Abstract
Purpose
During difficult airway management, oxygen insufflation through airway-exchange and intubating catheters (AEC/IC) can lead to life-threatening hyperinflation. Ventrain® was originally designed to facilitate emergency ventilation using active expiration through short, small-bore cannulas. Herein, we studied its efficacy (oxygenation and ventilation) and safety (avoidance of hyperinflation) in a long, small-bore AEC.
Methods
In six anesthetized pigs, the upper airway was obstructed, except for a 100 cm long, 3 mm internal diameter AEC. After apneic desaturation to a peripheral oxygen saturation (SpO2) of < 70%, ventilation through the AEC was started with Ventrain at an oxygen flow of 15 L·min−1, a frequency of 30 breaths·min−1, and an inspiration/expiration ratio of approximately 1:1. It was continued for ten minutes.
Results
Within one minute, severe hypoxia was reversed from a median [interquartile range] arterial saturation (SaO2) of 48 [34-56] % before initiation of Ventrain ventilation to 100 [99-100] % afterward (median difference 54%; 95% confidence interval [CI] 44 to 67; P = 0.028). In addition, hypercarbia was reversed from PaCO2 of 59 [53-61] mmHg to 40 [38-42] mmHg (median difference of −18 mmHg; 95% CI −21 to −15; P = 0.028). After ten minutes of Ventrain use, peak inspiratory and end-expiratory pressures were lower than during baseline pressure-controlled ventilation (8 [7-9] mmHg vs 12 [10-14] mmHg and −2 [−3 to +1] mmHg vs 4 [2 to 4] mmHg, respectively; P = 0.027 for both). No hemodynamic deterioration occurred.
Conclusion
Ventrain provides rapid reoxygenation and effective ventilation through a small-bore AEC in pigs with an obstructed airway. In clinical emergency situations of obstructed airways, this device may be able to overcome problems of unintentional hyperinflation and high intrapulmonary pressures when ventilating through long, small-bore catheters and could therefore minimize the risks of barotrauma and hemodynamic instability.
Résumé
Objectif
Pendant une prise en charge des voies aériennes difficile, l’insufflation d’oxygène via un échangeur de sonde de voie aérienne où une sonde d’intubation peut provoquer une hyperinflation potentiellement fatale. À l’origine, le Ventrain® a été conçu pour faciliter la ventilation d’urgence en utilisant l’expiration active via des canules courtes et de petit diamètre. Ici, nous avons étudié son efficacité (pour l’oxygénation et la ventilation) et son innocuité (évitement de l’hyperinflation) lorsqu’il était utilisé avec un échangeur de sonde long et de petit diamètre.
Méthode
Les voies aériennes supérieures de six porcs sous anesthésie ont été complètement obstruées, à l’exception d’un échangeur de sonde d’une longueur de 100 cm et d’un diamètre interne de 3 mm. Après avoir réalisé une désaturation apnéique jusqu’à une saturation d’oxygène (SpO2) périphérique < 70 %, la ventilation a été amorcée via l’échangeur de sonde avec un Ventrain à un débit d’oxygène de 15 L·min−1, une fréquence de 30 respirations·min−1, et un rapport d’inspiration/expiration d’environ 1:1. Cette expérience a été poursuivie pendant dix minutes.
Résultats
Après moins d’une minute, l’hypoxie grave a été neutralisée, passant d’une saturation artérielle (SaO2) médiane [écart interquartile] de 48 [34-56] % avant l’amorce de la ventilation avec le Ventrain à 100 [99-100] % après l’amorce du traitement (différence médiane 54 %; intervalle de confiance [IC] 95 % 44 à 67; P = 0,028). De plus, l’hypercarbie a été neutralisée, passant d’une PaCO2 de 59 [53-61] mmHg à 40 [38-42] mmHg (différence médiane de −18 mmHg; IC 95 % −21 à −15; P = 0,028). Après dix minutes d’utilisation du Ventrain, les pressions inspiratoire et télé-expiratoire maximales étaient plus basses que pendant la ventilation en pression contrôlée de base (8 [7-9] mmHg vs 12 [10-14] mmHg et −2 [−3 à +1] mmHg vs 4 [2 à 4] mmHg, respectivement; P = 0,027 pour les deux). Il n’y a eu aucune détérioration hémodynamique.
Conclusion
Le Ventrain permet une ré-oxygénation rapide et une ventilation efficace via un échangeur de sonde de voie aérienne de petit diamètre chez des porcs dont les voies aériennes sont obstruées. Dans les situations cliniques d’urgence de voies aériennes obstruées, ce dispositif pourrait permettre de surmonter les problèmes d’hyperinflation involontaire et de pressions intrapulmonaires élevées lorsqu’on ventile via de longues sondes à petit diamètre et ainsi minimiser les risques de barotraumatisme et d’instabilité hémodynamique.
Introduction
In cases where re-intubation is likely to be difficult, an airway-exchange catheter (AEC) can be placed through the endotracheal tube (ETT) prior to extubation.1 Should re-intubation prove necessary, an ETT can be advanced over the AEC. In case of a difficult intubation, a similar strategy of “railroading” an ETT over an intubating catheter (IC) is a basic principle of securing the airway. In addition to functioning as a guide for the ETT, some AECs and ICs have a central lumen through which oxygen can be delivered.
Flow resistance in a small-bore AEC or IC is high, however, so oxygen must be insufflated at high pressure. With conventional high-pressure (i.e., jet) ventilation devices that are used to inject oxygen through such catheters, the injected gas volume cannot be estimated. Also, because of its flow resistance, exhalation through small-bore catheters is limited. Thus, the absence of a patent airway allowing egress of air around the AEC/IC is a relative contraindication for oxygen injection into the lungs. Despite this contraindication, these catheters are still typically used in patients whose airways may become compromised due to obstruction (e.g., tissue swelling or hematoma).2 In such patients, expiration time is prolonged, and complete exhalation may not occur.3
Because of the inability to estimate inspiratory volumes and the risk of incomplete exhalation, injection of oxygen through an AEC or IC bears a significant risk of hyperinflation, barotrauma, hemodynamic deterioration, and even death.1,4,5 To decrease the risks associated with intrapulmonary/intrathoracic pressure buildup, the inspiration time must be minimized, leading to low minute volumes.1
Ventrain® (Ventinova Medical B.V., Eindhoven, The Netherlands) is a US Food and Drug Administration approved, manually operated, high-pressure ejector ventilator designed to be used with small-bore catheters in emergency airway situations (Figure).6-8 In contrast to pressure-regulated, high-pressure jet ventilators, this device is flow-regulated, thereby allowing control of inspiratory volumes. In addition, expiration can be actively facilitated by jet flow-generated suction, even through small-bore catheters. Previously published in vivo studies have demonstrated the efficacy of Ventrain on reoxygenation and ventilation when used with a short, small-bore transtracheal cannula (7.5 cm length, 2 mm inner diameter [ID]).9-11 Ventrain, however, may also be effective when used with much longer, small-bore catheters, such as an AEC/IC, and has achieved up to 7.5 L·min−1 ventilation during in vitro testing through a 100 cm long, 3 mm ID AEC across a broad range of compliances and resistances.12 To our knowledge, however, this has not yet been tested in vivo.
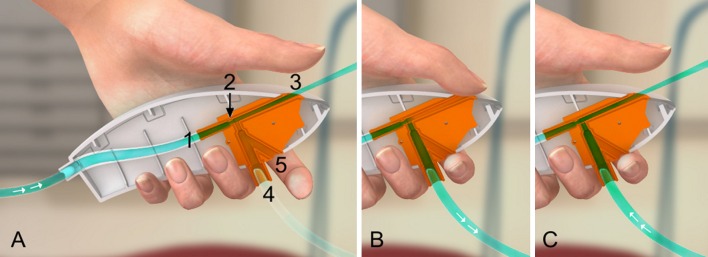
Figure.
A) A specially designed, optimized ejector is mounted inside the shell of the Ventrain (shown in a longitudinal section). Tubing coming from a pressure-compensated oxygen source (flowmeter or flow regulator attached to a wall outlet or an oxygen tank) is connected to the inlet (1). The oxygen flow is accelerated by a jet nozzle (2) and enters the exhaust pipe (3) at high speed, thereby creating subatmospheric pressure at the side-port (4) according to Bernoulli’s principle. A bypass (5) allows air to enter the ejector. Short tubing at the side-port connects the ejector by Luer lock to a small-bore catheter in the patient’s airway (not shown). So long as the bypass (functioning as the on/off switch) is open, there is no clinically relevant flow via the side-port to and from the catheter (= equilibration with the atmosphere). B) The bypass needs to be completely closed to activate Ventrain. By simultaneously and firmly closing the outlet of the exhaust pipe (functioning as the inspiration/expiration switch), oxygen is insufflated (= inspiration). C) Releasing the inspiration/expiration switch can reverse the flow through the small-bore catheter. Entrainment of gas by the high-speed flow of oxygen facilitates expiration and thus shortens the expiration time (= active expiration). Used with permission of Ventinova Medical, Eindhoven, The Netherlands
Accordingly, the goal of our study was to investigate the efficacy and safety of using Ventrain for reoxygenation and ventilation through a long AEC in a porcine model of a simulated completely obstructed upper airway and hypoxia. The primary aim was to assess whether efficient oxygenation (SaO2 > 95%) and ventilation (PaCO2 < 45 mmHg) could be restored within one minute. The secondary aim was to evaluate whether ventilation using Ventrain could be maintained safely during ten minutes of ventilation with regard to respiratory pressures and hemodynamic parameters.
Methods
The local animal welfare committee approved our study (no. 02-036/12; December 2012). After overnight fasting with free access to water, a convenience sample of six female pigs (weight range 26.5-29.8 kg) were premedicated with intramuscular ketamine 1200 mg and midazolam 10 mg. Anesthesia was induced with propofol 3 mg·kg−1, sufentanil 1 µg·kg−1, and pancuronium 0.2 mg·kg−1 via an intravenous cannula inserted into an ear vein. It was subsequently maintained with a continuous infusion of propofol 20 mg·kg−1·hr−1, sufentanil 1 µg·kg−1·hr−1, and pancuronium 0.1 mg·kg−1·hr−1. Following anesthesia induction, a lactated Ringer’s bolus of 20 mL·kg−1 and subsequent infusion of 10 mL·kg−1·hr−1 were administered intravenously. The adequacy of anesthesia was assessed according to hemodynamic variables. Incremental doses of intravenous sufentanil were given if clinically indicated.
Before the study, the pigs had been ventilated with conventional pressure-controlled ventilation (PCV), jet ventilation, and Ventrain according to different protocols for other, unrelated experiments.
Pressure-controlled ventilation via a 6.0 mm ID ETT (with the cuff inflated to 23 mmHg) was initiated with a fraction of inspired oxygen (FIO2) of 0.4. Ventilator settings were adjusted to achieve normocapnea, measured using sidestream end-tidal capnometry (Datex AS/3; Datex-Ohmeda, Helsinki, Finland).
The pigs were connected to a three-lead electrocardiogram, and peripheral oxygen saturation (SpO2) was monitored at the pigs’ tails with a pulse oximeter (Datex AS/3; Datex-Ohmeda, Helsinki, Finland). An 18G arterial catheter (ref FA-04018; Arrow, Reading, PA, USA) was inserted into the right carotid artery for arterial pressure monitoring and blood sampling. Pulmonary arterial pressure, mixed venous oxygen saturation, and cardiac output were monitored with a pulmonary artery catheter (ref 746HF8; Edwards Lifesciences Corporation, Irvine, CA, USA) that was advanced via a 9-French sheath (ref AH-09801; Arrow, Reading, PA, USA) into the right internal jugular vein.
Tracheal pressures were measured using a pressure-monitoring stylet [sheath of a guide wire (ref 502-563; Cordis, Miami Lakes, FL, USA) cut to a length of 50 cm] that was placed under fiberoptic guidance adjacent to the ETT with its tip just above the carina.
After baseline recordings, the ventilator was disconnected. A 100 cm long, 3 mm ID AEC (Cook Medical, Bloomington, IN, USA) was then placed in the trachea through a sealed Shiley double swivel connector (Covidien, Dublin, Ireland), simulating complete upper airway obstruction. The proper depth of insertion (i.e., above the the right upper lobe bronchial orifice) had been fibreoptically measured in advance.
When the SpO2 reached 70% (“zero minutes” = end of apnea), ventilation with Ventrain was started at an oxygen flow of 15 L·min−1 using a calibrated, pressure-compensated flowmeter (Draeger; Luebeck, Germany), a frequency of 30 breaths·min−1, and an inspiration/expiration (I:E) ratio of 1:1 guided by a metronome. Chest movements (as the primary criterion for an effective tidal volume) were continuously observed during this period. The I:E ratio was adjusted as needed based on intermittent tracheal pressure readings, aiming for 0 mmHg end-expiratory pressure.
Arterial blood samples and respiratory and hemodynamic data were collected at baseline, zero minutes (i.e, end of apnea), and at one, two, five, and ten minutes after resumption of Ventrain ventilation. After conclusion of the study protocol, the pigs were euthanized with an overdose of pentobarbital 50 mg·kg−1.
A separate in vitro protocol was added with a setup similar to that previously described6 to investigate flow-dependent insufflation (i.e., proximal driving) pressure and suction pressure when Ventrain was used with the studied AEC. The Ventrain was connected to a calibrated, pressure-compensated flowmeter and attached to an AEC as used in the in vivo protocol. Insufflation and suction pressures were measured at the side port of a T-piece connected proximally to the AEC at oxygen flows of 3, 6, 9, 12, and 15 L·min−1 using the Calibration Analyzer series RT 200 (Timeter Instrument Corporation, St. Louis, MO, USA). Each experiment was performed five times.
Statistical analysis
This pilot study evaluated the feasibility and safety of Ventrain used in combination with an AEC to re-oxygenate patients safely in cases of acutely obstructed airways. Ventilation with Ventrain was considered sufficient (i.e., feasible) if SaO2 was restored to > 95% within one minute after starting ventilation with Ventrain and if hypercapnia was reversed or prevented. Safety of ventilation with Ventrain was evaluated using peak inspiratory pressures and hemodynamic data. We expected that hyperinflation would lead to increased inspiratory pressures and deteriorated hemodynamic variables.
The primary endpoints for statistical analysis were SaO2 and PaCO2 at one minute compared with that at the end of apnea (as a measure of feasibility). The secondary end points were the respiratory and hemodynamic data during ten minutes of ventilation with Ventrain compared with the baseline PCV (as a measure of safety). Data are presented as the median [interquartile range (IQR)] unless specified otherwise and were analyzed using a Wilcoxon signed rank test. The median differences and 95% confidence intervals (CI) were calculated using the Hodges-Lehmann estimator. Statistical significance was defined at the level of P < 0.05 (two-sided). Analyses were performed using SPSS Statistics for Windows (version 23.0; IBM, Armonk, NY, USA).
Results
The median [IQR] minute volume for normoventilation of the six pigs during the initial period of PCV was 5.1 [4.2-5.9] L·min−1. Respiratory rate and tidal volume were 14 [12-15] breaths·min−1 and 384 [308-406] mL, respectively.
Respiratory measurements during the study period are shown in Table 1. During the period of apnea, the time to desaturation < SpO2 70% was one to two minutes. The median [IQR] PaO2 decreased to 36 [31-38] mmHg, and PaCO2 increased to 59 [53-61] mmHg. After initiation of ventilation with an FIO2 of 1.0 using Ventrain, the oxygenation and normocarbia were restored within one minute in all six pigs. The SaO2 increased from a median [IQR] of 48 [34-56] % to 100 [99-100] % (median difference 54; 95% CI 44 to 67; P = 0.028) with a corresponding increase in PaO2 to 318 [159-377] mmHg (P = 0.028). The median [IQR] PaCO2 decreased from 59 [53-61] mmHg to 40 [38-42] mmHg (median difference −18; 95% CI −21 to −15; P = 0.028).
Table 1.
Respiratory measurements at baseline (pressure control ventilation, pre-apnea), zero (end of apnea) and then one, two, five, and ten minutes after initiation of ventilation with Ventrain
| Baseline | 0 min | 1 min | 2 min | 5 min | 10 min | |
|---|---|---|---|---|---|---|
| SaO2 (%) | 100 [100-100] | 48 * [34-56] | 100 # [99-100] | 100 [100-100] | 100 [99-100] | 100 [100-100] |
| PaO2 (mmHg) | 218 [184-242] | 36 * [31-38] | 318 # [159-377] | 284 * [255-298] | 261 * [241-283] | 280 * [241-355] |
| PaCO2 (mmHg) | 38 [37-40] | 59 * [53-61] | 40 # [38-42] | 37 [35-40] | 36 [33-38] | 35 * [31-37] |
| PIP (mmHg) | 12 [10-14] | 0 * [0-0] | 8 *# [7-9] | 8 * [7-8] | 9 * [8-9] | 8 * [7-9] |
| EEP (mmHg) | 4 [2-4] | 0 * [0-0] | −2 * [−3-1] | −2 * [−2-1] | −2 * [−2-2] | −2 * [−3-1] |
Data are presented as median [interquartile range]
EEP = end-expiratory pressure; PaCO2 = arterial partial pressure of carbon dioxide; PaO2 = arterial partial pressure of oxygen; PIP = peak inspiratory pressure; SaO2 = arterial oxygen saturation; *P < 0.05 vs baseline, # P < 0.05 1 min vs 0 min
Oxygenation was maintained during the ten minutes of Ventrain ventilation, and PaCO2 declined further to 35 [31-37] mmHg at the end of the ten minutes (P = 0.028 compared to baseline), indicating mild hyperventilation.
Compared with the baseline PCV conditions, both peak inspiratory and end-expiratory pressures were lower when ventilating with Ventrain (baseline peak inspiratory pressures 12 [10-14] mmHg vs 8 [7- 9] mmHg after ten minutes of ventilation with Ventrain (P = 0.027) and baseline end-expiratory pressures 4 [2 to 4] mmHg vs −2 [−3 to 1] mmHg after ten minutes of ventilation with Ventrain (P = 0.027).
Hemodynamic data are presented in Table 2. These indices showed a change in measured variables during apnea, with increases in mean pulmonary artery pressure and decreases in mixed venous oxygen saturation. During ventilation with Ventrain, the heart rate and mean pulmonary artery pressure returned to baseline values, whereas the mean arterial blood pressure, mixed venous oxygen saturation, and cardiac output tended to increase slightly.
Table 2.
Hemodynamic measurements at baseline (pressure control ventilation, pre-apnea), zero (end of apnea) and then one, two, five, and ten minutes after initiation of ventilation with Ventrain.
| Baseline | 0 min | 1 min | 2 min | 5 min | 10 min | |
|---|---|---|---|---|---|---|
| HR (beats·min−1) | 108 [73-120] | 114 [93-129] | 100 [87-115] | 99 [84-123] | 102 [80-127] | 105 [80-126] |
| MAP (mmHg) | 55 [48-68] | 64 [57-86] | 71 *# [66-91] | 76 * [70-94] | 74 * [66-95] | 73 * [62-91] |
| Mean PAP (mmHg) | 16 [14-18] | 24 * [20-27] | 20 *# [18-24] | 18 [16-20] | 16 [14-18] | 16 [13-17] |
| SvO2 (%) | 50 [41-59] | 24 * [17-41] | 63 *# [51-69] | 64 * [56-73] | 66 * [51-76] | 65 * [51-76] |
| CO (L·min−1) | 3.4 [2.7-3.7] | 3.1 [2.4-3.9] | 3.3 [2.6-3.8] | 3.4 [2.9-4.6] | 3.9 * [3.2-4.3] | 3.9 * [3.2-4.9] |
Data are presented as median [interquartile range]
CO = cardiac output; HR = heart rate; MAP = mean arterial blood pressure; PAP = pulmonary artery pressure; SvO2 = mixed venous oxygen saturation; *P < 0.05 vs baseline, # P < 0.05 1 min vs 0 min
No clinical signs of barotrauma or hemodynamic deterioration were noted. Table 3 shows the flow dependence of the insufflation and suction pressures during the in vitro experimental series. Both pressures also increased with increasing flow.
Table 3.
In vitro insufflation and suction pressures measured proximally of the AEC at different oxygen flows
| Oxygen flow (L·min−1) |
3 | 6 | 9 | 12 | 15 |
|---|---|---|---|---|---|
| Insufflation pressure (mmHg) | 8 [7-8] | 23 [22-23] | 40 [40-40] | 62 [62-62] | 87 [87-88] |
| Suction pressure * (-mmHg) | 11 [11-12] | 32 [30-32] | 53 [52-53] | 72 [71-72] | 87 [87-87] |
Values are median [interquartile range]
* Note that all suction pressures are negative pressures. AEC = airway exchange catheter
Discussion
In the completely obstructed upper airway of our animal model, rapid reoxygenation and efficient ventilation were achieved through a 100 cm long, 3 mm ID AEC using Ventrain. No signs of barotrauma or hemodynamic disturbance were observed, suggesting that hyperinflation did not occur. Our findings may be useful for patients in whom an AEC/IC is in place to facilitate reintubation, but threading the ETT over the catheter is either difficult or has failed.
In a single-centre, cohort study, emergent re-intubation over an AEC during the postoperative period failed in two of nine (22%) cases.13 In a second, similar study, intubation over an AEC was unsuccessful in ten of 51 (20%) patients on the first attempt and failed after multiple attempts in four of 51 (8%) cases.14 If (re)intubation over an AEC/IC is difficult, ensuring sufficient and safe oxygenation of the patient becomes extremely challenging. A well-known option is jet ventilation through an AEC/IC. This option, however, has been associated with pulmonary barotrauma and subsequent hemodynamic deterioration. In one report of the use of an AEC, barotrauma occurred in 11% of patients in whom jet ventilation was used.4
Possible mechanisms leading to pneumothorax include trauma to the tracheobronchial tree by the AEC/IC, barotrauma associated with high intrapulmonary pressures, and hyperinflation following insufficient exhalation of insufflated gas.5,15 Importantly, non-pressure-limited insufflation of oxygen through an AEC/IC via connecting tubing directly attached to a flowmeter or flow regulator is also inherently dangerous because of the high piped (or tank) oxygen pressure even with low-flow oxygen.5
In contrast to high-pressure jet ventilation devices, Ventrain insufflates oxygen at a much lower pressure despite being driven by high pressure. Inside the functional centerpiece of Ventrain, high pressure at the jet nozzle is converted to high velocity of the flowing gas (Figure), so insufflation pressure is significantly lower compared with that associated with jet ventilation (Table 3). In a previous study where a 7.5 cm long, 2 mm ID transtracheal cannula was attached to Ventrain with a driving pressure of 2300 cm H2O (= 1725 mmHg), the actual insufflation pressure at the proximal end of the cannula was 140 cm H2O (= 105 mmHg) at an oxygen flow of 15 L·min−1.6 This situation is similar to the insufflation pressures we found in the present study when using Ventrain with the AEC studied: 87 mmHg at an oxygen flow of 15 L·min−1.
As insufflated gas can be evacuated effectively by jet flow-generated suction (i.e., the Bernoulli principle), intrapulmonary/intrathoracic pressures do not build up even during prolonged periods of ventilation with Ventrain if assisted expiration is properly applied.9 It remains extremely important, however, to observe chest wall movements to provide feedback to the operator regarding the adequacy of both the inspiration and expiration phases.
When connecting Ventrain to an oxygen source, the flow must be set depending on the patient’s characteristics. Flow control allows easy estimation of the volume injected over time (e.g., redirecting a flow of 15 L·min−1 to the patient for one second results in an insufflation volume of 0.25 L). Usually 12-15 L·min−1 is chosen in adults, whereas lower flow rates are advisable in children (especially small ones).16 Because the Bernoulli effect becomes more pronounced at increasing flow rates, expiratory support can be adjusted to the situational requirements (patient factors; length and ID of the catheter).
As recently published in vivo data suggest, Ventrain provides efficient reoxygenation and expiratory support when used with a short, small-bore transtracheal cannula in an otherwise highly or completely obstructed airway. In a completely open upper airway, however, Ventrain is less efficient for reoxygenation and ventilation.10,11
Of note, at the end of apnea (zero minutes), we showed that directly measured arterial oxygen saturation was even lower than the 70% saturation recorded using pulse oximetry. Several factors may have contributed to this finding. Pulse oximetry readings always lag behind true arterial oxygen levels due to algorithmic processing. In addition, pulse oximetry becomes increasingly inaccurate at low saturations. Because the trigger to start drawing blood in this study was a peripheral saturation reading of 70%, the blood collected for gas analysis was taken some seconds later. At already low partial oxygen pressures, a small decrease in oxygen pressure could lead to a large drop in oxygen saturation. Hence, the time lapse before sampling could increase the difference between peripheral and arterial oxygen saturation readings.
Furthermore, the PaO2 value during the intervention exceeded the baseline value. The PaO2, however, was lower than expected from a 0.4 increase in the FIO2 during pre-apnea PCV to 1.0 during ventilation with Ventrain. Several factors might have caused this discrepancy. Ventilation with Ventrain in this study protocol might not have been optimal as the settings used led to (calculated) tidal volumes of just 0.25 L. Not surprisingly, compared with the pre-apnea PCV with higher tidal volumes and lower frequencies, the peak inspiratory pressures were lower during ventilation with Ventrain. In addition, no positive end-expiratory pressure was applied, and assisted expiration, although observing the pigs’ chest walls to fall to their original position, resulted in a slightly negative end-expiratory pressure. Lastly, as the pigs had already been ventilated for other studies the same day, these prior interventions may have led to ventilator-induced lung injury, limiting oxygen uptake.
In our study setup, ventilation with an I:E ratio of approximately 1:1 at an oxygen flow of 15 L·min−1 led to a (calculated) minute volume of 7.5 L·min−1. The pigs required a minute volume of just 5.1 L·min−1 for normoventilation, however, which explains the hyperventilation seen during the intervention period.
Hemodynamic stability was preserved, suggesting that pulmonary hyperinflation and barotrauma were unlikely. Arterial blood pressure and cardiac output tended to increase slightly while ventilating with Ventrain, with no notable change in heart rate. Thus, a rise in stroke volume may be speculated. Lower inspiratory and end-expiratory airway pressures and the unintentional creation of a minimal negative intrathoracic pressure during assisted expiration might have augmented the preload. Negative end-expiratory pressure has been used during cardiopulmonary resuscitation with promising results.17 Our study, however, was not designed to investigate the detailaed hemodynamic effects of ventilation with Ventrain or to compare Ventrain ventilation with PCV.
One of the limitations of our study design is that, in real life, the I:E ratio cannot be adjusted by continuous intratracheal pressure monitoring, which would require a separate measurement channel. Therefore, it is crucial to observe chest movements meticulously in cases in which the upper airway is highly or completely obstructed. Intermittent pressure measurement via the lumen of an AEC/IC is also possible but, unfortunately, somewhat cumbersome. We chose to guide inspiratory and expiratory times by intratracheal pressures to homogenize ventilation among the pigs, which resulted in only small adjustments of the inspiratory and expiratory times. In light of clinical uncertainty and the risk of injury, a short equilibration phase between the ventilation cycles may be a pragmatic alternative to minimize the risk of undesired positive/negative pressure buildup in the patient’s lungs/chest.9
Another limitation to our study is the small sample size. In our opinion, however, the marked improvement in all of the animals while ventilating with Ventrain suggests a potential clinical value of the rescue strategy of using the lumen of an AEC/IC as an airway in case of intubation/reintubation problems in highly obstructed airways. We used pigs whose lung anatomy resembles that of the human lung, making this a generally well accepted in vivo model suitable for extrapolation to clinical practice. Obviously, a study such as this cannot be done in humans, but anecdotal reports reveal that Ventrain could be life-saving when used in combination with long, small-bore catheters.16
Although in the acute setting the primary goal is oxygenation, the fact that even normoventilation can be achieved with Ventrain implies an additional advantage over conventional methods of reoxygenation through an AEC/IC. Optimal oxygenation (both as rapid as possible and over longer periods of time) demands adequate gas exchange and therefore efficient ventilation.
In addition, the principle of flow control allows the operator to adapt to the situational requirements of the patient and the problematic airway. Decreasing the flow rate results in lower driving, as well as insufflation pressure.6 As a result, to minimize the risk of overinflation and barotrauma, it is recommendable that a lower flow rate be used at the start, increasing it stepwise as needed.
Our findings suggest that if acquiring an airway over an AEC/IC takes longer than anticipated (or while preparing for a surgical airway with an AEC/IC in tracheal position), rapid and adequate reoxygenation and sufficient ventilation can be achieved for at least ten minutes using Ventrain. In addition, in cases of critical airway obstruction, flow-controlled ventilation with Ventrain through an AEC/IC appears to be safe by providing control of insufflated and exhaled gas volume, which thereby controls intrapulmonary/intrathoracic pressure. Although we did not compare Ventrain with jet ventilation, the results suggest an advantage for Ventrain in this clinical situation. Admittedly, in a situation where the upper airway is not critically occluded, high-frequency jet ventilation might be more efficient. We would expect this to be true if the diameter of the patient’s airway is more than twice the diameter of the AEC, as it would minimize the risk of pulmonary hyperinflation.18 In clinical practice, it may be difficult to choose the optimal mode of rescue ventilation, and the choice would be dictated by the a priori risk of tissue swelling or hematoma and respective clinical signs. Even during rescue ventilation, the diameter of the surrounding airway can change, with the physician then having to consider switching to another ventilation method.
Nevertheless, it is important that general safety measures are always followed (i.e., proper placement of the AEC/IC and meticulous observation of the patient, which includes visual confirmation of the chest wall rising and falling to its original position). In the case of any ventilation uncertainty, switching to the equilibration mode of Ventrain is recommended.
In conclusion, in this preliminary study, Ventrain provided safe, effective oxygenation and ventilation through a long small-bore AEC in pigs with a simulated completely obstructed upper airway and hypoxia. Our results suggest potential clinical applicability and usefulness of Ventrain not only in combination with short, transtracheal cannulas but also long, small-bore AEC/IC when (re)intubation is difficult or has failed.
Acknowledgments
We thank the employees at the animal research facilities of the University Jena and Benjamin Preussler for their help and support in performing the study and Chris H. Van Leuvan (The Canberra Hospital, ACT, Australia) for the English editing. Maurice Theunissen, epidemiologist at the Maastricht University Medical Center, kindly provided his assistance in analyzing the data. The Figure was taken from animated instructions (www.ventrain.com) and was modified with permission of Ventinova Medical, Eindhoven, The Netherlands.
Competing interests
Dietmar Enk is the inventor of Ventrain and receives royalty payments from Ventinova Medical. The Maastricht University Medical Center has received royalty payments from Ventinova Medical.
Editorial responsibility
This submission was handled by Dr. Hilary P. Grocott, Editor-in-Chief, Canadian Journal of Anesthesia.
Author contributions
Michiel W.P. de Wolf contributed substantially to the analysis and interpretation of data and wrote the article. Reiner Gottschall, Niels P. Preussler, and Markus Paxian contributed substantially to conception and design of the manuscript and the acquisition of data. Dietmar Enk contributed substantially to all aspects of this manuscript, including conception and design; acquisition, analysis, and interpretation of data and drafting the article.
Funding
This work was supported by European funding, OP-Zuid [31R104].
References
- 1.Popat M, Mitchell V, Dravid R, Patel A, Swampillai C, Higgs A. Difficult Airway Society Guidelines for the management of tracheal extubation. Anaesthesia. 2012;67:318–340. doi: 10.1111/j.1365-2044.2012.07253.x. [DOI] [PubMed] [Google Scholar]
- 2.Cavallone LF, Vannucci A. Extubation of the difficult airway and extubation failure. Anesth Analg. 2013;116:368–383. doi: 10.1213/ANE.0b013e31827ab572. [DOI] [PubMed] [Google Scholar]
- 3.Dworkin R, Benumof JL, Benumof R, Karagianes TG. The effective tracheal diameter that causes air trapping during jet ventilation. J Cardiothorac Anesth. 1990;4:731–736. doi: 10.1016/S0888-6296(09)90012-6. [DOI] [PubMed] [Google Scholar]
- 4.Cooper RM. The use of an endotracheal ventilation catheter in the management of difficult extubations. Can J Anesth. 1996;43:90–93. doi: 10.1007/BF03015965. [DOI] [PubMed] [Google Scholar]
- 5.Duggan LV, Law JA, Murphy MF. Brief review: Supplementing oxygen through an airway exchange catheter: efficacy, complications, and recommendations. Can J Anesth. 2011;58:560–568. doi: 10.1007/s12630-011-9488-4. [DOI] [PubMed] [Google Scholar]
- 6.Hamaekers AE, Borg PA, Enk D. Ventrain: an ejector ventilator for emergency use. Br J Anaesth. 2012;108:1017–1021. doi: 10.1093/bja/aes033. [DOI] [PubMed] [Google Scholar]
- 7.Enk D. Patent US 8950400 B2. Gas flow reversing element, United States Patent and Trademark Office, February 10, 2015. Available from URL: https://www.google.si/patents/US8950400 (accessed September 2016).
- 8.Lang SA. Emergency airway management: What are the roles for surgical cricothyroidotomy and the Ventrain® device? Can J Anesth. 2016;63:997–998. doi: 10.1007/s12630-016-0669-z. [DOI] [PubMed] [Google Scholar]
- 9.Berry M, Tzeng Y, Marsland C. Percutaneous transtracheal ventilation in an obstructed airway model in post-apnoeic sheep. Br J Anaesth. 2014;113:1039–1045. doi: 10.1093/bja/aeu188. [DOI] [PubMed] [Google Scholar]
- 10.Hamaekers AE, van der Beek T, Theunissen M, Enk D. Rescue ventilation through a small-bore transtracheal cannula in severe hypoxic pigs using expiratory ventilation assistance. Anesth Analg. 2015;120:890–894. doi: 10.1213/ANE.0000000000000584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Paxian M, Preussler NP, Reinz T, Schlueter A, Gottschall R. Transtracheal ventilation with a novel ejector-based device (Ventrain) in open, partly obstructed, or totally closed upper airways in pigs. Br J Anaesth. 2015;115:308–316. doi: 10.1093/bja/aev200. [DOI] [PubMed] [Google Scholar]
- 12.Dias EM, Hamaekers A, Borg PA, Enk D. Adequate minute volume ventilation through a 100 cm long, 3 mm inner diameter airway exchange catheter by expiratory ventilation assistance (EVA): 19AP5-6. Eur J Anaesthesiol. 2012;29:239–240. doi: 10.1097/00003643-201206001-00796. [DOI] [Google Scholar]
- 13.McLean S, Lanam CR, Benedict W, Kirkpatrick N, Kheterpal S, Ramachandran SK. Airway exchange failure and complications with the use of the Cook Airway Exchange Catheter®: a single center cohort study of 1177 patients. Anesth Analg. 2013;117:1325–1327. doi: 10.1213/ANE.0b013e3182a7cd3d. [DOI] [PubMed] [Google Scholar]
- 14.Mort TC. Continuous airway access for the difficult extubation: the efficacy of the airway exchange catheter. Anesth Analg. 2007;105:1357–1362. doi: 10.1213/01.ane.0000282826.68646.a1. [DOI] [PubMed] [Google Scholar]
- 15.Harris K, Chalhoub M, Maroun R, Elsayegh D. Endotracheal tube exchangers: should we look for safer alternatives? Heart Lung. 2012;41:67–69. doi: 10.1016/j.hrtlng.2011.06.005. [DOI] [PubMed] [Google Scholar]
- 16.Willemsen MG, Noppens R, Mulder AL, Enk D. Ventilation with the Ventrain through a small lumen catheter in the failed paediatric airway: two case reports. Br J Anaesth. 2014;112:946–947. doi: 10.1093/bja/aeu125. [DOI] [PubMed] [Google Scholar]
- 17.Segal N, Parquette B, Ziehr J, Yannopoulos D, Lindstrom D. Intrathoracic pressure regulation during cardiopulmonary resuscitation: a feasibility case-series. Resuscitation. 2013;84:450–453. doi: 10.1016/j.resuscitation.2012.07.036. [DOI] [PubMed] [Google Scholar]
- 18.Buczkowski PW, Fombon FN, Lin ES, Russell WC, Thompson JP. Air entrainment during high-frequency jet ventilation in a model of upper tracheal stenosis. Br J Anaesth. 2007;99:891–897. doi: 10.1093/bja/aem312. [DOI] [PubMed] [Google Scholar]