Abstract
Decision-makers increasingly seek scientific guidance on investing in nature, but biodiversity remains difficult to estimate across diverse landscapes. Here, we develop empirically based models for quantifying biodiversity across space. We focus on agricultural lands in the tropical forest biome, wherein lies the greatest potential to conserve or lose biodiversity. We explore two questions, drawing from empirical research oriented toward pioneering policies in Costa Rica. First, can remotely sensed tree cover serve as a reliable basis for improved estimation of biodiversity, from plots to regions? Second, how does tropical biodiversity change across the land-use gradient from native forest to deforested cropland and pasture? We report on understory plants, nonflying mammals, bats, birds, reptiles, and amphibians. Using data from 67,737 observations of 908 species, we test how tree cover influences biodiversity across space. First, we find that fine-scale mapping of tree cover predicts biodiversity within a taxon-specific radius (of 30–70 m) about a point in the landscape. Second, nearly 50% of the tree cover in our study region is embedded in countryside forest elements, small (typically 0.05–100 ha) clusters or strips of trees on private property. Third, most species use multiple habitat types, including crop fields and pastures (to which 15% of species are restricted), although some taxa depend on forest (57% of species are restricted to forest elements). Our findings are supported by comparisons of 90 studies across Latin America. They provide a basis for a planning tool that guides investments in tropical forest biodiversity similar to those for securing ecosystem services.
Keywords: conservation science, countryside biogeography, ecosystem services, extinctions, species–area relationship
What is the potential of sustaining biodiversity and ecosystem services in agricultural landscapes? The future of biodiversity hinges on the answer, given the limited scope for expanding protected areas. Moreover, the generation and delivery of many vital ecosystem services occurs on local to regional scales in social–ecological systems where people make their livelihoods through cropping, grazing, forestry, and other rural activities. The answer is incomplete, but appears to be “high” (e.g., refs. 1–3). A further question, however, is how can this potential for conservation into the Anthropocene can be realized, with land use, other dimensions of global change, and rates of extinction intensifying rapidly worldwide (4–6) and weak institutions for protecting the global commons (7)?
Efforts to secure biodiversity and ecosystem services in rural landscapes are expanding and becoming more sophisticated (e.g., refs. 8 and 9). In the case of ecosystem services, both scientific and policy support for targeting investments have advanced rapidly (10, 11). In China, for example, 200 million people presently are being paid to engage in conservation and restoration activities; since 2000 these investments have resulted in many ecosystem service improvements, although not in biodiversity, at a national scale (12, 13). By contrast, scientific support for targeting biodiversity investments remains limited by relatively coarse and underdeveloped data, models, practical tools, and implementation efforts (14). This limitation is a bit ironic, because ecosystem services science was opened originally as a pathway for understanding human dependence on biodiversity and for motivating efforts to stem biodiversity loss (15–17).
Here we address two key questions for guiding investments in ecosystems services to benefit biodiversity. We seek answers to our questions in the context of Costa Rica, a nation remarkable for its rich biodiversity and for its innovative ecosystem services policies, launched in 1996, that presently pay landowners for services provided by forests ≥2.0 ha in size (18). The first question is whether tree cover estimates from satellites can serve as a reliable basis for improved estimation of biodiversity, on scales ranging from plots to regions. The second question is how biodiversity changes along the land-use gradient from native forest to deforested crop and pasture lands, again on scales ranging from plots to regions. We explore biodiversity at three levels: (i) the total number of species across a region (γ-diversity), broadly comparing forested and deforested areas; (ii) the total number of species at a point or plot on a landscape (α-diversity); and (iii) changes in species and abundances across different habitats (β-diversity). We draw lessons from an intensively studied region of Costa Rica and then generalize our findings through an intensive survey of studies across Latin America. We focus on the tropical forest biome because it hosts most of the world’s terrestrial biodiversity.
Results
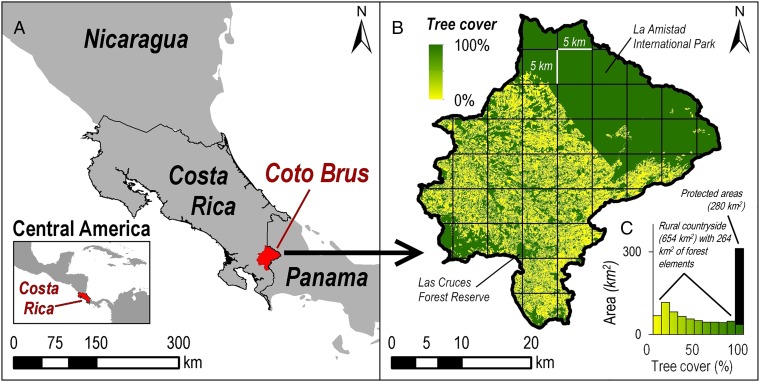
To guide investments in biodiversity across previously forested agricultural landscapes, we first asked what characteristics of a region can be remotely sensed using satellite images to quantify biodiversity across space. We focused on remotely sensing tree cover because it is a good predictor of biodiversity in deforested tropical regions (3, 19). Moreover, we start our approach by identifying tree cover at the finest spatial scales feasible using satellite images, for two reasons. First, previous studies of bats and birds suggest that biodiversity responds to changes in small areas, such as the area inside a circle with a 50- to 70-m radius (3, 19). Second, resampling or aggregating tree cover from smaller to larger scales is simple with spatial data. To estimate biodiversity for our regional case study, we created a finely detailed tree cover map for Coto Brus, Costa Rica (Fig. 1A) by measuring the surface area of land covered by large tree canopies (≥10 m in diameter). We identified all the tree cover across the 934-km2 Costa Rican canton (Fig. 1B).
Fig. 1.
Using tree cover, we spatially modeled biodiversity across a diverse region of Costa Rica. (A) First, we remotely sensed tree cover at a fine spatial resolution (0.61 m) across the canton of Coto Brus (934 km2). (B) Tree cover is concentrated in protected areas. (C) Nearly 50% of tree cover in Coto Brus is embedded in smaller countryside forest elements (0.05–100 ha), clusters, or strips of trees. We created models that predict biodiversity at a point in space, using the proportion of tree cover within a spatial scale specific to each taxonomic group.
When we used our remote sensing technique to estimate tree cover, fully 100% of the 46 randomly selected sites across the Coto Brus region agreed with ground observations of habitat classifications of forest and nonforest. Moreover, measurements of canopy cover taken inside each 0.05-ha site across the region showed no differences in canopy cover between forest sites inside a protected area (Las Cruces Forest Reserve) (Fig. 1B) and forest sites on private property (mean canopy cover ± SD, 84 ± 14% canopy cover, n = 23 sites). Sites in crop fields, mostly coffee plantations, and pastures had significantly less tree canopy cover than sites inside forests (mean canopy cover ± SD, 43 ± 34%; n = 23 sites), much of it in the form of banana plants and trees used for agricultural purposes.
According to our remotely sensed tree cover map, nearly 50% of the tree cover in Coto Brus is embedded in rural agricultural land, much of it in small and complexly configured “countryside forest elements” mostly on private property (Fig. 1C). These elements comprise trees lining property boundaries, scattered across pastures, and in winding, filamentous configurations over complex terrain in interconnected forms that typically cannot be considered isolated forest patches. Collectively, countryside forest elements cover ca. 264 km2, i.e., 40% of the 654 km2 of the land outside the protected areas (Fig. 1B).
Taken together, tree cover estimates at a fine scale reveal two broad habitat types in Coto Brus, where we conducted biodiversity sampling: (i) forest elements, both inside legally protected areas (public and private) and on legally unprotected private property, and (ii) crop fields and pastures, virtually all on legally unprotected private property. To quantify the total number of species in these two broad habitat types, we sampled forested sites (in both the Las Cruces Forest Reserve and in 2- to 100-ha countryside forest elements of various ages and shapes, n = 101 sites) and deforested sites (crop fields and pastures, n = 81 sites).
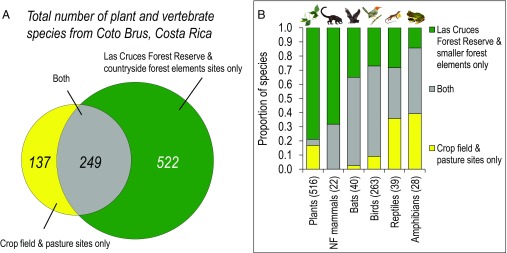
We made 67,737 plant and animal observations, for a total of 908 species at all sites. We found 771 species in the forest sites (including both the Las Cruces Forest Reserve and the countryside forest elements sites) and 386 species in crop fields and pastures (Fig. 2A). Only 4% of the 516 plant species were exotic, and all vertebrate species were native to the region. (We excluded humans, horses, cattle, cats, and dogs from all analyses). Overall, 58% of all species were encountered only in forest sites, 15% of species were encountered only in crop field and pasture sites, and 27% were observed in both (Fig. 2B). Plant species showed the greatest differences between the two broad habitat types in total number of species, with 407 species observed only in forest sites, 87 species observed only in crop field and pasture sites, and 22 species observed in both. Of the 392 vertebrate species, we observed 115 species only in forest sites, 50 species only in crop field and pasture sites, and 227 species in both (Fig. 2B).
Fig. 2.
(A) Of the 908 species recorded at all sites, 58% were observed only in forest sites, and 15% were observed only in crop field and pasture sites. (B) Taxonomic groups varied in their response to deforestation and use of crop fields and pastures. Overall, the total number of species in each broad habitat type of forest and nonforest, i.e., the γ-diversity of each habitat, was 771 species from all sites in forest elements (n = 101) and 386 species from all sites in crop fields and pastures (n = 81). NF mammals, nonflying mammals.
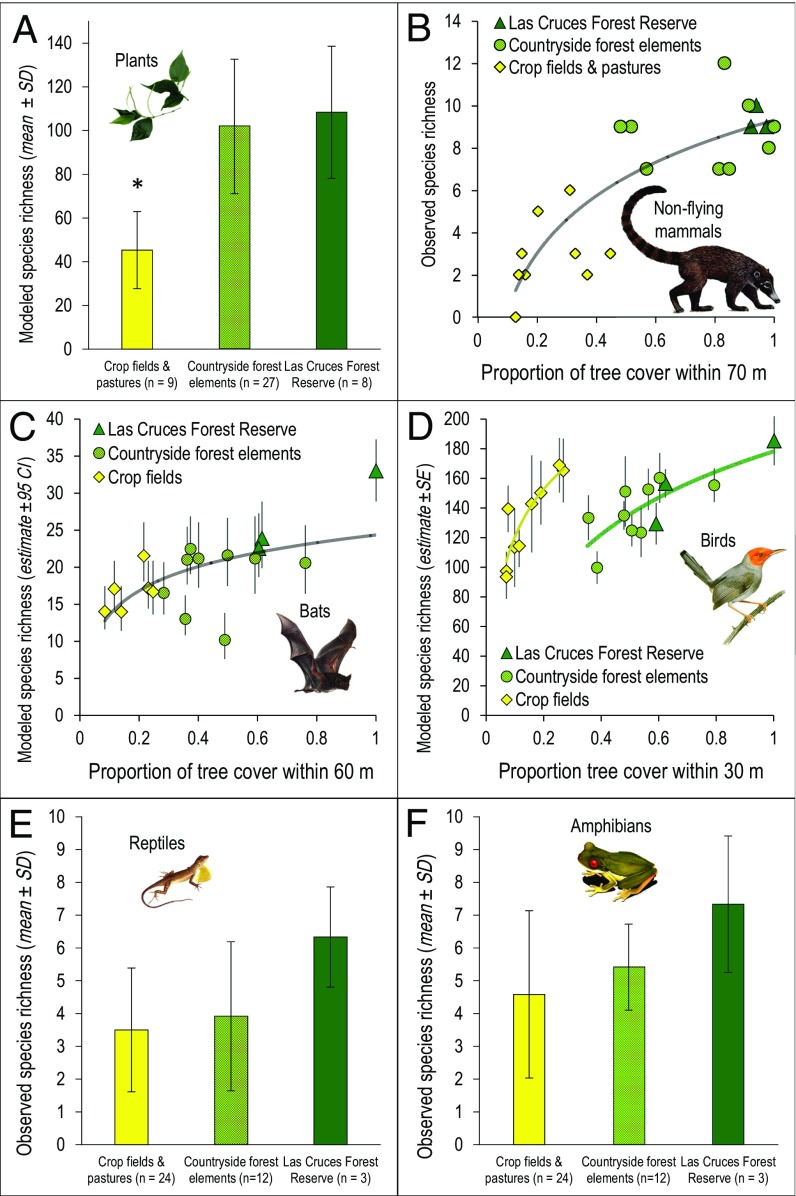
In four of the six taxonomic groups sampled (i.e., plants, nonflying mammals, bats, and birds) the number of species at a site (α-diversity) increased significantly as the amount of tree cover at a site increased. For example, the number of plant species was the same among forest sites located in the Las Cruces Forest Reserve and countryside forest elements, but sites located in crop fields and pastures contained about half the number of plant species (R2 = 0.40, P < 0.001, n = 44) (Fig. 3A and Table S1). Medium- to large-sized nonflying mammal species (>1 kg in size) were the vertebrates most sensitive to deforestation, never venturing into open areas (i.e. with <15% tree cover within 70 m) over nearly a year of continuous camera trapping, even when bait was present (R2 = 0.70, P < 0.001, n = 21) (Fig. 3B and Table S2). The number of bat species decreased by about a third in sites with no tree cover within 60 m (R2 = 0.32, P = 0.008, n = 18) (Fig. 3C and Table S3).
Fig. 3.
Increasing the proportion of local tree cover generally increases the total number of species at a point, i.e., the α-diversity. (A) The number of plant species was the same in sites located in forest elements (light and dark green) but was halved at sites in crop fields and pastures (yellow). (B–D) The number of nonflying mammal species at a point responded strongly to the amount of tree cover measured within 70 m of a site (B), whereas the numbers of bat (C) and bird (D) species at a point on a landscape were explained best by the amount of tree cover measured within 60 m and 30 m of the site, respectively. (E and F) The numbers of reptile (E) and amphibian (F) species did not increase significantly in sites with higher amounts of local tree cover, but a trend was observed.
Table S1.
Understory plant species richness by habitat type
| Variable | Estimate | SE | t value | P value |
| Intercept | 103.40 | 4.791 | 21.58 | <0.001 |
| Crop fields and pastures | −58.07 | 10.59 | −5.48 | <0.001 |
The number of plant species inside countryside forest elements and the Las Cruces Forest Reserve is nearly double the number of species observed in crop fields and pastures. No significant differences in species richness were recorded between countryside forest elements and the Las Cruces Forest Reserve.
Table S2.
Nonflying mammal species richness as a function of the amount of tree cover within 70 m
| Variable | Estimate | SE | t value | P value |
| Intercept | 9.30 | 0.60 | 15.57 | <0.001 |
| Ln* (proportion of tree cover at 70 m) | 3.89 | 0.57 | 6.89 | <0.001 |
The number of nonflying mammals increased as the proportion of tree cover increased within 70 m of a point of interest. The proportion of tree cover measured at other spatial scales was also significantly related to nonflying mammal species richness, but competitive models were discarded after comparing AICc.
Natural log of the proportion of tree cover within 70 m, measured as a proportion between 0 and 1.
Table S3.
Bat species richness as a function of the amount of tree cover within 40 m
| Variable | Estimate | SE | t value | P value |
| Intercept | 24.28 | 1.95 | 12.48 | <0.001 |
| Ln* (proportion of tree cover at 40 m) | 4.65 | 1.55 | 3.01 | 0.008 |
The number of bat species increased as the proportion of tree cover increased within 40 m of a point of interest. The proportion of tree cover measured at other spatial scales was also significantly related to bat species richness, but competitive models were discarded after comparing AICc.
Natural log of the proportion of tree cover within 40 m, measured as a proportion between 0 and 1.
The number of bird species at a site responded differently to the proportion of tree cover. The area of tree cover within a 30-m radius correlated best with the number of small bird species at a given point. However, because 24 bird species live exclusively in agricultural land and many species thrive in that habitat, especially as tree canopies increase in size (19; Fig. 2), two distinct species–area curves related to tree cover emerge (R2 = 0.67, P < 0.001, n = 21) (Fig. 3D and Table S4) for the two different, overlapping bird communities, as described further in a β-diversity analysis. The number of reptile and amphibian species did not vary significantly across sites, but trends suggest more species at sites with more tree cover (Fig. 3 E and F) and also show evidence of overlapping biological communities in further β-diversity analyses. Taken together, these results show that adding tree cover to a tropical forest region generally increases the number of species at a point or plot.
Table S4.
Bird species richness as a function of the amount of tree cover within 30 m
| Variable | Estimate | SE | t value | P value |
| Intercept | 235.05 | 17.26 | 13.62 | <0.001 |
| Habitat* (1 for forest elements) | −63.66 | 13.45 | −4.73 | <0.001 |
| Ln† (proportion of tree cover at 30 m) | 49.89 | 7.98 | 6.26 | <0.001 |
The number of birds increased as the proportion of tree cover increases within 30 m of a point of interest. The proportion of tree cover measured at other spatial scales was also significantly related to bird species richness, but competitive models were discarded after comparing AICc.
Habitat type i.e., crop fields and pastures or forest elements (including the Las Cruces Forest Reserve), had a significant effect on bird species richness.
Natural log of the proportion of tree cover within 30 m, measured as a proportion between 0 and 1.
We tested for changes in species and abundances across different habitats, i.e., β-diversity between sites, in two ways, first by comparing the relative abundances of each species from forest and nonforest sites, and second, by comparing abundance-based community similarity coefficients at forest and nonforest sites. First, the relative abundances of each species were calculated for sites in the Las Cruces Forest Reserve, forest sites in countryside forest elements, and sites in crop fields and pastures. After adjusting for sampling effort across multiple sites, a total of 30% of the 67,737 plant and animal observations were recorded inside the Las Cruces Forest Reserve, 38% in countryside forest elements, and 32% in crop fields and pastures (Fig. 4).
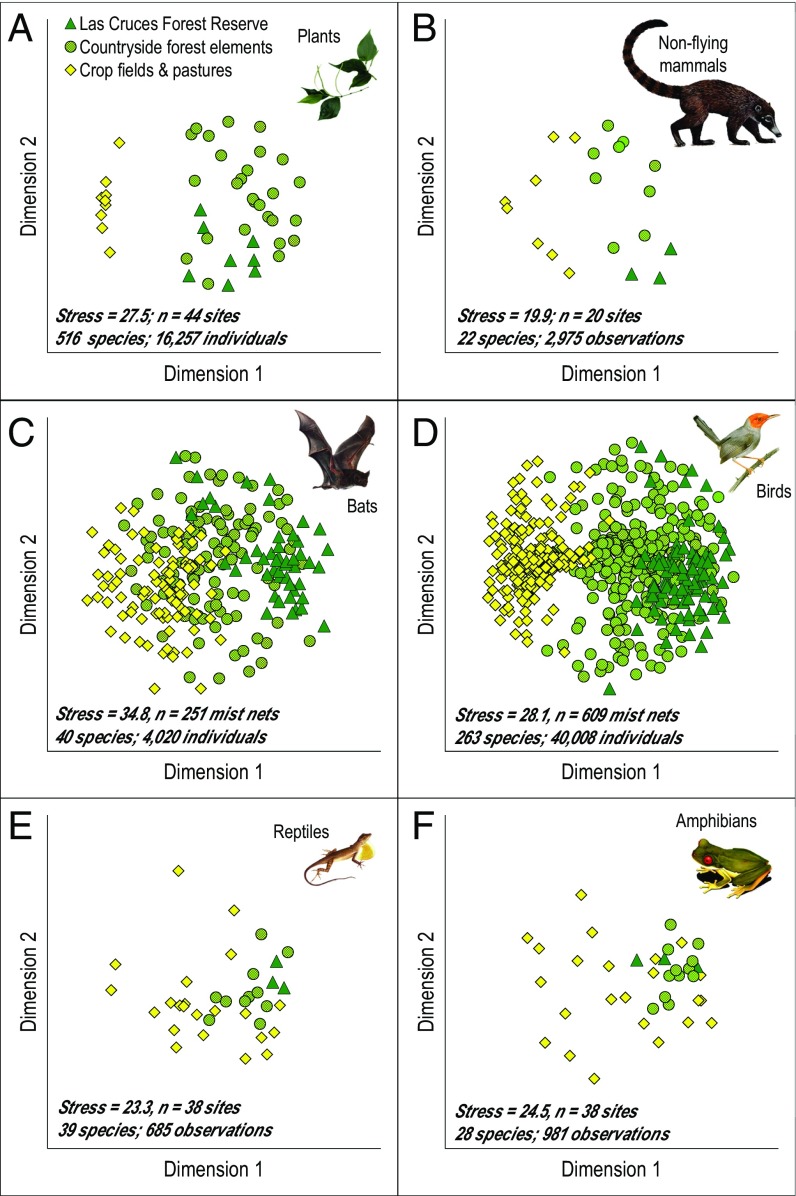
Fig. 4.
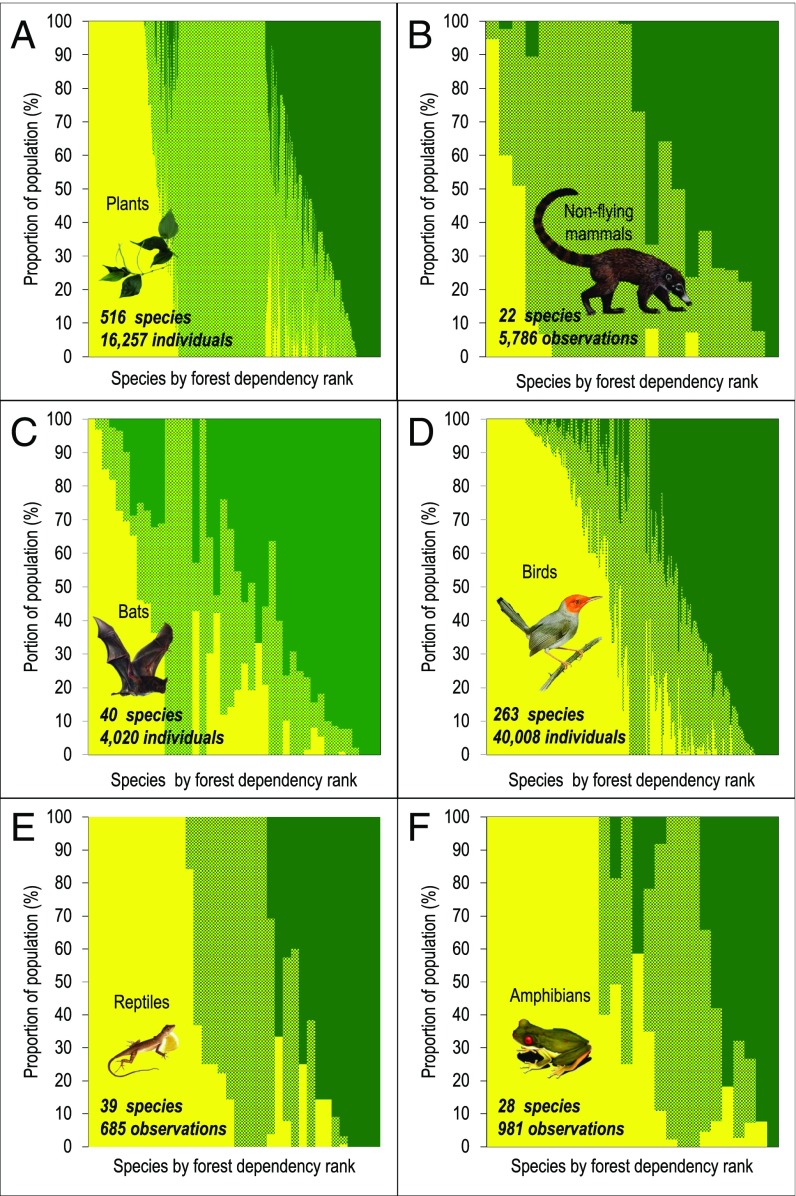
Plots A–F show changes in abundances for each species across different habitats (i.e., a measure of β-diversity) for 908 species. A total of 67,737 individual plants and animals were recorded in sites distributed across three broad categories: (i) Las Cruces Forest Reserve (dark green); (ii) countryside forest elements (light green); and (iii) crop fields and pastures (yellow). Most species used multiple habitat types, including crop fields and pastures. The forest-dependency rank ranges from forest avoidance (on the left side of the x axis) to forest dependence (on the right side of the x axis) and was determined by comparing relative abundance in the Las Cruces Forest Reserve (dark green) with that in crop fields and pastures (yellow). All proportions of populations account for differences in sampling effort among the three habitat types.
Species varied greatly in their dependence on forest, with 54% of the 908 species found in multiple habitat types (Fig. 4). Of the 516 plant species, we classified 75% as forest dependent because >50% of their population was observed in forest sites, inclusive of the Las Cruces Forest Reserve (Fig. 4A). Of the 22 nonflying mammal species, only three species (a rabbit, an opossum, and a squirrel) preferred crop fields and pastures with >50% of their observations occurring there; these data suggest that 86% of all nonflying mammal species are dependent on forest elements (Fig. 4B). Finally, assuming that a species is forest dependent if >50% of the individuals are observed in forest sites, inclusive of the Las Cruces Forest Reserve, we find that 88% of the 40 bat species, 61% of the 263 bird species, 59% of the 39 reptile species, and 54% of the 28 amphibian species are dependent on forest elements (Fig. 4 C–F). Note the decreasing order of sensitivity to deforestation.
Next, to test statistically for changes in species and abundances across different habitats, i.e., β-diversity, we compared abundance-based community similarity coefficients at forest and nonforest sites. All taxonomic groups differed significantly in their abundance-based community similarity coefficients when categorized into forest sites and nonforest sites. Permutational multivariate ANOVAs (PERMANOVAs) detected the strongest differences in abundance-based community composition between forest sites (including the Las Cruces Forest Reserve and countryside forest elements) and crop fields and pastures: plants, F1,42 = 9.60, P < 0.001 (Fig. 5A); nonflying mammals, F1,19 = 3.57, P = 0.005 (Fig. 5B); bats, F1,249 = 24.65, P < 0.001 (Fig. 5C); birds, F1,607 = 128.76, P < 0.001 (Fig. 5D); reptiles, F1,36 = 6.67, P < 0.001 (Fig. 5E); and amphibians, F1,36 = 5.60, P = 0.01 (Fig. 5F). Essentially, shifts in abundance and changes in species composition in all taxonomic groups from forest to nonforest has resulted in the formation of two overlapping biological communities in our study region—a larger, more biodiverse community associated with forest and a smaller, less biodiverse community associated with agricultural land (Figs. 2 and 5).
Fig. 5.
Significant shifts in community composition across different habitats (a measure of β-diversity) occur among all taxonomic groups. Shown in A–F are nonmetric multidimensional scaling plots that depict abundance-based community-similarity coefficients among all sites (or individual mist nets for this analysis in C and D). Closer proximity represents greater similarity in sites’ community composition. All taxonomic groups were significantly dissimilar in community composition when split into two broad habitat types: forest [including the Las Cruces Forest Reserve (dark green points) and countryside forest elements (light green points)] and crop fields and pastures (yellow points).
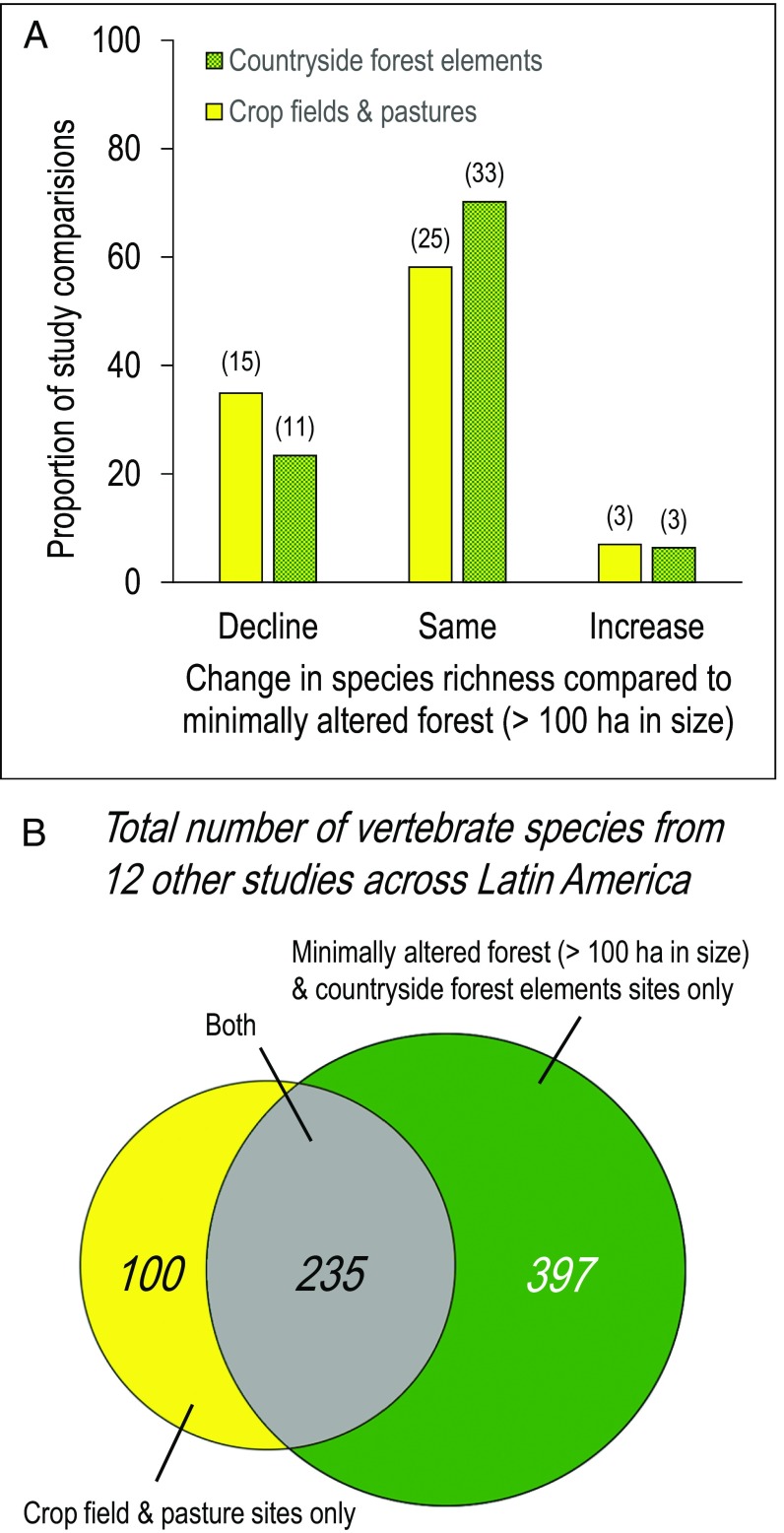
To generalize our findings beyond the study region in Costa Rica, we conducted a literature survey of countryside biogeographic studies of plants, nonflying mammals, bats, birds, reptiles, and amphibians in the tropical forest biome across Latin America. Of 857 reviewed articles, 19 (from Brazil, Costa Rica, Guatemala, Mexico, and Panama) met our criteria for study design and reporting of data (Table S5). We were able to extract data for 90 study comparisons of biodiversity change across different habitats representing the six taxonomic groups that we focused on in Costa Rica (Dataset S1). The 90 study comparisons tested for differences in the number of species (α-diversity) at sites in countryside forest elements or at sites in crop fields and pastures compared with the number of species at sites in minimally altered forest (>100 ha in size). In total, 29% of the 90 study comparisons reported a significant loss of species in crop fields and pastures compared with minimally altered forest, 68% reported no change, and 7% reported a significant increase in the number of species (Fig. 6A).
Table S5.
Search terms input into search engines to collect articles for the literature survey of countryside biogeography studies in Latin America
| capture* OR count OR inventory OR sampl* OR survey* OR trap* |
| AND |
| abundance* OR biodiversity OR densit* OR occupanc* OR richness* |
| AND |
| amphibia* OR arachnid* OR bird* OR hymenoptera OR lepidoptera OR mammal* OR plantae OR reptil* |
| AND |
| agricultur* conversion OR agricultur* disturbance OR agroecosystem* OR agroforest* OR “land use” OR plantation* OR banana* OR pineapple* OR pasture* OR sugar* OR coffee* OR oil palm OR rice OR orange* |
| AND |
| forest OR fragment* OR native OR natural OR preserv* OR reserve* OR undisturbed |
| AND |
| amazon* OR mexic* OR neotropic* |
A total of 857 research articles were reviewed, yielding 90 study comparisons.
Fig. 6.
Across Latin America, the biodiversity of the tropical forest biome is experiencing change and loss much in the same way as we report for Coto Brus, Costa Rica. (A) A survey of comparisons of species richness representing 3,041 species of plants, nonflying mammals, bats, birds, reptiles, and amphibians throughout Latin America revealed that sites located inside countryside forest elements usually support the same number of species as sites located in minimally altered forest. Comparisons of species richness in minimally altered forests and in crop fields and pastures revealed greater losses in species richness, but 58% of the comparisons between those habitat types were still statistically indistinguishable. (B) Community shift statistics from 29 studies and data from 12 studies of species occurrences throughout Latin America in three broad habitats for species of nonflying mammals (47 species), bats (62 species), birds (460 species), reptiles (67 species), and amphibians (96 species) confirmed that deforestation is leading to the formation of a novel, less biodiverse community that overlaps with a larger, more diverse biological community associated with tropical forests.
Moreover, of the 29 study comparisons that tested for shifts in abundances and/or changes in species composition between forested and deforested areas, 25 reported significant changes in β-diversity using various metrics. Finally, data published in 12 studies (20–31) of five vertebrate taxa (i.e., nonflying mammals, bats, birds, reptiles, and amphibians) supported the argument that two overlapping biological communities form with deforestation—one community associated with forest and a smaller, less biodiverse community associated with agricultural land (Fig. 6B). (Tests to determine general trends between taxonomic groups were not permitted because of data deficiency.)
Discussion
Protected areas span only 13% of the global land surface, and in reality these areas are only partially protected (32), suggesting that the future of terrestrial biodiversity depends in large part on the effect of agricultural use of unprotected lands (33–36). We developed an approach for quantifying the relationship between local tree cover and biodiversity (Fig. 3). Nearly 50% of the rural agricultural countryside of Coto Brus, our study region, is covered by trees, much of it in small forest elements of varied ages, shapes, and sizes along rivers, steep terrain, and property boundaries (Fig. 1B). These forest elements, together with the agricultural land in which they are embedded, support significant dimensions of biodiversity, whereas the adjacent crop fields and pastures support a different community (2, 3).
Despite the biodiversity found in deforested habitats, protected areas remain critical because most species at risk for extinction in the tropical forest biome require expansive forests. For example, 9% of 908 species in our study were completely dependent on the local protected area, occurring only in the Las Cruces Forest Reserve. Moreover, several local extinctions have occurred in the region because of habitat loss and poaching (37).
Our approach for modeling biodiversity across a tropical agricultural region can inform conservation policies by quantifying biodiversity using remotely sensed tree cover to scale from plots to regions. By selecting specific spatial scales to measure the area of tree cover for our α-diversity models that predict the number of species at a site, our approach accounts for forest configuration (38) and operationalizes the universal species–area relationship (39). Finally, the region we use as a case study has >10% natural habitat for most species; therefore large effects on biodiversity resulting from habitat fragmentation are not expected (40). Of course, our approach requires testing and adjustment based on the ecological contexts in which it might be applied. Moreover, our approach relies on high-quality inputs that will come from improvements in tree cover mapping and remote sensing. To replicate our approach, global estimates of tree cover are needed to guide investments in biodiversity and ecosystem services, especially in the tropical forest biome where biodiversity is concentrated and farms are small family businesses.
In practice, conservation applications of the predictive biodiversity models we present can be used to insulate protected areas and to diversify farmland in once-forested landscapes. Specifically, our models predicting the number of species at a site (α-diversity; Tables S2–S4) coupled with results that describe the kinds of species at a site (β-diversity) support arguments that increasing tree cover around existing forest reserves will lead to small gains in the number of species, but the species gained tend to be rare, unique, and at risk for extinction because of deforestation (2, 3, 41, 42) Our results also suggest that conservation efforts to diversify farmland by increasing tree cover in and around crop fields and pastures leads to large gains in the number of species, but the species gained tend to be common. Our results provide a theoretical basis for aligning conservation strategies to slow extinctions and for boosting some ecosystem services by buffering remaining forests with wildlife-friendly farmland and regenerating forests where politically feasible, given demands to farm the planet (43).
Our work supports valuing farming systems in Costa Rica and Latin America—including coffee plantations, pastures, and remnants of forests— holistically for the provision of biodiversity benefits as well as crops, forage, timber, carbon sequestration, water quality, scenic beauty, and other ecosystem services. In our study, remotely sensed tree cover serves as a reliable basis for estimating biodiversity, on scales from plots to regions. Specifically, we find that increasing the amount of tree cover on private property leads to increases in the richness of plant, nonflying mammal, bat, and bird species at the plot level. Moreover, tree cover on private property contributes to regional biodiversity by supporting the larger, more biologically diverse communities associated with tropical forests. Our survey of Latin American biodiversity and similar work on other continents, e.g., in an >2,000-y-old tropical agricultural system in the Western Ghats of India (44), demonstrate that it is possible to sustain patterns of relatively high biodiversity after deforestation when farming ecosystems are heterogeneous, incorporate elements of naturally occurring habitats, and grow perennial crops.
In grasslands and other biomes not dominated by forests or situated in the tropics, sustaining the conservation values of agricultural lands will depend on successfully identifying the features of agricultural lands linked with native biodiversity and local ecosystem services. In the context of Costa Rica, with an established program of payment for ecosystem services that has incentivized forest conservation and restoration on private property for 20 y, our approach of spatially linking tree cover at taxon-specific spatial scales to biodiversity patterns can be used as a basis for developing spatial planning tools to target investments in biodiversity and align values with goals to protect water, store carbon, and provide scenic beauty. We share a replicable approach for reducing extinctions caused by tropical deforestation and demonstrate it in the context of an existing policy that pays farmers for ecosystem services and biodiversity on their properties.
Methods
Study Area.
This research is being conducted in the rugged, hilly countryside of Coto Brus, a canton of Costa Rica, across agricultural landscapes spanning 700–1,350 m elevation. The region, originally tropical premontane wet forest, was heavily deforested in the 1960s and early 1970s (45). Today, the countryside comprises crop fields (mostly of coffee and diversified gardens), pasture, and trees scattered through crop fields and pastures, in strips bordering streams and property lines, and in forest elements of variable sizes (typically 0.05–100 ha). The landscape and biogeography are described more fully elsewhere (33, 37).
Tree Cover Estimates.
We created a finely detailed tree cover map by hand digitizing aerial photographs in Google Earth for the 934-km2 canton of Coto Brus. We used photographs taken in 2014 by commercial QuickBird satellites at a 0.61-m spatial resolution. We collected ground-truth data at 46 randomly selected plots measuring 500 m2 in three major habitat types: (i) nine sites in the Las Cruces Forest Reserve, the last large forest tract at midelevation and thus serving as a protected regional baseline; (ii) 14 sites in forest elements of various ages and sizes; and (iii) 23 sites in crop fields and pastures. In each site we surveyed vegetation to check whether the Google Earth tree-cover map matched the tree cover observed in the field.
Biodiversity.
To measure biodiversity, we identified and counted the abundances of plants and all vertebrate taxa: nonflying mammals, bats, birds, reptiles, and amphibians. We sampled plants and wildlife using standardized, systematic techniques appropriate for each taxon over extensive time periods (e.g., 1–5 y), with substantial trapping effort (i.e., ∼95,000 mist net hours and ∼15,000 trap nights), and at spatially independent sites across Coto Brus (i.e., 18–44 sites per taxa). We used sampling in the protected Las Cruces Forest Reserve as the regional baseline of minimally altered forest after sampling in La Amistad International Park (25 km away) revealed no differences in the total number of species recorded at that sampling site (i.e., in α-diversity) compared with sites in the Las Cruces Forest Reserve. We also conducted sampling in a suite of countryside forest elements of different ages and sizes and in deforested crop fields, mostly coffee plantations, and in pastures used for grazing. Biodiversity sampling of animals was conducted and approved under Stanford University's Institutional Animal Care and Use Committee (IACUC), Assurance Number: A3213-01, Protocol: 26920.
Understory Plant Biodiversity.
We used ground surveys to sample understory plants, identifying and counting all plants that fell inside a 1-m2 frame, including trees, shrubs, vines, forbs, grasses, and domestic plants. We selected sites randomly using Global Information System (GIS) software and replicated the 1-m2 sampling systematically 20 times in a total of 44 sites, each measuring 500 m2. We surveyed plants in four major habitat types: cattle pastures (three sites), coffee plantations (six sites), countryside forest elements (2- to 100-ha forest; 27 sites), and the Las Cruces Forest Reserve (eight sites). Understory plant biodiversity was aggregated for each site by pooling the 20 replicates of the 1-m2 frame inside each site. Plant species richness values for each site were modeled using techniques that account for detection biases (46).
Bird Biodiversity.
We sampled birds through constant-effort mist netting, using 20 12 × 2.6 m, 38-mm mesh ground-level mist nets in sites that covered 3–5 ha. We conducted sampling between January 25 and May 12 for 6 y (2007–2012) at 21 sites in three major habitat types: coffee plantation (nine sites), countryside forest elements (2- to 100-ha forest; nine sites), and in the Las Cruces Forest Reserve (three sites). We primarily captured passerines and other small birds, species that comprise the majority of the avifauna in the area. We fitted each captured bird with a unique aluminum leg ring, recorded the mist net location of capture, and released the bird at the capture site. Bird species richness values for each site were modeled using techniques that account for detection biases (46).
Bat Biodiversity.
We used constant-effort mist netting to sample bats, placing 20 12 × 2.6 m 38-mm mesh ground-level mist nets in a site that covered 3–5 ha. We conducted sampling between January 24 and March 28 for 4 y (2007–2010) at 18 sites in three major habitat types: coffee plantations (six sites), countryside forest elements (2- to 100-ha forest; nine sites), and the Las Cruces Forest Reserve (three sites). We sampled birds at all sites where bats were sampled. We primarily captured fruit bats; these species comprise the majority of the bats in the area (3). We fitted all captured bats with a unique collar, noted the net location of capture, and released bats at the capture site. Bat species richness values for each site were modeled using a Bayesian occupancy model that accounts for detection biases.
Nonflying Mammal Biodiversity.
We sampled medium-sized nonflying mammals (>1 kg in size) using both passive and baited camera traps. Camera trap protocols involved a motion-triggered camera trap fastened to a tree or post at 21 sites in four major habitat types of cattle pasture (three sites), coffee plantation (six sites), countryside forest elements (2- to 100-ha forest; nine sites), and the Las Cruces Forest Reserve (three sites). We operated camera traps without bait for nearly 1 y (September 2014–July 2015) and operated camera traps with bait (two bananas and three chicken gizzards, replenished daily) for a total of 14 d at each site. All cameras were at ground level. Species excluded from all analyses were humans, horses, cattle, cats, and dogs. Observed species richness of nonflying mammals at each site was used for analyses.
Reptile and Amphibian Biodiversity.
We sampled reptiles and amphibians using two complementary techniques: diurnal and nocturnal visual-encounter surveys and drift-fence trapping with pitfall and funnel traps. Six surveys for reptiles and amphibians took place during the wet and dry seasons over 3 y (2002–2004) at 39 sites in four major habitat types: cattle pasture (12 sites), coffee plantation (12 sites), countryside forest element (2- to 100-ha forest; 12 sites), and the Las Cruces Forest Reserve (three sites). We identified captured individuals but did not mark them and released all animals at the capture site. Observed reptile and amphibians species richness values for each site were used for further analyses.
Statistical Analyses.
To quantify how biodiversity changes along the land-use gradient from protected native forests to deforested agricultural plots, we explore biodiversity at three levels: (i) the total number of species in a region of forest and nonforest (the γ-diversity of each habitat), (ii) the total number of species at a point on a landscape (α-diversity), and (iii) similarity in species and abundances across different habitats (β-diversity).
To quantify changes in the total number of species at a point on a landscape, i.e., the α-diversity, we compared observed or modeled species richness estimates for each site and tested for differences among sites with different amounts of local tree cover. Plant and bird species richness was modeled using Chao species richness estimates, and bat species richness was estimated using a Bayesian occupancy model (46). We measured tree cover at each site in two ways. In the first, we used three habitat categories of Las Cruces Forest Reserve, countryside forest elements, and crop fields and pastures. In the later, more refined approach, we used the proportion of tree cover within a taxon-specific area, defined as the spatial scale at which species respond most strongly to a habitat variable (47) and hypothesized to be inclusive of configuration (38). We compared all linear models at different spatial scales using corrected Akaike Information Criteria (AICc). Including spatial structures in linear mixed effects models did not significantly influence results and was not done in final analyses.
To quantify changes in abundances across different habitats, the β-diversity, we calculated the relative abundance for each species in each habitat type and adjusted by sampling effort in space and time. Additionally, we tested statistically for changes in species and abundances across different habitats (β-diversity) by testing for differences among sites using abundance-based community similarity coefficients (46, 48). For each taxonomic group we aggregated captures and counts of all species by site and, for finer resolution in this analysis, by specific mist net for birds and bats. Each site or mist net was labeled by habitat type (i) Las Cruces Forest Reserve, (ii) countryside forest elements, or (iii) crop fields and pastures (Fig. 5). Using PERMANOVA tests, we tested for significant differences and clustering of abundance-based community similarity coefficients by habitat types.
Survey of Latin American Countryside Biogeography Studies.
To gather evidence to generalize our approach for modeling biodiversity on agricultural lands, we performed a survey of biogeographic studies in other agricultural landscapes where contiguous tropical forest once existed. We created a search term to target countryside biogeography studies in Latin America that conducted richness sampling of plants, nonflying mammals, bats, birds, reptiles, and/or amphibians (Table S5). All 857 studies returned by the search term were evaluated for inclusion by two independent readers to extract data (Dataset S1). We considered data only from studies with at least three sites in countryside forest elements or three sites in crop fields and/or pastures. Moreover, we required that studies consider at least three sites in minimally altered forest. We defined sites as minimally altered forest if they were located in continuous forest or in large forest fragments (>100 ha in size). Countryside forest elements were defined as small clusters or strips of trees (0.05–100 ha in size), secondary forests, riparian forest remnants, live fences, and agricultural fields left to fallow at least 5 y ago. Sites in crop fields and pastures were located on land that is currently being cultivated for growing crops or rearing animals and included land that had been used for agricultural purposes within 5 y.
Supplementary Material
Acknowledgments
We thank Federico Oviedo Brenes, Jeisson Figueroa Sandí, Paula Mesen Cabezas, and dozens of field assistants for help with surveys of tree cover and biodiversity; Luke Frishkoff and Hannah Frank for modeling bat species richness estimates; Ania Wrona and Leithen M’Gonigle for help with tree cover calculations; the dozens of landowners in Costa Rica who graciously allowed us to conduct field work on their property; and Paul R. Ehrlich, Gerardo Ceballos, and Rakan A. Zahawi for productive collaboration in the supportive environment of the Las Cruces Biological Station of the Organization for Tropical Studies. This work was supported by the Moore Family Foundation, the Winslow Foundation, the Haberfeld family, and Peter and Helen Bing. C.D.M. was supported by the Bing Fellowship in Honor of Paul Ehrlich, a National Science Foundation Graduate Research Fellowship, and a NatureNet Science Fellowship through The Nature Conservancy.
Footnotes
The authors declare no conflict of interest.
This paper results from the Arthur M. Sackler Colloquium of the National Academy of Sciences, “Coupled Human and Environmental Systems,” held March 14–15, 2016, at the National Academy of Sciences in Washington, DC. The complete program and video recordings of most presentations are available on the NAS website at www.nasonline.org/Coupled_Human_and_Environmental_Systems.
This article is a PNAS Direct Submission.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1604981113/-/DCSupplemental.
References
- 1.Rapidel B, DeClerck F, Le Coq JF, Beer J, editors. Ecosystem Services from Agriculture and Forestry: Measurement and Payment. Earthscan; London: 2011. [Google Scholar]
- 2.Frishkoff LO, et al. Loss of avian phylogenetic diversity in neotropical agricultural systems. Science. 2014;345(6202):1343–1346. doi: 10.1126/science.1254610. [DOI] [PubMed] [Google Scholar]
- 3.Mendenhall CD, Karp DS, Meyer CFJ, Hadly EA, Daily GC. Predicting biodiversity change and averting collapse in agricultural landscapes. Nature. 2014;509(7499):213–217. doi: 10.1038/nature13139. [DOI] [PubMed] [Google Scholar]
- 4.Tscharntke T, et al. Global food security, biodiversity conservation and the future of agricultural intensification. Biol Conserv. 2012;151:53–59. [Google Scholar]
- 5.Ceballos G, et al. Accelerated modern human-induced species losses: Entering the sixth mass extinction. Sci Adv. 2015;1(5):e1400253. doi: 10.1126/sciadv.1400253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Steffen W, Broadgate W, Deutsch L, Gaffney O, Ludwig C. The trajectory of the Anthropocene: The Great Acceleration. The Anthropocene Review. 2015;2(1):81–98. [Google Scholar]
- 7.Levin SA. Fragile Dominion: Complexity and the Commons. Perseus Publishing; New York: 2000. [Google Scholar]
- 8.Lipper L, Sakuyama T, Stringer R, Silberman D, editors. Payment for Environmental Services in Agricultural Landscapes: Economic Policies and Poverty Reduction in Developing Countries. Springer; New York: 2009. [Google Scholar]
- 9.Guerry AD, et al. Natural capital and ecosystem services informing decisions: From promise to practice. Proc Natl Acad Sci USA. 2015;112(24):7348–7355. doi: 10.1073/pnas.1503751112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kareiva P, Tallis H, Ricketts TH, Daily GC, Polasky S, editors. Natural Capital: Theory and Practice of Mapping Ecosystem Services. Oxford Univ Press; Oxford, UK: 2011. [Google Scholar]
- 11.Sharp R, et al. InVEST +VERSION+ User’s Guide (2015) The Natural Capital Project. Available at data.naturalcapitalproject.org/nightly-build/invest-users-guide/html/. Accessed May 1, 2016.
- 12.Daily GC, et al. Securing natural capital and human well-being: Innovation and impact in China. Acta Ecol Sin. 2013;33:669–676. [Google Scholar]
- 13.Ouyang Z, et al. Improvements in ecosystem services from investments in natural capital. Science. 2016;352(6292):1455–1459. doi: 10.1126/science.aaf2295. [DOI] [PubMed] [Google Scholar]
- 14.Pereira HM, et al. Scenarios for global biodiversity in the 21st century. Science. 2010;330(6010):1496–1501. doi: 10.1126/science.1196624. [DOI] [PubMed] [Google Scholar]
- 15.Ehrlich PR, Mooney HA. Extinction, substitution, and ecosystem services. Bioscience. 1983;33:248–254. [Google Scholar]
- 16.Barbier EB, Burgess JC, Folke C. Paradise Lost? The Ecological Economics of Biodiversity. Earthscan; London: 1994. [Google Scholar]
- 17.Daily GC, editor. Nature’s Services: Societal Dependence on Natural Ecosystems. Island; Washington, DC: 1997. [Google Scholar]
- 18.Asamblea Legislativa de la República de Costa Rica 1996. Ley Forestal 7575. pp. 1–36.
- 19.Mendenhall CD, Sekercioglu CH, Brenes FO, Ehrlich PR, Daily GC. Predictive model for sustaining biodiversity in tropical countryside. Proc Natl Acad Sci USA. 2011;108(39):16313–16316. doi: 10.1073/pnas.1111687108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Almeida-Gomes M, Rocha CFD. Landscape connectivity may explain anuran species distribution in an Atlantic forest fragmented area. Landsc Ecol. 2014;29(1):29–40. [Google Scholar]
- 21.Avila-Cabadilla LD, Stoner KE, Henry M, Añorve MYA. Composition, structure and diversity of phyllostomid bat assemblages in different successional stages of a tropical dry forest. For Ecol Manage. 2009;258(6):986–996. [Google Scholar]
- 22.Bael SA, Zambrano RV, Hall JS. Bird communities in forested and human-modified landscapes of Central Panama: A baseline survey for a native species reforestation treatment. International Journal of Biodiversity Science, Ecosystem Services & Management. 2013;9(4):281–289. [Google Scholar]
- 23.Bohn JL, Diemont SAW, Gibbs JP, Stehman SV, Mendoza Vega J. Implications of Mayan agroforestry for biodiversity conservation in the Calakmul Biosphere Reserve, Mexico. Agrofor Syst. 2014;88(2):269–285. [Google Scholar]
- 24.Cassano CR, Barlow J, Pardini R. Large mammals in an agroforestry mosaic in the Brazilian Atlantic forest. Biotropica. 2012;44(6):818–825. [Google Scholar]
- 25.Faria D, Baumgarten J. Shade cacao plantations (Theobroma cacao) and bat conservation in southern Bahia, Brazil. Biodivers Conserv. 2007;16(2):291–312. [Google Scholar]
- 26.Faria D. Phyllostomid bats of a fragmented landscape in the north-eastern Atlantic forest, Brazil. J Trop Ecol. 2006;22(05):531–542. [Google Scholar]
- 27.Garcia S, Finch DM, León GC. Patterns of forest use and endemism in resident bird communities of north-central Michoacán, Mexico. For Ecol Manage. 1998;110(1–3):151–171. [Google Scholar]
- 28.Marsden SJ, Whiffin M, Galetti M. Bird diversity and abundance in forest fragments and Eucalyptus plantations around an Atlantic forest reserve, Brazil. Biodiversity and Conservation. 2001;10(5):737–751. [Google Scholar]
- 29.Mendenhall CD, et al. Countryside biogeography of Neotropical reptiles and amphibians. Ecology. 2014;95(4):856–870. doi: 10.1890/12-2017.1. [DOI] [PubMed] [Google Scholar]
- 30.Santos-Filho M, Silva DJ, Sanaiotti TM. Edge effects and landscape matrix use by a small mammal community in fragments of semideciduous submontane forest in Mato Grosso, Brazil. Braz J Biol. 2008;68(4):703–710. doi: 10.1590/s1519-69842008000400004. [DOI] [PubMed] [Google Scholar]
- 31.Urbina-Cardona JN, Olivares-Pérez M, Reynoso VH. Herpetofauna diversity and microenvironment correlates across a pasture–edge–interior ecotone in tropical rainforest fragments in the Los Tuxtlas Biosphere Reserve of Veracruz, Mexico. Biol Conserv. 2006;132(1):61–75. [Google Scholar]
- 32.Ellis EC, Klein Goldewijk K, Siebert S, Lightman D, Ramankutty N. Anthropogenic transformation of the biomes, 1700 to 2000. Glob Ecol Biogeogr. 2010;19:589–606. [Google Scholar]
- 33.Daily GC, Ehrlich PR, Sánchez-Azofeifa GA. Countryside biogeography: Utilization of human-dominated habitats by the avifauna of southern Costa Rica. Ecol Appl. 2001;11:1–13. [Google Scholar]
- 34.Rosenzweig M. Win-Win Ecology: How the Earth’s Species Can Survive in the Midst of Human Enterprise. Oxford Univ Press; Oxford, UK: 2003. [Google Scholar]
- 35.Gardner TA, et al. Prospects for tropical forest biodiversity in a human-modified world. Ecol Lett. 2009;12(6):561–582. doi: 10.1111/j.1461-0248.2009.01294.x. [DOI] [PubMed] [Google Scholar]
- 36.Perfecto I, Vandermeer J, Wright A. Nature’s Matrix: Linking Agriculture, Conservation and Food Sovereignty. Earthscan; London: 2009. [Google Scholar]
- 37.Daily GC, Ceballos G, Pacheco J, Suzán G, Sánchez-Azofeifa A. Countryside biogeography of Neotropical mammals: Conservation opportunities in agricultural landscapes of Costa Rica. Conserv Biol. 2003;17:1814–1826. [Google Scholar]
- 38.Fahrig L. Rethinking patch size and isolation effects. J Biogeogr. 2013;40:1649–1663. [Google Scholar]
- 39.Harte J, Smith AB, Storch D. Biodiversity scales from plots to biomes with a universal species-area curve. Ecol Lett. 2009;12(8):789–797. doi: 10.1111/j.1461-0248.2009.01328.x. [DOI] [PubMed] [Google Scholar]
- 40.Rybicki J, Hanski I. Species-area relationships and extinctions caused by habitat loss and fragmentation. Ecol Lett. 2013;16(Suppl 1):27–38. doi: 10.1111/ele.12065. [DOI] [PubMed] [Google Scholar]
- 41.Balmford A, Green R, Phalan B. Land for food and land for nature? Daedalus. 2015;144(4):57–75. [Google Scholar]
- 42.Laurance WF, et al. Averting biodiversity collapse in tropical forest protected areas. Nature. 2012;489(7415):290–294. doi: 10.1038/nature11318. [DOI] [PubMed] [Google Scholar]
- 43.Kremen C. Reframing the land-sparing/land-sharing debate for biodiversity conservation. Ann N Y Acad Sci. 2015;1355:52–76. doi: 10.1111/nyas.12845. [DOI] [PubMed] [Google Scholar]
- 44.Ranganathan J, Daniels RJ, Chandran MD, Ehrlich PR, Daily GC. Sustaining biodiversity in ancient tropical countryside. Proc Natl Acad Sci USA. 2008;105(46):17852–17854. doi: 10.1073/pnas.0808874105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Zahawi RA, Duran G, Kormann U. Sixty-seven years of land-use change in southern Costa Rica. PLoS One. 2015;10(11):e0143554. doi: 10.1371/journal.pone.0143554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Chao A. Estimating the population size for capture-recapture data with unequal catchability. Biometrics. 1987;43(4):783–791. [PubMed] [Google Scholar]
- 47.Holland JD, Bert DG, Fahrig L. Determining the spatial scale of species’ response to habitat. Bioscience. 2004;54(3):227–233. [Google Scholar]
- 48.Chao A, Chazdon R, Colwell R, Shen TA. A new statistical approach for assessing similarity of species composition with incidence and abundance data. Ecol Lett. 2005;8:148–159. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.