Abstract
We report the case of an immunocompetent man who presented with fever and abdominal pain and was found to have a hepatic abscess and a peri-renal abscess with a computerized tomography scan. The hepatic abscess was drained percutaneously and cultures revealed the presence of Lactococcus lactis that was sensitive to penicillin. The patient was successfully treated with ceftriaxone and metronidazole with resolution of the abscesses. Further work-up revealed atrophic gastritis, vitamin B12 deficiency, periodontitis and gingivitis, suggesting a possible site of entry for the development of the abscesses.
Keywords: Lactococcus lactis, Intra-abdominal abscess, Pernicious anemia
Introduction
Lactococcus lactis is a facultative anaerobic, catalase-negative, gram-positive coccus widely used for the industrial production of fermented dairy products such as milk, cheese and yogurt [1]. Although Lactococcus lactis was considered to be non-pathogenic, it has been reported to cause human disease in the last two decades, in both immunocompetent and immunocompromised patients [2], [3], [4], [5], [6], [7], [8], [9], [10], [11], [12].
Pernicious anemia is a manifestation of autoimmune gastritis resulting from the destruction of gastric parietal cells and the associated lack of intrinsic factor to bind ingested vitamin B12 [13]. Malnutrition and micronutrient deficiencies, diabetes and poor oral hygiene are risk factors of periodontal diseases such as gingivitis and periodontitis.
We report an unusual case of L. lactis ssp cremoris infection in an immunocompetent male with severe periodontitis and pernicious anemia presenting with intra-abdominal abscesses.
Case report
A 46-year-old male of Greek origin, with no medical history, was admitted to the hospital because of fever and increasing abdominal pain. He was an active smoker and worked as heavy vehicle driver with international trips in Central Europe. He did not drink alcohol and used to consume semi-hard cheese. The patient was well until one month before admission when odontalgia developed, followed by chills, night sweats, arthralgias and myalgias. Partial improvement was noticed with paracetamol and ibuprofen. He reported anorexia and weight loss of 10 kg over a period of one month. Ten days before admission, fever with temperature up to 39.5oC and right upper quadrant pain developed along with diarrhea without presence of blood or mucus.
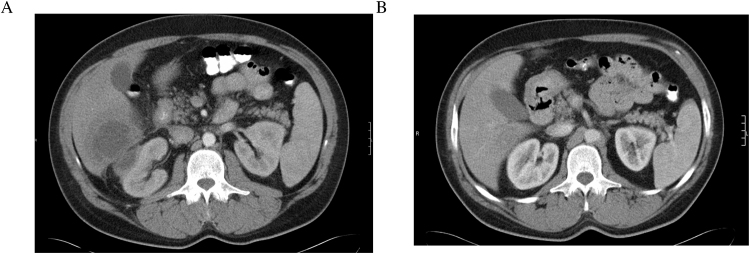
On admission, his temperature was 38 °C, the blood pressure 110/60 mmHg, the pulse 100 beats per minute, the respiratory rate was 14 breaths per minute and the oxygen saturation 97% while he was breathing ambient air. Physical examination revealed skin pallor, poor oral hygiene and gingivitis and right upper abdominal quadrant tenderness to palpation without rebound tenderness. The rest of the examination was normal. Initial laboratory findings demonstrated leukocytosis with neutrophilia (white blood cells 12,500/μL and neutrophils 10,000/μL with reference ranges 3800–10,500/μL and 1600–6500/μL, respectively), macrocytic anemia (hemoglobin 10.4 g/dL and mean corpuscular volume 104 fL with reference ranges 14–18 g/dL and 80–99 fL, respectively). The erythrocyte sedimentation rate was 113 mm in the first hour (reference range 1–15 mm/h) and the plasma C-reactive protein was 27.8 mg/dL (reference range 0.08-0.8 mg/dL). Liver function tests were almost normal with a gamma-glutamyl-transferase of 56 U/L (10–50 U/L). Serum amylase and urine analysis were normal. Ultrasonography of the upper abdomen demonstrated a hypoechoic hepatic lesion with distinct lobulated margins and irregular hyperechoic areas within the lesion. Abdominal computed tomography (CT) was performed after the oral and intravenous administration of contrast and confirmed the sonographic finding of a lobular lesion with a diameter of 10 cm inside the right hepatic lobe with a hypodense center and a contrast-enhancing peripheral rim; suggestive of a liver abscess [Fig. 1]. Moreover, a second abscess with a diameter of 2.5 cm was identified within the right peri-renal space without infiltration of the right kidney, as well as bowel wall thickening of the duodenum, the ascending colon and the terminal ileum.
Fig. 1.
Imaging Studies: (A) An abdominal CT scan with intravenous contrast showing a liver and a peri-renal abscess with a hypodense center and a contrast-enhancing peripheral rim. (B) An abdominal CT scan with intravenous contrast material was repeated after six-week of antimicrobial treatment and the intra-abdominal abscesses were not present.
Drainage of the liver abscess with a percutaneous catheter was performed and purulent fluid was drained and cultured. Lactococcus lactis ssp cremoris was isolated, and was sensitive to most antimicrobials, including penicillin. Ceftriaxone (1gram, once daily) and metronidazole (500 mg, three times daily) were intravenously administered for 2 weeks and the patient became afebrile on the 5th day. The drainage catheter was removed after a week, when the drained fluid was minimal. Blood and urine cultures were sterile. Examination of the stool did not yield any protozoa, enteropathogenic bacteria or Clostridium difficile. Serum antibodies and the analysis of the liver abscess aspirate were negative for Entamoeba histolytica. Transthoracic echocardiography was normal with no evidence of valvular vegetations. Further work up for macrocytic anemia was performed. The reticulocyte index was low (0.6%) and the blood smear showed anisocytosis, macrocytosis as well as hypersegmented neutrophils. Vitamin B12 level was low; 90 pg/mL (reference range 189–883 pg/mL) and the folate level was normal; 4.1 ng/mL (reference range 2.7–34 ng/mL). Antibodies against intrinsic factor and anti-parietal cell autoantibodies were positive (45.3 >25 units/mL and 1:160 respectively); indicative of pernicious anemia. Serum antibodies against Helicobacter pylori were negative. Endoscopic and histological investigation of the upper gastrointestinal tract revealed achlorhydria (pH 7-7.5) and atrophic gastritis. Colonoscopy showed diverticulosis of the sigmoid colon. Biopsies of the terminal ileum and ascending colon demonstrated edema and dilatation of the lymphatic vessels without signs of a specific disease. A dental exam and x-ray were performed because of the past history of odontalgia and showed severe periodontitis. Tests for antibodies to human immunodeficiency, hepatitis B and C viruses were negative. Cultures of the oral mucosa, gastric fluid and stool did not isolate L.lactis ssp cremoris.
After 2 weeks of intravenous antimicrobial treatment, the patient was discharged from the hospital on antimicrobial treatment with amoxicilin-clavulanic acid (1000/200 mg twice daily) for 4 more weeks as well as vitamin B12 to correct B12 deficiency. Two weeks after discharge, an abdominal CT was performed and revealed significant decrease of the intra-abdominal abscesses.
Discussion
Lactococcus lactis is a mesophilic and microaerophilic bacterium used widely in the fermentation of dairy products [14]. It is estimated that 1018 lactococci are annually consumed by humans [15]. Although L.lactis does not usually colonize the human mucosa, it is occasionally isolated from the oropharynx, intestines or vagina, particularly when unpasteurized dairy products have been consumed. Although L.lactis had not been considered a human pathogen, serious infections have been reported in almost any human body system (central nervous system, intra-abdominal or neck abscess, pneumonitis and empyema, endocarditis, urinary tract infection, cholangitis, catheter −related bacteremia, peritonitis, canaliculitis, septic arthitis, spondylodiscitis) the last two decades worldwide, regardless of patient age, gender and immune status. A Pubmed search demonstrated thirty-three cases of L.lactis-related infection, with L.lactis ssp cremoris identified in nineteen of these reports. Consumption of unpasteurized dairy products, presence of dental or periodontal or heart disease, immunosupression or damaged colonic mucosa were considered as potential risk factors for L.lactis infection.
Among the twenty-two cases of human infection by L.lactis ssp cremoris, 5 were liver abscess, 4 endocarditis, 3 peritonitis, 2 cerebral abscess, 2 neck abscess and a case of subdural empyema, canaliculitis, necrotizing pneumonia, purulent pleuritis, cholangitis and endocarditis complicated with spondylodiscitis were reported. Beta lactams were usually administered and disease resolution was achieved except in one case of endocarditis that led to death. Antimicrobial treatment for at least four weeks and percutaneous abscess drainage were used in all cases of liver abscess due to L.lactis ssp cremoris with complete recovery. Among the reported cases with L.lactis ssp cremoris liver abscess, ingestion of unpasteurized dairy products was not reported and neither immunodeficiency nor dental disease was present. However, two of these patients had colitis or diverticulitis, respectively. Liver abscess due to L.lactis subsp. lactis was also described in a patient with yogurt consumption and damaged colonic mucosa by bony fragment. In our case, the patient presented with a liver and a peri-renal abscesses but neither bacteremia nor endocarditis was documented. Percutaneous drainage of the liver abscess and six-week of antimicrobial treatment resulted in disease resolution for our patient.
To our knowledge, this is the first case report of multiple intra-abdominal abscesses due to L.lactis ssp cremoris. Even though the exact source of L.lactis was not found, we assume that severe periodontitis along with the digestion of fermented dairy products resulted in colonization of the gastrointestinal flora by L.lactis and subsequent bloodstream invasion.
In the present case, vitamin B12 deficiency secondary to achlorhydria was noted. Although vitamin B12 deficiency has been associated with apthous stomatitis [16], it is unknown whether periodontal disease is associated as well. Besides, a number of micronutrients such as vitamin A, vitamin C, folic acid, vitamin B12, iron and zinc influence the susceptibility of a host to infectious diseases as well as the course and outcome of these diseases [17]. Animal studies reported that vitamin B12 deficiency results in reduced macrophage migration and delayed phagocytosis [18]. Several infectious diseases were reported in patients with pernicious anemia such as salmonellosis, mucocutaneous candidiasis, syphilis, strongyloidiasis and infection by Neisseria meningitidis [19], [20], [21]. In the present case, impaired phagocytosis due to B12 deficiency might be associated with the severity of the disease caused by a less virulent microbe such as L.lactis ssp cremoris.
In conclusion, intra-abdominal abscesses due to L.lactis ssp cremoris are a rare clinical condition. Impaired barriers and immune defense mechanisms as in pernicious anemia, immunodeficiency, dental or periodontal disease, bowel disease along with the ingestion of fermented dairy products might be causes of severe infection by a less virulent bacterium like L.lactis ssp cremoris.
Consent
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or non-for-profit sectors.
Footnotes
Supplementary data associated with this article can be found, in the online version, at http://dx.doi.org/10.1016/j.idcr.2016.12.001.
Contributor Information
Konstantinos Fragkiadakis, Email: fragkiadakisk@hotmail.com.
Petros Ioannou, Email: petros_io@hotmail.com.
Emmanouil Barbounakis, Email: barbuman2003@yahoo.gr.
George Samonis, Email: samonis@med.uoc.gr.
Appendix A. Supplementary data
The following is Supplementary data to this article:
References
- 1.Ludwig W., Seewaldt E., Kilpper-Balz R., Schleifer K.H., Magrum L., Woese C.R. The phylogenetic position of Streptococcus and Enterococcus. J Gen Microbiol. 1985;131:543–551. doi: 10.1099/00221287-131-3-543. [DOI] [PubMed] [Google Scholar]
- 2.Pellizzer G., Benedetti P., Biavasco F., Manfrin V., Franzetti M., Scagnelli M. Bacterial endocarditis due to Lactococcus lactis subsp: cremoris: case report. Clin Microbiol Infect Off Publ Eur Soc Clin Microbiol Infect Dis. 1996;2:230–232. doi: 10.1016/s1198-743x(14)65148-x. [DOI] [PubMed] [Google Scholar]
- 3.Nakarai T., Morita K., Nojiri Y., Nei J., Kawamori Y. Liver abscess due to Lactococcus lactis cremoris. Pediatr Int Off J Japan Pediatr Soc. 2000;42:699–701. doi: 10.1046/j.1442-200x.2000.01294.x. [DOI] [PubMed] [Google Scholar]
- 4.Mat O., Rossi C., Beauwens R., Moenens F., Mestrez F., Muniz M.-C. Peritonitis due to Lactococcus cremoris in an automated peritoneal dialysis patient. Nephrol Dial Transplant Off Publ Eur Dial Transpl Assoc − Eur Ren Assoc. 2003;18:2690–2691. doi: 10.1093/ndt/gfg460. [DOI] [PubMed] [Google Scholar]
- 5.Koyuncu M., Acuner I.C., Uyar M. Deep neck infection due to Lactococcus lactis cremoris: a case report. Eur Arch Oto-Rhino-Laryngology Off J Eur Fed Oto-Rhino-Laryngological Soc Affil with Ger Soc Oto-Rhino-Laryngology − Head Neck Surg. 2005;262:719–721. doi: 10.1007/s00405-004-0889-7. [DOI] [PubMed] [Google Scholar]
- 6.Mofredj A., Beldjoudi S., Farouj N. Purulent pleurisy due to lactococcus lactis cremoris. Rev Mal Respir. 2006;23:485–486. doi: 10.1016/s0761-8425(06)71823-x. [DOI] [PubMed] [Google Scholar]
- 7.Leung D.Y.L., Kwong Y.Y.Y., Ma C.H., Wong W.M., Lam D.S.C. Canaliculitis associated with a combined infection of Lactococcus lactis cremoris and Eikenella corrodens. Jpn J Ophthalmol. 2006;50:284–285. doi: 10.1007/s10384-005-0318-5. [DOI] [PubMed] [Google Scholar]
- 8.Davies J., Burkitt M.D., Watson A. Ascending cholangitis presenting with Lactococcus lactis cremoris bacteraemia: a case report. J Med Case Rep. 2009;3:3. doi: 10.1186/1752-1947-3-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Topçu Y., Akıncı G., Bayram E., Hız M., Türkmen S. Brain abscess caused by Lactococcus lactis cremoris in a child. Eur J Pediatr. 2011;170:1603–1605. doi: 10.1007/s00431-011-1586-3. [DOI] [PubMed] [Google Scholar]
- 10.Buchelli-Ramirez H.L., Alvarez-Alvarez C., Rojo-Alba S., García-Clemente M., Cimadevilla-Suárez R., Pando-Sandoval A. Necrotising pneumonia caused by Lactococcus lactis cremoris. Int J Tuberc Lung Dis Off J Int Union Against Tuberc Lung Dis. 2013;17:565–567. doi: 10.5588/ijtld.12.0620. [DOI] [PubMed] [Google Scholar]
- 11.Saidane O., Mahmoud I., Saadi F., Souayah A., Zouari R. [Spondylodiscitis and Lactococcus cremoris endocarditis] Médecine Mal Infect. 2013;43:489–490. doi: 10.1016/j.medmal.2013.09.004. [DOI] [PubMed] [Google Scholar]
- 12.Inoue M., Saito A., Kon H., Uchida H., Koyama S., Haryu S. Subdural empyema due to Lactococcus lactis cremoris: case report. Neurol Med Chir (Tokyo) 2014;54:341–347. doi: 10.2176/nmc.cr2012-0440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Stabler S.P., Allen R.H. Vitamin B12 deficiency as a worldwide problem. Annu Rev Nutr. 2004;24:299–326. doi: 10.1146/annurev.nutr.24.012003.132440. [DOI] [PubMed] [Google Scholar]
- 14.Facklam R., Elliott J.A. Identification, classification, and clinical relevance of catalase-negative, gram-positive cocci, excluding the streptococci and enterococci. Clin Microbiol Rev. 1995;8:479–495. doi: 10.1128/cmr.8.4.479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bolotin A., Wincker P., Mauger S., Jaillon O., Malarme K., Weissenbach J. The complete genome sequence of the lactic acid bacterium lactococcus lactis ssp. lactis IL1403. Genome Res. 2001;11:731–753. doi: 10.1101/gr.169701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Weusten B.L., van de Wiel A. Aphthous ulcers and vitamin B12 deficiency. Neth J Med. 1998;53:172–175. doi: 10.1016/s0300-2977(98)00096-5. [DOI] [PubMed] [Google Scholar]
- 17.Bhaskaram P. Micronutrient malnutrition, infection, and immunity: an overview. Nutr Rev. 2002;60:S40–45. doi: 10.1301/00296640260130722. [DOI] [PubMed] [Google Scholar]
- 18.Thomaskutty K.G., Lee C.M. Interaction of nutrition and infection: macrophage activity in vitamin B12-deficient rats infected with Trypanosoma lewisi. J Natl Med Assoc. 1987;79:441–446. [PMC free article] [PubMed] [Google Scholar]
- 19.Stockbruegger R.W., Cotton P.B., Menon G.G., Beilby J.O., Bartholomew B.A., Hill M.J. Pernicious anaemia, intragastric bacterial overgrowth, and possible consequences. Scand J Gastroenterol. 1984;19:355–364. [PubMed] [Google Scholar]
- 20.Blizzard R.M., Gibbs J.H. Candidiasis: studies pertaining to its association with endocrinopathies and pernicious anemia. Pediatrics. 1968;42:231–237. [PubMed] [Google Scholar]
- 21.Sartin J.S. Neisseria sicca meningitis in a woman with nascent pernicious anemia. Am J Med. 2000;109:175–176. doi: 10.1016/s0002-9343(00)00438-1. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.