Abstract
BACKGROUND
Survival is significantly reduced by either hypotension or hypoxia during the prehospital management of major traumatic brain injury (TBI). However, only a handful of small studies have investigated the influence of the combination of both hypotension and hypoxia occurring together. Objective: In patients with major TBI, we evaluated the associations between mortality and prehospital hypotension and hypoxia, both separately and in combination.
METHODS
All moderate/severe TBI cases in the pre-implementation cohort of the Excellence in Prehospital Injury Care (EPIC) Study (a statewide, before/after, controlled study of the impact of implementing the prehospital TBI treatment guidelines) from 1/1/07–3/31/14 were evaluated [exclusions: age<10 years; prehospital oxygen saturation ≤10%; prehospital systolic blood pressure (SBP) <40 or >200mmHg]. The relationship between mortality and hypotension (SBP <90mmHg) and/or hypoxia (saturation <90%) was assessed using multivariable logistic regression, controlling for Injury Severity Score, head region severity, injury type (blunt versus penetrating), age, sex, race, ethnicity, payer, inter-hospital transfer, and trauma center.
RESULTS
Among the 13,151 cases that met inclusion criteria [Median age: 45; Male: 68.6%], 11,545 (87.8%) had neither hypotension nor hypoxia, 604 (4.6%) had hypotension only, 790 (6.0%) had hypoxia only, and 212 (1.6%) had both hypotension and hypoxia. Mortality for the four study cohorts was 5.6%, 20.7%, 28.1%, and 43.9%, respectively. The crude and adjusted odds ratios (cOR/aOR) for death within the cohorts, utilizing the patients with neither hypotension nor hypoxia as the reference, were 4.4/2.5, 6.6/3.0, and 13.2/6.1, respectively. Evaluation for an interaction between hypotension and hypoxia revealed that the effects are additive on the log odds of death.
CONCLUSION
In this statewide analysis of major TBI, combined prehospital hypotension/hypoxia were associated with dramatically increased mortality. This effect on survival persisted even after controlling for multiple potential confounders. In fact, the adjusted odds of death in patients with both hypotension and hypoxia was more than two times greater than those with either hypotension or hypoxia alone. These findings seem supportive of the emphasis on aggressive prevention and treatment of hypotension and hypoxia reflected in the current EMS TBI treatment guidelines but clearly reveal the need for further study to determine their impact on outcome.
INTRODUCTION
Background and Significance
Traumatic Brain Injury (TBI) is a massive public health problem leading to more than 50,000 deaths and enormous healthcare expenditures each year in the U.S.1,2 The Centers for Disease Control (CDC) estimates that at least 5.3 million Americans, approximately 2% of the U.S. population, are living with a major, permanent TBI-related disability.2,3
During the prehospital care of TBI patients, hypoxia occurs frequently4–9 and significantly increases mortality.6,7,10–16 It is independently associated with a higher risk of death even if the hypoxemic episode is reflected by only a single measurement of low oxygen saturation.10,12,17 Stocchetti found that the presence of prehospital hypoxia more than tripled the likelihood of death among victims of severe TBI.6 Hypotension is also very common early in the care of TBI7,10,11,18 and significantly impacts survival.6,10,11,14,15,18–39 A single episode of hypotension doubles mortality and this risk increases dramatically with repeated episodes (an odds ratio of 8.1 for death in one study).26
While the negative effect of hypotension and hypoxia have been well documented in the literature, little is known about the combination of both. Thus, it is unknown whether, together, they have no additional effect, an additive effect, or some intermediate impact on outcomes. Even though it is known that hypotension and hypoxia each independently increases mortality, this is not the same as showing that the combination of the two is additive in its effect in patients who actually experience both. In fact, some authors have suggested that since, at the cellular level, there are great similarities in the effect of hypoxia and hypotension (reduced oxygen deliver to the neuron), then having both may add little to the risk of death since the physiological insult may be similar with either or both.16,22,26 With the exception of a meta-analysis that had major issues with study heterogeneity and missing data,39 the reports that have looked at the impact of hypotension combined with hypoxia in TBI have had very few cases.6,16,22,26,28,35,40 Furthermore, even less is known about this issue in the prehospital setting. To our knowledge only two previous studies specifically evaluated the hypotension/hypoxia combination utilizing prehospital data.6,16 A key reason for evaluating the impact of blood pressure and oxygenation measured before hospital arrival is because the injured brain is so highly sensitive to changes in perfusion and oxygenation and the timeframe during which neuronal damage begins is so short. It is well established that secondary brain injury is initiated by even brief periods of compromised blood flow or hypoxia.20,22,23,28,35,40–43 Thus, decreased perfusion and/or hypoxia occurring during the prehospital time interval may have a profound impact on outcome.
Goal
The objective of this investigation was to evaluate the association between survival and prehospital hypotension, hypoxia, or both in patients with major TBI.41
Specific Hypothesis: In major TBI, the combination of both prehospital hypotension (SBP <90 mmHg) and hypoxia (oxygen saturation <90%) will have additional negative influence on survival compared to either factor alone.
MATERIALS AND METHODS
The EPIC Study has been described in detail elsewhere.41 It is funded by the National Institutes of Health [NINDS-R01NS071049 (“EPIC”) and 3R01NS071049-S1 (“EPIC4Kids”)] and, while not a randomized trial, it is registered at ClinicalTrials.gov (#NCT01339702). Rather than reiterating the details of the parent study, here we limit the description to the design attributes relevant to this specific evaluation.
Study Setting
The EPIC Study is evaluating the impact of implementing the prehospital TBI guidelines42–45 in patients with moderate or severe (“major”) TBI throughout the state of Arizona using a before-after, controlled, multisystem, observational design. The patients in this evaluation are in the pre-implementation cohort of EPIC (treated by an EMS agency between 1/1/2007 and 3/31/2014 without receiving EPIC study interventions). Cases in the interventional cohort were excluded for two reasons. First, inclusion of post-intervention cases in this observational evaluation would encroach on several of the main hypotheses of the primary parent study and the analysis plan does not allow for multiple “looks” at the interventional data. Second, since two of the emphases of guideline implementation are the prevention and aggressive treatment of hypotension and hypoxia, including post-implementation cases might significantly bias the results.
Study Design and Participants
The EPIC Database is made up of the subset of patients from the Arizona State Trauma Registry (ASTR) meeting EPIC Study criteria for major TBI (described below). The ASTR has detailed in-hospital data on all trauma patients taken to the eight state-designated level I trauma centers in Arizona. The EPIC Database contains both ASTR data and linked, detailed prehospital data. The necessary regulatory approvals for the EPIC project have been obtained from the Arizona Department of Health Services (ADHS) and the State Attorney General. The University of Arizona Institutional Review Board and the ADHS Human Subjects Review Board have approved the project and have determined that, by virtue of being a public health initiative, neither the interventions nor their evaluation constitute human subjects research and have approved the publication of de-identified data.
Inclusion criteria: Patients age 10 years or older with physical trauma who have trauma center diagnosis(es) consistent with TBI (either isolated or multisystem trauma that includes TBI) and meet at least one of the following definitions for moderate or severe TBI: a) Centers for Disease Control (CDC) Barell Matrix-Type 1, b) International Classification of Diseases-Version 9 (ICD-9) head region severity score ≥3, c) Abbreviated Injury Scale (AIS)-head region score ≥3.41
Exclusions for this analysis: Age <10 years, missing EMS systolic blood pressure (SBP), oxygen saturation, or other important confounders; lowest SBP <40mmHg or >200mmHg, oxygen saturation ≤10 percent, and patients who were transferred out of the reporting trauma center.
The age cutoff of less than 10 years was utilized primarily to simplify the analysis. Under age 10, hypotension is defined as an SBP < [70 mmHg + (age X 2)].43,45 Given that this represents only 6.8% of the EPIC population, this would markedly increase the complexity of the analysis without substantially adding to the size of the study cohort. Age <10 years also makes sense as an age cut-off since we are not yet looking at treatment (the purpose of the main study). The related cut-offs (like age <15 having ventilation rates = 20 BPM versus age ≥15 being 10 BPM) are not relevant to this analysis.
Interventions
This is an evaluation of the pre-implementation EPIC cohort and entails no interventions.
Outcome
The main outcome is survival to hospital discharge.41
Data Collection, Abstraction, and Linkage
The Arizona State Trauma Registry (ASTR) contains extensive trauma center data on all patients taken to the designated Level I trauma centers in the state. From the ASTR, all cases meeting study criteria (described above) are entered into the EPIC Database. Each participating Emergency Medical Services (EMS) agency then receives a list of the EPIC patients that were cared for in their system. The cases are matched by incident date, name, and other patient identifiers. Either scanned copies [paper-based patient care records (PCRs)] or electronic data files (electronic PCRs) are then sent to the EPIC Study Data Center. Database personnel then utilize a comprehensive data collection tool to abstract the data and enter it into the EPIC Database. This provides an extensive, linked dataset for study patients that includes both prehospital and trauma center data. The entire process of case identification, EMS/trauma center linkage, accessing EMS PCRs, trauma center and EMS data entry, data quality management, and the structure of the EPIC database are described in detail in the study methods paper.41 Over 20,000 cases have been enrolled in EPIC and over 31,000 EMS PCRs have been entered into the database. There are more PCRs than cases because many patients are cared for by more than one EMS agency. The successful linkage rate is exceptionally high (for example, throughout the study, the rate of cases with missing EMS SBP has been consistently <5%).
Blood pressure and oxygen saturation data were evaluated by including every documented prehospital measurement for each patient. This could include data from one or several EMS agencies in a given patient. Patients that had at least one SBP <90mmHg or oxygen saturation <90% within their entire set of prehospital measurements became, respectively, the group with “hypotension” or “hypoxia.” The “combined hypotension/hypoxia” cohort included all those who had at least one hypoxic measurement and at least one hypotensive measurement during the entire duration of their prehospital care.
Analysis
Continuous variables were summarized by median and interquartile range (IQR) within each of the two subgroups of patients who survived or died and also within each of the four groups defined by hypotension/hypoxia status (neither hypotension nor hypoxia, hypotension only, hypoxia only, and both hypotension and hypoxia). Categorical variables were summarized by frequency and proportion [with 95% confidence intervals (CI) when appropriate] with each of the subgroups described above. Association between mortality and hypotension/hypoxia status was examined by logistic regression, with or without adjustment, for important independent risk factors and potential confounders [age, sex, race, ethnicity, payment source, trauma type (blunt versus penetrating), head region injury score (International Classification of Diseases-Version 9 matched to Abbreviated Injury Scale), Injury Severity Score (ISS), inter-facility transfer, and treating trauma center]. Age, sex, race, ethnicity, head region injury score, ISS, and inter-facility transfer were included, a priori, in the model (regardless of whether they were found to be significant or not), while payment source, trauma type, and treating trauma center were included since they were found to be significant covariates. The effect of the age in the logistic regression was fitted non-parametrically using penalized thin plate regression splines through the generalized additive model,46 with the smoothing parameter chosen to optimize the Akaike information criterion (AIC). The software environment R was used for the analysis47 and the R package mgcv46,48 was used for the generalized additive model. The p values were calculated from a Wald-type test using the Bayesian covariance matrix.49 The fitted model was assessed by deviance residual plots as well as the area under the receiver operational characteristic (ROC) curve (AUC). The 95% Cis of the AUC were obtained by the Delong method.50 Collinearity was checked using variance inflation factors for the parametric terms and concurvity for the nonparametric term. Mixed effect models were used to assess the correlation of subjects treated by the same trauma center and multiple imputation procedures were used to evaluate the impact of missing covariates.
MAIN RESULTS
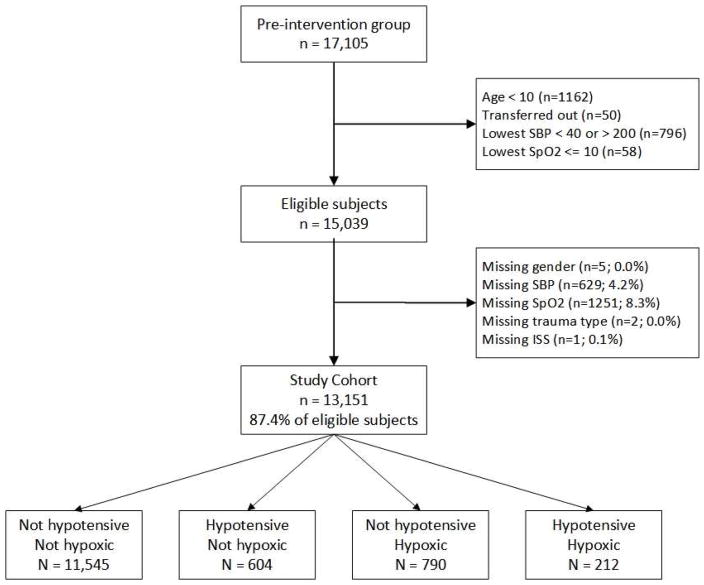
There were 17,105 subjects in the pre-intervention group (from 1/1/2007 through 3/31/2014) of which 13,151 cases (76.9%) met inclusion criteria (study cohort; Figure 1 shows the details of excluded cases). The median age was 45 years (IQR: 26, 64), 68.6% were male, and 8.2% died. Among those in the study group, 11,545 (87.8%) had neither hypotension nor hypoxia, 604 (4.6%) had hypotension only, 790 (6.0%) had hypoxia only, and 212 (1.6%) had both hypotension and hypoxia. Figure 2 shows the raw, unadjusted cohort mortality by the existence of neither hypotension nor hypoxia, hypotension only, hypoxia only, and both hypotension and hypoxia combined. The mortality rates range from a low of 5.6% for those with neither hypoxia nor hypotension to a high of 43.9% in patients with both. Table 1 summarizes the demographics and patient characteristics by survival status. Table 2 summarizes the same variables by hypotension and hypoxia status. All factors associated with risk of death were also associated with the hypotension/hypoxia status. The specific data by treating trauma center are not shown in Tables 1 or 2. Since absolute anonymity is required by state regulations and the IRB (for all subjects, EMS agencies, and hospitals), we are not able to report specific trauma center-related data, even generically. The reason for this is because trauma center patient volumes in Arizona are a matter of public record. Thus, presentation of these data could conceivably lead to certain hospital-specific information being inferred or identified (e.g., due to comparisons of the sizes of the 95% CIs). While the data are not shown, because treating trauma center was a significant confounder, we adjusted for it in the model.
Figure 1.
Details of Study Population Inclusion and Exclusion
SBP = systolic blood pressure; Sp02 = % oxygen saturation; trauma type = blunt or penetrating injury mechanism; ISS = Injury Severity Score
Figure 2.
Crude Mortality Rate by Hypotension/Hypoxia Status
Error bars represent 95% confidence intervals.
Table 1.
Patient and Injury Characteristics by Survival Status
| All# | Alive# | Dead# | ||
|---|---|---|---|---|
| Characteristics | 13151 | 12067 | 1084 | |
| Age (year) | 45 (26, 64) | 44 (25, 64) | 50 (28, 72) | |
| Male | No | 4135 (31.4%) | 3808 (31.6%) | 327 (30.2%) |
| Yes | 9016 (68.6%) | 8259 (68.4%) | 757 (69.8%) | |
| Race | Black | 386 (2.9%) | 358 (3%) | 28 (2.6%) |
| American Indian/Alaska Nat. | 1087 (8.3%) | 1007 (8.3%) | 80 (7.4%) | |
| Asian | 129 (1%) | 118 (1%) | 11 (1%) | |
| White | 9868 (75%) | 9047 (75%) | 821 (75.7%) | |
| Other | 1570 (11.9%) | 1444 (12%) | 126 (11.6%) | |
| Unknown | 111 (0.8%) | 93 (0.8%) | 18 (1.7%) | |
| Hispanic | No | 10083 (76.7%) | 9264 (76.8%) | 819 (75.6%) |
| Yes | 2743 (20.9%) | 2528 (20.9%) | 215 (19.8%) | |
| Unknown | 325 (2.5%) | 275 (2.3%) | 50 (4.6%) | |
| Payer | Private | 4292 (32.6%) | 4037 (33.5%) | 255 (23.5%) |
| AHCCCS/Medicaid | 3415 (26%) | 3165 (26.2%) | 250 (23.1%) | |
| Medicare | 2846 (21.6%) | 2544 (21.1%) | 302 (27.9%) | |
| Self Pay | 1698 (12.9%) | 1515 (12.6%) | 183 (16.9%) | |
| Other | 633 (4.8%) | 581 (4.8%) | 52 (4.8%) | |
| Unknown | 267 (2%) | 225 (1.9%) | 42 (3.9%) | |
| Trauma Type | Blunt | 12665 (96.3%) | 11782 (97.6%) | 883 (81.5%) |
| Penetrating | 486 (3.7%) | 285 (2.4%) | 201 (18.5%) | |
| Head Injury Severity Score (ICD) | 1–3 | 7182 (54.6%) | 7104 (58.9%) | 78 (7.2%) |
| 4 | 3874 (29.5%) | 3747 (31.1%) | 127 (11.7%) | |
| 5–6 | 1962 (14.9%) | 1099 (9.1%) | 863 (79.6%) | |
| Unknown | 133 (1%) | 117 (1%) | 16 (1.5%) | |
| Injury Severity Score (ICD) | 1–14 | 5372 (40.8%) | 5349 (44.3%) | 23 (2.1%) |
| 16–24 | 4381 (33.3%) | 4299 (35.6%) | 82 (7.6%) | |
| 25+ | 3398 (25.8%) | 2419 (20%) | 979 (90.3%) | |
| Hypotension | No | 12335 (93.8%) | 11469 (95%) | 866 (79.9%) |
| Yes | 816 (6.2%) | 598 (5%) | 218 (20.1%) | |
| Hypoxia | No | 12149 (92.4%) | 11380 (94.3%) | 769 (70.9%) |
| Yes | 1002 (7.6%) | 687 (5.7%) | 315 (29.1%) | |
| Hypotension and Hypoxia | No | 12939 (98.4%) | 11948 (99%) | 991 (91.4%) |
| Yes | 212 (1.6%) | 119 (1%) | 93 (8.6%) | |
| Inter-facility Transfer | No | 8890 (67.6%) | 8051 (66.7%) | 839 (77.4%) |
| Yes | 4176 (31.8%) | 3932 (32.6%) | 244 (22.5%) | |
| Unknown | 85 (0.6%) | 84 (0.7%) | 1 (0.1%) | |
median (interquartile range) for continuous variables and count (percentage) for categorical variables
Table 2.
Patient and Injury Characteristics by Hypotension/Hypoxia Status
| All# | No hypotension nor hypoxia# | Hypotension only# | Hypoxia only# | Both conditions# | ||
|---|---|---|---|---|---|---|
| Characteristics | 13151 | 11545 | 604 | 790 | 212 | |
| Death | No | 12067 (91.8%) | 10901 (94.4%) | 479 (79.3%) | 568 (71.9%) | 119 (56.1%) |
| Yes | 1084 (8.2%) | 644 (5.6%) | 125 (20.7%) | 222 (28.1%) | 93 (43.9%) | |
| Age (year) | 45 (26, 64) | 45 (26, 65) | 44 (25, 62) | 48 (28.2, 66) | 32.5 (21, 50) | |
| Male | No | 4135 (31.4%) | 3633 (31.5%) | 202 (33.4%) | 236 (29.9%) | 64 (30.2%) |
| Yes | 9016 (68.6%) | 7912 (68.5%) | 402 (66.6%) | 554 (70.1%) | 148 (69.8%) | |
| Race | Black | 386 (2.9%) | 341 (3%) | 11 (1.8%) | 31 (3.9%) | 3 (1.4%) |
| American Indian/Alaska Nat. | 1087 (8.3%) | 950 (8.2%) | 59 (9.8%) | 52 (6.6%) | 26 (12.3%) | |
| Asian | 129 (1%) | 114 (1%) | 6 (1%) | 7 (0.9%) | 2 (0.9%) | |
| White | 9868 (75%) | 8646 (74.9%) | 453 (75%) | 610 (77.2%) | 159 (75%) | |
| Other | 1570 (11.9%) | 1405 (12.2%) | 69 (11.4%) | 78 (9.9%) | 18 (8.5%) | |
| Unknown | 111 (0.8%) | 89 (0.8%) | 6 (1%) | 12 (1.5%) | 4 (1.9%) | |
| Hispanic | No | 10083 (76.7%) | 8837 (76.5%) | 456 (75.5%) | 625 (79.1%) | 165 (77.8%) |
| Yes | 2743 (20.9%) | 2430 (21%) | 124 (20.5%) | 145 (18.4%) | 44 (20.8%) | |
| Unknown | 325 (2.5%) | 278 (2.4%) | 24 (4%) | 20 (2.5%) | 3 (1.4%) | |
| Payer | Private | 4292 (32.6%) | 3782 (32.8%) | 190 (31.5%) | 243 (30.8%) | 77 (36.3%) |
| AHCCCS/Medicaid | 3415 (26%) | 2958 (25.6%) | 180 (29.8%) | 208 (26.3%) | 69 (32.5%) | |
| Medicare | 2846 (21.6%) | 2537 (22%) | 113 (18.7%) | 177 (22.4%) | 19 (9%) | |
| Self Pay | 1698 (12.9%) | 1487 (12.9%) | 82 (13.6%) | 101 (12.8%) | 28 (13.2%) | |
| Other | 633 (4.8%) | 552 (4.8%) | 22 (3.6%) | 44 (5.6%) | 15 (7.1%) | |
| Unknown | 267 (2%) | 229 (2%) | 17 (2.8%) | 17 (2.2%) | 4 (1.9%) | |
| Trauma Type | Blunt | 12665 (96.3%) | 11213 (97.1%) | 541 (89.6%) | 720 (91.1%) | 191 (90.1%) |
| Penetrating | 486 (3.7%) | 332 (2.9%) | 63 (10.4%) | 70 (8.9%) | 21 (9.9%) | |
| Head Injury Severity Score (ICD) | 1–3 | 7182 (54.6%) | 6573 (56.9%) | 284 (47%) | 274 (34.7%) | 51 (24.1%) |
| 4 | 3874 (29.5%) | 3476 (30.1%) | 139 (23%) | 208 (26.3%) | 51 (24.1%) | |
| 5–6 | 1962 (14.9%) | 1391 (12%) | 165 (27.3%) | 299 (37.8%) | 107 (50.5%) | |
| Unknown | 133 (1%) | 105 (0.9%) | 16 (2.6%) | 9 (1.1%) | 3 (1.4%) | |
| Injury Severity Score (ICD) | 1–14 | 5372 (40.8%) | 5090 (44.1%) | 137 (22.7%) | 132 (16.7%) | 13 (6.1%) |
| 16–24 | 4381 (33.3%) | 3986 (34.5%) | 168 (27.8%) | 198 (25.1%) | 29 (13.7%) | |
| 25+ | 3398 (25.8%) | 2469 (21.4%) | 299 (49.5%) | 460 (58.2%) | 170 (80.2%) | |
| Inter-facility Transfer | No | 8890 (67.6%) | 7662 (66.4%) | 410 (67.9%) | 641 (81.1%) | 177 (83.5%) |
| Yes | 4176 (31.8%) | 3808 (33%) | 191 (31.6%) | 144 (18.2%) | 33 (15.6%) | |
| Unknown | 85 (0.6%) | 75 (0.6%) | 3 (0.5%) | 5 (0.6%) | 2 (0.9%) | |
median (interquartile range) for continuous variables and count (percentage) for categorical variables
Logistic regression was used to examine the independent associations between hypotension/hypoxia status and mortality risk, controlling for potential confounders and significant risk measures (shown in Table 1). The results of the regression analysis are shown in Table 3. Figure 3 shows the crude (unadjusted) and adjusted odds ratios (cOR and aOR, respectively) for death for the sub-cohorts defined by hypotension/hypoxia status, utilizing the patients with neither hypotension nor hypoxia as the reference. Compared to this group, the cohort with both hypotension and hypoxia had a cOR for death of 13.2 (95% CI 10.0–17.5) and an aOR of 6.1 (95% CI 4.2–8.9). These represent at least a doubling of the corresponding odds ratios for either hypotension [cOR-4.4(3.6–5.5); aOR-2.5 (1.9–3.3)] or hypoxia [cOR-6.6 (5.6–7.9); aOR-3.0 (2.4–3.8)] alone (Figure 3). Testing for an interaction term between hypotension and hypoxia was not significant in the logistic regression model (p=0.427), indicating that the effects of hypotension and hypoxia were additive on the scale of log odds.
Table 3.
Logistic Regression Model for Death
| Covariates+ | OR | 95% CI | |
|---|---|---|---|
| Hypotension and Hypoxia Status | Neither hypotension nor Hypoxia | --- | --- |
| Hypotension only | 2.49 | (1.87, 3.32) | |
| Hypoxia only | 3.00 | (2.37, 3.78) | |
| Both conditions | 6.10 | (4.20, 8.86) | |
| Male | No | ||
| Yes | 0.98 | (0.82, 1.17) | |
| Race | Black | --- | --- |
| American Indian/Alaska Nat. | 1.82 | (1.00, 3.32) | |
| Asian | 1.31 | (0.51, 3.35) | |
| White | 1.72 | (1.02, 2.91) | |
| Other | 1.94 | (1.06, 3.56) | |
| Unknown | 2.23 | (0.89, 5.60) | |
| Hispanic | No | --- | --- |
| Yes | 0.73 | (0.56, 0.94) | |
| Unknown | 1.78 | (1.08, 2.93) | |
| Payer | Private | --- | --- |
| AHCCCS/Medicaid | 1.08 | (0.85, 1.37) | |
| Medicare | 1.29 | (0.97, 1.72) | |
| Self Pay | 2.49 | (1.89, 3.29) | |
| Other | 1.18 | (0.79, 1.76) | |
| Unknown | 2.75 | (1.62, 4.66) | |
| Trauma Type | Blunt | --- | --- |
| Penetrating | 4.73 | (3.55, 6.31) | |
| Head Injury Severity Score (ICD) | 1–3 | --- | --- |
| 4 | 1.34 | (0.96, 1.87) | |
| 5–6 | 12.35 | (9.05, 16.85) | |
| Unknown | 5.76 | (2.97, 11.16) | |
| Injury Severity Score (ICD) | 1–14 | --- | --- |
| 16–24 | 3.08 | (1.81, 5.25) | |
| 25+ | 12.93 | (7.82, 21.38) | |
| Inter-facility Transfer | No | --- | --- |
| Yes | 0.62 | (0.50, 0.77) | |
| Unknown | 0.25 | (0.03, 2.02) | |
Age was fitted non-parametrically and trauma center was also included (details not shown)
Figure 3.
Odds Ratios for Mortality by Hypotension/Hypoxia Status
Reference group was the cohort with neither hypotension nor hypoxia. Error bars represent 95% confidence intervals.
Deviance residual plots didn’t indicate any deviation from the model assumptions. The only continuous covariate in the model, age, was fitted nonparametrically. The AUC was estimated to be 0.938 (95% CI: 0.932–0.945), indicating a high discriminative ability of the model. In addition, no multicollinearity in the covariates was detected.
As a sensitivity analysis, random trauma center (TC) effects were added to the logistic regression model to explore the potential correlation among subjects treated by the same TC. There was minimal difference in the results: the largest change in the estimated ORs was 1.5% for the three groups of hypotension only, hypoxia only, and both conditions compared to the referent group of no hypotension nor hypoxia. Also the largest change in the standard error (SE) estimates for the three corresponding regression coefficients was 0.2%. As another sensitivity analysis, we applied the multiple imputation procedure to explore the effects of missing data and observed only small changes. The largest change in the estimated ORs was 10.5% and the lower limit of the 95% CI for each OR still stayed above 1.
LIMITATIONS
This study has limitations. First, the design is observational and we are unable to establish cause-and-effect relationships related to treatment. Thus, the results cannot be used to determine whether the treatment of hypotension or hypoxia are effective at reducing mortality (this is part of the primary hypothesis of the main, parent study). The current analysis simply allows us to identify associations between hypotension, hypoxia, and outcome. Second, there are some missing data. However, for a prehospital study, the rates for missing data are very low51 (Figure 1). In addition, the use of multiple imputation resulted in minimal differences in the analysis compared to that of the actual dataset. Third, the database contains only those measurements of blood pressure and oxygen saturation that were documented by EMS personnel and there is no way to independently verify the accuracy of the BP and oxygen saturation measurements. Thus, we cannot know for sure that all hypotensive or hypoxic patients were identified and, hence, there could be some misclassification of patients among the four groups (hypotension, hypoxia, neither, both). However, these issues related to data documentation and accuracy are true of essentially all EMS studies. One strength of EPIC is that the data are abstracted directly, consistently, and comprehensively from the PCRs. This level of scrutiny and consistency of data collection is rare in prehospital research.51 Fourth, there could have been some "leakage" in practice changes during the pre-implementation timeframe since the guidelines have been available for more than a decade. However, we believe it is very unlikely that this is a factor. We did a pre-study evaluation of TBI protocol changes and implementation prior to the EPIC Project implementation to identify whether partial or full implementation was occurring in Arizona. Information from 51 agencies (responsible for EMS response to 4.8 million residents/75% of the population) was gathered related to TBI EMS care. Only half had protocols specifying appropriate ranges for O2 saturation or blood pressure and only one-third had any specific treatment protocols. Even among agencies with TBI protocols, the monitoring and treatment recommendations were highly variable and no agency had implemented, or was planning to implement, the official TBI guidelines. Fifth, the definition for hypotension and hypoxia required only that there be at least a single low reading (<90mmHg/<90% saturation). Thus, the absence of time-sequence analysis means that we treated patients who may have had multiple low readings the same as cases that had only a single abnormal measurement. Finally, we did not evaluate whether interventions were performed in an attempt to prevent or treat blood pressure or oxygenation.
DISCUSSION
The detrimental effects of hypotension and hypoxia during the early care of patients with major TBI have been well established.6,7,10–40 However, there is almost nothing known about the impact of these factors when they both occur in patients prior to arrival at the hospital. This is because 1) the hypotension/hypoxia combination is an unusual occurrence and 2) studying this question requires the analysis of large numbers of TBI patients with linked prehospital data. While there are large trauma center-based databases that can be queried for ED and in-hospital information, these have limited or no prehospital data.22,38,39,52–57 Since the EPIC database has extensive prehospital data and is very large, it provides the opportunity to ask EMS-related questions in small patient subgroups.41
We have been able to find only two previous studies that reported specifically on the combined effect of prehospital hypotension and hypoxia on outcome.6,16 In Stocchetti’s investigation of 49 patients, 27 had an O2 saturation <90% on the scene and 12 had an SBP <100 mm Hg (their definition for hypotension).6 Unfortunately, the study does not report the number who had both hypotension and hypoxia. However, at least some of the patients must have had both because the authors concluded that “outcome was significantly worse in cases of hypotension, desaturation, or both.” They gave no information about the relative rates of mortality among the cohorts. Chi and associates studied 150 severe TBI patients who were transported by helicopter.16 Fourteen had only hypotension, 37 had only hypoxia (oxygen saturation <92%), and 6 had both. Mortality for cases with neither hypotension nor hypoxia was 20%. This compared to 8% for hypotension-only patients, 37% for hypoxia-only patients, and 24% for patients with both. These wide-ranging (and even paradoxical) results were likely due to the very small numbers and, thus, this study could make no conclusions about the impact of the combination of hypotension and hypoxia on outcome. Both Fearnside, et al11 and Stassen, et al40 also obtained prehospital clinical data in their evaluations of severe TBI. However, they made no comment at all about the relative impact of the combination of hypotension and hypoxia. In Chesnut’s classic study on secondary brain injury, they attempted to assess the impact of physiologic insults in the EMS setting.10 Unfortunately, the prehospital data were compromised by the fact that they did not actually obtain measurements of oxygenation. Rather, prehospital “hypoxia” was merely identified as the presence of cyanosis or apnea when this was documented by EMS personnel.10
The studies that report in-hospital data from the ED or the ICU give slightly more information about the combination of hypotension and hypoxia, but the findings have been quite variable and inconclusive. Manley studied 107 patients with TBI utilizing physiologic measurements in the ED and inpatient settings.26 Among the 14 patients who had both hypotension and hypoxia, they found that, “…the combination of hypotension and hypoxia were not additive.” Unfortunately, with such small numbers, the statistical power behind such a conclusion was very limited. Pigula evaluated 451 children with severe TBI in the National Pediatric Trauma Registry utilizing in-hospital physiologic parameters.22 Mortality was 61% among children with hypotension only, 21% with hypoxia only, and 85% among the small number (20) that had both hypotension and hypoxia. They concluded, “If both hypotension and hypoxia were found together, mortality was only slightly increased over those children with hypotension alone (p = 0.056).” Kohi found that the combination of hypotension and hypoxia in severe TBI patients was universally fatal.35 However, there were only 6 patients in this cohort and all of the measurements of blood pressure and oxygenation were obtained in the ICU. Thus, this study was reflective of patients with “late” hypotension and hypoxia, but provided no information about physiologic insults occurring earlier in the course and, perhaps, before irreversible injury had occurred. In a meta-analysis, McHugh reported on 465 combined hypotension/hypoxia patients and found a slight increase in mortality among patients who had both (54.6%) compared to those with hypotension only (48.5%).39 However, they utilized a mixture of ED admission data and an unspecified amount of EMS data. There was also significant heterogeneity among the investigations that were included in the final meta-analysis (e.g., differing definitions of hypotension). Furthermore, some of the studies had missing data rates exceeding 30%, creating substantial risks for selection bias.
In the current study of 13,151 major TBI patients, 604 (4.6%) experienced hypotension without hypoxia in the field, 790 (6.0%) had hypoxia without hypotension, and 212 (1.6%) suffered both. We believe this is the largest evaluation of prehospital hypotension and hypoxia yet conducted in TBI patients and this allowed us to examine detailed interactions that the previous studies could not (the largest report in the extant EMS literature had no more than 12 combined hypotension/hypoxia patients6,16). In the EPIC population, the combination of hypotension and hypoxia is associated with a dramatically-increased likelihood of dying (cOR-13.2, aOR-6.1) compared to the cohorts that have only hypotension (cOR-4.4, aOR-2.5) or hypoxia (cOR-6.6, aOR-3.0; Figure 3]. This means that the combination is associated with more than a doubling of the risk of death compared to having either alone. The clinical implications of this are further supported by the fact that there is no interaction on the log odds scale. In other words, hypoxia does not modify the effect of hypotension and, conversely, hypotension does not modify the effect of hypoxia. Thus, in patients who suffer both hypotension and hypoxia, the combination of these physiological insults has a profound impact on outcome with an additive influence on the log odds of death.
As stated in the study hypothesis, the primary focus of this evaluation was to identify whether the hypotension/hypoxia combination adds additional risk above that of either alone. However, this analysis also revealed another important finding—the associations between the secondary physiological insults and mortality are significantly stronger than have been generally reported. While there is variation, both the crude and adjusted odds of death for patients experiencing hypoxia alone have typically been around 2.7,10–13,17,20 However, in the EPIC population, the crude OR is 6.6 and the adjusted OR is 3.0 (Figure 3). Furthermore, the odds of mortality in patients with hypotension only have generally been in the range of 1.3 to 2.10,11,14,15,18–38 In contrast, we identified significantly higher odds of death in hypotensive patients (cOR-4.4, aOR-2.5; Figure 3). There are several potential reasons for this. First, perhaps the previous studies were simply too small to identify an accurate influence of these factors. Second, many of the studies that were dependent upon obtaining data from trauma center databases only had access to one or two prehospital vital signs measurements. Thus, it is unclear whether hypotension or hypoxia were reliably identified. This is because, in previous studies, it was unclear whether the EMS measurements recorded in the database were the first, last, highest, or lowest for each patient. By comparison, in the EPIC database, there is no limit to the number of vital signs measurements that can be recorded. For example, there are cases in the EPIC database that have more than 30 recorded prehospital blood pressures. Finally, most of the previous studies utilized blood pressure and oxygen saturation data obtained after arrival at the hospital. Thus, it is possible that the EPIC study, by specifically evaluating the prehospital treatment interval, has identified patients who become hypotensive and/or hypoxic earlier in their course. In this case, the effects of these insults may be magnified by occurring earlier, and perhaps lasting longer, and thus may impact the brain to a greater extent.
The design of the current study does not allow confident statements about the impact of EMS treatment aimed at preventing or reversing hypotension or hypoxia. However, it does bring up some interesting questions. Since the combination appears to be so detrimental, this raises the specter that if either hypoxia or hypotension can be prevented or treated, there may be the potential to significantly improve survival even if the other parameter is not improved. For example, the prevention of hypoxia by management of oxygenation may decrease a given patient’s risk of death from a highly fatal aOR of 6.1 (if they had suffered both) to a far more “favorable” aOR of 2.5 (having only suffered hypotension). The same might be relevant in the prevention and/or treatment of hypotension in a patient who has hypoxia that cannot be improved.
In summary, this statewide study evaluating prehospital hypotension and hypoxia in victims of major TBI found a greater risk for death from either of these insults than has generally been reported in the previous literature. Furthermore, the combination of both hypotension and hypoxia occurring prior to arrival at the hospital is associated with a dramatic increase in both the crude and adjusted odds of death when compared to either physiological insult alone. In fact, the effects are additive in their effect on the log odds of death. These findings seem supportive of the emphasis on aggressive prevention and treatment of hypotension and hypoxia reflected in the current EMS TBI treatment guidelines but clearly reveal the need for further study to determine their impact on outcome.42–45
Acknowledgments
Funding: Research reported in this publication was supported by the National Institute of Neurological Disorders And Stroke of the National Institutes of Health under Award Number R01NS071049. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Footnotes
Presented, in part, to the Society for Academic Medicine; May 16, 2013, Atlanta, Georgia and to the Resuscitation Science Symposium of the American Heart Association; Chicago, Illinois, November, 15, 2014.
Registration: This is an observational, non-interventional analysis of a subset of the data in the EPIC study. The parent study, while not a randomized clinical trial, is registered at ClinicalTrials.gov: #NCT01339702
Disclosures: The University of Arizona receives funding from the NIH supporting the EPIC Study. This includes support for the following authors: DWS, BJB, VC, BB, JBG, KRD, PDA, CV, and DS. No COI to report: CH, SMK, and TM.
Author Contributions: Study concept and design: DWS, BJB, JBG, KRD, PDA, CV, DS; Acquisition of the data: DWS, CH, BJB, VC, BB; Analysis and interpretation of the data: DWS, CH, BJB, VC, DS; Drafting of the manuscript: DWS, CH, BJB; Critical revision of the manuscript for important intellectual content: DWS, CH, BJB, VC, BB, JBG, KRD, SMK, PDA, CV, TM, DS; Statistical expertise: CH, DS; Obtained funding: DWS, BJB, JBG, KRD, CV, DS; Administrative, technical, or material support: VC, BB, TM.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Finkelstein E, Corso PS, Miller TR. The incidence and economic burden of injuries in the United States. Oxford ; New York: Oxford University Press; 2006. [Google Scholar]
- 2.Bell JMBM, Jenkins EL, Haarbauer-Krupa J. Traumatic Brain Injury In the United States: Epidemiology and Rehabilitation. National Center for Injury Prevention and Control, Division of Unintentional Injury Prevention, Centers for Disease Control; ; 2014. [Google Scholar]
- 3.Thurman DJ, Alverson C, Dunn KA, Guerrero J, Sniezek JE. Traumatic brain injury in the United States: A public health perspective. J Head Trauma Rehabil. 1999;14(6):602–615. doi: 10.1097/00001199-199912000-00009. [DOI] [PubMed] [Google Scholar]
- 4.Davis DP, Fakhry SM, Wang HE, et al. Paramedic rapid sequence intubation for severe traumatic brain injury: perspectives from an expert panel. Prehosp Emerg Care. 2007;11(1):1–8. doi: 10.1080/10903120601021093. [DOI] [PubMed] [Google Scholar]
- 5.Davis DP, Idris AH, Sise MJ, et al. Early ventilation and outcome in patients with moderate to severe traumatic brain injury. Crit Care Med. 2006;34(4):1202–1208. doi: 10.1097/01.CCM.0000208359.74623.1C. [DOI] [PubMed] [Google Scholar]
- 6.Stocchetti N, Furlan A, Volta F. Hypoxemia and arterial hypotension at the accident scene in head injury. J Trauma. 1996;40(5):764–767. doi: 10.1097/00005373-199605000-00014. [DOI] [PubMed] [Google Scholar]
- 7.Cooke RS, McNicholl BP, Byrnes DP. Early Management of Severe Head-Injury in Northern-Ireland. Injury-International Journal of the Care of the Injured. 1995;26(6):395–397. doi: 10.1016/0020-1383(95)00003-r. [DOI] [PubMed] [Google Scholar]
- 8.Davis DP, Dunford JV, Poste JC, et al. The impact of hypoxia and hyperventilation on outcome after paramedic rapid sequence intubation of severely head-injured patients. J Trauma. 2004;57(1):1–8. doi: 10.1097/01.ta.0000135503.71684.c8. discussion 8–10. [DOI] [PubMed] [Google Scholar]
- 9.Dunford JV, Davis DP, Ochs M, Doney M, Hoyt DB. Incidence of transient hypoxia and pulse rate reactivity during paramedic rapid sequence intubation. Ann Emerg Med. 2003;42(6):721–728. doi: 10.1016/s0196-0644(03)00660-7. [DOI] [PubMed] [Google Scholar]
- 10.Chesnut RM, Marshall LF, Klauber MR, et al. The role of secondary brain injury in determining outcome from severe head injury. J Trauma. 1993;34(2):216–222. doi: 10.1097/00005373-199302000-00006. [DOI] [PubMed] [Google Scholar]
- 11.Fearnside MR, Cook RJ, McDougall P, McNeil RJ. The Westmead Head Injury Project outcome in severe head injury. A comparative analysis of pre-hospital, clinical and CT variables. Br J Neurosurg. 1993;7(3):267–279. doi: 10.3109/02688699309023809. [DOI] [PubMed] [Google Scholar]
- 12.Marmarou A, Anderson RL, Ward JD, et al. Impact of Icp Instability and Hypotension on Outcome in Patients with Severe Head Trauma. J Neurosurg. 1991;75:S59–S66. [Google Scholar]
- 13.Silverston P. Pulse oximetry at the roadside: a study of pulse oximetry in immediate care. Bmj. 1989;298(6675):711–713. doi: 10.1136/bmj.298.6675.711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mayer TA, Walker ML. Pediatric head injury: the critical role of the emergency physician. Ann Emerg Med. 1985;14(12):1178–1184. doi: 10.1016/s0196-0644(85)81025-8. [DOI] [PubMed] [Google Scholar]
- 15.Ong L, Selladurai BM, Dhillon MK, Atan M, Lye MS. The prognostic value of the Glasgow Coma Scale, hypoxia and computerised tomography in outcome prediction of pediatric head injury. Pediatr Neurosurg. 1996;24(6):285–291. doi: 10.1159/000121057. [DOI] [PubMed] [Google Scholar]
- 16.Chi JH, Knudson MM, Vassar MJ, et al. Prehospital hypoxia affects outcome in patients with traumatic brain injury: a prospective multicenter study. J Trauma. 2006;61(5):1134–1141. doi: 10.1097/01.ta.0000196644.64653.d8. [DOI] [PubMed] [Google Scholar]
- 17.Jones PA, Andrews PJ, Midgley S, et al. Measuring the burden of secondary insults in head-injured patients during intensive care. J Neurosurg Anesthesiol. 1994;6(1):4–14. [PubMed] [Google Scholar]
- 18.Shutter LA, Narayan RK. Blood Pressure Management in Traumatic Brain Injury. Annals of Emergency Medicine. 2008;51(3, Supplement 1):S37–S38. doi: 10.1016/j.annemergmed.2007.11.013. [DOI] [PubMed] [Google Scholar]
- 19.Jankowitz BT, Adelson PD. Pediatric traumatic brain injury: past, present and future. Dev Neurosci. 2006;28(4–5):264–275. doi: 10.1159/000094153. [DOI] [PubMed] [Google Scholar]
- 20.Gentleman D. Causes and effects of systemic complications among severely head injured patients transferred to a neurosurgical unit. Int Surg. 1992;77(4):297–302. [PubMed] [Google Scholar]
- 21.Haddad S, Arabi Y, Al Shimemeri A. Initial management of traumatic brain injury. Middle East J Anesthesiol. 2005;18(1):45–68. [PubMed] [Google Scholar]
- 22.Pigula FA, Wald SL, Shackford SR, Vane DW. The effect of hypotension and hypoxia on children with severe head injuries. J Pediatr Surg. 1993;28(3):310–314. doi: 10.1016/0022-3468(93)90223-8. discussion 315–316. [DOI] [PubMed] [Google Scholar]
- 23.Kokoska ER, Smith GS, Pittman T, Weber TR. Early hypotension worsens neurological outcome in pediatric patients with moderately severe head trauma. J Pediatr Surg. 1998;33(2):333–338. doi: 10.1016/s0022-3468(98)90457-2. [DOI] [PubMed] [Google Scholar]
- 24.Miller JD, Becker DP. Secondary insults to the injured brain. J R Coll Surg Edinb. 1982;27(5):292–298. [PubMed] [Google Scholar]
- 25.Barton CW, Hemphill JC, Morabito D, Manley G. A novel method of evaluating the impact of secondary brain insults on functional outcomes in traumatic brain-injured patients. Acad Emerg Med. 2005;12(1):1–6. doi: 10.1197/j.aem.2004.08.043. [DOI] [PubMed] [Google Scholar]
- 26.Manley G, Knudson MM, Morabito D, Damron S, Erickson V, Pitts L. Hypotension, hypoxia, and head injury: frequency, duration, and consequences. Arch Surg. 2001;136(10):1118–1123. doi: 10.1001/archsurg.136.10.1118. [DOI] [PubMed] [Google Scholar]
- 27.Johnson DL, Boal D, Baule R. Role of apnea in nonaccidental head injury. Pediatr Neurosurg. 1995;23(6):305–310. doi: 10.1159/000120976. [DOI] [PubMed] [Google Scholar]
- 28.Price DJ, Murray A. The influence of hypoxia and hypotension on recovery from head injury. Injury. 1972;3(4):218–224. doi: 10.1016/0020-1383(72)90104-0. [DOI] [PubMed] [Google Scholar]
- 29.Michaud LJ, Rivara FP, Grady MS, Reay DT. Predictors of survival and severity of disability after severe brain injury in children. Neurosurgery. 1992;31(2):254–264. doi: 10.1227/00006123-199208000-00010. [DOI] [PubMed] [Google Scholar]
- 30.Levin HS, Aldrich EF, Saydjari C, et al. Severe head injury in children: experience of the Traumatic Coma Data Bank. Neurosurgery. 1992;31(3):435–443. doi: 10.1227/00006123-199209000-00008. discussion 443–434. [DOI] [PubMed] [Google Scholar]
- 31.Luerssen TG, Klauber MR, Marshall LF. Outcome from head injury related to patient's age. A longitudinal prospective study of adult and pediatric head injury. J Neurosurg. 1988;68(3):409–416. doi: 10.3171/jns.1988.68.3.0409. [DOI] [PubMed] [Google Scholar]
- 32.Miller JD, Sweet RC, Narayan R, Becker DP. Early insults to the injured brain. Jama. 1978;240(5):439–442. [PubMed] [Google Scholar]
- 33.Carrel M, Moeschler O, Ravussin P, Favre JB, Boulard G. Prehospital air ambulance and systemic secondary cerebral damage in severe craniocerebral injuries. Ann Fr Anesth Reanim. 1994;13(3):326–335. doi: 10.1016/s0750-7658(94)80041-3. [DOI] [PubMed] [Google Scholar]
- 34.Jeffreys RV, Jones JJ. Avoidable factors contributing to the death of head injury patients in general hospitals in Mersey Region. Lancet. 1981;2(8244):459–461. doi: 10.1016/s0140-6736(81)90786-8. [DOI] [PubMed] [Google Scholar]
- 35.Kohi YM, Mendelow AD, Teasdale GM, Allardice GM. Extracranial insults and outcome in patients with acute head injury--relationship to the Glasgow Coma Scale. Injury. 1984;16(1):25–29. doi: 10.1016/0020-1383(84)90110-4. [DOI] [PubMed] [Google Scholar]
- 36.Rose J, Valtonen S, Jennett B. Avoidable factors contributing to death after head injury. Br Med J. 1977;2(6087):615–618. doi: 10.1136/bmj.2.6087.615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Seelig JM, Klauber MR, Toole BM, Marshall LF, Bowers SA. Increased ICP and systemic hypotension during the first 72 hours following severe head injury. In: Miller JD, Teasdale GM, Rowan JO, editors. Intracranial Pressure VI. Berlin: Springer–Verlag; 1986. pp. 675–679. [Google Scholar]
- 38.Chesnut RM, Ghajar J, Maas AIR, et al. Part 2: Early indicators of prognosis in severe traumatic brain injury. J Neurotrauma. 2000;17(6–7):555. [Google Scholar]
- 39.McHugh GS, Engel DC, Butcher I, et al. Prognostic value of secondary insults in traumatic brain injury: results from the IMPACT study. J Neurotrauma. 2007;24(2):287–293. doi: 10.1089/neu.2006.0031. [DOI] [PubMed] [Google Scholar]
- 40.Stassen W, Welzel T. The prevalence of hypotension and hypoxaemia in blunt traumatic brain injury in the prehospital setting of Johannesburg, South Africa: A retrospective chart review. S Afr Med J. 2014;104(6):424–427. doi: 10.7196/samj.7494. [DOI] [PubMed] [Google Scholar]
- 41.Spaite DW, Bobrow BJ, Stolz U, et al. Evaluation of the impact of implementing the emergency medical services traumatic brain injury guidelines in Arizona: the Excellence in Prehospital Injury Care (EPIC) study methodology. Acad Emerg Med. 2014;21(7):818–830. doi: 10.1111/acem.12411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Guidelines for the management of severe traumatic brain injury. J Neurotrauma. 2007;24(Suppl 1):S1–106. doi: 10.1089/neu.2007.9999. [DOI] [PubMed] [Google Scholar]
- 43.Badjatia N, Carney N, Crocco TJ, et al. Guidelines for prehospital management of traumatic brain injury 2nd edition. Prehosp Emerg Care. 2008;12(Suppl 1):S1–52. doi: 10.1080/10903120701732052. [DOI] [PubMed] [Google Scholar]
- 44.Adelson PD, Bratton SL, Carney NA, et al. Guidelines for the acute medical management of severe traumatic brain injury in infants, children, and adolescents. Pediatr Crit Care Med. 2003;3(S3):S2–S81. doi: 10.1097/01.CCM.0000066600.71233.01. [DOI] [PubMed] [Google Scholar]
- 45.Kochanek PM, Carney N, Adelson PD, et al. Guidelines for the acute medical management of severe traumatic brain injury in infants, children, and adolescents. Pediatr Crit Care Med. (2) 2012 Jan;13(Suppl 1):S1–S82. doi: 10.1097/PCC.0b013e31823f435c. [DOI] [PubMed] [Google Scholar]
- 46.Wood SN. Generalized additive models : an introduction with R. Boca Raton, FL: Chapman & Hall/CRC; 2006. [Google Scholar]
- 47.R Core Team. [Accessed October 5, 2015];R: A language and environment for statistical computing. 2015 http://www.R-project.org/
- 48.Wood SN. Fast stable restricted maximum likelihood and marginal likelihood estimation of semiparametric generalized linear models. J R Stat Soc B. 2011;73:3–36. [Google Scholar]
- 49.Wood SN. On p-values for smooth components of an extended generalized additive model. Biometrika. 2013;100(1):221–228. [Google Scholar]
- 50.DeLong ER, DeLong DM, Clarke-Pearson DL. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics. 1988;44(3):837–845. [PubMed] [Google Scholar]
- 51.Spaite DW, Valenzuela TD, Meislin HW. Barriers to EMS System Evaluation: Problems Associated with Field Data Collection. Prehospital Disaster Med. 1993;8(S1):S35–S40. doi: 10.1017/s1049023x00040541. [DOI] [PubMed] [Google Scholar]
- 52.Chesnut RM, Marshall SB, Piek J, Blunt BA, Klauber MR, Marshall LF. Early and late systemic hypotension as a frequent and fundamental source of cerebral ischemia following severe brain injury in the Traumatic Coma Data Bank. Acta Neurochir Suppl (Wien) 1993;59:121–125. doi: 10.1007/978-3-7091-9302-0_21. [DOI] [PubMed] [Google Scholar]
- 53.MacKenzie EJ, Weir S, Rivara FP, et al. The value of trauma center care. J Trauma. 2010;69(1):1–10. doi: 10.1097/TA.0b013e3181e03a21. [DOI] [PubMed] [Google Scholar]
- 54.Ringdal KG, Lossius HM MTOS SahgoS, Trauma R. Feasibility of comparing core data from existing trauma registries in scandinavia. Reaching for a Scandinavian major trauma outcome study (MTOS) Scandinavian journal of surgery : SJS : official organ for the Finnish Surgical Society and the Scandinavian Surgical Society. 2007;96(4):325–331. doi: 10.1177/145749690709600412. [DOI] [PubMed] [Google Scholar]
- 55.Champion HR, Copes WS, Sacco WJ, et al. The Major Trauma Outcome Study: establishing national norms for trauma care. J Trauma. 1990;30(11):1356–1365. [PubMed] [Google Scholar]
- 56.Wardrope J. UK major trauma outcome study. BMJ. 1992;305(6860):1016. doi: 10.1136/bmj.305.6860.1016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Glance LG, Osler T. Beyond the major trauma outcome study: benchmarking performance using a national contemporary, population–based trauma registry. J Trauma. 2001;51(4):725–727. doi: 10.1097/00005373-200110000-00017. [DOI] [PubMed] [Google Scholar]