressed as mean ± SD or percentages. HFPEF = CHF with preserved LVEF; ACE = angiotensin-converting enzyme; ARBs = angiotensin II receptor blockers; PASP = pulmonary arterial systolic pressure; TAPSE = tricuspid annular plane systolic excursion.
After log transformation.
In a multivariate forward stepwise regression model including age, high diuretic dose, GFR-EPI, log NT-proBNP, CVP >5 mm Hg and ADHF within 1 year, only RRI (OR: 1.09; 95 CI: 1.03–1.16; p = 0.005) and CVP >5 mm Hg (OR: 2.62; 95% CI: 1.04–6.63; p = 0.041) remained significantly associated with WRF.
Best RRI Cutoff to Predict WRF
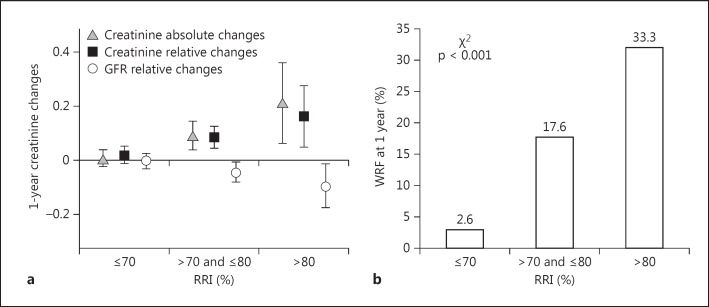
As shown in figure 1a, a progressive increase of absolute creatinine changes (p < 0.001 at ANOVA analysis) and a relative reduction of GFR-EPI (p < 0.05 at ANOVA analysis) were observed in the subgroup of patients with greater RRI values. Similarly, an increase in the risk of WRF occurrence was observed (fig. 1b). At ROC curve analysis, when WRF was considered, the RRI showed an area under the curve of 0.74 (95% CI 0.67–0.82). The best cutoff of RRI was 70, with a sensitivity of 91% and a specificity of 50%.
Fig. 1.
a One-year absolute (triangles) and relative (black squares) changes of creatinine and relative changes of the GFR (open circles) in the groups of patients according to the RRI values. At ANOVA analyses, p < 0.05 was observed for all variables. b The development of a WRF at 1 year (i.e. an increase in serum creatinine by ≥0.3 mg/dl from enrolment) in the same patient subgroups is shown. p refers to the χ2 test. Data are presented as the mean ± 95% CI.
Incremental Value of RRI in Detecting WRF
In order to assess the ability of RRI to improve reclassification, a logistic regression analysis was performed to obtain a reference model according to the number of events observed. This included age, CVP >5 mm Hg, HDD, ADHF within 1 year, GFR-EPI and logarithm of NT-BNP. In this model, only CVP (OR: 2.96; 95% CI: 1.16–7.54; p = 0.023) and ADHF within 1 year (OR: 2.88; 95% CI: 1.02–8.13; p = 0.046) remained associated with WRF. The addition of RRI >70 to the reference model, including these two variables, significantly improved reclassification according to both category-free net reclassification improvement (52%; 95% CI 16–88; p = 0.005) and integrated discrimination improvement (0.033; 95% CI 0.021–0.045; p = 0.006).
Clinical Relevance of WRF
As shown in table 2, patients with WRF when compared with those without WRF showed an increased probability of admission for ADHF. Moreover, at 1 year, they showed a greater absolute reduction of LVEF (-1 ± 4 vs. +2 ± 6% respectively, p = 0.024) and a greater occurrence of functional status worsening, defined as the increase of NYHA class ≥1 (16 vs. 32%, respectively, p = 0.023). Finally, during the follow-up (mean 36 ± 19 months), 38 patients died, and the rate of deaths among patients with WRF was 47%, whereas it was 9% among patients without WRF. WRF was associated with all-cause mortality at univariate (HR 6.28; 95% CI: 3.29–11.9; p < 0.001; C-index: 0.67) as well as at multivariate (HR: 2.24; 95% CI: 1.31–4.45; p = 0.021) Cox regression analysis after correction for age, NYHA class, LVEF and systolic arterial pressure at 1 year.
Discussion
The main finding of this study is that in CHF outpatients in stable clinical conditions, a high RRI is an independent predictor of WRF at 1 year of follow-up, thus strengthening its role in predicting cardiorenal syndrome progression.
In CHF patients, baseline renal function impairment, as well as its worsening, are common and are strongly associated with a greater morbidity and mortality [2]. Among the parameters that are useful to better evaluate renal function and stratify prognosis, we have recently proposed a parameter reflecting arterial renal resistances [7]. The information carried by RRI is strictly related to the pathophysiology of kidney circulation. The kidney is a profusely vascularized organ, which, unlike other organs, does not regulate blood flow mainly by oxygen demand. Reflex and neurohormonal mechanisms modulate renal resistances, but a number of other pathophysiological factors, such as an increased arterial stiffness, arterial atherosclerosis, renal parenchymal abnormalities, intra-abdominal and CVP, can also affect them [7,11]. Finally, renal resistances can remain permanently increased if a microvascular disease and vascular rarefaction occur as a result of vasoconstriction-related ischemia, oxidative stress, endothelial dysfunction and inflammatory cytokine activity [12]. All these mechanisms can increase RRI [6], thus explaining its ability in predicting not only HF but also renal dysfunction progression.
In our study, apart from RRI, CVP is the only other parameter independently associated with WRF. Over the last few years, among the factors causing renal injury in cardiorenal syndrome, the role of venous congestion has been widely demonstrated, following that of kidney perfusion and neurohormonal activity [11,13]. The effect of CVP on renal function seems to be mainly related to the increase in the efferent end glomerular capillary pressure that induces a reduction in the net filtration pressure and, consequently, a drop in the GFR [13]. Besides the effects on gradients of renal vasculature, an increased renal venous pressure also causes a rise in the interstitial pressure and an increase in arterial renal resistances. This is due to the low compliance of the interstitium and of the tight capsule of the kidney [11]. As a consequence, an increased CVP may cause both abnormalities in the GFR and an increase of RRI, thus leading to a vicious circle responsible for further reduction of renal function. On the other hand, in the absence of an increased CVP, the presence of a high RRI reflects the other main pathophysiological conditions underlying the risk of WRF, thus explaining its independent and incremental role in predicting WRF in our series of patients.
Contrary to our previous results [14], we did not find an association between the loop diuretic dose and WRF. However, it is worth noting that the need for a high, or increased, loop diuretic dose is strictly related to RRI. For this reason, the lack of an independent association of the loop diuretic dose does not seem to be relevant because the diuretic dose seems to be mainly influenced by a high CVP and a high RRI [15].
Limitations of the Study
In our study, changes in creatinine levels were considered the primary endpoint. This endpoint has been widely demonstrated to be associated with a worse prognosis in ADHF as well as in CHF [2,16], and this has also been confirmed in our series. Moreover, in the assessment of WRF, we considered not only absolute changes of creatinine but also its relative changes from baseline and the relative changes of GFR. However, this endpoint could be limited in patients with greater baseline creatinine levels who also present greater baseline values of RRI due by to the greater changes of creatinine levels that can be observed.
Conclusions
RRI provides additional important information about cardiorenal pathophysiology, reflecting the hemodynamic effect of HF on renal function, and can help identify those patients who are more likely to encounter both HF and renal dysfunction progression.
Statement of Ethics
This research was ethically conducted in accordance with the Declaration of Helsinki. The study protocol was approved by the local ethics committee, and the enrolled patients gave their informed written consent.
Disclosure Statement
The authors declare that they have no conflicts of interest to disclose.
Acknowledgment
We would like to thank Anna Cavallo for her helpful cooperation in collecting blood samples and in planning scheduled patient follow-up.
References
- 1.Ronco C, Haapio M, House AA, Anavekar N, Bellomo R. Cardiorenal syndrome. J Am Coll Cardiol. 2008;52:1527–1539. doi: 10.1016/j.jacc.2008.07.051. [DOI] [PubMed] [Google Scholar]
- 2.Damman K, Tang WH, Testani JM, McMurray JJ. Terminology and definition of changes renal function in heart failure. Eur Heart J. 2014;35:3413–3416. doi: 10.1093/eurheartj/ehu320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Radermacher J, Ellis S, Haller H. Renal resistance index and progression of renal disease. Hypertension. 2002;39:699–703. doi: 10.1161/hy0202.103782. [DOI] [PubMed] [Google Scholar]
- 4.Ennezat PV, Maréchaux S, Six-Carpentier M, Pinçon C, Sediri I, Delsart P, Gras M, Mounier-Véhier C, Gautier C, Montaigne D, Jude B, Asseman P, Le Jemtel TH. Renal resistance index and its prognostic significance in patients with heart failure with preserved ejection fraction. Nephrol Dial Transplant. 2011;26:3908–3913. doi: 10.1093/ndt/gfr116. [DOI] [PubMed] [Google Scholar]
- 5.Doi Y, Iwashima Y, Yoshihara F, Kamide K, Hayashi SI, Kubota Y, Nakamura S, Horio T, Kawano Y. Renal resistive index and cardiovascular and renal outcomes in essential hypertension. Hypertension. 2012;60:770–777. doi: 10.1161/HYPERTENSIONAHA.112.196717. [DOI] [PubMed] [Google Scholar]
- 6.Bigé N, Lévy PP, Callard P, Faintuch JM, Chigot V, Jousselin V, Ronco P, Boffa JJ. Renal arterial resistive index is associated with severe histological changes and poor renal outcome during chronic kidney disease. BMC Nephrol. 2012;13:139. doi: 10.1186/1471-2369-13-139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ciccone MM, Iacoviello M, Gesualdo L, Puzzovivo A, Antoncecchi V, Doronzo A, Monitillo F, Citarelli G, Paradies V, Favale S. The renal arterial resistance index: a marker of renal function with an independent and incremental role in predicting heart failure progression. Eur J Heart Fail. 2014;16:210–216. doi: 10.1002/ejhf.34. [DOI] [PubMed] [Google Scholar]
- 8.McMurray JJ, Adamopoulos S, Anker SD, et al. ESC guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: The Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail. 2012;14:803–869. doi: 10.1093/eurjhf/hfs105. [DOI] [PubMed] [Google Scholar]
- 9.Rudski LG, Lai WW, Afilalo J, Hua L, Handschumacher MD, Chandrasekaran K, Solomon SD, Louie EK, Schiller NB. Guidelines for the echocardiographic assessment of the right heart in adults: a report from the American Society of Echocardiography endorsed by the European Association of Echocardiography, a registered branch of the European Society of Cardiology, and the Canadian Society of Echocardiography. J Am Soc Echocardiogr. 2010;23:685–713. doi: 10.1016/j.echo.2010.05.010. [DOI] [PubMed] [Google Scholar]
- 10.Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF, 3rd, Feldman HI, Kusek JW, Eggers P, Van Lente F, Greene T, Coresh J. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150:604–612. doi: 10.7326/0003-4819-150-9-200905050-00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Braam B, Cupples WA, Joles JA, Gaillard C. Systemic arterial and venous determinants of renal hemodynamics in congestive heart failure. Heart Fail Rev. 2012;17:161–175. doi: 10.1007/s10741-011-9246-2. [DOI] [PubMed] [Google Scholar]
- 12.Chade AR. Renal vascular structure and rarefaction. Compr Physiol. 2013;3:817–831. doi: 10.1002/cphy.c120012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Verbrugge FH, Grieten L, Mullens W. Management of the cardiorenal syndrome in decompensated heart failure. Cardiorenal Med. 2014;4:176–188. doi: 10.1159/000366168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Iacoviello M, Puzzovivo A, Monitillo F, Saulle D, Lattarulo MS, Guida P, et al. Independent role of high central venous pressure in predicting worsening of renal function in chronic heart failure outpatients. Int J Cardiol. 2013;162:261–263. doi: 10.1016/j.ijcard.2012.06.088. [DOI] [PubMed] [Google Scholar]
- 15.Iacoviello M, Doronzo A Paradies V, Antoncecchi V, Monitillo F, Citarelli G, Leone M, Puzzovivo A, Gesualdo L, Ciccone MM. The independent association between altered renal arterial resistance and loop diuretic dose in chronic heart failure outpatients. IJC Heart Vasc. 2015;7:119–123. doi: 10.1016/j.ijcha.2015.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Damman K, Navis G, Voors AA, Asselbergs FW, Smilde TD, Cleland JG, van Veldhuisen DJ, Hillege HL. Worsening renal function and prognosis in heart failure: systematic review and meta-analysis. J Card Fail. 2007;13:599–608. doi: 10.1016/j.cardfail.2007.04.008. [DOI] [PubMed] [Google Scholar]