Abstract
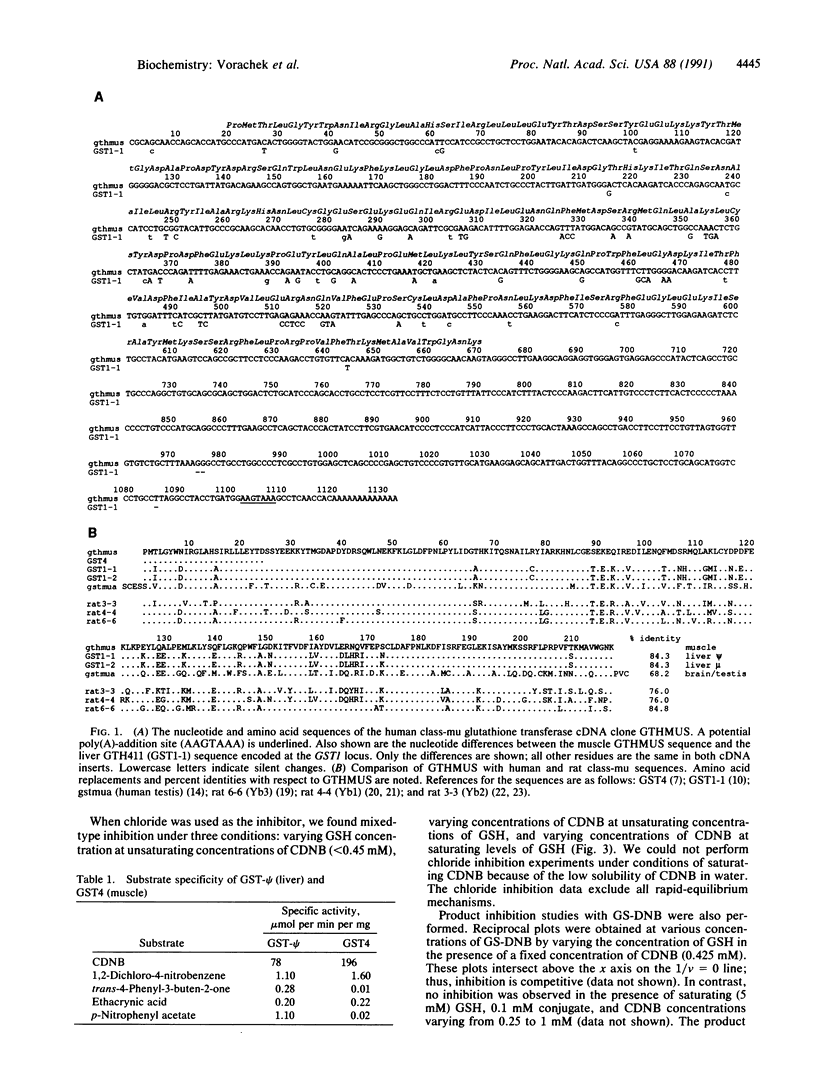
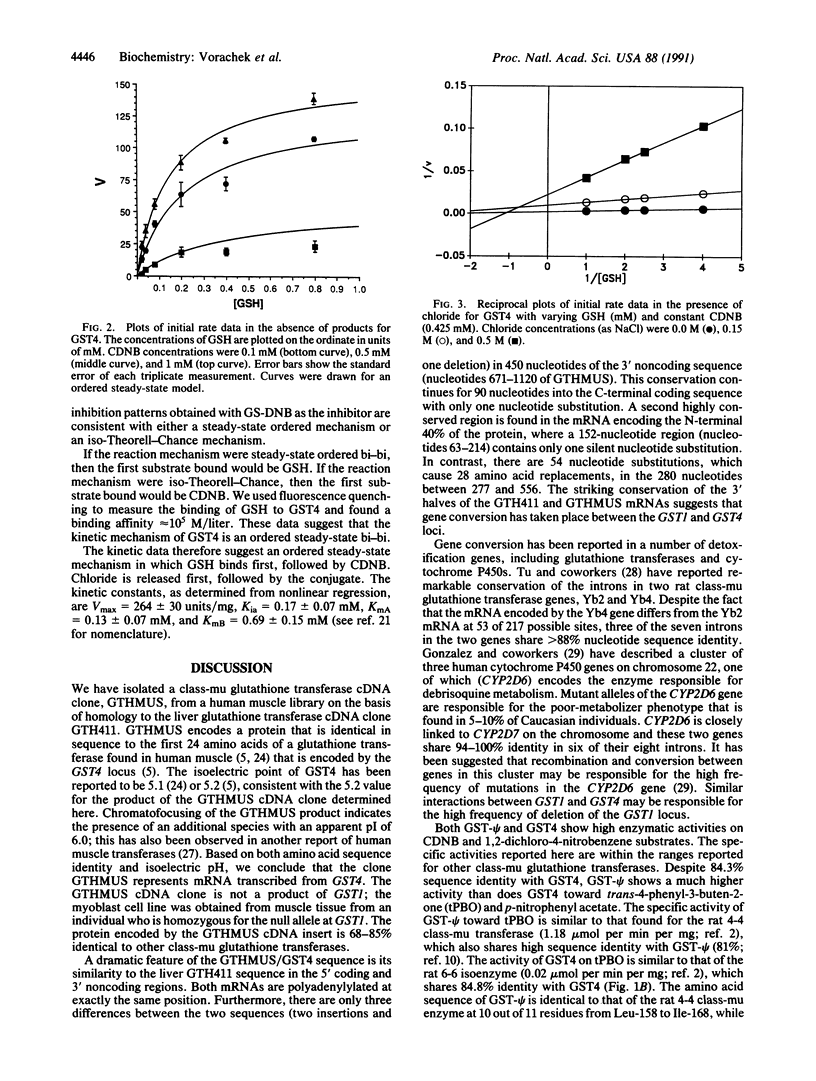
A class-mu glutathione transferase cDNA clone, GTHMUS, was isolated from human myoblasts and its sequence was determined. The sequence predicts a protein of molecular weight 25,599 whose 24 amino-terminal residues are identical to those of the class-mu isoenzyme expressed from the GST4 locus. The GTHMUS cDNA shares 93.7% nucleotide sequence identity with a human liver cDNA clone, GTH411, that is encoded at the GST1 locus. Comparison of the liver and muscle cDNA sequences shows two regions of remarkable sequence conservation: a 140-nucleotide region in the 5' coding portion of the molecule that has a single silent nucleotide substitution, and a 550-nucleotide region, including the entire 3' noncoding region, that has only three nucleotide substitutions or deletions. This sequence conservation suggests that gene conversion has occurred between the human GST1 and GST4 glutathione transferase gene loci. The human muscle and liver glutathione transferase clones GTHMUS and GTH411 have been expressed in Escherichia coli. The kinetic mechanism of the muscle enzyme was examined in product inhibition studies. The inhibition patterns are best modeled by a steady-state ordered bi-bi reaction mechanism. Glutathione is the first substrate bound and chloride ion is the first product released. Chloride ion inhibits the muscle enzyme.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abramovitz M., Listowsky I. Selective expression of a unique glutathione S-transferase Yb3 gene in rat brain. J Biol Chem. 1987 Jun 5;262(16):7770–7773. [PubMed] [Google Scholar]
- Alin P., Mannervik B., Jörnvall H. Cytosolic rat liver glutathione transferase 4-4. Primary structure of the protein reveals extensive differences between homologous glutathione transferases of classes alpha and mu. Eur J Biochem. 1986 Apr 15;156(2):343–350. doi: 10.1111/j.1432-1033.1986.tb09588.x. [DOI] [PubMed] [Google Scholar]
- Board P. G. Biochemical genetics of glutathione-S-transferase in man. Am J Hum Genet. 1981 Jan;33(1):36–43. [PMC free article] [PubMed] [Google Scholar]
- Board P. G. Gene deletion and partial deficiency of the glutathione S-transferase (ligandin) system in man. FEBS Lett. 1981 Nov 30;135(1):12–14. doi: 10.1016/0014-5793(81)80933-7. [DOI] [PubMed] [Google Scholar]
- Board P. G., Suzuki T., Shaw D. C. Human muscle glutathione S-transferase (GST-4) shows close homology to human liver GST-1. Biochim Biophys Acta. 1988 Apr 14;953(3):214–217. doi: 10.1016/0167-4838(88)90027-1. [DOI] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Campbell E., Takahashi Y., Abramovitz M., Peretz M., Listowsky I. A distinct human testis and brain mu-class glutathione S-transferase. Molecular cloning and characterization of a form present even in individuals lacking hepatic type mu isoenzymes. J Biol Chem. 1990 Jun 5;265(16):9188–9193. [PubMed] [Google Scholar]
- DeJong J. L., Chang C. M., Whang-Peng J., Knutsen T., Tu C. P. The human liver glutathione S-transferase gene superfamily: expression and chromosome mapping of an Hb subunit cDNA. Nucleic Acids Res. 1988 Sep 12;16(17):8541–8554. doi: 10.1093/nar/16.17.8541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ding G. J., Ding V. D., Rodkey J. A., Bennett C. D., Lu A. Y., Pickett C. B. Rat liver glutathione S-transferases. DNA sequence analysis of a Yb2 cDNA clone and regulation of the Yb1 and Yb2 mRNAs by phenobarbital. J Biol Chem. 1986 Jun 15;261(17):7952–7957. [PubMed] [Google Scholar]
- Ding G. J., Lu A. Y., Pickett C. B. Rat liver glutathione S-transferases. Nucleotide sequence analysis of a Yb1 cDNA clone and prediction of the complete amino acid sequence of the Yb1 subunit. J Biol Chem. 1985 Oct 25;260(24):13268–13271. [PubMed] [Google Scholar]
- Habig W. H., Jakoby W. B. Assays for differentiation of glutathione S-transferases. Methods Enzymol. 1981;77:398–405. doi: 10.1016/s0076-6879(81)77053-8. [DOI] [PubMed] [Google Scholar]
- Hirrell P. A., Hume R., Fryer A. A., Collins M. F., Drew R., Bradwell A. R., Strange R. C. Studies on the developmental expression of glutathione S-transferase isoenzymes in human heart and diaphragm. Biochim Biophys Acta. 1987 Oct 15;915(3):371–377. doi: 10.1016/0167-4838(87)90022-7. [DOI] [PubMed] [Google Scholar]
- Ivanetich K. M., Goold R. D. A rapid equilibrium random sequential bi-bi mechanism for human placental glutathione S-transferase. Biochim Biophys Acta. 1989 Sep 14;998(1):7–13. doi: 10.1016/0167-4838(89)90111-8. [DOI] [PubMed] [Google Scholar]
- Jakoby W. B. The glutathione S-transferases: a group of multifunctional detoxification proteins. Adv Enzymol Relat Areas Mol Biol. 1978;46:383–414. doi: 10.1002/9780470122914.ch6. [DOI] [PubMed] [Google Scholar]
- Kimura S., Umeno M., Skoda R. C., Meyer U. A., Gonzalez F. J. The human debrisoquine 4-hydroxylase (CYP2D) locus: sequence and identification of the polymorphic CYP2D6 gene, a related gene, and a pseudogene. Am J Hum Genet. 1989 Dec;45(6):889–904. [PMC free article] [PubMed] [Google Scholar]
- Lai H. C., Qian B., Grove G., Tu C. P. Gene expression of rat glutathione S-transferases. Evidence for gene conversion in the evolution of the Yb multigene family. J Biol Chem. 1988 Aug 15;263(23):11389–11395. [PubMed] [Google Scholar]
- Lai H. C., Tu C. P. Rat glutathione S-transferases supergene family. Characterization of an anionic Yb subunit cDNA clone. J Biol Chem. 1986 Oct 15;261(29):13793–13799. [PubMed] [Google Scholar]
- Pabst M. J., Habig W. H., Jakoby W. B. Glutathione S-transferase A. A novel kinetic mechanism in which the major reaction pathway depends on substrate concentration. J Biol Chem. 1974 Nov 25;249(22):7140–7147. [PubMed] [Google Scholar]
- Schramm V. L., McCluskey R., Emig F. A., Litwack G. Kinetic studies and active site-binding properties of glutathione S-transferase using spin-labeled glutathione, a product analogue. J Biol Chem. 1984 Jan 25;259(2):714–722. [PubMed] [Google Scholar]
- Seidegård J., Guthenberg C., Pero R. W., Mannervik B. The trans-stilbene oxide-active glutathione transferase in human mononuclear leucocytes is identical with the hepatic glutathione transferase mu. Biochem J. 1987 Sep 15;246(3):783–785. doi: 10.1042/bj2460783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seidegård J., Pero R. W., Miller D. G., Beattie E. J. A glutathione transferase in human leukocytes as a marker for the susceptibility to lung cancer. Carcinogenesis. 1986 May;7(5):751–753. doi: 10.1093/carcin/7.5.751. [DOI] [PubMed] [Google Scholar]
- Seidegård J., Pero R. W. The hereditary transmission of high glutathione transferase activity towards trans-stilbene oxide in human mononuclear leukocytes. Hum Genet. 1985;69(1):66–68. doi: 10.1007/BF00295531. [DOI] [PubMed] [Google Scholar]
- Seidegård J., Vorachek W. R., Pero R. W., Pearson W. R. Hereditary differences in the expression of the human glutathione transferase active on trans-stilbene oxide are due to a gene deletion. Proc Natl Acad Sci U S A. 1988 Oct;85(19):7293–7297. doi: 10.1073/pnas.85.19.7293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shatzman A. R., Rosenberg M. Efficient expression of heterologous genes in Escherichia coli. The pAS vector system and its applications. Ann N Y Acad Sci. 1986;478:233–248. doi: 10.1111/j.1749-6632.1986.tb15534.x. [DOI] [PubMed] [Google Scholar]
- Singh S. V., Ahmad H., Kurosky A., Awasthi Y. C. Purification and characterization of unique glutathione S-transferases from human muscle. Arch Biochem Biophys. 1988 Jul;264(1):13–22. doi: 10.1016/0003-9861(88)90564-4. [DOI] [PubMed] [Google Scholar]
- Singh S. V., Kurosky A., Awasthi Y. C. Human liver glutathione S-transferase psi. Chemical characterization and secondary-structure comparison with other mammalian glutathione S-transferases. Biochem J. 1987 Apr 1;243(1):61–67. doi: 10.1042/bj2430061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki T., Coggan M., Shaw D. C., Board P. G. Electrophoretic and immunological analysis of human glutathione S-transferase isozymes. Ann Hum Genet. 1987 May;51(Pt 2):95–106. doi: 10.1111/j.1469-1809.1987.tb01051.x. [DOI] [PubMed] [Google Scholar]
- Warholm M., Guthenberg C., Mannervik B. Molecular and catalytic properties of glutathione transferase mu from human liver: an enzyme efficiently conjugating epoxides. Biochemistry. 1983 Jul 19;22(15):3610–3617. doi: 10.1021/bi00284a011. [DOI] [PubMed] [Google Scholar]



