Abstract
The intracellular facultative bacteria Piscirickettsia salmonis is one of the most important pathogens of the Chilean aquaculture. However, there is a lack of information regarding the whole genomic transcriptional response according to different extracellular environments. We used next generation sequencing (NGS) of RNA (RNA-seq) to study the whole transcriptome of an isolate of P. salmonis (FAVET-INBIOGEN) using a cell line culture and a modified cell-free liquid medium, with or without iron supplementation. This was done in order to obtain information about the factors there are involved in virulence and iron acquisition. First, the isolate was grown in the Sf21 cell line; then, the bacteria were cultured into a cell-free liquid medium supplemented or not with iron. We identified in the transcriptome, genes associated with type IV secretion systems, genes related to flagellar structure assembly, several proteases and sigma factors, and genes related to the development of drug resistance. Additionally, we identified for the first time several iron-metabolism associated genes including at least two iron uptake pathways (ferrous iron and ferric iron uptake) that are actually expressed in the different conditions analyzed. We further describe putative genes that are related with the use and storage of iron in the bacteria, which have not been previously described. Several sets of genes related to virulence were expressed in both the cell line and cell-free culture media (for example those related to flagellar structure; such as basal body, MS-ring, C-ring, proximal and distal rod, and filament), which may play roles in other basic processes rather than been restricted to virulence.
Introduction
In Chile, Piscirickettsiosis is the most important bacterial disease in salmonid fish and causes high mortalities and significant economic losses during the sea water phase (approximately U$ 100 million per year) (reviewed in [1]). The etiologic agent is P. salmonis, a facultative intracellular Gram-negative bacterium [2], phylogenetically related to Francisella, Coxiella, and Legionella [3]. The bacterium is a non-motile coccoid organism with an approximate diameter of 0.5–1.5 μm that is often a pleomorphic, fastidious, and facultative intracellular pathogen in host cells. It has been seen replicating inside intracytoplasmic vacuoles in macrophages of head kidney of infected salmon. In vitro can be cultured in cell lines as CHSE-214 (Chinook salmon embryo), macrophage/monocyte (RTS-11) or Sf21 (Spodoptera frugiperda; ECACC 89070101) cell culture, where P. salmonis has high titers) [4]; and also in cell-free culture media, as CHAB agar, AUSTRAL-SRS broth or MC medium [5–7].
The virulence levels of P. salmonis have been described using mortality curves in different salmonid species [8–11]. Several genes have been used to assess virulence based on the available P. salmonis genome. The presence of a locus encoding a toxin-antitoxin system (TA) was detected and named ps-tox-antox. Expression of the ps-tox gene was highly toxic in transformed Escherichia coli, showing minimal growth after 8 hours compared to those cells carrying the expression vector with both, the ps-antox and ps-tox genes, which showed a normal growth curve [12]. Using a stressful cell-free culture media (Marine broth, Difco), Marshall et al [13] described that P. salmonis produced a large amount of cellular aggregates that form a typical biofilm structure that act as an adaptive strategy for survival and persistence under stress conditions in the marine environment. Using RT-qPCR, the expression of an operon associated with biofilm formation (mazEF) was demonstrated, reaching its highest levels of expression during the early stage of formation of this structure [13]. In addition, the in vitro expression of bacterial genes in the RT11 cell line show that four virulence genes were expressed (dotA, dotB, icmK and icmE; that are homologous to the type IV secretion system Dot/Icm) in P. salmonis. [14]. A recent experiment using the SHK-1 line, suggested that P. salmonis significantly increased the gene expression of two homologues of the clpB and bipA, when compared with a cell free media [15]. These genes show increased expression allowing the pathogen to evade the cellular immune response and replicate inside macrophages [15].
The study of the transcriptome is essential for the functional interpretation of many molecular components of cells that are related to iron metabolism and virulence, thus improving the knowledge about disease pathogenesis [16]. The transcriptome of a cell corresponds to the complete set of transcripts for a specific developmental stage or under a particular physiological condition [17]. Currently, next generation sequencing (NGS) of RNA (RNA-seq) has emerged as a powerful tool for gene expression studies that allows the discovery of novel genes that have not been previously annotated and the mapping of transcriptional structures of a variety of bacterial species [18]. RNA-seq has been used to characterize in vitro differences at the whole transcriptome level between field strains of Salmonella enteritidis with high and low pathogenicity where the latter showed a unique transcriptional profile characterized by a significant reduction in the expression of several transcriptional regulators, genes involved in virulence mechanimsm and in the protection against osmotic and oxidative stress [19]. RNA-seq has also been used to study the transcriptomic response in other phylogenetically related bacteria, such as Legionella pneumophila [20,21]. Mycobacterium marinum [22], which is another pathogen causing disease in fish, show a similar mechanism of persistency like P. salmonis.
The iron metabolism pathways are known in several Gram negative bacteria, but this information is lacking in P. salmonis. For example the transcriptomic analysis of Pseudomonas fluorescens Pf using RNAseq revealed that genes encoding functions related to iron homeostasis, a number of TonB-dependent receptor systems, and some inner-membrane transporters were significantly up-regulated in response to iron limitation. Conversely, the expression of genes involved in hydrogen cyanide production and flagellar biosynthesis were down-regulated under these conditions [23]. The transcriptional response of the Gram negative bacterium Campylobacter jejuni was also characterized using RNA-seq. The results identified 77 iron-activated genes and 50 iron-repressed genes that were involved in iron acquisition or oxidative stress defense, revealing that iron is an important regulator of flagellar biogenesis [24]. This demonstrate the usefulness of this approach for discovery new metabolic pathways in prokaryotes.
In this paper, we are aimed at gain knowledge on the pathways related to several mechanisms used for iron metabolism and virulence in P. salmonis. We present for the first time information concerning the transcriptomic response of P. salmonis via NGS technology and the pathways involved in the growth of this bacterium in different culture conditions. In particular, considering media lacking iron, which is expected to be an important factor involved in bacterial growth [25]. We perform a series of experiments using different cultures (a cell line and a liquid medium supplemented or not with iron) to describe the full transcriptomic response of P. salmonis under these conditions.
Materials and Methods
Bacterial strain and experimental culture conditions
In order to develop a highly proliferative intracellular environment, a cryopreserved isolate of P. salmonis (FAVET-INBIOGEN) was propagated in the Sf21 cell line (Spodoptera frugiperda; ECACC 89070101) [4,26] in antibiotic-free Grace’s medium supplemented with 10% fetal calf serum. Infected monolayers were incubated at 26°C in 25-cm2 culture flasks. Bacterial cells were harvested when the infected monolayers reached 95–100% cytopathic effects (CE). A supplemented liquid medium modified from Henriquez et al. [5] was used that contained 8 g L-1 yeast extract, 4 g L-1peptone from meat, 1.32 g L-1 (NH4)2 SO4, 0.1 g L-1 MgSO4, 6.3 g L-1 K2HPO4, 9 g L-1 NaCl, 0.08 g L-1 CaCl2, 3% glucose and 0.03 g L-1 FeSO4.
Experimental design
Sf21 cells from 4 culture flasks containing monolayers infected with P. salmonis that reached 95–100% CE were centrifuged. Then, an aliquot of 2 ml from the supernatant named “CC medium” (cell culture medium) was centrifuged at 4000 x g for 10 min at 4°C. The pellet was used for total RNA extraction. The rest of the supernatant was also centrifuged, and the pellet was cultured in liquid medium supplemented or not with FeSO4 named “CFC-I medium” (cell-free culture medium with iron supplementation) and “CFC-N medium” (cell-free culture medium without iron supplementation), respectively. The infected media were incubated at 25°C and 100 rpm. After 24 h, a pool of bacterial pelleted from each condition (CFC-I and CFC-N), was obtained for total RNA extraction.
RNA isolation procedures
Total RNA was extracted using TRIzol (Invitrogen) following the manufacturer’s instructions. The RNA pellets dissolved in diethylpyrocarbonate (DEPC)-treated water were quantified using the Qubit® fluorometer (Invitrogen) and stored at -80°C. The quality of RNA extraction was analyzed according to the RNA quality number (RQN) using the Fragment Analyzer®.
Library preparation and transcriptome sequencing
To sequence the transcriptome, RNA samples were treated with DNase I (Fermentas) and purified with the RNeasy MinElute Cleanup Kit (Qiagen) to remove traces of DNase I. Depletion of rRNA was accomplished using the Ribo-Zero™ Kit (Bacteria)–Low Input. Purification of the rRNA-depleted samples was accomplished with the RNeasy MinElute Cleanup Kit (Qiagen). RNA-seq libraries of the samples were prepared using the ScriptSeq™ v2 RNA-Seq Library Preparation Kit (Epicentre®) according to the manufacturer’s protocol. The assessment of the library quantity and quality were completed using qPCR (Illumina) and the Fragment Analyzer®, respectively. The samples were sequenced using the v2 Reagent Kit (300 cycles PE kit) with NGS equipment (Illumina Miseq®).
Mapping of reads and description of transcriptomes
The reads obtained from the CFC and CC media samples were mapped to the annotated genome of the FAVET-INBIOGEN (Whole Genome Shotgun (WGS) project accession number LELB01000000) isolate using the CLC Genomics Workbench. The P. salmonis transcriptomes were characterized according to the functional classification of the Rast server [27] and Kyoto Encyclopedia of Genes and Genomes (KEEG) through the automatic metabolic pathway annotation server KAAS [28]. According to Humphrys et al [29], we defined two different cut-offs to asses a minimal gene expression following sequencing. The minimal gene expression was considering for a gene with a minimum of a RPKM (Reads Per Kilobase of transcript per Million mapped reads) ≥ 0.1 and a minimum of 10 mapped reads. Additionally, to assess the relative abundance of genes between both grown conditions, we used the test of proportion through Kal’s test statistic using CLC Genomic Workbench [30], The genes were considered as differentially expressed with FDR (false discovery rate) ≤ 0.01.
Results
Comparisons between transcriptomes obtained under different culture conditions
P. salmonis from the CC medium had 2188 genes with a minimal level expression (RPKM ≥ 0.1 and a minimum of 10 mapped reads). In the CFC-I medium, P. salmonis expressed 2264 genes and 2296 in the CFC-N medium with a minimal level expression.
We validate some of the genes annotated in the RNA-seq using a RT-qPCR. The chosen genes were ferrous iron transport protein B (feoB), ferric uptake regulation protein (fur), bacterioferritin (bfr), Ferrichrome transport ATP-binding protein (fhuC), RNA polymerase sigma factor RpoS (rpoS) and Ferrochelatase, protoheme ferro-lyase (hemH).
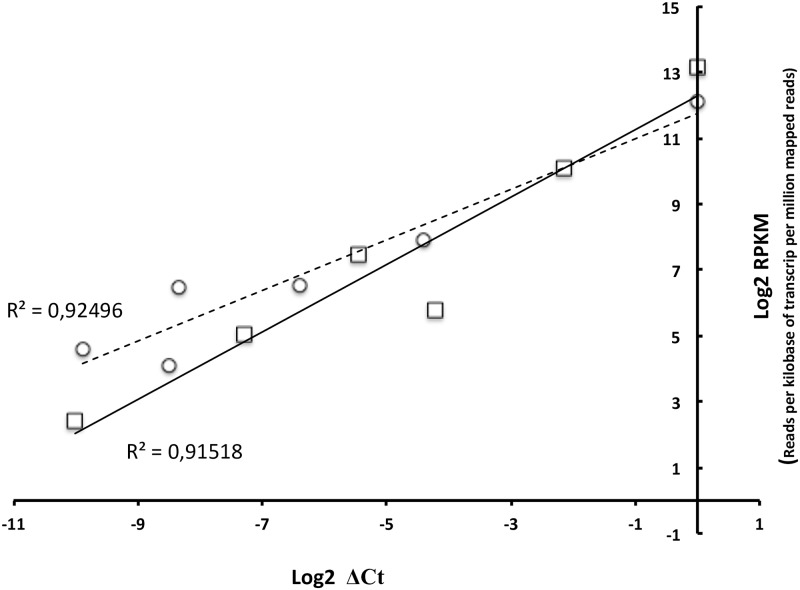
For the 6 genes used, there was a high correlation between Ct and RPKM (Read per kilobase per million mapped reads) values for each gene in both samples (Fig 1).
Fig 1. Correlation between RT-qPCR and RNA-seq data for six genes (feoB, fur, bfr, fhuC, rpoS and hemH) expressed by P. salmonis in both types of cultures.
The tendency line and determination coefficient (R2) are showed for both cultures.
A total of 1205 genes were differentially expressed, where 876 genes were up regulated and 328 genes were down regulated in CFC-I culture medium compare to CC culture medium with FDR ≤ 0.01. The number of differentially expressed genes related to virulence is presented in Table 1. When we compared CFC-I with CFC-N, this analysis revealed 323 genes differentially expressed (FDR ≤ 0.01), with 145 genes up regulated when iron was supplemented in the media. A description of the number of genes related to virulence, differentially expressed presented in Table 2.
Table 1. Number of genes up and down regulated in the transcriptome from CFC medium compare to CC medium.
| Up regulated genes | Down regulated genes | |
|---|---|---|
| Flagellar structures | 1 | 3 |
| Iron metabolism | 23 | 7 |
| Proteases | 6 | 10 |
| Drug resistance | 8 | 8 |
| Oxidative stress | 20 | 6 |
| T4SS | 0 | 8 |
| Chaperones | 5 | 3 |
| Sigma factors | 6 | 1 |
Table 2. Number of genes up and down regulated in the transcriptome from CFC-I medium compare to CFC-N medium.
| Up regulated genes | Down regulated genes | |
|---|---|---|
| Flagellar structures | 0 | 0 |
| Iron metabolism | 5 | 4 |
| Proteases | 1 | 2 |
| Drug resistance | 5 | 2 |
| Oxidative stress | 3 | 4 |
| T4SS | 2 | 0 |
| Chaperones | 1 | 1 |
| Sigma factors | 1 | 1 |
Pathways associated with virulence and iron metabolism
We sought to divide the analysis in two main mechanisms that the bacteria could use to interact with the host. In particular, we analyzed virulence factors such as flagellar structure, type IV secretion system, sigma factors, proteases, chaperones and mobile proteins elements, as described previously. We also analyzed the pathways related with uptake, utilization and storage of the iron (see below).
Virulence-associated genes
Flagellar structures
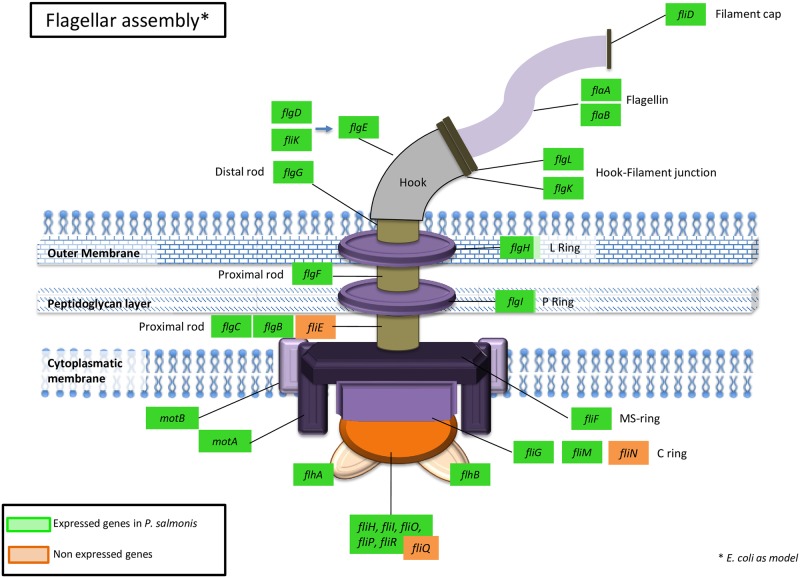
Although this pathogen has been described as a non-motile bacterium, a total of 46 genes involved in flagellum biosynthesis, assembly, structural organization, function, and the flagella motor genes were expressed in P. salmonis under different conditions (Fig 2). Among these, three genes were differentially expressed in CC medium when it was compared with the CFC-I medium: flagellar motor rotation protein MotB_2, Flagellar hook-associated protein FlgL and Chemotaxis regulator-transmits chemoreceptor signals to flagelllar motor components CheY_3, which were 4.7, 1.9 and 1.6 fold more expressed in CC medium (FDR ≤ 0.01) (S1 Table). When we compared CFC-I with CFC-N, we did not find any genes differential expressed between treatments.
Fig 2. Representative genes associated with the flagella in the transcriptome.
The image shows the set of genes associated with the flagellar structure that are expressed by P. salmonis in all conditions tested. The green square represents genes expressed in the different media, whereas the orange square represents genes that did not show expression., in the different grown conditions analyzed.
All these flagella related genes have been involved in protein export (especially of virulence factors) in non-motile bacteria. Several proteins that form the flagellar metabolic pathway may export other proteins to the extracellular space, especially those linked to virulence [31,32]. Maezawa et al. [33] reported the existence of hundreds of flagellar-expressed hooks and basal body structures in a bacterium that lacked the filament portion of the flagellum, supporting previous suggestions of the possible specialization of these genes in protein export from the bacterium to the host. Abby and Rocha [34] suggested that non-flagellar type III secretion systems (injectisomes) evolved from the bacterial flagellum. Additionally, P. salmonis can produce extracellular products (EPCs), at least some of which are thermo-labile exotoxins that probably play a role in the pathogenesis of Piscirickettsiosis [35]. Overall, the results showed that P. salmonis expressed several genes that were associated with the flagellar structure; However, the fliC gene that encodes the filament of the flagellum was not found in the annotated genome. Moreover, the subunits flaA and flaB which also encode the filament of the flagella were expressed. This results show that P. salmonis is expressing another host-cell adaptation system (i.e., the type III secretion system), which likely delivery virulence factors such as flagellar component and exotoxins into eukaryotic cells. Further analysis of this secretion system are required in order to understand the interaction of these factors in the host.
The type IV secretion system
Data from our experiment showed that the FAVET-INBIOGEN isolate expressed 24 genes annotated in the icm/dot system. Among this group, eight genes were differentially expressed in CC medium (FDR ≤ 0.01) compared with CFC-I (S1 Table), and two were differentially expressed between CFC-I and CFC-N (S2 Table). The group of genes that form the icm (intracellular multiplication)/dot (defective organelle trafficking) system have been described as a type IV secretion system (T4SS) that is responsible for intracellular survival and multiplication. T4SS has been described in L. pneumophila and C. burnetii as one of the most important virulence factors of these pathogens [36–39], which are both pathogens, are phylogenetically related to P. salmonis.
Proteases
Bacterial pathogens express a group of proteases that contribute to virulence either directly due to host interactions or indirectly (e.g., allowing replication in the host environment) [40]. Because P. salmonis is a facultative intracellular bacterium, we would expect to find expression of alternative proteases related to these processes that have not been previously described in this bacterium. Indeed, the transcriptome of the FAVET-INBIOGEN isolate revealed 20 genes (e.g., clpP, clpA, clpS and clpX) encoding proteases and chaperons that were expressed under all culture conditions and would contribute to bacterial adaptation to the hostile conditions produced by host cells [41]. Moreover, clpB, whose expression has been described previously in P. salmonis, was differentially expressed in the CC medium compared with CFC-I medium (FDR ≤ 0.01) (S1 Table), but it was not differentially expressed between CFC-N and CFC-I.
Sigma factors
Bacterial pathogens have alternative sigma factors that regulate the expression of virulence genes and virulence-associated genes in response to a particular stimuli, which can be critical for host establishment [42]. Alternative sigma factors are a class of proteins that constitute essential dissociable subunits of the prokaryotic RNA polymerase [43]. The association of appropriate alternative sigma factors with the core RNA polymerase provides a mechanism for cellular responses mediated through the redirection of transcription initiation [44].
The transcriptomic responses of the FAVET-INBIOGEN isolate under all culture conditions showed 10 genes related with sigma factors. Seven were differentially expressed between CC medium and CFC-I (S1 Table), and two between CFC-I and CFC-N (S2 Table). Among those expressed genes; rpoE, encoding the sigma factor E (σE) that controls specific functions involved in the interaction of the bacterium with the host (e.g., synthesis, assembly and homeostasis of lipopolysaccharides and outer membrane porins including several proteases) [45]; rpoD, which has been described as a housekeeping gene in Gram-negative bacteria; rpoH, which is associated with the heat-shock response; rpoS, which contributes to bacterial survival in stressful environments [44]; and rpoN, which has been reported to be involved in flagellar regulation [46].
Chaperones
Molecular chaperones are essential for the survival of all three kingdoms of life and have been shown to play key roles in the integration of cellular and organismal responses to environmental fluctuation. Heat shock proteins (Hsp) comprise a subgroup of molecular chaperones that are induced in response to adverse environmental conditions. These proteins have been reported for several bacteria during infection processes, and thus form a first line of attack that helps consolidate pathogen virulence [47]. Similar to many other pathogenic bacteria, we expect that P. salmonis would express several groups of chaperones that were important for the invasion process in the host. Indeed, numerous genes encoding Hsp33, Hsp40, Hsp60, Hsp70 and Hsp90 family proteins were annotated in the transcriptomes of P. salmonis. The genes encoding the heat shock protein GrpE, GroES, GroEL, Hsp90 and Hsp70, which were more expressed in the CC medium, and the gene encoding the Hsp33, hsp40 and “Protein export cytoplasm chaperone” proteins were more expressed in the CFC-I medium (FDR ≤ 0.01) (S1 Table). When we compared CFC-I with CFC-N only 2 genes were not consistently differentially expressed, hsp40 which was more expressed in CFC-I and hsp90 which was more expressed in CFC-N (FDR ≤ 0.01) (S2 Table).
Mobile protein elements
The analysis of the transcriptomes in this study revealed more than forty annotations defined as "mobile protein elements", which are defined as DNA segments encoding enzymes and other proteins that allow movement of the genomic DNA [48]. Sixteen of them were differentially expressed between CC medium and CFC-I (S1 Table), and seven between CFC with and without iron supplementation (S2 Table) (FDR ≤ 0.01).
Iron metabolism-associated genes
As mentioned before, few studies are available in the literature regarding iron metabolism in P. salmonis. Thus, to characterize the mechanisms underlying iron homeostasis in this bacterium is important, in order to understand more deeply how the bacteria interact with the host. In all the media used, a total of 41 genes related to iron homeostasis were expressed and sorted into four functional groups: uptake/transport, intracellular utilization, storage and.iron regulation (S1–S3 Figs).
Iron uptake
The transcriptomic response of P. salmonis was associated with three pathways involved with the transport for siderophores. Both transcriptomes contained homologues of vibrioferrin (pvsA, pvsB, pvsC, pvsD, pvsE, and pvuA), a member of the carboxylate class of siderophores that has been described in marine a bacteria [49,50]. Among them, pvsB, pvsC, pvsD, pvsE were differentially expressed in CC medium versus the CFC-I medium (FDR ≤ 0.01) (S1 Table). Additionally, a homologue of fhuC, which is involved in the iron uptake by siderophores of the hydroxamate class, was also represented in both transcriptomes, but more expressed in CC medium (FDR ≤ 0.01) (S1 Table). A homologue of the feoB gene and other of the feoA gene that are take up Fe2+ directly were expressed in both cultures media. In addition, a homologous gene fecD that is involved in iron uptake by siderophores of the dicitrate class was also contained in all transcriptomes analyzed of the bacteria (associated with CC, CFC-I, CFC-N), but without showing statistical differences between both grown conditions.
When we compared CFC-I with CFC-N two genes were differentially expressed (feoA and pvuA) which were more expressed in CFC-N (FDR ≤ 0.01) (S2 Table).
Iron utilization
Iron-sulphur [Fe-S] clusters and ferredoxins (fdx) play an important role in cellular processes, such as bacterial respiration, cofactor biosynthesis and gene expression [51]. As expected, the [Fe-S] cluster assembly pathway was represented in both growth conditions assayed for P. salmonis by genes encoding for the following proteins: a cysteine desulphurase that catalyzes the release of sulphur from L-cysteine, which is one of the essential components of the CFC medium; the SufE protein that accepts the sulphur molecule that is necessary for [Fe-S] cluster assembly [52]; an ATPase SufC protein and two assembly proteins (SufB and SufD) [53]; a gene encoding a likely iron binding protein from the SufA family; and a gene associated with the iron binding protein IscA, which is involved in [Fe-S] cluster assembly. All these genes were over represented in CFC-I medium in relation to CC medium (FDR ≤ 0.01) (S1 Table). More importantly, we detected the expression of iscR, which is a necessary regulator of [Fe-S] cluster assembly [54,55] and that was 6-fold more represented in CFC-I medium than CC medium (FDR ≤ 0.01) (S1 Table).
Comparing CFC-I with CFC-N there were four genes differentially expressed (pvuA, 4Fe-4S ferredoxin-iron sulfur binding, IscR and probable iron binding protein from the HesB_IscA_SufA family) (FDR ≤ 0.01), which three of them more expressed in CFC-I (S2 Table).
Iron storage
As expected, all the transcriptomes contained the bfr gene that encodes bacterioferritin, which was 1.4 fold more expressed in CFC-I medium than CC medium (FDR ≤ 0.01) (S1 Table), and 1.06 fold more expressed in CFC-I than CFC-N medium (S2 Table). This protein is involved in intracellular iron storage and has been described as the most important storage protein for iron in bacteria [56,57].
Iron regulation
Similar to many other prokaryotes, iron homeostasis in P. salmonis appear to be regulated by the fur gene. The fur gene that encodes the main transcriptional regulator of iron genes [58,59] was almost 3-fold more represented in CFC-I medium when compared with the CC medium (FDR ≤ 0.01) (S1 Table). However, the “RNA-binding protein hfq” gene was also differentially expressed in all comparison (S1 and S2 Tables). This gene has been described to encode a chaperone of a small RNA named RhyB that was also responsible for iron homeostasis, albeit at a different level than fur [60,61]. A further analysis of the small RNA will be required in order to understand more deeply this mechanism.
Oxidative stress
High concentrations of iron result in an oxidant environment that catalyzes reactive species of oxygen, mainly through the Fenton reaction. These molecules are toxic causing cell death [62,63]. Several expressed genes were observed under both conditions, e.g: alkyl hydroperoxidase D (ahpD), alkyl hydroperoxide reductase C (ahpC), catalase (kat) and superoxide dismutase (sodB), which are associated with protection of the cell against oxidative stress and were up regulated in the CFC-I medium compared with CC medium (FDR ≤ 0,01) (S1 Table). Other seven genes were differentially expressed between CFC-I and CFC-N, four of them were more expressed in CFC with iron supplementation (FDR ≤ 0.01) (S2 Table).
Discussion
Genes associated with virulence processes
Our results showed that 46 genes were associated with flagellar biosynthesis, assembly, structural organization, and function (S3 Table). To date, P. salmonis has been described as a non-motile bacterium [1]. However, in this type of facultative intracellular symbiotic bacteria the presence of the flagella is unnecessary and energetically expensive unless proteins involved in the flagella pathway are also involved in other essential functions for the bacterium or the host [31].
During in vivo infection, P. salmonis replicates in the phagocyte preventing phagosome fusion with the lysosome. Therefore, the bacterium requires the expression of the necessary metabolic machinery to survive under these conditions [1,64,65] The expression of the Dot/Icm type IV secretion system (icmK, icmE, dotA and dotB) was previously described in P. salmonis [14]. Our results demonstrated that those dot/icm gene homologues were expressed both during in vitro cell line infection and in cell-free media, suggesting putative constitutive expression, confirming thereby the hypothesis previously suggested by Gomez et al. [14]. In L. pneumophila, this Type IV secretion system shows cytopathogenicity and hemolytic properties in addition to being essential for the induction of apoptosis [66,67]. To understand and analyze the functions of proteins delivered into host cells by this mechanism, it is important to improve the knowledge of the mechanisms related with P. salmonis pathogenesis.
The stressful conditions of the phagosome (i.e., oxidative burst and fusion of the phagosome with lysosomes) are capable of inducing the expression of bacterial chaperones (heat shock proteins, Hsp) and proteases, which also participate in the survival strategies of intracellular pathogens. Our results suggest similar survival strategies due to the expression of genes that are homologues of the most important classes of Hsps described in bacteria (Hps33, DnaJ, DnaK, HtpG, GroEL, GroES and GrpE proteins). All these proteins are involved in protein folding processes in E. coli, B. suis, C. jejuni and S. enterica serovar Typhimurium [47,68–70]. At present, DnaK protein is the best characterized bacterial chaperone. Mutations in dnaK increase the sensitivity of normally resistant S. aureus strains to oxacillin and methicillin; moreover, mutations in dnaK or dnaJ in E. coli make the cells susceptible to fluoroquinolones [71].
The FAVET-INBIOGEN isolate also expressed the clpP and clpB genes that encoded ATP-dependent proteases that were described as important virulence factors in numerous bacterial pathogens [72]. The represented clpA and clpX genes in the transcriptomes have been described in Gram-negative bacteria [40]. These genes encode two ATPase-chaperones that interact with the ClpP protease [41]. In L. pneumophila, the ClpP homologue is required for cell division and several transmission traits, including stress tolerance, cell shortening, sodium sensitivity, cytotoxicity and intracellular multiplication [73]. Moreover, the ClpB protease contributes to resistance to various stress conditions, intracellular multiplication and virulence in F. tularensis [74]. The presence of the clpB gene has been described in the P. salmonis showing significantly increased expression during infection of the SHK-1 cell line compared to the cell-free medium, indicating that clpB could also be expressed during propagation in host cells permitting the adaptation to the hostile conditions in the host [15]. We found similar results in our study, i.e. the clpB gene was significantly more expressed in the CC medium (Sf21 cell line), thereby confirming the hypothesis of Isla et al. [15]. Despite the fact that phenotypic protein expressions have not yet have been demonstrated in P. salmonis, data concerning its presence in the genome and its gene expression will allow the study of alternative mechanisms of survival in the host.
Metabolic pathways and iron uptake
Previously, four putative genes encoding siderophore-related proteins involved in iron metabolism and transport (one hemH gene, two tonB genes, and one fur gene) were reported in the published genome of P. salmonis [75]. The results of the transcriptome analysis of our isolate revealed two pathways for iron uptake: through siderophores and iron ferrous uptake.
Several Gram-negative bacteria synthesize and excrete low-molecular-weight compounds that chelate iron with high affinity that are called siderophores [76]. Siderophores are synthesized and secreted for iron acquisition; for example, muscle tissues expressing carrier proteins in Atlantic salmon were recently demonstrated to have a significantly higher bacterial load and became a niche where P. salmonis could acquire iron [25]. The FAVET-INBIOGEN isolate expressed genes encoding a protein system homologous to a system present in V. parahaemolyticus that was involved in the biosynthesis of vibrioferrin [77]. Because P. salmonis is a Gram-negative bacterium, the expressed genes also encoded proteins of the TonB system, which is a sophisticated mechanism that supplies the required energy for ferri-siderophores uptake. The TonB system includes an integral membrane protein (ExbB), a membrane-anchored periplasmatic protein (ExbD) and the TonB protein [78,79].
The feo system is the main route described for the uptake of Fe+2 and is associated with the growth of bacteria under anaerobic or microaerobic [80,81] and to a lesser extent aerobic conditions. This system was described in E. coli K-12; however, it has also been described in Salmonella [82], Campylobacter [83], Francisella [84] and Legionella [85]. The expression of feoB serves as an alternative pathway for iron uptake by P. salmonis.
The RNA-seq data show that bacterioferritin (bfr) is a mechanism that the bacteria is using for iron storage. This is a ubiquitous system used for the other facultative intracellular bacteria, such as Salmonella and B. abortus [86,87]. When intracellular iron concentrations are very high, this family of proteins also plays an antioxidant role and sequesters iron away from oxygen free radicals [88].
To assembly the [Fe-S] cluster (in addition to serving as a source of iron), the bacterium requires a source of sulphur. The sulphur is supplied by L-cysteine, an amino acid with a thiol side chain, which is an important supplemental component of the CFC medium. After transport into the cytoplasm, the cysteine desulphurase catalyzes the release of sulphur from L-cysteine, which is necessary for [Fe-S] cluster assembly [89]. Cysteine supplementation is not required for intracellular growth in macrophages, mammalian cell lines, or natural amoebic hosts, indicating that cysteine and other essential amino acids are obtained directly from the host [90].
An environment with excess iron together with reactive oxygen species (ROS) via the Fenton reaction causes oxidative stress. This stress increases the damage associated with [Fe–S] clusters, protein carbonylation, Cys/Met-residue oxidation, membrane lipid peroxidation, and DNA damage. The main strategy employed is the production of enzymes that degrade ROS species to maintain stress levels within a range of tolerance [91]. Our bacteria expressed alkyl hydroperoxidase D (ahpD) and alkyl hydroperoxide reductase C (ahpC), which provide significant antioxidant protection and have been described in various bacteria [92–94]. Additionally, Chung et al. [95] demonstrated that deletion of the ahpC genes alone in V. parahaemolyticus did not significantly affect the survival of this pathogen; thus, bacteria require more than one of these antioxidant molecules. Resistance against oxidative stress through biofilm formation was studied in C. jejuni, where the over expression of ahpC was associated with a decrease in biofilm formation; conversely, an ahpC mutant increased biofilm formation [96].
Finally, one of the most important factors involved in the development of resistance to antibiotics is the ability of bacteria to share genetic resources through lateral gene transfer (LGT) [97]. Drug resistance genes are commonly associated with mobile genetic elements (the mobilome) that can be transferred between bacteria by plasmids, integrons and transposons [98].
Final remarks
Several sets of virulence-associated genes were expressed under the experimental conditions studied here; however, the response of the bacteria was similar in different media. Virulence-associated genes were expressed in the cell line culture and also in the cell-free medium supplemented with iron, indicating that these genes could play a role in basic processes rather than specifically in pathogenesis. Furthermore, the expressed genes related to flagellar structure could play a similar role to a type III secretion system during pathogenic processes, supporting the production of extracellular products by P. salmonis [35], but still this require further analysis considering the interaction with the host.
Overall, this study provides new insights into the functional mechanisms of P. salmonis. This information will be important for improving our basic knowledge of this bacterium to gain better prevention and control strategies against Piscirickettsiosis in Chile.
Supporting Information
(XLSX)
(XLSX)
(XLSX)
(A) Suggested genetic organization of siderophore synthesis and transport genes transcribed from the P. salmonis genome that could represent a putative operon. The RPKM values of each of genes that were expressed in the pathway are showed for the CFC-I growth condition. (B) Siderophore synthesis is accomplished by the PvsA, PvsB, PvsD and PvsE proteins. The PvsC protein transports the siderophore to the extracellular space, where it captures Fe+3 and is uptaked by the outer membrane protein PvuA. (OM: outer membrane; CM: cytoplasmatic membrane).
(TIFF)
(A) The organization of tonB, ferric aerobactin, ABC transporter and the iron (III) dicitrate transport system permease protein FecD genes could represent an operon separated from the exbD/tolQ and motA/tolQ/exbB proton channel family protein genes. The RPKM values of each of genes that were expressed in the pathway are showed for the CFC-I growth condition. (B) There are two putative genes encoding transmembrane cytoplamatic proteins which transport ferri-siderophores from the periplasmic space into the cytoplasm where these molecules are reduced. (OM: outer membrane; CM: cytoplasmatic membrane).
(TIFF)
The RPKM values of each of genes that were expressed in the pathway are showed for the CFC-I growth condition. When the cell has high levels of [Fe-S] clusters, the IscR protein would capture a cluster in its structure, which is a signal for transcriptional repression of sufB, sufC, sufD, and sufS.
(TIFF)
Acknowledgments
This work was financially supported by the Chilean Government through FONDEF-IDEA (14I10090) to V. Martinez. Alvaro Machuca acknowledge the support of scholarships for National PhD students from CONICYT.
Data Availability
Genome assembly files are available from the NCBI database (Whole Genome Shotgun (WGS) project accession number LELB01000000). All other relevant data are within the paper and its Supporting Information files.
Funding Statement
This work was financially supported by the Chilean Government through FONDEF-IDEA (14I10090) to V. Martinez. Alvaro Machuca acknowledges the support of scholarships for National PhD students from CONICYT.
References
- 1.Rozas M, Enriquez R (2014) Piscirickettsiosis and Piscirickettsia salmonis in fish: a review. J Fish Dis 37: 163–188. 10.1111/jfd.12211 [DOI] [PubMed] [Google Scholar]
- 2.Fryer JL, Lannan CN, Giovannoni SJ, Wood ND (1992) Piscirickettsia salmonis gen. nov., sp. nov., the causative agent of an epizootic disease in salmonid fishes. Int J Syst Bacteriol 42: 120–126. 10.1099/00207713-42-1-120 [DOI] [PubMed] [Google Scholar]
- 3.Liu R, Ochman H (2007) Origins of flagellar gene operons and secondary flagellar systems. J Bacteriol 189: 7098–7104. 10.1128/JB.00643-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Birkbeck TH, Griffen AA, Reid HI, Laidler LA, Wadsworth S (2004) Growth of Piscirickettsia salmonis to high titers in insect tissue culture cells. Infect Immun 72: 3693–3694. 10.1128/IAI.72.6.3693-3694.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Henriquez M, Gonzalez E, Marshall SH, Henriquez V, Gomez FA, et al. (2013) A novel liquid medium for the efficient growth of the salmonid pathogen Piscirickettsia salmonis and optimization of culture conditions. PLoS One 8: e71830 10.1371/journal.pone.0071830 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mikalsen J, Skjaervik O, Wiik-Nielsen J, Wasmuth MA, Colquhoun DJ (2008) Agar culture of Piscirickettsia salmonis, a serious pathogen of farmed salmonid and marine fish. FEMS Microbiol Lett 278: 43–47. 10.1111/j.1574-6968.2007.00977.x [DOI] [PubMed] [Google Scholar]
- 7.Yanez AJ, Valenzuela K, Silva H, Retamales J, Romero A, et al. (2012) Broth medium for the successful culture of the fish pathogen Piscirickettsia salmonis. Dis Aquat Organ 97: 197–205. 10.3354/dao02403 [DOI] [PubMed] [Google Scholar]
- 8.Lannan CN, F JL. (1994) Extracellular survival of Piscirickettsia salmonis. J Fish Dis 17: 545–548. [Google Scholar]
- 9.House ML, Bartholomew JL, Winton JR, Fryer JL (1999) Relative virulence of three isolates of Piscirickettsia salmonis for coho salmon Oncorhynchus kisutch. Dis Aquat Organ 35: 107–113. 10.3354/dao035107 [DOI] [PubMed] [Google Scholar]
- 10.Smith PA, Pizarro P, Ojeda P, Contreras J, Oyanedel S, et al. (1999) Routes of entry of Piscirickettsia salmonis in rainbow trout Oncorhynchus mykiss. Dis Aquat Organ 37: 165–172. 10.3354/dao037165 [DOI] [PubMed] [Google Scholar]
- 11.Smith PA, Rojas ME, Guajardo A, Contreras J, Morales MA, et al. (2004) Experimental infection of coho salmon Oncorhynchus kisutch by exposure of skin, gills and intestine with Piscirickettsia salmonis. Dis Aquat Organ 61: 53–57. 10.3354/dao061053 [DOI] [PubMed] [Google Scholar]
- 12.Gómez FA, Cardenas C, Henriquez V, Marshall SH (2011) Characterization of a functional toxin-antitoxin module in the genome of the fish pathogen Piscirickettsia salmonis. FEMS Microbiol Lett 317: 83–92. 10.1111/j.1574-6968.2011.02218.x [DOI] [PubMed] [Google Scholar]
- 13.Marshall SH, Gomez FA, Ramirez R, Nilo L, Henriquez V (2012) Biofilm generation by Piscirickettsia salmonis under growth stress conditions: a putative in vivo survival/persistence strategy in marine environments. Res Microbiol 163: 557–566. 10.1016/j.resmic.2012.08.002 [DOI] [PubMed] [Google Scholar]
- 14.Gomez FA, Tobar JA, Henriquez V, Sola M, Altamirano C, et al. (2013) Evidence of the presence of a functional Dot/Icm type IV-B secretion system in the fish bacterial pathogen Piscirickettsia salmonis. PLoS One 8: e54934 10.1371/journal.pone.0054934 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Isla A, Haussmann D, Vera T, Kausel G, Figueroa J (2014) Identification of the clpB and bipA genes and an evaluation of their expression as related to intracellular survival for the bacterial pathogen Piscirickettsia salmonis. Vet Microbiol 173: 390–394. 10.1016/j.vetmic.2014.08.014 [DOI] [PubMed] [Google Scholar]
- 16.Oshlack A, Robinson MD, Young MD (2010) From RNA-seq reads to differential expression results. Genome Biol 11: 220 10.1186/gb-2010-11-12-220 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.McGettigan PA (2013) Transcriptomics in the RNA-seq era. Curr Opin Chem Biol 17: 4–11. 10.1016/j.cbpa.2012.12.008 [DOI] [PubMed] [Google Scholar]
- 18.Pinto AC, Melo-Barbosa HP, Miyoshi A, Silva A, Azevedo V (2011) Application of RNA-seq to reveal the transcript profile in bacteria. Genet Mol Res 10: 1707–1718. [DOI] [PubMed] [Google Scholar]
- 19.Shah DH (2014) RNA sequencing reveals differences between the global transcriptomes of Salmonella enterica serovar enteritidis strains with high and low pathogenicities. Appl Environ Microbiol 80: 896–906. 10.1128/AEM.02740-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Weissenmayer BA, Prendergast JG, Lohan AJ, Loftus BJ (2011) Sequencing illustrates the transcriptional response of Legionella pneumophila during infection and identifies seventy novel small non-coding RNAs. PLoS One 6: e17570 10.1371/journal.pone.0017570 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Fryer JL, Hedrick RP (2003) Piscirickettsia salmonis: a Gram-negative intracellular bacterial pathogen of fish. J Fish Dis 26: 251–262. [DOI] [PubMed] [Google Scholar]
- 22.Wang S, Dong X, Zhu Y, Wang C, Sun G, et al. (2013) Revealing of Mycobacterium marinum transcriptome by RNA-seq. PLoS One 8: e75828 10.1371/journal.pone.0075828 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lim CK, Hassan KA, Tetu SG, Loper JE, Paulsen IT (2012) The effect of iron limitation on the transcriptome and proteome of Pseudomonas fluorescens Pf-5. PLoS One 7: e39139 10.1371/journal.pone.0039139 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Butcher J, Stintzi A (2013) The transcriptional landscape of Campylobacter jejuni under iron replete and iron limited growth conditions. PLoS One 8: e79475 10.1371/journal.pone.0079475 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dettleff P, Bravo C, Patel A, Martinez V (2015) Patterns of Piscirickettsia salmonis load in susceptible and resistant families of Salmo salar. Fish Shellfish Immunol 45: 67–71. 10.1016/j.fsi.2015.03.039 [DOI] [PubMed] [Google Scholar]
- 26.Vaughn JL, Goodwin RH, Tompkins GJ, McCawley P (1977) The establishment of two cell lines from the insect Spodoptera frugiperda (Lepidoptera; Noctuidae). In Vitro 13: 213–217. [DOI] [PubMed] [Google Scholar]
- 27.Aziz RK, Bartels D, Best AA, DeJongh M, Disz T, et al. (2008) The RAST Server: rapid annotations using subsystems technology. BMC Genomics 9: 75 10.1186/1471-2164-9-75 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Moriya Y, Itoh M, Okuda S, Yoshizawa AC, Kanehisa M (2007) KAAS: an automatic genome annotation and pathway reconstruction server. Nucleic Acids Res 35: W182–185. 10.1093/nar/gkm321 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Humphrys MS, Creasy T, Sun Y, Shetty AC, Chibucos MC, et al. (2013) Simultaneous transcriptional profiling of bacteria and their host cells. PLoS One 8: e80597 10.1371/journal.pone.0080597 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kal A, Van Zonneveld A, Benes V, Van Den Berg M, Koerkamp M, et al. (1999) Dynamics of gene expression revealed by comparison of serial analysis of gene expression transcript profiles from yeast grown on two different carbon sources. Mol Biol Cell 10: 1859–1872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Toft C, Fares MA (2008) The evolution of the flagellar assembly pathway in endosymbiotic bacterial genomes. Mol Biol Evol 25: 2069–2076. 10.1093/molbev/msn153 [DOI] [PubMed] [Google Scholar]
- 32.Kearns DB (2010) A field guide to bacterial swarming motility. Nat Rev Microbiol 8: 634–644. 10.1038/nrmicro2405 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Maezawa K, Shigenobu S, Taniguchi H, Kubo T, Aizawa S, et al. (2006) Hundreds of flagellar basal bodies cover the cell surface of the endosymbiotic bacterium Buchnera aphidicola sp. strain APS. J Bacteriol 188: 6539–6543. 10.1128/JB.00561-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Abby SS, Rocha EP (2012) The non-flagellar type III secretion system evolved from the bacterial flagellum and diversified into host-cell adapted systems. PLoS Genet 8: e1002983 10.1371/journal.pgen.1002983 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rojas ME, Galleguillos M, Diaz S, Machuca A, Carbonero A, et al. (2013) Evidence of exotoxin secretion of Piscirickettsia salmonis, the causative agent of Piscirickettsiosis. J Fish Dis 36: 703–709. 10.1111/jfd.12019 [DOI] [PubMed] [Google Scholar]
- 36.De Buck E, Anne J, Lammertyn E (2007) The role of protein secretion systems in the virulence of the intracellular pathogen Legionella pneumophila. Microbiology 153: 3948–3953. 10.1099/mic.0.2007/012039-0 [DOI] [PubMed] [Google Scholar]
- 37.Newton HJ, Ang DK, van Driel IR, Hartland EL (2010) Molecular pathogenesis of infections caused by Legionella pneumophila. Clin Microbiol Rev 23: 274–298. 10.1128/CMR.00052-09 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Beare PA, Gilk SD, Larson CL, Hill J, Stead CM, et al. (2011) Dot/Icm type IVB secretion system requirements for Coxiella burnetii growth in human macrophages. MBio 2: e00175–00111. 10.1128/mBio.00175-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lifshitza Z, B D; Schwartzc K; Shumanc H; Pupkob T; et al. (2014) Identification of Novel Coxiella burnetii Icm/Dot Effectors and Genetic Analysis of Their Involvement in Modulating a Mitogen- Activated Protein Kinase Pathway. Infect Immun 82: 3740–3752. 10.1128/IAI.01729-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ingmer H, Brondsted L (2009) Proteases in bacterial pathogenesis. Res Microbiol 160: 704–710. 10.1016/j.resmic.2009.08.017 [DOI] [PubMed] [Google Scholar]
- 41.Kress W, Maglica Z, Weber-Ban E (2009) Clp chaperone-proteases: structure and function. Res Microbiol 160: 618–628. 10.1016/j.resmic.2009.08.006 [DOI] [PubMed] [Google Scholar]
- 42.Bashyam MD, Hasnain SE (2004) The extracytoplasmic function sigma factors: role in bacterial pathogenesis. Infect Genet Evol 4: 301–308. 10.1016/j.meegid.2004.04.003 [DOI] [PubMed] [Google Scholar]
- 43.Kazmierczak MJ, Wiedmann M, Boor KJ (2005) Alternative sigma factors and their roles in bacterial virulence. Microbiol Mol Biol Rev 69: 527–543. 10.1128/MMBR.69.4.527-543.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Dong T, Schellhorn HE (2010) Role of RpoS in virulence of pathogens. Infect Immun 78: 887–897. 10.1128/IAI.00882-09 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Rhodius VA, Suh WC, Nonaka G, West J, Gross CA (2006) Conserved and variable functions of the sigmaE stress response in related genomes. PLoS Biol 4: e2 10.1371/journal.pbio.0040002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Jacobi S, Schade R, Heuner K (2004) Characterization of the alternative sigma factor sigma54 and the transcriptional regulator FleQ of Legionella pneumophila, which are both involved in the regulation cascade of flagellar gene expression. J Bacteriol 186: 2540–2547. 10.1128/JB.186.9.2540-2547.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Neckers L, Tatu U (2008) Molecular chaperones in pathogen virulence: emerging new targets for therapy. Cell Host Microbe 4: 519–527. 10.1016/j.chom.2008.10.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Frost LS, Leplae R, Summers AO, Toussaint A (2005) Mobile genetic elements: the agents of open source evolution. Nat Rev Microbiol 3: 722–732. 10.1038/nrmicro1235 [DOI] [PubMed] [Google Scholar]
- 49.Amin SA, Green DH, Kupper FC, Carrano CJ (2009) Vibrioferrin, an unusual marine siderophore: iron binding, photochemistry, and biological implications. Inorg Chem 48: 11451–11458. 10.1021/ic9016883 [DOI] [PubMed] [Google Scholar]
- 50.Zamora-Pantoja DR, Quinones-Ramirez EI, Fernandez FJ, Vazquez-Salinas C (2013) Virulence factors involved in the pathogenesis of Vibrio parahaemolyticus. Reviews in Medical Microbiology 24: 41–47. [Google Scholar]
- 51.Lill R (2009) Function and biogenesis of iron-sulphur proteins. Nature 460: 831–838. 10.1038/nature08301 [DOI] [PubMed] [Google Scholar]
- 52.Trotter V, Vinella D, Loiseau L, De Choudens SO, Fontecave M, et al. (2009) The CsdA cysteine desulphurase promotes Fe/S biogenesis by recruiting Suf components and participates to a new sulphur transfer pathway by recruiting CsdL (ex-YgdL), a ubiquitin-modifying-like protein. Molecular Microbiology 74: 1527–1542. [DOI] [PubMed] [Google Scholar]
- 53.Wollers S, Layer G, Garcia-Serres R, Signor L, Clemancey M, et al. (2010) Iron-sulfur (Fe-S) cluster assembly: the SufBCD complex is a new type of Fe-S scaffold with a flavin redox cofactor. J Biol Chem 285: 23331–23341. 10.1074/jbc.M110.127449 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Johnson DC, Dean DR, Smith AD, Johnson MK (2005) Structure, function, and formation of biological iron-sulfur clusters. Annu Rev Biochem 74: 247–281. 10.1146/annurev.biochem.74.082803.133518 [DOI] [PubMed] [Google Scholar]
- 55.Raulfs EC, O'Carroll IP, Dos Santos PC, Unciuleac MC, Dean DR (2008) In vivo iron-sulfur cluster formation. Proc Natl Acad Sci U S A 105: 8591–8596. 10.1073/pnas.0803173105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Crow A, Lawson TL, Lewin A, Moore GR, Le Brun NE (2009) Structural basis for iron mineralization by bacterioferritin. J Am Chem Soc 131: 6808–6813. 10.1021/ja8093444 [DOI] [PubMed] [Google Scholar]
- 57.Shvyreva US, Tutukina MN, Ozoline ON (2011) Bacterioferritin: Properties, structural and functional organization of the dps gene regulatory region. Biophysics 56: 795–802. [PubMed] [Google Scholar]
- 58.Troxell B, Hassan HM (2013) Transcriptional regulation by Ferric Uptake Regulator (Fur) in pathogenic bacteria. Front Cell Infect Microbiol 3: 59 10.3389/fcimb.2013.00059 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Fillat MF (2014) The FUR (ferric uptake regulator) superfamily: diversity and versatility of key transcriptional regulators. Arch Biochem Biophys 546: 41–52. 10.1016/j.abb.2014.01.029 [DOI] [PubMed] [Google Scholar]
- 60.Aiba H (2007) Mechanism of RNA silencing by Hfq-binding small RNAs. Curr Opin Microbiol 10: 134–139. 10.1016/j.mib.2007.03.010 [DOI] [PubMed] [Google Scholar]
- 61.Masse E, Salvail H, Desnoyers G, Arguin M (2007) Small RNAs controlling iron metabolism. Curr Opin Microbiol 10: 140–145. 10.1016/j.mib.2007.03.013 [DOI] [PubMed] [Google Scholar]
- 62.Palyada K, Sun YQ, Flint A, Butcher J, Naikare H, et al. (2009) Characterization of the oxidative stress stimulon and PerR regulon of Campylobacter jejuni. BMC Genomics 10: 481 10.1186/1471-2164-10-481 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Poole K (2012) Stress responses as determinants of antimicrobial resistance in Gram-negative bacteria. Trends Microbiol 20: 227–234. 10.1016/j.tim.2012.02.004 [DOI] [PubMed] [Google Scholar]
- 64.Rojas V, Galanti N, Bols NC, Marshall SH (2009) Productive infection of Piscirickettsia salmonis in macrophages and monocyte-like cells from rainbow trout, a possible survival strategy. J Cell Biochem 108: 631–637. 10.1002/jcb.22295 [DOI] [PubMed] [Google Scholar]
- 65.Rojas V, Galanti N, Bols NC, Jimenez V, Paredes R, et al. (2010) Piscirickettsia salmonis induces apoptosis in macrophages and monocyte-like cells from rainbow trout. J Cell Biochem 110: 468–476. 10.1002/jcb.22560 [DOI] [PubMed] [Google Scholar]
- 66.Zink SD, Pedersen L, Cianciotto NP, Abu-Kwaik Y (2002) The Dot/Icm type IV secretion system of Legionella pneumophila is essential for the induction of apoptosis in human macrophages. Infect Immun 70: 1657–1663. 10.1128/IAI.70.3.1657-1663.2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Zhu W, Banga S, Tan Y, Zheng C, Stephenson R, et al. (2011) Comprehensive identification of protein substrates of the Dot/Icm type IV transporter of Legionella pneumophila. PLoS One 6: e17638 10.1371/journal.pone.0017638 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Calloni G, Chen T, Schermann SM, Chang HC, Genevaux P, et al. (2012) DnaK functions as a central hub in the E. coli chaperone network. Cell Rep 1: 251–264. 10.1016/j.celrep.2011.12.007 [DOI] [PubMed] [Google Scholar]
- 69.Evstigneeva ZG, Solov'eva NA, Sidel'nikova LI (2001) [Structure and functions of chaperones and chaperonins (Review)]. Prikl Biokhim Mikrobiol 37: 5–18. [PubMed] [Google Scholar]
- 70.Vabulas RM, Raychaudhuri S, Hayer-Hartl M, Hartl FU (2010) Protein folding in the cytoplasm and the heat shock response. Cold Spring Harb Perspect Biol 2: a004390 10.1101/cshperspect.a004390 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Evans CG, Chang L, Gestwicki JE (2010) Heat shock protein 70 (hsp70) as an emerging drug target. J Med Chem 53: 4585–4602. 10.1021/jm100054f [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Frees D, Brondsted L, Ingmer H (2013) Bacterial proteases and virulence. Subcell Biochem 66: 161–192. 10.1007/978-94-007-5940-4_7 [DOI] [PubMed] [Google Scholar]
- 73.Li XH, Zeng YL, Gao Y, Zheng XC, Zhang QF, et al. (2010) The ClpP protease homologue is required for the transmission traits and cell division of the pathogen Legionella pneumophila. Bmc Microbiology 10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Meibom KL, Dubail I, Dupuis M, Barel M, Lenco J, et al. (2008) The heat-shock protein ClpB of Francisella tularensis is involved in stress tolerance and is required for multiplication in target organs of infected mice. Mol Microbiol 67: 1384–1401. 10.1111/j.1365-2958.2008.06139.x [DOI] [PubMed] [Google Scholar]
- 75.Yanez AJ, Molina C, Haro RE, Sanchez P, Isla A, et al. (2014) Draft Genome Sequence of Virulent Strain AUSTRAL-005 of Piscirickettsia salmonis, the Etiological Agent of Piscirickettsiosis. Genome Announc 2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Faraldo-Gomez JD, Sansom MS (2003) Acquisition of siderophores in gram-negative bacteria. Nat Rev Mol Cell Biol 4: 105–116. 10.1038/nrm1015 [DOI] [PubMed] [Google Scholar]
- 77.Tanabe T, Funahashi T, Nakao H, Miyoshi SI, Shinoda S, et al. (2003) Identification and Characterization of Genes Required for Biosynthesis and Transport of the Siderophore Vibrioferrin in Vibrio parahaemolyticus. Journal of Bacteriology 185: 6938–6949. 10.1128/JB.185.23.6938-6949.2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Chu BC, Peacock RS, Vogel HJ (2007) Bioinformatic analysis of the TonB protein family. Biometals 20: 467–483. 10.1007/s10534-006-9049-4 [DOI] [PubMed] [Google Scholar]
- 79.Mikael LG, Pawelek PD, Labrie J, Sirois M, Coulton JW, et al. (2002) Molecular cloning and characterization of the ferric hydroxamate uptake (fhu) operon in Actinobacillus pleuropneumoniae. Microbiology 148: 2869–2882. 10.1099/00221287-148-9-2869 [DOI] [PubMed] [Google Scholar]
- 80.Miethke M (2013) Molecular strategies of microbial iron assimilation: from high-affinity complexes to cofactor assembly systems. Metallomics 5: 15–28. 10.1039/c2mt20193c [DOI] [PubMed] [Google Scholar]
- 81.Kim H, Lee H, Shin D (2012) The FeoA protein is necessary for the FeoB transporter to import ferrous iron. Biochem Biophys Res Commun 423: 733–738. 10.1016/j.bbrc.2012.06.027 [DOI] [PubMed] [Google Scholar]
- 82.Cartron ML, Maddocks S, Gillingham P, Craven CJ, Andrews SC (2006) Feo—transport of ferrous iron into bacteria. Biometals 19: 143–157. 10.1007/s10534-006-0003-2 [DOI] [PubMed] [Google Scholar]
- 83.Naikare H, Palyada K, Panciera R, Marlow D, Stintzi A (2006) Major role for FeoB in Campylobacter jejuni ferrous iron acquisition, gut colonization, and intracellular survival. Infect Immun 74: 5433–5444. 10.1128/IAI.00052-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Ramakrishnan G, Sen B, Johnson R (2012) Paralogous outer membrane proteins mediate uptake of different forms of iron and synergistically govern virulence in Francisella tularensis tularensis. J Biol Chem 287: 25191–25202. 10.1074/jbc.M112.371856 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Cianciotto NP (2007) Iron acquisition by Legionella pneumophila. Biometals 20: 323–331. 10.1007/s10534-006-9057-4 [DOI] [PubMed] [Google Scholar]
- 86.Almiron MA, Ugalde RA (2010) Iron homeostasis in Brucella abortus: the role of bacterioferritin. J Microbiol 48: 668–673. 10.1007/s12275-010-0145-3 [DOI] [PubMed] [Google Scholar]
- 87.Carrondo MA (2003) Ferritins, iron uptake and storage from the bacterioferritin viewpoint. EMBO J 22: 1959–1968. 10.1093/emboj/cdg215 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Velayudhan J, Castor M, Richardson A, Main-Hester KL, Fang FC (2007) The role of ferritins in the physiology of Salmonella enterica sv. typhimurium: a unique role for ferritin B in iron-sulphur cluster repair and virulence. Mol Microbiol 63: 1495–1507. 10.1111/j.1365-2958.2007.05600.x [DOI] [PubMed] [Google Scholar]
- 89.Ohtsu I, Wiriyathanawudhiwong N, Morigasaki S, Nakatani T, Kadokura H, et al. (2010) The L-cysteine/L-cystine shuttle system provides reducing equivalents to the periplasm in Escherichia coli. J Biol Chem 285: 17479–17487. 10.1074/jbc.M109.081356 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Ewann F, Hoffman PS (2006) Cysteine metabolism in Legionella pneumophila: characterization of an L-cystine-utilizing mutant. Appl Environ Microbiol 72: 3993–4000. 10.1128/AEM.00684-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Cabiscol E, Tamarit J, Ros J (2000) Oxidative stress in bacteria and protein damage by reactive oxygen species. Int Microbiol 3: 3–8. [PubMed] [Google Scholar]
- 92.Michan C, Manchado M, Dorado G, Pueyo C (1999) In vivo transcription of the Escherichia coli oxyR regulon as a function of growth phase and in response to oxidative stress. J Bacteriol 181: 2759–2764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Cornelis P, Wei Q, Andrews SC, Vinckx T (2011) Iron homeostasis and management of oxidative stress response in bacteria. Metallomics 3: 540–549. 10.1039/c1mt00022e [DOI] [PubMed] [Google Scholar]
- 94.Clarke TE, Romanov V, Chirgadze YN, Klomsiri C, Kisselman G, et al. (2011) Crystal structure of alkyl hydroperoxidase D like protein PA0269 from Pseudomonas aeruginosa: homology of the AhpD-like structural family. BMC Struct Biol 11: 27 10.1186/1472-6807-11-27 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Chung CH, Ma TY, Fen SY, Wong HC (2014) Activities of Alkyl Hydroperoxide Reductase Subunits C1 and C2 of Vibrio parahaemolyticus against Different Peroxides. Appl Environ Microbiol 80: 7398–7404. 10.1128/AEM.02701-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Oh E, Jeon B (2014) Role of alkyl hydroperoxide reductase (AhpC) in the biofilm formation of Campylobacter jejuni. PLoS One 9: e87312 10.1371/journal.pone.0087312 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Stokes HW, Gillings MR (2011) Gene flow, mobile genetic elements and the recruitment of antibiotic resistance genes into Gram-negative pathogens. FEMS Microbiol Rev 35: 790–819. 10.1111/j.1574-6976.2011.00273.x [DOI] [PubMed] [Google Scholar]
- 98.Wellington EM, Boxall AB, Cross P, Feil EJ, Gaze WH, et al. (2013) The role of the natural environment in the emergence of antibiotic resistance in gram-negative bacteria. Lancet Infect Dis 13: 155–165. 10.1016/S1473-3099(12)70317-1 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(XLSX)
(XLSX)
(XLSX)
(A) Suggested genetic organization of siderophore synthesis and transport genes transcribed from the P. salmonis genome that could represent a putative operon. The RPKM values of each of genes that were expressed in the pathway are showed for the CFC-I growth condition. (B) Siderophore synthesis is accomplished by the PvsA, PvsB, PvsD and PvsE proteins. The PvsC protein transports the siderophore to the extracellular space, where it captures Fe+3 and is uptaked by the outer membrane protein PvuA. (OM: outer membrane; CM: cytoplasmatic membrane).
(TIFF)
(A) The organization of tonB, ferric aerobactin, ABC transporter and the iron (III) dicitrate transport system permease protein FecD genes could represent an operon separated from the exbD/tolQ and motA/tolQ/exbB proton channel family protein genes. The RPKM values of each of genes that were expressed in the pathway are showed for the CFC-I growth condition. (B) There are two putative genes encoding transmembrane cytoplamatic proteins which transport ferri-siderophores from the periplasmic space into the cytoplasm where these molecules are reduced. (OM: outer membrane; CM: cytoplasmatic membrane).
(TIFF)
The RPKM values of each of genes that were expressed in the pathway are showed for the CFC-I growth condition. When the cell has high levels of [Fe-S] clusters, the IscR protein would capture a cluster in its structure, which is a signal for transcriptional repression of sufB, sufC, sufD, and sufS.
(TIFF)
Data Availability Statement
Genome assembly files are available from the NCBI database (Whole Genome Shotgun (WGS) project accession number LELB01000000). All other relevant data are within the paper and its Supporting Information files.