Abstract
In an attempt to develop Complementary and Alternative Medicine (CAM) for the treatment of diabetes and related complications, the antidiabetic potential of the mature unripe fruits of Coccinia grandis (CGF) was evaluated. Oxidative stress and glycation plays an important role in manifesting of diabetes and vascular complications. Agents with antioxidant and antiglycation properties may retard these pathological alterations. In this study, the edible plant Coccinia grandis was assessed for in vitro estimation of antioxidant and antiglycation potential and its insulinotrophic properties in RINm5F cells. Antioxidant activity was evaluated as DPPH (1,1-diphenyl-2-picrylhydrazyl), hydrogen peroxide and superoxide anion scavenging activities, whereas the protein glycation inhibitory potential was evaluated using in vitro albumin-fructose glycation model. Glycation inhibition was estimated by different biochemical parameters viz. fructosamine, protein carbonyl group and protein aggregation using thioflavin T fluorescence. C. grandis extract exerted a dose dependent radical scavenging activity and exhibited a significant antiglycation potential. The extract also showed a significant insulinotrophic property with 1.28 and 1.71-fold increase in insulin release when compared to control at 0.25 and 0.50 mg/mL, respectively. These data suggest the possible antidiabetic role of CGF extract, presumably by its antioxidant, antiglycation and insulin secretory effects. Present findings provide experimental evidence that the fruits of C. grandis have potential antidiabetic activity which might be used as a functional food and safe remedy for the treatment of diabetes and associated complications. This study also revealed that the plant can be a promising source for development of natural antiglycating agents and novel insulin secretagogues.
Keywords: antioxidant, polyphenols, Coccinia grandis, advanced glycation end products (AGEs), antiglycation, RINm5F cells
Graphical abstract
1. Introduction
The total predicted increase in numbers of people with diabetes from 2012 to 2030 is about 180 million (371.33 million to 551.87 million, respectively), an astonishing increase of 48% from 2012 at an annual growth of 2.7%, which is twice the annual growth of the total world adult population. Forty-two per cent of the anticipated absolute global increase of 180 million people with diabetes is projected to occur in India and China alone. According to International Diabetes Federation (IDF), nearly 5 million people died due to diabetes and its related complications in 2012.1 Therefore, novel concepts in the management of diabetes have aroused a curiosity among researchers throughout the world. In countries such as India and China, use of herbal medicines is a very common practice from ancient time, and herbal medicines are considered to be much safer and less expensive therapeutic strategies for the treatment of various diseases. A proper scientific investigation of traditional herbal remedies can provide valuable leads for the development of alternative drugs and strategies for the management of diabetes.2 Role of herbs in the management and control of diabetes has emerged fast over the years with the discovery of hypoglycemic effect of Bitter Melon (Mormodica charantia).3, 4
The human body is constantly subjected to significant oxidative stress as a result of an imbalance between antioxidative protective systems and the formation of reactive oxygen species (ROS) including free radicals. Experimental investigations as well as clinical and epidemiological findings has provided evidence supporting the role of reactive oxygen species (ROS) such as singlet oxygen, superoxide anions (O2•−), hydrogen peroxide (H2O2), and hydroxyl radical (OH•) in the etiology of diabetes, cardiovascular diseases, aging etc.5 Medicinal plants and herbs are a promising and diverse source of natural antioxidants. Many plant extracts and plant products have been identified as good protectors against the free radicals by triggering antioxidant gene expression. For that account natural antioxidants from plant sources have been viewed as promising therapeutic drugs.6
Advanced glycation end-products (AGEs) are the final products derived from the Maillard reaction, which is a non-enzymatic glycation of free amino groups by sugars and aldehydes. AGE formation begins under hyperglycemic or oxidative stress conditions and is characterized by conversion of reversible Schiff-base adducts to covalently bound Amadori products, which undergo further rearrangements that terminate in the formation of irreversibly bound compounds known as AGEs.7, 8 The interaction of AGEs with receptors for AGEs (RAGE) directly activates multiple intracellular signaling, gene expression, and the secretory pro-inflammatory molecules accompanied by increasing free radicals that contribute towards pathologic complications related to diabetes.9 Aminoguanidine (AG), a well-known antiglycating agent, inhibits the formation of AGEs and prevents the development of diabetic complications in animal models of diabetes. However, previous studies have shown that AG has serious adverse effects in diabetes patients, such as myocardial infarction, congestive heart failure, arterial fibrillation, anemia and gastrointestinal disturbance.10 Therefore, attention has been focused on the antioxidant and antiglycation properties of phytochemicals present in fruits and vegetables due to their lower toxicity and fewer side effects than synthetic drugs. Natural antioxidants and AGE inhibitors may have promising therapeutic potential in delaying the onset and preventing diabetic complications. Many studies have shown the beneficial effect of plant-based diets for the inhibition of protein glycation in vitro and in vivo.
Diabetes mellitus is a chronic metabolic disorder characterized by hyperglycemia. It is believed that absolute or relative insulin deficiency due to the inadequate beta cell mass is the cause of hyperglycemia. Type 1 diabetes results from the destruction of pancreatic beta-cells by beta-cell-specific autoimmune responses, and type 2 diabetes results from the progressive loss of beta-cell mass and function. Therefore, strategies that preserve or restore beta-cell mass and function are logical therapeutic approaches for the treatment of diabetes.11 In type I diabetes, or insulin dependent DM (IDDM), the body has little or no insulin secretory capacity and depends on exogenous insulin to prevent metabolic disorders and death. While conventional treatments such as sulfonylureas, metformin and thiazolidinedones are effective, they have several limitations, including adverse side effects, secondary failure or the inability to halt further loss of insulin secretory capacity. Newer and cheaper medications are therefore needed. One solution is to use herbal remedies, which appear to be widely used with relatively few documented side effects. In this continuing search for new antidiabetic compounds from plants, the present study investigated the antioxidant, antiglycation and insulinotrophic properties of Coccinia grandis (L.) fruits in vitro and its possible role in prevention of diabetic complications.12
Coccinia grandis (L.) Voigt belongs to the Cucurbitaceae family and grows abundantly in India. It is a climbing perennial herb, growing throughout India especially in warmer and humid climatic conditions. It is widely used in traditional treatment of diabetes.13 The fruits are used for culinary purposes as a vegetable. Scientific investigations have supported the efficacy of leaf and root extracts in amelioration of diabetic complications.14, 15 C. indica leaves have been reported to stimulate insulin secretion in diabetic rats.16 To the best of our knowledge, there were no reports in the scientific literature on antiglycation and insulin secreting activity of mature unripe Coccinia grandis fruits (CGF). Hence, this study was aimed at investigating the antioxidant and antiglycation effect of CGF in vitro as well as to examine the effect of CGF on insulin secretion and its cytoprotective effect using insulin-secreting Rat insulinoma clone m5F (RINm5F) cells.

Kingdom: Plantae
Order: Cucurbitales
Family: Cucurbitaceae
Genus: Coccinia
Species: C. grandis
Binomial name: C. grandis (L) J. Voigt
2. Materials and methods
2.1. Cell line, chemicals and reagents
Roswell Park Memorial Institute (RPMI-1640) was procured from Gibco and Rat insulinoma clone m5F (RINm5F) Rattus norvegicus (rat) cell line was obtained from the American Type Culture Collection (ATCC). Bovine serum albumin (BSA), aminoguanidine hydrochloride (AG), nitroblue tetrazolium (NBT), 1-deoxy-1-morpholino-d-fructose (1-DMF), guanidine hydrochloride, thioflavin T reagent [4-(3,6-dimethyl-1,3-benzothiazol-3-ium-2-yl)-N,N-dimethylaniline chloride], 5,5′-dithiobis(2-nitrobenzoic acid) (DTNB) and l-cysteine were purchased from Sigma-Aldrich Co. (St. Louis, MO, USA). Fructose, potassium dihydrogen orthophosophate, DPPH, dipotassium hydrogen phosphate, sodium azide, trichloroacetic acid, hydrochloric acid, urea, ethyl alcohol, ethyl acetate, sodium chloride were procured from Qualigens Pvt. Ltd., Mumbai, India. All other chemicals and reagents used were of analytical grade. All other chemicals and solvents were of analytical reagent grade. Water was purified by Milli Q Water Purification System, Millipore, USA.
2.2. Plant materials
Coccinia grandis (L.) Voigt mature unripe whole fruits were collected from Southern part of India (Kancheepuram District, Tamil Nadu, India) during the month of July 2015 and the pharmacognostic authentication was done by Department of Plant Sciences, University of Madras, Chennai-600 025. The fruits were cut in to small slices, air dried under shade, pulverized to fine powder using a laboratory scale cutting mill.
2.3. Extraction procedure
The extract was prepared using the methods described by Olgica et al.17 with minor modifications. Methanol extract was chosen because it has been reported to be the best solvents for the extraction of antioxidant compounds.17 The dried, ground plant material was extracted by maceration with methanol. Fifty g of plant material was soaked with 500 mL of the solvent for 24 h at room temperature in a shaker. The sample was filtered through filter paper. The residue from the filtration was extracted again, twice, using the same procedure. The filtrates obtained were combined and then evaporated to dryness using a rotary evaporator at 40 °C. The obtained extract was stored in sterile sample tube at -20 °C.
2.4. Phytochemical analysis of CGF extract
2.4.1. Qualitative phytochemical analyses of CGF
Preliminary phytochemical analyses of the CGF extract were done using standard procedure of Sofowora18 and Harbourne.19
2.4.2. Determination of total phenolics
The amount of total phenolics in CGF extract was determined by the method of Singleton et al.20 with minor modifications. One hundred μL of crude extract (20 μg/mL) was mixed with 0.2 mL of Folin-Ciocalteu reagent, 2 mL of distilled water and 1 mL of 15% Na2CO3. The mixture was measured at 765 nm using UV-Visible spectrophotometer (T60U, PG Instruments Limited, UK) after 2 h at room temperature. Gallic acid was used as a standard and the total phenolics were expressed as mg of gallic acid equivalent (mg GAE) per g of extract (dry weight).
2.4.3. Determination of total flavonoids
Total flavonoid content was determined using the method of Chang et al.21 with some modifications using quercetin as the standard. A calibration curve of quercetin was prepared in the range of 0–200 μg/mL. Briefly, extract (0.5 mL) and standard (0.5 mL) were placed in different test tubes and to each 10% aluminum chloride (0.1 mL), 1 M potassium acetate (0.1 mL), 80% methanol (1.5 mL) and distilled water (2.8 mL) were added and mixed. A blank was prepared in the same manner where 0.5 mL of distilled water was used instead of the sample or standard, and the amount of aluminum chloride was also replaced by distilled water. All tubes were incubated at room temperature for 30 min. The absorbance was taken at 415 nm using UV-Visible spectrophotometer. The concentration of flavonoid was expressed as mg quercetin equivalent (QE) per gram of extract.
2.4.4. Determination of saponins and dietary fiber content
Saponins in CGF extract was determined by standard method as described by Anhawange et al.22 The saponins were calculated as mg per g of extract (dry weight). The dietary fiber content was analyzed by the enzymatic-gravimetric method of Asp et al.23 and expressed as g per 100 g of plant material.
2.5. Antioxidant and free radical scavenging activities of CGF (cell free systems)
2.5.1. DPPH free radical scavenging activity
The DPPH assay was performed as described by Koleva et al.24 Approximately 10 μL of different concentrations (12.5–250 μg/mL) of test sample solutions were added to 190 μL DPPH (150 μM) in methanol solution. The solutions were later vortex mixed and incubated for 20 min at 37 °C. The solvent alone was considered “blank.” The decrease in absorbance of test mixtures (due to quenching of DPPH free radicals) was determined at 517 nm using UV-Visible spectrophotometer; Butylated hydroxyanisole (BHA) (12.5–250 μg/mL) was used as the standard.
2.5.2. Assay of hydrogen peroxide (H2O2) scavenging activity
The ability of the extract to scavenge hydrogen peroxide was determined according to the method of Ruch et al.25 A solution of hydrogen peroxide (2 mM) was prepared in phosphate buffer (pH 7.4). Hydrogen peroxide concentration was determined spectrophotometrically from absorption at 230 nm. Extract samples (12.5–250 μg/mL) were added to hydrogen peroxide solution (0.6 mL). Absorbance of hydrogen peroxide at 230 nm was determined after 10 min against a blank solution containing phosphate buffer without hydrogen peroxide using UV–Visible spectrophotometer. The percentage of scavenging of hydrogen peroxide of both the extract and standard compound Butylated hydroxyanisole (BHA) was determined using the following formula:
where A0 is absorbance of the Control, and A1 is the absorbance in the presence of the Sample or Standard (BHA).
2.5.3. Assay of superoxide radical-scavenging activity
Measurement of superoxide anion-scavenging activity of the extract was based on the method described by Liu et al.26 with minor modifications. The reaction mixture consisted of 1.0 mL of Nitroblue tetrazolium (NBT), 1.0 ml of NADH and 0.5 mL of an appropriately diluted sample (12.5–250 μg/mL). The reaction was initiated by addition of 100 μl of Phenazine methosulfate (PMS) to the mixture. The tubes were incubated at ambient temperature for 5 min and the absorbance was measured at 560 nm in a Spectrophotometer. The absorbance of the control was determined by replacing the sample with methanol. BHA was used as positive control. Decreased absorbance of the reaction mixture indicated increased superoxide anion scavenging activity. The percentage inhibition of superoxide anion generation was calculated using the following formula:
where A0 is absorbance of the Control, and A1 is absorbance of the Sample or Standard (BHA).
2.6. In vitro antiglycation assay with BSA-fructose model
Glycated BSA was done according to the method of Sharma et al.27 with minor modifications. Briefly, 10 mg/mL BSA (0.50 mL) was incubated with 0.46 mL of 500 mM fructose in 100 mM phosphate buffered-saline (pH 7.4) containing 0.02% sodium azide at 37 °C for 4 weeks. Before incubation, 0.04 mL of different concentrations of CGF extract (0.0625, 0.125, 0.25 and 0.50 mg/mL) and Aminoguanidine (AG) (final concentration: 0.50 mg/mL, positive control) were added into the reaction mixtures. The formation of fluorescent AGEs was measured by using a spectrofluorometer. The fluorescent intensity was measured at an excitation wavelength of 355 nm and emission wavelength of 460 nm. The percentage of fluorescent AGE formation was calculated as follows:
where FC and FCB were the fluorescent intensity of control with fructose and blank of control without fructose, FS and FSB were the fluorescent intensity of sample with fructose and blank of sample without fructose.
2.6.1. Determination of fructosamine
The concentration of fructosamine, the Amadori product in glycated albumin samples and controls (after incubation for 7, 14, 21, and 28 days) was determined using a nitroblue tetrazolium (NBT) assay according to Ardestani and Yazdanparast.28 Briefly, glycated BSA (10 μL) was incubated with 0.5 mM NBT (90 μL) in 100 mM carbonate buffer, pH 10.4 at 37 °C. The absorbance was recorded at 530 nm. The level of fructosamine was calculated by using the different absorption at the time point of 10 and 15 min. The level of fructosamine was calculated from a standard curve prepared using 1-deoxy-1-morpholinofructose (1-DMF).
2.6.2. Determination of protein carbonyl contents
The protein carbonyl contents were measured according to the method of Meeprom et al.29 with minor modifications. In brief, glycated BSA (0.10 mL) was incubated with 0.40 mL of 10 mM 2,4-dinitrophenylhydrazine (DNPH) in 2.5 M HCl at room temperature for 60 min. Subsequently, glycated BSA was precipitated by 0.50 mL of 20% (w/v) trichloroacetic acid (TCA), left on ice for 5 min and centrifuged at 10,000 g at 4 °C for 10 min. The pellet was washed three times using 1:1 (v/v) ethanol:ethyl acetate mixture (1 mL). The final pellet was dissolved in 6 M guanidine hydrochloride (0.25 mL). The absorbance was recorded at 370 nm. The level of protein carbonyl contents was calculated by using an absorption coefficient of 22,000 M−1 cm−1. The results were expressed as nmol carbonyls/mg protein.
2.6.3. Determination of protein aggregation
Amyloid cross β-structure, a common marker for protein aggregation was measured by using thioflavin T, a marker for amyloid cross β structure according to Tupe and Agte with minor modifications.30 Thioflavin (32 mM) was dissolved in glycine- NaOH buffer (50 mM, pH 8.5). Glycated samples (100 μL, as described in 2.6) were incubated with thioflavin T solution (3 mL) for 1 h, and the fluorescence was measured at an excitation and emission wavelength of 435 nm and 485 nm, respectively (slit, 10 nm), with appropriate blanks devoid of thioflavin T. The results were expressed as arbitrary units (AU).
2.7. In vitro cytoprotective effect of CGF extract
RINm5F cells were cultured in RPMI-1640 medium supplemented with 10% fetal bovine serum, 1% antibiotic-antimycotic solution and incubated at 37 °C in a humidified atmosphere containing 5% CO2. RINm5F cells (2 × 105) were incubated with CGF extract (Concentration: 0.0625 and 0.50 mg/mL) for 24 h. After the incubation period, the medium was removed. Cells were treated with alloxan at 9 mM (which caused about 50% of cell death) for 1 h.31 At the end of experiment, cell viability was assessed by MTT assay as previously described.32
2.8. Determination of the effect of CGF extract on insulin secretion
Insulin secretion by RINm5F cells was determined by the method of Persaud et al.33 The RINm5F insulinoma cells (1 × 105 cells/mL) were suspended in RPMI-1640 media supplemented with 10% FBS, 10 mM HEPES and 1 mM sodium pyruvate (200 μL) into 96-well plates. Cells were incubated at 37 °C in a 5% CO2 incubator for 48 h, after which the medium was exchanged for a glucose-free Krebs-Ringer bicarbonate (KRB) buffer (135 mM NaCl, 3.6 mM KCl, 5 mM NaHCO3, 0.5 mM NaH2PO4, 0.5 mM MgCl2 and 1.5 mM CaCl2), pH 7.4 supplemented with 1 mg/ml BSA and 10 mM HEPES for a further 2 h of incubation. The medium was subsequently replaced with 100 μL of glucose-free KRB containing plant extract (0.0625 and 0.50 mg/mL) glibenclamide (0.1–10 μM) or plain medium, and incubated at 37 °C in a 5% CO2 for 1 h. Glibenclamide was dissolved in DMSO and further diluted with Kreb's Ringer buffer. The final concentration of DMSO (0.5% w/v) did not affect RINm5F cell viability. Aliquots (10 μL) were removed from each well, centrifuged (2000 rpm for 5 min, at 4 °C), and assayed for insulin with Mercodia Rat Insulin ELISA kit as per Manufacturer's protocol. Briefly, samples (10 μL) were mixed with 100 μL enzyme conjugate solution and incubated for 2 h on a shaker at 37 °C. The plate was washed thrice with buffer to remove unbound enzyme labeled antibody, mixed with 200 μL of 3,3′,5,5′-tetramethylbenzidine (TMB) and incubated for 15 min at room temperature. The reaction was stopped with 50 μL of 0.5 M sulphuric acid and the absorbance was measured at 450 nm using Microplate Reader (BioTek Instruments, Inc.). The insulin content of supernatants was determined using the following formula:34
2.9. Data analysis
All experiments were performed in triplicate. The statistical analyses of the results were evaluated by using analysis of variance (ANOVA) followed by Duncan's post hoc analysis. p-Values less than 0.05 were set as the level of significance. All analyses were undertaken in SPSS 16 (Statistical Package for Social Science). Data are presented as the mean ± standard deviation of mean (SD).
3. Results
3.1. Phytochemical analysis of CGF
Phytochemical screening of CGF extract revealed the presence of flavonoids, alkaloids, glycosides, saponins, steroids, terpenoids, tannins and phenolic compounds (Table 1). It is well known that phenolic compounds belong to the bioactive components of plant products and have good health-promoting activities. Saponins are the glycosidic compounds found in most of the plants and have been reported to possess anticarcinogenic and antifungal activity. In this study, the total phenolic content, flavoniod contents, saponins and dietary fiber contents were detected in CGF extract, and the results are shown in Table 2.
Table 1.
Preliminary phytochemical screening of CGF extract.
| S.No. | Phytochemical constituent | Test | Inference |
|---|---|---|---|
| 1. | Alkaloids | Wagner's Test Mayer's Test |
a |
| 2. | Carbohydrates | Molisch's Test Fehling's Test |
a |
| 3. | Flavonoids | Shinoda Test | a |
| 4. | Glycosides | Borntrager's Test Legal's Test Keller–Killiani Test |
a |
| 5. | Saponins | Froth Test | a |
| 6. | Steroids | Salkowski reaction | a |
| 7. | Tannins and Phenolic compounds | Ferric Chloride Test Lead Acetate Test Nitric Acid Test |
a |
| 8. | Terpenoids | Libermann–Burchard's test | a |
Indicates presence of phytochemical constituents.
Table 2.
Contents of total phenolics, flavonoids, saponins and Dietary Fiber in methanol extract of Coccinia grandis fruits (CGF).
| Parameter | Concentration |
|---|---|
| Yield of extract (%) | 7.68 ± 0.58 |
| Total Phenolic Contentsa | 15.47 ± 1.12 |
| Total Flavonoid Contentsb | 6.82 ± 0.67 |
| Total Saponinsc | 0.12 ± 0.02 |
| Soluble Dietary Fiberd | 11.65 ± 0.93 |
| Insoluble Dietary Fiberd | 38.42 ± 1.56 |
Each value is expressed as mean ± SD from minimum of three independent experiments.
Data expressed as milligram of gallic acid equivalent (mg GAE) per g of extract (dry weight).
Data expressed as milligram of quercetin equivalent (mg QE) per g of extract (dry weight).
Data expressed as milligram per g of extract (fresh weight).
Data expressed as gram per 100 g of plant material.
3.2. Antioxidant and free radical scavenging activities of CGF
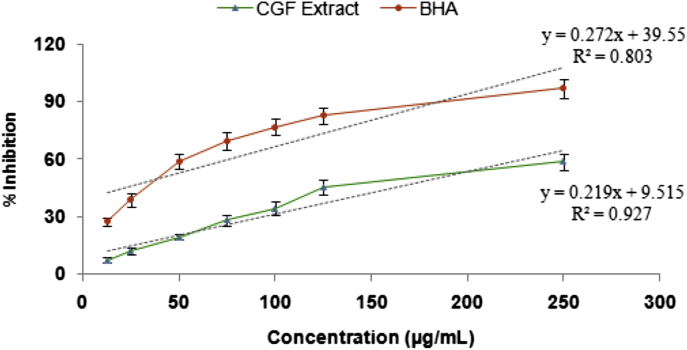
The DPPH radicals scavenging activity demonstrate the effect of CGF extract as antioxidant through their hydrogen donating ability, which reduces the stable violet DPPH radical to the yellow DPPH-H. A high percentage of radical scavenging indicated a strong antioxidant activity in the tested sample. The extracts showed concentration dependent antiradical activity. Furthermore, the extract which contained the considerable amount of total phenolics and flavonoids, showed a significant effect in inhibiting DPPH, reaching 60% at a concentration of 250 μg/mL. The IC50 value (the extract concentration with 50% scavenging activity) of scavenging activities on DPPH radical was to be 0.165 mg/mL (Fig. 1).
Fig. 1.
DPPH free radical scavenging activity of methanol extract of CGF at different concentrations. BHA was used as a reference antioxidant. Values are expressed as mean ± standard deviation of three independent experiments.
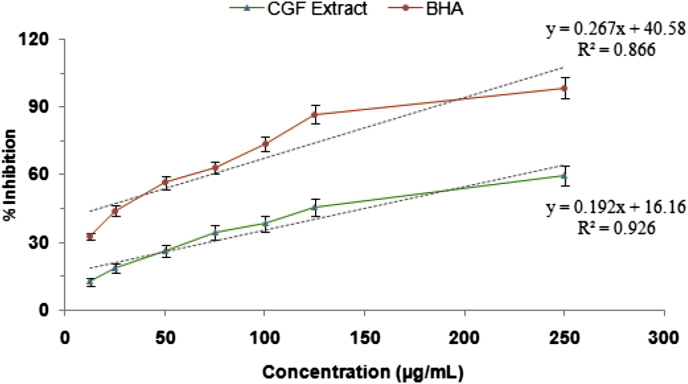
Hydrogen peroxide scavenging activity of the CGF extract was tested and it was increased steadily with the increasing concentrations. The extract significantly scavenged up to 59.64 ± 4.22% hydrogen peroxide at a concentration of 250 μg/mL. The ability of the CGF extract and BHA (a reference compound) to scavenge H2O2 was compared and presented in Fig. 2.
Fig. 2.
Hydrogen peroxide scavenging activity of methanol extract of CGF at different concentrations. BHA was used as a reference antioxidant. Values are expressed as mean ± standard deviation of three independent experiments.
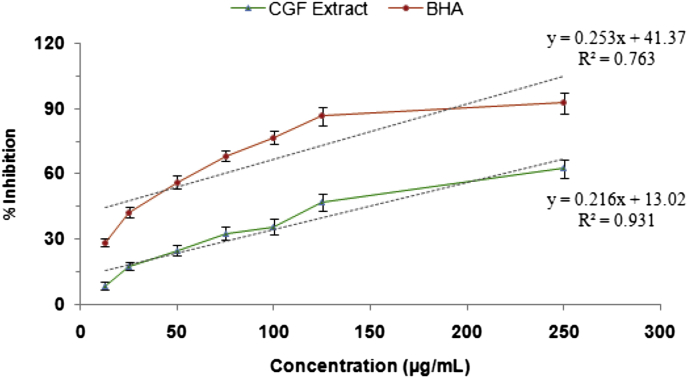
Superoxide anion radical is an initial radical and plays an important role in the formation of other reactive oxygen-species such as hydroxyl radical, hydrogen peroxide or singlet oxygen in living systems. Fig. 3 shows the superoxide anion radical scavenging activity of CGF extract and BHA. Significant superoxide anion radical scavenging activities were evident at all the tested concentrations of the CGF. The scavenging activity was increased with increasing concentrations.
Fig. 3.
Superoxide radical scavenging activity of methanol extract of CGF at different concentrations. BHA was used as a reference antioxidant. Values are expressed as mean ± standard deviation of three independent experiments.
3.3. Effect of CGF extract on the formation of fluorescence AGEs
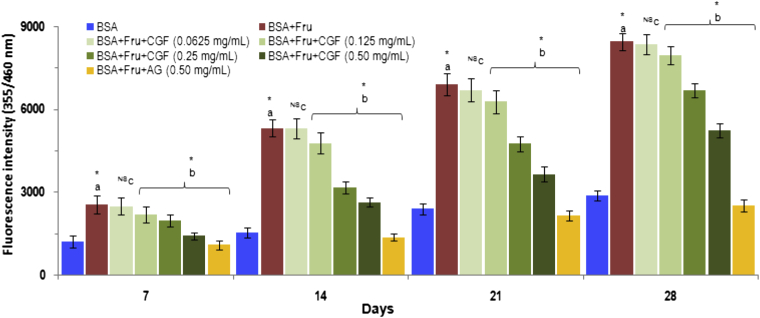
Advanced Glycation End-Products (AGEs) are signaling proteins associated with several vascular and neurological complications in diabetic patients. In the present study, the effect of CGF extract on the formation of fluorescent AGEs in glycation model systems composed by BSA and fructose treated at 37 °C for 4 weeks was evaluated. As shown in Fig. 4, the formation of AGEs was observed weekly by the measurement of increasing fluorescent intensity. The significant increase in fluorescent intensity in BSA incubated with fructose was seen during 4 weeks of the incubation. The results demonstrated that addition of CGF extract into BSA-fructose in vitro model significantly reduced the formation of fluorescent AGE in a concentration-dependent manner.
Fig. 4.
The effect of CGF and aminoguanidine on fluorescent AGE formation in BSA-fructose system. Data are expressed as mean ± SD of results obtained from minimum of three independent experiments. a∗ represents significant increase at p < 0.05 when compared to BSA values while b∗ represents significant decrease at p < 0.05 when compared to BSA + Fru values. cNS represents no significant difference between BSA + Fru and BSA + Fru + CGF. BSA: Bovine serum albumin; Fru: Fructose; CGF: Coccinia grandis (L.) Extract; AG: aminoguanidine.
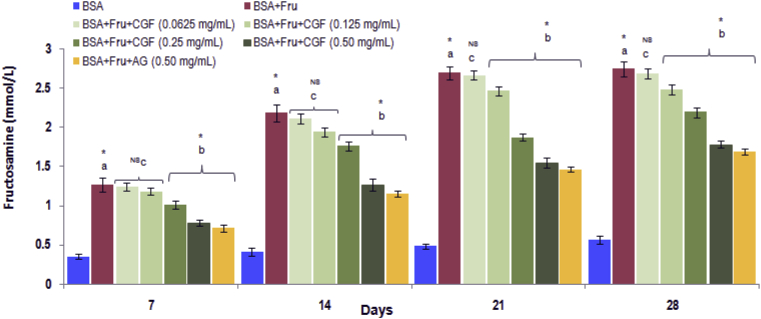
3.3.1. Effect of CGF extract on fructosamine level
The levels of fructosamine in glycated samples in the presence and absence of CGF extract are shown in Fig. 5. Maximum fructosamine was formed in glycated albumin (positive control) as compared with the negative control. Treatment of glycated samples with CGF extract showed a significant reduction of fructosamine level (p < 0.05). At day 28, the percentage reduction of fructosamine level by CGF (0.50 mg/mL) was found to be 35.03% while AG at the same concentration of had the reduction of 38.32%.
Fig. 5.
The effect of CGF and aminoguanidine on the levels fructosamine in BSA-fructose system. Data are expressed as mean ± SD of results obtained from minimum of three independent experiments. a∗ represents significant increase at p < 0.05 when compared to BSA values while b∗ represents significant decrease at p < 0.05 when compared to BSA + Fru values. cNS represents no significant difference between BSA + Fru and BSA + Fru + CGF. BSA: Bovine serum albumin; Fru: Fructose; CGF: Coccinia grandis (L.) Extract; AG: aminoguanidine.
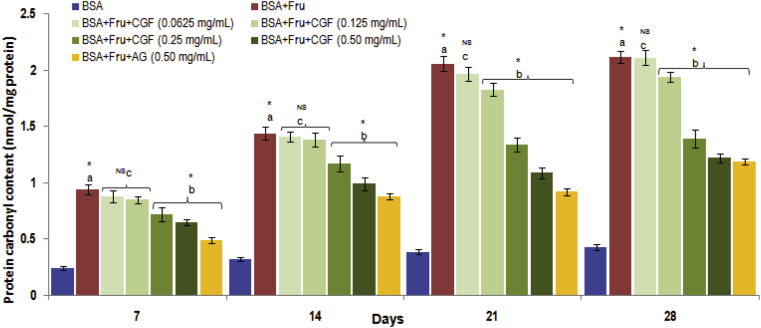
3.3.2. The effect of CGF on protein oxidation
To access the protein oxidation mediated by glycation process, the levels of protein carbonyl content were measured for the duration of 4 weeks. As shown in Fig. 6, the carbonyl content of fructose-glycated BSA was significantly increased, whereas CGF treatment significantly suppressed an increase in protein carbonyl content of fructose-glycated BSA. When comparing with fructose-glycated BSA at week 4, the percentage reduction of carbonyl content by CGF extract (0.50 mg/mL) was found to be 42.45%.
Fig. 6.
The effect of CGF and aminoguanidine on the levels on the level protein carbonyl in BSA-fructose system. Data are expressed as mean ± SD of results obtained from minimum of three independent experiments. a∗ represents significant increase at p < 0.05 when compared to BSA values while b∗ represents significant decrease at p < 0.05 when compared to BSA + Fru values. cNS represents no significant difference between BSA + Fru and BSA + Fru + CGF. BSA: Bovine serum albumin; Fru: Fructose; CGF: Coccinia grandis (L.) Extract; AG: aminoguanidine.
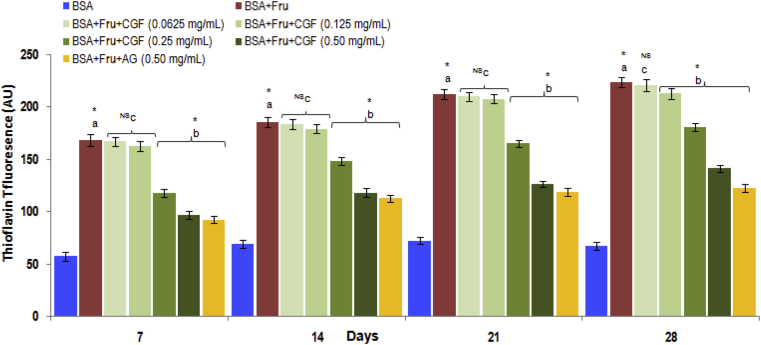
3.3.3. Effect of CGF on protein aggregation
Glycation of albumin induces the formation of amyloid fibrils which contain a cross beta structure. The property of amyloid fibril binding to specific dye thioflavin T, was used to quantify the level of amyloid in glycated albumin samples. The results are depicted in Fig. 7. It was observed that CGF extract showed significant inhibition to amyloid aggregation (36.86% at 0.50 mg/mL concentration on 28th day), indicating their potential to prevent conformation changes in albumin during glycation. The reason behind decrease in β-amyloid formation may be the low-molecular-weight bioactive compounds such as polyphenols, phenolic acids or flavonoids which have anti-amyloidogenic effects and are likely to be present in the extract.
Fig. 7.
The effect of CGF and aminoguanidine on the levels on the level on the level protein aggregation measured as thioflavin T fluorescence in BSA-fructose system. Data are expressed as mean ± SD of results obtained from minimum of three independent experiments. a∗ represents significant increase at p < 0.05 when compared to BSA values while b∗ represents significant decrease at p < 0.05 when compared to BSA + Fru values. cNS represents no significant difference between BSA + Fru and BSA + Fru + CGF. BSA: Bovine serum albumin; Fru: Fructose; CGF: Coccinia grandis (L.) Extract; AG: aminoguanidine.
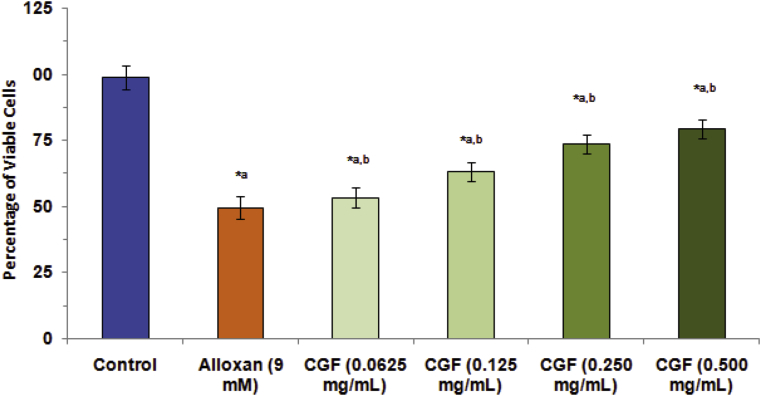
3.4. In vitro cytoprotective effect of CGF extract
The protective effect of CGF extract against alloxan-induced cell death was studied by MTT assay. The half maximal inhibitory concentration (9 mM) of alloxan on cell viability was used for studying the cytoprotective effect of the extract. It was found that alloxan, a free radical producing agent, decreased the number of RINm5F cells. The results of MTT assay (Fig. 8) showed that pretreatments of CGF extract provided a significant protection from alloxan-induced RINm5F cell damage. The extract significantly increased the cell viability to 79.45 ± 3.68% at the concentration of 0.50 mg/mL, when compared to alloxan alone treated cells. The concentration of the extract was limited at 0.50 mg/mL to avoid toxicity on cell viability that may be caused by the extract itself.
Fig. 8.
Cytoprotective effect of CGF extract measured as % cell viability by MTT assay. Pre-treatment with CGF extract was performed on alloxan-induced RINm5F cell damage. The IC50 of alloxan (9 mM) was used to induce cell death after pre-treatment with CGF extract. In panel, Control is normal cells whereas Alloxan is cells treated with alloxan alone. In panel CGFs cells were treated with CGF extract at concentrations of 62.5, 125, 250 and 500 μg/mL, respectively for 24 h before exposure of alloxan. Values are expressed as mean ± SD of results obtained from minimum of three independent experiments. *ap < 0.05 statistically significant difference compared to control. *b p < 0.05 statistically significant difference compared to alloxan.
3.5. Effect of CGF extract on insulin secretion
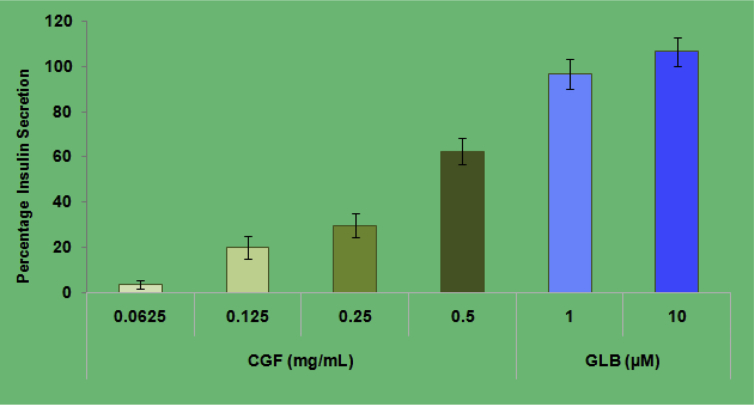
In the present study, RINm5F cells were used to evaluate the effect of total methanol extract from C. grandis (CGF) on insulin secretion in vitro and the data were shown in Fig. 9. A concentration dependent increase in insulin secretion was observed for CGF, with significant (p < 0.05) increase from 1.193 ± 0.18 μg/L to 1.98 ± 0.24 μg/L (2.84 ± 1.4% to 70.69 ± 8.7%) from concentrations of 62.5 μg/ml to 500 μg/ml. Glibenclamide also significantly (p < 0.05) increased insulin secretion in a concentration dependent manner.
Fig. 9.
Insulin secreted by RIN-m5F pancreatic cells (A) expressed as percentage of untreated control cells ± SD of results obtained from minimum of three independent experiments to the CGF extract and glibenclamide (positive control) in glucose free medium.
4. Discussion
Oxidation of DNA, lipids and proteins by ROS play an important role in diabetes, cardiovascular disease, cancer, immune and inflammatory disorders and many other diseases related to the aging process.35 Diets biased towards plant-based components are known to provide high amounts of antioxidant phytochemicals, which offer protection against reactive oxygen species (ROS)-induced cellular damage.36 Phytochemical screening is of paramount importance in identifying new source of therapeutically valuable compounds having medicinal significance, to make the best and judicious use of available natural wealth.37 This study has focused on both the antioxidant and antidiabetic effect of C. grandis. Screening of the CGF extract revealed the presence of phytochemicals such as phenols, tannins, flavonoids, saponins, steroids, and alkaloids. The phenolic compounds are one of the largest and most ubiquitous groups of plant metabolites and attracted a great attention in relation to their potential for beneficial effects on health. Thus, the antioxidant and hypoglycemic activity of the CGF extract as recorded in this study might be attributed to the presence of phenolic compounds and flavonoids.
The dietary fiber content of CGF extract was analyzed by the enzymatic-gravimetric method and the extract showed good amounts of both soluble (11.65 ± 0.93 mg/g) and insoluble fibers (38.42 ± 1.56 mg/g). Oral administration of pectin isolated from C. grandis fruits was shown to have a significant hypoglycemic effect in normal rats. Pectin is a soluble fiber that affects blood glucose levels either by decreasing the transit time or indirectly through the production of short chain fatty acids (SCFAs). Acetate, propionate, and butyrate are the commonly produced SCFAs obtained by anaerobic fermentation of dietary fiber components by the microflora in the large intestine. Thus, the high dietary fiber content of CGF extract recorded in the present study correlate the beneficial effects of CGF in the diabetic condition.38, 39
The DPPH free radical scavenging model can be used to evaluate the antioxidant activity in a relatively short time. DPPH is a stable free radical and accepts either an electron or OH• to become a stable diamagnetic molecule.40 CGF extract exhibited dose-dependent DPPH radical scavenging activity. The extract, which contained the considerable amount of total phenolics and flavonoids, showed a significant effect in inhibiting DPPH, reaching ∼ 60% at a concentration of 250 μg/mL. The IC50 value (the amount of antioxidant material required to scavenge 50% of free radical in the assay system) for DPPH scavenging by the CGF extract was found to be 165.69 μg/mL. Similarly, significant H2O2 and Superoxide anion radical scavenging activities of CGF were evident at all the tested concentrations. The scavenging activity increased with increasing concentration. The decrease in H2O2 concentration could help to reduce hydroxyl radical formation and therefore reduce the effect caused by oxidative damage. The DPPH radical, H2O2 and superoxide radical scavenging activities of CGF indicates the strong capacity of its constituents that can maintain sound health and protect the human body from oxidative damage, which can induce cancer, arteriosclerosis, arthritis, cirrhosis, inflammation and diabetes, etc.
Advanced Glycation End-Products (AGEs) are ubiquitous signaling proteins related with vascular and neurological complications of diabetes. They include various compounds formed by the Maillard reaction, which is a non-enzymatic glycation of free amino groups by sugars and aldehydes. AGE formation begins under hyperglycemic or oxidative stress conditions and is characterized by conversion of reversible Schiff-base adducts to covalently bound Amadori products, which undergo further rearrangements that terminate in the formation of irreversibly bound compounds known as AGEs.41 Detection and measurement techniques for AGEs have been gradually improved over the past few years, which hold a promise for future bedside management of patients with diabetes.42 The reported role of AGEs in vascular complications of diabetes and cardiovascular disease also prompted the development of pharmacological inhibitors of their effects, giving rise to many experimental activities and a number of both preclinical and clinical studies.8
In the present study, the effect of CGF extract on various parameters indicative of multiple steps of albumin glycation and mitigation of protein oxidation were assessed. The results showed that CGF extract efficiently inhibited fluorescent AGE formation. A significant increase of fructosamine, protein carbonyl content and aggregation of proteins in BSA were observed when the protein was glycated by fructose. When CGF extract was added to the same systems, it significantly suppressed these processes. The antiglycation effect of CGF might be due to presence of high content of polyphenolic (15.47 ± 1.12 mg GAE/g of extract, dry weight) compounds, total flavonoids (6.82 ± 0.67 mgQE/g of extract, dry weight) and saponins (0.12 ± 0.02 mg/g of extract fresh weight). The reduction of free radical generation by antioxidant activity of polyphenols may highlight major mechanisms for the prevention of AGE formation.43 Recent studies shown that polyphenolic compounds from the edible plants may play a protective role against monosaccharide induced protein glycation.44 Kusirisin et al. have reported a strong correlation between the polyphenolic content in the plant extracts and the ability to inhibit protein glycation.45 According to the results obtained, it may be postulated that CGF extract can inhibit glycation by various strategies such as (i) blocking the early glycation product (fructosamine), (ii) reducing the generation of reactive carbonyl or dicarbonyl groups either from fructosamine or glucose and (iii) inhibiting the AGEs formation. The results of the study put forward the protective effect of polyphenolic compounds of CGF against glycation induced cellular damage and inhibition of AGEs.
Albumin, the most abundant circulating protein in the plasma undergoes glycative alteration and the elevated level of glycated albumin (2–3 fold) plays a vital role in the pathogenesis of vascular complications.46 Glycation induced modification of albumin by fructose in prolonged incubation has been reported to form various aggregates and amyloid-like structures causing decrease in α-helices and increase in β-sheet content.47 The long-term accumulation of amyloid cross β-structures in the tissues may cause the progression of pancreatic islet amyloidosis which directly destroys β-cell and impairs insulin secretion.48 Hence, the effect of CGF extract on the structural modifications of BSA was evaluated. The property of amyloid fibril binding to specific dye thioflavin T, was used to quantify the level of amyloid in glycated albumin samples. It was observed that CGF extract showed significant inhibition to amyloid aggregation during the course of experiment, 7–28 days, indicating their potential to prevent conformational changes in albumin during glycation. The reason behind decrease in β-amyloid formation may be the low-molecular-weight bioactive compounds such as polyphenols, phenolic acids or flavonoids present in the extract which have anti-amyloidogenic effects.49 The results of this study corroborate the effective antiglycation action of CGF extract as they not only counteracted with the initial glycation reaction and its progression but also prevented amyloid cross β-structures in BSA in vitro. This beneficial effect might help to reduce the risk of developing diabetes complications.
This report further investigated the cytoprotective potential of CGF extract against alloxan-induced cell death using RINm5F cells using MTT assay. The results of MTT assay (Fig. 8) showed that pretreatments of CGF extract provided a significant protective effect from alloxan-induced RINm5F cell damage. The results suggest that the cytoprotective effect of CGF extract against alloxan may be partly derived from the free radical scavenging activity of its antioxidant compounds. An antidiabetic agent could exert a beneficial effect in the diabetic situation by enhancing insulin secretion and/or by improving/mimicking insulin action.50 In this study, RINm5F cells have been used for studying the effect of insulin secretagogues and the insulin concentrations of the cell culture supernatant were determined. CGF extract at 0.250 mg/mL and 0.50 mg/mL concentrations has significantly increased insulin secretion and stimulated 1.28 and 1.71-fold increase in insulin secretion. The lower concentrations (0.0625 and 0.125 mg/mL) did not increase the insulin secretion significantly.
The flavonoids and glycosides present in C. indica leaves are reported to have antidiabetic effects.51, 52 Triterpenes present in the toluene extract of C. indica were shown to reduce alloxan-induced β-cell damage and increase insulin secretion. Oral administration of pectin isolated from C. indica fruits was shown to have a significant hypoglycemic effect in normal rats.53 Pectin is a soluble fiber that affects blood glucose levels either by decreasing the transit time or indirectly through the production of short chain fatty acids.38, 39 From the results of this study, it can be inferred that the synergistic action of the bioactive constituents present in the CGF extract might contributed to the observed bioactivity.
5. Conclusion
With the prevalence of Coccinia grandis used both for diabetes treatment and food, it is important to know and investigate the possible mechanisms of its antidiabetic activity. This study investigated the potential antidiabetic activity of Coccinia grandis, focussing on antioxidant activity in cell free systems; antiglycation in BSA-fructose in vitro model; insulin secretion and safety through cytotoxicity assay. From the results obtained, it can be concluded that the Coccinia grandis mature unripe fruit extract possesses profound antioxidant, antiglycation, insulin secretory activities. However, as this is only preliminary study, further studies are in progress to identify the active constituents and in vivo oral rodent efficacy studies in a disease model.
Conflicts of interest statement
The authors declare no conflict of interest.
Funding
There is no external funding for this project.
Authors' contributions
Mrs. PM performed the experiments, analyzed/interpreted data and wrote the manuscript.
Dr. AP assisted in performing experiments and writing the manuscript.
Dr.SM contributed to the concept, designed experiments, analyzed/interpreted data and finalized the manuscript.
Footnotes
Peer review under responsibility of The Center for Food and Biomolecules, National Taiwan University.
References
- 1.Kaplan Warren. 2013. Priority medicines for Europe and the world “A Public Health Approach to Innovation”; pp. 8–10. Update on 2004 Background Paper, Background Paper6.4 Diabetes. [Google Scholar]
- 2.Ambady R., Chamukuttan S. Early diagnosis and prevention of diabetes in developing countries. Rev Endocr Metab Disord. 2008;9:193–201. doi: 10.1007/s11154-008-9079-z. [DOI] [PubMed] [Google Scholar]
- 3.Welihinda J., Karunanayake E.H., Sheriff M.H., Jayasinghe K.S. The effect of Momordica charantia on the glucose tolerance in maturity onset diabetes. J Ethnopharmacol. 1986;17:277–282. doi: 10.1016/0378-8741(86)90116-9. [DOI] [PubMed] [Google Scholar]
- 4.Ali L., Khan A.K., Mamun M.I. Studies on hypoglycemic effects of fruit pulp, seed, and whole plant of Momordica charantia on normal and diabetic model rats. Planta Medica. 1993;59:408–412. doi: 10.1055/s-2006-959720. [DOI] [PubMed] [Google Scholar]
- 5.Surveswaran Siddharthan, Cai Yi-Zhong, Corke Harold, Sun Mei. Systematic evaluation of natural phenolic antioxidants from 133 Indian medicinal plants. Food Chem. 2007;102:938–953. [Google Scholar]
- 6.Luo Wei, Zhao Mouming, Yang Bao, Ren Jiaoyan, Shen Guanglin, Rao Guohua. Antioxidant and antiproliferative capacities of phenolics purified from Phyllanthus emblica L. fruit. Food Chem. 2011;126:277–282. [Google Scholar]
- 7.Thornalley P.J. Dicarbonyl intermediates in the maillard reaction. Ann N Y Acad Sci. 2005;1043:111–117. doi: 10.1196/annals.1333.014. [DOI] [PubMed] [Google Scholar]
- 8.Nenna A., Nappi F., Avtaar Singh S.S. Pharmacologic approaches against advanced glycation end products (AGEs) in diabetic cardiovascular disease. Res Cardiovasc Med. 2015;23:e26949. doi: 10.5812/cardiovascmed.4(2)2015.26949. Available online. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Caengprasath N., Ngamukote S., Makynen K., Adisakwattana S. The protective effects of pomelo extract (Citrus Grandis L. Osbeck) against fructose-mediated protein oxidation and glycation. EXCLI J. 2013;12:491–502. [PMC free article] [PubMed] [Google Scholar]
- 10.Friedman E.A. Evolving pandemic diabetic nephropathy. Rambam Maimonides Med J. 2010;1:e0005. doi: 10.5041/RMMJ.10005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ritzel R.A. Therapeutic approaches based on beta-cell mass preservation and/or regeneration. Front Biosci. 2009;14:1835–1850. doi: 10.2741/3345. [DOI] [PubMed] [Google Scholar]
- 12.Venkatesh S., Dayanand Reddy G., Reddy Y.S.R., Sathyavathy D., Madhava Reddy B. Effect of Helicteres isora root extracts on glucose tolerance in glucose-induced hyperglycemic rats. Fitoterapia. 2004;75:364–367. doi: 10.1016/j.fitote.2003.12.025. [DOI] [PubMed] [Google Scholar]
- 13.Venkateswaran S., Pari L. Effect of Coccinia indica on blood glucose, insulin and key hepatic enzymes in experimental diabetes. Pharm Biol. 2002;3:165–170. [Google Scholar]
- 14.Venkateswaran S., Pari L. Effect of Coccinia indica leaf extract on plasma antioxidants in Streptozotocin-induced experimental diabetes in rats. Phytother Res. 2003;17:605–608. doi: 10.1002/ptr.1195. [DOI] [PubMed] [Google Scholar]
- 15.Akhtar M.A., Rashid M., Wahed M.I., Islam R. Comparison of long term antihyperglycemic and hypolipidemic effects between Coccinia cordifolia (Linn.) and Catharanthus roseus (Linn.) in alloxan-induced diabetic rats. Res J Med Med Sci. 2007;2:29–34. [Google Scholar]
- 16.Kumar G.P., Sudheesh S., Vijayalakshmi N.R. Hypoglycaemic effect of Coccinia indica: mechanism of action. Planta Med. 1993;59:330–332. doi: 10.1055/s-2006-959693. [DOI] [PubMed] [Google Scholar]
- 17.Stefanovic Olgica D., Tesic Jelena D., Comic Ljiljana R. Melilotus albus and Dorycnium herbaceum extracts as source of phenolic compounds and their antimicrobial, antibiofilm, and antioxidant potentials. J Food Drug Anal. 2015;23:417–424. doi: 10.1016/j.jfda.2015.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sofowora A. Medicinal Plants and Traditional Medicine in Africa. 2nd Edn. Spectrum Books Ltd; Nigeria, Ibadan: 1993. p. 150. [Google Scholar]
- 19.Harbourne J.B. 3rd Edn. Chapman & Hall Pub; London, UK: 1998. Phytochemical Methods: A Guide to Modern Techniques of Plant Analysis. [Google Scholar]
- 20.Singleton V.L., Orthofer R., Lamuela-Raventos R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. In: Packer L., editor. Vol 299. Academic Press; San Diego, CA: 1999. pp. 152–178. (Methods in Enzymology, Oxidant and Antioxidants (Part A)). [Google Scholar]
- 21.Chang C., Yang M., Wen H., Chern J. Estimation of total flavonoid content in propolis by two complementary colorimetric methods. J Food Drug Anal. 2002;10:178–182. [Google Scholar]
- 22.Anhawange A.A., Ajibola V.O., Oniye S.J. Chemical studies of seeds of Moronga oleifera (Lam) and detarium microcarpum (Guill and Sperr) J Biol Sci. 2004;4:711–715. [Google Scholar]
- 23.Asp N.G., Johansson C.G., Hallmer H., Siljestroem M. Rapid enzymic assay of insoluble and soluble dietary fiber. J Agric Food Chem. 1983;31:476–482. doi: 10.1021/jf00117a003. [DOI] [PubMed] [Google Scholar]
- 24.Koleva, van Beek T.A., Linssen J.P., de Groot A., Evstatieva L.N. Screening of plant extracts for antioxidant activity: a comparative study on three testing methods. Phytochem Anal. 2002;13:8–17. doi: 10.1002/pca.611. [DOI] [PubMed] [Google Scholar]
- 25.Ruch R.J., Cheng S.J., Klaunig J.E. Prevention of cytotoxicity and inhibition of intracellular communication by antioxidant catechins isolated from Chinese green tea. Carcinogenesis. 1989;10:1003–1008. doi: 10.1093/carcin/10.6.1003. [DOI] [PubMed] [Google Scholar]
- 26.Liu F., Ooi V.E.C., Chang S.T. Free radical scavenging activity of mushroom polysaccharide extracts. Life Sci. 1997;60:763–771. doi: 10.1016/s0024-3205(97)00004-0. [DOI] [PubMed] [Google Scholar]
- 27.Sharma S.D., Pandey B.N., Mishra K.P., Sivakami S. Amadori product and age formation during nonenzymatic glycosylation of bovine serum albumin in vitro. J Biochem Mol Biol Biophys. 2002;6:233–242. doi: 10.1080/10258140290031554. [DOI] [PubMed] [Google Scholar]
- 28.Ardestani A., Yazdanparast R. Cyperus rotundus suppresses AGE formation and protein oxidation in a model of fructose-mediated protein glycoxidation. Int J Biol Macromol. 2007;41:572–578. doi: 10.1016/j.ijbiomac.2007.07.014. [DOI] [PubMed] [Google Scholar]
- 29.Meeprom A., Sompong W., Chan C.B., Adisakwattana S. Isoferulic acid, a new anti-glycation agent, inhibits fructose- and glucose-mediated protein glycation in vitro. Molecules. 2013;18:6439–6454. doi: 10.3390/molecules18066439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tupe R.S., Agte V.V. Role of zinc along with ascorbic acid and folic acid during long-term in vitro albumin glycation. Br J Nutr. 2010;103:370–377. doi: 10.1017/S0007114509991929. [DOI] [PubMed] [Google Scholar]
- 31.Kumkrai P., Weeranantanapan O., Chudapongse N. Antioxidant, α-glucosidase inhibitory activity and sub-chronic toxicity of Derris reticulata extract: its antidiabetic potential. BMC Complement Altern Med. 2015;15:35. doi: 10.1186/s12906-015-0552-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods. 1983;65:55–63. doi: 10.1016/0022-1759(83)90303-4. [DOI] [PubMed] [Google Scholar]
- 33.Persaud J., Al-majed H., Raman A., Jones P.M. Gymnema sylvestre stimulates insulin release in vitro by increased membrane permeability. J Endocrinol. 1999;163:207–212. doi: 10.1677/joe.0.1630207. [DOI] [PubMed] [Google Scholar]
- 34.Olaokun O.O., McGaw L.J., Awouafack M.D., Eloff J.N., Naidoo V. The Potential role of GLUT4 transporters and insulin receptors in the hypoglycaemic activity of Ficus lutea acetone leaf extract. BMC Complementary Altern Med. 2014;14:269. doi: 10.1186/1472-6882-14-269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rother K.I. Diabetes treatment: bridging the divide. New Engl J Med. 2007;356:1499–1501. doi: 10.1056/NEJMp078030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Shi J., Ho C.T., Shahidi F. CRC Press; Boca Raton, FL: 2005. Asian Functional Foods. [Google Scholar]
- 37.Mungole Arvind J., Awati Ravi, Chaturvedi Alka, Zanwar Prakash. Preliminary phytochemical screening of Ipomoea obscura (L) – a hepatoprotective medicinal plant. Int J Pharm Tech Res. 2010;24:2307–2312. [Google Scholar]
- 38.Smith J.G., Yokoyama W.H., German J.B. Butyric acid from the diet: actions at the level of gene expression. Crit Rev Food Sci. 1998;38:259–297. doi: 10.1080/10408699891274200. [DOI] [PubMed] [Google Scholar]
- 39.Gao Z., Yin J., Zhang J. Butyrate improves insulin sensitivity and increases energy expenditure in mice. Diabetes. 2009;58:1509–1517. doi: 10.2337/db08-1637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Yermilov V., Rubio J., Becchi M., Friesen M.D., Pignatelli B., Ohshima H. Formation of 8-nitroguanine by the reaction of guanine with peroxynitrite in vitro. Carcinogenesis. 1995;16:2045–2050. doi: 10.1093/carcin/16.9.2045. [DOI] [PubMed] [Google Scholar]
- 41.Ulrich P., Cerami A. Protein glycation, diabetes, and aging. Recent Prog Horm Res. 2001;56:1–21. doi: 10.1210/rp.56.1.1. [DOI] [PubMed] [Google Scholar]
- 42.Tanaka K., Nakayama M., Kanno M. Skin autofluorescence is associated with the progression of chronic kidney disease: a prospective observational study. PLoS One. 2013;8:e83799. doi: 10.1371/journal.pone.0083799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wu C.H., Huang S.M., Lin J.A., Yen G.C. Inhibition of advanced glycation end product formation by foodstuffs. Food Funct. 2011;2:224–234. doi: 10.1039/c1fo10026b. [DOI] [PubMed] [Google Scholar]
- 44.Elosta A., Ghous T., Ahmed N. Natural products as anti-glycation agents: possible therapeutic potential for diabetic complications. Curr Diabetes Rev. 2012;8:92–108. doi: 10.2174/157339912799424528. [DOI] [PubMed] [Google Scholar]
- 45.Kusirisin W., Srichairatanakool S., Lerttrakarnnon P. Antioxidative activity, polyphenolic content and anti-glycation effect of some Thai medicinal plants traditionally used in diabetic patients. Med Chem. 2009;5:139–147. doi: 10.2174/157340609787582918. [DOI] [PubMed] [Google Scholar]
- 46.Guerin-Dubourg A., Catan A., Bourdon E., Rondeau P. Structural modifications of human albumin in diabetes. Diabetes Metabolism. 2012;38:171–178. doi: 10.1016/j.diabet.2011.11.002. [DOI] [PubMed] [Google Scholar]
- 47.Bouma B., Kroon-Batenburg L.M., Wu Y.P. Glycation induces formation of amyloid cross-beta structure in albumin. J Biol Chem. 2003;278:41810–41819. doi: 10.1074/jbc.M303925200. [DOI] [PubMed] [Google Scholar]
- 48.Marzban L., Verchere C.B. The role of islet amyloid polypeptide in type 2 diabetes. Can J Diabetes. 2004;28:39–47. [Google Scholar]
- 49.Tupe R.S., Sankhe N.M., Shaikh S.A. Nutraceutical properties of dietary plants extracts: prevention of diabetic nephropathy through inhibition of glycation and toxicity to erythrocytes and HEK293 cells. Pharm Biol. 2015;53:40–50. doi: 10.3109/13880209.2014.910236. [DOI] [PubMed] [Google Scholar]
- 50.Gray A.M., Flatt P.R. Nature's own pharmacy: the diabetes perspective. Proc Nutr Soc. 1997;56:507–517. doi: 10.1079/pns19970051. [DOI] [PubMed] [Google Scholar]
- 51.Kuriyan R., Rajendran R., Bantwal G., Kurpad A.V. Effect of supplementation of Coccinia cordifolia extract on newly detected diabetic patients. Diabetes Care. 2008;31:216–220. doi: 10.2337/dc07-1591. [DOI] [PubMed] [Google Scholar]
- 52.Ajay S.S. Hypoglycemic activity of Coccinia indica (Cucurbitaceae) leaves. Int J Pharm Tech Res. 2009;1:892–893. [Google Scholar]
- 53.Gurukar M.S., Mahadevamma S., Chilkunda N.D. Renoprotective effect of Coccinia indica fruits and leaves in experimentally induced diabetic rats. J Med Food. 2013;16:839–846. doi: 10.1089/jmf.2012.2689. [DOI] [PMC free article] [PubMed] [Google Scholar]