Abstract
Li-Fraumeni syndrome (LFS) is a cancer predisposition disorder caused by germline mutations in TP53 that can lead to increased mitochondrial metabolism in patients. However, the implications of altered mitochondrial function for tumorigenesis in LFS are unclear. Here, we have reported that genetic or pharmacologic disruption of mitochondrial respiration improves cancer-free survival in a mouse model of LFS that expresses mutant p53. Mechanistically, inhibition of mitochondrial function increased autophagy and decreased the aberrant proliferation signaling caused by mutant p53. In a pilot study, LFS patients treated with metformin exhibited decreases in mitochondrial activity concomitant with activation of antiproliferation signaling, thus reproducing the effects of disrupting mitochondrial function observed in LFS mice. These observations indicate that a commonly prescribed diabetic medicine can restrain mitochondrial metabolism and tumorigenesis in an LFS model, supporting its further consideration for cancer prevention in LFS patients.
Introduction
Growing evidence suggests that the mitochondrion plays a critical role in cancer development through various processes including metabolism, ROS generation, and cell signaling (1, 2). Increased oxidative phosphorylation and fatty acid oxidation have been observed in diffuse large B cell lymphoma as well as chronic lymphocytic leukemia, and the master regulator of mitochondrial biogenesis PGC-1α promotes cancer metastasis (3, 4). Furthermore, mitochondrial DNA–depleted (mtDNA-depleted) cancer cells cannot metastasize until they acquire host mtDNA (5). Mitochondria appear to be essential for cancer progression; however, their role in de novo tumor formation remains unclear.
Li-Fraumeni syndrome (LFS) is an autosomal dominant cancer predisposition syndrome caused predominately by germline mutations of TP53, encoding the tumor suppressor protein p53 (6). We previously reported that LFS family members who carried TP53 mutations have increased oxidative metabolism (7). These findings were confirmed in a mouse model of LFS that demonstrated the distinction between p53 null and p53 mutant states in regulating the mitochondria (8, 9). Although the role of mitochondria in tumorigenesis in LFS remains unclear, their function is tightly integrated with cell survival and proliferation, suggesting their involvement. Therefore, we hypothesized that disrupting mitochondrial function in an LFS model would affect de novo tumorigenesis. Furthermore, we explored the feasibility of attenuating mitochondrial activity in LFS patients utilizing metformin, a well-tolerated antidiabetic medication that is known to inhibit respiration, but has not been demonstrated as doing so in humans.
Results and Discussion
Genetic disruption of mitochondria delays de novo tumorigenesis.
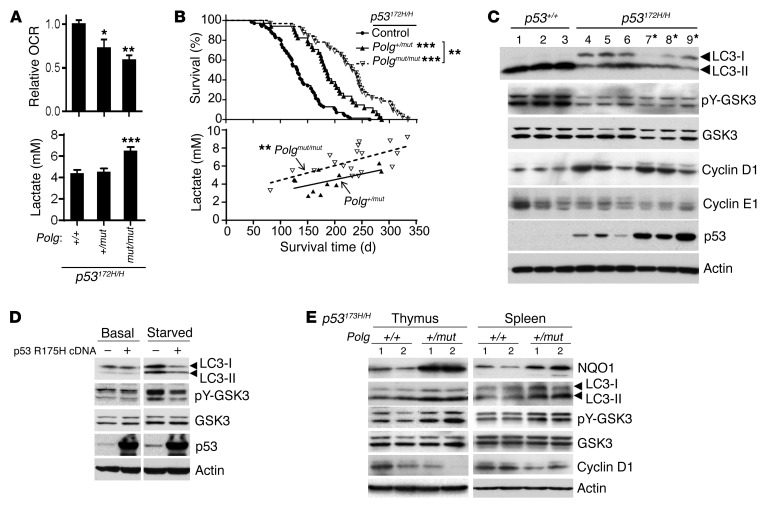
Mitochondrial function in an established mouse model of LFS (p53172H/H), created by knockin of the mouse equivalent of human p53 R175H hotspot mutation (10), was directly disrupted by introducing the DNA polymerase γ D257A mutant (Polgmut) to generate frequent mtDNA mutations (11). Because the majority of p53172H/H mice develop thymic lymphomas, we measured mitochondrial respiration of thymus tissue, which revealed decreased oxygen consumption rate (OCR) in a Polgmut allele dose-dependent manner (Figure 1A). This decrease in oxidative metabolism was associated with increased blood lactate levels (Figure 1A). Similarly, decreased mitochondrial complex I, but increased glucose transporter 4 and hexokinase 2, which promote glycolysis, were observed in the skeletal muscle of Polg mutant mice (Supplemental Figure 1; supplemental material and unedited blots available online with this article; doi:10.1172/JCI88668DS1). This metabolic reprogramming markedly increased cancer-free survival of LFS mice. Compared with p53172H/H mice, the median survival time of Polg+/mut p53172H/H and Polgmut/mut p53172H/H double-mutant mice was increased by 40% and 79%, respectively (Figure 1B). Further demonstrating the physiological effect of mitochondrial inhibition on p53172H/H mouse survival, a positive correlation between blood lactate level and survival time was observed in the double-mutant mice (Figure 1B). In addition, the increased cancer-free survival time by Polg mutation in homozygous p53172H/H mice was also reproduced in heterozygous p53172R/H mice, which have a much longer life span and more closely model the genotype that causes LFS in humans (Supplemental Figure 2A).
Figure 1. Genetic disruption of mitochondria reverses aberrant proliferation signaling and increases survival of p53172H/H mice.
(A) OCRs of approximately 10-week-old mouse thymus cells (n = 5–7) and lactate in tail blood (average age approximately 12 weeks) (n = 14–19). (B) Upper panel shows Kaplan-Meier survival plot of p53172H/H control (n = 70), Polg+/mut p53172H/H (n = 34), and Polgmut/mut p53172H/H (n = 32) mice. Lower panel shows correlation between blood lactate level and survival time of Polg+/mut p53172H/H (r = 0.6, P = 0.057, n = 11) and Polgmut/mut p53172H/H (r = 0.7, P < 0.01, n = 18) mice. *P < 0.05; **P < 0.01; ***P < 0.001. (C) lmmunoblots of approximately 10-week-old p53+/+ and p53172H/H thymus tissues. Asterisks (lanes 7–9) indicate enlarged thymus samples with lymphoma. (D) Primary human myoblasts transduced with empty vector (–) or human p53 R175H cDNA (+) were serum starved for 3 hours prior to immunoblotting. (E) lmmunoblots of thymus and spleen samples obtained from age-matched Polg+/+ and Polg+/mut mice in p53172H/H background (lane 1, approximately 10 weeks old; lane 2, approximately 20 weeks old). Statistical differences by 1-way ANOVA (A), log-rank test (B, upper), and Pearson correlation coefficient test (B, lower).
Inhibiting respiration reverses aberrant proliferation signaling in LFS thymus.
More than 70% of p53172H/H mice developed lymphomas in the thymus (Supplemental Table 1); we therefore examined this tissue to identify biomarkers of mitochondrial regulated cell signaling. One mechanism of p53 tumor suppression is activation of autophagy, while mutant p53, including the human R175H mutation, can act in a protumorigenic manner by inhibition of autophagy (12, 13). Indeed, the thymic tissue of p53172H/H mice showed decreased levels of autophagosome membrane–associated LC3-II (lanes 4–9 versus 1–3, Figure 1C). Autophagy and associated endoplasmic reticulum stress can activate glycogen synthase kinase 3α/β by stimulating its autophosphorylation at tyrosine 276/216 (pY-GSK3) (14–16). Active GSK3 is necessary for downregulating cyclin D1 (17), a critical driver for cell proliferation that promotes tumorigenesis, including lymphoma formation (18, 19). Accordingly, increased cyclin D1 was inversely correlated with decreased pY-GSK3 and autophagy in p53172H/H mice along with reduced autophagy (lanes 4–9, Figure 1C). Cyclin E1, also, like cyclin D1, involved in G1 cell cycle regulation, remained unchanged in p53172H/H mice, indicating a specific defect in the cyclin D1 pathway (Figure 1C). To further demonstrate the effect of p53 R172H mutant on this signaling pathway, we examined the response of human primary myoblasts to serum deprivation. After 3 hours of serum starvation, control cells displayed increased autophagy and pY-GSK3, both of which were blocked by expressing human p53 R175H (homologous to mouse R172H) (Figure 1D). In vivo, LFS mouse thymus tissue also showed changes that were consistent with the inhibitory effects of mutant p53 on autophagy and GSK3 signaling (Supplemental Figure 3).
One direct effect of inhibiting mitochondrial respiration is lower mitochondrial membrane potential, which can induce mitophagy/autophagy, potentially disrupting proliferation signaling in LFS mice. Because homozygous Polgmut/mut mice have accelerated involution of the thymus, we focused on the heterozygous Polg+/mut, which causes no overt phenotype (11), but conferred a substantial increase in cancer-free survival of p53172H/H mice. Notably, the Polg+/mut state caused decreases in both mitochondrial complex I subunit and activity levels in mutant p53 R172H thymus and spleen (Supplemental Figure 4). This disruption of mitochondria induced autophagy and pY-GSK3 as well as NAD(P)H dehydrogenase quinone 1 (NQO1) levels, while cyclin D1 was downregulated despite the presence of mutant p53 (Figure 1E). Polg mutation also activated the autophagy/GSK/cyclin D1 pathway in approximately 1-year-old heterozygous p53172R/H mice, indicating the preservation of this cell signaling in the genotype modeling LFS patients (Supplemental Figure 2B).
Metformin can reproduce the effects of genetically disrupting mitochondrial function in LFS mice.
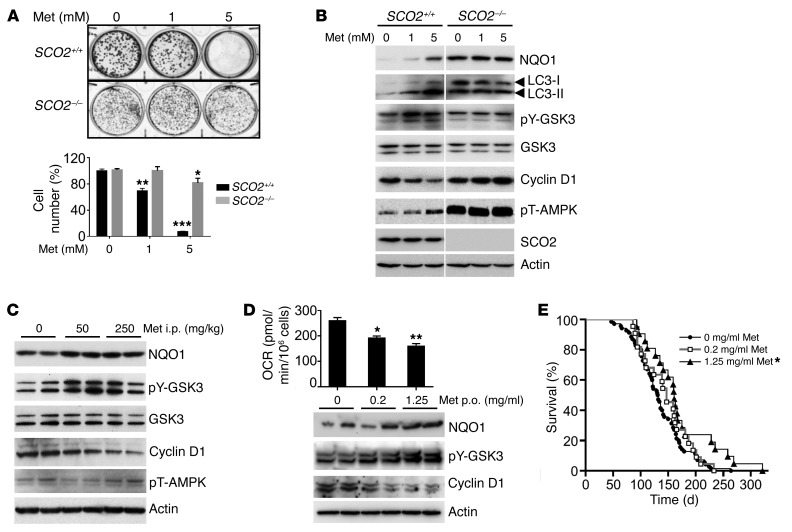
Based on its known inhibitory effect on mitochondria and excellent safety profile in humans, metformin represented an ideal therapeutic agent for a test of whether it can reproduce the antiproliferation signaling induced by Polg mutation. Because metformin has pleiotropic effects on cells, we utilized a pair of isogenic respiratory-proficient and -deficient cell lines to demonstrate that its antiproliferation effect acts via inhibition of respiration. Metformin inhibited the proliferation of wild-type human colon cancer HCT116 cells, but did not markedly affect nonrespiring cells with homozygous deletion of synthesis of cytochrome c oxidase 2 (SCO2–/–), an essential factor for the assembly of respiratory complex IV (Figure 2A) (20). In parallel, metformin reproduced the in vivo cell signaling changes caused by mitochondrial disruption only in respiring SCO2+/+ cells (Figure 2B). Despite the high concentration of metformin used in vitro, the autophagy/GSK3/cyclin D1 signaling components remained unchanged in SCO2–/– cells with high basal levels of NQO1 and phosphorylated AMPK (pT-AMPK) (Figure 2B). It is notable that, although pT-AMPK is thought to be a major target of metformin, pY-GSK3 appeared to be more sensitive to metformin than pT-AMPK in respiring cells, as seen by its strong induction at 1 mM compared with 5 mM for pT-AMPK (Figure 2B).
Figure 2. Metformin induces antiproliferation signaling by inhibiting respiration and prevents tumorigenesis.
(A) Crystal violet staining and quantification of SCO2+/+ and SCO2–/– HCT116 cells after metformin (Met) treatment for 10 days (n = 4–6). (B) HCT116 cells treated with metformin overnight prior to immunoblotting. pT-AMPK is from a separate gel using the same samples. Representative of 3 experiments. (C) p53172H/H mice (approximately 10 weeks old) were injected (i.p.) with metformin daily for 1 week prior to thymus immunoblotting. (D) OCR and immunoblots of thymus from approximately 7-week-old p53172H/H mice treated with metformin by mouth (p.o) in drinking water for 3 weeks (n = 6–7). (E) Kaplan-Meier survival plot of p53172H/H mouse treated with metformin in drinking water (n = 21–70). Statistical difference by 2-way ANOVA (A), 1-way ANOVA (D), and log-rank test (E). *P < 0.01; **P < 0.001; ***P < 0.0001.
Questions have been raised about the clinical relevance of in vitro findings using millimolar concentrations of metformin versus the micromolar range plasma levels observed in patients on therapeutic doses of metformin (21). Thus, we examined whether treating LFS mice with metformin at human therapeutic levels would induce antiproliferation signaling (22). The i.p. injection of metformin at 250 mg/kg body weight reproduced the autophagy/GSK3/cyclin D1 signaling cascade, as observed in cultured cells (Figure 2C). Notably, pY-GSK and cyclin D1 levels were again more sensitive to metformin than pT-AMPK (Figure 2C). This was consistent with the report that pT-AMPK induction by metformin cannot be detected in tissues in contrast with cultured cells (23).
Based on these findings, we tested the feasibility of a cancer-free survival study in p53172H/H mice treated with metformin in drinking water. A dose of 1.25 mg/ml metformin in drinking water was previously shown to result in plasma concentrations within the therapeutic range observed in humans and to inhibit xenograft growth in mice (22, 24). The thymus of mice fed metformin showed a dose-dependent decrease in oxygen consumption and the induction of antiproliferation signaling cascade (Figure 2D). Initiating treatment with 1.25 mg/ml metformin after the mice were weaned (~4 weeks old) increased its median and mean cancer-free survival times by 22% and 27%, respectively, while 0.2 mg/ml metformin treatment showed only a trend of increased survival time (Figure 2E). Although less effective in prolonging cancer-free survival than the genetic approach, the improvement in survival by metformin treatment was nonetheless remarkable, as lymphomas can develop early in p53172H/H mice.
Metformin inhibits mitochondrial function and activates antiproliferation signaling in LFS patients.
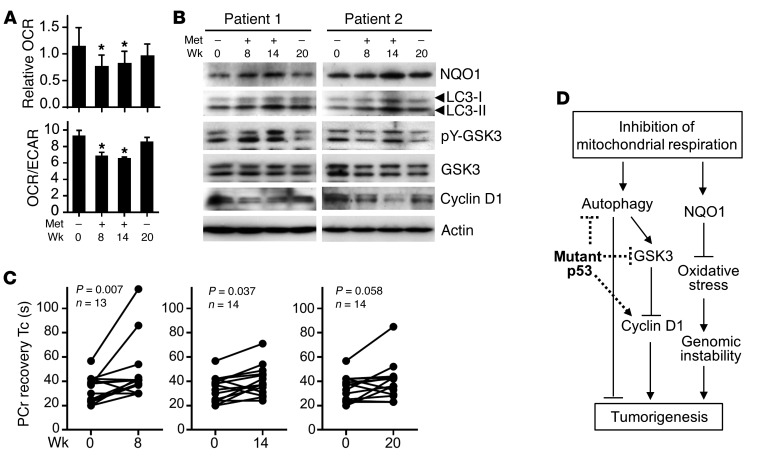
We next examined whether metformin could elicit similar effects in LFS patients. As part of a clinical protocol examining the safety and tolerability of metformin, healthy LFS patients carrying germline TP53 mutations were examined for mitochondrial function and antiproliferation biomarkers before (week 0), during (weeks 8 and 14), and after (week 20) metformin treatment (Supplemental Table 2 and Supplemental Figure 5A) (25). Blood samples were obtained at the indicated time points, and mononuclear cells (MNCs) were isolated for simultaneous measurements of OCR and extracellular acidification rate (ECAR) as markers of oxidative and glycolytic metabolism, respectively. Metformin treatment at the maximum dose of 2,000 mg daily at weeks 8 and 14 substantially reduced OCR as well as the OCR to ECAR ratio as an index of oxidative metabolism, while 6 weeks of medication washout at week 20 resulted in recovery of respiration (Figure 3A and Supplemental Figure 5B). As a control, we confirmed that the acute removal of metformin in preparation for the assay does not affect the inhibition (Supplemental Figure 6). The metformin treatment reproduced the pattern of increased NQO1, LC3-II, and pY-GSK3 activation and decreased cyclin D1 in MNCs (Figure 3B). Finally, we measured skeletal muscle phosphocreatine (PCr) recovery kinetics after exercise in vivo as a marker of oxidative phosphorylation capacity using phosphorus-31 magnetic resonance spectroscopy (31P-MRS) (7). Treatment with metformin (weeks 8 and 14) increased the PCr recovery time constant, providing in vivo evidence of decreased mitochondrial activity that is necessary for regenerating PCr in muscle (Figure 3C).
Figure 3. Metformin inhibits mitochondria and activates antiproliferation signaling in LFS patients.
(A) Effect of metformin on OCR and OCR/ECAR ratio of LFS patient blood MNCs (n = 13–14). (B) Representative immunoblots of MNCs from 2 out of 6 examined patients. (C) Effect of metformin treatment on PCr recovery time constant (Tc) after exercise in patient skeletal muscle. (D) Mitochondrial inhibition counteracts tumor-promoting activities of mutant p53 (dotted lines). Statistical differences by 1-way ANOVA (A) and Wilcoxon matched-pairs signed rank test (C). *P < 0.01.
In the current study, we have demonstrated that the genetic perturbation of mitochondrial respiration in a mouse model of LFS can markedly improve cancer-free survival in association with inhibition of the aberrant proliferation signaling caused by p53 mutation (Figure 3D). Pharmacologically inhibiting mitochondrial respiration using metformin also improved cancer-free survival and reproduced the cell-signaling cascade observed in the genetic model. Importantly, treatment of LFS patients with metformin inhibited respiration in blood MNCs and evoked similar anti–cell proliferation signaling that could serve as treatment biomarkers. Furthermore, we have provided in vivo evidence that metformin can decrease mitochondrial activity in the tissue of LFS patients, which is known to give rise to rhabdomyosarcoma, a cancer associated with the syndrome.
One possible mechanism by which mitochondrial disruption prevented tumorigenesis is through increased oxidative stress. However, the overall decrease in cellular ROS levels associated with decreased nuclear NRF2 suggested that increased oxidative stress may not be the major mechanism underlying our observations (Supplemental Figure 7). The marked improvement in survival of p53172H/H mice by the homozygous mutation of Polg could be explained by increased apoptosis, but this was not detected in the heterozygous Polg mutant state, as previously reported (Supplemental Figure 8) (11, 26). Although our study was not designed to address whether inhibiting respiration prevents cancer specifically in LFS, our findings are in agreement with the growing evidence indicating the importance of mitochondria in tumorigenesis. In this regard, inhibiting the expression of citrate transport protein (CTP), a gene that is induced by a gain-of-function p53 mutant and increases mitochondrial respiration, blunts the accelerated xenograft growth of both p53 mutant and p53 null cancer cells (27). Such observations point to the essential role of the mitochondrion in tumorigenesis regardless of p53 status and intrinsic mitochondrial function.
In summary, our study provides evidence that disrupting mitochondrial respiration can delay cancer development in LFS, a cancer predisposition syndrome for which there is currently no therapeutic option. Despite epidemiologic evidence of decreased cancer incidence associated with metformin treatment in diabetic patients, biomarkers correlating its treatment effect to tumor suppression have not been identified. By using a LFS mouse model with concurrent translation of the findings to patients, our study provides in vivo evidence of the inhibitory effects of metformin on mitochondria and provides a mechanistic rationale for investigating the chemopreventive efficacy of metformin in populations at high risk of cancer.
Methods
Statistics.
GraphPad Prism software (version 6.02) was used for 1-way ANOVA test for multiple comparisons, the log-rank test for animal survival, the Pearson correlation coefficient test, and the Wilcoxon matched-pairs signed rank test (2-tailed) for before and after metformin treatment in patients. Values are shown as mean ± SEM. A P value of less than 0.05 was considered significant.
Study approval.
All animal procedures were approved by the Animal Care and Use Committee of the NHLBI-NIH. All human subjects provided written informed consent. The IRB of the NCI and NHLBI approved the protocols (ClinicalTrials.gov NCT01981525 and NCT00406445).
See Supplemental Methods for additional information.
Author contributions
PYW, JL, JGK, MFS, SLT, JZ, JHP, and PMH designed and performed the experimental studies. FLW, PLM, MP, CMA, SAS, ATF, and PMH designed and performed the clinical studies. RDH and CMB managed patients. PYW, JL, and PMH wrote the manuscript.
Supplementary Material
Acknowledgments
This work was supported by the Division of Intramural Research, NHLBI-NIH (HL005101 to P.M. Hwang). We thank J. Stolinski, R. Hill, and H. Merkle for MRS studies; J. Chavez, R. Fleischmann, A. Merriweather, and C. King for pathology services; T. Finkel for helpful comments and the Polg+/mut mouse created by T.A. Prolla; M.A. Waclawiw for advice with statistical analysis; and N.A. Long and W.M. Kamp for technical and editorial assistance.
Footnotes
Conflict of interest: The authors have declared that no conflict of interest exists.
Reference information:J Clin Invest. 2017;127(1):132–136. doi:10.1172/JCI88668.
Contributor Information
Ping-yuan Wang, Email: wangp4@nhlbi.nih.gov.
Jie Li, Email: jie.li4@nih.gov.
Ju-Gyeong Kang, Email: kangju@mail.nih.gov.
Matthew F. Starost, Email: starostm@ors.od.nih.gov.
Jie Zhuang, Email: zhuangj1@mail.nih.gov.
Ji-Hoon Park, Email: Ji-hoon.park@nih.gov.
Michael Pollak, Email: michael.pollak@mcgill.ca.
References
- 1.Weinberg F, et al. Mitochondrial metabolism and ROS generation are essential for Kras-mediated tumorigenicity. Proc Natl Acad Sci U S A. 2010;107(19):8788–8793. doi: 10.1073/pnas.1003428107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Weinberg SE, Chandel NS. Targeting mitochondria metabolism for cancer therapy. Nat Chem Biol. 2015;11(1):9–15. doi: 10.1038/nchembio.1712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Caro P, et al. Metabolic signatures uncover distinct targets in molecular subsets of diffuse large B cell lymphoma. Cancer Cell. 2012;22(4):547–560. doi: 10.1016/j.ccr.2012.08.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.LeBleu VS, et al. PGC-1α mediates mitochondrial biogenesis and oxidative phosphorylation in cancer cells to promote metastasis. Nat Cell Biol. 2014;16(10):992–1003. doi: 10.1038/ncb3039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tan AS, et al. Mitochondrial genome acquisition restores respiratory function and tumorigenic potential of cancer cells without mitochondrial DNA. Cell Metab. 2015;21(1):81–94. doi: 10.1016/j.cmet.2014.12.003. [DOI] [PubMed] [Google Scholar]
- 6.Malkin D, et al. Germ line p53 mutations in a familial syndrome of breast cancer, sarcomas, and other neoplasms. Science. 1990;250(4985):1233–1238. doi: 10.1126/science.1978757. [DOI] [PubMed] [Google Scholar]
- 7.Wang PY, et al. Increased oxidative metabolism in the Li-Fraumeni syndrome. N Engl J Med. 2013;368(11):1027–1032. doi: 10.1056/NEJMoa1214091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zhuang J, Wang PY, Huang X, Chen X, Kang JG, Hwang PM. Mitochondrial disulfide relay mediates translocation of p53 and partitions its subcellular activity. Proc Natl Acad Sci U S A. 2013;110(43):17356–17361. doi: 10.1073/pnas.1310908110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kamp WM, Wang PY, Hwang PM. TP53 mutation, mitochondria and cancer. Curr Opin Genet Dev. 2016;38:16–22. doi: 10.1016/j.gde.2016.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lang GA, et al. Gain of function of a p53 hot spot mutation in a mouse model of Li-Fraumeni syndrome. Cell. 2004;119(6):861–872. doi: 10.1016/j.cell.2004.11.006. [DOI] [PubMed] [Google Scholar]
- 11.Kujoth GC, et al. Mitochondrial DNA mutations, oxidative stress, and apoptosis in mammalian aging. Science. 2005;309(5733):481–484. doi: 10.1126/science.1112125. [DOI] [PubMed] [Google Scholar]
- 12.Morselli E, et al. Mutant p53 protein localized in the cytoplasm inhibits autophagy. Cell Cycle. 2008;7(19):3056–3061. doi: 10.4161/cc.7.19.6751. [DOI] [PubMed] [Google Scholar]
- 13.Bieging KT, Mello SS, Attardi LD. Unravelling mechanisms of p53-mediated tumour suppression. Nat Rev Cancer. 2014;14(5):359–370. doi: 10.1038/nrc3711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lochhead PA, Kinstrie R, Sibbet G, Rawjee T, Morrice N, Cleghon V. A chaperone-dependent GSK3beta transitional intermediate mediates activation-loop autophosphorylation. Mol Cell. 2006;24(4):627–633. doi: 10.1016/j.molcel.2006.10.009. [DOI] [PubMed] [Google Scholar]
- 15.Chiara F, et al. Chemotherapeutic induction of mitochondrial oxidative stress activates GSK-3α/β and Bax, leading to permeability transition pore opening and tumor cell death. Cell Death Dis. 2012;3:e444. doi: 10.1038/cddis.2012.184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Han G, Casson RJ, Chidlow G, Wood JP. The mitochondrial complex I inhibitor rotenone induces endoplasmic reticulum stress and activation of GSK-3β in cultured rat retinal cells. Invest Ophthalmol Vis Sci. 2014;55(9):5616–5628. doi: 10.1167/iovs.14-14371. [DOI] [PubMed] [Google Scholar]
- 17.Diehl JA, Cheng M, Roussel MF, Sherr CJ. Glycogen synthase kinase-3beta regulates cyclin D1 proteolysis and subcellular localization. Genes Dev. 1998;12(22):3499–3511. doi: 10.1101/gad.12.22.3499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tsikitis M, Zhang Z, Edelman W, Zagzag D, Kalpana GV. Genetic ablation of Cyclin D1 abrogates genesis of rhabdoid tumors resulting from Ini1 loss. Proc Natl Acad Sci U S A. 2005;102(34):12129–12134. doi: 10.1073/pnas.0505300102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lovec H, Grzeschiczek A, Kowalski MB, Möröy T. Cyclin D1/bcl-1 cooperates with myc genes in the generation of B-cell lymphoma in transgenic mice. EMBO J. 1994;13(15):3487–3495. doi: 10.1002/j.1460-2075.1994.tb06655.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Matsumoto T, Wang PY, Ma W, Sung HJ, Matoba S, Hwang PM. Polo-like kinases mediate cell survival in mitochondrial dysfunction. Proc Natl Acad Sci U S A. 2009;106(34):14542–14546. doi: 10.1073/pnas.0904229106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chandel NS, et al. Are metformin doses used in murine cancer models clinically relevant? Cell Metab. 2016;23(4):569–570. doi: 10.1016/j.cmet.2016.03.010. [DOI] [PubMed] [Google Scholar]
- 22.Memmott RM, Mercado JR, Maier CR, Kawabata S, Fox SD, Dennis PA. Metformin prevents tobacco carcinogen--induced lung tumorigenesis. Cancer Prev Res (Phila) 2010;3(9):1066–1076. doi: 10.1158/1940-6207.CAPR-10-0055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Martin-Montalvo A, et al. Metformin improves healthspan and lifespan in mice. Nat Commun. 2013;4:2192. doi: 10.1038/ncomms3192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wheaton WW, et al. Metformin inhibits mitochondrial complex I of cancer cells to reduce tumorigenesis. Elife. 2014;3:e02242. doi: 10.7554/eLife.02242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Walcott FL, et al. Abstract 29: Design of a phase I chemoprevention study of metformin and Li-Fraumeni syndrome (LFS) Cancer Research. 2014;74(23):29. doi: 10.1158/1538-7445.CANSUSC14-29. doi:10.1158/1538-7445.CANSUSC14-29. [DOI] [Google Scholar]
- 26.Trifunovic A, et al. Somatic mtDNA mutations cause aging phenotypes without affecting reactive oxygen species production. Proc Natl Acad Sci U S A. 2005;102(50):17993–17998. doi: 10.1073/pnas.0508886102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kolukula VK, et al. SLC25A1, or CIC, is a novel transcriptional target of mutant p53 and a negative tumor prognostic marker. Oncotarget. 2014;5(5):1212–1225. doi: 10.18632/oncotarget.1831. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.