Abstract
DNA methylation and histone modifications interact to modulate gene expression in biological organisms. The histone demethylase IBM1 suppresses DNA methylation and gene silencing, primarily by targeting genic regions in the Arabidopsis genome. The chromatin regulator EDM2 is also required for prevention of genic DNA methylation because it maintains IBM1 expression by promoting IBM1 mRNA distal polyadenylation. Loss-of-function ibm1 and edm2 mutant plants display a wide range of developmental defects, but little is known about which developmentally important genes are regulated by IBM1 and EDM2. Here, we show that both ibm1 and edm2 mutants display defects in production of stomatal lineage cells, which is linked to DNA hypermethylation of the ERECTA family genes, including ER, ERL1 and ERL2. Stomatal phenotypes and DNA methylation levels of ER genes in ibm1 and edm2 mutants are restored by mutations in the genes encoding the histone methyltransferase KYP and DNA methyltransferase CMT3. Our data demonstrate that a specific plant developmental context is influenced by IBM1-regulated histone modification and DNA methylation on the gene body region of the ERECTA receptors.
KEY WORDS: ER receptors, DNA methylation, Stomatal development, IBM1 histone demethylase, EDM2 chromatin regulator
Summary: The histone demethylase IBM1 regulates Arabidopsis stomatal development by suppressing DNA methylation on the gene body region of ERECTA receptors.
INTRODUCTION
DNA cytosine methylation and histone modification closely interact to silence gene expression (Fuks, 2005). Histone 3 lysine 9 (H3K9) dimethylation is associated with heterochromatin formation, DNA hypermethylation and thereby gene silencing in both plants and mammals (Badeaux and Shi, 2013; Liu et al., 2010). INCREASE IN BOSAI METHYLATION 1 (IBM1) encodes a histone demethylase that contains a JmjC-domain and belongs to the JHDM2/KDM3 family (Klose et al., 2006; Saze et al., 2008). Mutations in IBM1 not only result in increased H3K9 dimethylation, but also cause ectopic DNA methylation, mainly in gene bodies but not transposons (Inagaki et al., 2010; Miura et al., 2009). Recently, the putative chromatin regulator ENHANCED DOWNY MILDEW 2 (EDM2) was found to function upstream of IBM1 to maintain the expression of functional IBM1 by promoting the distal polyadenylation of its transcripts (Lei et al., 2014). Consistent with this direct genetic interaction between IBM1 and EDM2, almost 90% of hypermethylated loci in edm2 mutant plants overlapped with those in the ibm1 mutant (Lei et al., 2014). As a consequence of the extensive DNA hypermethylation in gene bodies, both edm2 and ibm1 mutations affect plant development in many aspects. The edm2 and ibm1 mutants show abnormally curved leaves, arrested flowers, reduced fertility and aborted seed development (Saze et al., 2008). EDM2 also promotes floral transition by acting upstream of FLOWERING LOCUS C (FLC) (Tsuchiya and Eulgem, 2010a) and facilitates the growth features related to vegetative phase change in Arabidopsis (Tsuchiya and Eulgem, 2010b). Thus far, little progress has been made towards understanding the specific downstream genes regulated by IBM1 and EDM2 in plant developmental programs.
The RNA-directed DNA methylation (RdDM) pathway is required for de novo DNA methylation and maintenance of methylation in the CHH (where H is A, T or C) sequence context (Matzke et al., 2015; Zhang and Zhu, 2011; Law and Jacobsen, 2010). RdDM requires tightly coordinated activities of DOMAINS REARRANGED METHYLTRANSFERASE 2 (DRM2), RNA POLYMERASE II-RELATED RNA POLYMERASE IV and V (Pol IV and V) (Wierzbicki et al., 2008; Zheng et al., 2009), RNA-DEPENDENT RNA POLYMERASE 2 (RDR2), DICER-LIKE 3 (DCL3), ARGONAUTE 4 and 6 (AGO4/6), and many other proteins (Matzke et al., 2015). Maintenance of CG and CHG methylation requires MET1 and CMT3, respectively (He et al., 2011; Law and Jacobsen, 2010). DNA methylation by the plant-specific DNA methyltransferase CMT3 (Bartee et al., 2001; Lindroth et al., 2001) is guided by dimethylation of histone H3 at lysine 9 (H3K9me2) (Malagnac et al., 2002), which is catalyzed by the histone methyltransferase KRYPTONITE (KYP; also known as SUVH4) (Jackson et al., 2002; Ebbs et al., 2005). Mutations in the histone H3K9me2 demethylase IBM1 cause widespread gene body CHG methylation and developmental defects in Arabidopsis (Saze et al., 2008). The defects of ibm1 mutants are fully suppressed by cmt3 and kyp mutations, but not by mutations in the RdDM machinery (Miura et al., 2009; Saze et al., 2008).
Stomata are microscopic pores in the epidermis of leaves, stems and many other aerial organs in plants that control gas exchange between plants and the atmosphere. The production and patterning of stomata are controlled by a relatively linear signaling pathway (reviewed by Lau and Bergmann, 2012; Pillitteri and Dong, 2013; Pillitteri and Torii, 2012), which is initiated by extracellular peptide ligands in the EPIDERMAL PATTERNING FACTOR (EPF) family (Rowe and Bergmann, 2010; Torii, 2012); this signal is perceived by a receptor-like protein, TOO MANY MOUTHS (TMM) (Nadeau and Sack, 2002), and the LEUCINE-RICH REPEAT (LRR) receptor-like kinase family, including ERECTA (ER), ER-LIKE 1 (ERL1) and ER-LIKE 2 (ERL2) (Shpak et al., 2005). The ligand-receptor signaling is then delivered by a canonical MAP kinase cascade to modulate cytoplasmic and nuclear machineries in stomatal production and division patterning. Mutations in these signaling components, including the EPF peptides and ER receptors, lead to mis-patterned stomatal distribution and overproliferation of the stomatal lineage cells (Hara et al., 2007, 2009; Hunt and Gray, 2009; Shpak, 2013; Torii, 2012).
To investigate how epigenetic modifications, e.g. histone H3K9 methylation and DNA methylation, might affect plant development, we examined the loss-of-function Arabidopsis mutants ibm1 and edm2. We found that these mutants bear an increased number of stomatal lineage cells in the leaf epidermis. We show that the stomatal defects in edm2 mutants are caused by the impaired expression of IBM1. We further elucidated that three ER genes (ER, ERL1 and ERL2), but not other stomatal genes, are hypermethylated in the absence of IBM1 or EDM2, which in turn results in lowered transcript level and thus abnormal stomatal patterning. The ER receptors belong to the large gene family of LRR receptor-like kinases and play important roles in cell division, proliferation and differentiation in various plant developmental processes (Shpak, 2013; van Zanten et al., 2009). Our results therefore provide new insights into the ER gene family as an integration point where the chromatin regulators IBM1 and EDM2 modulate plant development at an epigenetic level.
RESULTS
Stomatal lineage divisions are overproduced in ibm1 and edm2 mutants
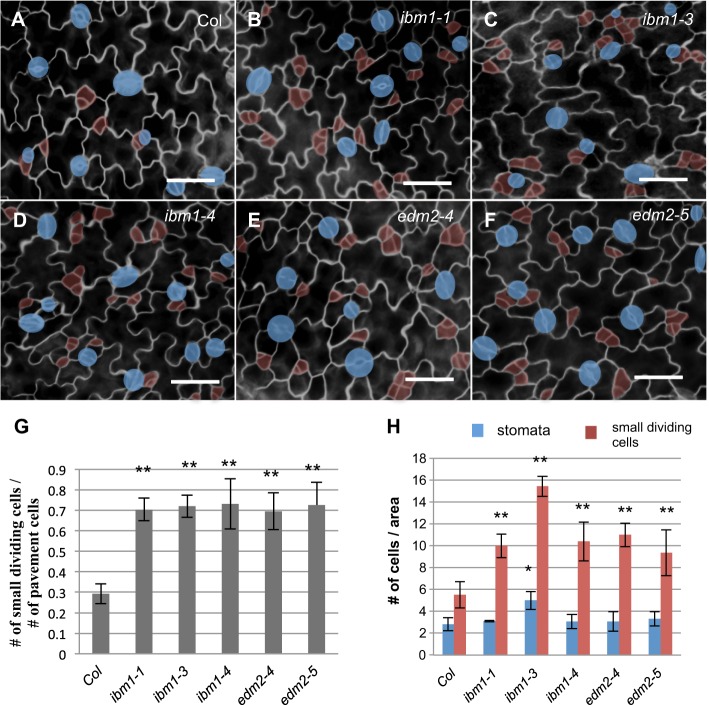
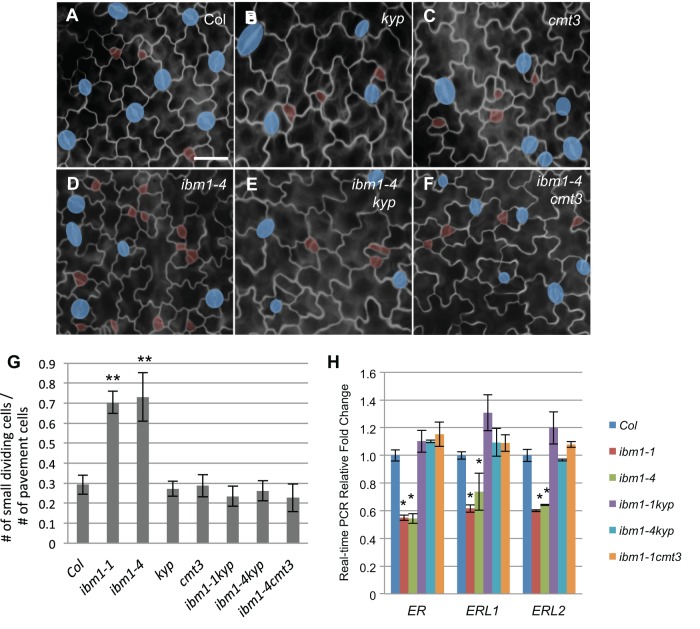
The loss-of-function mutants of ibm1 and edm2 show a variety of developmental defects in Arabidopsis leaves, flowers and seeds (Eulgem et al., 2007; Saze et al., 2008; Tsuchiya and Eulgem, 2010a,b). Here, we examined stomatal development and patterning defects. The lines under analysis are three independent alleles, ibm1-1, ibm1-3 and ibm1-4 (Lei et al., 2014; Fan et al., 2012), and two alleles of edm2-4 and edm2-5 (Lei et al., 2014). We found that all of these mutants produce more cell divisions, probably within the stomatal lineage, in the 3-dpg (days post-germination) adaxial cotyledons (Fig. 1A-F). The number of small dividing cells in ibm1 and edm2 mutants was about twice that of the Columbia (Col) wild-type plants (Fig. 1G,H) and the elevated stomatal lineage divisions persist in older leaves (16-dpg cotyledons and 12-dpg true leaves) of ibm1 and er mutants, but not in Col (Figs S1, S2).
Fig. 1.
ibm1 and edm2 mutants generate more stomatal divisions in the leaf epidermis. (A-F) Epi-fluorescence images of 3-dpg adaxial cotyledons of wild type (Col; A), ibm1-1 (B), ibm1-3 (C), ibm1-4 (D), edm2-4 (E) and edm2-5 (F). The images in A and D are also shown in Fig. 4D and Fig. 4F, respectively, because these experiments were performed concurrently. Stomata are shaded blue; small dividing cells are highlighted in red. Remarkably elevated numbers of small dividing cells were evident in ibm1 and edm2 mutants. Cell outlines were stained with FM1-43. Scale bars: 20 μm. (G) Histogram showing the ratio of total number of small dividing cells relative to that of pavement cells in different genotypes. (H) Quantification of stomata and small dividing cells in the indicated plants. For each sample, cells were counted from similarly positioned areas of 122,500 μm2 in 3-dpg cotyledons. In G and H, data are mean±s.d., n=6 individual cotyledons for each line. This quantification method applies to all other figures that demonstrate stomatal phenotype in this study. *P<0.05, **P<0.01; Student's t-test with Bonferroni correction was used to compare the mutant values with those of Col.
To identify visually the small dividing cells in ibm1 and edm2, we crossed two marker lines, MUTEp-nucGFP (nuclear GFP driven by the MUTE promoter) and TMMp-GFP (GFP driven by the TMM promoter), into the ibm1-1 mutant. The expression of MUTEp-nucGFP marks the meristemoids that are differentiating into guard mother cells (Pillitteri et al., 2007) and TMMp-GFP labels the stomatal lineage cells (Nadeau and Sack, 2002). As expected, the small dividing cells in ibm1-1 are positive for both markers (Fig. 2A-D) and the numbers are greatly increased compared with the wild type (Col) (Fig. 2E).
Fig. 2.
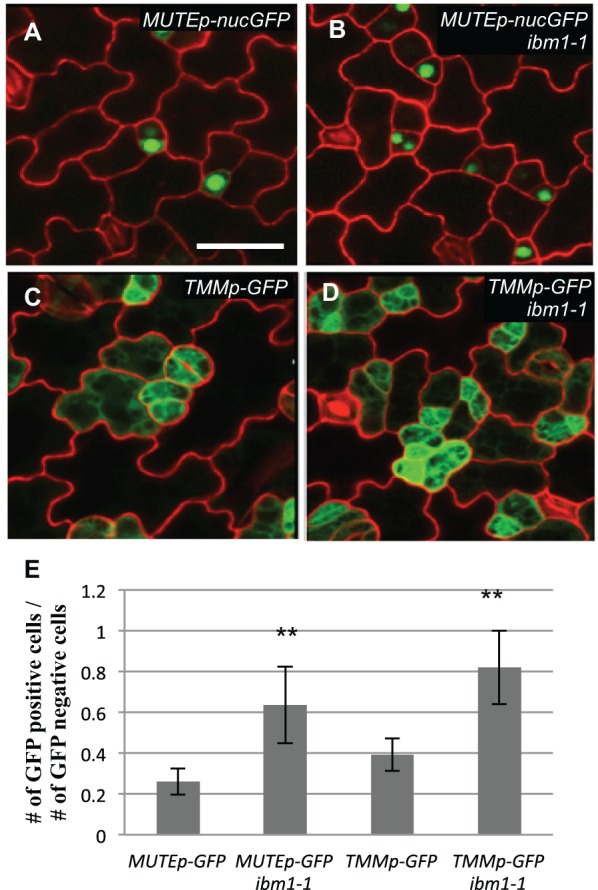
Overproduction of stomatal lineage cells in ibm1 mutants. (A-D) Confocal images of 3-dpg adaxial cotyledons expressing cell identity markers (green). Cell outlines are stained with FM4-64 (red). Scale bar: 30 μm (in A, for A-D). (A,B) The meristemoid marker MUTEp-nucGFP (nuclear GFP) in Col (A) and ibm1-1(B). (C,D) The stomatal lineage marker TMMp-GFP in Col (C) and ibm1-1 (D). More cells expressing GFP were evident in ibm1-1 mutants. (E) Histogram showing the ratio of the number of GFP-positive cells over that of GFP negative. *P<0.05, **P<0.01; Student's t-test with Bonferroni correction (n=6; ±s.d.) was used to compare the marker lines in ibm1-1 with the wild type.
Stomatal defects in edm2 can be rescued by ectopic expression of the IBM1-Long transcripts
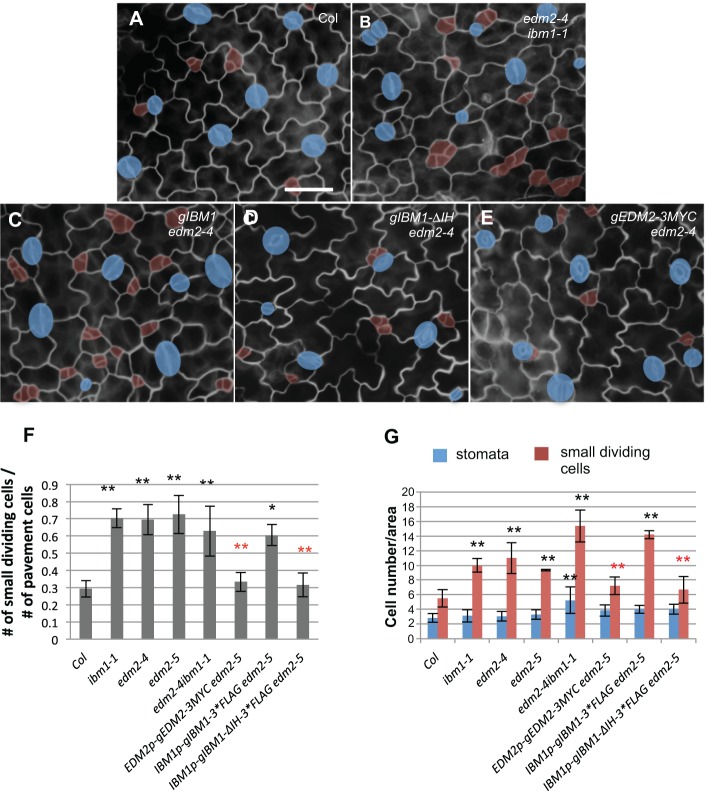
The putative chromatin regulator EDM2 functions to regulate the expression of IBM1 (Lei et al., 2014). To investigate the genetic interaction between ibm1 and edm2 in stomatal development, we generated a double mutant of ibm1-1 edm2-4 and found that the stomatal phenotype of the double mutant resembled that of the single mutants of ibm1-1 or edm2-4 (Fig. 3A,B,F,G). Because ibm1-1 edm2-4 appeared to grow smaller leaves, the number of cells per area in the double mutant appeared to be relatively high (Fig. 3G), but the ratio of stomatal lineage cells versus pavement cells (more reflective of stomatal development) was comparable to that of the single mutants (Fig. 3F). These data suggested that EDM2 and IBM1 do indeed function in the same genetic pathway for proper stomatal production.
Fig. 3.
Stomata defects in edm2 are caused by impaired expression of IBM1. (A-E) Epi-fluorescence images of 3-dpg adaxial cotyledons of Col (A), edm2-4 ibm1-1 (B), IBM1-gDNA in edm2-4 (C; full-length genomic IBM1), gIBM1-ΔIH in edm2-4 (D; the intron region deleted from gIBM1) and EDM2-gDNA-3*Myc in edm2-4 (E; full-length genomic EDM2). The control image (A) is also shown in Fig. 6A because these experiments were performed concurrently. The double mutant edm2-4 ibm1-1 produced more stomatal lineage cells. The full-length IBM1 failed to rescue edm2, but the intron-minus version did. Blue shades indicate stomata, and small dividing cells are highlighted by red. Scale bar: 30 μm (in A, for A-E). (F) Histogram showing the ratio of the total number of small dividing cells relative to that of the pavement cells in different genotypes. The double mutant edm2-4 ibm1-1 resembles the single mutants of ibm1-1 and edm2-4. Successful complementation was found by EDM2 transgene, as well as truncated IBM1 transgene (ΔIH), but not by the full-length IBM1. *P<0.05, **P<0.01; Student's t-test with Bonferroni correction. Black asterisks indicate significantly different from Col and red asterisks indicate significantly different from edm2-5. (G) Quantification of the total number of stomata and small dividing cells in the indicated mutants. The same format of statistical analysis is used as in F.
The detectable transcripts of IBM1 contain at least two variants (short and long) and EDM2 is required for the accumulation of the long version (IBM1-L) (Lei et al., 2014). EDM2 binds to the intronic heterochomatin region of IBM1 gene, and blocks the proximal polyadenylation that produces IBM1-S to promote distal polyadenylation, which produces IBM1-L (Lei et al., 2014). Previous work showed that when the intronic heterochromatin region of IBM1 was deleted, the accumulation of IBM1-L no longer required EDM2 (Lei et al., 2014). Here, we found that, indeed, when driven by the endogenous promoter, the genomic region of the IBM1 gene with a 764-bp deletion of the intronic heterochromatin fragment (gIBM1-ΔIH), which produces functional IBM1-L (Lei et al., 2014), largely rescued the stomatal defects of edm2 (Fig. 3C,D,F-G). By contrast, the wild-type version of IBM1 (gIBM1) failed to rescue the defects of edm2-4 in stomatal development (Fig. 3C). As a control, the genomic EDM2 fragment rescued the stomatal phenotype in edm2 mutants (Fig. 3E-G). Taken together, these data suggested that the elevated number of stomatal asymmetric cell divisions in edm2 mutants is caused by deficient expression of functional IBM1-L transcripts.
Hypermethylation and silencing of the ER family genes in ibm1 and edm2
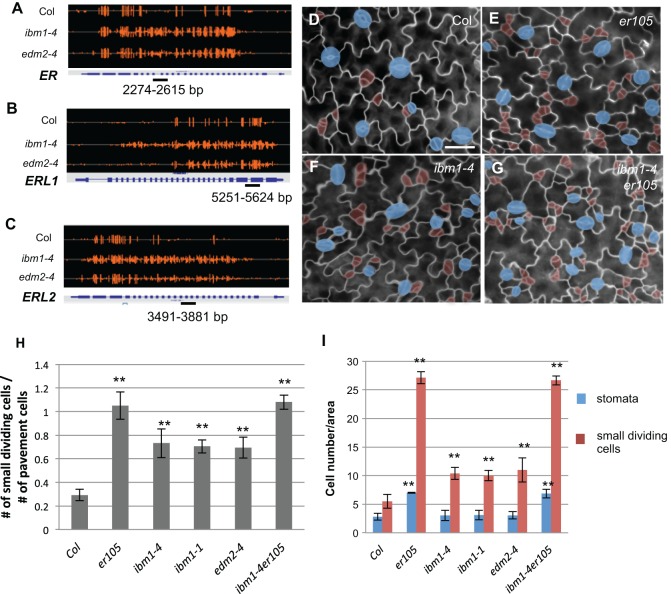
It was reported that IBM1, as a histone demethylase, functions to prevent gene body methylation and that ibm1 loss-of-function mutants bear ectopic H3K9me2 and CHG hypermethylation in the gene body of thousands of active genes (Inagaki et al., 2010). The leaf epidermal patterning in ibm1 and edm2 to a certain extent phenocopied that of the loss-of-function mutants of the negative regulators in the signaling pathway during stomatal development, e.g. erecta (er105) (Shpak et al., 2004), tmm (Nadeau and Sack, 2002), epf1 (Hara et al., 2007) and epf2 (Hara et al., 2009; Hunt and Gray, 2009), though they did not show major defects in stomatal patterning (clusters) as was observed in epf1 and tmm mutants. We scanned the whole-genome bisulfite sequencing database (Lei et al., 2014) and examined the DNA methylation levels of the stomatal development-related genes in different genetic backgrounds (Fig. 4A-C; Table S1). Among them, the coding regions of ER, ERL1 and ERL2 contain moderately methylated cytosine sites in Col (Fig. 4A-C). Interestingly, in ibm1-4 and edm2-4 mutants, the DNA methylation levels in ER, ERL1 and ERL2 were dramatically elevated (>25 fold) compared with those in the wild type (Col) (Fig. 4A-C; Table S1).
Fig. 4.
ER genes are hypermethylated in ibm1 and edm2 mutants. (A-C) Integrated genome viewer snapshots showing the DNA methylation status of ER (A), ERL1 (B) and ERL2 (C) in the wild type (Col), and in ibm1-4 and edm2-4 mutants. Orange bars indicate the sites of cytosine methylation and the height of the bars indicates the relative DNA methylation level. The genomic regions where bisulfite sequencing was performed are underlined. (D-G) Epi-fluorescence images of 3-dpg adaxial cotyledon of Col (D), er105 (E), ibm1-4 (F) and ibm1-4 er105 (G). The double mutant resembles er105. Cell outlines are marked by FM1-43 staining. Scale bar: 30 μm (in D, for D-G). The images in D and F are also shown in Fig. 1A and Fig. 1D, respectively, because these experiments were performed concurrently. (H) Histogram showing the ratio of the total number of small dividing cells relative to that of the pavement cells in different genotypes. (I) Quantification of the total number of stomata and small dividing cells. Data in H and I are mean±s.d. *P<0.05, **P<0.01, compared with the wild type (Col) by Student's t-test with Bonferroni correction (n=6).
By contrast, most of the other stomata-related genes that we examined did not show an obvious increase (fold change <2) in methylation level in ibm1 and edm2 mutants (Table S1, a few exceptions showed two- to tenfold change). These genes include those encoding the transcription factors SPCH (MacAlister et al., 2007), MUTE (Pillitteri et al., 2007), FAMA (Ohashi-Ito and Bergmann, 2006), FOUR LIPS (Lai et al., 2005) and SCREAM (Kanaoka et al., 2008), and those encoding the signaling molecules TMM (Nadeau and Sack, 2002), EPF1 (Hara et al., 2007) and EPF2 (Hara et al., 2009; Hunt and Gray, 2009), as well as STOMAGEN (Kondo et al., 2010; Sugano et al., 2010), MKK7 and 9 (Lampard et al., 2009), MPK3 and 6 (Wang et al., 2007). MYB88 is an exception that showed CHG elevation in ibm1 mutants, but loss of MYB88 alone does not produce a discernable stomatal phenotype (Lai et al., 2005). Interestingly, a CHG methylation reduction was found in SPCH (Table S1), though the transcription level of SPCH was not obviously affected (Fig. 5E), therefore the possible connection to ibm1 stomatal overproduction cannot be assumed.
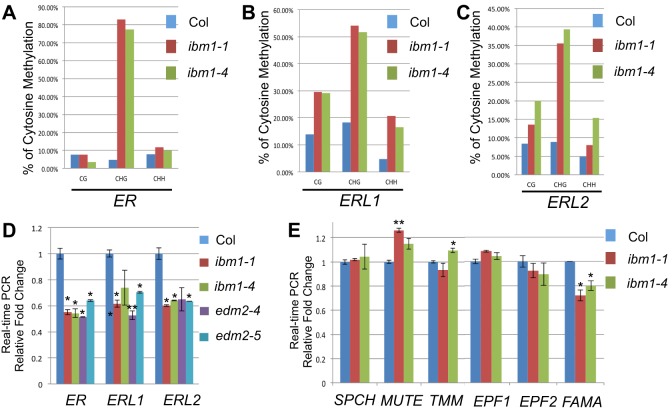
Fig. 5.
The ibm1 mutations affect the DNA methylation levels of ER genes. (A-C) Individual locus bisulfite sequencing analyses of CG, CHG, CHH methylation in ER (A), ERL1 (B) and ERL2 (C) in Col, ibm1-1 and ibm1-4. At least ten clones were selected from each sample. Striking methylation elevation in CHGs was found in all three ER genes when IBM1 is defective. (D,E) Transcript level of stomata-related genes in Col and in ibm1 and edm2 mutants. The expression of ER genes was suppressed in ibm1 and edm2 mutants. Values are mean±s.d. Data were collected from three biological replicates and the expression fold changes were normalized to the transcript level in Col. *P<0.05, **P<0.01, compared with the wild type (Col) by Student's t-test with Bonferroni correction; n=3 replicates.
The stomatal defects in ibm1 and edm2 mutants to a certain extent resembled those of the ERECTA loss-of-function mutant er105 (Shpak et al., 2005) (Fig. 4D-G), in that the stomatal entry divisions were considerably increased (red shades in Fig. 4E,F vs 4D; quantification in Fig. 4H,I) and the total numbers of stomata were mildly elevated (Fig. 4I). We crossed ibm1 mutants to er105 and found that the double mutants ibm1-4 er105 (Fig. 4G) and ibm1-1 er105 (Fig. S3A,B) both showed a similar level of stomatal phenotype to that of er105 (Fig. 4H,I). At the whole-plant level, 3-week-old plants of ibm1-1 er105 mainly bear morphological features of er105 (Fig. S3B). These data strongly suggested that er is epistatic to ibm1 in stomatal and early leaf development. To improve our understanding of the DNA methylation change in the three sequence contexts (CG, CHG and CHH) in ibm1, we performed bisulfite sequencing on individual loci of ER, ERL1 and ERL2 genes in Col, ibm1-1 and ibm1-4 mutants, respectively. The genomic regions examined by bisulfite sequencing are underlined in Fig. 4A-C. Consistent with the whole-genome methylation data, the individual locus methylation data confirmed that the gene bodies of ER, ERL1 and ERL2 are hypermethylated in both ibm1-1 and ibm1-4 mutants (Fig. 5A-C). It is also evident that the three tested ER genes showed the most dramatic elevation in CHG methylation, compared with CG and CHH methylation, when IBM1 was disrupted (Fig. 5A-C). The promoter regions of the ER family genes were not obviously subjected to DNA methylation regulation in Col, ibm1 and edm2 plants (Fig. S4).
To correlate DNA methylation with gene expression level, we then analyzed transcription level of the ER genes. Real-time PCR experiments using 3-dpg seedlings showed that, in line with the elevated DNA methylation levels, expression levels of the ER genes were reduced by about 40% in ibm1 and edm2 mutants (Fig. 5D). By contrast, the expression levels of other stomatal regulatory genes mentioned above, including positive regulators (SPCH and FAMA) and negative regulators (EPF1, EPF2 and TMM), were not consistently increased in the two alleles of ibm1 mutants (Fig. 5E). The expression of MUTE was slightly enhanced (Fig. 5E), probably owing to overproduced stomatal lineage cells in ibm1. These data suggest that the stomatal defects in ibm1 and edm2 mutants are mainly caused by the decreased expression of three ER genes.
We further analyzed other receptor-like kinases that are phylogenetically related to the ER receptor family (Shiu and Bleecker, 2001): the brassinosteroid receptor BRI1 (Wang et al., 2001), the CLV1 receptor in shoot meristem development (Clark et al., 1997) and the TMK1 receptor that is thought to sense auxin signaling (Dai et al., 2013; Xu et al., 2014). None of them showed discernable changes of DNA methylation in gene body and promoter regions (Fig. S5). Recently, the receptor-like kinase family SOMATIC EMBRYOGENESIS RECEPTOR KINASE (SERK), which has four members, was found to bind to ER receptors to regulate stomatal development (Meng et al., 2015). SERK3 (also called BAK1) interacts with BRI1 to mediate brassinosteroid signaling in Arabidopsis (Li et al., 2002; Nam and Li, 2002). Three SERKs (1, 2 and 3) seemed to be mildly hypermethylated in ibm1 and edm2 mutants (Fig. S6A). However, their transcription levels were apparently elevated as demonstrated by real-time PCR (Fig. S6B). Therefore, considering the important fact that er ibm1 resembles er, the stomatal phenotypes of ibm1 and edm2 are not likely to be caused by elevated expression of SERK genes, but could be related to high DNA methylation and low transcription levels of ER genes.
cmt3 and kyp mutations are epistatic to ibm1 and edm2 in stomatal development
Previous reports showed that the ectopic cytosine methylation phenotype of ibm1 depends on CMT3 DNA methyltransferase- and the KYP histone methyltransferase-mediated H3K9 dimethylation, and cmt3 and kyp mutations suppressed almost all detectable developmental defects in ibm1 mutants (Saze et al., 2008). Therefore, we crossed ibm1 and edm2 mutants with kyp and cmt3 to create the corresponding double mutants (Fig. 6; Fig. 7). We found that, differing from the increased stomatal lineage divisions in ibm1 and edm2, stomatal development and patterning of the single kyp and cmt3 mutants were indistinguishable from the wild type (Col) (Fig. 6A-C). Furthermore, the double mutants ibm1-4 kyp and ibm1-4 cmt3 did not exhibit the stomatal phenotype seen in ibm1-4, instead resembling those of kyp, cmt3 and Col (Fig. 6D-F). Similar effects were also seen when another allele of ibm1 was used (ibm1-1 kyp; Fig. S7A,B). These data suggested that both KYP and CMT3 are required for the ibm1-induced stomatal phenotypes. Similarly, the double mutants of edm2-5 kyp and edm2-4 cmt3 restored the stomatal phenotypes of edm2-4 back to the wild-type level (Fig. 7).
Fig. 6.
cmt3 and kyp4 mutations suppress the stomatal defects in ibm1 mutants. (A-F) Epi-fluorescence microscope images show 3-dpg adaxial cotyledons of Col (A), kyp (B), cmt3 (C), ibm1-4 (D), ibm1-4 kyp (E) and ibm1-4 cmt3 (F). The control image (A) is also shown in Fig. 3A because these experiments were performed concurrently. Scale bar: 30 μm (in A, for A-F). cmt3 and kyp were epistatic to ibm1 mutations. (G) Histogram displaying the ratio of the total number of small dividing cells over that of the pavement cells in different mutant backgrounds. *P<0.05, **P<0.01; one-way ANOVA with Bonferroni multiple comparison (n=6; ±s.d.). (H) Real-time PCR analyses for the expression level of ER genes in the indicated mutants. The expression fold changes were normalized to the transcript level in Col. Note the expression of ER genes recovered to the wild-type level when kyp and cmt3 were intragressed into the ibm1 mutants. *P<0.05, **P< 0.01 compared with the wild type (Col) by Student's t-test with Bonferroni correction (n=3 replicates).
Fig. 7.
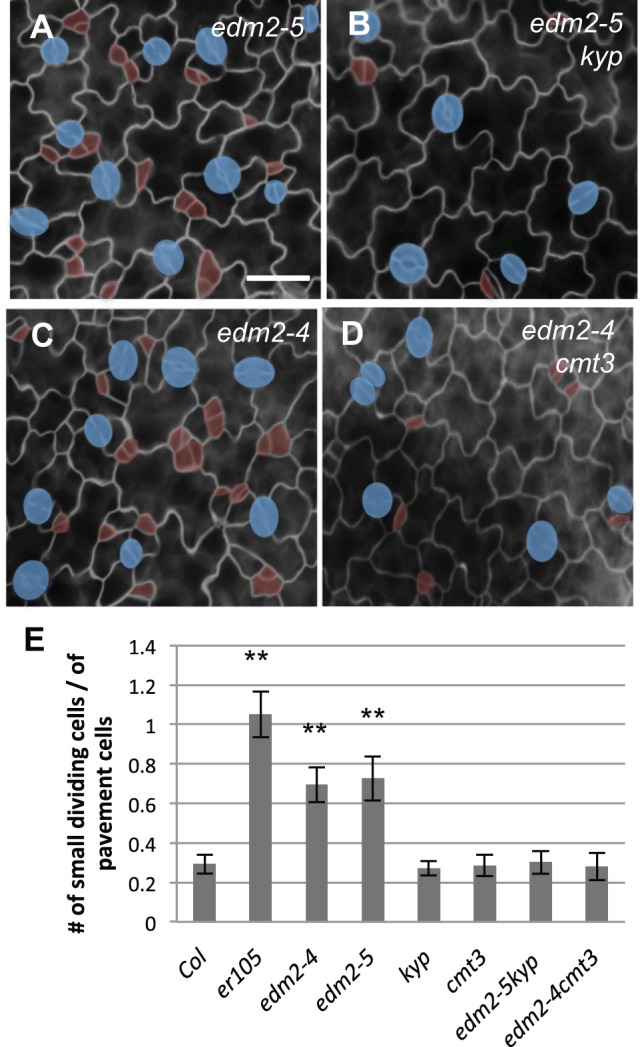
cmt3 and kyp mutations suppress the stomatal defects in edm2 mutants. (A-D) Epi-fluorescence microscope images show 3-dpg adaxial cotyledons of edm2-5 (A), edm2-5 kyp (B), edm2-4 (C) and edm2-4 cmt3 (D). Scale bar: 30 μm (in A, for A-D). cmt3 and kyp were epistatic to edm2 mutations. (E) Histogram displaying the ratio of the total number of small dividing cells over that of the pavement cells in different mutant backgrounds. *P<0.05, **P<0.01; one-way ANOVA with Bonferroni multiple comparison (n=6; ±s.d.).
To further support the hypothesis that the recovered stomatal phenotype, when kyp and cmt mutations were introgressed into ibm1 mutant plants, is related to ER gene expression, we performed real-time PCR assays. Our data showed that the expression levels of ER, ERL1 and ERL2 were lower in ibm1, but when combined with the kyp and cmt3 mutations, the decreased expression of the ER family genes recovered to near wild-type level in the double mutants of ibm1 kyp and ibm1 cmt3 (Fig. 6H).
We also found that a loss-of-function mutation in the RdDM component AGO4 did not exhibit any obvious stomatal defects in division, production and patterning (Fig. S8A,B). As expected, ago4 did not suppress the stomatal defects of the ibm1 mutant (Fig. S8C-F) supporting the hypothesis that ibm1 is epistatic to ago4 in stomatal development (Fig. S9).
DISCUSSION
Hypermethylation of ER genes in ibm1 and edm2
The whole-genome bisulfite sequencing data revealed increased DNA methylation levels in the coding regions of ER, ERL1 and ERL2 in ibm1 and edm2 mutants. Our individual locus bisulfite sequencing data confirmed that, in ibm1 mutants, all three ER genes were hypermethylated, particularly at CHG sites (Fig. 5A-C). The suggestion that ER genes are putative targets of IBM1 in stomatal development is consistent with the characteristics of the typical genes that are under the control of IBM1 in Arabidopsis. These genes often encode long transcripts (the full-length genomic region of ER is about 6 kb) and IBM1-mediated suppression of DNA methylation occurs mainly at CHG sites (Miura et al., 2009).
Many studies have investigated how ER receptor-like kinases play pleiotropic functions in plant development, physiological processes and responses to growth and environmental stimuli (Shpak, 2013; van Zanten et al., 2009), but not much is known about how expression patterns and levels of the ER gene are modulated. A recent study showed that 26 introns in the ER gene are important for its mRNA accumulation and efficient translation. It was hypothesized that these introns might affect the elongation of the poly(A) tail of ER mRNA or the assembly of a spliceosome that might promote ER transcription and translation (Karve et al., 2011). Here, we propose another possibility that these introns could recruit other regulatory factors, e.g. IBM1 and/or EDM2 chromatin factors, which antagonize histone modification- and DNA methylation-induced gene silencing for sustained expression of ER in plant development.
Epigenetic regulation of genes that modulate stomatal lineage population
Our study showed that the IBM1 H3K9me2 demethylase is necessary for preventing the gene body DNA methylation of three ER genes. DNA hypermethylation of the ER genes in ibm1 mutants depended on the DNA methyltransferase CMT3, but not on the RdDM pathway. By contrast, it was recently reported that the DNA methylation level of EPF2, encoding a signal peptide that functions upstream of the ER receptors (Hunt and Gray, 2009), was controlled through another mechanism, the DNA demethylase ROS1 (Yamamuro et al., 2014). ros1 loss-of-function mutants produced overproliferated stomatal lineage cells because the EPF2 promoter was hypermethylated and silenced. The stomatal phenotype of ros1 can be counteracted by RdDM mutations (Yamamuro et al., 2014). The CMT3 and RdDM pathways seemed to work in a specific manner on target genes; ER genes were hypermethylated in ibm1 mutants, but not in ros1 mutants (Fig. S11), and EPF2 was hypermethylated in ros1 mutants, but not in ibm1 (Table S1). Taken together, the initiation and maintenance of the stomatal lineage cells, a specialized stem cell population in plants (Matos and Bergmann, 2014), are ensured by two independent mechanisms: ROS1-mediated DNA demethylation on the EPF2 promoter and IBM1-mediated histone demethylation on the bodies of ER genes. These epigenetic mechanisms dramatically differ from those in mammalian pluripotent stem cells in which chromatin regulators mainly target and regulate pluripotency transcription factors (Luo et al., 2014; Papp and Plath, 2013). So far, no direct evidence supports the hypothesis that the key stomatal differentiation transcription factors SPCH, MUTE, FAMA and SCRM are under active DNA methylation control, though possible regulations of chromatin modifiers that enforce terminal guard cell integrity are emerging (Lee et al., 2014; Matos et al., 2014).
Expression pattern of IBM1 and its effect on ER genes at the tissue level
It is interesting to note that, in ibm1 and edm2 mutants, the CHG hypermethylation in the genic regions of all three ER genes resulted in downregulation of their expression levels to about 50-60% of the wild-type level. The combined effect of three ER genes in ibm1 seemed to produce relatively mild stomatal phenotypes, and no drastic developmental or patterning defects that might be detrimental to plant growth, in contrast to those found in er erl1 erl2 triple mutants (Fig. S10; severe stomatal clustering and seedlings are lethal). Even when compared with the er105 mutant, ibm1 stomatal phenotypes are less severe (Fig. 4). These data seem to suggest that epigenetic factors (e.g. DNA methylation and histone modifications), compared with genetic disruptions, are more flexible and plastic in regulating gene expression.
Besides elevated stomatal lineage numbers, the er mutants also display prominent defects at the whole-plant level, including enlarged shoot apical meristem size (Mandel et al., 2014), short plant stature and compact inflorescence (Shpak, 2013; Torii et al., 1996). These phenotypes were not evident in ibm1 and edm2 mutants (Saze et al., 2008). We also found that the IBM1 expression levels vary a lot in different tissues in Arabidopsis and the highest expression level of IBM1-L was found in inflorescences (Fig. S12). Our bisulfite sequencing data suggested that the ER gene was similarly hypermethylated in ibm1 flowers (Fig. S13A,B), and the expression levels of the three ER genes were reduced (Fig. S13C). These data suggest that the absence of defects in inflorescence architecture in ibm1 mutants might be explained by the small decrease in the levels of ER expression that were not sufficient to cause apical growth defects.
MATERIALS AND METHODS
Plant materials and growth conditions
The Arabidopsis homozygous loss-of-function mutants ibm1-1 (point mutation) and ibm1-4 (SALK_035608) were described previously (Saze et al., 2008). The third allele ibm1-3 (SALK_023533) was a gift from Dr Ligeng Ma (College of Life Sciences, Capital Normal University, Beijing) er105 and other mutants of the ER family were obtained from Dr Keiko Torii (Shpak et al., 2005). Other plant materials obtained from the Arabidopsis Biological Resource Center (http://www.arabidopsis.org) were confirmed by genotyping and sequencing. The ecotype Columbia (Col-0, labeled as Col in the figures and text) was used as the wild type unless otherwise noted. The TMM transcriptional marker was described by Nadeau and Sack (2002) and the MUTE promoter driving nuclear GFP was obtained from Dr Dominique Bergmann and used previously by Dong et al. (2009).
Arabidopsis seeds were sterilized with 5% household bleach solution and rinsed with sterilized double-distilled H2O more than six times. Clean seeds were then dispersed on ½ MS (Murashige–Skoog) agar plates and incubated at 4°C for a day before being transferred to a standard plant growth chamber (23°C with16-h light/8-h dark cycles). When needed, seedlings were transferred to soil for continued growth under similar conditions.
Microscopic observation and phenotype quantification
To examine stomatal phenotypes, Arabidopsis cotyledons (3-, 8- and 16-dpg) and true leaves (12-dpg) were stained with FM1-43 (Invitrogen T35356, 1 μg/ml), a fluorescent probe that integrates into the plasma membrane to outline cell shape. Seedlings were examined on an Olympus BX53 fluorescence microscope equipped with a GFP filter.
For quantification, the genetic materials used for counting cells in 3-dpg cotyledons were grown at the same time and under the same growth conditions to ensure the materials across the board are fully comparable in most of the figures. The stomatal lineage cells under division were counted as ‘small dividing cells’. The epidermal cells are classified into three groups: pavement cells (larger than a mature guard cell and showing at least one obvious lobe), guard cells, and small dividing cells. To quantify stomatal phenotype, typically, cells from six independent cotyledons from six individual plants were collected for each sample. An area of 122,500 μm2 (350×350 μm) from each sample was documented for cell counting. To test statistical significance, Student's t-test combined with Bonferroni correction was generally used. When more than two groups were compared, one-way ANOVA and Bonferroni multiple comparison were used.
To visualize TMM and MUTE transcriptional reporters (TMMp-GFP and MUTEp-nucGFP) in the ibm1 mutants, a Leica SP5 confocal microscope was used to document the GFP expression pattern in the 3-dpg seedlings counterstained with FM4-64 (Invitrogen; F34653). The excitation/emission spectra were: GFP 488 nm/500-530 nm and FM4-64 561 nm/570-652 nm.
Identification of hypermethylated genes
The whole-genome bisulfite sequencing data we used for analyses were reported previously (Lei et al., 2014). To compare the DNA methylation level of each target gene related to stomatal development, their genomic sequences, including the promoter and the coding regions, were scanned for methylated cytosines in CG, CHG and CHH contexts. We collected the numbers of methylated cytosines in each context to indicate DNA methylations levels. These numbers were calculated and compared between the wild type (Col) and the related mutants (ibm1, edm2 and others).
Real-time PCR
The RNeasy Plant Mini Kit (Qiagen) was used to extract total RNAs from 14-dpg Arabidopsis plants. After the removal of DNA, 1 μg of total RNAs served as template for synthesizing the first strand cDNAs with a One-Step SYBR PrimeScript RT-PCR Kit II (Perfect Real Time) (Takara). Real-time PCR was performed following the manufacturer's instructions. The first strand cDNA mixture was diluted four times and 2 μl was used as template in a 25 μl of PCR reaction with iQ SYBR Green Supermix (Bio-Rad). The PCR reactions were performed following this protocol: pre-incubation at 95°C for 5 min followed by 40 cycles of denaturation at 95°C for 15 s, annealing at 56°C for 30 s, and extension at 72°C for 30 s. The primers used are listed in Table S2. Reactions were carried out on a iQ5 Multicolour Real-Time PCR Detection System (Bio-Rad). The comparative threshold cycle (Ct) method was used to determine the relative transcript levels and the expression levels of ACTIN2 were used as an internal control.
Individual locus bisulfite sequencing
The genomic DNA of Arabidopsis plants (14-dpg) was extracted by a DNeasy Plant Mini Kit (Qiagen). About 600 ng of genomic DNAs were aliquoted for each cytosine conversion reaction, which is achieved by using an Epitect Bisulfite Kit (Qiagen) and following the manufacturer's instructions. Out of 25 μl of the reaction mixture, 2 μl of bisulfite-treated DNA was used for nested PCR, which was programmed as follows: initial denaturation at 94°C for 3 min followed by 40 cycles of denaturation at 95°C for 15 s, annealing at 58°C for 30 s, and extension 72°C for 40 s using an ExTaq DNA polymerase (Takara). The resulting PCR products were ligated to pMD18-T vector (Takara) following the manufacturer's instructions. At least 15 clones were picked for sequencing to determine the DNA methylation level at each tested region.
Acknowledgements
We thank Jian Liang and Cheng Zhao for their technical support at the PSC and Dr Honggui La for helpful discussion.
Footnotes
Competing interests
The authors declare no competing or financial interests.
Author contributions
Y.W. and X.X performed experiments and analyzed data. Y.W., J.D. and J.-K.Z. designed experiments and wrote the manuscript.
Funding
This research project was supported by funding from the Chinese Academy of Sciences; and grants from the National Institute of General Medical Sciences [R01 GM070795, R01 GM059138 and R01 GM109080]. Deposited in PMC for release after 12 months.
Supplementary information
Supplementary information available online at http://dev.biologists.org/lookup/doi/10.1242/dev.129932.supplemental
References
- Badeaux A. I. and Shi Y. (2013). Emerging roles for chromatin as a signal integration and storage platform. Nat. Rev. Mol. Cell Biol. 14, 211-224. 10.1038/nrm3545 [DOI] [PubMed] [Google Scholar]
- Bartee L., Malagnac F. and Bender J. (2001). Arabidopsis cmt3 chromomethylase mutations block non-CG methylation and silencing of an endogenous gene. Genes Dev. 15, 1753-1758. 10.1101/gad.905701 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark S. E., Williams R. W. and Meyerowitz E. M. (1997). The CLAVATA1 gene encodes a putative receptor kinase that controls shoot and floral meristem size in Arabidopsis. Cell 89, 575-585. 10.1016/S0092-8674(00)80239-1 [DOI] [PubMed] [Google Scholar]
- Dai N., Wang W., Patterson S. E. and Bleecker A. B. (2013). The TMK subfamily of receptor-like kinases in Arabidopsis display an essential role in growth and a reduced sensitivity to auxin. PLoS ONE 8, e60990 10.1371/journal.pone.0060990 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong J., MacAlister C. A. and Bergmann D. C. (2009). BASL controls asymmetric cell division in Arabidopsis. Cell 137, 1320-1330. 10.1016/j.cell.2009.04.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ebbs M. L., Bartee L. and Bender J. (2005). H3 lysine 9 methylation is maintained on a transcribed inverted repeat by combined action of SUVH6 and SUVH4 methyltransferases. Mol. Cell. Biol. 25, 10507-10515. 10.1128/MCB.25.23.10507-10515.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eulgem T., Tsuchiya T., Wang X.-J., Beasley B., Cuzick A., Tör M., Zhu T., McDowell J. M., Holub E. and Dangl J. L. (2007). EDM2 is required for RPP7-dependent disease resistance in Arabidopsis and affects RPP7 transcript levels. Plant J. 49, 829-839. 10.1111/j.1365-313X.2006.02999.x [DOI] [PubMed] [Google Scholar]
- Fan D., Dai Y., Wang X., Wang Z., He H., Yang H., Cao Y., Deng X. W. and Ma L. (2012). IBM1, a JmjC domain-containing histone demethylase, is involved in the regulation of RNA-directed DNA methylation through the epigenetic control of RDR2 and DCL3 expression in Arabidopsis. Nucleic Acids Res. 40, 8905-8916. 10.1093/nar/gks647 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuks F. (2005). DNA methylation and histone modifications: teaming up to silence genes. Curr. Opin. Genet. Dev. 15, 490-495. 10.1016/j.gde.2005.08.002 [DOI] [PubMed] [Google Scholar]
- Hara K., Kajita R., Torii K. U., Bergmann D. C. and Kakimoto T. (2007). The secretory peptide gene EPF1 enforces the stomatal one-cell-spacing rule. Genes Dev. 21, 1720-1725. 10.1101/gad.1550707 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hara K., Yokoo T., Kajita R., Onishi T., Yahata S., Peterson K. M., Torii K. U. and Kakimoto T. (2009). Epidermal cell density is autoregulated via a secretory peptide, EPIDERMAL PATTERNING FACTOR 2 in Arabidopsis leaves. Plant Cell Physiol. 50, 1019-1031. 10.1093/pcp/pcp068 [DOI] [PubMed] [Google Scholar]
- He X. J., Chen T. and Zhu J. K. (2011). Regulation and function of DNA methylation in plants and animals. Cell Res. 21, 442-465. 10.1038/cr.2011.23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunt L. and Gray J. E. (2009). The signaling peptide EPF2 controls asymmetric cell divisions during stomatal development. Curr. Biol. 19, 864-869. 10.1016/j.cub.2009.03.069 [DOI] [PubMed] [Google Scholar]
- Inagaki S., Miura-Kamio A., Nakamura Y., Lu F., Cui X., Cao X., Kimura H., Saze H. and Kakutani T. (2010). Autocatalytic differentiation of epigenetic modifications within the Arabidopsis genome. EMBO J. 29, 3496-3506. 10.1038/emboj.2010.227 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jackson J. P., Lindroth A. M., Cao X. and Jacobsen S. E. (2002). Control of CpNpG DNA methylation by the KRYPTONITE histone H3 methyltransferase. Nature 416, 556-560. 10.1038/nature731 [DOI] [PubMed] [Google Scholar]
- Kanaoka M. M., Pillitteri L. J., Fujii H., Yoshida Y., Bogenschutz N. L., Takabayashi J., Zhu J.-K. and Torii K. U. (2008). SCREAM/ICE1 and SCREAM2 specify three cell-state transitional steps leading to arabidopsis stomatal differentiation. Plant Cell 20, 1775-1785. 10.1105/tpc.108.060848 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karve R., Liu W., Willet S. G., Torii K. U. and Shpak E. D. (2011). The presence of multiple introns is essential for ERECTA expression in Arabidopsis. RNA 17, 1907-1921. 10.1261/rna.2825811 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klose R. J., Kallin E. M. and Zhang Y. (2006). JmjC-domain-containing proteins and histone demethylation. Nat. Rev. Genet. 7, 715-727. 10.1038/nrg1945 [DOI] [PubMed] [Google Scholar]
- Kondo T., Kajita R., Miyazaki A., Hokoyama M., Nakamura-Miura T., Mizuno S., Masuda Y., Irie K., Tanaka Y., Takada S. et al. (2010). Stomatal density is controlled by a mesophyll-derived signaling molecule. Plant Cell Physiol. 51, 1-8. 10.1093/pcp/pcp180 [DOI] [PubMed] [Google Scholar]
- Lai L. B., Nadeau J. A., Lucas J., Lee E. K., Nakagawa T., Zhao L., Geisler M. and Sack F. D. (2005). The Arabidopsis R2R3 MYB proteins FOUR LIPS and MYB88 restrict divisions late in the stomatal cell lineage. Plant Cell 17, 2754-2767. 10.1105/tpc.105.034116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lampard G. R., Lukowitz W., Ellis B. E. and Bergmann D. C. (2009). Novel and expanded roles for MAPK signaling in Arabidopsis stomatal cell fate revealed by cell type-specific manipulations. Plant Cell 21, 3506-3517. 10.1105/tpc.109.070110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lau O. S. and Bergmann D. C. (2012). Stomatal development: a plant's perspective on cell polarity, cell fate transitions and intercellular communication. Development 139, 3683-3692. 10.1242/dev.080523 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Law J. A. and Jacobsen S E. (2010). Establishing, maintaining and modifying DNA methylation patterns in plants and animals. Nat. Rev. Genet. 11, 204-220. 10.1038/nrg2719 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee E. K., Lucas J. R., Goodrich J. and Sack F. D. (2014). Arabidopsis guard cell integrity involves the epigenetic stabilization of the FLP and FAMA transcription factor genes. Plant J. 78, 566-577. 10.1111/tpj.12516 [DOI] [PubMed] [Google Scholar]
- Lei M., La H., Lu K., Wang P., Miki D., Ren Z., Duan C.-G., Wang X., Tang K., Zeng L. et al. (2014). Arabidopsis EDM2 promotes IBM1 distal polyadenylation and regulates genome DNA methylation patterns. Proc. Natl. Acad. Sci. USA 111, 527-532. 10.1073/pnas.1320106110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li J., Wen J., Lease K. A., Doke J. T., Tax F. E. and Walker J. C. (2002). BAK1, an Arabidopsis LRR receptor-like protein kinase, interacts with BRI1 and modulates brassinosteroid signaling. Cell 110, 213-222. 10.1016/S0092-8674(02)00812-7 [DOI] [PubMed] [Google Scholar]
- Lindroth A. M., Cao X., Jackson J. P., Zilberman D., McCallum C. M., Henikoff S. and Jacobsen S. E. (2001). Requirement of CHROMOMETHYLASE3 for maintenance of CpXpG methylation. Science 292, 2077-2080. 10.1126/science.1059745 [DOI] [PubMed] [Google Scholar]
- Liu C., Lu F., Cui X. and Cao X. (2010). Histone methylation in higher plants. Annu. Rev. Plant Biol. 61, 395-420. 10.1146/annurev.arplant.043008.091939 [DOI] [PubMed] [Google Scholar]
- Luo C., Dong J., Zhang Y. and Lam E. (2014). Decoding the role of chromatin architecture in development: coming closer to the end of the tunnel. Front. Plant Sci. 5, 374 10.3389/fpls.2014.00374 [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacAlister C. A., Ohashi-Ito K. and Bergmann D. C. (2007). Transcription factor control of asymmetric cell divisions that establish the stomatal lineage. Nature 445, 537-540. 10.1038/nature05491 [DOI] [PubMed] [Google Scholar]
- Malagnac F., Bartee L. and Bender J. (2002). An Arabidopsis SET domain protein required for maintenance but not establishment of DNA methylation. EMBO J. 21, 6842-6852. 10.1093/emboj/cdf687 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mandel T., Moreau F., Kutsher Y., Fletcher J. C., Carles C. C. and Eshed Williams L. (2014). The ERECTA receptor kinase regulates Arabidopsis shoot apical meristem size, phyllotaxy and floral meristem identity. Development 141, 830-841. 10.1242/dev.104687 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matos J. L. and Bergmann D. C. (2014). Convergence of stem cell behaviors and genetic regulation between animals and plants: insights from the Arabidopsis thaliana stomatal lineage. F1000Prime Rep. 6, 53 10.12703/P6-53 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matos J. L., Lau O. S., Hachez C., Cruz-Ramírez A., Scheres B. and Bergmann D. C. (2014). Irreversible fate commitment in the Arabidopsis stomatal lineage requires a FAMA and RETINOBLASTOMA-RELATED module. ELife 3 10.7554/eLife.03271 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matzke M. A., Kanno T. and Matzke A. J. (2015). RNA-directed DNA methylation: The evolution of a complex epigenetic pathway in flowering plants. Annu. Rev. Plant Biol. 266, 243-267. 10.1146/annurev-arplant-043014-114633 [DOI] [PubMed] [Google Scholar]
- Meng X., Chen X., Mang H., Liu C., Yu X., Gao X., Torii K. U., He P. and Shan L. (2015). Differential function of Arabidopsis SERK family receptor-like kinases in stomatal patterning. Curr. Biol. 25, 2361-2372. 10.1016/j.cub.2015.07.068 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miura A., Nakamura M., Inagaki S., Kobayashi A., Saze H. and Kakutani T. (2009). An Arabidopsis jmjC domain protein protects transcribed genes from DNA methylation at CHG sites. EMBO J. 28, 1078-1086. 10.1038/emboj.2009.59 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nadeau J. A. and Sack F. D. (2002). Control of stomatal distribution on the Arabidopsis leaf surface. Science 296, 1697-1700. 10.1126/science.1069596 [DOI] [PubMed] [Google Scholar]
- Nam K. H. and Li J. (2002). BRI1/BAK1, a receptor kinase pair mediating brassinosteroid signaling. Cell 110, 203-212. 10.1016/S0092-8674(02)00814-0 [DOI] [PubMed] [Google Scholar]
- Ohashi-Ito K. and Bergmann D. C. (2006). Arabidopsis FAMA controls the final proliferation/differentiation switch during stomatal development. Plant Cell 18, 2493-2505. 10.1105/tpc.106.046136 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papp B. and Plath K. (2013). Epigenetics of reprogramming to induced pluripotency. Cell 152, 1324-1343. 10.1016/j.cell.2013.02.043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pillitteri L. J. and Dong J. (2013). Stomatal development in Arabidopsis. Arabidopsis Book 11, e0162 10.1199/tab.0162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pillitteri L. J. and Torii K. U. (2012). Mechanisms of stomatal development. Annu. Rev. Plant Biol. 63, 591-614. 10.1146/annurev-arplant-042811-105451 [DOI] [PubMed] [Google Scholar]
- Pillitteri L. J., Sloan D. B., Bogenschutz N. L. and Torii K. U. (2007). Termination of asymmetric cell division and differentiation of stomata. Nature 445, 501-505. 10.1038/nature05467 [DOI] [PubMed] [Google Scholar]
- Rowe M. H. and Bergmann D. C. (2010). Complex signals for simple cells: the expanding ranks of signals and receptors guiding stomatal development. Curr. Opin. Plant Biol. 13, 548-555. 10.1016/j.pbi.2010.06.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saze H., Shiraishi A., Miura A. and Kakutani T. (2008). Control of genic DNA methylation by a jmjC domain-containing protein in Arabidopsis thaliana. Science 319, 462-465. 10.1126/science.1150987 [DOI] [PubMed] [Google Scholar]
- Shiu S.-H. and Bleecker A. B. (2001). Receptor-like kinases from Arabidopsis form a monophyletic gene family related to animal receptor kinases. Proc. Natl. Acad. Sci. USA 98, 10763-10768. 10.1073/pnas.181141598 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shpak E.-D. (2013). Diverse roles of ERECTA family genes in plant development. J. Integr. Plant Biol. 55, 1238-1250. 10.1111/jipb.12108 [DOI] [PubMed] [Google Scholar]
- Shpak E. D., Berthiaume C. T., Hill E. J. and Torii K. U. (2004). Synergistic interaction of three ERECTA-family receptor-like kinases controls Arabidopsis organ growth and flower development by promoting cell proliferation. Development 131, 1491-1501. 10.1242/dev.01028 [DOI] [PubMed] [Google Scholar]
- Shpak E. D., McAbee J. M., Pillitteri L. J. and Torii K. U. (2005). Stomatal patterning and differentiation by synergistic interactions of receptor kinases. Science 309, 290-293. 10.1126/science.1109710 [DOI] [PubMed] [Google Scholar]
- Sugano S. S., Shimada T., Imai Y., Okawa K., Tamai A., Mori M. and Hara-Nishimura I. (2010). Stomagen positively regulates stomatal density in Arabidopsis. Nature 463, 241-244. 10.1038/nature08682 [DOI] [PubMed] [Google Scholar]
- Torii K. U. (2012). Mix-and-match: ligand-receptor pairs in stomatal development and beyond. Trends Plant Sci. 17, 711-719. 10.1016/j.tplants.2012.06.013 [DOI] [PubMed] [Google Scholar]
- Torii K. U., Mitsukawa N., Oosumi T., Matsuura Y., Yokoyama R., Whittier R. F. and Komeda Y. (1996). The Arabidopsis ERECTA gene encodes a putative receptor protein kinase with extracellular leucine-rich repeats. Plant Cell 8, 735-746. 10.1105/tpc.8.4.735 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsuchiya T. and Eulgem T. (2010a). The Arabidopsis defense component EDM2 affects the floral transition in an FLC-dependent manner. Plant J. 62, 518-528. 10.1111/j.1365-313X.2010.04169.x [DOI] [PubMed] [Google Scholar]
- Tsuchiya T. and Eulgem T. (2010b). Co-option of EDM2 to distinct regulatory modules in Arabidopsis thaliana development. BMC Plant Biol. 10, 203 10.1186/1471-2229-10-203 [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Zanten M., Snoek L. B., Proveniers M. C. G. and Peeters A. J. (2009). The many functions of ERECTA. Trends Plant Sci. 14, 214-218. 10.1016/j.tplants.2009.01.010 [DOI] [PubMed] [Google Scholar]
- Wang Z.-Y., Seto H., Fujioka S., Yoshida S. and Chory J. (2001). BRI1 is a critical component of a plasma-membrane receptor for plant steroids. Nature 410, 380-383. 10.1038/35066597 [DOI] [PubMed] [Google Scholar]
- Wang H., Ngwenyama N., Liu Y., Walker J. C. and Zhang S. (2007). Stomatal development and patterning are regulated by environmentally responsive mitogen-activated protein kinases in Arabidopsis. Plant Cell 19, 63-73. 10.1105/tpc.106.048298 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wierzbicki A. T., Haag J. R. and Pikaard C. S. (2008). Noncoding transcription by RNA polymerase Pol IVb/Pol V mediates transcriptional silencing of overlapping and adjacent genes. Cell 135, 635-648. 10.1016/j.cell.2008.09.035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu T., Dai N., Chen J., Nagawa S., Cao M., Li H., Zhou Z., Chen X., De Rycke R., Rakusova H. et al. (2014). Cell surface ABP1-TMK auxin-sensing complex activates ROP GTPase signaling. Science 343, 1025-1028. 10.1126/science.1245125 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamuro C., Miki D., Zheng Z., Ma J., Wang J., Yang Z., Dong J. and Zhu J.-K. (2014). Overproduction of stomatal lineage cells in Arabidopsis mutants defective in active DNA demethylation. Nat. Commun. 5, 4062 10.1038/ncomms5062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang H. and Zhu J. K. (2011). RNA-directed DNA methylation. Curr. Opin. Plant Biol. 14, 142-147. 10.1016/j.pbi.2011.02.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng B., Wang Z., Li S., Yu B., Liu J.-Y. and Chen X. (2009). Intergenic transcription by RNA polymerase II coordinates Pol IV and Pol V in siRNA-directed transcriptional gene silencing in Arabidopsis. Genes Dev. 23, 2850-2860. 10.1101/gad.1868009 [DOI] [PMC free article] [PubMed] [Google Scholar]