Abstract
The aim of the study was to evaluate antibacterial activity of composite materials modified with calcium fluoride against cariogenic bacteria S. mutans and L. acidophilus. One commercially available conventional light-curing composite material containing fluoride ions (F2) and two commercially available flowable light-curing composite materials (Flow Art and X-Flow) modified with 1.5, 2.5, and 5.0 wt% anhydrous calcium fluoride addition were used in the study. Composite material samples were incubated in 0.95% NaCl at 35°C for 3 days; then dilution series of S. mutans and L. acidophilus strains were made from the eluates. Bacteria dilutions were cultivated on media afterwards. Colony-forming unit per 1 mL of solution (CFU/mL) was calculated. Composite materials modified with calcium fluoride highly reduced (p < 0.001) bacteria growth compared to commercially available composite materials containing fluoride compounds. The greatest reduction in bacteria growth was observed for composite materials modified with 1.5% wt. CaF2. All three tested composite materials showed statistically greater antibacterial activity against L. acidophilus than against S. mutans.
1. Introduction
In dentistry nowadays, as well as in any other branch of medicine, constant development in material science is observed. The ideal restorative material would not only perfectly restore hard dental tissues, but also possess antibacterial properties, as the most common reason for filling removal is secondary caries [1, 2]. Secondary caries is caused by bacterial infection due to microleakage, bacteria presence in dentinal tubules, or poor adhesion of composite material to hard dental tissues [3, 4]. All of the above may lead to pulp infection and cause postoperative complications. Antibacterial properties of restorative materials are of major clinical importance and would allow for less invasive hard dental tissue preparation, highly influencing positive treatment outcome.
In order to reduce microorganisms proliferation on the tooth-resin interface and around dental fillings, various chemical compounds were added to the materials composition: fluoride compounds, chlorhexidine digluconate (CHG), chlorhexidine diacetate (CHA), nanoparticles of amorphous calcium phosphate (NACP), quaternary ammonium dimethacrylate (QADM), 12-methacryloyloxydodecylpyridinium bromide (MDBP), methacryloxylethylcetyl dimethyl ammonium chloride (DMAE-CB), cetylpyridinium chloride (CPC), quaternary ammonium polyethylenimine (PEI), 2-dimethyl-2-dodecyl-1-methacryloxyethyl ammonium iodine (DDMAI), and furanone derivatives [5, 6].
Composite materials due to surface roughness [7–9] and residual monomers released after polymerization [10] favor bacterial colonization much more than other dental materials like amalgam, gold alloys, or glass ionomer cements [11–13]. Bacteria present in biofilm also induce further adhesion of microorganism to composite filling [14, 15]. Also, even correctly applied composite restorations with time undergo hydrolytic degradation due to water sorption, swelling, plasticization, or enzymatic decomposition. The latter may be induced by enzymes, present in saliva or bacteria, as well as by endogenous enzymes present in dentine—metalloproteinases—that cause collagen fibres hydrolysis leading to microleakage [16]. All of the above mentioned result in degradation of filling material, lack of marginal adaptation, and bacterial colonization of enamel, dentine, and cementum leading to restoration replacement and further loss of hard dental tissues [7, 17–23].
Cariostatic effect of fluoride ions is widely known and involves reduction of demineralization, promoting of remineralization, and incorporating fluoride ions in enamel structure as fluoroapatites and hydroxyl-fluoroapatites. Fluoride prevents demineralization process and enhances remineralization of enamel. Both processes take place in low concentration of fluoride ions in enamel amounting up to 0.03 ppm [8, 14, 20, 24–27]. Due to higher organic substance amount in dentine, the fluoride amount indispensable to enhance both processes should be 10 times higher than for enamel [28]. Fluoride release from dental composites extensively limits secondary caries progression [29]. Altogether, fluoride ions by enhancing enamel toughness and increasing its solubility in acidic environment [24, 28, 30–38] control caries process [21].
Kulshrestha et al. [29] performed research on the influence of CaF2 nanoparticles (CaF2-NPs) on bacteria, both in vitro and in vivo. The results showed strong antibacterial activity of CaF2-NPs against S. mutans: almost 90% reduction of biofilm formation, reduced bacteria acid, and exopolysaccharides production. In low pH environment, fluoride ions bind hydrogen ions creating hydrofluoric acid that penetrates bacterial membrane. Hydrofluoric acid inside bacteria dissociates and causes acidification of cytoplasm and inhibits enzymes (enolase and ATPase) [29, 39, 40]. Fluorides in very high concentrations (3040–5700 ppm) cause bacteria cell death [24].
Moreover, fluoride also adversely influences metabolism and adhesion of bacteria cells [25, 26, 30, 31, 41]. Microbes showed decrease of adhesion to biofilm and greater sensitivity to acidic environment in presence of calcium fluoride. The in vivo investigation in rats revealed that, after exposure to calcium fluoride nanoparticles, S. mutans adhesion to tooth surface decreased. Additionally, CaF2 nanoparticles restrain biofilm formation and as a consequence they reduce caries lesions development, due to great fluoride ions release and its influence on bacteria.
Various fluoride salts were added to organic matrix of composite materials (NaF, CaF2, KF, SrF2, and SnF2), yet the fluoride ion release decreased substantially with time, while mechanical properties were deteriorated [36, 38]. The headway in dental material science should focus on developing restorative material that would combine antibacterial and regenerative properties towards hard dental tissues as well as possess optimal mechanical parameters [8].
2. Aim of the Study
The purpose was to evaluate antibacterial activity of composite materials Flow Art and X-Flow modified with calcium fluoride against cariogenic bacteria S. mutans and L. acidophilus.
3. Materials and Methods
3.1. Sample Preparation
Two flowable composite materials were used in the study: Flow Art (Arkona, Poland) and X-Flow (Dentsply, Germany) and one conventional composite material F2 (Arkona, Poland).
Flow Art is a light-cured composite that consists of dimetacrylic organic matrix (bisphenol A dimethacrylate, dimethacrylate urethane, and triethylene glycol dimethacrylate) containing inorganic fillers (barium-aluminium-silicate glass, pyrogenic silica) and additions (photoinitiator, coinitiator, inhibitor, stabilizers, and pigments). Mineral fillers constitute up to 60% of the composite.
X-Flow is a flowable, light-curing composite that consists of strontium-alumino-sodium fluoro-phosphor-silicate glass, di- and multifunctional acrylate and methacrylate resins, diethylene glycol dimethacrylate (DGDMA), highly dispersed silicon dioxide, UV stabilizer, ethyl-4-dimethylaminobenzoate, camphorquinone, butylated hydroxyl-toluene (BHT), iron pigments, and titanium dioxide. The filler load was 60% in weight and 38% in volume. Filler size (D 50) was 1.6 micrometers [42].
F2 is conventional light-curing composite containing fluoride ions and Bis-GMA, TEGDMA, UDMA, Bis-EMA, and filler (barium-aluminium-borosilicate glass, fluoro-barium-silicate glass, and pyrogenic silica). The filler load is 77% by weight.
Both flowable composite materials were modified with anhydrate powder of calcium fluoride addition (Arcos Organics, Belgium) and 3 study groups were established as presented in Table 1.
Table 1.
Flow Art and X-Flow study groups.
| Composite material | Group 0 | Group I | Group II | Group III |
|---|---|---|---|---|
| Flow Art (FA) |
Unmodified (0 FA) |
+1.5% wt CaF2 (I FA) | +2.5% wt CaF2 (II FA) | +5% wt CaF2 (III FA) |
| X-Flow (XF) |
Unmodified (0 XF) |
+1.5% wt CaF2 (I XF) | +2.5% wt CaF2 (II XF) | +5% wt CaF2 (III XF) |
F2 composite material, as the one containing fluoride ions, was not modified with CaF2 addition.
The samples were prepared using cylindrical silicone molds of 3 mm height and 5 mm in diameter. Composite material was applied into the mold in layering technique and polymerized with halogen polymerizing lamp at light intensity of 800 mW/cm2 (Megalux Soft-Start/Mega-PHYSIK Gmbh & Co. KG, Germany). The manufacturer's instructions were as follows:
Flow Art: 3 mm material layer was polymerized for 30 s; one layer was applied.
X-Flow: 1.5 mm material layer was polymerized for 20 s; two layers were applied.
F2: 3 mm material layer was polymerized for 30 s; one layer was applied.
For each study group (3 study groups) and bacteria strain (2 bacteria strains) 15 samples were prepared. The total number of samples equals 90 (3 × 2 × 15 = 90). From each sample two experiments were performed.
3.2. Microbank System
Microbiological studies were conducted on two reference strains: Streptococcus mutans ATCC 25175 and Lactobacillus acidophilus ATCC 4356. The strains were stored in Microbank systems (Biocorp, Poland). The method was based on ceramic balls placed in containers (cryotube). The vial was inoculated with 24-hour pure bacteria culture (McFarland standard 0.5). The bacteria were attached to ceramic balls in cryopreservation media. After 15-minute immersion in cryopreservation media, each ball (with bacteria) was covered with media. Vials can be stored in a freezer in temperatures ranging from −20°C to −70°C or in liquid nitrogen. This system is designed to simplify the storage and retrieval of bacterial cultures.
3.3. Serial Dilution of Bacteria Strains
3.3.1. Bacterial Colonies without Composite Sample
The strains from Microbank were revived on proliferating media: Columbia agar (Becton-Dickinson, USA) for S. mutans in 5% CO2-enriched conditions—GENbox CO2 (BioMérieux, France)—and for L. acidophilus in anaerobic conditions—GENbox anaer (BioMérieux, France). After 18-hour cultivation, bacterial emulsion in McFarland standard 0.5 was prepared. Serial dilutions of bacterial emulsion were made: 0.5 mL of dilution was added to 4.5 mL 0.9% NaCl resulting in 10 times diluted bacteria emulsion (10−1). Then dilutions were prepared by transferring 0.5 mL solution to the subsequent tubes. Next, 100 μL of dilution was cultured on the growth medium and incubated for 24 h in 35°C. This procedure allowed us to calculate the number of bacterial colonies for given dilutions of bacterial strait without the influence of composite on bacteria.
3.3.2. Bacterial Colonies with Composite Sample
The prepared samples of the composite material of Groups 0, I, II, and III were placed in 2.5 mL of 0.95% NaCl and incubated at 35°C for 3 days. Then, the material was removed from the eluate and serial dilutions of the bacteria strains were prepared. Into 1.8 mL of eluate, 200 μL of the strain was introduced obtaining dilutions, in which fluoride ions released from composite materials were found effective against tested bacteria. Serial dilutions prepared for S. mutans and L. acidophilus, for both Flow Art and X-Flow composite materials, are presented in Tables 2 and 3.
Table 2.
Serial dilutions for S. mutans.
| Control group | Group I | Group II | Group III |
|---|---|---|---|
| 10−3–10−6 | 10−2–10−5 | 10−2–10−5 | 10−2–10−5 |
Table 3.
Serial dilutions for L. acidophilus.
| Control group | Group I | Group II | Group III |
|---|---|---|---|
| 10−1–10−4 | 10−0–10−3 | 10−0–10−3 | 10−0–10−3 |
In case of F2 composite material, the quantifiable bacteria colonies appeared in different dilutions. Serial dilutions are presented in Table 4.
Table 4.
Serial dilutions for F2 composite material.
| S. mutans | L. acidophilus |
|---|---|
| 10−3–10−5 | 10−1–10−4 |
3.4. Bacteria Incubation
Bacteria strains were incubated for 24 hours in eluates. S. mutans and L. acidophilus bacteria emulsion in saline incubated in the same conditions used in the control group. Next, the control and 100 μL of bacteria dilution in eluate were cultivated on media to establish bacteria susceptibility: S. mutans on MH agar (Becton-Dickinson, USA) and L. acidophilus on composite media (90% Iso-Sensitest Agar + 10% Rogosa Agar) (OXOID, UK). The strains were incubated for 24 hours at 35°C. Next, bacterial colonies in the studied samples and the control groups were counted by calculating the number of bacteria per 1 mL of solution, according to the following formula:
| (1) |
3.5. Statistical Analysis
Statistical analysis comprised the Shapiro-Wilk W tests for normality, Levene's test for the homogeneity of variances, and the Mann–Whitney U test (due to large dispersion of the data and heterogeneity of variances). Furthermore, in order to test the differences depending on the species of bacteria, type of composite material, the percentage concentration of calcium fluoride, and the specific distribution of CFU, zero-inflated Poisson regression with robust standard errors was used. A level of p < 0.05 was considered statistically significant. All statistical procedures were performed with the use of Stata®/Special Edition, release 14.1 (StataCorp LP, College Station, Texas, USA).
4. Results
After microbiological studies, bacteria cell number was counted in 1 mL of solution (CFU/1 mL). Distribution of bacteria cells for tested composite materials and bacteria were presented in Figures 1 –3.
Figure 1.
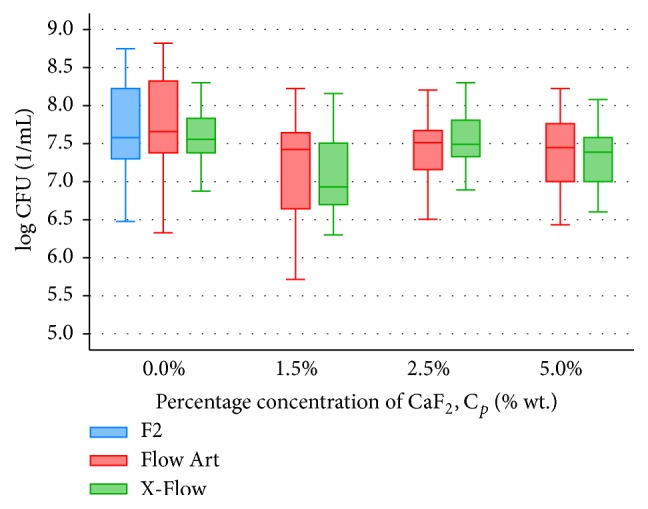
Bacteria S. mutans distribution in 1 mL of solution (CFU/1 mL), where composite material samples (F2, Flow Art, and X-Flow) were stored (n = 30).
Figure 2.
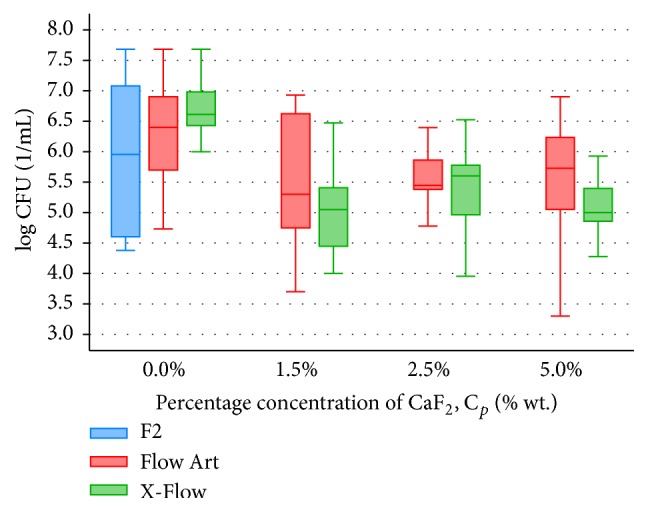
Bacteria L. acidophilus distribution in 1 mL of solution (CFU/1 mL), where composite material samples (F2, Flow Art, and X-Flow) were stored (n = 30).
Figure 3.
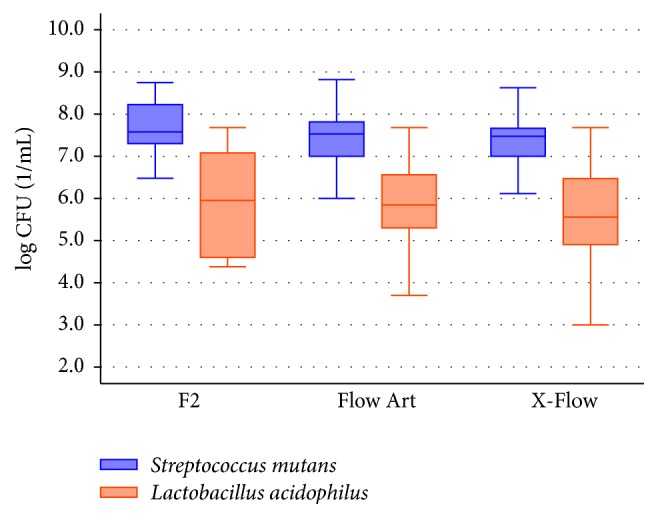
Comparison of bacteria S. mutans and L. acidophilus susceptibility/sensitivity to the composite materials, both modified and unmodified (F2: n = 60; Flow Art: n = 240; and X-Flow: n = 240).
The significantly highest (p = 0.025) antibacterial activity against S. mutans, among all tested commercially available composite materials (F2, Flow Art, and X-Flow), was shown in X-Flow (Me CFU: 3.60∗107/mL), while the lowest was in Flow Art (Me CFU: 4.20∗107/mL) (Figure 1).
Composite materials Flow Art and X-Flow modified with calcium fluoride showed comparable antibacterial activity against S. mutans; any differences were statistically insignificant (p = 0.396) (Figure 1). The highest antibacterial activity against S. mutans, for both modified composites, was observed after addition of 1.5 wt% CaF2 (Figure 1). Increasing the amount of CaF2 (over 1.5 wt%) did not influence antibacterial activity of both tested materials.
Antibacterial properties against L. acidophilus of three tested materials (unmodified, control groups) were comparable and statistically insignificant (p = 0.980) (Figure 2).
When comparing antibacterial activity of fluoride-based composite materials, Flow Art and X-Flow, against L. acidophilus, the statistically highest (p < 0.001) activity presented X-Flow containing 1.5 wt% CaF2 (Me CFU: 9∗104/mL) (Figure 2). Increasing the amount of CaF2 (over 1.5 wt%) did not influence antibacterial activity of both tested materials.
All three tested composite materials (control and study groups altogether) show statistically greater antibacterial activity against L. acidophilus than against S. mutans (F2, p < 0.001; Flow Art, p < 0.001; and X-Flow, p < 0.001) (Figure 3).
5. Discussion
Secondary caries emerges on the filling-hard dental tissues interface. The bacteria used in the study, S. mutans and L. acidophilus, are the mainly involved in the carious process. The former one is responsible for the initiation of the process and the latter for its development [3, 7, 21, 29, 43–45].
According to many researches, 55 to 70% of fillings need replacement due to secondary caries, making it the most common reason for application of new restoration [3, 4, 10, 21, 46, 47]. The present study showed that conventional composites demonstrate little antibacterial activity against S. mutans and L. acidophilus, adversely affecting treatment outcome. Similar conclusions may be drawn based on scientific paper reviews [2, 8, 10, 48].
Bernardo et al. [2] assessed that secondary caries almost 3 times more often concerns composite material fillings than amalgams. Imazato [12] noted the increased risk of gingivitis when subgingival lesions are filled with composite materials due to their low antibacterial activity and substantial plaque deposition on their surface comparing to other restorative materials. Beyth et al. [8] and Nedeljkovic et al. [10] stated that composite materials are widely used restorative materials for their excellent esthetics and mechanical properties, but the issue of increased occurrence of the secondary caries remains unsolved. Many researchers [2, 4, 10, 48] suggest the need for development of antibacterial properties of composite materials.
For the present study, X-Flow composite material was chosen because it is very popular material with good clinical performance. While Flow Art and F2 composites exhibit also good mechanical characteristics and have similar composition, the latter contains relatively high amount of fluoride glass. Both composite materials without fluoride compounds (Flow Art) and with low fluoride content (X-Flow contains around 1% of F−) showed little antibacterial activity. Comparison of two unmodified materials Flow Art and X-Flow and F2 composite material revealed statistically higher antibacterial activity against S. mutans for X-Flow and the lowest for Flow Art. The F2 composite material that, according to manufacturer, contained fluoride glass (fluoride content is 5–7%) acts weaker than the material with no fluoride ions in composition (X-Flow). Antibacterial activities of unmodified composite materials X-Flow, Flow Art, and F2 against L. acidophilus were also evaluated. No statistical differences have been shown between tested materials, though all unmodified materials (X-Flow, Flow Art, and F2) presented significantly higher antibacterial activity against L. acidophilus than against S. mutans.
A number of researches [14, 26, 32, 38, 39, 49–52] proved that the highest amount of fluoride ions is released as follows: glass ionomer cements, resin modified glass ionomer cements, compomers, and the least composite materials. Authors suggest that modification of dental materials with CaF2-based compound could be major issue that limits dental caries development.
Calcium fluoride was used by Xu et al. [53], who incorporated CaF2 nanoparticles into polymer matrix of composite material. The CaF2 nanoparticles content was at 20%, at general content of the filler 65%. The amount of fluoride ions released from the material was approximately the same or higher than glass ionomer cements for about 2 months. Moreover, the material released phosphate and calcium ions that induced fluoroapatites formation and reduces secondary caries occurrence. The authors explained the result of their study with the size of calcium fluoride particles. Addition of nanoparticles of 1 μm and smaller resulted in the substantial increase (20 times greater) in surface area of modified composite material compared to conventional composite material modified with calcium fluoride powder [26].
Galvan et al. [54] observed that releasing fluoride ions dental materials showed the highest activity on the first day after polymerization, followed by dropping for the next 90 days of the experiment remaining minimum level of F− ion release. It is expected that such materials would serve as fluoride reservoirs and will remain cariostatic.
The results of the study indicated improvement in antibacterial properties of modified composite materials. Both materials, Flow Art and X-Flow, showed the highest activity when 1.5 wt% CaF2 was added. Comparison of CFU median for control groups of Flow Art and X-Flow with CFU median for Groups I FA and I XF against S. mutans showed significant improvement in antibacterial activities in groups modified with CaF2. Similar results were obtained for L. acidophilus; increasing CaF2 over 1.5 wt% did not decrease statistically CFU/mL number, remaining the same antibacterial activity. Groups modified with CaF2 possess higher antibacterial activity than F2 material containing fluoride glass.
In the present study, in order to enhance antibacterial activity of composite materials, calcium fluoride was introduced to their composition and microbiological study was performed afterwards. Most research focus on the influence of fluoride ions on S. mutans. Given proved cariogenic activity of S. mutans as well as Lactobacillus spp., both of those bacteria have been tested.
Statistical analysis revealed that all tested composite materials, both modified and unmodified, had statistically significant greater antibacterial activity against L. acidophilus than against S. mutans (p < 0.001). Naorungroj et al. [30] also compared sensitivity of S. mutans and L. acidophilus to composite materials releasing fluoride ions, but the results of their study were vague. One material showed the same level of antibacterial activity against both bacteria, while for the other two materials the activity varied. The differences in results can be explained by the differentiated resin material composition, which is not given by manufacturers in detail. Moreover, those ingredients may exhibit divers influence on bacterial cells. Naorungroj et al. [30] used enamel discs and agar diffusion method, while the present study measured influence of composite eluates on bacterial growth.
6. Conclusions
Within the limitations of the study the following conclusions can be made:
Commercially available composite materials limit bacteria growth, even higher than composite material with increased fluoride content (F2).
Composite materials modified with calcium fluoride reduced bacteria growth stronger than commercially available composite materials containing fluoride compounds. The greatest reduction in bacteria growth was observed for composite materials modified with 1.5% wt. CaF2.
All tested composite materials exhibit statistically greater activity against L. acidophilus than S. mutans.
Competing Interests
The authors declare that they have no competing interests.
References
- 1.Abt E. The risk of failure is higher for composites than for amalgam restorations. Journal of Evidence-Based Dental Practice. 2008;8(2):83–84. doi: 10.1016/j.jebdp.2008.03.007. [DOI] [PubMed] [Google Scholar]
- 2.Bernardo M., Luis H., Martin M. D., et al. Survival and reasons for failure of amalgam versus composite posterior restorations placed in a randomized clinical trial. The Journal of the American Dental Association. 2007;138(6):775–783. doi: 10.14219/jada.archive.2007.0265. [DOI] [PubMed] [Google Scholar]
- 3.Jokstad A. Secondary caries and microleakage. Dental Materials. 2016;32(1):11–25. doi: 10.1016/j.dental.2015.09.006. [DOI] [PubMed] [Google Scholar]
- 4.Mjor I. A. Secondary caries: a literature review with case reports. Quintessence International. 2003;31:165–179. [PubMed] [Google Scholar]
- 5.Szram A., Póltorak K., Podlewska M., Sokołowski J., Łukomska-Szymańska M. Selected antibacterial components in dental materials—literature review. Protetyka Stomatologiczna. 2015;65(5):476–481. [Google Scholar]
- 6.Półtorak K., Podlewska M., Szram A., Sokołowski J., Łukomska-Szymańska M. Composite materials with antimicrobal properties-literature review. e-Dentico. 2016;60(2):84–89. [Google Scholar]
- 7.Mehdawi I. M., Young A. Antibacterial Composite Restorative Materials for Dental Applications. Sawston, UK: Woodhead Publishing Limited; 2012. [Google Scholar]
- 8.Beyth N., Farah S., Domb A. J., Weiss E. I. Antibacterial dental resin composites. Reactive and Functional Polymers. 2014;75(1):81–88. doi: 10.1016/j.reactfunctpolym.2013.11.011. [DOI] [Google Scholar]
- 9.Chau N. P. T., Pandit S., Jung J.-E., Jeon J.-G. Evaluation of Streptococcus mutans adhesion to fluoride varnishes and subsequent change in biofilm accumulation and acidogenicity. Journal of Dentistry. 2014;42(6):726–734. doi: 10.1016/j.jdent.2014.03.009. [DOI] [PubMed] [Google Scholar]
- 10.Nedeljkovic I., Teughels W., De Munck J., Van Meerbeek B., Van Landuyt K. L. Is secondary caries with composites a material-based problem? Dental Materials. 2015;31(11):e247–e277. doi: 10.1016/j.dental.2015.09.001. [DOI] [PubMed] [Google Scholar]
- 11.Bojar W., Kobylecki W., Tyski S., Zareba T., Kobyłecki W. Porównanie właściwości przeciwbakteryjnych wybranych materiałów do wypełnień. Nowa Stomatologia. 2000;1-2:13–16. [Google Scholar]
- 12.Imazato S. Antibacterial properties of resin composites and dentin bonding systems. Dental Materials. 2003;19(6):449–457. doi: 10.1016/S0109-5641(02)00102-1. [DOI] [PubMed] [Google Scholar]
- 13.Farrugia C., Camilleri J. Antimicrobial properties of conventional restorative filling materials and advances in antimicrobial properties of composite resins and glass ionomer cements—a literature review. Dental Materials. 2015;31(4):e89–e99. doi: 10.1016/j.dental.2014.12.005. [DOI] [PubMed] [Google Scholar]
- 14.Sungurtekin-Ekci E., Ozdemir-Ozenen D., Duman S., Acuner I. C., Sandalli N. Antibacterial surface properties of various fluoride-releasing restorative materials in vitro. Journal of Applied Biomaterials and Functional Materials. 2015;13(2):e169–e173. doi: 10.5301/jabfm.5000212. [DOI] [PubMed] [Google Scholar]
- 15.Yamamoto K., Ohashi S., Taki E., Hirata K. Adherence of oral streptococci to composite resin of varying surface roughness. Dental Materials Journal. 1996;15(2):201–204. doi: 10.4012/dmj.15.201. [DOI] [PubMed] [Google Scholar]
- 16.Moon P. C., Weaver J., Brooks C. N. Review of matrix metalloproteinases' effect on the hybrid dentin bond layer stability and chlorhexidine clinical use to prevent bond failure. The Open Dentistry Journal. 2010;4(1):147–152. doi: 10.2174/1874210601004010147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Craig R., Sakaguchi R., Powers J. Craig's Restorative Dental Materials. 13th. Philadelphia, Pa, USA: Elsevier; 2012. [Google Scholar]
- 18.Sadowsky S. J. An overview of treatment considerations for esthetic restorations: a review of the literature. Journal of Prosthetic Dentistry. 2006;96(6):433–442. doi: 10.1016/j.prosdent.2006.09.018. [DOI] [PubMed] [Google Scholar]
- 19.Lin B., Jaffer F., Duff M., Tang Y., Santerre J. Identifying enzyme activities within human saliva which are relevant to dental resin composite biodegradation. Biomaterials. 2005;26(20):4259–4264. doi: 10.1016/j.biomaterials.2004.11.001. [DOI] [PubMed] [Google Scholar]
- 20.Garcia A. H., Martínez-Lozano M. A., Cabanes-Vila J., Barjau-Escribano A., Fos-Galve P. Composite resins. A review of the materials and clinical indications. Medicina Oral, Patología Oral y Cirugía Bucal. 2006;11(2):E215–E220. [PubMed] [Google Scholar]
- 21.Li Y., Carrera C., Chen R., et al. Degradation in the dentin-composite interface subjected to multi-species biofilm challenges. Acta Biomaterialia. 2014;10(1):375–383. doi: 10.1016/j.actbio.2013.08.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cury J. A., de Oliveira B. H., dos Santos A. P. P., Tenuta L. M. A. Are fluoride releasing dental materials clinically effective on caries control? Dental Materials. 2016;23(3):323–333. doi: 10.1016/j.dental.2015.12.002. [DOI] [PubMed] [Google Scholar]
- 23.Kosior P. Uwalnianie Jonów Fluorkowych Z Wybranych Materiałów Stomatologicznych w Warunkach Doświadczalnych I Klinicznych. Wrocław, Poland: Rozpr. Doktorska; 2005. [Google Scholar]
- 24.Whitford G. M. Matabolism and toxicity of fluoride. Monographs in Oral Science. 1996;16:1–153. doi: 10.1159/000425104. [DOI] [PubMed] [Google Scholar]
- 25.Browne D., Whelton H., O'Mullane D. Fluoride metabolism and fluorosis. Journal of Dentistry. 2005;33(3):177–186. doi: 10.1016/j.jdent.2004.10.003. [DOI] [PubMed] [Google Scholar]
- 26.Xu H. H. K., Weir M. D., Sun L., et al. Strong nanocomposites with Ca, PO4, and F release for caries inhibition. Journal of Dental Research. 2010;89(1):19–28. doi: 10.1177/0022034509351969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hashimoto M., Nakamura K., Kaga M., Yawaka Y. Crystal growth by fluoridated adhesive resins. Dental Materials. 2008;24(4):457–463. doi: 10.1016/j.dental.2007.04.014. [DOI] [PubMed] [Google Scholar]
- 28.Kaczmarek U. Mechanizmy kariostatyczne fluoru. Czasopismo Stomatologiczne. 2005;58(6):404–413. [Google Scholar]
- 29.Kulshrestha S., Khan S., Hasan S., Khan M. E., Misba L., Khan A. U. Calcium fluoride nanoparticles induced suppression of Streptococcus mutans biofilm: an in vitro and in vivo approach. Applied Microbiology and Biotechnology. 2016;100(4):1901–1914. doi: 10.1007/s00253-015-7154-4. [DOI] [PubMed] [Google Scholar]
- 30.Naorungroj S., Wei H.-H., Arnold R. R., Swift E. J., Jr., Walter R. Antibacterial surface properties of fluoride-containing resin-based sealants. Journal of Dentistry. 2010;38(5):387–391. doi: 10.1016/j.jdent.2010.01.005. [DOI] [PubMed] [Google Scholar]
- 31.Li X., Wang J., Joiner A., Chang J. The remineralisation of enamel: a review of the literature. Journal of Dentistry. 2014;42(supplement 1):S12–S20. doi: 10.1016/s0300-5712(14)50003-6. [DOI] [PubMed] [Google Scholar]
- 32.Sikorska-Jaroszyńska M. H., Czelej G. Fluor w Stomatologii i Medycynie. Lublin, Poland: Wydawnictwo Czelej; 2000. [Google Scholar]
- 33.Burbank B. D., Slater M., Kava A., et al. Ion release, fluoride charge of and adhesion of an orthodontic cement paste containing microcapsules. Journal of Dentistry. 2015;45:32–38. doi: 10.1016/j.jdent.2015.11.009. [DOI] [PubMed] [Google Scholar]
- 34.Fejerskov O., Ekstrand J., Burt B. A. Fluoride in Dentistry. Copenhagen, Denmark: Munksgaard; 1996. [Google Scholar]
- 35.Davis H. B., Gwinner F., Mitchell J. C., Ferracane J. L. Ion release from, and fluoride recharge of a composite with a fluoride-containing bioactive glass. Dental Materials. 2014;30(10):1187–1194. doi: 10.1016/j.dental.2014.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Furtos G., Cosma V., Prejmerean C., et al. Fluoride release from dental resin composites. Materials Science and Engineering: C. 2005;25(2):231–236. doi: 10.1016/j.msec.2005.01.016. [DOI] [Google Scholar]
- 37.Szczepańska J. Contemporary view on the fluoride prophylaxis—some aspects of cariostatic fluoride action. Nowa Stomatologia. 2003;8(24):80–83. [Google Scholar]
- 38.Moreau J. L., Xu H. H. K. Fluoride releasing restorative materials: effects of pH on mechanical properties and ion release. Dental Materials. 2010;26(11):e227–e235. doi: 10.1016/j.dental.2010.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Wiegand A., Buchalla W., Attin T. Review on fluoride-releasing restorative materials—fluoride release and uptake characteristics, antibacterial activity and influence on caries formation. Dental Materials. 2007;23(3):343–362. doi: 10.1016/j.dental.2006.01.022. [DOI] [PubMed] [Google Scholar]
- 40.Wang Z., Shen Y., Haapasalo M. Dental materials with antibiofilm properties. Dental Materials. 2014;30(2):e1–e16. doi: 10.1016/j.dental.2013.12.001. [DOI] [PubMed] [Google Scholar]
- 41.Błaszczyk I., Ratajczak-Kubiak E., Birkner E. Korzystne i szkodliwe działanie fluoru. Farmacja Polska. 2009;18(9):623–626. [Google Scholar]
- 42. http://www.dentsply.eu/bausteine.net/f/7293/SCXflow20030616.pdf?fd=2.
- 43.Strużycka I. Biofilm—współczesne spojrzenie na etiologię próchnicy. Dental Forum. 2010;1(38):73–79. [Google Scholar]
- 44.Kołwzan B. Analiza zjawiska biofilmu—warunki jego powstawania i funkcjonowania. Ochrona Środowiska. 2011;33:3–14. [Google Scholar]
- 45.Cummins D. The development and validation of a new technology, based upon 1.5% arginine, an insoluble calcium compound and fluoride, for everyday use in the prevention and treatment of dental caries. Journal of Dentistry. 2013;41(supplement 2):S1–S11. doi: 10.1016/j.jdent.2010.04.002. [DOI] [PubMed] [Google Scholar]
- 46.Wang Y., Samoei G. K., Lallier T. E., Xu X. Synthesis and characterization of new antibacterial fluoride-releasing monomer and dental composite. ACS Macro Letters. 2013;2(1):59–62. doi: 10.1021/mz300579y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Cheng L., Weir M. D., Xu H. H. K., et al. Antibacterial and physical properties of calcium-phosphate and calcium-fluoride nanocomposites with chlorhexidine. Dental Materials. 2012;28(5):573–583. doi: 10.1016/j.dental.2012.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Imazato S. Bio-active restorative materials with antibacterial effects: new dimension of innovation in restorative dentistry. Dental Materials Journal. 2009;28(1):11–19. doi: 10.4012/dmj.28.11. [DOI] [PubMed] [Google Scholar]
- 49.Patil S. A., Goud K. M., Sajjan G. S. Fluoride release from various restorative materials over a 3 month period—an in vitro study. Journal of Pierre Fauchard Academy. 2012;26(1):30–34. [Google Scholar]
- 50.Pereira P. N. R., Inokoshi S., Tagami J. In vitro secondary caries inhibition around fluoride releasing materials. Journal of Dentistry. 1998;26(5-6):505–510. doi: 10.1016/s0300-5712(98)00008-6. [DOI] [PubMed] [Google Scholar]
- 51.Hicks J., Garcia-Godoy F., Donly K., Flaitz C. Fluoride-releasing restorative materials and secondary caries. Journal of the California Dental Association. 2003;31(3):229–245. [PubMed] [Google Scholar]
- 52.Hotwani K., Thosar N., Baliga S., Bundale S., Sharma K. Antibacterial effects of hybrid tooth colored restorative materials against Streptococcus mutans: an in vitro analysis. Journal of Conservative Dentistry. 2013;16(4):319–322. doi: 10.4103/0972-0707.114358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Xu H. H. K., Moreau J. L., Sun L., Chow L. C. Strength and fluoride release characteristics of a calcium fluoride based dental nanocomposite. Biomaterials. 2008;29(32):4261–4267. doi: 10.1016/j.biomaterials.2008.07.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Galvan R. I., Robertello F. J., Lynde T. A. In vitro comparison of fluoride release of six direct core materials. The Journal of Prosthetic Dentistry. 2000;83(6):629–633. doi: 10.1067/mpr.2000.107192. [DOI] [PubMed] [Google Scholar]


