Abstract
Sequestration of Plasmodium falciparum parasites within the placenta often leads to an accumulation of macrophages within the intervillous space and increased production of tumor necrosis factor alpha (TNF-α), a cytokine associated with placental pathology and poor pregnancy outcomes. P. falciparum glycosylphosphatidylinositol (GPI) anchors have been shown to be the major parasite component that induces TNF-α production by monocytes and macrophages. Antibodies against P. falciparum GPI (anti-PfGPI), however, can inhibit the induction of TNF-α and inflammation. Thus, the study was undertaken to determine whether anti-PfGPI antibodies down-regulate inflammatory-type changes in the placentas of women with malaria. Anti-PfGPI immunoglobulin M (IgM) and IgG levels were measured in 380 pregnant women with or without placental malaria, including those who delivered prematurely and at term. Results showed that anti-PfGPI antibody levels increased with gravidity and age and that malaria infection boosted anti-PfGPI antibodies in pregnant women. However, no association was found between anti-PfGPI antibodies and placental TNF-α levels or the presence of acute or chronic placental malaria. Furthermore, anti-PfGPI antibody levels were similar in women with preterm and full-term deliveries and were not associated with an increase in infant birth weight. Thus, these results fail to support a strong role for anti-PfGPI antibodies in the prevention of chronic placental malaria infections and malaria-associated poor birth outcomes.
Plasmodium falciparum infection during pregnancy can have detrimental consequences for both the mother and the fetus. Pathology associated with malaria during pregnancy is due in part to the ability of the parasite to render infected erythrocytes adhesive to host receptors expressed within the placenta, resulting in sequestration of parasitized erythrocytes in the intervillous space (IVS) (2). One of the consequences of parasite sequestration in the placenta is the increased migration and recruitment of monocytes and macrophages to the IVS (21, 37). Monocytes and macrophages isolated in the IVS of malaria-infected women secreted significantly higher levels of tumor necrosis factor alpha (TNF-α) and several β-chemokines in vitro than similar cells isolated from the IVS of uninfected women (37). Such findings emphasize the role of maternal blood cells, particularly macrophages, as important sources of inflammatory mediators during placental malaria infection (37).
Although inflammatory cytokines are beneficial in controlling and eliminating parasites (29, 42), excessive concentrations of these cytokines, particularly TNF-α, play a significant role in exacerbating clinical symptoms (22, 27) as well as influencing placental pathogenesis and pregnancy outcome (13, 31). Experimental studies have shown that Plasmodium vinckei infection in pregnant mice increases serum TNF-α levels that are associated with fetal death and abortions (9). Recently, we reported that malaria-infected women who had preterm deliveries (PTD) possessed significantly higher placental TNF-α concentrations than malaria-infected women who delivered at term and uninfected women with PTD (38). Thus, identifying parasite factors that lead to excessive production of TNF-α in pregnant women is important.
Malaria toxins released during schizogony have been shown to stimulate TNF-α and nitric oxide production by macrophages (30, 41) and are considered to be important for the development of malaria-related pathology. Purification and chemical analysis of components in an extract of asexual-stage parasites has identified the parasites' glycosylphosphatidylinositol (GPI) anchors as the main TNF-α-inducing malaria toxin (33). Parasite GPIs can initiate signal transduction cascades in various host cells, notably macrophages (32, 39, 40), leading to the secretion of TNF-α and nitric oxide (40, 43). Moreover, P. falciparum GPIs elicit strong host responses by up-regulating the expression of adhesion molecules on endothelial cells, thereby increasing the cytoadherence of trafficking leukocytes and malaria-infected erythrocytes (35).
Interestingly, the administration of antibodies specific to P. falciparum GPIs (anti-PfGPI antibodies) in mice prevents the induction of both interleukin-1β and TNF-α (34), suggesting that anti-PfGPI antibodies may ameliorate some of the clinical symptoms of malaria. There is mounting evidence that elevated levels of anti-PfGPI antibodies can confer protection against clinical episodes of malaria in humans (24). Experiments with rodents also demonstrate that a GPI-based antitoxic vaccine protects against several malaria complications such as acidosis, pulmonary edema, and even death (36), causing optimism that a similar GPI-based vaccine can also be developed for humans. Since high numbers of parasites sequester in the placenta, it is likely that large amounts of GPI are released during schizogony that could activate macrophages to secrete TNF-α. However, if anti-PfGPI immunoglobulin G (IgG) antibodies are present, they might aid in controlling the level of inflammation in the IVS of the placenta. Thus, the goal of the present study was to determine the prevalence of anti-PfGPI IgM and IgG antibodies in pregnant women and whether they correlated with placental TNF-α concentrations and the risk of low-birth-weight (LBW) babies or PTD.
MATERIALS AND METHODS
Subjects and study design.
Women residing in the capital city of Yaoundé who delivered at the Biyem-Assi Hospital between August 1996 and August 1997 and at Central Hospital from September 1997 to August 2001 were consecutively recruited as part of a comprehensive immunological study on placental malaria and its effects on pregnancy outcome. The purpose of the study was explained to each subject by a member of the hospital staff, and only after obtaining informed verbal consent was a woman enrolled. A questionnaire was used to record maternal age, gravidity, and use of antimalarial drugs during pregnancy. After delivery, information about the newborn was recorded. Infants born between the 28th and 37th weeks of gestation were considered premature. In addition, 42 nonpregnant, malaria-negative women living in Yaoundé were enrolled for comparison. The study was approved by the Institutional Review Board, Georgetown University, and the National Ethical Committee, Cameroon.
Among the entire group of pregnant women recruited, 66 women had PTD. To compare PTD with full-term deliveries (FTD), a representative group of 314 women with FTD was selected for comparison by frequency matching with respect to placental malaria infection. A description of the 380 women in this study is provided in Table 1. The majority of these women were included in previous studies that evaluated the effect of malaria on changes of leukocyte distribution, chemokine and cytokine secretion in the placenta (37), and cytokine changes in the placentas of women with PTD and FTD (38). Since these data were reported previously, they are not repeated herein.
TABLE 1.
Characteristics of women in the study
| Parameter | Nonpregnant malaria-negative women | Pregnant women
|
P value | |
|---|---|---|---|---|
| Placental malaria negative | Placental malaria positive | |||
| No. of women | 42 | 203 | 177 | |
| Maternal age (yr, mean ± SD) | 26.4 ± 7.0 | 25.0 ± 5.4 | 26.3 ± 6.0 | 0.069 |
| Reported chemoprophylaxis usage (% of women) | 89.2 | 86.4 | 0.727 | |
| Primigravidae (%) | 24.1 | 34.5 | 0.027 | |
| Placental parasitemia (median) | 0 | 1.08 | ||
| Interquartile range (%) | 0.3-4.4 | |||
| Peripheral parasitemia (median) | 0 | 0.05 | ||
| Interquartile range (%) | 0-0.4 | |||
| % PTD | 15.3 | 19.0 | 0.352 | |
| Infant birth weight (g, mean ± SD) | 3,201 ± 658 | 2,993 ± 639 | <0.0001 | |
Collection and analysis of placental samples.
Immediately following delivery, a sample of heparinized maternal peripheral blood as well as a sample of placental blood was collected (37). A biopsy sample of placental tissue (2 cm by 2 cm by 2 cm) was collected; a portion was used to prepare impression smears for parasitological analysis, and another biopsy sample was fixed in 10% neutral buffered formalin for histology. Peripheral and placental blood smears and impression smears of placental tissues were prepared, stained, and examined for the presence of malaria parasites. Percentages of parasitemia were determined based on peripheral blood and impression smears. Based on the presence or absence of parasites in placental impression and blood smears and/or histological sections, women were considered to be either positive or negative for placental malaria at the time of delivery. Plasma was collected from maternal peripheral and placental blood samples and cryopreserved until used.
Determining TNF-α concentrations in placental plasma.
TNF-α levels in placental plasma were determined by using an OptEIA enzyme-linked immunosorbent assay kit (BD Pharmingen, San Diego, Calif.) with an assay sensitivity of 5 pg of TNF-α/ml. Since the purpose of the study was to determine whether anti-PfGPI antibodies reduced TNF-α levels in the IVS of the placenta, placental, rather than peripheral, plasma was used in the assay. In general, cytokines exert their effects locally, and previous studies have shown that for women with placental malaria, cytokine levels are higher in the placental blood than in the peripheral blood (18).
Histopathology.
Histological studies were conducted with placental tissues that had been fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned, and stained with hematoxylin-eosin. All sample slides were coded and read by two individuals who were blind to the study conditions. Based on the system of classification developed by Bulmer and colleagues (8), women were classified as being uninfected, having an active malaria infection, having an active chronic infection, or having had a past infection based on the presence and localization of P. falciparum-infected erythrocytes and/or hemozoin pigment within histological tissue sections (8).
Measurement of anti-PfGPI IgM and IgG antibodies in peripheral plasma.
Anti-PfGPI IgM and IgG antibodies were measured in peripheral plasma since peripheral blood constantly supplies antibodies to the IVS, where they can neutralize the toxic effects of parasite GPIs. Placental plasma was not used in this part of the study because parasite antigens in placental blood samples can form immune complexes that would affect the results. Previous studies have found no significant difference in antibody levels to other malarial antigens in peripheral and placental sera (28).
P. falciparum GPI was purified by high-performance liquid chromatography as described previously (24). Purified GPI was dissolved in methanol and was coated at 2 ng/well into flat-bottomed 96-well microtiter plates (Maxisorp, Nunc, Denmark). The plates were incubated overnight at 37°C, washed once with phosphate-buffered saline (pH 7.2) with 0.05% Tween 20 (the washing buffer used throughout), and blocked with Tris-buffered saline (pH 7.4) containing 0.5% casein. After a 1-h incubation at 37°C, the plates were washed three times. Plasma samples were diluted 1:100 in diluting buffer (Tris-buffered saline with 0.5% casein and 0.05% Tween 20), and 50 μl was added to each well. The plates were incubated at 37°C for 1 h and then washed five times. Diluted conjugates (horseradish peroxidase-conjugated anti-human IgM from Southern Biotech, Birmingham, Ala., and horseradish peroxidase-conjugated anti-human IgG from KPL, Inc., Gaithersburg, Md.) were applied, incubated for 1 h at 37°C, and washed. Peroxidase substrate (ABTS Microwell; BioFX Laboratories, Inc., Owings Mills, Md.) was added, and plates were incubated for 30 min. Optical densities (ODs) were read at 450 nm. A sample that was previously determined to have high antibody levels to P. falciparum GPI was used as a positive control. Pooled plasma from individuals not exposed to malaria was used as the negative control. Samples were considered to be anti-PfGPI IgM or IgG positive if the ODs were greater than the mean + 2 standard deviations of the negative controls.
Statistical analyses.
Pearson's χ2 test was employed for univariate, between-group comparisons of binomial variables (e.g., chemoprophylaxis use during pregnancy, percent primigravidae, PTD, and antibody responders). The Wilcoxon signed-rank test was used to compare peripheral and placental parasitemias for each subject. The Wilcoxon rank sum test was used in univariate, between-group comparisons of continuous variables, including age and infant birth weight, and for analysis of anti-PfGPI IgM and IgG levels. The Kruskal-Wallis test was used to examine anti-PfGPI IgM and IgG levels in relation to histological results. The Spearman's correlation coefficient was used to determine the association between anti-PfGPI antibody concentrations and (i) maternal age, (ii) parasite density in the placenta, (iii) placental TNF-α concentrations, (iv) infant birth weight, and (v) gestational age. A logistic regression model was used to evaluate the influence of maternal age, gravidity, malaria infection, infant birth weight, and gestational age on anti-PfGPI IgG levels. The P values of type 3 logistic regression statistics are reported. P values of <0.05 were considered to be significant.
RESULTS
Women included in the study.
A total of 42 nonpregnant and 203 pregnant malaria-negative women and 177 pregnant women with placental malaria were evaluated (Table 1). Mean ages were similar among the three groups of women. Consistent with previous studies, median parasitemias were greater in the placenta than in peripheral blood (P < 0.0001). Those with placental malaria were more likely to be primigravidae (P = 0.027) and to deliver babies with lower mean birth weights than those without placental malaria (P < 0.0001) (Table 1). Clearly, placental malaria had a negative impact on pregnancy outcome.
Anti-PfGPI antibodies in nonpregnant and pregnant women.
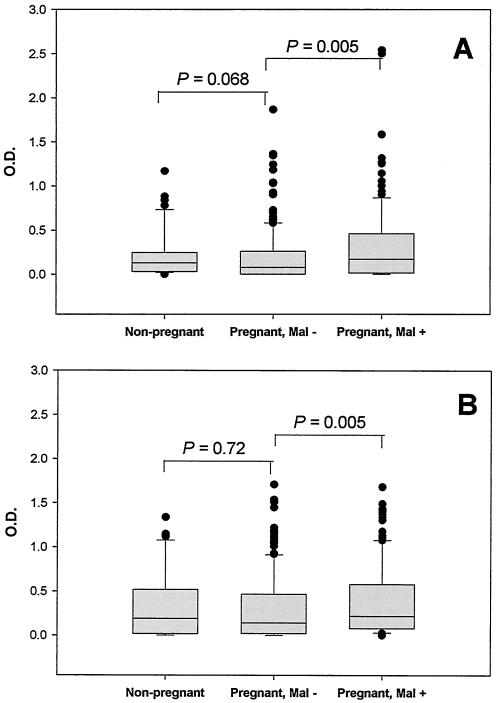
The prevalence of anti-PfGPI IgM was significantly lower in pregnant malaria-negative women than in nonpregnant malaria-negative women (P < 0.0001) (Table 2). The concentrations of anti-PfGPI IgM antibodies were also lower among pregnant women, with the difference being borderline significant (P = 0.068) (Fig. 1A). No significant difference in either the prevalence or the amount of anti-PfGPI IgG antibodies was found between nonpregnant and pregnant women who were malaria negative (Table 1 and Fig. 1B).
TABLE 2.
Proportion of women with anti-PfGPI antibodies
| Characteristic and group | No. of women | Anti-PfGPI IgM
|
Anti-PfGPI IgG
|
||
|---|---|---|---|---|---|
| % Prevalence | Pa | % Prevalence | Pa | ||
| Malaria negative | |||||
| Pregnant women | 203 | 72 | <0.0001 | 83 | 0.75 |
| Nonpregnant women | 42 | 98 | 80 | ||
| Placental malaria | |||||
| Pregnant women, malaria negative | 203 | 72 | 0.13 | 83 | <0.0001 |
| Pregnant women, malaria positive | 177 | 80 | 98 | ||
| Malaria negative | |||||
| Primigravidae | 48 | 63 | 0.12 | 73 | 0.036 |
| Multigravidae | 155 | 75 | 86 | ||
| Malaria positive | |||||
| Primigravidae | 60 | 78 | 0.73 | 95 | 0.40 |
| Multigravidae | 117 | 81 | 97 | ||
| Age | |||||
| Pregnant women ≤20 yr | 75 | 70 | 0.27 | 88 | 0.63 |
| Pregnant women >20 yr | 305 | 77 | 90 | ||
Determined by Pearson's χ2 test.
FIG. 1.
Anti-PfGPI antibody levels in malaria-negative, nonpregnant women and in pregnant women without and with placental malaria (Mal − and Mal +, respectively). Anti-PfGPI IgM (A) and IgG (B) levels were measured in the peripheral plasma, and results from antibody-positive women are displayed in box plots. The horizontal line within each box represents the median OD value, with the upper and lower vertical bars indicating the 90th and 10th percentiles, respectively. Anti-PfGPI IgM and IgG levels were significantly elevated in women with placental malaria compared to those without placental malaria. The Wilcoxon rank sum test was used for between-group comparisons.
Among pregnant women, the prevalence of anti-PfGPI IgG, but not IgM, antibodies was higher in women with placental malaria than in those without (98% versus 83%, respectively; χ2 = 19.14, P < 0.0001) (Table 2). The concentrations of both anti-PfGPI IgM and IgG antibodies were also significantly higher in women with placental malaria (P = 0.005 for both) (Fig. 1). Furthermore, a low yet statistically significant positive correlation was observed between placental parasite densities and levels of anti-PfGPI IgM (Spearman's ρ = 0.18, P = 0.002) and anti-PfGPI IgG (Spearman's ρ = 0.15, P = 0.005). These results demonstrate that the presence of placental P. falciparum parasites was associated with an increase in the production of anti-PfGPI IgM and IgG antibodies in pregnant women.
Anti-PfGPI IgM and IgG antibodies increase with gravidity and age.
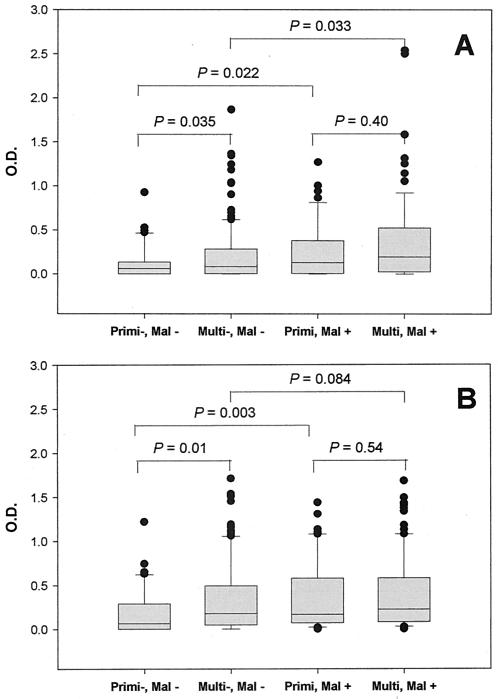
In Yaoundé, being primigravida and ≤20 years of age are risk factors for placental malaria (44). Thus, the influence of gravidity and age on anti-PfGPI antibodies was investigated. Analysis of the serological results from secundigravidae revealed they did not differ significantly from those of multigravid women. Thus, secundigravidae were included in the multigravid group. The prevalences of anti-PfGPI IgM antibodies were similar in malaria-negative primigravidae and multigravidae (χ2 = 2.36, P = 0.12), but the prevalence of anti-PfGPI IgG antibodies was significantly higher in multigravid women (86% for multigravidae versus 73% for primigravidae; χ2 = 4.38, P = 0.036) (Table 2). In addition, the levels of both anti-PfGPI IgM and IgG antibodies were significantly higher for multigravidae than for primigravidae among malaria-negative women (IgM medians, 0.082 versus 0.059, respectively [P = 0.035]; IgG medians, 0.181 versus 0.064, respectively [P = 0.010]) (Fig. 2). Thus, in malaria-negative women, the prevalence and amount of anti-PfGPI IgG and the amount of IgM antibodies were significantly higher among multigravidae. However, among women with placental malaria, neither the prevalence nor levels of anti-PfGPI IgM and IgG differed significantly between primigravidae and multigravidae (Table 2 and Fig. 2).
FIG. 2.
Comparison of anti-PfGPI IgM and IgG levels between primigravidae (Primi) and multigravidae (Multi). Anti-PfGPI IgM (A) and IgG (B) levels in peripheral plasma of primigravidae and multigravidae, with or without placental malaria (Mal + and Mal −, respectively), are displayed in box plots. The horizontal line within each box represents the median OD value, with the upper and lower vertical bars indicating the 90th and 10th percentiles, respectively. Both anti-PfGPI IgM and IgG levels were significantly lower in malaria-negative primigravidae than in malaria-negative multigravidae. The Wilcoxon rank sum test was employed for between-group comparisons.
No significant difference in the prevalence of anti-PfGPI IgM antibodies was found in women ≤20 years and those >20 years of age (Table 2), and anti-PfGPI IgM levels did not differ significantly between the two groups (medians, 0.099 versus 0.078, respectively; P = 0.150). Further analysis found no association between anti-PfGPI IgM antibodies and age (Spearman's ρ = −0.017, P = 0.77). These results, however, contrast with those for anti-PfGPI IgG. Although the prevalences of anti-PfGPI IgG antibodies were similar (P = 0.63) (Table 2), among antibody-positive women, those who were >20 years of age possessed significantly higher levels of anti-PfGPI IgG than women ≤20 years old (medians, 0.195 versus 0.102, respectively; P = 0.018). In addition, a low yet statistically significant correlation between age and the level of anti-PfGPI IgG antibodies was found (Spearman's ρ = 0.10, P = 0.044). Thus, among pregnant women, anti-PfGPI levels were significantly higher in both multigravidae and women >20 years of age.
Lack of correlation between anti-PfGPI antibodies, TNF-α, and stage of placental malaria infection.
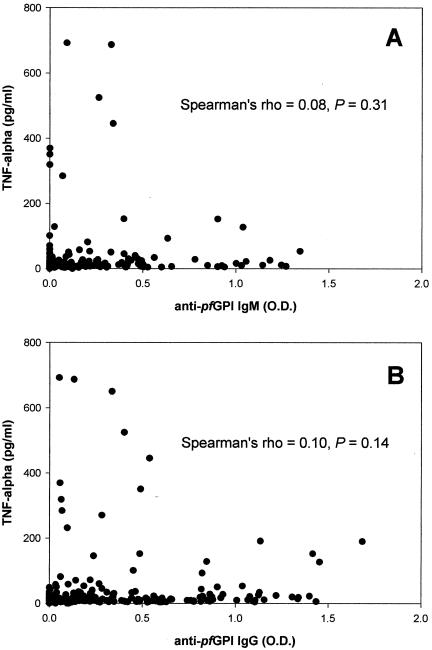
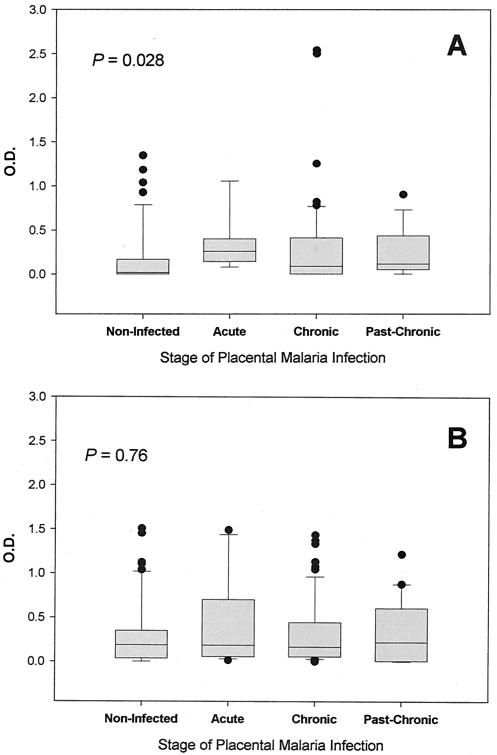
Since levels of anti-PfGPI IgG antibodies were significantly higher in multigravidae and anti-PfGPI antibodies can counteract the ability of P. falciparum to stimulate the induction of TNF-α (4, 34), the potential role of anti-PfGPI antibodies in regulating placental TNF-α concentrations was investigated. Results are summarized in Fig. 3. No significant correlation was found between TNF-α levels in placental plasma and either anti-PfGPI IgM antibodies (Spearman's ρ = 0.08, P = 0.31) (Fig. 3A) or anti-PfGPI IgG antibodies (Spearman's ρ = 0.10, P = 0.14) (Fig. 3B). Although anti-PfGPI IgM levels were higher in women with acute placental infections than in women without placental infections (P = 0.013) (Fig. 4A), no significant association between anti-PfGPI IgG levels and the presence of acute, chronic, or past placental malarial infections was observed (Fig. 4B). Taken together, these results show that anti-PfGPI IgM and IgG antibodies do not have a significant impact on placental TNF-α levels or modulate the inflammatory response in the placentas of Cameroonian women.
FIG. 3.
Scatter plots between TNF-α and anti-PfGPI antibodies. Anti-PfGPI IgM (A) and IgG (B) levels were plotted against their corresponding placental TNF-α concentrations within the same individual. Spearman's rho coefficient was measured to determine the degree of correlation between the variables.
FIG. 4.
Comparison of anti-PfGPI IgG and IgM levels in women with different stages of placental malaria infection. Anti-PfGPI IgM (A) and IgG (B) levels in the peripheral plasma of women with no evidence of placental infection or with acute, chronic, or past infections are displayed in box plots. The horizontal line within each box represents the median OD value, with the upper and lower vertical bars indicating the 90th and 10th percentiles, respectively. The Kruskal-Wallis test was employed for among-group comparisons.
Anti-PfGPI antibodies and birth outcomes.
In Yaoundé, placental malaria is a significant risk factor for PTD and LBW infants in primigravidae but not in multigravidae. Thus, in this part of the study, only data from primigravidae were used in the analysis. Results showed that anti-PfGPI IgM and IgG antibody levels were similar between women with LBW and normal-birth-weight infants (IgM medians, 0.192 versus 0.126, respectively [P = 0.49]; IgG medians, 0.151 versus 0.175, respectively [P = 0.72]). No significant correlation was found between infant birth weight and levels of either anti-PfGPI IgM (Spearman's ρ = −0.027, P = 0.84) or anti-PfGPI IgG (Spearman's ρ = −0.034, P = 0.52). Although malaria-infected women with FTD had higher levels of anti-PfGPI IgM and IgG than women with PTD, the difference was not statistically significant (IgM medians, 0.176 versus 0.143, respectively [P = 0.77]; IgG medians, 0.216 versus 0.189, respectively [P = 0.57]). There was also no significant correlation between the number of weeks of gestation and the level of anti-PfGPI IgM or anti-PfGPI IgG antibodies (Spearman's ρ = −0.023, P = 0.81, and Spearman's ρ = −0.014, P = 0.85, respectively).
A logistic regression model was used to simultaneously examine the effects of maternal age, gravidity, presence of placental malaria, birth weight outcome, and gestational age on the levels of anti-PfGPI IgM and IgG antibodies in all the pregnant women studied. Only the presence of placental malaria remained significantly associated with anti-PfGPI IgM levels (P = 0.002), and maternal age and placental malaria were significantly associated with the levels of anti-PfGPI IgG antibodies (P = 0.004 and 0.030, respectively) after adjusting for other covariates.
DISCUSSION
Both antiparasitic and antidisease or antitoxic immune responses are important in mediating immunity against malaria. Most research on vaccine development has focused on the prevention of infection by targeting proteins expressed on the surface of asexual-stage parasites (15). Once an individual becomes infected, however, the severity of symptoms is largely dependent on the virulence of the parasite strain(s) and the extent of the host's inflammatory response (1, 16). A role for P. falciparum GPI anchors in malaria pathogenesis has been reported previously (14, 33), as these molecules are able to elicit proinflammatory immune responses through the induction of TNF-α from host monocytes and macrophages (43). Anti-PfGPI antibodies have been associated with protection against clinical malaria in children, most likely through antidisease immunity (24).
Malaria symptoms are more frequent and severe during pregnancy (7, 11). An increase in the number of macrophages migrating to the IVS of the placenta during malaria infection has been linked to PTD (21). These cells are a significant source of TNF-α in the placenta (37), and elevated levels of TNF-α have been observed in malaria-infected women with PTD (38). Since anti-PfGPI antibodies can neutralize the induction of TNF-α in macrophages in vitro (4, 34), it was hypothesized that high levels of anti-PfGPI antibodies in pregnant women might correlate with decreased levels of TNF-α within the placenta and that in turn would be associated with improved pregnancy outcomes. However, in the present study, no significant correlation was found between the concentrations of circulating anti-PfGPI antibodies and placental TNF-α concentrations. Anti-PfGPI antibody levels also did not differ in women for whom there was no histological evidence of having placental malaria during pregnancy, women with chronic infections, and women who had cleared their infections (Fig. 4). The only exception was the elevated levels of anti-PfGPI IgM in women with acute placental malaria infections (Fig. 4A), which was not surprising since carbohydrate antigens from parasites are known to induce IgM responses early in infection (26). There was also no significant difference in anti-PfGPI antibody levels in malaria-infected women who delivered prematurely and at term.
In the present study, the malaria infection status of the women and the levels of anti-PfGPI antibodies were evaluated only at the time of delivery. The prevalence of malaria peaks around the 12th to the 16th week of pregnancy (20). It is therefore plausible that in some women who may have been infected with malaria early in pregnancy, the anti-PfGPI antibodies that were induced helped prevent pathology, and then antibody levels waned before delivery, since it was recently reported that the human immune response to P. falciparum GPI is transient (6). Thus, a role for anti-PfGPI antibodies in conferring protection against placental malaria cannot be totally discounted. However, the results presented herein fail to provide evidence for a significant role for anti-PfGPI antibodies in conferring protection against chronic placental malaria infection and poor birth outcomes. The association between placental malaria infection and increased levels of anti-PfGPI IgM and IgG antibodies in pregnant women may simply be due to antibody boosting that has been shown to be dependent upon exposure to antigen (10).
Maternal humoral responses generally remain unaffected by pregnancy (19). In the present study, a significant decrease in both the prevalence and levels of anti-PfGPI IgM antibodies was found in noninfected pregnant women compared to nonpregnant women (Table 2 and Fig. 1A), whereas anti-PfGPI IgG responses were similar (Table 2 and Fig. 1B). These findings are in direct contrast to those previously reported by Nambei and colleagues (25), where IgM responses to the merozoite surface protein 1 antigen were increased among parturients compared to nonpregnant women (25). These results demonstrate, as reported previously, that changes in IgM responses during pregnancy may vary depending on the antigen (12, 23). Although anti-PfGPI IgM antibodies may be able to inhibit the induction of TNF-α (3), protection against clinical malaria is primarily mediated by cytophilic IgG antibodies and not IgM (5). Thus, a reduced anti-PfGPI IgM response in pregnant women may be of minimal clinical importance.
The acquisition of anti-PfGPI antibodies is age dependent (10, 17). Recently, Hudson Keenihan and colleagues (17) cited age as an important factor that influences both the quantity and quality of the IgG immune response towards P. falciparum GPI (17). A common theme that is emerging is that children from areas of malaria endemicity usually possess low concentrations of anti-PfGPI antibodies. However, those few who are able to mount a strong response are less likely to experience symptoms of clinical malaria, including anemia and fever (17, 24). While the protective effect of anti-PfGPI is evident in children, it is unclear if the same effect holds true for adults (17). In the present study, the concentrations of anti-PfGPI IgG antibodies were significantly higher among pregnant women >20 years of age than among pregnant women ≤20 years old, demonstrating that anti-PfGPI antibodies do increase with age. Since multigravidae are generally older than primigravidae, it was not surprising to find that multigravidae also possessed significantly higher concentrations of anti-PfGPI IgM and IgG antibodies than did primigravidae (Table 2 and Fig. 2). Indeed, results from the logistic regression model identified maternal age and placental malaria as the only significant factors to influence the level of anti-PfGPI antibodies after adjusting for other variables.
In summary, our results indicate that placental malaria boosts anti-PfGPI antibody levels and that anti-PfGPI antibody concentrations increase with gravidity and age. These levels, however, did not correlate with placental TNF-α concentrations, placental pathology, or pregnancy outcomes. Thus, while anti-PfGPI antibodies may have a clinically significant role that has yet to be identified, these results do not provide evidence that they influence events that lead to malaria-associated poor birth outcomes.
Acknowledgments
We express our gratitude to all the Cameroonian women who participated in this study. We are indebted to the entire technical staff and technicians of the Biotechnology Center of the University of Yaoundé, including the medical students who were involved in sample collection, for their immense help.
This project has been funded by the NIAID, NIH, through the Human Immune Resistance to Malaria in Endemic Areas (HIRE-Malaria) Program (no. UO1AI 43888).
Editor: W. A. Petri, Jr.
REFERENCES
- 1.Allan, R. J., A. Rowe, and D. Kwiatkowski. 1993. Plasmodium falciparum varies in its ability to induce tumor necrosis factor. Infect. Immun. 61:4772-4776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Andrews, K. T., and M. Lanzer. 2002. Maternal malaria: Plasmodium falciparum sequestration in the placenta. Parasitol. Res. 88:715-723. [DOI] [PubMed] [Google Scholar]
- 3.Bate, C. A., and D. Kwiatkowski. 1994. Inhibitory immunoglobulin M antibodies to tumor necrosis factor-inducing toxins in patients with malaria. Infect. Immun. 62:3086-3091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bate, C. A., and D. Kwiatkowski. 1994. A monoclonal antibody that recognizes phosphatidylinositol inhibits induction of tumor necrosis factor alpha by different strains of Plasmodium falciparum. Infect. Immun. 62:5261-5266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bouharoun-Tayoun, H., and P. Druilhe. 1992. Plasmodium falciparum malaria: evidence for an isotype imbalance which may be responsible for delayed acquisition of protective immunity. Infect. Immun. 60:1473-1481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Boutlis, C. S., P. K. Fagan, D. C. Gowda, M. Lagog, C. S. Mgone, M. J. Bockarie, and N. M. Anstey. 2003. Immunoglobulin G (IgG) responses to Plasmodium falciparum glycosylphosphatidylinositols are short-lived and predominantly of the IgG3 subclass. J. Infect. Dis. 187:862-865. [DOI] [PubMed] [Google Scholar]
- 7.Brabin, B. J. 1991. The risks and severity of malaria in pregnant women, p. 1-34. In Applied field research in malaria. Report no. 1. World Health Organization, Geneva, Switzerland.
- 8.Bulmer, J. N., F. N. Rasheed, N. Francis, L. Morrison, and B. M. Greenwood. 1993. Placental malaria. I. Pathological classification. Histopathology 22:211-218. [DOI] [PubMed] [Google Scholar]
- 9.Clark, I. A., and G. Chaudhri. 1988. Tumor necrosis factor in malaria-induced abortion. Am. J. Trop. Med. Hyg. 39:246-249. [DOI] [PubMed] [Google Scholar]
- 10.De Souza, J. B., J. Todd, G. Krishegowda, D. C. Gowda, D. Kwiatkowski, and E. M. Riley. 2002. Prevalence and boosting of antibodies to Plasmodium falciparum glycosylphosphatidylinositols and evaluation of their association with protection from mild and severe clinical malaria. Infect. Immun. 70:5045-5051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Diagne, N., C. Rogier, B. Cisse, and J.-F. Trape. 1997. Incidence of clinical malaria in pregnant women exposed to intense perennial transmission. Trans. R. Soc. Trop. Med. Hyg. 91:166-170. [DOI] [PubMed] [Google Scholar]
- 12.Fievet, N., M. Cot, C. Chougnet, B. Maubert, J. Bickii, B. Dubois, J. Y. Lehesran, Y. Frobert, F. Migot, F. Romain, et al. 1995. Malaria and pregnancy in Cameroonian primigravidae—humoral and cellular immune responses to Plasmodium falciparum blood-stage antigens. Am. J. Trop. Med. Hyg. 53:3612-3617. [DOI] [PubMed] [Google Scholar]
- 13.Fried, M., R. O. Muga, A. O. Misore, and P. E. Duffy. 1998. Malaria elicits type 1 cytokines in the human placenta: IFN-γ and TNF-α associated with pregnancy outcomes. J. Immunol. 160:2523-2530. [PubMed] [Google Scholar]
- 14.Gerold, P., A. Dieckmann-Schuppert, and R. Schwarz. 1994. Glycosylphosphatidylinositols synthesized by asexual erythrocytic stages of the malarial parasite, Plasmodium falciparum. Candidates for plasmodial glycosylphosphatidylinositol membrane anchor precursors and pathogenicity factors. J. Biol. Chem. 269:2597-2606. [PubMed] [Google Scholar]
- 15.Good, M. F. 2001. Towards a blood-stage vaccine for malaria: are we following all the leads? Nat. Rev. Immunol. 1:117-125. [DOI] [PubMed] [Google Scholar]
- 16.Gupta, S., A. V. Hill, D. Kwiatkowski, A. M. Greenwood, B. M. Greenwood, and K. P. Day. 1994. Parasite virulence and disease patterns in Plasmodium falciparum malaria. Proc. Natl. Acad. Sci. USA 91:3715-3719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hudson Keenihan, S. N., S. Ratiwayanto, S. Soebianto, Krisin, H. Marwoto, G. Krishnegowda, D. C. Gowda, M. J. Bangs, D. J. Fryauff, T. L. Richie, S. Kumar, and J. K. Baird. 2003. Age-dependent impairment of IgG responses to glycosylphosphatidylinositol with equal exposure to Plasmodium falciparum among Javanese migrants to Papua, Indonesia. Am. J. Trop. Med. Hyg. 69:36-41. [PubMed] [Google Scholar]
- 18.Jakobsen, P. H., F. N. Rasheed, J. N. Bulmer, M. Theisen, R. G. Ridley, and B. M. Greenwood. 1998. Inflammatory reactions in placental blood of Plasmodium falciparum-infected women and high concentrations of soluble E-selectin and a circulating P. falciparum protein in the cord sera. Immunology 93:264-269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Luppi, P. 2003. How immune mechanisms are affected by pregnancy. Vaccine 21:3352-3357. [DOI] [PubMed] [Google Scholar]
- 20.McGregor, I. A. 1984. Epidemiology, malaria, and pregnancy. Am. J. Trop. Med. Hyg. 33:517-525. [DOI] [PubMed] [Google Scholar]
- 21.Menendez, C., J. Ordi, M. R. Ismail, P. J. Ventura, J. J. Aponte, E. Kahigwa, F. Font, and P. L. Alonso. 2000. The impact of placental malaria on gestational age and birth weight. J. Infect. Dis. 181:1740-1745. [DOI] [PubMed] [Google Scholar]
- 22.Miller, L. H., D. I. Baruch, K. Marsh, and O. K. Doumbo. 2002. The pathogenic basis of malaria. Nature 415:673-678. [DOI] [PubMed] [Google Scholar]
- 23.Mvondo, J. L., M. A. James, A. J. Sulzer, and C. C. Campbell. 1992. Malaria and pregnancy in Cameroonian women. Naturally acquired antibody responses to asexual blood-stage antigens and the circumsporozoite protein of Plasmodium falciparum. Trans. R. Soc. Trop. Med. Hyg. 86:486-490. [DOI] [PubMed] [Google Scholar]
- 24.Naik, R. S., O. H. Branch, A. S. Woods, M. Vijaykumar, D. J. Perkins, B. L. Nahlen, A. A. Lal, R. J. Cotter, C. E. Costello, C. F. Ockenhouse, E. A. Davidson, and D. C. Gowda. 2000. Glycosylphosphatidylinositol anchors of Plasmodium falciparum: molecular characterization and naturally elicited antibody response that may provide immunity to malaria pathogenesis. J. Exp. Med. 192:1563-1575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Nambei, W. S., M. Goumbala, A. Spiegel, A. Diéye, R. Perraut, and O. Garraud. 1998. Imbalanced distribution of IgM and IgG antibodies against Plasmodium falciparum antigens and merozoite surface protein-1 (MSP1) in pregnancy. Immunol. Lett. 61:197-199. [DOI] [PubMed] [Google Scholar]
- 26.Nyame, A. K., F. A. Lewis, B. L. Doughty, R. Correa-Oliveira, and R. D. Cummings. 2003. Immunity to schistosomiasis: glycans are potential antigenic targets for immune intervention. Exp. Parasitol. 104:1-13. [DOI] [PubMed] [Google Scholar]
- 27.Odeh, M. 2001. The role of tumour necrosis factor-α in the pathogenesis of complicated falciparum malaria. Cytokine 14:11-18. [DOI] [PubMed] [Google Scholar]
- 28.Rasheed, F. N., J. N. Bulmer, D. T. Dunn, C. Menendez, M. F. Jawla, A. Jepson, P. H. Jakobsen, and B. M. Greenwood. 1993. Suppressed peripheral and placental blood lymphoproliferative responses in first pregnancies: relevance to malaria. Am. J. Trop. Med. Hyg. 48:154-160. [DOI] [PubMed] [Google Scholar]
- 29.Rockett, K. A., M. M. Awburn, B. B. Aggarwal, W. B. Cowden, and I. A. Clark. 1992. In vivo induction of nitrite and nitrate by tumor necrosis factor, lymphotoxin, and interleukin-1: possible roles in malaria. Infect. Immun. 60:3725-3730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rockett, K. A., D. Kwiatkowski, C. A. Bate, M. M. Awburn, E. J. Rockett, and I. A. Clark. 1996. In vitro induction of nitric oxide by an extract of Plasmodium falciparum. J. Infect. 32:187-196. [DOI] [PubMed] [Google Scholar]
- 31.Rogerson, S. J., H. C. Brown, E. Pollina, E. T. Abrams, E. Tadesse, V. M. Lema, and M. E. Molyneux. 2003. Placental tumor necrosis factor alpha but not gamma interferon is associated with placental malaria and low birth weight in Malawian women. Infect. Immun. 71:267-270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ropert, C., and T. R. Gazzinelli. 2000. Signaling of immune system cells by glycosylphosphatidylinositol (GPI) anchor and related structures derived from parasitic protozoa. Curr. Opin. Microbiol. 3:395-403. [DOI] [PubMed] [Google Scholar]
- 33.Schofield, L., and F. Hackett. 1993. Signal transduction in host cells by a glycosylphosphatidylinositol toxin of malaria parasites. J. Exp. Med. 177:145-153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Schofield, L., I. Vivas, and F. Hackett. 1993. Neutralizing monoclonal antibodies to glycosylphosphatidylinositol, the dominant TNF-α-inducing toxin of Plasmodium falciparum: prospects for the immunotherapy of severe malaria. Ann. Trop. Med. Parasitol. 87:617-626. [DOI] [PubMed] [Google Scholar]
- 35.Schofield, L., S. Novakovic, P. Gerold, R. T. Schwarz, M. J. McConville, and S. D. Tachado. 1996. Glycosylphosphatidylinositol toxin of Plasmodium upregulates intercellular adhesion molecule-1, vascular cell adhesion molecule-1, and E-selectin expression in vascular endothelial cells and increases leukocyte and parasite cytoadherence via tyrosine kinase-dependent signal transduction. J. Immunol. 156:1886-1896. [PubMed] [Google Scholar]
- 36.Schofield, L., M. C. Hewitt, K. Evans, M. Siomos, and P. H. Seeberger. 2002. Synthetic GPI as a candidate anti-toxic vaccine in a model of malaria. Nature 418:785-789. [DOI] [PubMed] [Google Scholar]
- 37.Suguitan, A. L., Jr., R. G. F. Leke, G. Fouda, A. Zhou, L. Thuita, S. Metenou, J. Fogako, R. Megnekou, and D. W. Taylor. 2003. Changes in the levels of chemokines and cytokines in the placentas of women with Plasmodium falciparum malaria. J. Infect. Dis. 188:1074-1082. [DOI] [PubMed] [Google Scholar]
- 38.Suguitan, A. L., Jr., T. J. Cadigan, T. A. Nguyen, A. Zhou, R. J. I. Leke, S. Metenou, L. Thuita, R. Megnekou, J. Fogako, R. G. F. Leke, and D. W. Taylor. 2003. Malaria-associated cytokine changes in the placenta of women with pre-term deliveries in Yaoundé, Cameroon. Am. J. Trop. Med. Hyg. 69:574-581. [PubMed] [Google Scholar]
- 39.Tachado, S. D., P. Gerold, R. Schwarz, S. Novakovic, M. McConville, and L. Schofield. 1997. Signal transduction in macrophages by glycosylphosphatidylinositols of Plasmodium, Trypanosoma, and Leishmania: activation of protein tyrosine kinases and protein kinase C by inositolglycan and diacylglycerol moieties. Proc. Natl. Acad. Sci. USA 94:4022-4027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Tachado, S. D., P. Gerold, M. J. McConville, T. Baldwin, D. Quilici, R. T. Schwarz, and L. Schofield. 1997. Glycosylphosphatidylinositol toxin of Plasmodium induces nitric oxide synthase expression in macrophages and vascular endothelial cells by a protein tyrosine kinase-dependent and protein kinase C-dependent signaling pathway. J. Immunol. 156:1897-1907. [PubMed] [Google Scholar]
- 41.Taverne, J., C. A. Bate, D. A. Sarkar, A. Meager, G. A. Rook, and J. H. Playfair. 1990. Human and murine macrophages produce TNF in response to soluble antigens of Plasmodium falciparum. Parasite Immunol. 12:33-43. [DOI] [PubMed] [Google Scholar]
- 42.Troye-Blomberg, M., G. Anderson, L. N. Otoo, S. Jepsen, and B. M. Greenwood. 1985. Production of IL-2 and IFN-gamma by T cells from malaria patients in response to Plasmodium falciparum or erythrocyte antigens in vitro. J. Immunol. 135:3498-3504. [PubMed] [Google Scholar]
- 43.Vijaykumar, M., R. S. Naik, and D. C. Gowda. 2001. Plasmodium falciparum glycosylphosphatidylinositol-induced TNF-α secretion by macrophages is mediated without membrane insertion or endocytosis. J. Biol. Chem. 276:6909-6912. [DOI] [PubMed] [Google Scholar]
- 44.Zhou, A., R. Megnekou, R. Leke, J. Fogako, S. Metenou, B. Trock, D. W. Taylor, and R. G. F. Leke. 2002. Prevalence of Plasmodium falciparum infection in pregnant Cameroonian women. Am. J. Trop. Med. Hyg. 67:566-570. [DOI] [PubMed] [Google Scholar]