Key Points
Neonatal and adult CD8+ T cells adopt different fates after infection because they are derived from distinct progenitor cells.
Lin28b may regulate the developmental switch from fetal to adult CD8+ T cells.
Abstract
During the ontogeny of the mammalian immune system, distinct lineages of cells arise from fetal and adult hematopoietic stem cells (HSCs) during specific stages of development. However, in some cases, the same immune cell type is produced by both HSC populations, resulting in the generation of phenotypically similar cells with distinct origins and divergent functional properties. In this report, we demonstrate that neonatal CD8+ T cells preferentially become short-lived effectors and adult CD8+ T cells selectively form long-lived memory cells after infection because they are derived from distinct progenitor cells. Notably, we find that naïve neonatal CD8+ T cells originate from a progenitor cell that is distinguished by expression of Lin28b. Remarkably, ectopic expression of Lin28b enables adult progenitors to give rise to CD8+ T cells that are phenotypically and functionally analogous to those found in neonates. These findings suggest that neonatal and adult CD8+ T cells belong to separate lineages of CD8+ T cells, and potentially explain why it is challenging to elicit memory CD8+ T cells in early life.
Introduction
Neonates often generate incomplete immunity against intracellular bacteria and viruses. Because CD8+ T cells play a critical role in protecting the host against these pathogens, it is important to understand how and why neonatal CD8+ T cells respond to infection differently than in adults. Recent studies suggest that neonatal CD8+ T cells fail to become memory cells because of an inherent propensity to rapidly proliferate and become terminally differentiated after antigenic stimulation.1-3 However, the underlying basis for these age-related differences remains unknown.
Several models might explain why neonatal CD8+ T cell adopt fates different from those of adults during infection. First, the “proliferation model” posits that developmental changes in the CD8+ T-cell response relate to differences in homeostatic proliferation before infection. When naïve CD8+ T cells enter a lymphopenic environment, they divide rapidly in response to homeostatic cytokines and upregulate phenotypic markers (CD44, CD122) indicative of cell differentiation.4,5 Thus, because newborn mice are nearly devoid of peripheral CD8+ T cells, it is possible that neonatal CD8+ T cells are less likely to develop into memory CD8+ T cells because the starting population is more differentiated than adults before infection.
Another possibility relates to the distinct hematopoietic stem cell (HSC) lineages that generate neonatal and adult CD8+ T cells (“origin model”). Although neonatal CD8+ T cells are derived from fetal liver HSCs that colonize the thymus during midgestation (approximately embryonic day [e] 13), adult CD8+ T cells are produced from bone marrow (BM) HSCs that seed the thymus just before birth (∼e20). Importantly, fetal HSCs turn over more rapidly6 and preferentially give rise to innatelike lymphocytes compared with adult HSCs.7 Thus, it is also possible that neonatal CD8+ T cells fail to form memory cells because they are created from distinct progenitor cells.
To discriminate between the proliferation and origin models, we compared neonatal and adult CD8+ T cells that had undergone equivalent homeostatic proliferation in the periphery, or were at the same stage of development in the thymus. We also compared T-cell maturation by fetal and adult precursors in the adult thymus and examined whether fetal-derived CD8+ T cells respond differently to infection than their adult counterparts. Collectively, our data reject the proliferation model and support the origin model, and imply that neonatal and adult CD8+ T cells adopt different fates after infection because they belong to separate lineages of naïve CD8+ T cells derived from distinct progenitors.
Materials and methods
Mice
B6-Ly5.2/Cr mice were purchased from Charles River Laboratories (Frederick, MD). TCR transgenic mice specific for the HSV-1 glycoprotein B498-505 peptide SSIEFARL8 (gBT-I mice) were provided by Janko Nikolich-Zugich (University of Arizona, Tucson, AZ) and crossed with Thy1.1 or C57BL/6 mice purchased from Jackson Laboratories (Bar Harbor, ME). Neonatal and adult gBT-I animals were used at 6 to 8 days old and at 2 to 4 months old, respectively. Mice with a tetracycline-inducible copy of human LIN28B on a C57BL/6 background (iLin28b mice) were obtained from George Daley (Harvard University, Cambridge, MA).9 Male mice were used for all experiments, and mice were housed under specific pathogen-free conditions at Cornell University College of Veterinary Medicine, accredited by the Assessment and Accreditation of Laboratory Animal Care.
Antibodies and flow cytofluorimetric analysis
Antibodies were purchased from eBioscience (San Diego, CA), Biolegend (San Diego, CA), Invitrogen (Carlsbad, CA), or BD Biosciences (Mountain View, CA). Sheep anti-human Lin28b was obtained from R&D Systems (Minneapolis, MN), and Alexa Fluor 488 rabbit-anti-sheep from Jackson Immunoresearch (West Grove, PA). Flow cytofluorimetric data were acquired using DiVa software from an LSRII equipped with 4 lasers (BD Biosciences). Analysis was performed with FlowJo (Tree Star, Ashland, OR).
Cell sorting
To purify subsets of CD44hiCD122hi and CD44loCD122lo CD8+ T cells from neonatal and adult gBT-I mice, CD8+ T cells were enriched using anti-CD8α microbeads (Miltenyi Biotec) and were subsequently labeled with antibodies against CD4, CD8, CD44, and CD122 and sorted to >95% purity on a fluorescence-activated cell sorting (FACS) Aria III (BD Biosciences). To purify single-positive (SP; CD8+CD4–) thymocytes from neonatal and adult gBT-I mice, CD4+ thymocytes were depleted by negative immunomagnetic selection via biotinylated antibody followed by streptavidin-coated microbeads (Miltenyi Biotec). Thymocytes were labeled with antibodies against CD4, CD8, Vα2, and Vβ8, and FACS-sorted to >90% purity. To isolate donor CD8+ T cells from chimeric mice, CD4+, CD19+, MHC-II+, and Ter119+ cells were depleted by negative immunomagnetic selection. Cells were labeled with antibodies against CD8, CD4, CD45.1, CD45.2, Thy1.1, and Thy1.2, and FACS-sorted to >98% purity for adoptive cotransfers. Antibodies against Vα2 and Vβ8 were added for sorts intended for RNA isolation.
In vitro stimulation of CD8+ T cells
SP CD8+ thymocytes were FACS-sorted from neonatal and adult gBT-I mice and labeled with carboxyfluorescein diacetate succinimidyl ester, as described,1 and cells were stimulated with plate-bound anti-CD3 (5 μg/mL) and anti-CD28 (20 μg/mL).
Intrathymic injection
To compare the progeny of different aged progenitor cells, we adapted a protocol from Adkins B.10 B6-Ly5.2 recipient mice were sublethally irradiated with 600 rads and injected intrathymically with 1 × 106 double-negative (DN, CD8–CD4–) thymocytes from fetal gBT-I Thy1.2 mice (14 days’ gestation) and adult gBT-I Thy1.1 mice. CD8–CD4– thymocytes were isolated from adult mice by depleting CD8+ and CD4+ cells using negative magnetic separation. Because 100% of thymocytes on embryonic day 14 are CD8–CD4– cells,10 whole thymocyte preparations were used from fetal gBT-I mice. Splenic CD8+ T cells were recovered 4 weeks posttransfer and cotransferred into new B6-Ly5.2 (CD45.1+Thy1.2+) recipients.
Adoptive cotransfers
To compare primary and secondary responses to infection, FACS-sorted splenic neonatal and adult CD44hiCD122hi or CD44loCD122lo CD8+ cells, or donor CD8+ cells from thymic transfers or BM chimeras were combined at a 1:1 ratio. Combined cells were suspended at 2 × 105 cells per mL of PBS and 100 ul of cells was injected i.v. into adult B6-Ly5.2 recipient mice. The next day, recipient mice were infected with WT LM-gB (5 × 103 CFU, i.v.) and later challenged with WT LM-gB (5 × 104 CFU, i.v.), as described.1
Bone marrow chimeras
Single and mixed BM chimeras were generated by obtaining BM from congenically marked gBT-I (Thy1.1+CD45.2+) and gBT-I iLin28b (Thy1.2+CD45.2+) mice. CD4+ and CD8+ T cells were depleted by magnetic separation, and 1 × 107 cells were injected (5 × 106 cells per donor for mixed chimeras) into lethally irradiated (990 rads) 7- to 8-week-old adult B6-Ly5.2 recipients. Forty-eight hours posttransplantation, mice were administered 1 mg/mL doxycycline with 10 mg/mL sucrose via drinking water to induce LIN28B expression for 8 weeks during reconstitution.
RNA sequencing
For mRNA sequencing, libraries were generated using 25 to 200 ng total RNA with the NEBNext Ultra RNA Library Prep Kit for Illumina (NEB) and sequenced on an Illumina HiSequation 2500, generating 100-nucleotide reads. The analysis was performed as previously described.3 Reads were aligned with Tophat11 (mm9), and CuffDiff was used to quantify the abundance and differential expression of each mRNA, using a false discovery rate of 5% for significance.12 Small RNA libraries were generated using 50 to 100 ng of total RNA using the NEBNext Small RNA Library Kit (NEB), and sequenced on an Illumina HiSequation 2500, generating 50-nucleotide reads. The analysis was performed as previously described.3 miRNAs were mapped and quantified using MirDeep213 (MirBase version 21) with Bowtie14 (mm9). The data have been deposited in the GEO Small Read Archive15 under accession number GSE80597.
Clustering was performed using the partitioning around the medoids method16 in R. Gene ontology was performed with DAVID17,18 to find enriched biological process terms with Benjamini-corrected P values < .05. Gene sets for enrichment statistics were downloaded from the Molecular Signatures Database (MSDB).19 For each cluster, we found the number of genes present in both the cluster and the data set (b), the total number of genes present in that cluster (n), the number of genes in the MSDB data set (B), and the total number of genes that had been clustered (N). Enrichment was calculated as (b/n)/(B/N). One-sided Fisher exact tests were used to measure significance.
Statistical analysis
Statistical analysis was performed using Prism (GraphPad Software, Inc, La Jolla, CA). Error bars represent standard error of the mean. Significance was determined by Student t test or one-way analysis of variance followed by Tukey multiple comparisons test. Significance is denoted as: *P < .05, **P < .01, ***P < .001, and ****P < .0001.
Results
Neonatal and adult CD8+ T cells exhibit distinct phenotypes
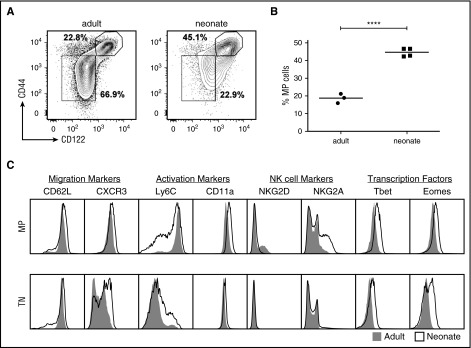
We first sought to test the proliferation model and determine whether differences in homeostatic proliferation contribute to cell-intrinsic changes in neonatal CD8+ T cells. To examine this possibility, we stained CD8+ T cells from uninfected neonatal and adult gBT-I mice for CD44 and CD122, which are used to define cells that have undergone significant amounts of homeostatic proliferation. Such CD44hiCD122hi are referred to as memory phenotype naïve cells, because they display certain characteristics of memory cells without having encountered foreign antigen.20,21 The neonatal CD8+ T cells were comprised of twofold more CD44hiCD122hi memory phenotype (MP) cells, whereas the adult CD8+ T cells were made up of more CD44loCD122lo true naïve (TN) cells (Figure 1A-B). Importantly, we did not observe significant numbers of MP cells in the thymi of neonatal mice, and their appearance in the periphery coincided with the relative number of CD8+ T cells that are actively dividing, as measured by Ki-67 expression (supplemental Figure 1, available on the Blood Web site). Together, these data are consistent with earlier reports indicating that large number of CD8+ T cells undergo homeostatic expansion in early life.22,23 We also compared the phenotype of neonatal and adult MP CD8+ T cells in the periphery, and observed multiple age–related differences in the expression of natural killer cell receptors, activation markers, and T-box transcription factors (Figure 1C). Thus, neonatal CD8+ T cells that have undergone homeostatic expansion in the periphery express a distinct phenotypic signature, even when compared with matched adult counterparts (Figure 1C).
Figure 1.
Memory phenotype CD8+T cells are more abundant in early life and express different surface markers and transcription factors. (A) Surface expression of CD44 and CD122 by splenic CD8+ T cells from uninfected neonatal and adult gBT-I. (B) Statistical analysis of the percentages of MP cells from neonatal and adult gBT-I mice. (C) Comparison of various surface markers and transcription factors expressed in TN and MP cells from uninfected neonatal (line) or adult (shaded) gBT-I mice. Data are representative of at least 3 experiments. Significance was determined by Student t test (****P < .0001).
TN and MP CD8+ T cells from different aged mice express distinct gene expression profiles
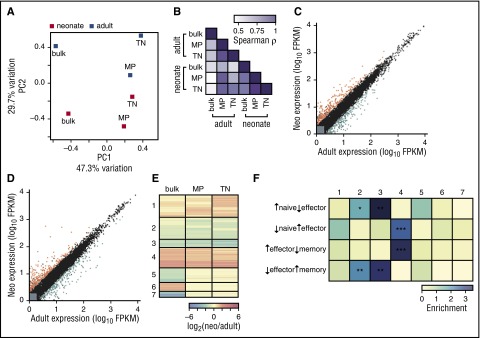
We previously found that splenic neonatal CD8+ T cells upregulate genes needed for cell cycle progression and effector function,3 similar to adult MP cells.24 Thus, gene expression differences observed between bulk adult and neonatal CD8+ T cells could be explained by the different proportions of MP and TN cells. To investigate this possibility, we sequenced transcriptomes from TN, MP, and bulk splenic cells from neonatal and adult mouse CD8+ T cells (supplemental Table 1). Instead, we found that adult cells were distinct from neonatal cells for all populations, which we visualized using principal component analysis (Figure 2A-B; see supplemental Figure 2 for transcripts contributing to differences). Hundreds of genes were differentially expressed between adults and neonates in both the TN and MP populations (Figure 2C-D), several of which were consistent with protein abundances observed in Figure 1C. Clustering genes by their expression patterns showed that most genes had equivalent expression differences between adults and neonates (clusters 1-4), whereas the remainder had age-dependent expression changes in the bulk CD8+ population alone (clusters 5-7, Figure 2E; supplemental Table 1). Upon comparing the genes in each cluster (Figure 2E) with gene sets that characterize CD8+ T cells at different infection states,19,25 we found that genes upregulated in neonates in both MP and TN cells (cluster 4) are significantly enriched in genes typifying effector cells (Figure 2F) and gene ontology terms related to cell division (P < 10−8, supplemental Table 1). Similarly, genes downregulated in neonates in both MP and TN cells (clusters 2 and 3) are enriched in naïve and memory cell genes, consistent with previous data.3 Most gene expression differences between adults and neonates therefore cannot be attributed simply to the increase in MP cells observed in early life.
Figure 2.
Both TN and MP CD8+T cells from uninfected neonatal gBT-I mice express different genes from adults. (A) Principal component analysis on RNA-Seq data. Mean FPKM values from well-expressed genes were used from adult and neonatal bulk (total), TN, and MP cells. Bulk adult, MP adult, and MP neonatal samples consist of 3 pooled biological replicates; bulk neonatal, TN adult, and TN neonatal samples consist of 2 pooled biological replicates. The percentage of the overall variation accounted for by principal components 1 (x-axis) and 2 (y-axis) is indicated for each axis. Gene loadings for principal components 1 and 2 are shown in supplemental Figure 2. (B) Color-coded pairwise Spearman rank correlation coefficients comparing FPKM values for genes that are significantly differentially expressed between adults and neonates in at least 1 sample; P < 1015 for all comparisons. (C) Gene expression values for adults and neonates in MP cells. Gray indicates lowly expressed genes, black indicates nondifferentially expressed genes, orange indicates the 126 genes upregulated in neonatal cells, and blue indicates the 159 genes upregulated in adult cells. (D) Gene expression values for adults and neonates in TN cells, where 204 genes are upregulated in neonatal cells and 195 genes are upregulated in adult cells. (E) Clustering of genes in all samples. Fold-change differences for significantly differentially expressed genes were calculated between adults and neonates. Clustering was performed to identify genes with similar differences in expression in each sample; fold-change for each gene is plotted in each sample, and genes are shown in their clusters. (F) Genes in each cluster were compared with genes that define naïve cells before infection and effector or memory cells after infection. Enrichment was calculated as number of genes in each cluster compared with the number expected. Significance was determined by Fisher exact tests; *P < .05, **P < .005, ***P < .0005. See supplemental Table 1 for gene expression values and clustering.
TN and MP CD8+ T cells from different aged mice respond differently to infection
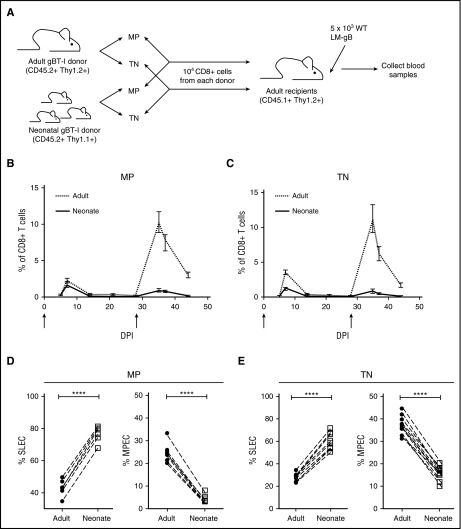
Next, we wanted to address whether neonatal CD8+ T cells preferentially become terminally differentiated during an immune response because a larger proportion of the naïve pool has undergone homeostatic proliferation before infection. We cotransferred equivalent phenotypic subsets (MP or TN cells) from neonatal and adult gBT-I mice into the same adult recipient mice (Figure 3A) and tracked their response to infection. Regardless of their initial phenotype, neonatal donor cells preferentially differentiated into short-lived effectors (SLECs; KLRG1hiCD127lo) during the effector stage (Figure 3D-E) and failed to transition into the long-lived memory pool. As a consequence, the memory recall response was almost entirely dominated by adult donor–derived cells (Figure 3B-C). Collectively, these data argue against the hypothesis that age-related differences in homeostatic proliferation underlie the altered behavior of different-aged CD8+ T cells after infection.
Figure 3.
Both TN and MP CD8+T cells from neonatal gBT-I mice preferentially become short-lived effectors after infection. (A) Schematic of experimental design: TN and MP cells were sorted from congenically marked gBT-I adult (CD45.2, Thy1.2) and neonatal (CD45.2, Thy1.1) mice and adoptively cotransferred (1:1 ratio) into adult recipient mice (CD45.1, Thy1.2). These recipients were infected with 5 × 103 colony-forming unit (CFU) LM-gB and serially bled to examine primary CD8+ T-cell responses. Recipients were also challenged at 28 dpi with 5 × 104 WT LM-gB to assess the memory recall response. Arrows indicate days of infections. Relative numbers and ratios of TN (B) and MP (C) donor cells from different aged mice at various times after infection. Percentages of MP (D) and TN (E) donor cells from different aged mice that display a short-lived effector cell (SLEC) or memory precursor effector cell (MPEC) phenotype at the peak of the primary response (7 dpi). Data are representative of 2 experiments (n = 9-12 mice/group). Significance was assessed by Student t test (****P < .0001). DPI, days postinfection.
Single positive CD8+CD4– thymocytes in neonatal mice exhibit unique genetic properties compared with adults
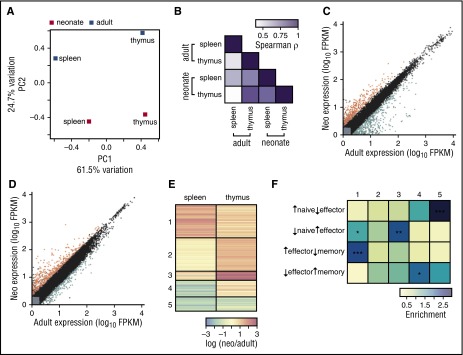
We next sought to test the origin model and determine whether cell-intrinsic differences between neonatal and adult CD8+ T cells relate to their distinct HSC origin. We compared gene expression profiles in single-positive (SP) CD8+ T cells from neonatal and adult thymi to compare transcriptomes at the same stage of thymic development, and to control for age-related differences in maturation and proliferation (supplemental Table 1; validated protein levels of selected genes in supplemental Figure 3). Overall, we found greater variation between splenic and thymic CD8+ T cells, regardless of age (Figure 4A-B). Widespread gene expression differences do exist between adults and neonates in both types of cells (Figure 4C-D and supplemental Table 1), however, indicating that neonatal CD8+ T cells exhibit a divergent pattern of gene expression even at the time they are initially created. To gain insight into these differences, we clustered genes based on their coexpression patterns (Figure 4E-F; supplemental Table 1). Genes with differential expression between adults and neonates in the spleen generally have similar differences in the thymus (clusters 1 and 5), yet additional genes show differences in the thymus only. Interestingly, genes that are upregulated only in the neonatal thymus (cluster 3) are enriched in effector-cell genes, suggesting that neonatal CD8+ T cells are more effectorlike. Genes that are downregulated in splenic neonates with little difference in thymic cells (cluster 4) have memorylike expression, whereas genes downregulated in neonates in both the thymus and spleen (cluster 5) are enriched for genes found in naïve cells. Overall, differences in gene expression between neonatal and adult CD8+ T cells, beginning at the cells’ generation and continuing throughout the course of development, suggest neonatal CD8+ T cells represent a distinct and more effectorlike population of lymphocytes that associates with their progenitor cell origin.
Figure 4.
Neonatal single-positive CD8+T cells from the thymus express different genes and proliferate faster than adults. (A-F) RNAseq was performed on neonate and adult gBT-I CD8+ thymocytes and splenocytes. (A) Principal component analysis. Mean FPKM values from well-expressed genes were used from adult and neonatal naïve splenic and thymic CD8+ T cells. The naïve splenic adult sample consists of 3 pooled biological replicates; the remaining samples consist of 2 pooled biological replicates. The percentage of the overall variation accounted for by principal components 1 (x-axis) and 2 (y-axis) is indicated for each axis. Gene loadings are shown for principal components 1 and 2 in supplemental Figure 4. (B) Color-coded pairwise Spearman rank correlation coefficients comparing FPKM values for genes that are significantly differentially expressed between adults and neonates in at least one sample; P < 1015 for all comparisons. (C) Gene expression values for adults and neonates in splenic cells. Gray indicates lowly expressed genes, black indicates nondifferentially expressed genes, orange indicates the 118 genes upregulated in neonatal cells, and blue indicates the 153 genes upregulated in adult cells. (D) Gene expression values for adults and neonates in thymic cells, where 264 genes are upregulated in neonatal cells and 199 genes are upregulated in adult cells. Clustering of genes in naïve splenic and thymic CD8+ T-cell transcriptomes. Fold-change differences for significantly differentially expressed genes were calculated between adults and neonates. (E) Clustering was performed to identify genes with similar differences in expression in each sample; fold-change for each gene is plotted in each sample, and genes are shown in their clusters. (F) Genes in each cluster were compared with genes that define naïve, effector, or memory cells. Enrichment was calculated as number of genes in each cluster compared with the number expected. See supplemental Table 1 for gene expression values and clustering.
To uncover functional differences between thymic CD8+ T cells from neonatal and adult mice, we compared the ability of SP CD8+ T cells from neonatal and adult thymi to proliferate after in vitro stimulation. We found that neonatal SP CD8+ T cells had divided significantly more than their adult counterparts (supplemental Figure 5). Thus, the enhanced ability of peripheral neonatal CD8+ T cells to proliferate after stimulation appears to originate in their immediate precursors in the thymus.
Fetal progenitors give rise to mature CD8+ T cells that adopt distinctive fates after infection
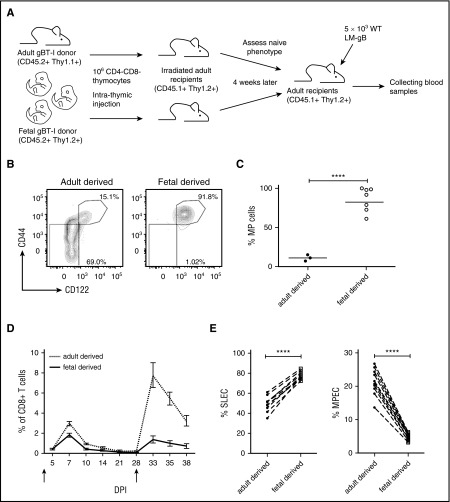
To confirm that thymic precursors in neonate and adult mice give rise to distinct lineages of CD8+ T cells, we intrathymically injected equivalent numbers of DN thymocytes from fetal and adult gBT-I mice into adult recipient mice, allowing us to control for age-related differences in the thymic stromal environment (Figure 5A). Four weeks after injection, we collected the peripheral cells and compared their phenotype. Strikingly, nearly 100% of fetal-derived CD8+ T cells were MP cells, whereas adult-derived CD8+ T cells were comprised of many more TN cells (Figure 5B-C). To determine whether the progeny of fetal and adult precursors behaved similarly to neonatal and adult CD8+ T cells after microbial challenge, we cotransferred fetal- and adult-derived CD8+ T cells into adult recipient mice and compared their ability to respond to infection. We found that fetal-derived cells exhibited a terminally differentiated phenotype at the peak of the primary response (7dpi), and failed to survive during later stages of the infection (Figure 5D-E). As a result, the memory recall response was largely comprised of adult-derived CD8+ T cells (Figure 5D). Collectively, these data support the origin model, and suggest that neonatal and adult CD8+ T cells adopt different fates after infection because they are derived from different progenitor cells.
Figure 5.
Fetal progenitors give rise to mature CD8+T cells that adopt different fates after infection. (A) Schematic of experimental design: CD8–CD4– progenitors from congenically marked gBT-I adult (CD45.2, Thy1.1) and neonatal (CD45.2, Thy1.2) mice were transferred by intrathymic injection into sublethally irradiated congenic recipient mice (CD45.1, Thy1.2) separately. Four weeks later, progenitor-derived splenic CD8+ T cells were sorted and adoptively co-transferred (1:1 ratio) into new congenic recipient mice (CD45.1, Thy1.2). These recipients were infected with 5 × 103 LM-gB and serially bled to examine CD8+ T-cell responses. (B-C) Naïve phenotypes of progenitor-derived CD8+ T cells before adoptive cotransfer. (B) Representative plots displaying CD44 and CD122 expression by gBT-I progenitor-derived CD8+ T cells 4 weeks post–intrathymic transfer in blood. (C) Statistical analysis of the percentages of MP cells from gBT-I adult and fetal progenitor-derived cells. (D) Relative numbers of gBT-I adult (dashed) and fetal (solid) progenitor-derived donor CD8+ T cells postinfection and re-challenge (5 × 104 WT LM-gB); arrows indicate days of infections. (E) Percentages of gBT-I adult and fetal progenitor-derived donor CD8+ T cells that are SLECs and MPECs. Data are representative of 2 experiments (n = 4-11 mice/group) (****P < .0001).
Lin28b reprograms adult CD8+ T cells to behave more like neonatal cells
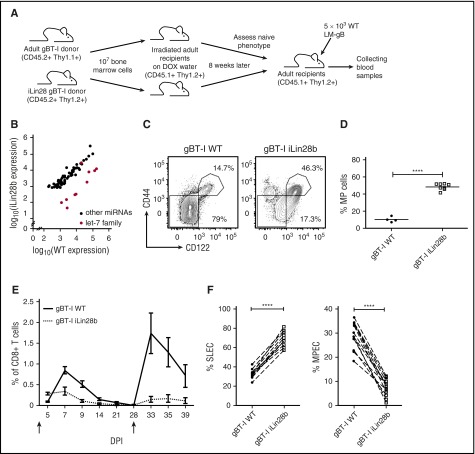
Previous reports suggest that many key biological differences between fetal and adult HSCs can be attributed to changes in the expression of Lin28b, a conserved and developmentally regulated RNA-binding protein that represses the let-7 microRNA,26,27 among other roles. Importantly, Lin28b is selectively expressed in fetal progenitors (supplemental Figure 6), and overexpression of Lin28b in adult BM cells promotes the development of fetal-like B cells (B-1a B cells, marginal zone B cells) and innate-like T cells (NKT cells, γ δ T cells).28 If Lin28b underlies key developmental-related differences in CD8+ T cells, we reasoned that ectopic expression of Lin28 in adult progenitor cells would result in the development of “neonatal-like” cells. To test this, we generated BM chimeras by reconstituting lethally irradiated mice with gBT-I WT or iLin28b hematopoietic cells (Figure 6A). After reconstitution, robust expression of Lin28b was confirmed in CD8+ T cells (supplemental Figure 7A-B), and its expression was sufficient to downregulate all let-7 family members except let-7c, which is not subject to control by Lin2829 (Figure 6B; supplemental Table 1). Phenotypic analysis of CD8+ T cells from chimeric mice revealed that forced expression of Lin28b in adult progenitor cells promotes the generation of MP cells that develop after thymic egress, reminiscent of neonatal cells (Figure 6C-D; supplemental Figure 7C-D). Notably, in mixed BM chimeras, wild-type (WT) CD8+ T cells produced in the same thymus as iLin28b CD8+ T cells did not give rise to more MP CD8+ T cells (supplemental Figure 8), suggesting that Lin28b drives the development of MP CD8+ T cells in a cell-intrinsic manner.
Figure 6.
Lin28b reprograms adult CD8+T cells to behave more like neonatal cells. (A) Schematic of experimental design: congenic gBT-I adult (Thy1.1) and iLin28b gBT-I adult (Thy1.2) BM cells were transferred into lethally irradiated congenic WT recipient mice (CD45.1) separately. Lin28b expression was induced by DOX in drinking water. Eight weeks later, splenic donor CD8+ T cells were sorted and adoptively cotransferred into congenic WT recipient mice (CD45.1). These recipients were infected with 5 × 103 WT LM-gB and serially bled to examine CD8+ T-cell responses. (B-D) Naïve phenotypes of gBT-I WT or gBT-I iLin28b CD8+ T cells before adoptive cotransfer. (B) Expression of miRNAs in adult WT gBT-I and adult iLin28b splenic donor CD8+ T cells at 8 weeks after reconstitution. (C) Representative plots showing CD44 and CD122 expression by gBT-I BM-derived splenic CD8+ T cells 8 weeks after reconstitution. (D) Statistical analysis of the percentages of MP cells from WT gBT-I adult and iLin28b adult CD8+ T cells. (E) Relative numbers of iLin28b gBT-I adult (dashed) and WT gBT-I adult (solid) BM-derived donor CD8+ T cells postinfection and re-challenge (5 × 104 WT LM-gB); arrows indicate days of infections. (F) Percentages of gBT-I adult and fetal progenitor-derived donor CD8+ T cells that are SLECs and MPECs. Data are representative of 2 experiments (****P < .0001).
To determine whether Lin28b expression influences the fate of CD8+ T cells after infection, we adoptively cotransferred equal numbers of WT and iLin28b CD8+ T cells from BM chimeras into adult recipients, and infected the recipients (Figure 6A). The data revealed that WT cells predominate as the immune response progressed. The few iLin28b CD8+ cells that remained at day 28 responded poorly to secondary challenge, and the recall response was entirely comprised of WT cells (Figure 6E). We also examined changes in the expression of KLRG1 and CD127 at the peak of the primary response, which revealed that most iLin28b cells expressed a SLEC phenotype, whereas WT cells exhibited a balance of SLEC and MPEC phenotypes (Figure 6F). Together, these data demonstrate that ectopic expression of Lin28b in adult progenitor cells enables them to give rise to CD8+ T cells that are phenotypically and functionally analogous to those found in neonates, indicating that increased expression of Lin28b in neonatal progenitor cells underlies their preferential terminal differentiation into SLECs during infection.
Discussion
Previous work has demonstrated that the ontogeny of the immune system does not progress in a linear manner from fetal life to adulthood.30-34 Instead, the prevailing model argues that distinct “steps” or “layers” develop sequentially from progenitor cells that are metabolically and epigenetically distinct.35-39 As a consequence, unique populations of immune cells (B1a-B cells and dendritic epidermal T cells) are generated during different windows of development.40-42 In this report, we provide compelling support for the existence of a distinct layer of CD8+ T cells that is produced in early life and demonstrate that neonatal and adult CD8+ T cells respond differently to infection because they are derived from distinct progenitor cells.
The “layered immune hypothesis” may also be relevant to the ontogeny of MP CD8+ T cells. Previous work has suggested that MP cells are driven by lymphopenia-induced proliferation during early stages of development.22,43 However, our work demonstrates that fetal-derived CD8+ T cells preferentially acquire a memory phenotype, even when placed in the same environment as adult-derived cells (Figure 5). These findings suggest that MP cell development cannot be attributed solely to age-related changes in the environment and indicates that cell-intrinsic differences between neonatal and adult CD8+ T cells should also be considered. It will be important to determine whether sensitivity to homeostatic cytokines and/or proximity toward IL-7– and IL-15–dependent niches in lymphoid tissue is linked to their developmental origin. Indeed, perhaps MP cells in young adult mice are derived largely from the fetal layers of HSCs.
Intrathymic transfer experiments allowed us to control for differences in the amount of post–thymic maturation. As recent thymic emigrants (RTEs) make up a larger percentage of naïve cells in neonatal mice (100%) compared with adult mice (∼10%-20%)44 and skew toward the short-lived effector lineage after infection,45 there has been some speculation that neonatal CD8+ T cells produce fewer memory cells because they have yet to undergo a sufficient amount of post–thymic maturation. Although we were unable to directly compare neonatal and adult RTEs because of dramatic differences in the rates of homeostatic proliferation, it is worth pointing out that we have compared the progeny of fetal and adult progenitor cells after giving these cells the same length of post–thymic maturation (∼4 weeks), and we still observed cell-intrinsic differences (Figure 5). Thus, although the age of the cell clearly influences its ability to respond to stimulation, the age of the animal in which the cell is created may ultimately be a greater determining factor for how a cell will respond after infection.
What are the molecular mechanisms that underlie the developmental switch from fetal to adult CD8+ T-cell production? Our work points toward a role for Lin28b in this process, which is highly expressed in fetal animals and then downregulated later in development. Previous reports have suggested that many posttranscriptional and metabolic differences between fetal and adult HSCs can be attributed to the expression of Lin28b.26,46,47 One way in which Lin28b regulates fetal HSCs is by blocking the biogenesis of let-7 miRNAs, which control the developmental timing in a wide range of species.28,48-51 More recently, let-7 has gained attention for its ability to behave as a tumor suppressor in human cancer cells by repressing cell proliferation,52-54 potentially by targeting cell cycle factors and genes in the mTOR pathway.9 In this report, we demonstrate that ectopic Lin28b expression in adult progenitor cells gives rise to CD8+ T cells that undergo more rapid proliferation and effector cell differentiation (Figure 6). To our knowledge, this is the first study demonstrating how Lin28 expression alters the CD8+ T-cell response to infection. However, additional studies are required to determine the degree to which Lin28b is controlling CD8+ T-cell responses by reducing expression of let-7, enhancing the translation of metabolic enzymes55 or other factors.56,57 It will be essential to examine more closely the changes in gene expression at the individual cell level to uncover the molecular circuitry regulated by Lin28b and let-7 in CD8+ T cells. We are currently using both gain- and loss-of-function gene targeting approaches to identify these networks. Clearly, more work studying the role of let-7/Lin28 in CD8+ T-cell development is warranted.
It is intriguing to speculate whether Lin28b was co-opted during the evolution of mammalian immune system to serve as rheostat of effector and memory cell differentiation during stages of life. A common feature of lymphocytes derived from Lin28b+ fetal HSCs is that they tend to respond more rapidly to infection than their adult-derived counterparts.1,58 Responding with a more vigorous primary CD8+ T cell response may help protect the host at critical stages of immune development, but likely comes at the cost of not being able to form long-lived memory cells. However, because the neonatal CD8+ T-cell repertoire is extremely limited, it is likely that neonatal cells are of lesser value in the memory pool, because more “fit” CD8+ T cells will arise later in adulthood. Although this model remains to be tested, our data provide insight into the developmental-related factors that influence effector and memory CD8+ T-cell differentiation during periods of life.
Acknowledgments
The authors thank Dorian La Tocha (Director of the Flow Cytometry Core) for expert sorting assistance and Jennifer Grenier (RNA sequencing Core) for generation ofA sequencing data and help with analysis.
The Cytometry Core is supported in part by the Empire State Stem Cell Fund, New York State Department of Health, and Contract #C026718. The RNA sequencing was performed on the Illumina HiSeq 2500, which is funded from National Institutes of Health, Office of the Director grant 1S10OD010693-01 and supported by Core B of P50HD076210. This work was supported by the National Institutes of Health, National Institute of Allergy and Infectious Disease grants R01AI105265 and R01AI110613 (B.D.R.); National Institutes of Health, National Institute of General Medical Sciences grants R01GM105668 and P50HD076210 (A.G.); the Cornell Center for Vertebrate Genomics (B.D.R. and A.G.); and National Science Foundation Graduate Research Fellowship award 2011078441 (E.M.W.).
Footnotes
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Authorship
Contribution: J.W., E.M.W., and N.B.W. performed experiments, analyzed all data, and wrote the manuscript; N.L.S. provided experimental assistance and aided in writing the manuscript; and A.G. and B.D.R. helped design experiments, interpret data, and assisted in writing the paper.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
The current affiliation for E.M.W. is Department of Molecular, Cell, and Developmental Biology, University of California, Los Angeles, Los Angeles, CA.
Correspondence: Brian D. Rudd, Department of Microbiology and Immunology, C5 147 Veterinary Medical Center, Cornell University, Ithaca, NY 14853; e-mail: bdr54@cornell.edu; and Andrew Grimson, Department of Molecular Biology and Genetics, 455 Biotechnology Building, Cornell University, Ithaca, NY 14853; e-mail: agrimson@cornell.edu.
References
- 1.Smith NL, Wissink E, Wang J, et al. Rapid proliferation and differentiation impairs the development of memory CD8+ T cells in early life. J Immunol. 2014;193(1):177-184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Reynaldi A, Smith NL, Schlub TE, Venturi V, Rudd BD, Davenport MP. Modeling the dynamics of neonatal CD8(+) T-cell responses. Immunol Cell Biol. 2016;94(9):838-848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wissink EM, Smith NL, Spektor R, Rudd BD, Grimson A. MicroRNAs and Their Targets Are Differentially Regulated in Adult and Neonatal Mouse CD8+ T Cells. Genetics. 2015;201(3):1017-1030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Takada K, Jameson SC. Naive T cell homeostasis: from awareness of space to a sense of place. Nat Rev Immunol. 2009;9(12):823-832. [DOI] [PubMed] [Google Scholar]
- 5.Surh CD, Sprent J. Homeostasis of naive and memory T cells. Immunity. 2008;29(6):848-862. [DOI] [PubMed] [Google Scholar]
- 6.Bowie MB, McKnight KD, Kent DG, McCaffrey L, Hoodless PA, Eaves CJ. Hematopoietic stem cells proliferate until after birth and show a reversible phase-specific engraftment defect. J Clin Invest. 2006;116(10):2808-2816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Barber CL, Montecino-Rodriguez E, Dorshkind K. Reduced production of B-1-specified common lymphoid progenitors results in diminished potential of adult marrow to generate B-1 cells. Proc Natl Acad Sci USA. 2011;108(33):13700-13704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mueller SN, Heath W, McLain JD, Carbone FR, Jones CM. Characterization of two TCR transgenic mouse lines specific for herpes simplex virus. Immunol Cell Biol. 2002;80(2):156-163. [DOI] [PubMed] [Google Scholar]
- 9.Zhu H, Shyh-Chang N, Segrè AV, et al. ; DIAGRAM Consortium; MAGIC Investigators. The Lin28/let-7 axis regulates glucose metabolism. Cell. 2011;147(1):81-94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Adkins B. Developmental regulation of the intrathymic T cell precursor population. J Immunol. 1991;146(5):1387-1393. [PubMed] [Google Scholar]
- 11.Trapnell C, Pachter L, Salzberg SL. TopHat: discovering splice junctions with RNA-Seq. Bioinformatics. 2009;25(9):1105-1111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Trapnell C, Hendrickson DG, Sauvageau M, Goff L, Rinn JL, Pachter L. Differential analysis of gene regulation at transcript resolution with RNA-seq. Nat Biotechnol. 2013;31(1):46-53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Friedländer MR, Chen W, Adamidi C, et al. Discovering microRNAs from deep sequencing data using miRDeep. Nat Biotechnol. 2008;26(4):407-415. [DOI] [PubMed] [Google Scholar]
- 14.Langmead B, Trapnell C, Pop M, Salzberg SL. Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol. 2009;10(3):R25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Barrett T, Wilhite SE, Ledoux P, et al. NCBI GEO: archive for functional genomics data sets--update. Nucleic Acids Res. 2013;41(Database issue):D991-D995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Reynolds AP, Richards G, de la Iglesia B, Rayward-Smith VJ. Clustering Rules: A Comparison of Partitioning and Hierarchical Clustering Algorithms. J Math Model Algorithms. 2006;5(4):475-504. [Google Scholar]
- 17.Huang W, Sherman BT, Lempicki RA. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc. 2009;4(1):44-57. [DOI] [PubMed] [Google Scholar]
- 18.Huang W, Sherman BT, Lempicki RA. Bioinformatics enrichment tools: paths toward the comprehensive functional analysis of large gene lists. Nucleic Acids Res. 2009;37(1):1-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Subramanian A, Tamayo P, Mootha VK, et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA. 2005;102(43):15545-15550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Goldrath AW, Bogatzki LY, Bevan MJ. Naive T cells transiently acquire a memory-like phenotype during homeostasis-driven proliferation. J Exp Med. 2000;192(4):557-564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kieper WC, Jameson SC. Homeostatic expansion and phenotypic conversion of naïve T cells in response to self peptide/MHC ligands. Proc Natl Acad Sci USA. 1999;96(23):13306-13311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Min B, McHugh R, Sempowski GD, Mackall C, Foucras G, Paul WE. Neonates support lymphopenia-induced proliferation. Immunity. 2003;18(1):131-140. [DOI] [PubMed] [Google Scholar]
- 23.Schönland SO, Zimmer JK, Lopez-Benitez CM, et al. Homeostatic control of T-cell generation in neonates. Blood. 2003;102(4):1428-1434. [DOI] [PubMed] [Google Scholar]
- 24.Goldrath AW, Luckey CJ, Park R, Benoist C, Mathis D. The molecular program induced in T cells undergoing homeostatic proliferation. Proc Natl Acad Sci USA. 2004;101(48):16885-16890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kaech SM, Hemby S, Kersh E, Ahmed R. Molecular and functional profiling of memory CD8 T cell differentiation. Cell. 2002;111(6):837-851. [DOI] [PubMed] [Google Scholar]
- 26.Thornton JE, Gregory RI. How does Lin28 let-7 control development and disease? Trends Cell Biol. 2012;22(9):474-482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jiang S, Baltimore D. RNA-binding protein Lin28 in cancer and immunity. Cancer Lett. 2016;375(1):108-113. [DOI] [PubMed] [Google Scholar]
- 28.Yuan J, Nguyen CK, Liu X, Kanellopoulou C, Muljo SA. Lin28b reprograms adult bone marrow hematopoietic progenitors to mediate fetal-like lymphopoiesis. Science. 2012;335(6073):1195-1200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Triboulet R, Pirouz M, Gregory RI. A Single Let-7 MicroRNA Bypasses LIN28-Mediated Repression. Cell Reports. 2015;13(2):260-266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Krow-Lucal ER, McCune JM. Distinct functional programs in fetal T and myeloid lineages. Front Immunol. 2014;5:314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Krow-Lucal ER, Kim CC, Burt TD, McCune JM. Distinct functional programming of human fetal and adult monocytes. Blood. 2014;123(12):1897-1904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mold JE, McCune JM. At the crossroads between tolerance and aggression: Revisiting the “layered immune system” hypothesis. Chimerism. 2011;2(2):35-41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mold JE, Venkatasubrahmanyam S, Burt TD, et al. Fetal and adult hematopoietic stem cells give rise to distinct T cell lineages in humans. Science. 2010;330(6011):1695-1699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mold JE, Michaëlsson J, Burt TD, et al. Maternal alloantigens promote the development of tolerogenic fetal regulatory T cells in utero. Science. 2008;322(5907):1562-1565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kim I, Saunders TL, Morrison SJ. Sox17 dependence distinguishes the transcriptional regulation of fetal from adult hematopoietic stem cells. Cell. 2007;130(3):470-483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Mochizuki-Kashio M, Mishima Y, Miyagi S, et al. Dependency on the polycomb gene Ezh2 distinguishes fetal from adult hematopoietic stem cells. Blood. 2011;118(25):6553-6561. [DOI] [PubMed] [Google Scholar]
- 37.Park IK, Qian D, Kiel M, et al. Bmi-1 is required for maintenance of adult self-renewing haematopoietic stem cells. Nature. 2003;423(6937):302-305. [DOI] [PubMed] [Google Scholar]
- 38.Hock H, Hamblen MJ, Rooke HM, et al. Gfi-1 restricts proliferation and preserves functional integrity of haematopoietic stem cells. Nature. 2004;431(7011):1002-1007. [DOI] [PubMed] [Google Scholar]
- 39.Hock H, Meade E, Medeiros S, et al. Tel/Etv6 is an essential and selective regulator of adult hematopoietic stem cell survival. Genes Dev. 2004;18(19):2336-2341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Havran WL, Allison JP. Origin of Thy-1+ dendritic epidermal cells of adult mice from fetal thymic precursors. Nature. 1990;344(6261):68-70. [DOI] [PubMed] [Google Scholar]
- 41.Havran WL, Allison JP. Developmentally ordered appearance of thymocytes expressing different T-cell antigen receptors. Nature. 1988;335(6189):443-445. [DOI] [PubMed] [Google Scholar]
- 42.Hayakawa K, Hardy RR, Herzenberg LA, Herzenberg LA. Progenitors for Ly-1 B cells are distinct from progenitors for other B cells. J Exp Med. 1985;161(6):1554-1568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Akue AD, Lee JY, Jameson SC. Derivation and maintenance of virtual memory CD8 T cells. J Immunol. 2012;188(6):2516-2523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Fink PJ. The biology of recent thymic emigrants. Annu Rev Immunol. 2013;31:31-50. [DOI] [PubMed] [Google Scholar]
- 45.Makaroff LE, Hendricks DW, Niec RE, Fink PJ. Postthymic maturation influences the CD8 T cell response to antigen. Proc Natl Acad Sci USA. 2009;106(12):4799-4804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Shyh-Chang N, Daley GQ. Lin28: primal regulator of growth and metabolism in stem cells. Cell Stem Cell. 2013;12(4):395-406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Copley MR, Babovic S, Benz C, et al. The Lin28b-let-7-Hmga2 axis determines the higher self-renewal potential of fetal haematopoietic stem cells. Nat Cell Biol. 2013;15(8):916-925. [DOI] [PubMed] [Google Scholar]
- 48.Reinhart BJ, Slack FJ, Basson M, et al. The 21-nucleotide let-7 RNA regulates developmental timing in Caenorhabditis elegans. Nature. 2000;403(6772):901-906. [DOI] [PubMed] [Google Scholar]
- 49.Lagos-Quintana M, Rauhut R, Lendeckel W, Tuschl T. Identification of novel genes coding for small expressed RNAs. Science. 2001;294(5543):853-858. [DOI] [PubMed] [Google Scholar]
- 50.Rodriguez A, Griffiths-Jones S, Ashurst JL, Bradley A. Identification of mammalian microRNA host genes and transcription units. Genome Res. 2004;14(10A):1902-1910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kloosterman WP, Plasterk RH. The diverse functions of microRNAs in animal development and disease. Dev Cell. 2006;11(4):441-450. [DOI] [PubMed] [Google Scholar]
- 52.Takamizawa J, Konishi H, Yanagisawa K, et al. Reduced expression of the let-7 microRNAs in human lung cancers in association with shortened postoperative survival. Cancer Res. 2004;64(11):3753-3756. [DOI] [PubMed] [Google Scholar]
- 53.Kuehbacher A, Urbich C, Zeiher AM, Dimmeler S. Role of Dicer and Drosha for endothelial microRNA expression and angiogenesis. Circ Res. 2007;101(1):59-68. [DOI] [PubMed] [Google Scholar]
- 54.Barh D, Malhotra R, Ravi B, Sindhurani P. MicroRNA let-7: an emerging next-generation cancer therapeutic. Curr Oncol. 2010;17(1):70-80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Shyh-Chang N, Zhu H, Yvanka de Soysa T, et al. Lin28 enhances tissue repair by reprogramming cellular metabolism. Cell. 2013;155(4):778-792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wilbert ML, Huelga SC, Kapeli K, et al. LIN28 binds messenger RNAs at GGAGA motifs and regulates splicing factor abundance. Mol Cell. 2012;48(2):195-206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Zeng Y, Yao B, Shin J, et al. Lin28A Binds Active Promoters and Recruits Tet1 to Regulate Gene Expression. Mol Cell. 2016;61(1):153-160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Montecino-Rodriguez E, Dorshkind K. New perspectives in B-1 B cell development and function. Trends Immunol. 2006;27(9):428-433. [DOI] [PubMed] [Google Scholar]