Abstract
The purpose of this qualitative study is to elucidate stakeholder perceptions of, and institutional practices related to cell-based therapies and products (CTP) regulation and commercialization in Canada. The development of reproducible, safe and effective CTPs is predicated on regulatory and commercialization environments that enable innovation. Manufacturing processes constitute a critical step for CTP development in this regard. The road from CTP manufacturing to translation in the clinic, however, has yet to be paved. This study aims to fill an empirical gap in the literature by exploring how CTP manufacturing facilities navigate Canadian regulatory and commercialization environments, which together drive the translation of novel CTPs from bench to bedside. Using the multi-level model of practice-driven institutional change proposed by Smets et al., we demonstrate how CTP manufacturing practices are governed by established standards, yet meaningfully shape higher-order regulatory and commercial norms in CTP research and development. We identify four key themes that undergird such processes of innovation: 1) managing regulatory uncertainty, which stems from an inability to classify CTPs within existing regulatory categories for approval and commercialization purposes; 2) building a ‘business case’ whereby a CTP's market potential is determined in large part by proving its safety and effectiveness; 3) standardizing manufacturing procedures that mobilize CTPs from a research and development phase to a commercialization one; and 4) networking between researchers and regulators to develop responsible commercialization processes that reflect the uniqueness of CTPs as distinct from other biologics and medical devices.
Keywords: Regenerative medicine, Commercialization, Cell-based therapies, Regulation, Institutional change, Canada
1. Background
Regenerative Medicine (RM) products are novel, diverse, and complex. They are intended to “repair, replace or restore diseased, damaged or missing tissues” (Bailey et al., 2014) and can range from drugs, medical devices, to cell therapies. Cell-based products and therapies (CTP) are a specific subset of RM products, and have demonstrated promise towards improving individual and population health as well as fostering bio-economies (Heathman et al., 2015). The clinical translation process of CTPs, not unlike pharmaceutical drugs and medical devices, proceeds sequentially in three primary phases. First, researchers working in RM and other stem cell-focused research fields amount sufficient pre-clinical evidence (proof of concept) and clinical evidence (safety and efficacy in the context of indication) for proposed applications of CTPs. Manufacturing can then proceed at a credited GMP facility, where standardized procedures ensure quality control in the production of the CTP or other RM product. Regulation and commercialization make up the final stages in this translational process, whereby CTPs are evaluated for safety/efficacy as well as commercial viability, respectively. Safety and efficacy of the CTP underpin its ‘value proposition’—that is “the benefits [of the CTP] offered to the customer, minus the cost and risk” (Ginty et al., 2011). This ‘value proposition’ in turn determines the CTP's reimbursement and, ultimately, its commercial viability (Ginty et al., 2011). It is on the latter phases of CTP translation (manufacturing, regulation and commercialization) that this paper explores in the Canadian context.
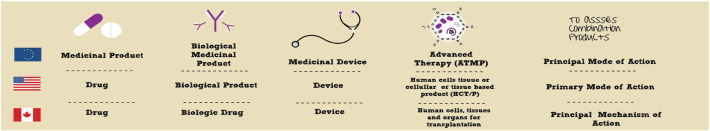
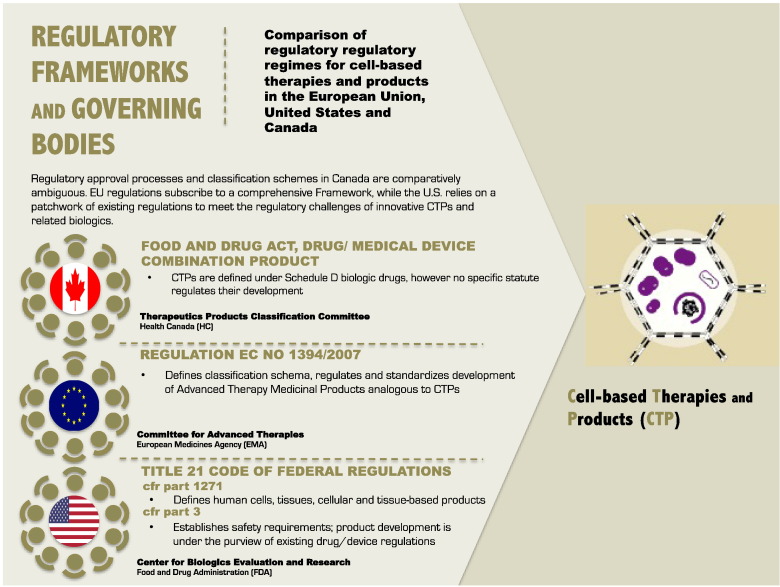
Regulation and commercialization therefore play important gatekeeping roles in the clinical translation, and market introduction of CTPs in Canada and other jurisdictions to be sure (Abbasalizadeh and Baharvand, 2013). Regulatory bodies such as Health Canada (CA), the Food and Drug Administration (U.S.) and the European Medicines Agency (EU) are responsible for evaluating CTP safety. The regulatory frameworks that have since emerged in each jurisdiction differ considerably in the ways in which they classify CTPs (Fig. 1), assess their safety and borrow from existing regulatory procedures meant for pharmaceutical drugs or medical devices to regulate CTPs and other novel RM therapeutics. Fig. 2 summarizes the regulatory mechanisms in each of these jurisdictions, while a detailed comparative analysis of their regulatory frameworks has been published by the lead author elsewhere (Beak and Isasi, 2014).
Fig. 1.
Regulatory classification schemas for RM products in the European Union, United States and Canada.
Fig. 2.
Cross comparison of regulatory regimes and relevant statutes for CTPs in the European Union, United States and Canada.
Designing national regulatory and commercialization environments that are conducive to innovation yet remain globally competitive therefore continues to be both a priority and a challenge for many countries (von Tigerstrom, 2015; Nature Editorials, 2015). This is particularly true in Canada. The case-by-case, conditional market approval process in Canada testifies to this, and which favours innovation by accelerating the licensing process. Indeed, it was this process that facilitated the regulatory approval of Prochymal™, the first stem cell product in the world (Viswanathan and Bubela, 2015).
Fostering a commercially-friendly, regulatory environment not only involves mandating regulatory compliance, but also balancing investment risks associated with RM technology development and adoption (French et al., 2013). One such risk derives from the novelty and manufacturing technicality of CTPs. Canada addresses this in part by investing in facilities that reflect “good manufacturing practices” (GMP) (French et al., 2013, Webster et al., 2011, Eriksson and Webster, 2015, Rosemann, 2014, Webster and Eriksson, 2008). Highly networked, multi-stakeholder initiatives assess regulatory pathways, mobilize knowledge and standardize GMPs across Canada (e.g. CellCAN: http://www.cellcan.com/en/) to enhance the clinical translation of CTPs.
Substantial challenges persist in the effort to create regulatory and commercial environments that drive innovation of CTPs from research and development, to approval and eventually commercialization. Considering the “diversity of cell types, disease indications and delivery pathways make a ‘one-size-fits-all’ manufacturing platform” unlikely for CTPs (Ginty et al., 2011). Several initiatives (e.g. CCRM) address these challenges by proposing improved regulatory frameworks that proportionally differentiate between products based on their scientific and socio-economic relevance (Heathman et al., 2015). Like other emerging biologics, field-level practices in CTP research and development inform the higher-order regulatory and commercial standards that now govern CTP innovation. It is currently unknown, however, how CTP manufacturing facilities navigate these regulatory and commercialization challenges to successfully bring such products to market. In depth exploration of practice-driven changes in the regulatory and commercialization environments would lend valuable insight into how CTP facilities navigate them, while shedding light on the techno-social applications both developers and regulators envision for CTPs in the clinic.
This study elucidates the perceptions and practices of CTP developers, regulators, manufacturers and commercial stakeholders in Canada, and is furthermore guided by the following research question: What facets of the regulatory and commercial environments for CTP development enable translation and innovation of CTPs in Canada? We aim to understand how various stakeholders—particularly GMP-CTP facilities—co-produce regulatory and commercialization norms for CTP innovation in their daily activities. While we focus on the Canadian context, the insights gained herein mirror those of RM regulators and commercial actors in other jurisdictions. Our study highlights how other countries may construct practice-level solutions to challenges in regulation and commercialization of CTPs, as well as for related RM products generally.
2. Conceptual framework: a multi-level model of practice driven institutional change
Regulatory and commercial standards currently in place for CTP research and development have largely drawn from practice-level changes in the CTP manufacturing sphere. In line with this observed phenomenon, we used the multi-level model of practice-driven institutional change developed by Smets et al. (2012). This framework provides a lens through which to examine how Canadian cell-manufacturing GMP facilities work within the confines of current regulatory and commercialization norms, while creating new ones through their daily practice. The Smets et al. framework focuses on how small changes in everyday practices lead to institutional, field-level changes experienced downstream on the CTP research and development continuum. For the purposes of this study we consider practices, “patterns of activities that are given thematic coherence by shared meanings and understandings” (Smets et al., 2012). Smets et al. point to three overlapping mechanisms that explain the relationship between practices: situated improvising, normative network reorientation and unobtrusive embedding. We investigate the multi-level model of practice driven institutional change at GMP facilities in Canada Table 1, and demonstrate how the interactions between these levels lead to higher-order regulatory and commercialization change. A description of the three mechanisms are provided below.
Table 1.
Characteristics of 6 Canadian GMP facilities.
| Facility | Affiliations/partners/networks/funders | Principle aims (values; mission statement) | Main research activities | Cell production/manufacturing activities | Quality control characteristics of facility/set up |
|---|---|---|---|---|---|
| Cell and Tissue Innovative Research Centre (CTRIC) Edmonton 2009 |
Affiliations and funders University of Alberta; Canada Foundation for Innovation; Alberta Enterprise and Advanced Education |
|
|
To meet regulatory requirements, each batch of cell and tissue product produced must be tested against pre-approved product specifications. Raw materials used in processing may require testing before use. CTIRC's GMP Quality Control Laboratory is equipped to provide these services. | |
| Orsino Facility for Cell Therapy Toronto 2006 |
Affiliations Princess Margaret Hospital; University of Toronto; Princess Margaret Hospital Foundation; Toronto General and Western Hospital Foundation |
|
The CTP has been involved in cell-based clinical trials for both anti-cancer therapies and for regenerative medicine. To-date the program has manufactured cellular products for four clinical trials. |
|
Three independent, class 10,000 clean rooms, each equipped with a biological safety cabinet, incubators, refrigerators, cell processing equipment and computer work stations; cryopreservation facility featuring multiple liquid nitrogen storage tanks; abundant support space for quality control/quality assurance; raw materials storage and quarantine spaces |
|
Funding Canada Foundation for Innovation Grants; Ontario Innovation Trust; Ontario Research Fund along with generous donations from philanthropists. | |||||
| Centre for Commercialization of Regenerative Medicine (CCRM) Toronto |
Affiliations Centres of Excellence for Commercialization and Research (CECR); The Networks of Centres of Excellence of Canada (NCE); MaRS Innovation (MI); MaRS Discovery District (MaRS DD); Ontario Stem Cell Initiative (OSCI); Stem Cell Network (SCN); McEwen Centre for Regenerative Medicine; and McGill's Centre of Genomics and Policy (CGP) |
Create and sustain a global nexus for Regenerative Medicine (RM) commercialization by unifying dynamic business leadership with high value innovative translational technology platforms based on demonstrated excellence in fundamental stem cell and biomaterial science. |
|
|
“Scale up” solutions are further tested in GMP facilities (Orsino Cell Therapy Facility and Sprott Centre for Stem Cell GMP Facility). Facilities include
|
|
Funding Industry sponsors in Regenerative Medicine including pharmaceutical, devices, reagents, tools, biomaterials and cell therapies. The industry consortium consists of large multinationals, small-medium enterprises and emerging biotechnology companies. (http://ccrm.ca/industry-consortium) | |||||
| Sprott Centre for Stem Cell Research Ottawa 2006 |
Affiliations University of Ottawa |
|
|
|
|
|
Funding Canada Foundation for Innovation; Ontario Innovation Trust; Kresge Foundation; Ottawa Hospital Foundation; Ontario Research and Development Challenge Fund; Genome Canada, the Canadian Institutes of Health Research, and the Stem Cell Network (2006 press release). | |||||
| Centre of Excellence for Cellular Therapy (CETC) Montreal 2010 |
Affiliations and funding Hôpital Maisonneuve-Rosement; Université de Montréal |
Conduct leading-edge research on stem cells, the immune system and cancer, with the goal of developing treatments for a number of diseases such as leukemia, lymphoma, myeloma, macular degeneration, articular reconstruction, diabetes, Parkinson's disease and Alzheimer's disease. |
The CETC consists of three separate branches: Branches 1 + 2 Studies on patients who have undergone transplants Branch 3 Preclinical and post-transplant studies. The preclinical laboratory will be used to develop new procedures or cellular therapy strategies and to ensure screening, validation and final quality control prior to transferring the new procedures to clinical application. |
|
Biosecurity clearance level BSL2 or confined to level NC2. Handling procedures are conducted under a biological safety hood. The CETC is equipped with highly sophisticated instruments such as:
|
| LOEX (Experimental Organogenesis Laboratory); Centre multidisciplinaire de développement du génie tissulaire; Quebec City 2009 |
Affiliations Université Laval bioengineers; Clinical research physicians from Hôpital du Saint-Sacrement or other hospitals |
|
|
Organogenesis and tissue reconstruction | These cells and tissues are produced in the clean room suite in a research center of 5000 m2 dedicated to tissue reconstruction and stem cell research. It includes 3 cell culture areas comprised each of an airlock, an incubator room, and a cell culture room with flow hood, microscope, centrifuge, cell counter, cold storage room, a preparation room and other spaces such as gowning, a corridor and an airlock entrance. Access to the area is strictly controlled, within a building that also has its own controlled access. The clean room suite allows the production of cells and tissues in a controlled environment based on GMP requirements. |
| Fonds de la recherche en santé du Québec (FRSQ) Natural Sciences and Engineering Research Council of Canada (NSERC); Medical Research Council of Canada (MRC); Fondation des pompiers du Québec pour les grands brûlés (FPQGB); Fondation de l'Hôpital du Saint-Sacrement | |||||
Situated improvising refers to emerging practices meant to circumvent workplace inefficiencies, which challenge contextual norms that stakeholders involved in the management process under study use to ‘get the job done’. In this way, staff members working within CTP manufacturing facilities mitigate uncertainty surrounding regulatory and commercialization requirements by developing tailored practices. Staff engage in ‘lesson learning’ while developing such practices (Carlile, 2002). Tailored practices may span (inter)national contexts, since CTP development requires that facilities adapt to regulatory frameworks used outside their own jurisdiction. Finally, while their overarching aim is to facilitate the development process, situated improvising also serves to diffuse practices conducive to innovation within the field generally.
The process through which emerging practices are diffused outside the organization is described as normative network reorientation. This involves the way in which practices are legitimized and endorsed by leading stakeholders in the field. As such, stakeholders constitute “referent audiences who matter” in diffusing emerging practices (Smets et al., 2012).
Finally, unobtrusive embedding is a form of “creeping standardization” that is ongoing and initiated from the bottom up. While dependent upon stakeholder consensus, unobtrusive embedding within CTP facilities refers to the ways in which standardizing GMP occurs uninterruptedly and unopposed. Unobtrusive embedding defines the communicative processes by which investigators and facility staff manage regulatory and commercialization challenges. They do so using strategies that can be identified through the optic of situated improvising and normative network reorientation. It also represents a form of boundary work (Zietsma and Lawrence, 2010). The diffusion of standardized GMP practices redefine regulatory and commercialization environments, thereby creating a ‘shared context’ (Zietsma and Lawrence, 2010) for CTP development. Consequently, the boundaries redefined by unobtrusive embedding challenge the dominant paradigms of regulation and commercialization that precedes them.
3. Methods
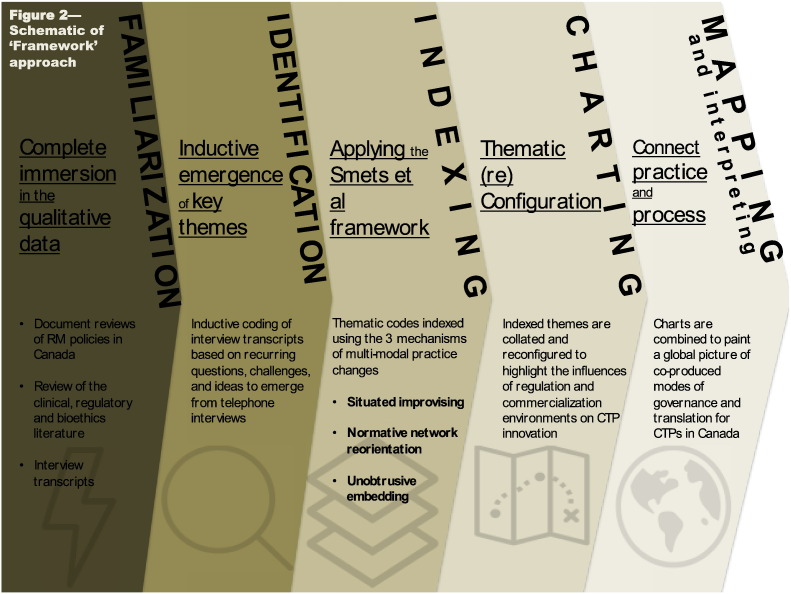
This qualitative description study adopts the ‘framework approach’ proposed by Ritchie and Spencer (2002) (Fig. 3). The authors affirm that the ‘Framework’ approach “involves a systematic process of sifting, charting and sorting material according to key issues and themes… The five key stages to qualitative data analysis involve in ‘Framework’ are:
-
1.
Familiarization
-
2.
Identifying a thematic framework
-
3.
Indexing
-
4.
Charting
-
5.
Mapping and interpretation (this being the stage at which the key objectives of qualitative analysis are addressed)” (Ritchie and Spencer, 2002, p.310).
Fig. 3.
Graphical schematic of the ‘Framework’ approach to qualitative data analysis for applied policy research adopted in the study.
The ‘Framework’ approach was explicitly designed to embed qualitative research traditions within applied policy research. It is conducive to exploring the relationships between regulatory policy and practice, foci we adopt in the context of RM innovation in this paper. Our study furthermore adheres to the critical appraisal requirements for qualitative research outlined by Kuper et al. (2008). Using the theoretical scaffolding of the Smets et al. framework, this study maps the multi-level model of practice driven institutional change onto the CTP manufacturing and regulatory practices of all 6 existing CTP facilities in Canada. The perceptions and experiences from CTP investigators, manufacturing staff, technology transfer officers as well as members of the national regulatory authority in Canada involved at various stages of CTP development (i.e. research, manufacturing, regulation and/or commercialization) were included in this study. The framework approach further enabled us to take into account the local specificities unique to each facility studied, as well as the similarities across them.
3.1. Participant recruitment
We used a combination of purposeful and snowball sampling strategies to recruit participants (Patton, 2014). Cell sampling strategies (Robinson, 2013) also enabled us to select participants based on a priori categories we defined in relation to staff roles and responsibilities at each facility we studied. Between 4 and 5 participants with the following expertise were recruited from each facility based on these categories: CTP manufacturing, regulatory affairs, management, science, medicine, and commercialization (in particular technology transfer officers). In addition, we interviewed staff at the national regulatory health agency (Health Canada) Table 2. We shared our cell sampling strategy with recruited participants, who then referred prospective participants they believed met one or more of our cell sampling criteria.
Table 2.
Interviewee roles within 6 GMP facilities and 1 federal regulatory body in Canada (N = 27).
| Regulator | CTP scientist/investigator | Technology transfer officer | CEO/associate director(s) | Scientific project manager/coordinator | Commercialization/business development and licensing | |
|---|---|---|---|---|---|---|
| GMP facility | ||||||
| LOEX | 1 | 1 | 2 | |||
| CCRM | 2 | 1 | 2 | |||
| CETC | 1 | 1 | 1 | |||
| Osino Cell Therapy Program | 3 | 1 | ||||
| CTIRC | 1 | 1 | 1 | |||
| Sprott Centre | 2 | 1 | ||||
| Regulatory body | ||||||
| Health Canada | 3 | |||||
| Professional body | ||||||
| CellCAN | 1 | |||||
3.2. Data collection
We achieved theoretical saturation and maximum variation (Bowen, 2008) after conducting 27 semi-structured interviews over the phone. Some participants worked within the facilities directly, while others worked in partnership. We developed an initial interview guide based on the conceptual framework proposed by Smets et al. (Appendix A) in which interview questions focused on clinical translation issues. We developed a separate interview guide for technology transfer officers (Appendix B) and regulatory officers (Appendix C) pursuant to commercialization and regulatory issues, respectively. Telephone interviews lasted approximately 45–60 min, were recorded and transcribed verbatim. Matrix tables as well as preliminary summaries of each case (GMP facility) were developed for preliminary analysis over the course of data collection (Miles et al., 2014, Lauckner et al., 2012).
3.3. Data analysis
We used template analysis to better understand how CTP facilities navigate and co-construct regulatory and commercial environments (Brooks et al., 2014). With N'Vivo software, we developed, and then expanded a codebook based on preliminary findings from the interview data. Codes were clustered and initial themes identified for each facility. These codes were further synthesized through memoing to develop a thematic map (Miles et al., 2014). As is customary for template analysis, we agreed to create a second version of the template in order to ensure appropriate coverage of emergent themes (Brooks et al., 2014). The data was then re-coded according to this new template. Two team members (KC, RI) discussed all codes and maps after successive iterations of the codebook. We then reviewed text segments from the coded excerpts for each potential theme to ensure the internal homogeneity (extent to which data that belong to a category or theme hold together) and external heterogeneity (extent to which differences between categories or themes are bold and clear) of themes (Patton, 2015). The analysis then mapped these template categories onto the conceptual themes proposed by Smets et al. Template analysis allowed us to demonstrate how the emergent themes interrelated practices, networking and standardization of CTP development, and reinforced the ways in which regulatory and commercialization environments were co-constructed.
4. Findings
Four major themes emerged in exploring what shapes regulatory and commercial environments for CTPs, and how they influence innovation: 1) managing regulatory uncertainty; 2) building a business case; 3) standardizing manufacturing; and 4) networking. We nuance each of these themes in the subsequent sections.
4.1. Managing regulatory uncertainty
Under the Food and Drugs Act (1985), CTPs are generally classified as ‘advance medicinal products’ and follow diverse regulatory pathways depending on their degree of manipulation (Viswanathan and Bubela, 2015). Given the novelty, complexity and diversity of CTPs, it was noted that the conventional pharmaceutical (chemical-based) drug paradigm was ill suited for these products. Interviewees underscored the unpredictability of cellular behavior and the difficulty of categorizing CTPs within the existing regulatory framework as a result. Thus, uncertainty was not only described as an intrinsic characteristic of cell-based products, but also a prominent feature of the Canadian regulatory framework (Von Tigerstom, 2015).
Cells are variable and they're alive. So they can change. So when it comes to efficacy testing, sometimes, it can be difficult. Sometimes you don't necessarily know what the cells are doing in your therapeutic. You just know that it works. (JUN26)
For one participant, Health Canada's flexible, case-by-case approach to CTP approval (Viswanathan and Bubela, 2015) “allow[s] us to interpret those regulations in a more friendly manner for cell therapy products”,(JUN16). In contrast, other participants argued this approach contributes to the regulatory uncertainty often attributable to clinical translation delays. Building a ‘regulatory case’ for CTPs within a regulatory environment that is in a perpetual state of flux proved difficult for many CTP developers. The industry's reliance on guidelines (as opposed to legislation) exacerbated this uncertainty, to say nothing of the guidelines' pharmaceutical-specific orientation (Ridgway, 2015, von Tigerstrom, 2015) that could be inappropriate for regulating biological products such as CTPs.
The regulatory environment is based mostly for drugs where there are very robust procedures for production, but here, we're talking about cell therapy. So the therapy is a cell. So the handling of the cell and the robustness of the process, there's more variability in the processes too. So, the criteria for actually approving a procedure for clinical trials had to adapt to the fact that we're not talking about a drug, we're talking about a cell. (SEP03)
Demonstrating safety and efficacy is an added component to building a ‘regulatory case’, and occurs primarily alongside regulators at pre-CTA meetings. Developers described this process as a ‘guessing game’ with a steep “learning curve”.
I think people wish that there was an easier more formal mechanism to get those answers directly from Health Canada. But I think it is always a struggle that you maybe don't want to reveal your whole process to them and you don't want to ask them questions that make you look like you don't know what you're talking about. So you're often are kind of making educated guesses. (SEP25)
The regulatory authority is an important facilitator in this process, and ongoing consultation with Health Canada was highlighted as a central strategy for managing uncertainty, particularly when “you may be doing something that has not been seen before. [Health Canada] may not necessarily have an answer ready” (JUN10).
Global competiveness was highlighted as another important feature of the Canadian framework that promotes regulation as a necessary step towards innovation:
One of the things that we pitched to companies when we're telling them about Canada is that Health Canada is relatively more progressive to cell therapy regulations than the FDA, or even the EU. (AUG22)
Lack of harmonization between jurisdictions and regulatory frameworks, however, continues to be barrier in the competitive market according to one participant, who compared Health Canada regulations to those at the FDA: “There is a sense in the community, broadly, that the FDA has higher standards than other jurisdictions and I hear people say that a lot, at meetings and stuff. Interestingly, I've seen a couple of specific examples with our own product where Health Canada had a higher standard than the FDA did.” (SEP25)
These differences may be also linked to confusion between manufacturing standardization and harmonization. Whereas standardization is a process intended to unify practices and policies, harmonization seeks convergence across these process in order to identify strategies that foster cross-jurisdictional collaboration (Isasi, 2009, Isasi and Knoppers, 2009, Isasi and Knoppers, 2011). One regulator nuanced this important distinction:
I don't think we are talking about necessarily regulatory harmonization, but more regulatory convergence. (….) Harmonization again makes it sound like we would be more having exactly the same (regulatory) frameworks and that's not possible. So, again (convergence), it's more (…) (about) the same types of steps, the same sort of processes versus the same regulations. (OCT23)
Managing uncertainty is implicated in both regulatory and commercialization efforts for CTPs. Uncertainty with respect to product classification and how best to put forth a strong ‘regulatory case’ can act as barriers to CTP innovation and widespread commercial availability. In the next section, we discuss how some participants perceived the significance of building a strong ‘business case’ in the commercialization process of a new CTP.
4.2. Building a business case
The CTP translation process is characterized by intertwined scientific, regulatory and commercialization challenges. One participant reasoned that, “the regulatory paths are not necessarily well-defined in the sense that, or specifically adapted to stem cell therapeutics and that is slowing down the commercialization” (JUN26). As with the regulatory framework, uncertainty in commercialization manifested in general reluctance to invest in cell therapies. While regulators supported investigators in building a ‘regulatory case’, there was less support for developers hoping to commercialize CTPs through building a successful ‘business case’: “There's absolutely no appetite and no money in Canada to pursue those sorts of [commercial] endeavours. The money is extremely conservative and risk averse, and tends to shy away from cell therapy products (JUL11). Moreover, interviewees were wary of gaps in the clinical translation cycle, and their role in bridging this gap:
Just like any commercialization for anything in the life sciences, there's always a gap, which we refer to as the valley of death in the commercialization process, sort of the proof of concept and clinical trials, actually to get on the market and the industry and often times, it's lack of funding. So it may be, it may not necessarily be, whose responsibility it is to bridge that gap is up for debate, but engaging people in that debate, whether or not the government can facilitate that by providing additional support, resources, whether it's financial or technical, there's certainly a gap that needs to be addressed on some degree. (JUN26)
The archetype business case of the ‘blockbuster’ drug that meets a large commercial need (OCT07) further complicates the case building for CTPs. This is especially true of CTPs that target orphan/rare diseases, and are therefore far less commercially viable than ‘blockbuster’ drugs. The CTP's ‘business case’ is often inappropriately compared and disadvantaged from a commercial standpoint as a result:
There's a perception that a business model or a business case can be made for some cell therapy products. I think that some of it is misguided because a lot of, not a lot, but some cell therapy will never become commercialized because there isn't a particularly good need for it. I′ve seen this happen over the past few years, particularly with regards to bone marrow transplantation. (JUL11)
Participants felt building a ‘business case’ and finding the necessary financial partners to develop and commercialize a potential CTP to be an arduous process. In contrast to Canada's reputation as an enabling regulatory environment, the commercialization environment was characterized as risk-averse in comparison to other countries:
Now, I don't want to sound negative or pessimistic, but what I′m trying to convey is the need for a sense of realism here and that might lead to ways to address the issue [restrictive commercial environment]. I mean we could be leaders in the cell therapy field globally if there was a pool of money that was available that could be used and leveraged to promote this sort of activity [commercialization of CTPs in Canada]. That would solve it overnight. (JUL11)
Academic-industry relationships in CTP development add another layer of commercial complexity, especially as they bring competing objectives into sharp relief: “The goal of the regulatory body is to protect the public health. But there's also a secondary objective of the jurisdictions to get new products on the market to help their population.” (AUG29). For some participants, integrating the private sector and academic research was seen as a last resort:
(…) Once the private sector is involved, it changes the whole, it becomes a business approach rather than an academic approach (…) and that means that decisions are made for purely business reasons, which are very frustrating. I have been involved in a major phase 3 cell development program, which looked incredibly promising and extremely good phase 3 data, all indicators looking spot on, it's a major international company funding this, and two thirds into the way through, they just pull the plug because they decided that their priorities are elsewhere, and just purely economical business, the whole program is shut down. (JUN10)
That the pressure to commercialize encourages academic-industry partnerships was seen as a culturally changing force:
Increasingly, academics have been sort of forced to consider commercialization. All of their granting initiatives have, most of the granting initiatives need to have a commercial aspect to it. That's not something that academics are always keen or interested in pursuing. (JUN27)
Albeit a hallmark of CTP commercialization and ensuring a strong ‘business case’, intellectual property was viewed as a restrictive process that can delimit scientific freedom. As one interviewee noted, a ‘patent thicket’ can mask areas where further research and intellectual opportunities are needed in RM:
There's a very high burden of proof to demonstrate novelty and originality and to show that you're not infringing on prior art, and that you're not just recycling an idea that's been there and that's been done for something else. So I think demonstrating that you really do have something novel is a very high bar now, I think higher than it used to be. And there are a lot of costs associated with it. (JUN 16)
Assessing the market is an intrinsic part of shaping a successful commercial strategy, along with ensuring strategic jurisdictional IP protection. Overcoming the “patent thicket” (JUN27) requires a strategic understanding of the patent landscape such that protection for the CTP can be filed in the most commercially promising jurisdiction:
You also need to look at the competitive space. Who are your competitors, where are they operating, where are they selling, where can you most effectively block them with your IP…So what people often default to is they file everywhere and that becomes extremely expensive as it moves through the process, but they don't want to leave out any jurisdictions in case, later on, a business strategy indicates that they really should have filed in Japan. (JUN27)
Yet expertise in filing commercially strategic patents among CTP developers was markedly lacking. One participant described the consequences of this:
(…) there's an educational gap that is there with researchers. They often don't know how to protect their IP and sometimes have published something…I think they're kind of in a tough situation and because, they are researchers and they need to publish for obvious reasons, but also sometimes there might be regret with publishing something that could potentially have led to some IP. So I think, again, there is a lack of, just, knowledge in the whole process early on, and it's usually at stages where they're not even thinking about product development. (JUN17)
In some cases, obtaining IP protection may not be warranted. Alternative protections were suggested to broaden the value of a potential product or therapy while circumventing the patent thicket, for example clinical adoption: “If you move a cell therapy to the clinic quickly and everyone in the world considers it to be the gold standard treatment, then, everyone is so far behind that having a patent is really not all that important (…) the patenting is only one parameter in creating a viable business.” (SEP09)
Although technology transfer offices (TTOs) are implicated in securing patent protection and licensing/servicing agreements, few respondents rated TTO performance favorably:
(…) technology transfer offices usually aren't that helpful, simply because they are extremely biased and extremely frugal…The frugality makes them biased. They're only going to advance so many ideas because they only have a limited budget that they can go through a whole patenting process with. (JUL09)
At the same time, TTO officers highlighted their own challenges in dealing with academic researchers:
Researchers tend to have a certain level of data and certainty about their new technology before they are willing to share it. However, for patent purposes, the amount of data and proof that you require is a little bit lower than what is required for a scientific publication. So we do try to do a lot of outreach and ensure that researchers know to come and at least talk to us before they publish or present. It's also very challenging, often times, when researchers have students, they want the students to go to a conference and present an abstract, which really only contains a small part of their technology, but it may be enough to impede their chances for a patent. So, again, we try to work with them early on in the process. The flip side of that is that if we're filing for patent applications too early without sufficient data, the applications may not be as strong as they need to be to get through the whole application process and results in an un-issued patent. (OCT06)
While researchers aim to broaden the value of CTPs beyond IP protection, our respondents felt overwhelmed by the pressure to commercialize CTPs (Levesque et al., 2014), and reported few other options other than academic-industry partnerships to help them build successful business ‘cases’.
4.3. Standardized manufacturing
It was not surprising that the development of robust standards to accelerate the clinical translation of CTPs were among participants' major concerns given their association with GMP facilities. The goal of the manufacturing process is to produce scalable, comparable, safe and effective products (French et al., 2013, Webster et al., 2011). Similar to their strategy for managing regulatory uncertainty, participants demonstrated ‘lesson learning’ and ‘pooling of resources’ to overcome the substantial resources needed to standardize production. Facility staff felt, however, that regulatory and technical standards were designed to meet the resource capacities of the industry/private sector, which differs considerably from academia. Such standards directly affect compliance, particularly with respect to GMP requirements. Staff questioned the possibility of aligning GMP and standardized manufacturing practices across facilities: “Well, there needs to be no alignment between GMP facilities. Because again, the definition of GMP is largely empirical and evidence-based (JUL09).”Participants further highlighted the role of standardized manufacturing in addressing funding and expertise issues:
Toxicology testing, safety testing, (inaudible) agent testing, those are all good things that are required by regulation, but if you were working in a lab where you have no experience in the translational trajectory, then, you wouldn't be able to, it would be quite a hassle to being a product to market. You need to have the right collaborators; you need to be able to pay for consultants, to give the advice that you need to get to get the paperwork to meet the requirements. (SEP03)
Indeed, limited resources were identified as a major obstacle for GMP facilities in particular:
Well, it's two things. Usually, it's money to build the facility, which is a big amount of money, and then they get some money for research, that's another allotment of funds, but there is no money to ensure compliance. So they try to do compliance by stealing some money from research or somehow covering or filling the gap or hoping that they can get their clients to fill that gap. (AUG29)
Yet, there was consensus that resource barriers were not insurmountable:
Yes, I think what I′d like to add is we can solve this problem overnight. All it needs is money. We have expertise. I mean the scientific expertise around the country is formidable. The clinical expertise around the country is formidable. We have a strong history of doing phase one, two, three clinical trials with drugs. So drug development, there's a long history of that. And that's a model for moving this forward. We can easily put those components together. We've got a hundred million dollars invested through federal money and other money, in infrastructure, GMP facilities and so on. All we need is funding to overcome the valley of death (…) (JUL11)
CTP manufacturing standards have not been developed in Canada or elsewhere as yet, but are actively negotiated at the practice level among GMP facility staff, researchers and regulators. Indeed, the need to develop more uniform ways to achieve GMP compliance and to align manufacturing practices to that end was only partially recognized by stakeholders.
4.4. Networking
Networking helped establish a common set of principles for CTP clinical translation to be applied across facilities, and was especially useful for managing the regulatory and commercial uncertainty discussed at length above. The infancy of the RM field and novelty of CTP development were determining factors that enabled participants to create a highly networked community:
So, having to work in a field that's really at the edge of science, all these scientists have worked very collegially, all together, since the beginning. And so they started really to see that working together, bringing resources together, would allow them to have more efficient operation with the idea of reducing the costs, reducing also the risk of the methodologies, and also making all those practices, best practices, robust, and also, reproducible from center to center. (OCT03)
Pooling resources and expertise within networks, namely by sharing ‘lessons learned’, also helped to fill gaps in regulatory guidance for CTP developers. While common standards are developed primarily to reduce costs associated with developing CTPs, they also aim to simplify, if not clarify, the current regulatory environment through a process of ‘lessons learning’. Initiatives such as CellCAN illustrate this:
So, with CellCan, what we're trying to do is to have some sort of a, make our own GMP guidelines a little less gray. And so we're coming up with sort of our own standards and hopefully, that would become the standard in Canada (…) that's sort of the plan we have. So the guidelines are (…) kind of gray, and vague, (and then) will be less vague and a little more detailed (…) (OCT09)
Networking was effective for engaging with the regulatory authority to raise awareness of the regulatory issues that facilities face:
Well, we hope to actually open a more direct and a more continuous dialogue with Health Canada, so that there's, again, a better understanding, but from both sides. Both sides have to understand what is the challenge and so that both sides can find a middle ground where [the regulation] specific enough, but not too specific so that it doesn't start to prevent a therapy to go through the system. So it's an equilibrium that has to be reached, but at the end, it's really CellCan's second role will be definitely to favor, to induce and to find, to create opportunities for continuous dialogue with Health Canada. (OCT03)
More direct engagement with regulators led to a series of workshops and conferences where stakeholders jointly developed guidance documents specific to cell therapies (JUN16), and proposed templates for interpreting regulatory requirements. Some regulators remained cautious, fearing other stakeholders would undermine their legitimacy in the absence of formal, national guidance:
I think our role, through Health Canada, again, is through what we do as the regulators. The approval, to be seen as an enabler, not a barrier to research and allowing these clinical trials and reviewing the clinical trials, as well as our role when eventually it comes to submissions in the future…Again, we have to make sure that we know what our role is and we don't go out beyond what we have the authority to do as the regulator and as Health Canada. (OCT23)
Networking also identified potential market opportunities and financial partners. For instance:
So the reason [name of facility] has become an opinion leader is that we have knowledge of the entire breadth of the regenerative medicine space, from research-based discovery and what is happening at universities right through commercial products that are already on the market as well as everything else in-between. Because we also have a very broad network in the academic and research space as well as the industry space, we are able to connect with anyone in the industry that may have any questions. And so we developed a very deep expertise and understanding of the market dynamics around cell therapies. And we're then able to craft the technologies that we develop to meet market needs, more specifically future market needs. And because we have this expertise we created, the broader regenerative medicine community at large looks at us as a key opinion leader in the space. (AUG22)
The ‘network’ can also provide ‘insider’ information not otherwise accessible, such as emerging information technologies not yet available on the market:
Yeah, so, in the example of [name of facility], we have an agreement that allows to see disclosures and IT related to regenerative medicine technologies, we evaluate that IP with respect to its commercial potential, we provide that IP in some cases to industry consortium members so they can either license it from institutions directly or we decide that the technology requires further development and upon this development, it will have further value, in which case we in-license it from the technology transfer offices. We create companies around the technologies by bundling technologies from many different places to make the companies we create more competitive and, as part of that, the institutions and the technology transfer offices benefit from the royalties and equity-related relationships. (JUL02)
Finally, networking confers mutual support among CTP stakeholders, and leverages collective strengths required to overcome both regulatory and commercial hurdles. The same collaborative spaces that facilitate understanding of the regulatory and commercial norms also reshape the frameworks used to guide these norms. This is particularly true of new standardization practices resulting from networks and ‘lesson learning’.
5. Discussion
Taken together, the three organizational mechanisms (situated improvising, normative network reorientation and unobtrusive embedding) can be identified in the regulatory and commercialization environments for CTP development in Canada. The bidirectional relationship between regulation and commercialization is a noteworthy finding in this regard, and the particular ways in which standardized practices are sustained through networking, lesson learning and co-construction of both business and regulatory ‘cases’. Surprisingly, the same collaborative spaces that gave rise to co-constructed, standardized practices among CTP developers and regulators also established boundaries meant to differentiate CTP regulation and commercialization from other biologics such as pharmaceutical drugs.
The Stem Cell Network, CellCAN and the Centre for Commercialization of Regenerative Medicine (CCRM) initiatives, amongst others, embody networking in action. We found CTP standardization arose as the byproduct of an ongoing, parallel learning processes among GMP facilities. Situated improvising helped explain the onset of this learning process, whereby developers and regulators forged partnerships to build successful ‘cases’ in preparation for regulatory approval. This partnership alleviated much of the uncertainty that surrounded approval processes. The pre-CTA meetings, in particular, marked a reconciliatory point for investigators and regulators. Regulators acknowledged their limited expertise and knowledge of emerging CTPs, and thereby consulted CTP developers working in GMP facilities on developing best practices to inform regulation.
Networking resulted in two primary outcomes based on our findings. First, regulatory guidelines and best practices emerged from a bidirectional, ‘lesson learning’ process in which the exchange of information between CTP developers and regulators contributed to policy development. Although some aspects of the regulatory framework are still under development, networking served to bring academic researchers involved in CTP development and the RM industry into closer confines to ensure future CTPs adhere to forthcoming regulatory standards. Networking allowed for discussions and understanding of unique (if not conflicting) logics and cultures that distinguish academia from the industrial sector. While some participants expressed caution in facilitating such close academic-industry partnerships, others reinforced how networking was instrumental in assisting CTP developers with building strategic ‘business cases’. Networking with industry was furthermore promulgated as a strategic option for navigating an especially complex IP landscape. We found CTP developers reversed the typical sequence of innovation. That is, it was commonplace to secure investors after filing a patent application. Identifying investors vis-à-vis networking prior to developing CTPs therefore supplanted the need for TTOs who, according to some participants, lacked the necessary resources and expertise to assist developers in overcoming commercialization hurdles.
Second, networking between CTP development and regulatory/commercial actors signaled a change in the organizational culture of emerging biotechnology regulation in Canada. It effectively challenged the traditional, asymmetric relationship between the ‘regulator’ and ‘regulated’. Health Canada developed best practice guidelines in consultation with CTP developers, and were inspired by the daily practices at CTP-GMP facilities. The trust inculcated through sustained collaboration and change in regulatory ‘culture’ for CTPs is attributable in part to the small community of RM innovators, policy makers and commercial actors.
6. Strengths and limitations of the study
Our study presented some limitations. Both team members (RI, KC) reviewed summaries and matrix tables for each facility, however, further triangulation of other sources of data was not feasible due to the proprietary nature of some commercialization information (Shenton, 2004). Second, it was not possible to recruit all participants knowledgeable in commercialization from some facilities. Because interviews are the primary qualitative data source, our inability to include all key stakeholders in the CTP development, manufacturing, regulatory and commercial process for each facility presented the greatest limitation. Technology transfer officers did not participate in the case of one facility because they were minimally involved in the facility's commercialization activities. These recruitment challenges could have impacted the maximum variation sample intended with including all relevant CTP manufacturing, regulatory and commercial stakeholders. In another case, we were not able to recruit the facility's director or CEO, but we obtained relevant qualitative data on commercialization experiences from other interviewees who held high-level positions at this facility.
7. Conclusion
This study aimed to better understand the relationship between the regulatory and commercialization environments for CTPs in Canada, and elucidate their combined effects on CTP innovation. To that end, we focused on Canadian CTP-GMP facilities and the national regulatory authority. The conceptual framework proposed by Smets et al. was used to illustrate how regulatory and commercialization practices shape field-level changes in CTP development based on the daily practices of CTP-GMP facilities. Our study highlights how practices, networking and standardization efforts are intertwined within the regulatory and commercialization environments for CTPs in Canada. Networking initiatives are vital tools to compensate for limited resources, namely funding and expertise in CTP development among investigators and facility staff. Participants in our study were successful networkers in part because of the relatively small community that comprises CTP development. The size of the CTP community and their open relationships with regulatory authorities were fundamental factors enabling innovation in this field. Networking, insofar as it involves legitimating key regulatory and research stakeholders is essential for facilitating product development. Alongside efforts to align academic and industry priorities, support from other stakeholders such as patient advocacy groups also helped investigators and GMP facilities assess the need for specific CTPs. This enhanced investigators' ability to establish persuasive regulatory and business ‘cases’ which, ultimately, dictates CTP adoption.
Through co-production of regulatory standards and practices, CTP developers and regulators engaged in what we discovered was a unique reverse governance process. Put simply, regulators relied on the scientific expertise of CTP researchers and developers to help establish a regulatory framework for governing the future of CTP innovation. The merits of such bidirectional governance are twofold: regulations for emerging biotechnologies may better reflect the operational and innovation-oriented priorities of scientists working at their helm, while also encouraging collaboration among the various stakeholders involved at each point in the biotechnology innovation continuum.
Lastly, participants agreed Canada has historically, and will continue to be a global leader in the CTP and RM fields
because, from an international perspective, Canadians are viewed as very friendly and helpful. And so if you combine the three, the trifecta of expertise and a global reputation for expertise and excellence (…) combined with being Canadian, that's actually made our ability to grow very rapidly (…) (AUG22)
This is in no small part due to the way regulation and commercialization environments have emerged as co-produced modes of governance within the RM field in Canada.
Appendix A. General interview guide based on the conceptual framework proposed by Smets et al.
| As you may know, our research project, entitled “The clinical translation of stem cell research in Canada: the role of stem cell biorepositories in shaping the policy, commercialization and regulatory environments” aims to understand how practices within GMP cell manufacturing facilities shape current policy, regulatory and commercialization frameworks, both nationally and globally. We would therefore like to ask you questions regarding the lessons you have learned as a scientist working in a biorepository focused on the clinical translation of stem cell research, particularly with respect to regulatory requirements that tend to shape the clinical translation of stem cell products. We would like to learn as well your thoughts regarding national and international initiatives aimed at standardizing the use of stem cell products in clinical research. More specifically, for this interview, we would like to cover the following issues:
|
| Experiences with policy and regulatory requirements and lessons learned |
|
|
|
|
|
|
| Commercialization issues |
|
|
|
|
|
|
| Relationships with stakeholders and networking initiatives in the course of the development process |
|
|
|
|
|
|
Appendix B. Interview guide for technology transfer
| As you may know, our research project, entitled “The clinical translation of stem cell research in Canada: the role of stem cell biorepositories in shaping the policy, commercialization and regulatory environments" aims to understand how practices within GMP cell manufacturing facilities shape current policy, regulatory and commercialization frameworks, both nationally and globally. We would therefore like to ask you questions regarding your role as regulators with respect to the clinical translation of stem cell research, particularly with respect to regulatory requirements that tend to shape the clinical translation of stem cell products. We would like to learn as well your thoughts regarding national and international initiatives aimed at standardizing the use of stem cell products in clinical research. More specifically, for this interview, we would like to cover the following issues:
|
| Challenges regarding the commercialization of stem cell-based products and therapies |
|
|
|
|
|
| Networking activities |
|
|
|
|
|
|
| Wider policy/regulatory environment |
|
|
|
|
| Is there anything you would like to add? |
Appendix C. Interview guide for regulatory officers
| As you may know, our research project, entitled “The clinical translation of stem cell research in Canada: the role of stem cell biorepositories in shaping the policy, commercialization and regulatory environments” aims to understand how practices within GMP cell manufacturing facilities shape current policy, regulatory and commercialization frameworks, both nationally and globally. We would therefore like to ask you questions regarding your role as regulators with respect to the clinical translation of stem cell research, particularly with respect to regulatory requirements that tend to shape the clinical translation of stem cell products. We would like to learn as well your thoughts regarding national and international initiatives aimed at standardizing the use of stem cell products in clinical research. |
More specifically, for this interview, we would like to cover the following issues:
|
| Challenges regarding the commercialization of stem cell-based products and therapies |
|
|
|
|
|
|
| What steps did you take to address these problems? How did that work out? |
|
|
|
|
|
|
|
|
|
|
References
- Abbasalizadeh S., Baharvand H. Technological progress and challenges towards cGMP manufacturing of human pluripotent stem cells based therapeutic products for allogeneic and autologous cell therapies. Biotechnol. Adv. 2013;31(8):1600–1623. doi: 10.1016/j.biotechadv.2013.08.009. [DOI] [PubMed] [Google Scholar]
- Anon . 1985. Food and Drugs Act, Canada. [Google Scholar]
- Bailey A.M., Mendicino M., Au P. An FDA perspective on preclinical development of cell-based regenerative medicine products. Nat. Biotechnol. 2014;32(8):721–723. doi: 10.1038/nbt.2971. [DOI] [PubMed] [Google Scholar]
- Beak C., Isasi R. Regenerative medicine: socio-ethical challenges and regulatory approaches. In: Joly Y., Knoppers B.M., editors. Routledge Handbook of Medical Law and Ethics. Routledge; New York, NY: 2014. pp. 244–268. [Google Scholar]
- Bowen G.A. Naturalistic inquiry and the saturation concept: a research note. Qual. Res. 2008;8(1):137–152. [Google Scholar]
- Brooks J. The utility of template analysis in qualitative psychology research. Qual. Res. Psychol. 2014;12(2):202–222. doi: 10.1080/14780887.2014.955224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carlile P.R. A pragmatic view of knowledge and boundaries: boundary objects in new product development. Organ. Sci. 2002;13(4):442–455. [Google Scholar]
- Eriksson L., Webster A. Standardizing work as a recursive process: shaping the embryonic stem cell field. New Genet. Soc. 2015;34(1):72–88. [Google Scholar]
- French A., Buckler R.L., Brindley D.A. Commercialization of regenerative medicine: learning from spin-outs. Rejuvenation Res. 2013;16(2):164–170. doi: 10.1089/rej.2013.1423. [DOI] [PubMed] [Google Scholar]
- Ginty P.J. Regenerative medicine, resource and regulation: lessons learned from the remedi project. Regen. Med. 2011;6(2):241–253. doi: 10.2217/rme.10.89. [DOI] [PubMed] [Google Scholar]
- Heathman T.R.J. The translation of cell-based therapies: clinical landscape and manufacturing challenges. Regen. Med. 2015;10(1):49–64. doi: 10.2217/rme.14.73. [DOI] [PubMed] [Google Scholar]
- Isasi R., Knoppers B.M. From banking to international governance: fostering innovation in stem cell research. Stem Cells Int. 2011;2011:498132. doi: 10.4061/2011/498132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Isasi R.M. Policy interoperability in stem cell research: demystifying harmonization. Stem Cell Rev. 2009;5(2):108–115. doi: 10.1007/s12015-009-9067-z. [DOI] [PubMed] [Google Scholar]
- Isasi R.M., Knoppers B.M. Governing stem cell banks and registries: emerging issues. Stem Cell Res. 2009;3(2–3):96–105. doi: 10.1016/j.scr.2009.05.003. [DOI] [PubMed] [Google Scholar]
- Kuper A., Lingard L., Levinson W. Critically appraising qualitative research. Br. Med. J. 2008;337 doi: 10.1136/bmj.a1035. a1035–a1035. [DOI] [PubMed] [Google Scholar]
- Lauckner H., Paterson M., Krupa T. The Qualitative Report 2012. 17(Article 25) 2012. Using constructivist case study methodology to understand community development processes: proposed methodological questions to guide the research process; pp. 1–22. [Google Scholar]
- Levesque M. Stem cell research funding policies and dynamic innovation: a survey of open access and commercialization requirements. Stem Cell Rev. 2014;10(4):455–471. doi: 10.1007/s12015-014-9504-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miles M.B., Huberman M.A., Saldaña J. Qualitative data analysis. A methods sourcebook. Z. Pers. 2014;28(4):485–487. [Google Scholar]
- Nature Editorials Fetal tissue research under threat. Nature. 2015;528(December 10):163–164. doi: 10.1038/528163a. [DOI] [PubMed] [Google Scholar]
- Patton M.Q. fourth ed. SAGE Publications; 2014. Qualitative Research and Evaluation Methods. [Google Scholar]
- Patton M.Q. fourth ed. SAGE Publications; Saint Paul: 2015. Qualitative Research and Evaluation Methods: Theory and Practice. [Google Scholar]
- Ridgway A. BMC Blog Network; 2015. Stem cell research & therapy celebrates its fifth anniversary. [Google Scholar]
- Ritchie J., Spencer L. Qualitative data analysis for applied policy research. In: Huberman M., Miles M.B., editors. The Qualitative Researcher's Companion. SAGE Publications; 2002. [Google Scholar]
- Robinson O.C. Sampling in interview-based qualitative research: a theoretical and practical guide. Qual. Res. Psychol. 2013;11(1):25–41. [Google Scholar]
- Rosemann A. Standardization as situation-specific achievement: regulatory diversity and the production of value in intercontinental collaborations in stem cell medicine. Soc. Sci. Med. 2014;122:72–80. doi: 10.1016/j.socscimed.2014.10.018. [DOI] [PubMed] [Google Scholar]
- Shenton A. Strategies for ensuring trustworthiness in qualitative research projects. Educ. Inf. 2004;22:63–75. [Google Scholar]
- Smets M., Morris T., Greenwood R. From practice to field: a multilevel model of practice driven institutional change. Acad. Manag. J. 2012;55(4):877–904. [Google Scholar]
- von Tigerstrom B. Human tissue legislation and a new medical paradigm: governing tissue engineering in Canada. McGill J. Law Health. 2015;8(2):S1–S56. [Google Scholar]
- Viswanathan S., Bubela T. Current practices and reform proposals for the regulation of advanced medicinal products in Canada. Regen. Med. 2015;10(5):647–663. doi: 10.2217/rme.15.28. [DOI] [PubMed] [Google Scholar]
- Von Tigerstom B. Revising the regulation of stem cell-based therapies: critical assessment of potential models. Food Drug Law J. 2015;70(2):315–337. [PubMed] [Google Scholar]
- Webster A., Eriksson L. Governance-by-standards in the field of stem cells: managing uncertainty in the world of “basic innovation” uncertainty in the world of “basic innovation”. New Genet. Soc. 2008;27(2):99–111. [Google Scholar]
- Webster A., Haddad C., Waldby C. Experimental heterogeneity and standardisation: stem cell products and the clinical trial process. BioSocieties. 2011;6(4):401–419. [Google Scholar]
- Zietsma C., Lawrence T.B. Institutional work in the transformation of an organizational field: the interplay of boundary work and practice work. Adm. Sci. Q. 2010;55(2):189–221. [Google Scholar]