Abstract
Although the global climate is changing at an unprecedented rate, links between weather and infectious disease have received little attention in high income countries. The “El Niño Southern Oscillation” (ENSO) occurs irregularly and is associated with changing temperature and precipitation patterns. We studied the impact of ENSO on infectious diseases in four census regions in the United States. We evaluated infectious diseases requiring hospitalization using the US National Hospital Discharge Survey (1970–2010) and five disease groupings that may undergo epidemiological shifts with changing climate: (i) vector-borne diseases, (ii) pneumonia and influenza, (iii) enteric disease, (iv) zoonotic bacterial disease, and (v) fungal disease. ENSO exposure was based on the Multivariate ENSO Index. Distributed lag models, with adjustment for seasonal oscillation and long-term trends, were used to evaluate the impact of ENSO on disease incidence over lags of up to 12 mo. ENSO was associated more with vector-borne disease [relative risk (RR) 2.96, 95% confidence interval (CI) 1.03–8.48] and less with enteric disease (0.73, 95% CI 0.62–0.87) in the Western region; the increase in vector-borne disease was attributable to increased risk of rickettsioses and tick-borne infectious diseases. By contrast, ENSO was associated with more enteric disease in non-Western regions (RR 1.12, 95% CI 1.02–1.15). The periodic nature of ENSO may make it a useful natural experiment for evaluation of the impact of climatic shifts on infectious disease risk. The impact of ENSO suggests that warmer temperatures and extreme variation in precipitation events influence risks of vector-borne and enteric disease in the United States.
Keywords: El Nino Southern Oscillation, infection, epidemiology, climate change, distributed lag models
The global environment is changing at an unprecedented rate and in a manner that is attributable in large part to climate change mediated by anthropogenic “greenhouse gases” (1). Direct linkage between the physical environment and infection risk has been recognized since the time of Hippocrates (∼400 BCE) (2, 3). Such linkages (e.g., surges in diarrheal disease and vector-borne disease risk with changing weather patterns) are evident in low income countries with limited health-enhancing infrastructure (4–6). The impact of the physical environment on infection risk has been less well-studied in high income countries perhaps because of the perception that wealth, strong public health institutions, and strong infrastructure mitigate such risks (7). For example, Brunkard et al. evaluated dengue virus seroprevalence on the Texas–Mexico border and demonstrated compellingly that wealth, and the infrastructure it buys, can have important protective effects against infectious disease, even within a given geographical area (8). However, risk mitigation is not equivalent to risk elimination, and, as climate and weather continue to shift ever more rapidly, infectious disease control practitioners in high, middle, and low income countries all need to develop improved understanding of the impact such shifts may have on infectious disease epidemiology.
Although it would be desirable to directly measure the effects of long-term anthropogenic climate change on infectious disease risk, the long time scale of these changes and their unprecedented and global nature make the design of “controlled studies” challenging (9). The interactions between environment, ecosystems, and disease dynamic systems are highly complex and depend on threshold effects for disease emergence and persistence; consequently, model-based projections are associated with great uncertainty (10). The El Niño Southern Oscillation (ENSO) is a major, irregular, periodic global climatic phenomenon that results from thermal inversion in the Pacific Ocean (11). ENSO has profound effects on weather and climate worldwide (12) and causes regional surges in rainfall in the United States (particularly in the “Great Basin” in the Southwest and also along the Gulf of Mexico) (13, 14), with increased temperatures in northwestern states (15). However, ENSO is associated with anomalous precipitation (both unusually wet and unusually dry) and temperatures (warmer than usual in northern states, cooler than usual in southern states) throughout the continental United States (14, 16), with effects predominantly on wintertime weather (14). ENSO events may become more frequent under climate change scenarios (17–19). Furthermore, although ENSO effects are global, there is variability in the magnitude of impact of ENSO effects, with some regions considered “teleconnected” to ENSO (i.e., they experience climatic anomalies linked to ENSO anomalies despite being many thousands of kilometers away) whereas other regions are nonteleconnected (12, 14). As such, ENSO represents a useful natural experiment that may be used to infer effects of altered climatic patterns on a variety of phenomena, including conflict (12), vegetation abundance (17), health of fisheries (20), agricultural productivity (21), and infectious diseases (22).
Most attempts to evaluate ENSO effects on infectious disease occurrence have occurred in relatively small geographic areas (22–24) or in low or middle income countries (5, 25–27). A systematic review of data on ENSO and health by Kovats et al. did identify several studies in high income countries, with most of these focusing on arboviral disease risk in Australia (28); studies of ENSO impact on viral respiratory disease have been performed in California (29), including one multicity study (24, 29), and the impact of ENSO on plague occurrence has been evaluated in New Mexico (29). As such, evaluations of the potential impact of ENSO on changing infectious disease dynamics across multiple regions in high income countries remain limited.
We combined ENSO exposure data with the National Hospital Discharge Survey (NHDS), a large, nationally representative, multiregional hospital discharge survey from the United States (30). Our objectives were as follows: (i) to evaluate associations between ENSO and infectious disease hospitalization risk in multiple US regions, and (ii) to evaluate between-region heterogeneity in such effects and, in particular, to identify differences in ENSO effect between the more teleconnected Western region of the US and other regions.
Methods
Data Sources.
Candidate disease groupings.
We identified five disease groupings of interest that could reasonably be expected to undergo epidemiological shifts due to ENSO, either because of profound seasonality, or because disease natural history is dependent on animal reservoirs or vectors that are sensitive to shifts in precipitation and temperature. The five groupings were as follows: (i) vector-borne disease (including both arthropod-borne viral disease and tick-borne and rickettsial diseases), (ii) pneumonia and influenza, (iii) enteric disease, (iv) zoonotic bacterial diseases, and (v) fungal disease.
National Hospital Discharge Survey.
Estimates of hospitalized case burden and total discharge volume by month and census region were derived from the NHDS (30), a national survey conducted on an annual basis by the US Centers for Disease Control and Prevention from 1970 to 2010 (41 complete years of data). The survey consists of diagnostic and demographic data for a probability sample of patient discharge records (31). Cases of infectious diseases of interest were identified and grouped by month and region, based on International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) codes outlined in Table 1. Due to the complex sampling design of the NHDS, case counts and total discharges were estimated using included sample weights.
Table 1.
International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) codes used to identify infectious disease groupings in hospitalization datasets
| Disease grouping | ICD-9-CM code |
| Vector-borne disease | 060-066; 080-088 |
| Pneumonia and influenza | 480-488 |
| Enteric diseases | 001-009 |
| Zoonotic bacterial diseases | 020-027 |
| Fungal disease | 110-118 |
Vector-borne disease groupings were further subdivided into arthropod-borne viral disease (060-066) and Rickettsioses and other arthropod-borne diseases (080-088) in exploratory analyses.
El Niño Southern Oscillation indices.
ENSO is a complex phenomenon manifested by changes in sea surface temperature and atmospheric pressure over the Pacific Ocean and can be characterized using a large number of measures. Our primary exposure of interest was the National Oceanic and Atmospheric Administration (NOAA) Multivariable ENSO Index (MEI), a widely used bimonthly indicator of ENSO activity that incorporates surface wind, sea surface temperature, air temperature, and cloudiness data into a single index. The MEI is scaled with values ranging from −3 (lower values consistent with “La Niña” events) to +3 (higher values consistent with ENSO events). However, we evaluated the robustness of associations identified with MEI by performing sensitivity analyses in which nine other potential measures of ENSO (including monthly average sea surface temperature measurements from several Pacific Ocean regions, the Southern Oscillation Index, Equatorial Southern Oscillation Index, and Oceanic Niño Index) were used as exposures. These indices, and links to data sources and supporting materials, can be found in Table S1.
Table S1.
Name and source of time series for indices of El Niño Southern Oscillation (ENSO) used in analyses
| Index name | Source | Further details and notes | Main website |
| Multivariate El Niño Index | http://www.esrl.noaa.gov/psd/enso/mei/table.html | Bimonthly index that incorporates surface wind, sea-surface temperature, surface-air temperature and cloudiness into a single multivariable index. Scaled from approximately −3 to +3 with higher values being more “El Niño-like.” | http://www.esrl.noaa.gov/psd/enso/mei/ |
| NOAA Southern Oscillation Index | http://www.cpc.ncep.noaa.gov/data/indices/soi | Based on gradient in sea-level pressure between Darwin and Tahiti (standardized Tahiti sea-level pressure − standardized Darwin sea-level pressure). For details see “How is the SOI Calculated” at http://www.cpc.ncep.noaa.gov/data/indices/Readme.index.shtml#SOICALC. | http://www.cpc.ncep.noaa.gov/data/indices/ |
| Australian Southern Oscillation Index | ftp://ftp.bom.gov.au/anon/home/ncc/www/sco/soi/soiplaintext.html | Standardized gradient in Tahiti–Darwin sea-level pressure, as with NOAA. Calculated differently; scaled such that lower values more ENSO-like, higher values more “La Niña-like.” For details see http://www.bom.gov.au/climate/glossary/soi.shtml | http://www.bom.gov.au/climate/current/soi2.shtml |
| Southern Oscillation Index, 3-month running average | http://www.cpc.ncep.noaa.gov/data/indices/soi.3m.txt | Three-month moving average of NOAA Southern Oscillation Index | http://www.cpc.ncep.noaa.gov/data/indices/ |
| Equatorial Southern Oscillation Index, 3-month running mean | http://www.cpc.ncep.noaa.gov/data/indices/reqsoi.3m.for | Calculated as for SOI, but standardized differences are between the eastern equatorial Pacific Ocean (80°W–130°W, 5°N–5°S) and Indonesia at (90°E–140°E, 5°N–5°S). | http://www.cpc.ncep.noaa.gov/data/indices/ |
| Nino 12 | http://www.esrl.noaa.gov/psd/data/correlation/nina1.data | Extreme eastern tropical Pacific Ocean sea-surface temperature measured at (0°–10°S, 90°W–80°W). | http://www.esrl.noaa.gov/psd/data/climateindices/list/#mei |
| Nino 3 | http://www.esrl.noaa.gov/psd/data/correlation/nina3.data | Sea-surface temperature measured in the eastern tropical Pacific Ocean (5°N–5°S)(150°W–90°W). | http://www.esrl.noaa.gov/psd/data/climateindices/list/#mei |
| Nino 4 | http://www.esrl.noaa.gov/psd/data/correlation/nina4.data | Central tropical Pacific Ocean sea-surface temperature measured at (5°N–5°S) (160°E–150°W). | http://www.esrl.noaa.gov/psd/data/climateindices/list/#mei |
| Nino 34 | http://www.esrl.noaa.gov/psd/data/correlation/nina34.data | Sea-surface temperature measured in the east–central tropical Pacific Ocean (5°N–5°S)(170°W–120°W). | http://www.esrl.noaa.gov/psd/data/climateindices/list/#mei |
| Oceanic Niño Index | http://www.cpc.ncep.noaa.gov/data/indices/oni.ascii.txt | Moving average of anomalies in eastern region Pacific Ocean sea-surface temperatures. Base period for standardization is changeable; 30-year windows are used. | http://www.cpc.ncep.noaa.gov/products/analysis_monitoring/ensostuff/ensoyears.shtml |
Note: All indices are available for entirety of 1970–2010 study period.
Statistical Methods.
Regional variation in disease incidence, temporal trends, and confidence intervals for incidence were estimated using Poisson regression models, with weighted monthly case counts as the outcome and weighted discharge estimates used as model offsets.
The effects of ENSO on disease risk at lags of up to 12 mo were evaluated using distributed lag nonlinear lag models (4, 32, 33). Briefly, these models represent exposure as a two-dimensional risk plane, known as a cross-basis, defined by level of exposure × lag (32, 33). The cross-basis can be incorporated along with other covariates into multivariable regression models, to generate three-dimensional estimates of risk as a function of exposure level and lag (33). We assumed that the effects of ENSO at a given lag would be linear, but lag structure was modeled as a cubic polynomial. ENSO effects were evaluated at the most extreme (most “El Niño-like” value) of a given index, with the midpoint between “El Niño-like” and “La Niña-like” values used as the referent (relative risk = 1.0) in models.
Because nonlinear trends in incidence, and significant seasonality, were identified in initial exploratory analyses for all disease groupings, we modeled long-term trends by incorporating linear and quadratic temporal terms into models and by including fast Fourier transforms to adjust for disease seasonality (3). Models were constructed for the following: (i) pooled national estimates, (ii) all census regions excluding the Western United States census region, and (iii) each of the four census regions individually (Northeast, Midwest, South, and West) (34). Effects were assessed through generation of three-dimensional risk surfaces and contour plots and by estimating the cumulative (integrated) relative risk (RR) of disease occurrence over the entire 12-mo period downstream from an ENSO-like signal (33).
Because we hypothesized a priori that ENSO effects would be stronger in the teleconnected Western region than in other regions, we evaluated differential effects using a “ratio of ratios” (RoR) approach, with the integrated relative risk (RR) of disease in the Western region divided by the integrated RR in non-Western regions. The 95% credible intervals were approximated through bootstrap resampling of the 95% confidence limits for Western and non-Western relative risk (RR), calculating ratios of resampled values, and identifying the 2.5th and 97.5th percentile ranks of ratios. We performed 10,000 bootstrap replicates for each ratio. We also used the metaanalytic Q-statistic to evaluate heterogeneity in effects between Western and non-Western regions (35). Metaanalytic evaluations were performed, and initial descriptive Poisson models were constructed, in Stata version 10.1 (Stata Corporation); distributed lag nonlinear lag models were constructed using the dlnm package in R (R Project); bootstrap resampling was accomplished in Microsoft Excel version 14.1 using the NORMINV command (Microsoft Corporation).
Results
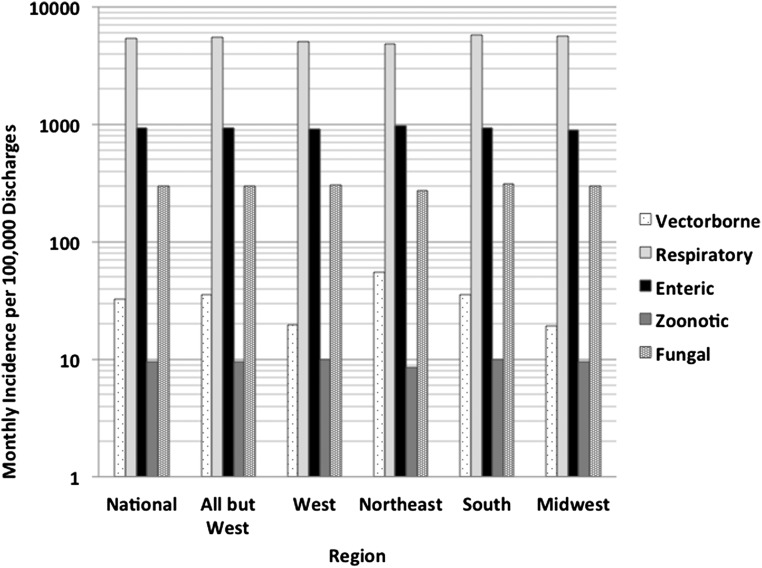
Disease group-specific monthly rates of discharge are presented in Fig. 1. Discharges due to respiratory illness (pneumonia and influenza) were most frequent whereas discharges due to zoonoses were least frequent. Although fractions of discharges due to these diseases varied across the four health regions, there were no differences between regions with respect to the rank-ordering of discharge frequency (Fig. 1).
Fig. 1.
Disease incidence by region. Rates of disease in five disease groupings per 100,000 discharges, in each census region, plotted on a log10 scale. Respiratory disease (pneumonia and influenza) was most common. Zoonotic bacterial disease was least common. Marked between-region variation in vector-borne disease is visible even on a log scale, with hospitalizations 2.7 times higher in the Northeastern region than the Western region.
Significant nonlinear multiyear temporal trends and significant seasonal oscillation were observed for all disease groupings, as was significant interregional variation in disease frequency (Table 2). The largest between-region difference in disease burden was seen for vector-borne disease in the Northeast region, which was 2.7 times higher than that in the referent Western region, consistent with the known high incidence of Lyme disease in this area (36).
Table 2.
Temporal trends, seasonal oscillation, and regional variation in disease risk for five disease groupings
| Model covariate | Vector-borne disease | Pneumonia and influenza | Enteric infectious diseases | Zoonotic bacterial diseases | Fungal diseases | |||||
| Incidence rate ratio (95% CI) | P value | Incidence rate ratio (95% CI) | P value | Incidence rate ratio (95% CI) | P value | Incidence rate ratio (95% CI) | P value | Incidence rate ratio (95% CI) | P value | |
| Year | 0.997 (0.996–0.999) | 0.001 | 1.055 (1.055–1.056) | <0.001 | 0.944 (0.944–0.944) | <0.001 | 0.918 (0.915–0.920) | <0.001 | 0.704 (0.703–0.704) | <0.001 |
| Year-squared | 1.00034 (1.00030–1.00037) | <0.001 | 0.999 (0.999–1.000) | <0.001 | 1.002 (1.002–1.002) | <0.001 | 1.001 (1.001–1.001) | <0.001 | 1.007 (1.007–1.007) | <0.001 |
| Sine (month) | 0.741 (0.738–0.745) | <0.001 | 1.256 (1.266–1.256) | <0.001 | 1.153 (1.151–1.154) | <0.001 | 0.867 (0.860–0.875) | <0.001 | 0.973 (0.970–0.974) | <0.001 |
| Cosine (month) | 0.606 (0.603–0.609) | <0.001 | 1.274 (1.273–1.274) | <0.001 | 1.070 (1.069–1.071) | <0.001 | 0.880 (0.872–0.888) | <0.001 | 1.029 (1.027–1.030 | <0.001 |
| Northeast region | 2.68 (2.65–12.71) | <0.001 | 0.979 (0.978–0.980) | <0.001 | 1.093 (1.091–1.096) | <0.001 | 0.84 (0.82–0.85) | <0.001 | 0.85 (0.85–0.86) | <0.001 |
| Southern region | 1.72 (1.70–1.74) | <0.001 | 1.14 (1.14–1.15) | <0.001 | 1.044 (1.042–1.046) | <0.001 | 1.03 (1.01–1.04) | 0.003 | 1.06 (1.05–1.06) | <0.001 |
| Midwestern region | 0.99 (0.98–1.01) | 0.282 | 1.18 (1.18–1.19) | <0.001 | 1.031 (1.029–1.033) | <0.001 | 0.90 (0.88–0.92) | <0.001 | 0.91 (0.91–0.92) | <0.001 |
CI, confidence interval. Western region incidence used as referent for regional effects.
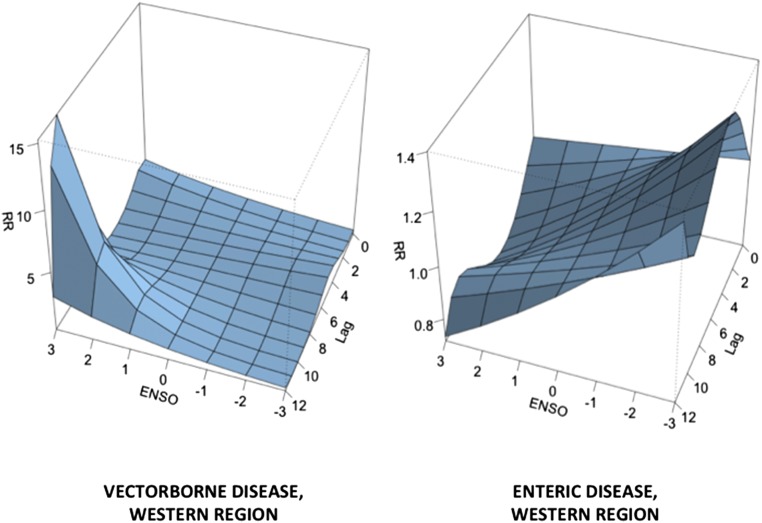
No trend was seen over the study period in the intensity of ENSO indices or interval between ENSO events. At the national level, no significant effect of ENSO was seen in any disease grouping (Table 3). However, in analyses restricted to the Western region, ENSO (defined as MEI = +3) was associated with a cumulative increase in vector-borne disease risk [RR 2.96, 95% confidence interval (CI) 1.03–8.48] and a cumulative decrease in enteric disease risk (0.73, 95% CI 0.62–0.87) over the subsequent 12-mo period (Fig. 2). By contrast, ENSO was associated with an increased risk of enteric disease in non-Western regions as a whole (RR 1.12, 95% CI 1.02–1.15), an effect driven mostly by a marked increase in risk in the Northeastern region (RR 1.15, 95% CI 1.01–1.32). Other regional effects included decreases in zoonotic bacterial disease risk in the Midwest region (RR 0.36, 95% CI 0.13–0.97) and fungal disease risk in the South (RR 0.80, 95% CI 0.66–0.97).
Table 3.
Cumulative relative risk with strong El Niño signal over a 12-mo lag period
| Geographic region | Vector-borne disease | Pneumonia and influenza | Enteric diseases | Zoonotic bacterial diseases | Fungal diseases | |||||
| Relative risk | 95% CI | Relative risk | 95% CI | Relative risk | 95% CI | Relative risk | 95% CI | Relative risk | 95% CI | |
| All regions | 1.11 | 0.77–1.60 | 1.01 | 0.96–1.06 | 1.04 | 0.96–1.13 | 0.84 | 0.50–1.42 | 0.92 | 0.80–1.05 |
| All except west | 1.06 | 0.74–1.51 | 1.02 | 0.96–1.07 | 1.12 | 1.02–1.22* | 0.98 | 0.55–1.72 | 0.91 | 0.79–1.04 |
| West | 2.96 | 1.03–8.48* | 0.99 | 0.91–1.07 | 0.73 | 0.62–0.87* | 0.43 | 0.11–1.68 | 0.92 | 0.69–1.23 |
| Northeast | 0.96 | 0.53–1.72 | 0.97 | 0.91–1.04 | 1.15 | 1.01–1.32* | 1.26 | 0.44–3.59 | 0.94 | 0.71–1.24 |
| South | 0.42 | 0.08–2.16 | 1.03 | 0.96–1.10 | 1.06 | 0.94–1.20 | 1.36 | 0.57–3.26 | 0.80 | 0.66–0.97* |
| Midwest | 0.89 | 0.40–1.99 | 1.04 | 0.97–1.11 | 1.09 | 0.96–1.24 | 0.36 | 0.13–0.97* | 1.01 | 0.81–1.25 |
| Ratio of ratios† | 2.80 | 0.91–8.52 | 0.97 | 0.88–1.07 | 0.65 | 0.54–0.79 | 0.44 | 0.11–1.99 | 1.02 | 0.74–1.41 |
Indicates 95% confidence limits that exclude the null.
Ratio of RR in Western region to RR in non-Western regions with 95% credible intervals derived via bootstrap resampling.
Fig. 2.
The 3D risk surfaces for ENSO and disease in the Western Region. Strong ENSO signals are associated with a surge in risk of vector-borne disease at a 12-mo lag, whereas the same signals are associated with a decrease in enteric disease risk at 12- and 2- to 6-mo lags. Multivariate ENSO Index values are plotted on the x axis (+3 = more El Niño-like), whereas lag (months) is plotted on the y axis and relative risk on the z axis. Note the difference in z axis scales between the two plots.
The relative-relative risk of vector-borne disease in the Western region compared with non-Western regions was 2.80, but 95% credible intervals (0.91–8.52) overlapped the null, and a metaanalytic Q-statistic did not provide strong evidence for heterogeneity (0.99 on 1 degree of freedom, P = 0.39). By contrast, the RoR for enteric disease in Western vs. non-Western regions did not overlap the null (RoR 0.65, 95% credible interval 0.54–0.79), with strong statistical evidence of heterogeneity (Q-statistic 22.48 on 1 degree of freedom, P < 0.001). No other significant heterogeneity was seen between Western and non-Western regions.
Exploratory analyses on Western region vector-borne disease effects suggested that increase in risk was likely attributable to changes in Rickettsioses and other tick-borne infections (cumulative RR 3.27, 95% CI 0.98–10.91), rather than arboviral disease (cumulative RR 1.32, 95% CI 0.03–61.38). We performed a series of sensitivity analyses on ENSO effects using alternate indices of El Niño (Tables S2–S7). For vector-borne disease and enteric disease analyses, findings remained robust when alternate indices were used, except that analyses using Nino12 (sea surface temperature in easternmost Pacific Ocean regions) were associated with imprecise confidence intervals. Fungal disease effects noted in the Southern region were not robust when alternate ENSO indices were used. Zoonotic bacterial disease risk estimates for the Midwestern region were identical in direction and similar in magnitude to those seen in the base case analysis, but confidence limits for some indices overlapped the null.
Table S2.
Effects of El Niño on vector-borne disease risk, Western region
| Index | Range (La Niña-like, referent, El Niño-like) | RR | 95% CI |
| MEI (base case) | −3, 0, +3 | 2.96 | 1.03–8.48 |
| Southern Oscillation Index, NOAA | +4.8, −0.6, −6 | 15.81 | 4.20–59.55 |
| Southern Oscillation Index, Australia | +30, 0, −30 | 10.49 | 3.18–34.54 |
| SOI NOAA, 3-month moving average | +2, −1, −3 | 18.30 | 4.61–72.65 |
| Equatorial SOI, 3-month moving average | +3, 0, −3 | 12.98 | 3.72–45.36 |
| Nino 12 | 19, 23.5, 28 | 35.71 | 0–undefined* |
| Nino 3 | 23, 26, 29 | 3.34 | 1.17–9.50 |
| Nino 4 | 26, 28, 30 | 13.68 | 3.85–48.62 |
| Nino 34 | 24, 26.5, 29 | 4.49 | 1.89–10.64 |
| Oceanic Nino Index | −2, 0, 2 | 4.65 | 1.87–11.56 |
Effects noted to be significant in base case analyses were subject to sensitivity analyses with nine alternate measures of ENSO used as exposures instead of MEI. RR denote relative risk of 12-month lagged exposures in distributed lag model, evaluated at the index-specific extreme numerical value of the given index that was most “El Niño-like.” Range midpoints were used as referent, with most “La Niña-like” values at the opposite end of the range from most “El Niño-like.” Asterisks mark analyses in which 95% confidence limits overlapped the null when the alternate index was used.
Table S7.
Effects of El Nino on zoonotic disease risk, Midwestern region
| Index | Range (La Niña-like, referent, El Niño-like) | RR | 95% CI |
| MEI (base case) | −3, 0, +3 | 0.35 | 0.13–0.97 |
| Southern Oscillation Index, NOAA | +4.8, −0.6, −6 | 0.29 | 0.10–0.88 |
| Southern Oscillation Index, Australia | +30, 0, −30 | 0.38 | 0.15–1.00* |
| SOI NOAA, 3-month moving average | +2, −1, −3 | 0.29 | 0.10–0.91 |
| Equatorial SOI, 3-month moving average | +3, 0, −3 | 0.34 | 0.11–1.04* |
| Nino 12 | 19, 23.5, 28 | 0 | 0–0.07 |
| Nino 3 | 23, 26, 29 | 0.25 | 0.08–0.74 |
| Nino 4 | 26, 28, 30 | 0.52 | 0.20–1.32* |
| Nino 34 | 24, 26.5, 29 | 0.43 | 0.21–0.88 |
| Oceanic Nino Index | −2, 0, 2 | 0.48 | 0.22–1.02* |
Effects noted to be significant in base case analyses were subject to sensitivity analyses with nine alternate measures of ENSO used as exposures instead of MEI. RR denote relative risk of 12-month lagged exposures in distributed lag model, evaluated at the index-specific extreme numerical value of the given index that was most “El Niño-like.” Range midpoints were used as referent, with most “La Niña-like” values at the opposite end of the range from most “El Niño-like.” Asterisks mark analyses in which 95% confidence limits overlapped the null when the alternate index was used.
Table S3.
Effects of El Nino on enteric disease risk, non-Western regions
| Index | Range (La Niña-like, referent, El Niño-like) | RR | 95% CI |
| MEI (base case) | −3, 0, +3 | 1.12 | 1.02–1.22 |
| Southern Oscillation Index, NOAA | +4.8, −0.6, −6 | 1.18 | 1.07–1.29 |
| Southern Oscillation Index, Australia | +30, 0, −30 | 1.17 | 1.07–1.27 |
| SOI NOAA, 3-month moving average | +2, −1, −3 | 1.18 | 1.07–1.30 |
| Equatorial SOI, 3-month moving average | +3, 0, −3 | 1.13 | 1.03–1.23 |
| Nino 12 | 19, 23.5, 28 | 1.62 | 0.82–3.18* |
| Nino 3 | 23, 26, 29 | 1.10 | 1.003–1.21 |
| Nino 4 | 26, 28, 30 | 1.11 | 1.03–1.20 |
| Nino 34 | 24, 26.5, 29 | 1.09 | 1.02–1.16 |
| Oceanic Nino Index | −2, 0, 2 | 1.12 | 1.05–1.19 |
Effects noted to be significant in base case analyses were subject to sensitivity analyses with nine alternate measures of ENSO used as exposures instead of MEI. RR denote relative risk of 12-month lagged exposures in distributed lag model, evaluated at the index-specific extreme numerical value of the given index that was most “El Niño-like.” Range midpoints were used as referent, with most “La Niña-like” values at the opposite end of the range from most “El Niño-like.” Asterisks mark analyses in which 95% confidence limits overlapped the null when the alternate index was used.
Table S4.
Effects of El Nino on enteric disease risk, Western region
| Index | Range (La Niña-like, referent, El Niño-like) | RR | 95% CI |
| MEI (base case) | −3, 0, +3 | 0.73 | 0.62–0.87 |
| Southern Oscillation Index, NOAA | +4.8, −0.6, −6 | 0.71 | 0.59–0.85 |
| Southern Oscillation Index, Australia | +30, 0, −30 | 0.73 | 0.61–0.86 |
| SOI NOAA, 3-month moving average | +2, −1, −3 | 0.69 | 0.57–0.83 |
| Equatorial SOI, 3-month moving average | +3, 0, −3 | 0.72 | 0.60–0.85 |
| Nino 12 | 19, 23.5, 28 | 0.21 | 0.05–0.83 |
| Nino 3 | 23, 26, 29 | 0.70 | 0.58–0.84 |
| Nino 4 | 26, 28, 30 | 0.76 | 0.66–0.87 |
| Nino 34 | 24, 26.5, 29 | 0.79 | 0.70–0.88 |
| Oceanic Nino Index | −2, 0, 2 | 0.79 | 0.70–0.89 |
Effects noted to be significant in base case analyses were subject to sensitivity analyses with nine alternate measures of ENSO used as exposures instead of MEI. RR denote relative risk of 12-month lagged exposures in distributed lag model, evaluated at the index-specific extreme numerical value of the given index that was most “El Niño-like.” Range midpoints were used as referent, with most “La Niña-like” values at the opposite end of the range from most “El Niño-like.” Asterisks mark analyses in which 95% confidence limits overlapped the null when the alternate index was used.
Table S5.
Effects of El Nino on enteric disease risk, Northeast region
| Index | Range (La Niña-like, referent, El Niño-like) | RR | 95% CI |
| MEI (base case) | −3, 0, +3 | 1.15 | 1.01–1.31 |
| Southern Oscillation Index, NOAA | +4.8, −0.6, −6 | 1.22 | 1.06–1.41 |
| Southern Oscillation Index, Australia | +30, 0, −30 | 1.21 | 1.07–1.38 |
| SOI NOAA, 3-month moving average | +2, −1, −3 | 1.23 | 1.06–1.42 |
| Equatorial SOI, 3-month moving average | +3, 0, −3 | 1.14 | 1.00–1.30* |
| Nino 12 | 19, 23.5, 28 | 0.76 | 0.26–2.17* |
| Nino 3 | 23, 26, 29 | 1.06 | 0.92–1.22* |
| Nino 4 | 26, 28, 30 | 1.23 | 1.11–1.38 |
| Nino 34 | 24, 26.5, 29 | 1.13 | 1.02–1.24 |
| Oceanic Nino Index | −2, 0, 2 | 1.14 | 1.04–1.26 |
Effects noted to be significant in base case analyses were subject to sensitivity analyses with nine alternate measures of ENSO used as exposures instead of MEI. RR denote relative risk of 12-month lagged exposures in distributed lag model, evaluated at the index-specific extreme numerical value of the given index that was most “El Niño-like.” Range midpoints were used as referent, with most “La Niña-like” values at the opposite end of the range from most “El Niño-like.” Asterisks mark analyses in which 95% confidence limits overlapped the null when the alternate index was used.
Table S6.
Effects of El Nino on fungal disease risk, Southern region
| Index | Range (La Niña-like, referent, El Niño-like) | RR | 95% CI |
| MEI (base case) | −3, 0, +3 | 0.80 | 0.66–0.97 |
| Southern Oscillation Index, NOAA | +4.8, −0.6, −6 | 0.92 | 0.73–1.14* |
| Southern Oscillation Index, Australia | +30, 0, −30 | 0.95 | 0.78–1.16* |
| SOI NOAA, 3-month moving average | +2, −1, −3 | 0.92 | 0.72–1.15* |
| Equatorial SOI, 3-month moving average | +3, 0, −3 | 0.82 | 0.65–1.04* |
| Nino 12 | 19, 23.5, 28 | 0.56 | 0.09–3.62* |
| Nino 3 | 23, 26, 29 | 0.84 | 0.67–1.06* |
| Nino 4 | 26, 28, 30 | 0.78 | 0.65–0.94 |
| Nino 34 | 24, 26.5, 29 | 0.85 | 0.74–0.99 |
| Oceanic Nino Index | −2, 0, 2 | 0.87 | 0.75–1.02* |
Effects noted to be significant in base case analyses were subject to sensitivity analyses with nine alternate measures of ENSO used as exposures instead of MEI. RR denote relative risk of 12-month lagged exposures in distributed lag model, evaluated at the index-specific extreme numerical value of the given index that was most “El Niño-like.” Range midpoints were used as referent, with most “La Niña-like” values at the opposite end of the range from most “El Niño-like.” Asterisks mark analyses in which 95% confidence limits overlapped the null when the alternate index was used.
Discussion
The irregular periodicity of ENSO makes this phenomenon a useful natural experiment for evaluating the impact of changing temperature and precipitation patterns on infectious disease risk (28). Our hypothesis a priori had been that ENSO would be associated with changes in the epidemiology of infectious diseases in the more teleconnected Western US region than in other regions. Indeed, we did observe a large (but imprecise) impact of ENSO on vector-borne disease risk in the Western region of the United States, without changes in disease burden in other regions. The impact of ENSO was concentrated at a lag of 10–12 mo and seemed to be largely on tick-borne disease. This finding is consistent with potential effects of precipitation and elevated temperatures on tick vectors as well as increased rodent disease reservoirs (37–40) although such observations have not been universal (41).
However, our findings on enteric disease risk were more complex. We identified heterogeneous effects of ENSO on risk, with an unexpected decrease in enteric disease risk in Western regions, but an increase in risk in other regions taken together. If this finding is valid, it yields a key insight into the potential impact of environmental impacts on infectious disease risk: A given environmental change may result in different effects on disease risk in different ecosystems. It has been noted that both unusually wet and unusually dry conditions may enhance enteric disease risk (42). In the Southwestern United States, major metropolitan areas are dependent on reservoirs and aquifers (43, 44), which may be depleted in drought conditions, with concentration of nutrients and pathogens; by contrast, other regions that depend more heavily on surface waters may be vulnerable to runoff that attends heavier rainfalls. Although we have regarded Western regions as teleconnected to ENSO and used non-Western regions as controls, ENSO has impacts on the weather across the continental United States, and, as such, the effects we observe are plausible. Furthermore, there is dissociation between regionally predominant ENSO effects and the boundaries of US census regions used in this analysis; the Northwestern United States experiences less precipitation under ENSO influence whereas the Southwestern United States experiences heavier precipitation (13, 14); because these regions are aggregated in our analysis, it is difficult to determine the precise mechanism whereby ENSO would change risk.
Similarly, the regional impacts on zoonotic bacterial disease and fungal disease risk that we observed in Midwestern and Southern regions could represent true effects although the latter effect was not robust in the face of sensitivity analyses. Given strong precipitation effects (positive and negative) associated with ENSO across the US Gulf Coast and Ohio River Valley (13), it should be clear that our consideration of only Western regions as ENSO-teleconnected represents a heuristic approach. Further verification and characterization of effects we report will require analyses at more localized geographic scales that will make it possible to identify specific environmental contributors to risk, as well as interaction between multiple exposures at different lags.
Furthermore, the absence of other clear effects of ENSO on disease risk for other disease groupings could reflect a true absence of effects, could reflect lack of statistical power to find such differences, or could represent nondifferential misclassification of exposure due to the large geographic regions under study and dissociation between boundaries of ENSO effects and census regions. Notably, we failed to identify impacts of ENSO on pneumonia and influenza risk, in contrast to the work of Choi et al., who found ENSO to be associated with attenuation of influenza risk in California (24). Our use of hospitalized case data means that we are of necessity studying only a small subset of disease cases of interest, all of which are more likely to have been managed in the outpatient setting or to have involved not seeking medical care at all (45).
Finally, it is important to note that our study focuses on changes in risk of diseases, most of which are endemic, rather than emergence of novel infectious diseases. Climatic change could favor the emergence of novel infectious diseases, by shifting communicable disease reproductive numbers past threshold values that permit disease emergence, allowing imported infectious diseases to become endemic rather than undergo stochastic extinction (46). Some infectious diseases [e.g., legionellosis (47) and West Nile virus infection (48)] either were initially recognized or truly emerged in North America, during the period under study. However, the lack of microbial specificity in our dataset and the relative rarity of such events limit our ability to study the impact of climate on disease emergence using the approach outlined above.
We identified impacts of ENSO on infection risk, with the implication that ongoing climate change may also impact infectious disease epidemiology in the United States. The magnitude and direction of such effects may be regionally variable. This finding highlights the importance of better understanding the linkages between environment and infection risk in high income regions and the need for investment in robust public health surveillance systems that are able to detect changing disease burdens. The transdisciplinary nature of the questions begged by our analysis can be answered through collaboration between data scientists and modelers and across the disciplines of epidemiology, ecology, and climate science.
Footnotes
This paper results from the Arthur M. Sackler Colloquium of the National Academy of Sciences, “Coupled Human and Environmental Systems,” held March 14–15, 2016, at the National Academy of Sciences in Washington, DC. The complete program and video recordings of most presentations are available on the NAS website at www.nasonline.org/Coupled_Human_and_Environmental_Systems.
The authors declare no conflict of interest.
This article is a PNAS Direct Submission.
Data deposition: The master dataset and accompanying documentation can be accessed at https://figshare.com/articles/Master_Dataset_ENSO_Paper/3398881.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1604980113/-/DCSupplemental.
References
- 1.Solomon S, et al., editors. Climate Change 2007: The Physical Science Basis. Cambridge Univ Press; Cambridge, UK: 2007. [Google Scholar]
- 2.Pappas G, Kiriaze IJ, Falagas ME. Insights into infectious disease in the era of Hippocrates. Int J Infect Dis. 2008;12(4):347–350. doi: 10.1016/j.ijid.2007.11.003. [DOI] [PubMed] [Google Scholar]
- 3.Fisman DN. Seasonality of infectious diseases. Annu Rev Public Health. 2007;28:127–143. doi: 10.1146/annurev.publhealth.28.021406.144128. [DOI] [PubMed] [Google Scholar]
- 4.Eisenberg MC, Kujbida G, Tuite AR, Fisman DN, Tien JH. Examining rainfall and cholera dynamics in Haiti using statistical and dynamic modeling approaches. Epidemics. 2013;5(4):197–207. doi: 10.1016/j.epidem.2013.09.004. [DOI] [PubMed] [Google Scholar]
- 5.Hales S, Weinstein P, Souares Y, Woodward A. El Niño and the dynamics of vectorborne disease transmission. Environ Health Perspect. 1999;107(2):99–102. doi: 10.1289/ehp.9910799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Siraj AS, et al. Altitudinal changes in malaria incidence in highlands of Ethiopia and Colombia. Science. 2014;343(6175):1154–1158. doi: 10.1126/science.1244325. [DOI] [PubMed] [Google Scholar]
- 7.Greer A, Ng V, Fisman D. Climate change and infectious diseases in North America: The road ahead. CMAJ. 2008;178(6):715–722. doi: 10.1503/cmaj.081325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Brunkard JM, et al. Dengue fever seroprevalence and risk factors, Texas-Mexico border, 2004. Emerg Infect Dis. 2007;13(10):1477–1483. doi: 10.3201/eid1310.061586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hsiang SM, Burke M, Miguel E. Quantifying the influence of climate on human conflict. Science. 2013;341(6151):1235367. doi: 10.1126/science.1235367. [DOI] [PubMed] [Google Scholar]
- 10.Semenza JC, Menne B. Climate change and infectious diseases in Europe. Lancet Infect Dis. 2009;9(6):365–375. doi: 10.1016/S1473-3099(09)70104-5. [DOI] [PubMed] [Google Scholar]
- 11.Climate.gov 2016 El Niño & La Niña (El Niño-Southern Oscillation) (National Oceanic and Atmospheric Administration, Washington, DC). Available at https://www.climate.gov/enso. Accessed May 9, 2016.
- 12.Hsiang SM, Meng KC, Cane MA. Civil conflicts are associated with the global climate. Nature. 2011;476(7361):438–441. doi: 10.1038/nature10311. [DOI] [PubMed] [Google Scholar]
- 13.Ropelewski CF, Halpert MS. Global and regional scale precipitation patterns associated with El Niño/Southern Oscillation. Mon Weather Rev. 1987;115:1606–1626. [Google Scholar]
- 14.Becker EJ, Berbery EH, Higgins RW. Understanding the characteristics of daily precipitation over the United States using the North American Regional Reanalysis. J Clim. 2009;22(23):6268–6286. [Google Scholar]
- 15.Halpert MS, Ropelewski CF. Anomalous temperature patterns associated with the Southern Oscillation. J Clim. 1992;5:577–593. [Google Scholar]
- 16.Climate Prediction Center 1997 State El Niño Impacts (National Weather Service, Silver Spring, MD). Available at www.cpc.ncep.noaa.gov/products/analysis_monitoring/ensostuff/states/states.html. Accessed May 12, 2016.
- 17.Ingram JC, Dawson TP. Climate change impacts and vegetation response on the island of Madagascar. Philos Trans A Math Phys Eng Sci. 2005;363(1826):55–59. doi: 10.1098/rsta.2004.1476. [DOI] [PubMed] [Google Scholar]
- 18.Latif M, Keenlyside NS. El Nino/Southern Oscillation response to global warming. Proc Natl Acad Sci USA. 2009;106(49):20578–20583. doi: 10.1073/pnas.0710860105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Timmermann A, et al. Increased El Niño frequency in a climate model forced by future greenhouse warming. Nature. 1999;398:694–697. [Google Scholar]
- 20.Kilduff DP, Di Lorenzo E, Botsford LW, Teo SL. Changing central Pacific El Niños reduce stability of North American salmon survival rates. Proc Natl Acad Sci USA. 2015;112(35):10962–10966. doi: 10.1073/pnas.1503190112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Stige LC, et al. The effect of climate variation on agro-pastoral production in Africa. Proc Natl Acad Sci USA. 2006;103(9):3049–3053. doi: 10.1073/pnas.0600057103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jury MR. Climate influence on dengue epidemics in Puerto Rico. Int J Environ Health Res. 2008;18(5):323–334. doi: 10.1080/09603120701849836. [DOI] [PubMed] [Google Scholar]
- 23.Zaraket H, Saito R, Tanabe N, Taniguchi K, Suzuki H. Association of early annual peak influenza activity with El Niño southern oscillation in Japan. Influenza Other Respi Viruses. 2008;2(4):127–130. doi: 10.1111/j.1750-2659.2008.00047.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Choi KM, Christakos G, Wilson ML. El Niño effects on influenza mortality risks in the state of California. Public Health. 2006;120(6):505–516. doi: 10.1016/j.puhe.2006.01.011. [DOI] [PubMed] [Google Scholar]
- 25.Mantilla G, Oliveros H, Barnston AG. The role of ENSO in understanding changes in Colombia’s annual malaria burden by region, 1960-2006. Malar J. 2009;8:6. doi: 10.1186/1475-2875-8-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lama JR, Seas CR, León-Barúa R, Gotuzzo E, Sack RB. Environmental temperature, cholera, and acute diarrhoea in adults in Lima, Peru. J Health Popul Nutr. 2004;22(4):399–403. [PubMed] [Google Scholar]
- 27.Pascual M, Rodó X, Ellner SP, Colwell R, Bouma MJ. Cholera dynamics and El Niño-Southern Oscillation. Science. 2000;289(5485):1766–1769. doi: 10.1126/science.289.5485.1766. [DOI] [PubMed] [Google Scholar]
- 28.Kovats RS, Bouma MJ, Hajat S, Worrall E, Haines A. El Niño and health. Lancet. 2003;362(9394):1481–1489. doi: 10.1016/S0140-6736(03)14695-8. [DOI] [PubMed] [Google Scholar]
- 29.Ebi KL, Exuzides KA, Lau E, Kelsh M, Barnston A. Association of normal weather periods and El Niño events with hospitalization for viral pneumonia in females: California, 1983-1998. Am J Public Health. 2001;91(8):1200–1208. doi: 10.2105/ajph.91.8.1200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.National Center for Health Statistics 2016 National Hospital Discharge Survey (US Centers for Disease Control and Prevention, Atlanta, GA). Available at www.cdc.gov/nchs/nhds/. Accessed May 9, 2016.
- 31.DeFrances CJ, Lucas CA, Buie VC, Golosinskiy A. 2006 national hospital discharge survey. Natl Health Stat Rep. 2008;2008(5):1–20. [PubMed] [Google Scholar]
- 32.Brown KA, Daneman N, Arora P, Moineddin R, Fisman DN. The co-seasonality of pneumonia and influenza with Clostridium difficile infection in the United States, 1993-2008. Am J Epidemiol. 2013;178(1):118–125. doi: 10.1093/aje/kws463. [DOI] [PubMed] [Google Scholar]
- 33.Gasparrini A, Armstrong B, Kenward MG. Distributed lag non-linear models. Stat Med. 2010;29(21):2224–2234. doi: 10.1002/sim.3940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Dowell SF, Kupronis BA, Zell ER, Shay DK. Mortality from pneumonia in children in the United States, 1939 through 1996. N Engl J Med. 2000;342(19):1399–1407. doi: 10.1056/NEJM200005113421904. [DOI] [PubMed] [Google Scholar]
- 35.Deeks J, Altman D. Effect measures for meta-analysis of trials with binary outcomes. In: Egger M, Davey Smith G, Altman DG, editors. Systematic Reviews in Health Care: Meta-Analysis in Context. BMJ Publishing Group; London: 1995. pp. 313–335. [Google Scholar]
- 36.Orloski KA, Hayes EB, Campbell GL, Dennis DT. Surveillance for Lyme disease: United States, 1992-1998. MMWR Surveillance Summaries. 2000;49(3):1–11. [PubMed] [Google Scholar]
- 37.Ostfeld RS, Canham CD, Oggenfuss K, Winchcombe RJ, Keesing F. Climate, deer, rodents, and acorns as determinants of variation in lyme-disease risk. PLoS Biol. 2006;4(6):e145. doi: 10.1371/journal.pbio.0040145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ostfeld RS, et al. Effects of acorn production and mouse abundance on abundance and Borrelia burgdorferi infection prevalence of nymphal Ixodes scapularis ticks. Vector Borne Zoonotic Dis. 2001;1(1):55–63. doi: 10.1089/153036601750137688. [DOI] [PubMed] [Google Scholar]
- 39.McCabe GJ, Bunnell JE. Precipitation and the occurrence of lyme disease in the northeastern United States. Vector Borne Zoonotic Dis. 2004;4(2):143–148. doi: 10.1089/1530366041210765. [DOI] [PubMed] [Google Scholar]
- 40.Schulz M, Mahling M, Pfister K. Abundance and seasonal activity of questing Ixodes ricinus ticks in their natural habitats in southern Germany in 2011. J Vector Ecol. 2014;39(1):56–65. doi: 10.1111/j.1948-7134.2014.12070.x. [DOI] [PubMed] [Google Scholar]
- 41.Schulze TL, Jordan RA, Schulze CJ, Hung RW. Precipitation and temperature as predictors of the local abundance of Ixodes scapularis (Acari: Ixodidae) nymphs. J Med Entomol. 2009;46(5):1025–1029. doi: 10.1603/033.046.0508. [DOI] [PubMed] [Google Scholar]
- 42.Nichols G, Lane C, Asgari N, Verlander NQ, Charlett A. Rainfall and outbreaks of drinking water related disease and in England and Wales. J Water Health. 2009;7(1):1–8. doi: 10.2166/wh.2009.143. [DOI] [PubMed] [Google Scholar]
- 43. NW-QAN (2014) Regional Assessment of Groundwater Quality in the Basin and Range, Rio Grande, Coastal Basins, and Central Valley Aquifer Systems of the Southwestern United States (US Geological Survey, Reston, VA). Available at water.usgs.gov/nawqa/studies/praq/swpa/. Accessed May 15, 2016.
- 44.Foti R, Ramirez JA, Brown TC. 2012 Vulnerability of U.S. Water Supply to Shortage: A Technical Document Supporting the Forest Service 2010 RPA Assessment (US Department of Agriculture, Fort Collins, CO). Gen. Tech. Rep. RMRS-GTR-295. Available at www.fs.fed.us/rm/pubs/rmrs_gtr295.pdf. Accessed May 9, 2014.
- 45.Majowicz SE, et al. Estimating the under-reporting rate for infectious gastrointestinal illness in Ontario. Can J Public Health. 2005;96(3):178–181. doi: 10.1007/BF03403685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Harvell CD, et al. Climate warming and disease risks for terrestrial and marine biota. Science. 2002;296(5576):2158–2162. doi: 10.1126/science.1063699. [DOI] [PubMed] [Google Scholar]
- 47.Fraser DW, et al. Legionnaires’ disease: Description of an epidemic of pneumonia. N Engl J Med. 1977;297(22):1189–1197. doi: 10.1056/NEJM197712012972201. [DOI] [PubMed] [Google Scholar]
- 48.Centers for Disease Control and Prevention (CDC) West Nile virus activity: Eastern United States, 2001. MMWR Morb Mortal Wkly Rep. 2001;50(29):617–619. [PubMed] [Google Scholar]