Abstract
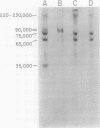
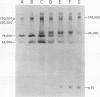
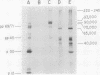
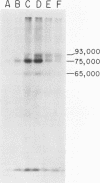
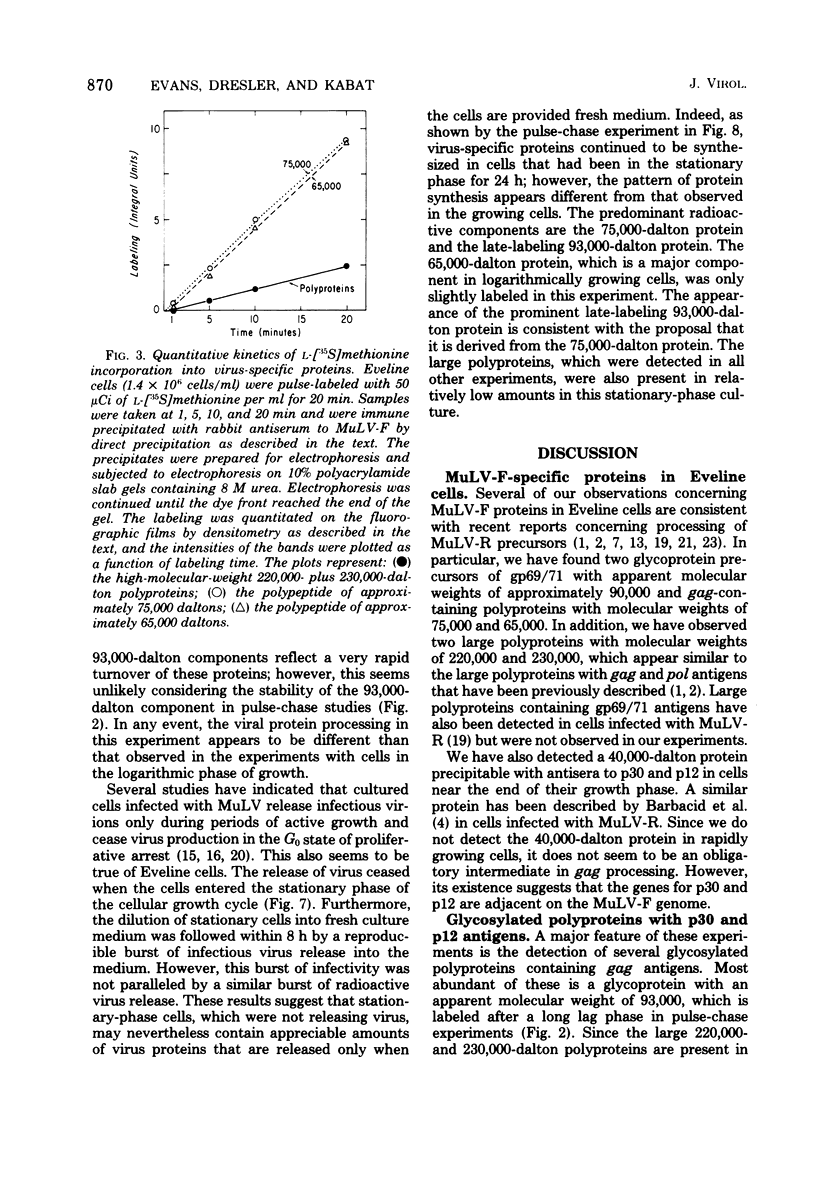
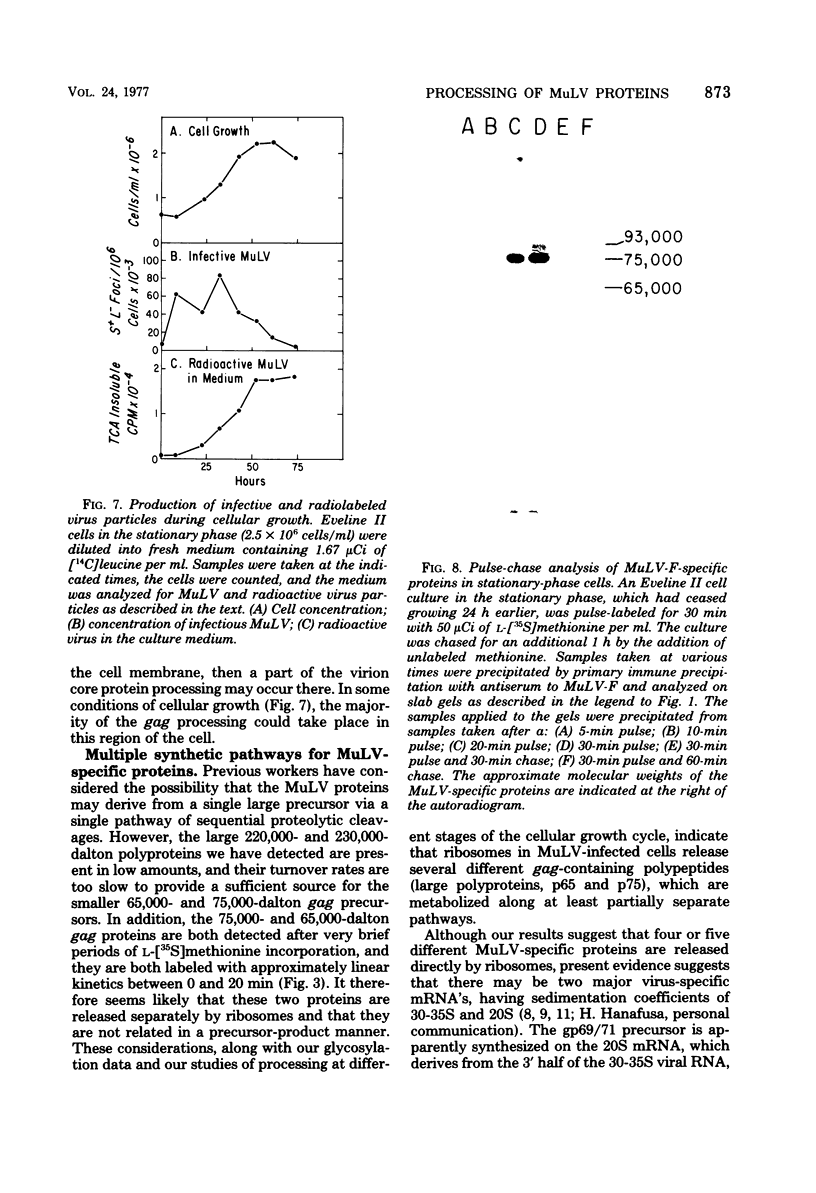
Synthesis and post-translational processing of murine leukemia virus proteins were analyzed in a murine cell line (Eveline) that produces large amounts of Friend lymphatic leukemia virus. Immunoprecipitation of l-[35S]methionine-labeled cell extracts demonstrated that several different virus-specific proteins antigenically related to the virion core (gag) proteins p12 and p30 become radioactive within 1 min of labeling and exhibit labeling kinetics characteristic of primary translation products. The most abundant of these were proteins with molecular weights of 75,000 and 65,000. There were, in addition, two large glycosylated polyproteins with apparent molecular weights of 220,000 and 230,000, which were precipitated by antisera to p30 or p12 but not by antiserum to the major envelope glycoproteins gp69/71. Several lines of evidence, including labeling with d-[3H]glucosamine and binding to insolubilized lectins, suggested that the 75,000-dalton internal core polyprotein is slowly processed to form a glycoprotein with an apparent molecular weight of 93,000. On the contrary, the 65,000-dalton protein appeared to be an immediate precursor to the virion core proteins. Its processing can involve intermediates containing p30 and p12 antigens with molecular weights of 50,000 and 40,000; however, the latter did not appear to be obligatory intermediates. The detection of the 40,000-dalton protein suggested that the genes for p30 and p12 are adjacent on the viral genome. These results indicated that there are several pathways of synthesis and post-translational processing of polyprotein precursors to the gag proteins and that several of these polyproteins are glycosylated. A comparison of gag precursor processing in rapidly growing, slowly growing, and stationary cells indicated that different pathways are favored under different conditions of cell growth. Our analysis of envelope glycoprotein synthesis has confirmed the existence of two rapidly labeled 90,000-dalton glycoproteins, which appear to be precursors to the envelope glycoproteins gp69/71.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arcement L. J., Karshin W. L., Naso R. B., Jamjoom G., Arlinghaus R. B. Biosynthesis of Rauscher leukemia viral proteins: presence of p30 and envelope p15 sequences in precursor polypeptides. Virology. 1976 Feb;69(2):763–774. doi: 10.1016/0042-6822(76)90504-3. [DOI] [PubMed] [Google Scholar]
- Avrameas S., Ternynck T. The cross-linking of proteins with glutaraldehyde and its use for the preparation of immunoadsorbents. Immunochemistry. 1969 Jan;6(1):53–66. doi: 10.1016/0019-2791(69)90178-5. [DOI] [PubMed] [Google Scholar]
- Barbacid M., Stephenson J. R., Aaronson S. A. gag Gene of mammalian type-C RNA tumour viruses. Nature. 1976 Aug 12;262(5569):554–559. doi: 10.1038/262554a0. [DOI] [PubMed] [Google Scholar]
- Bolognesi D. P., Luftig R., Shaper J. H. Localization of RNA tumor virus polypeptides. I. Isolation of further virus substructures. Virology. 1973 Dec;56(2):549–564. doi: 10.1016/0042-6822(73)90057-3. [DOI] [PubMed] [Google Scholar]
- Bonner W. M., Laskey R. A. A film detection method for tritium-labelled proteins and nucleic acids in polyacrylamide gels. Eur J Biochem. 1974 Jul 1;46(1):83–88. doi: 10.1111/j.1432-1033.1974.tb03599.x. [DOI] [PubMed] [Google Scholar]
- Famulari N. G., Buchhagen D. L., Klenk H. D., Fleissner E. Presence of murine leukemia virus envelope proteins gp70 and p15(E) in a common polyprotein of infected cells. J Virol. 1976 Nov;20(2):501–508. doi: 10.1128/jvi.20.2.501-508.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gielkens A. L., Van Zaane D., Bloemers H. P., Bloemendal H. Synthesis of Rauscher murine leukemia virus-specific polypeptides in vitro. Proc Natl Acad Sci U S A. 1976 Feb;73(2):356–360. doi: 10.1073/pnas.73.2.356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Ledbetter J., Nowinski R. C., Emery S. Viral proteins expressed on the surface of murine leukemia cells. J Virol. 1977 Apr;22(1):65–73. doi: 10.1128/jvi.22.1.65-73.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mueller-Lantzsch N., Fan H. Monospecific immunoprecipitation of murine leukemia virus polyribosomes: identification of p30 protein-specific messenger RNA. Cell. 1976 Dec;9(4 Pt 1):579–588. doi: 10.1016/0092-8674(76)90040-4. [DOI] [PubMed] [Google Scholar]
- Naso R. B., Arcement L. J., Karshin W. L., Jamjoom G. A., Arlinghaus R. B. A fucose-deficient glycoprotein precursor to Rauscher leukemia virus gp69/71. Proc Natl Acad Sci U S A. 1976 Jul;73(7):2326–2330. doi: 10.1073/pnas.73.7.2326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nermut M. V., Frank H., Schäfer W. Properties of mouse leukemia viruses. 3. Electron microscopic appearance as revealed after conventional preparation techniques as well as freeze-drying and freeze-etching. Virology. 1972 Aug;49(2):345–358. doi: 10.1016/0042-6822(72)90487-4. [DOI] [PubMed] [Google Scholar]
- Panem S., Kirsten W. H. Release of mouse leukemia-sarcoma virus from synchronized cells. J Natl Cancer Inst. 1973 Feb;50(2):563–565. doi: 10.1093/jnci/50.2.563. [DOI] [PubMed] [Google Scholar]
- Paskind M. P., Weinberg R. A., Baltimore D. Dependence of Moloney murine leukemia virus production on cell growth. Virology. 1975 Sep;67(1):242–248. doi: 10.1016/0042-6822(75)90421-3. [DOI] [PubMed] [Google Scholar]
- Pawson T., Martin G. S., Smith A. E. Cell-free translation of virion RNA from nondefective and transformation-defective Rous sarcoma viruses. J Virol. 1976 Sep;19(3):950–967. doi: 10.1128/jvi.19.3.950-967.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schäfer W., Seifert E. Production of a potent complement-fixing murine leukemia virus-antiserum from the rabbit and its reactions with various types of tissue culture cells. Virology. 1968 Jun;35(2):323–328. doi: 10.1016/0042-6822(68)90273-0. [DOI] [PubMed] [Google Scholar]
- Shapiro S. Z., Strand M., August J. T. High molecular weight precursor polypeptides to structural proteins of Rauscher murine leukemia virus. J Mol Biol. 1976 Nov 15;107(4):459–477. doi: 10.1016/s0022-2836(76)80078-2. [DOI] [PubMed] [Google Scholar]
- Sherton C. C., Evans L. H., Polonoff E., Kabat D. Relationship of Friend murine leukemia virus production to growth and hemoglobin synthesis in cultured erythroleukemia cells. J Virol. 1976 Jul;19(1):118–125. doi: 10.1128/jvi.19.1.118-125.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steeves R. A. Editorial: Spleen focus-forming virus in Friend and Rauscher leukemia virus preparations. J Natl Cancer Inst. 1975 Feb;54(2):289–297. doi: 10.1093/jnci/54.2.289. [DOI] [PubMed] [Google Scholar]
- Zaanie D., Gielkens A. L., Dekker-michielsen M. J., Bloemers H. P. Virus-specific precursor polypeptides in cells infected with Rauscher leukemia virus. Virology. 1975 Oct;67(2):544–552. doi: 10.1016/0042-6822(75)90454-7. [DOI] [PubMed] [Google Scholar]
- von der Helm K., Duesberg P. H. Translation of Rous sarcoma virus RNA in a cell-free system from ascites Krebs II cells. Proc Natl Acad Sci U S A. 1975 Feb;72(2):614–618. doi: 10.1073/pnas.72.2.614. [DOI] [PMC free article] [PubMed] [Google Scholar]