Abstract
Differences in infection kinetics and host response between Burkholderia multivorans and Burkholderia cenocepacia were demonstrated in a pulmonary infection model in BALB/c mice. B. multivorans persisted in the lung, while B. cenocepacia was cleared. Indirect immunofluorescence and electron microscopy of B. multivorans-infected lungs localized bacteria to macrophages. Clearance of B. cenocepacia was associated with greater interleukin-1β and neutrophil responses than the responses induced by B. multivorans.
Members of the Burkholderia cepacia complex (BCC) are opportunistic pathogens in patients with cystic fibrosis (CF) (8). In Canada, the outcome of infection with members of the BCC appears to be species dependent (16). The worst prognoses are associated with Burkholderia cenocepacia infections, which have the highest rates of transmissibility and mortality. Burkholderia multivorans infections are also common among CF patients, but these infections are associated with lower rates of transmissibility and mortality. The mechanisms underlying the pathogenetic differences in these two species are poorly understood (7). Many potential virulence factors for BCC clinical isolates have been identified and evaluated, including a type III secretion system, the cable pilus, flagellin, and ornibactin (9, 13, 15, 18, 19, 21). However, these determinants are typically studied in epidemic CF isolates and therefore almost always represent a virulence determinant for B. cenocepacia. A cross-species comparison of BCC virulence in a pulmonary model has not been done previously.
We used a model of pulmonary infection that shows clear differences between infection with B. cenocepacia and infection with B. multivorans. In this intranasal infection model of BALB/c mice, B. cenocepacia strain C6433 promoted a more vigorous host response and was rapidly cleared from the lung, whereas B. multivorans strain C5568 established a persistent infection in pulmonary macrophages.
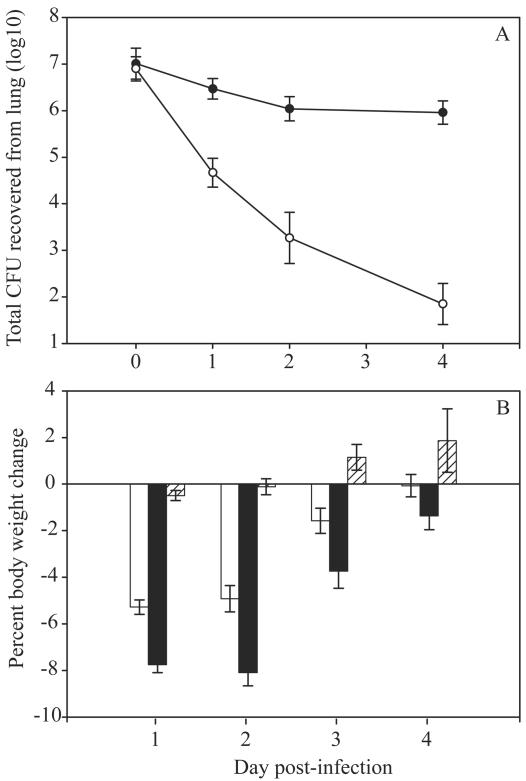
B. multivorans C5568 and B. cenocepacia C6433 were recovered from patients with CF and are part of the Canadian B. cepacia Research and Referral Repository. They were stored at −70°C in Mueller-Hinton broth. Animal studies were approved by the University of British Columbia Animal Care Committee (UBC-ACC no. A98-0138). Intranasal infection experiements were conducted with healthy female mice as previously described (5). All results are expressed below as means ± standard errors of the means. All data were analyzed by the Mann-Whitney test for nonparametric, unpaired values. P values of <0.05 were considered statistically significant. B. multivorans C5568 persisted in the lungs of BALB/c mice, while B. cenocepacia C6433 was cleared (Fig. 1A). Mice infected with B. multivorans sustained a pulmonary infection for 4 days. In contrast, B. cenocepacia was cleared quickly from the lungs of infected mice.
FIG. 1.
Pulmonary bacterial load and body weight change after infection with B. multivorans and B. cenocepacia. BALB/c mice were intranasally challenged with ∼1.0 × 107 CFU of bacteria or sterile PBS. (A) Quantitative bacteriology of the lung assessed 3, 24, 48, and 96 h postinfection. •, B. multivorans C5568; ○, B. cenocepacia C6433. (B) Animals were weighed daily. The bars indicate the body weight change from preinfection values on day 0. Open bars, B. multivorans C5568; solid bars, B. cenocepacia C6433; cross-hatched bars, sterile PBS. The data are the mean ± standard error of the mean for six animals at each time point. The total number of animals studied decreased over time as animals were removed for analysis.
The general health of animals was assessed daily by using a three-point system based upon weight, food consumption, and general appearance. Animals were observed for their responses to infection and were considered unwell when two of the following three observations were made: >7% weight loss, <3 g of food consumed daily, or appearance of systemic illness (ruffled coats, huddled position, lethargy). B. cenocepacia C6433 infection caused more weight loss than B. multivorans C5568 infection (Fig. 1B). The mean weight loss in animals infected with B. cenocepacia was significantly greater (P = 0.004) than the mean weight loss in animals infected with B. multivorans on days 1 through 3 of the experiment. A higher proportion of B. cenocepacia-infected animals than of animals given B. multivorans displayed other signs of systemic illness on days 1 and 2 postinfection. On day 1, 62.5% of the mice (30 of 48 mice) challenged with B. cenocepacia but only 27.6% of the mice (13 of 47 mice) challenged with B. multivorans appeared to be ill. On day 2 postinfection, 60.6% of the mice (20 of 33 mice) challenged with B. cenocepacia appeared to be ill, compared with 32.0% of the mice (8 of 25 mice) infected with B. multivorans.
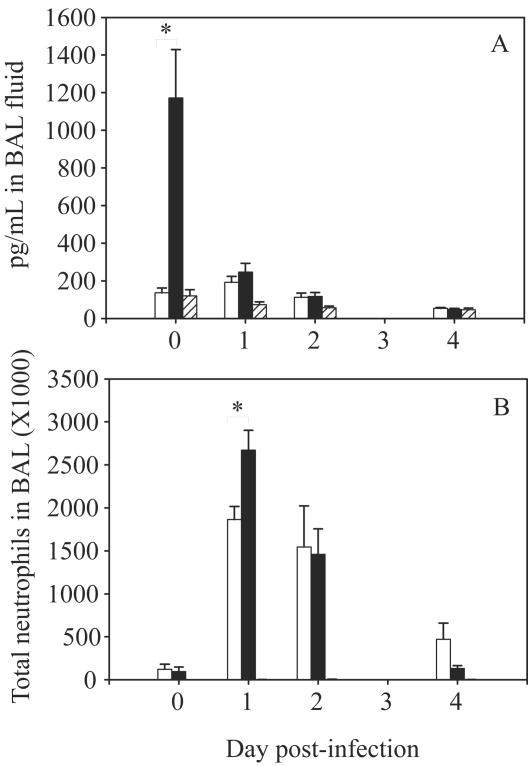
Analyses of pulmonary cells and cytokines were carried out with groups of three mice per time point. Bronchoalveolar lavage (BAL) was performed on euthanized mice in situ as previously described (4). Cells were counted with a hemocytometer; 100-μl aliquots were deposited onto glass slides by using a cytocentrifuge and were stained with Hemacolor (EM Diagnostic Systems, Gibbstown, N.J.) for differential counting. B. cenocepacia C6433-infected mice displayed greater pulmonary host responses than mice infected with B. multivorans C5568 displayed (Fig. 2). The number of neutrophils in the lungs of infected mice peaked on day 1 of infection for both the B. multivorans- and B. cenocepacia-challenged groups (Fig. 2B). The maximal number of neutrophils was significantly greater in the BAL fluid of mice infected with B. cenocepacia than in the BAL fluid of mice infected with B. multivorans (P = 0.0173). The number of neutrophils in the lungs of both groups diminished quickly thereafter, and there was no difference between B. multivorans- and B. cenocepacia-challenged mice in the recovery phase. The number of macrophages recovered from the BAL fluid of infected mice increased on day 1, peaked on day 2 of infection, and was sustained at day 4. There was no difference in the numbers of macrophages recovered from the BAL fluids of mice infected with the two strains (data not shown).
FIG. 2.
Pulmonary host response following bacterial infection. BALB/c mice were intranasally challenged with ∼1.0 × 107 CFU of B. multivorans strain C5568 (open bars), ∼1.0 × 107 CFU of B. cenocepacia strain C6433 (solid bars), or sterile PBS (cross-hatched bars). BAL was performed in situ at 3, 24, 48, and 96 h postinfection. (A) Pulmonary IL-1β profile; (B) pulmonary neutrophil population. The data are the mean ± standard error of the mean for six animals at each time point. An asterisk indicates a significant difference between the C5568- and C6433-infected groups (P < 0.0001 for IL-1β; P = 0.0173 for neutrophils).
BAL fluid was frozen at −70°C until an enzyme-linked immunosorbent assay analysis for mouse interleukin-1β (IL-1β), macrophage inflammatory protein 2 (MIP-2), and tumor necrosis factor alpha (R&D Systems, Minneapolis, Minn.) was performed. All three cytokines exhibited peak recoverable levels for the lavage at 3 h postchallenge, and there was a quick decline in the detectable levels thereafter (Fig. 2A). The level of IL-1β in B. cenocepacia-challenged mice was significantly higher (P < 0.0001) than the level detected in B. multivorans-challenged mice 3 h postinfection. The levels of MIP-2 and tumor necrosis factor alpha in B. cenocepacia-challenged mice did not differ significantly from those in B. multivorans-challenged mice on any day of infection (data not shown).
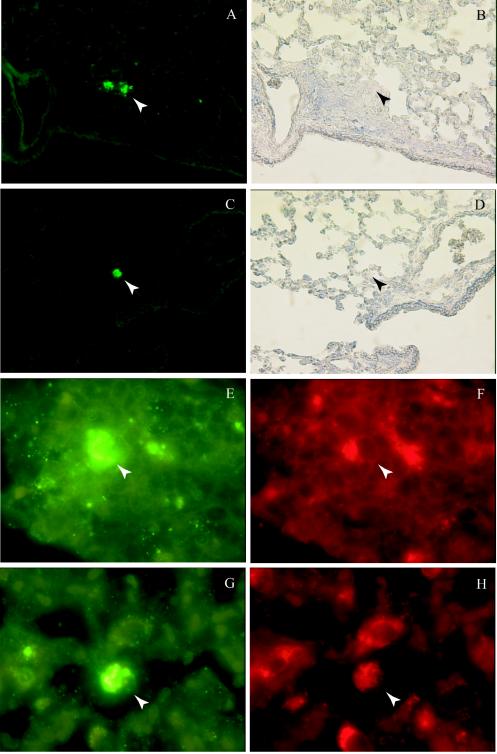
Lung tissue was removed on day 4 of infection and used for immunofluorescent localization of bacteria. Positive staining was observed only in B. multivoran-infected lungs, in accordance with the number of CFU. Lungs and tracheae were excised in toto from euthanized mice, fixed in acetone-methanol, dehydrated, and embedded in paraffin. The antibodies used in this study were rabbit polyclonal sera generated in our laboratory against B. multivorans FC147, rat anti-mouse Mac-3 monoclonal antibody (BD Biosciences), Alexa 488-conjugated goat anti-rabbit antibody, and Alexa 594-conjugated goat anti-rat antibody (Molecular Probes, Eugene, Oreg.). All antibodies were diluted 1/500 in blocking buffer (phosphate-buffered saline [PBS] containing 0.5% [vol/vol] Tween 20 and 5% [vol/vol] normal goat serum). Histological sections were blocked in PBS containing 1% normal goat serum and 0.5% Tween 20 (Sigma). Samples were incubated with primary and secondary antibodies, in sequence, for 1 h at room temperature. Some sections were counterstained with Mayer's hematoxylin (Sigma Diagnostics, St. Louis, Mo.). A persistent B. multivorans infection was localized primarily with Mac-3-positive macrophages in the lungs on day 4 of infection. Lung sections were either immunostained for bacteria prior to Mayer's hematoxylin counterstaining or immunostained for both bacteria and Mac-3 antigen (Fig. 3). Sections immunostained with the B. multivorans-specific polyclonal antibody showed that 39.0% of the bacteria localized to lymphoid aggregates and that 7.3% of these bacteria were organized as bronchus-associated lymphoid tissue (Fig. 3A and B); 56.1% of the bacteria localized with isolated macrophages (Fig. 3C and D), and 4.9% localized with bronchiolar epithelium (data not shown). Similar patterns of bacterium-host association were observed in the sections subjected to double immunostaining. In these sections, 76.5% of the bacteria localized to areas that were also positive for the Mac-3 antigen; 28.8% of these double-positive loci were cells within lymphoid aggregates (Fig. 3E and F), and 51.9% were alveolar macrophages (Fig. 3G and H). Also, 5.8% of the double-positive cells appeared to be in bronchiolar epithelia, whereas 13.5% of the double-positive cells occurred in areas where there was collapse or excessive erythrocyte penetration.
FIG. 3.
Indirect immunofluorescent localization of persistent B. multivorans C5568. BALB/c mice were intranasally challenged with ∼1.0 × 107 CFU of B. multivorans strain C5568, and lung tissue was processed for immunohistological analysis on day 4 of the infection. Sections were either immunostained for bacteria prior to Mayer's hematoxylin counterstaining (A to D) or double immunostained for both bacteria and the Mac-3 antigen without counterstaining (E to H). A positive bacterial signal (green) was localized primarily in lymphoid aggregates (A and B) or with mononuclear cells in the airspace (C and D). Double immunostaining showed that the positive bacterial signal (green) frequently colocalized with Mac-3-positive cells (red) in these areas (lymphoid aggregates [E and F] and macrophages [G and H]), suggesting that macrophages were the primary host cells interacting with B. multivorans strain C5568 at this point in infection.
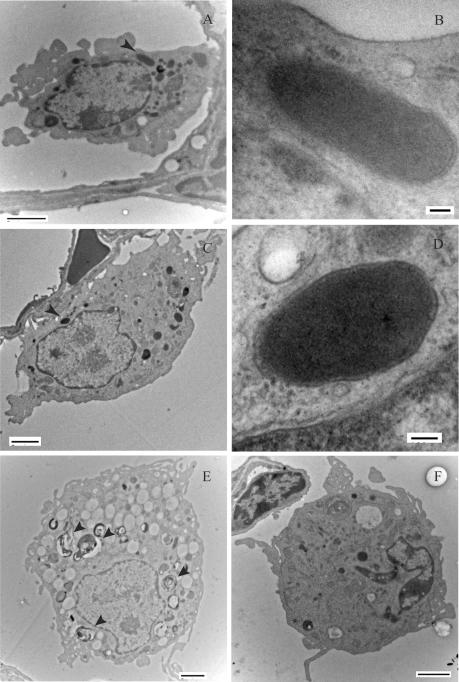
Day 4 B. multivorans-infected lung samples were diced to obtain 1-mm cubes, processed, and stained as previously described (20), except for an initial 1.5-h fixation with 1% glutaraldehyde and the exclusion of tannic acid. Ultrathin sections were viewed with a Hitachi H7600 transmission electron microscope. Electron-dense bacteria were observed in membrane-bound vacuoles of macrophages in the alveolar space (Fig. 4A to D) of B. multivorans-challenged mice. Bacteria were distinguished from cytoplasmic granules by morphology and by the distinct presence of both bacterial outer membranes and vacuolar membranes. Macrophages in B. multivorans-challenged lung samples showed an increased presence of cytoplasmic vacuoles, regardless of whether they contained bacteria; some samples had phagolysosomes containing degraded material (Fig. 4E). No bacteria were observed in conducting airways or lymphoid aggregates by transmission electron microscopy. Macrophages from PBS-challenged lungs appeared to be slightly smaller than macrophages from infected lungs, and they had few cytoplasmic vacuoles and no phagolysosomes (Fig. 4F). No bacteriomorphic, electron-dense, membrane-bound structures were observed in macrophages or any other cell type in PBS-challenged lungs.
FIG. 4.
Transmission electron microscopy of B. multivorans C5568-infected lung (A to E) and PBS-challenged lung (F) at day 4. (A and C) Electron-dense bacteria (arrows) within membrane-bound vacuoles of alveolar macrophages on the fourth day of infection. (B and D) Bacteria were distinguished from cytoplasmic granules by morphology and by the presence of distinct bacterial outer membranes and vacuolar membranes. Panels B and D are high-magnification micrographs of bacteria in panels A and C, respectively. (E) Macrophages in an infected lung were often highly vacuolized, and some cells contained phagolysosomes (arrows). (F) Alveolar macrophage from PBS-challenged lung on day 4. Lower-magnification micrographs were taken at magnifications of ×8,830 to ×15,100, and higher-magnification micrographs were taken at magnifications of ×101,000. (A, C, E, and F) Bars = 2 μm; (B and D) bars = 100 nm.
Here we describe a pulmonary model of BCC infection in an immunocompetent murine host, which differentiates members of two clinically relevant BCC species on the basis of in vivo persistence and toxicity. In this model, persistent B. multivorans is found in association with pulmonary macrophages, while B. cenocepacia is cleared from the lung. The latter organism may be more readily eliminated because it induces a strong inflammatory response from the host cells. The differences in the dynamics of the host responses may facilitate dissection of the mechanisms of BCC infection.
A difference in the host responses to the two BCC strains was observed within hours of pulmonary instillation. Both B. multivorans C5568 and B. cenocepacia C6433 induced peak levels of proinflammatory cytokines in the lung by 3 h postchallenge. However, the level of IL-1β induced by B. cenocepacia was significantly higher than the level induced by B. multivorans. B. cenocepacia infection also resulted in significantly greater pulmonary neutrophil recovery at day 1. Although MIP-2 is associated with neutrophil recruitment (3), the pulmonary levels of this cytokine were not appreciably affected. It is possible that the difference in the number of pulmonary neutrophils 24 h postinfection reflected the levels of IL-1β at 3 h postchallenge, likely generated by resident alveolar macrophages (6). The rapid clearance of B. cenocepacia from the lung could be attributed to the increased expression of IL-1β, which plays a role not only in neutrophil mobilization but also in granule exocytosis and elastase release (1, 14), thereby presenting amplified neutrophil activation in response to B. cenocepacia.
The observation that B. cenocepacia C6433 is rapidly cleared from the murine lung initially appears to be inconsistent with the clinical finding that B. cenocepacia infections in CF patients are chronic and notoriously difficult to eradicate (7). This observation may be related to the use of an inbred strain which is resistant to infection with Pseudomonas, a closely related pathogen, and the absence of the Cftr mutation and the resulting complicated immune scenario in the CF lung (17). In CF, the more phlogistic agents (such as B. cenocepacia) induce a proinflammatory host response that does not effectively clear the provocative stimulus but rather induce pulmonary toxicity. In this model, rapid clearance of B. cenocepacia was accompanied by a level of host toxicity and immunological response not demonstrated in any B. multivorans-challenged mice. The differential host damage and immune stimulation may highlight key elements of virulence and host responses in BCC infection in CF patients.
Both indirect immunofluorescent staining and transmission electron microscopy of infected lungs showed persistent B. multivorans in association with host macrophages on day 4 of infection, indicating that the intracellular localization may protect the bacteria and have a role in diminishing the host response. Whether B. multivorans actively invades host cells or evades host-mediated killing after phagocytosis is unclear. The immunofluorescent localization of persistent bacteria in lymphoid aggregates in the lungs suggests that host macrophages migrate to these areas to present antigen after internalization of bacteria (2, 10). Studies of the host response to Mycobacterium infection in rabbits and guinea pigs have shown that bacilli are found in the bronchus-associated lymphoid tissue during disease (11, 12), which is evidence that pulmonary lymphoid tissue is a site that pathogens do encounter after lung infection. The possibility that these sites could provide an immune-privileged niche for B. multivorans will be explored by localization of bacteria in these structures at a later infection time point.
We established a model of pulmonary infection in immunocompetent mice which discriminates B. multivorans strains from B. cenocepacia strains on the basis of persistence and host response. We propose that B. multivorans C5568 is capable of persisting in the host by establishing an intracellular infection in macrophages and that B. cenocepacia C6433 is cleared rapidly from the host because it is not able to gain such a foothold and in the process induces a more vigorous and damaging host response than strain C5568. This model system also has the capacity to evaluate BCC virulence determinant mutants. In future experiments with these two strains and various pulmonary host cell populations we will examine the molecular relationship between pathogen and host. Such studies should provide further information about the precise mechanisms of intracellular survival and may instruct novel therapeutic approaches.
Acknowledgments
We thank Elaine Humphries and Garnet Martens at the University of British Columbia Bioimaging Facility and Faye Sirianni at St. Paul's Hospital for their help with the transmission electron microscopy work.
This work was supported by an operating grant (to D.P.S.) and traineeships (to K.K.C., D.J.D., and K.L.M.) from the Canadian CF Foundation and by grants from the B.C. Lung Association and the Canadian Institutes for Health Research (to D.P.S.).
Editor: J. T. Barbieri
REFERENCES
- 1.Brandolini, L., R. Bertini, C. Bizzarri, R. Sergi, G. Caselli, D. Zhou, M. Locati, and S. Sozzani. 1996. IL-1 beta primes IL-8-activated human neutrophils for elastase release, phospholipase D activity, and calcium flux. J. Leukoc. Biol. 59:427-434. [DOI] [PubMed] [Google Scholar]
- 2.Breel, M., M. Van der Ende, T. Sminia, and G. Kraal. 1988. Subpopulations of lymphoid and non-lymphoid cells in bronchus-associated lymphoid tissue (BALT) of the mouse. Immunology 63:657-662. [PMC free article] [PubMed] [Google Scholar]
- 3.Broxmeyer, H. E., S. Cooper, G. Cacalano, N. L. Hague, E. Bailish, and M. W. Moore. 1996. Involvement of interleukin (IL) 8 receptor in negative regulation of myeloid progenitor cells in vivo: evidence from mice lacking the murine IL-8 receptor homologue. J. Exp. Med. 184:1825-1832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cheung, D. O. Y., K. Halsey, and D. P. Speert. 2000. Role of pulmonary alveolar macrophages in defense of the lung against Pseudomonas aeruginosa. Infect. Immun. 68:4585-4592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chu, K. K., D. J. Davidson, T. K. Halsey, J. W. Chung, and D. P. Speert. 2002. Differential persistence among genomovars of the Burkholderia cepacia complex in a murine model of pulmonary infection. Infect. Immun. 70:2715-2720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Faccioli, L. H., G. E. Souza, F. Q. Cunha, S. Poole, and S. H. Ferreira. 1990. Recombinant interleukin-1 and tumor necrosis factor induce neutrophil migration “in vivo” by indirect mechanisms. Agents Actions 30:344-349. [DOI] [PubMed] [Google Scholar]
- 7.Govan, J. R., and V. Deretic. 1996. Microbial pathogenesis in cystic fibrosis: mucoid Pseudomonas aeruginosa and Burkholderia cepacia. Microbiol. Rev. 60:539-574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Govan, J. R. W. 2000. Infection control in cystic fibrosis: methicillin-resistant Staphylococcus aureus, Pseudomonas aeruginosa and the Burkholderia cepacia complex. J. R. Soc. Med. 93:40-45. [PMC free article] [PubMed] [Google Scholar]
- 9.Hutchison, M. L., I. R. Poxton, and J. R. Govan. 1998. Burkholderia cepacia produces a hemolysin that is capable of inducing apoptosis and degranulation of mammalian phagocytes. Infect. Immun. 66:2033-2039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kolopp-Sarda, M. N., M. C. Bene, N. Massin, J. J. Moulin, and G. C. Faure. 1994. Immunohistological analysis of macrophages, B-cells and T-cells in the mouse lung. Anat. Rec. 239:150-157. [DOI] [PubMed] [Google Scholar]
- 11.Lugton, I. 1999. Mucosa-associated lymphoid tissues as sites for uptake, carriage and excretion of tubercle bacilli and other pathogenic mycobacteria. Immunol. Cell Biol. 77:364-372. [DOI] [PubMed] [Google Scholar]
- 12.Racz, P., K. Tenner-Racz, Q. N. Myrvik, B. T. Shannon, and S. H. Love. 1978. Sinus reactions in the hilar lymph node complex of rabbits undergoing a pulmonary cell-mediated immune response: sinus macrophage clumping reaction, sinus lymphocytosis and immature sinus histiocytosis. J. Reticuloendothel. Soc. 24:499-525. [PubMed] [Google Scholar]
- 13.Sajjan, U. S., F. A. Sylvester, and J. F. Forstner. 2000. Cable-piliated Burkholderia cepacia binds to cytokeratin 13 of epithelial cells. Infect. Immun. 68:1787-1795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Smith, R. J., B. J. Bowman, and S. C. Speziale. 1986. Interleukin-1 stimulates granule exocytosis from human neutrophils. Int. J. Immunopharmacol. 8:33-40. [DOI] [PubMed] [Google Scholar]
- 15.Sokol, P., P. Darling, D. E. Woods, E. Mahenthiralingam, and C. Kooi. 1999. Role of ornibactin biosynthesis in the virulence of Burkholderia cepacia: characterization of pvdA, the gene encoding l-ornithine N5-oxygenase. Infect. Immun. 67:4443-4455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Speert, D. P., D. Henry, P. Vandamme, M. Corey, and E. Mahenthiralingam. 2002. Epidemiology of Burkholderia cepacia complex in patients with cystic fibrosis, Canada. Emerg. Infect. Dis. 8:181-187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Stotland, P. K., D. Radzioch, and M. M. Stevenson. 2000. Mouse models of chronic lung infection with Pseudomonas aeruginosa: models for the study of cystic fibrosis. Pediatr. Pulmonol. 30:413-424. [DOI] [PubMed] [Google Scholar]
- 18.Tomich, M., A. Griffith, C. A. Herfst, J. L. Burns, and C. D. Mohr. 2003. Attenuated virulence of a Burkholderia cepacia type III secretion mutant in a murine model of infection. Infect. Immun. 71:1405-1415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tomich, M., C. A. Herfst, J. W. Golden, and C. D. Mohr. 2002. Role of flagella in host cell invasion by Burkholderia cepacia. Infect. Immun. 70:1799-1806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Walker, D. C., A. R. Behzad, and F. Chu. 1995. Neutrophil migration through preexisting holes in the basal laminae of alveolar capillaries and epithelium during streptococcal pneumonia. Microvasc. Res. 50:397-416. [DOI] [PubMed] [Google Scholar]