Abstract
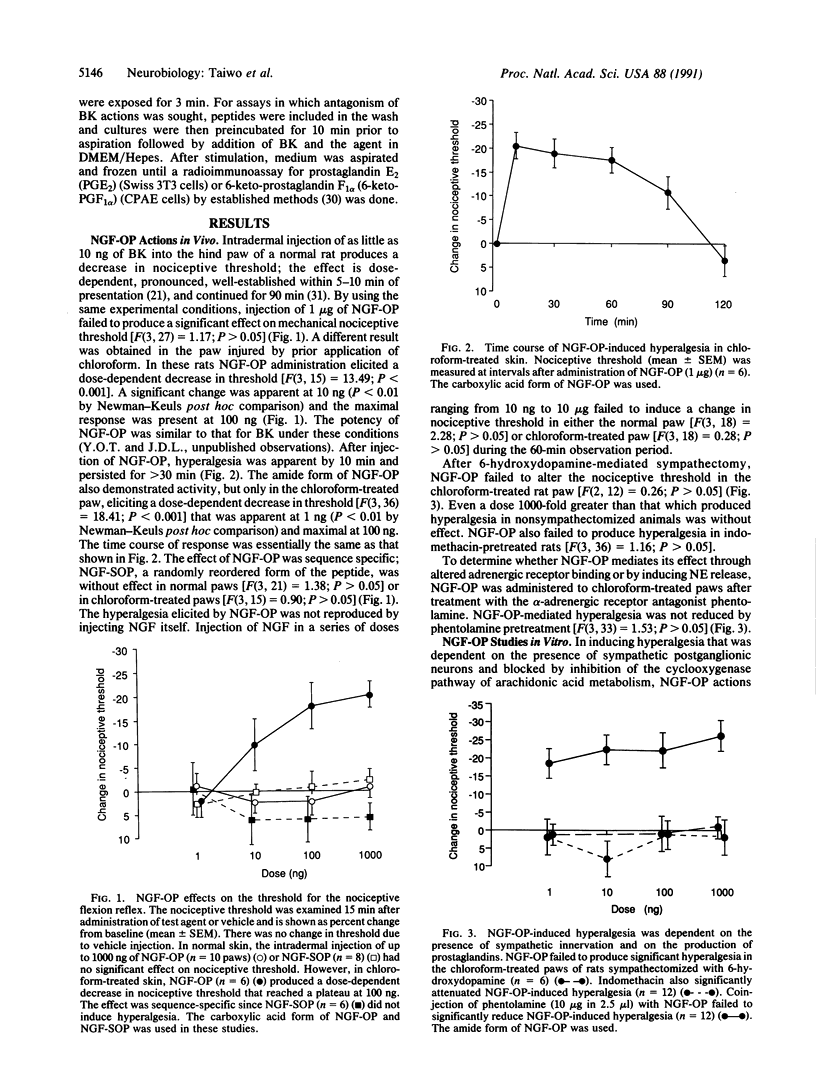
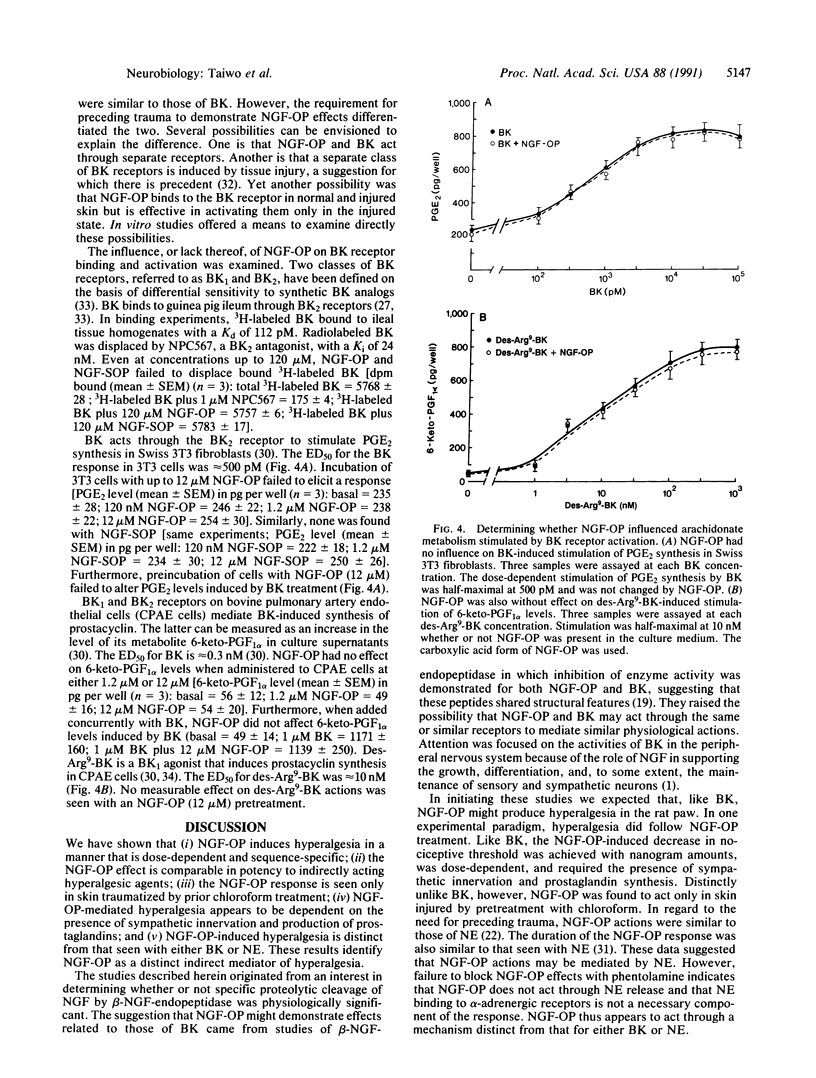
Nerve growth factor (NGF) in the mouse submandibular gland undergoes cleavage of its amino-terminal octapeptide when salivation is induced by epinephrine. The significance of this event is uncertain; cleaved NGF demonstrates bioactivity and no function has been attributed to the octapeptide produced (NGF-OP; Ser-Ser-Thr-His-Pro-Val-Phe-His). Enzyme inhibition studies indicating structural relatedness of NGF-OP and bradykinin (BK) prompted us to determine whether NGF-OP would elicit BK-like actions. We found that like BK, NGF-OP induced a decrease in mechanical nociceptive threshold (i.e., produced hyperalgesia) in the hairy skin of the rat. This effect was dose-dependent and sequence-specific; like BK it was attenuated by sympathectomy and indomethacin pretreatment. However, NGF-OP actions appeared to be distinct from those for BK in that tissue injury was required for NGF-OP to induce hyperalgesia. Furthermore, we found no evidence that NGF-OP bound to or activated BK receptors. Our data indicate that NGF-OP is a distinct mediator of hyperalgesia. We suggest that NGF-OP alters pain threshold in the injured target regions of NGF-responsive neurons.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aloe L., Alleva E., Böhm A., Levi-Montalcini R. Aggressive behavior induces release of nerve growth factor from mouse salivary gland into the bloodstream. Proc Natl Acad Sci U S A. 1986 Aug;83(16):6184–6187. doi: 10.1073/pnas.83.16.6184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barde Y. A. Trophic factors and neuronal survival. Neuron. 1989 Jun;2(6):1525–1534. doi: 10.1016/0896-6273(89)90040-8. [DOI] [PubMed] [Google Scholar]
- Bjerre B., Björklund A., Mobley W. A stimulatory effect by nerve growth factor on the regrowth of adrenergic nerve fibres in the mouse peripheral tissues after chemical sympathectomy with 6-hydroxydopamine. Z Zellforsch Mikrosk Anat. 1973 Dec 31;146(1):15–43. doi: 10.1007/BF00306757. [DOI] [PubMed] [Google Scholar]
- Bjerre B., Björklund A., Mobley W., Rosengren E. Short- and long-term effects of nerve growth factor on the sympathetic nervous system in the adult mouse. Brain Res. 1975 Aug 29;94(2):263–277. doi: 10.1016/0006-8993(75)90061-x. [DOI] [PubMed] [Google Scholar]
- Bothwell M. A., Wilson W. H., Shooter E. M. The relationship between glandular kallikrein and growth factor-processing proteases of mouse submaxillary gland. J Biol Chem. 1979 Aug 10;254(15):7287–7294. [PubMed] [Google Scholar]
- Burton L. E., Wilson W. H., Shooter E. M. Nerve growth factor in mouse saliva. Rapid isolation procedures for and characterization of 7 S nerve growth factor. J Biol Chem. 1978 Nov 10;253(21):7807–7812. [PubMed] [Google Scholar]
- Conklin B. R., Burch R. M., Steranka L. R., Axelrod J. Distinct bradykinin receptors mediate stimulation of prostaglandin synthesis by endothelial cells and fibroblasts. J Pharmacol Exp Ther. 1988 Feb;244(2):646–649. [PubMed] [Google Scholar]
- Farmer S. G., Burch R. M., Dehaas C. J., Togo J., Steranka L. R. [Arg1-D-Phe7]-substituted bradykinin analogs inhibit bradykinin- and vasopressin-induced contractions of uterine smooth muscle. J Pharmacol Exp Ther. 1989 Feb;248(2):677–681. [PubMed] [Google Scholar]
- Finch L., Haeusler G., Thoenen H. A comparison of the effects of chemical sympathectomy by 6-hydroxydopamine in newborn and adult rats. Br J Pharmacol. 1973 Feb;47(2):249–260. doi: 10.1111/j.1476-5381.1973.tb08322.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fitzgerald M., Wall P. D., Goedert M., Emson P. C. Nerve growth factor counteracts the neurophysiological and neurochemical effects of chronic sciatic nerve section. Brain Res. 1985 Apr 15;332(1):131–141. doi: 10.1016/0006-8993(85)90396-8. [DOI] [PubMed] [Google Scholar]
- Heumann R., Korsching S., Bandtlow C., Thoenen H. Changes of nerve growth factor synthesis in nonneuronal cells in response to sciatic nerve transection. J Cell Biol. 1987 Jun;104(6):1623–1631. doi: 10.1083/jcb.104.6.1623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heumann R., Lindholm D., Bandtlow C., Meyer M., Radeke M. J., Misko T. P., Shooter E., Thoenen H. Differential regulation of mRNA encoding nerve growth factor and its receptor in rat sciatic nerve during development, degeneration, and regeneration: role of macrophages. Proc Natl Acad Sci U S A. 1987 Dec;84(23):8735–8739. doi: 10.1073/pnas.84.23.8735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heumann R. Regulation of the synthesis of nerve growth factor. J Exp Biol. 1987 Sep;132:133–150. doi: 10.1242/jeb.132.1.133. [DOI] [PubMed] [Google Scholar]
- Hogue-Angeletti R., Bradshaw R. A., Marshall G. R. The synthesis and characterization of the amino-terminal octapeptide of mouse nerve growth factor. Int J Pept Protein Res. 1974;6(5):321–328. [PubMed] [Google Scholar]
- Ishii D. N., Shooter E. M. Regulation of nerve growth factor synthesis in mouse submaxillary glands by testosterone. J Neurochem. 1975 Dec;25(6):843–851. doi: 10.1111/j.1471-4159.1975.tb04416.x. [DOI] [PubMed] [Google Scholar]
- Korsching S., Thoenen H. Developmental changes of nerve growth factor levels in sympathetic ganglia and their target organs. Dev Biol. 1988 Mar;126(1):40–46. doi: 10.1016/0012-1606(88)90236-9. [DOI] [PubMed] [Google Scholar]
- Korsching S., Thoenen H. Nerve growth factor in sympathetic ganglia and corresponding target organs of the rat: correlation with density of sympathetic innervation. Proc Natl Acad Sci U S A. 1983 Jun;80(11):3513–3516. doi: 10.1073/pnas.80.11.3513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lembeck F., Popper H., Juan H. Release of prostaglandins by bradykinin as an intrinsic mechanism of its algesic effect. Naunyn Schmiedebergs Arch Pharmacol. 1976 Jul;294(1):69–73. doi: 10.1007/BF00692786. [DOI] [PubMed] [Google Scholar]
- Levi-Montalcini R. The nerve growth factor 35 years later. Science. 1987 Sep 4;237(4819):1154–1162. doi: 10.1126/science.3306916. [DOI] [PubMed] [Google Scholar]
- Levine J. D., Taiwo Y. O., Collins S. D., Tam J. K. Noradrenaline hyperalgesia is mediated through interaction with sympathetic postganglionic neurone terminals rather than activation of primary afferent nociceptors. Nature. 1986 Sep 11;323(6084):158–160. doi: 10.1038/323158a0. [DOI] [PubMed] [Google Scholar]
- Lindholm D., Heumann R., Meyer M., Thoenen H. Interleukin-1 regulates synthesis of nerve growth factor in non-neuronal cells of rat sciatic nerve. Nature. 1987 Dec 17;330(6149):658–659. doi: 10.1038/330658a0. [DOI] [PubMed] [Google Scholar]
- Marceau F., Lussier A., Regoli D., Giroud J. P. Pharmacology of kinins: their relevance to tissue injury and inflammation. Gen Pharmacol. 1983;14(2):209–229. doi: 10.1016/0306-3623(83)90001-0. [DOI] [PubMed] [Google Scholar]
- McPherson G. A. Analysis of radioligand binding experiments. A collection of computer programs for the IBM PC. J Pharmacol Methods. 1985 Nov;14(3):213–228. doi: 10.1016/0160-5402(85)90034-8. [DOI] [PubMed] [Google Scholar]
- Meier R., Becker-André M., Götz R., Heumann R., Shaw A., Thoenen H. Molecular cloning of bovine and chick nerve growth factor (NGF): delineation of conserved and unconserved domains and their relationship to the biological activity and antigenicity of NGF. EMBO J. 1986 Jul;5(7):1489–1493. doi: 10.1002/j.1460-2075.1986.tb04387.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merrifield B. Solid phase synthesis. Science. 1986 Apr 18;232(4748):341–347. doi: 10.1126/science.3961484. [DOI] [PubMed] [Google Scholar]
- Mobley W. C., Rutkowski J. L., Tennekoon G. I., Gemski J., Buchanan K., Johnston M. V. Nerve growth factor increases choline acetyltransferase activity in developing basal forebrain neurons. Brain Res. 1986 Jul;387(1):53–62. doi: 10.1016/0169-328x(86)90020-3. [DOI] [PubMed] [Google Scholar]
- Mobley W. C., Schenker A., Shooter E. M. Characterization and isolation of proteolytically modified nerve growth factor. Biochemistry. 1976 Dec 14;15(25):5543–5552. doi: 10.1021/bi00670a019. [DOI] [PubMed] [Google Scholar]
- Munson P. J., Rodbard D. Ligand: a versatile computerized approach for characterization of ligand-binding systems. Anal Biochem. 1980 Sep 1;107(1):220–239. doi: 10.1016/0003-2697(80)90515-1. [DOI] [PubMed] [Google Scholar]
- Murphy R. A., Saide J. D., Blanchard M. H., Young M. Nerve growth factor in mouse serum and saliva: role of the submandibular gland. Proc Natl Acad Sci U S A. 1977 Jun;74(6):2330–2333. doi: 10.1073/pnas.74.6.2330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Regoli D., Barabé J. Pharmacology of bradykinin and related kinins. Pharmacol Rev. 1980 Mar;32(1):1–46. [PubMed] [Google Scholar]
- Rich K. M., Luszczynski J. R., Osborne P. A., Johnson E. M., Jr Nerve growth factor protects adult sensory neurons from cell death and atrophy caused by nerve injury. J Neurocytol. 1987 Apr;16(2):261–268. doi: 10.1007/BF01795309. [DOI] [PubMed] [Google Scholar]
- Sung C. P., Arleth A. J., Shikano K., Berkowitz B. A. Characterization and function of bradykinin receptors in vascular endothelial cells. J Pharmacol Exp Ther. 1988 Oct;247(1):8–13. [PubMed] [Google Scholar]
- Taiwo Y. O., Goetzl E. J., Levine J. D. Hyperalgesia onset latency suggests a hierarchy of action. Brain Res. 1987 Oct 13;423(1-2):333–337. doi: 10.1016/0006-8993(87)90858-4. [DOI] [PubMed] [Google Scholar]
- Taiwo Y. O., Levine J. D. Characterization of the arachidonic acid metabolites mediating bradykinin and noradrenaline hyperalgesia. Brain Res. 1988 Aug 23;458(2):402–406. doi: 10.1016/0006-8993(88)90487-8. [DOI] [PubMed] [Google Scholar]
- Taniuchi M., Clark H. B., Johnson E. M., Jr Induction of nerve growth factor receptor in Schwann cells after axotomy. Proc Natl Acad Sci U S A. 1986 Jun;83(11):4094–4098. doi: 10.1073/pnas.83.11.4094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallace L. J., Partlow L. M. alpha-Adrenergic regulation of secretion of mouse saliva rich in nerve growth factor. Proc Natl Acad Sci U S A. 1976 Nov;73(11):4210–4214. doi: 10.1073/pnas.73.11.4210. [DOI] [PMC free article] [PubMed] [Google Scholar]



