Abstract
Background:
Serum concentrations of antimullerian hormone (AMH) correlate with ovarian response during assisted reproduction treatment (ART) cycles.
Objective:
This retrospective study attempted to evaluate the selection of ovarian stimulation protocols based on serum AMH levels in patients and its impact on the results of ART.
Materials and Methods:
Based on AMH levels, the patients with tubal factor infertility were divided in three groups of normal, low and high AMH levels. Oocyte, good embryo number and pregnancy rate in each group were analyzed.
Results:
Using agonist and antagonist protocols, an increase in serum AMH led to higher number of oocytes and better quality embryos. At all low, normal and high AMH levels, the agonist protocol led to a more significant increase in the number of oocytes than the antagonist protocol (p<0.05). The number of high quality embryos significantly increased by the agonist protocol than antagonist protocol in women with normal AMH levels of 1.3-2.6 ng/ml (p=0.00). Moreover, the results for the number of high quality embryos at AMH ˃2.6 ng/ml was in favor of the antagonist protocol (p=0.00). The results showed the lowest pregnancy rate at AMH ˂1.3 ng/ml. At AMH ˃2.6 ng/ml, there was a significant increase in pregnancy rate through the antagonist protocol (p=0.04).
Conclusion:
Findings of this study suggested that the ART results are predictable, taking into account the AMH levels. The protocol specific to each patient can be used given the AMH level in each individual. This is because the results of each protocol depend on individual conditions.
Key Words: AMH, Ovarian stimulation protocols, Oocyte, Embryo, Pregnancy rate
Introduction
(COH) plays an important role in reproductive medicine. That is why the selection of an appropriate ovarian stimulation strategy can improve the results of assisted reproductive techniques. Although the two common ovarian stimulation protocols (GnRH agonist and GnRH antagonist) exhibit similar implantation and pregnancy rates, each protocol entails specific characteristics. In fact, the agonist protocol leads to higher number of oocytes per cycle, whereas the antagonist protocol curtails the gonadotropin dose, shortening the stimulation period and saving the treatment costs, which in turn leads to patient comfort (1-4).
Therefore, a suitable treatment strategy should be selected according to the ovarian response required for each person by taking the patient’s conditions into account. Anti-mullerian hormone (AMH) is a predictor of ovarian response used to select the agonist and antagonist protocols. This can maximize the success rate of assisted reproductive techniques, while enhancing the safety of ovarian stimulation practices (5). AMH is a subcategory of TGFβ produced by the granulosa cells of pre-antral and antral follicles (6). AMH can prevent the growth and recruitment of primordial follicles by reducing the sensitivity of follicles to FSH (7). The direct relationship between serum AMH levels and the number of antral follicles has been demonstrated (8).
Furthermore, the AMH levels do not fluctuate during the menstrual cycle (9, 10). AMH is considered as an indicator that can be employed as the best option during assisted reproductive treatment (11). There are very few studies on the application of AMH to predict the quality of oocytes and embryos (12). Based on the AMH level in each patient, the COH can improve the clinical pregnancy rate and minimize the harm associated with ovarian response (13). According to previous studies, different results have been obtained by using agonist and antagonist protocols in normal, low and high AMH levels (14). The COH may give rise to certain problems due to different ovarian responses to FSH (15).
This extensive study intended to compare the number of oocytes, quality of embryo and ultimately pregnancy rate through the agonist and antagonist protocols in patients with different AMH levels. There have been no studies so far comparing the results of both protocols in normal, low and high responders based on serum AMH levels.
Materials and methods
This retrospective study was conducted on 243 patients under ICSI treatment in the private assisted reproduction (Laleh Hospital) ICSI during 2012-2014.
Characteristics
The patients included women with tubal factor infertility at age less than 40 years, normal menstrual cycle, presence of two ovaries, with no history of ovarian surgery, chemotherapy, endocrine disorders and hormonal therapy. The preliminary analysis involved infertility details, medical history, past surgeries and obstetric examinations one month prior to the treatment. Furthermore, the trans-vaginal ultrasonography was performed to assess the local pathology and hormonal profile, including FSH, TSH and serum AMH levels in 2-3 days of menstrual cycle.
Ovarian stimulation protocols
According to clinical diagnosis, the patients underwent ovarian stimulation through GnRH agonist and antagonist protocols. The GnRH agonist protocol involved daily subcutaneous injection of 0.1 mg triptorelin acetat (decapeptyl, Ferring Pharmaceuticals, Netherlands) in the mid-luteal phase (day 20) and menstrual cycle before the stimulation. The ovarian stimulation was obtained from day 2 or 3 of the cycle by subcutaneous injection of recombinant human FSH (Gonal-F, Merk Serono, Germany) at a fixed dose of 150-225 IU and then continued by injection of hCG.
The ovarian response was monitored through serial ultrasonography and evaluation of serum estradiol levels. GnRH antagonist protocol performed by administration of gonadotropin rFSH on day 2 of the cycle along with GnRH antagonist cetrorelix (Cetrotide, ASTA Medica, Amsterdam, The Netherlands; 0.25 mg/d, s.c) on day 9 post-stimulation. In all patients, the ovulation stimulation was done with 5000-10000 units of hCG, leading to a follicle diameter of 19 mm and a desirable level of serum estradiol. The oocytes were retrieved through transvaginal ultrasound-guided technique 36 hours after administration of hCG.
The intracytoplasmic sperm injection was performed following the oocyte retrieval and oocytes denudation. The incidence of fertilization was assessed 19-21 hr after the injection in the presence of two pronuclei. The quality of embryo was recorded based on the number of blastomeres and percentage of fragmentation 42-44 hr after the injection, which was then comparatively studied. This study attempted to compare the number of high quality embryos characterized by (i) 4 or 5 blastomeres on the 2nd and minimum 7 blastomeres on the 3rd day after fertilization, (ii) absence of multi-nucleotide blastomeres, and (iii) fragmentation of less than 20% on 2nd and 3rd days after fertilization.
During the assessment of results, the patients were divided on the basis of serum AMH into three groups: 1) AMH ˂1.3 ng/ml, 2) AMH between 1.3-2.6 ng/ml and 3) AMH ˃2.6 ng/ml (14). This study exclusively compared the number of oocytes, high quality embryos and pregnancy rates using two different protocols at three AMH levels.
Statistical analysis
SPSS software (SPSS, version 21 for windows; SPSS Inc., Chicago. IL), ANOVA including Tukey internal test was used to compare quantitative variables between the groups and 2for qualitative variables. p˂0.05 was considered statistically significant.
Results
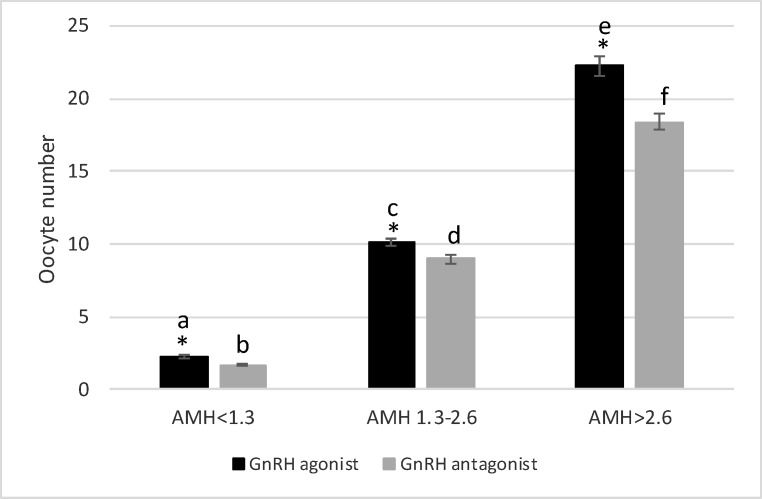
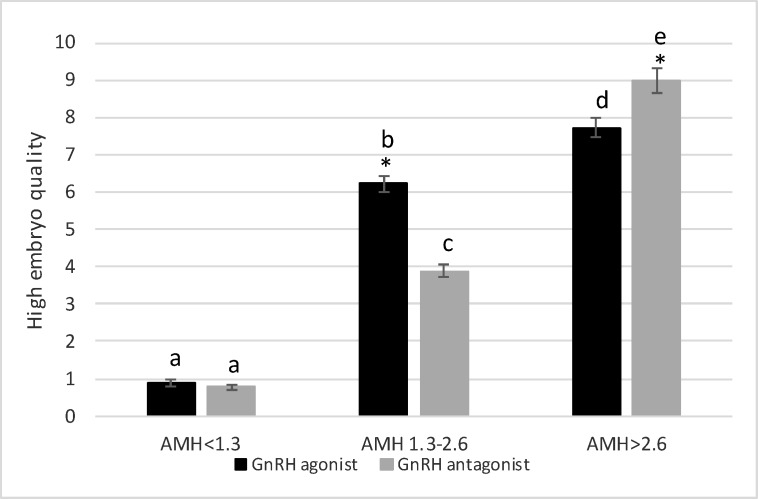
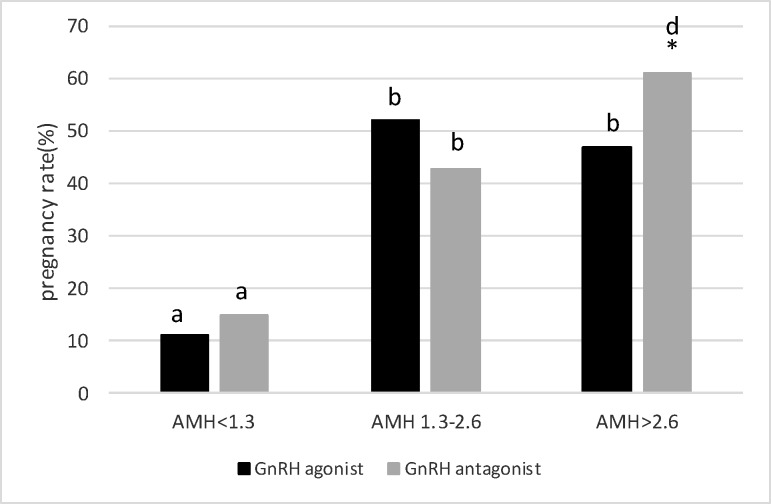
The data on age and hormonal profiles have been listed in table I, where there are no significant differences between the two protocols in these three groups. The results indicated that the number of oocytes was significantly higher when using the agonist protocol in women with AMH of 1.3-2.6 ng/ml compared to the antagonist protocol (10.16±0.27 vs. 9.01±0.3, p=0.006) (Figure 1). The number of high quality embryos showed a dramatic increase by the agonist protocol than the antagonist protocol (6.23±0.2 vs. 3.88±0.17, p=0.00) (Figure 2). Despite the difference in the two protocols in the numbers of oocytes and high quality embryos, the pregnancy rates did not show any significant difference. Nonetheless, the pregnancy rate was higher in the agonist protocol than the antagonist protocol (52% and 43%, respectively) (Figure 3).
Table I.
Mean (standard deviation) age, number of oocytes retrieved, embryo number, FSH, TSH levels in the GnRH-a long protocol vs. GnRH-ant protocol group
| Variables |
GnRH-a long protocol
(n = 123) |
GnRH-ant protocol
(n = 120) |
p-value | |
|---|---|---|---|---|
| Age | 36 ± 4.07 | 37 ± 3.6 | 0.11 | |
| Serum FSH (IU/L) | ||||
| AMH <1.3 (ng/ml) | 9.1 ± 0.45 (41) | 8.58 ± 0.64 (39) | 0.26 | |
| AMH 1.3-2.6 | 6.65 ± 0.9 (42) | 6.9 ± 0.61 (40) | 0.33 | |
| AMH>2.6 | 7.27 ± 0.71 (40) | 7.61 ± 0.82 (41) | 0.39 | |
| Serum TSH (IU/L) | ||||
| AMH<1.3 (ng/ml) | 2.66 ± 1.01 | 2.91 ± 0.98 | 0.79 | |
| AMH 1.3-2.6 | 2.2 ± 0.34 | 2.67 ± 0.56 | 0.10 | |
| AMH>2.6 | 2.83 ± 0.58 | 2.39 ± 0.43 | 0.68 | |
| BMI (kg/m2) | ||||
| AMH<1.3 (ng/ml) | 24 ± 1.5 | 23 ± 1.2 | 0.14 | |
| AMH 1.3-2.6 | 22 ± 1.1 | 22 ± 1.17 | 0.81 | |
| AMH>2.6 | 21 ± 1.2 | 22 ± 1.4 | 0.43 | |
FSH: follicle-stimulating hormone
TSH: thyroid-stimulating hormone
AMH: anti-mullerian hormone
BMI: body mass index
Figure 1.
Oocyte retrieval in three AMH levels with GnRH agonist protocol vs antagonist protocol. *shows statistical difference between two protocols (p˂0.05). The letter shows statistical difference within and between groups (p˂0.05
Figure 2.
High embryo quality in three AMH levels with GnRH agonist protocol vs antagonist protocol. *shows statistical difference between two protocols (p˂0.05). The letter shows statistical difference within and between groups (p˂0.05
Figure 3.
Pregnancy percentage in three AMH levels with GnRH agonist protocol vs antagonist protocol. *shows statistical difference between two protocols (p˂0.05). The letter shows statistical difference within and between groups (P˂ 0.05
In women with low AMH levels (˂1.3 ng/ml), the number of oocytes obtained by the agonist protocol was significantly higher than that by the antagonist protocol (2.23±0.11 vs. ˂2, p˂0.001) (Figure 1). Nevertheless, there was no significant difference between the agonist protocol (0.88±0.08) and the antagonist protocol (0.78±0.07) in the number of high quality embryos, p=0.34 (Figure 2). Although the number of oocytes and high quality embryos is in favor of the agonist protocol, the pregnancy rate in the antagonist protocol is higher than in the agonist protocol, indicating an insignificant difference (15% vs. 11%, respectively) (Figure 3).
In women with high ovarian response (AMH ˃2.6 ng/ml), there was a high number of oocytes obtained by agonist protocol, indicating a significant increase compared to antagonist protocol (22.26±0.64 vs. 18.41±0.55, p=0.00) (Figure 1). The comparison of the number of embryos showed different patterns. In fact, the number of high quality embryos was significantly higher when using the antagonist protocol, compared to the agonist protocol (9±0.32 vs. 7.71±0.26, p=0.002) (Figure 2). Similar to the number of high quality embryos, the pregnancy rate significantly increased when using the antagonist protocol, compared to the agonist protocol (61% vs. 47%, respectively) (Figure 3).
The comparison of AMH levels against the results of ovarian stimulation, quality of embryo and pregnancy rate demonstrated that an increase in AMH level significantly increased the number of oocytes and good quality embryos when using both protocols. In fact, the highest number of oocytes and high quality embryos in both protocols were observed at the highest AMH level, whereas, the lowest AMH level was associated with the lowest number of oocytes and embryos. The pregnancy rate patterns varied in different AMH levels in these two protocols. In fact, the highest pregnancy rate at normal AMH level was observed in agonist protocol, even though the difference was insignificant in comparison of high AMH level.
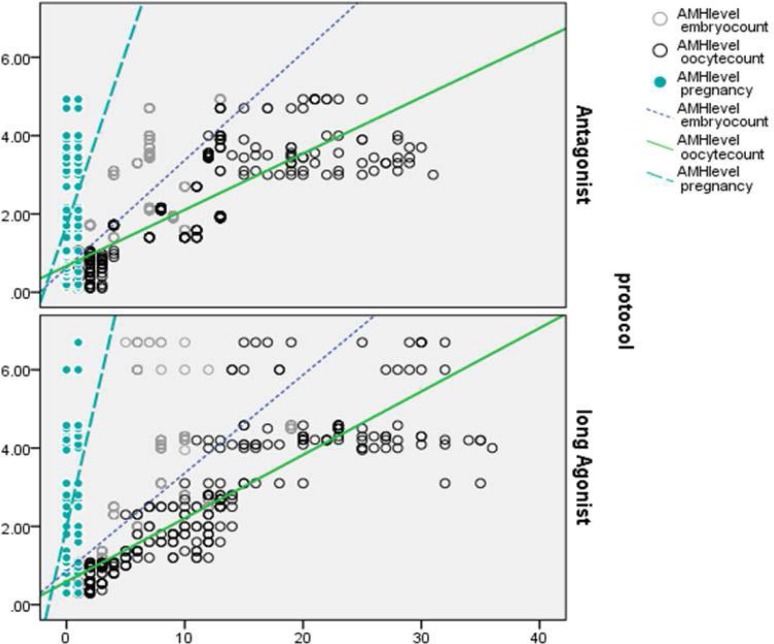
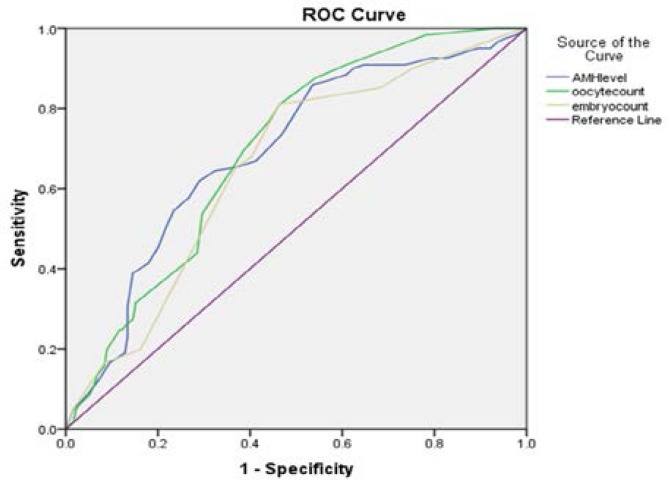
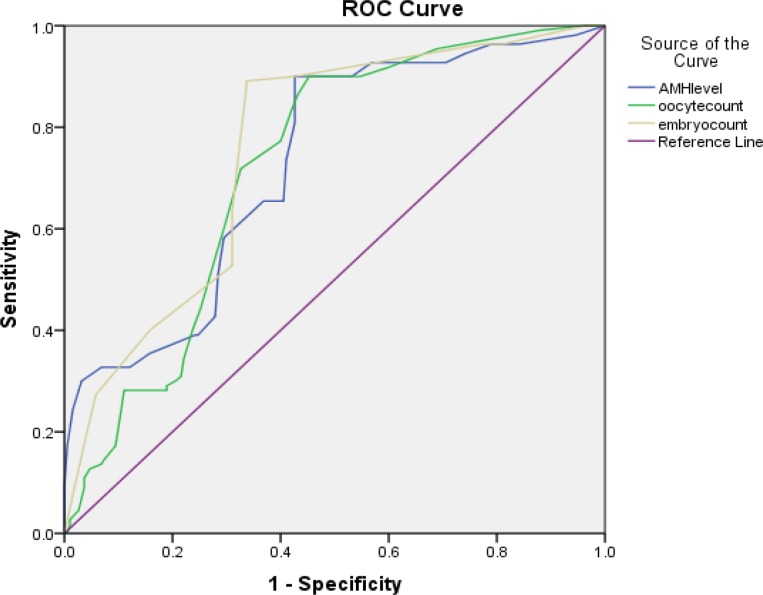
The comparison of the normal and high AMH level groups, however, showed a significant increase in comparison of the lowest AMH level. In the antagonist protocol, the highest pregnancy rate was observed at the highest AMH level, indicating a significantly higher increase than the other two groups of AMH (1.3-2.6 and ˂1.3). Moreover, the pregnancy rate at normal AMH level increased significantly higher than the lowest level (˂1.3 ng/ml). In total, the levels of serum AMH in all patients undergoing antagonist protocol stimulation showed a positive correlation with the number of retrieved oocytes (r=0.82, p<0.01), embryo count (r=0.66, p<0.01) and pregnancy (r=0.32, p<0.01) (Figure 4). Also, the levels of AMH in patients who received agonist protocol showed a positive correlation with the number of retrieved oocytes (r=0.86, p<0.01), embryo count (r=0.68, p<0.01) and pregnancy (r=0.36, p<0.01) (Figure 1). Furthermore, according to the stimulation protocols used, AMH also was an accurate marker of pregnancy in both agonist and antagonist protocol (AUC of 0.69 and 0.72, respectively) (Figure 5, 6).
Figure 4.
Correlations between AMH level, oocyte and embryo with positive pregnancy in both antagonist and agonist protocol.
Figure 5.
Comparison of predictive values for positive pregnancy using the ROC curve analysis in antagonist protocol
Figure 6.
Comparison of predictive values for positive pregnancy using the ROC curve analysis in agonist protocol
Discussion
In this study, the results of GnRH agonist and GnRH antagonist protocols were explored based on serum AMH levels. It was proven that an increase in serum AMH level led to a higher number of oocytes and embryos. These results were consistent with the findings of other studies. The serum AMH level can be adopted as a predictor of ovarian response in ART cycles (5, 16). Ovarian response is an important success factor in ART results. The two common protocols of ovarian stimulation are associated with different ovarian responses, each entailing a unique characteristic (17).
Just as demonstrated in this study, the use of two different protocols in women with low serum AMH levels led to different ovarian responses. In this regard, some studies preferred the GnRH agonist, whereas others the GnRH protocol for patients with low AMH levels or a history of weak ovarian response (18, 19).
According to the current study, although the GnRH agonist yielded a greater number of oocytes, it did not increase the number of high quality embryos compared to the GnRH antagonist. Moreover, the pregnancy rate was higher when the antagonist protocol was used. What ultimately matters in selecting the ovarian stimulation protocol is the outcomes. At first glance, the agonist protocol might indicate poorer results in this category of patients, but the outcomes of the antagonist protocol tended to be more satisfactory at the end. Similar studies which preferred the antagonist protocol in patients with low AMH levels indicate treatment costs decline and higher patients’ comfort, owing to a reduction in gonadotropin dose and canceled cycles. By the same token, a broader study has been conducted on ovarian stimulation protocol in women with low AMH levels. Despite more intense stimulation through the agonist protocol, there was no difference in the number of oocytes, canceled cycles and pregnancy rate through the antagonist protocol (20).
Similar to our findings, the previous study introduced the antagonist protocol as the front line of treatment for people with low AMH levels (21). Contrary to our findings, Malmusi et al, Sun and Zhu preferred the agonist protocol due to a higher number of oocytes and embryos in women with low AMH levels (22, 23). Regardless of the type of protocol, there was a very low pregnancy rate in this category of patients. This could be due to defects in granulosa cells, which produce the AMH as mentioned earlier. The defects in the supporting cells decreased the quality of oocytes, which in turn led to lower implantation and pregnancy in women with low AMH levels (24, 25).
According to our findings, the agonist protocol in women with normal AMH levels leads to a greater number of oocytes and embryos (2). Moreover, the pregnancy rate was higher than that in the antagonist protocol (26, 27). Some studies have suggested that the agonist protocol in patients with normal ovarian response is associated with higher number of oocytes, implantation and pregnancy rates, as well as an increase in live births and reduction in canceled cycles (28). A few studies prefer the agonist protocol over the antagonist protocol in normal respondents because of the negative impact of the latter on the performance of major endometrial proteins (29). In this regard, there have been other studies rejecting that statement on the grounds that endometrial receptivity in the antagonist protocol is similar to the natural endometrial cycle (30). According to our findings, however, the agonist protocol yielded more favorable results in women with normal AMH levels and ovarian responses.
The previous studies have suggested mixed results on high levels of serum AMH and selecting the type of treatment protocol (31). In the selection of agonist protocol for ovarian stimulation in patients with high AMH levels, Nelson et al and Manno et al reported an increase in pregnancy rate. Moreover, it was accompanied by an increase in the incidence of ovarian hyperstimulation syndrome (OHSS) and canceled cycles (17, 32). In contrast to these results, the molecular studies on the oocytes, obtained through the antagonist protocol in patients with high AMH levels, indicated that the gene expressions of ATPase and BMP15, as two indicators of oocyte quality, enhanced through the antagonist protocol (4). Therefore, the antagonist protocol can be the more desirable option in high AMH cases. In an analysis, La Marca and Sunkara argued that the antagonist protocol in patients with high ovarian responses curtails the risk of OHSS, because the use of antagonist protocol depends on the patient’s endocrine conditions (14). Hence, it may lead to lower number of follicles during ovarian stimulation. Moreover, it entails a shorter treatment period and higher pregnancy rate than the agonist protocol (33).
These results are consistent with our findings, where the number of high quality embryos and pregnancy rates significantly increased despite the lower number of oocytes obtained by the antagonist protocol. Therefore, the antagonist protocol is the more suitable option for patients with high serum AMH levels. According to a positive correlation of AMH levels with the number of retrieved oocytes, embryo count and pregnancy rate in both different protocols, AMH is an adequate predictor of ovarian response and ART outcomes. Further AMH-tailored protocol selection improved ART outcomes. These findings are similar to Hamdine et al findings (15).
This retrospective study generally demonstrated that the agonist protocol is the more suitable option for the normal range of serum AMH given the number of oocytes and high quality embryos leading to higher pregnancy rate. Since low AMH levels provide no prognosis for ovarian response and pregnancy rate, the selection of a good protocol can improve the pregnancy rate in patients who barely respond to treatment due to excessive ovarian stimulation.
Hence, the administration of high-dose gonadotropin and agonist protocol are not associated with improvement of outcomes. It is better to select the antagonist as the more ideal ovarian stimulation strategy for low AMH level given the higher pregnancy rate. Since the antagonist protocol in patients with high AMH levels leads to greater patient comfort and lower risks associated with ovarian hyper-stimulation and above all, yielding more desirable outcomes, it can be regarded as a safe technique for ovarian stimulation.
Conclusion
According to these results, it can be concluded that the ovarian stimulation protocol should be selected based on the patient’s’ specific characteristics. Moreover, the prediction of ovarian response and pregnancy rate can curtail the treatment costs and pave the way for successful assisted reproductive techniques.
Acknowledgements
This study was supported by Anatomy Department of Iran University of Medical Sciences and also IVF Center of Laleh Hospital. We would like to thank the people from IVF Laboratory in IVF Center of Laleh Hospital especially Ms. Alenabi.
Conflict of interest
There is no conflict of interest in this study.
References
- 1.Mochtar MH. The effect of an individualized GnRH antagonist protocol on folliculogenesis in IVF/ICSI. Hum Reprod. 2004;19:1713–1718. doi: 10.1093/humrep/deh334. [DOI] [PubMed] [Google Scholar]
- 2.Depalo R, Lorusso F, Palmisano M, Bassi E, Totaro I, Vacca M, et al. Follicular growth and oocyte maturation in GnRH agonist and antagonist protocols for in vitro fertilisation and embryo transfer. Gynecol Endocrinol. 2009;25:328–334. doi: 10.1080/09513590802617762. [DOI] [PubMed] [Google Scholar]
- 3.Greco E, Litwicka K, Ferrero S, Baroni E, Sapienza F, Rienzi L, et al. GnRH antagonists in ovarian stimulation for ICSI with oocyte restriction: a matched, controlled study. Reprod Biomed Online. 2007;14:572–578. doi: 10.1016/s1472-6483(10)61048-6. [DOI] [PubMed] [Google Scholar]
- 4.Hoseini FS, Noori Mugahi SM, Akbari-Asbagh F, Eftekhari-Yazdi P, Aflatoonian B, Aghaee-Bakhtiari SH, et al. A randomized controlled trial of gonadotropin releasing hormone agonist versus gonadotropin-releasing hormone antagonist in Iranian infertile couples: oocyte gene expression. Daru. 2014;22:67–76. doi: 10.1186/s40199-014-0067-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Aflatoonian A, Oskouian H, Ahmadi SH, Oskouian L. Prediction of high ovarian response to controlled ovarian hyperstimulation: anti-Müllerian hormone versus small antral follicle count (2-6 mm) J Assist Reprod Genet. 2009;26:319–325. doi: 10.1007/s10815-009-9319-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Durlinger AL, Visser JA, Themmen AP. Regulation of ovarian function: the role of anti-Müllerian hormone. Reproduction. 2002;124:601–609. doi: 10.1530/rep.0.1240601. [DOI] [PubMed] [Google Scholar]
- 7.Durlinger AL, Gruijters MJ, Kramer P, Karels B, Kumar TR, Matzuk MM, et al. Anti-Müllerian hormone attenuates the effects of FSH on follicle development in the mouse ovary. Endocrinology. 2001;142:4891–4899. doi: 10.1210/endo.142.11.8486. [DOI] [PubMed] [Google Scholar]
- 8.Gruijters MJ, Visser JA, Durlinger AL, Themmen AP. Anti-Müllerian hormone and its role in ovarian function. Mol Cell Endocrinol. 2003;211:85–90. doi: 10.1016/j.mce.2003.09.024. [DOI] [PubMed] [Google Scholar]
- 9.Cook CL, Siow Y, Taylor S, Fallat ME. Serum müllerian-inhibiting substance levels during normal menstrual cycles. Fertil Steril. 2000;73:859–861. doi: 10.1016/s0015-0282(99)00639-1. [DOI] [PubMed] [Google Scholar]
- 10.Hehenkamp WJ, Looman CW, Themmen AP, de Jong FH, Te Velde ER, Broekmans FJ. Anti-Müllerian hormone levels in the spontaneous menstrual cycle do not show substantial fluctuation. J Clin Endocrinol Metab. 2006;91:4057–4063. doi: 10.1210/jc.2006-0331. [DOI] [PubMed] [Google Scholar]
- 11.Nelson SM, Yates RW, Fleming R. Serum anti-Mu¨ llerian hormone and FSH: prediction of live birth and extremes of response in stimulated cycles-implications for individualization of therapy. Hum Reprod. 2007;22:2414–2421. doi: 10.1093/humrep/dem204. [DOI] [PubMed] [Google Scholar]
- 12.Majumder K, Gelbaya TA, Laing I, Nardo LG. The use of anti-Müllerian hormone and antral follicle count to predict the potential of oocytes and embryos. Eur J Obstet Gynecol Reprod Biol. 2010;150:166–170. doi: 10.1016/j.ejogrb.2010.02.029. [DOI] [PubMed] [Google Scholar]
- 13.Yates AP, Rustamov O, Roberts SA, Lim HY, Pemberton PW, Smith A, et al. Anti-Mullerian hormone-tailored stimulation protocols improve outcomes whilst reducing adverse effects and costs of IVF. Hum Reprod. 2011;26:2353–2362. doi: 10.1093/humrep/der182. [DOI] [PubMed] [Google Scholar]
- 14.La Marca A, Sunkara SK. Individualization of controlled ovarian stimulation in IVF using ovarian reserve markers: from theory to practice. Hum Reprod Update. 2014;20:124–140. doi: 10.1093/humupd/dmt037. [DOI] [PubMed] [Google Scholar]
- 15.Hamdine O, Eijkemans MJ, Lentjes EW, Torrance HL, Macklon NS, Fauser BC, et al. Ovarian response prediction in GnRH antagonist treatment for IVF using anti-Müllerian hormone. Hum Reprod. 2015;30:170–8. doi: 10.1093/humrep/deu266. [DOI] [PubMed] [Google Scholar]
- 16.Seifer DB, MacLaughlin DT, Christian BP, Feng B, Shelden RM. Early follicular serum müllerian-inhibiting substance levels are associated with ovarian response during assisted reproductive technology cycles. Fertil Steril. 2002;77:468–471. doi: 10.1016/s0015-0282(01)03201-0. [DOI] [PubMed] [Google Scholar]
- 17.Al-Inany H1, Aboulghar M. GnRH antagonist in assisted reproduction: a Cochrane review. Hum Reprod. 2002;17:874–885. doi: 10.1093/humrep/17.4.874. [DOI] [PubMed] [Google Scholar]
- 18.Nelson SM. Biomarkers of ovarian response: current and future applications. Fertil Steril. 2013;99:963–969. doi: 10.1016/j.fertnstert.2012.11.051. [DOI] [PubMed] [Google Scholar]
- 19.Marci R, Caserta D, Dolo V, Tatone C, Pavan A, Moscarini M. GnRH antagonist in IVF poor-responder patients: results of a randomized trial. Reprod Biomed Online. 2005;11:189–193. doi: 10.1016/s1472-6483(10)60957-1. [DOI] [PubMed] [Google Scholar]
- 20.Copperman AB, Benadiva C. Optimal usage of the GnRH antagonists: a review of the literature. Reprod Biol Endocrinol. 2013;11:20–33. doi: 10.1186/1477-7827-11-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pu D, Wu J, Liu J. Comparisons of GnRH antagonist versus GnRH agonist protocol in poor ovarian responders undergoing IVF. Hum Reprod. 2011;11:2742–2749. doi: 10.1093/humrep/der240. [DOI] [PubMed] [Google Scholar]
- 22.Malmusi S, La Marca A, Giulini S, Xella S, Tagliasacchi D, Marsella T, et al. Comparison of a gonadotropin-releasing hormone (GnRH) antagonist and GnRH agonist flare-up regimen in poor responders undergoing ovarian stimulation. Fertil Steril. 2005;84:402–406. doi: 10.1016/j.fertnstert.2005.01.139. [DOI] [PubMed] [Google Scholar]
- 23.Sun Y, Zhu YM. Gonadotropin-releasing hormone antagonist protocol in patients with risk of poor response to ovarian stimulation in IVF-ET. Zhejiang Da Xue Xue Bao Yi Xue Ban. 2009;38:305–310. doi: 10.3785/j.issn.1008-9292.2009.03.014. [DOI] [PubMed] [Google Scholar]
- 24.Ebner T, Sommergruber M, Moser M, Shebl O, Schreier-Lechner E, Tews G. Basal level of anti-Müllerian hormone is associated with oocyte quality in stimulated cycles. Hum Reprod. 2006;21:2022–2026. doi: 10.1093/humrep/del127. [DOI] [PubMed] [Google Scholar]
- 25.Taghavi SA, Ashrafi M, Mehdizadeh M, Karimian L, Joghataie MT, Aflatoonian R. Toll-like receptors expression in follicular cells of patients with poor ovarian response. Int J Fertil Steril. 2014;8:183–192. [PMC free article] [PubMed] [Google Scholar]
- 26.Yang S1, Chen XN, Qiao J, Liu P, Li R, Chen GA, et al. Comparison of GnRH antagonist fixed protocol and GnRH agonists long protocol in infertile patients with normal ovarian reserve function in their first in vitro fertilization-embryo transfer cycle. Zhonghua Fu Chan Ke Za Zhi. 2012;47:245–249. [PubMed] [Google Scholar]
- 27.Johnston-MacAnanny EB, DiLuigi AJ, Engmann LL, Maier DB, Benadiva CA, Nulsen JC. Selection of first in vitro fertilization cycle stimulation protocol for good prognosis patients: gonadotropin releasing hormone antagonist versus agonist protocols. J Reprod Med. 2011;56:12–16. [PubMed] [Google Scholar]
- 28.Grow D, Kawwass JF, Kulkarni AD, Durant T, Jamieson DJ, Macaluso M. GnRH agonist and GnRH antagonist protocols: comparison of outcomes among good-prognosis patients using national surveillance data. Reprod Biomed Online. 2014;29:299–304. doi: 10.1016/j.rbmo.2014.05.007. [DOI] [PubMed] [Google Scholar]
- 29.Meng Y1, Guo Y, Qian Y, Guo X, Gao L, Sha J, et al. Effects of GnRH antagonist on endometrial protein profiles in the window of implantation. Proteomics. 2014;14:2350–2359. doi: 10.1002/pmic.201400145. [DOI] [PubMed] [Google Scholar]
- 30.Haouzi D, Assou S, Dechanet C, Anahory T, Dechaud H, De Vos J, et al. Controlled ovarian hyperstimulation for in vitro fertilization alters endometrial receptivity in humans: protocol effects. Biol Reprod. 2010;82:679–686. doi: 10.1095/biolreprod.109.081299. [DOI] [PubMed] [Google Scholar]
- 31.Anckaert E, Smitz J, Schiettecatte J, Klein BM, Arce JC. The value of anti-Mullerian hormone measurement in the long GnRH agonist protocol: association with ovarian response and gonadotrophin-dose adjustments. Hum Reprod. 2012;27:1829–1839. doi: 10.1093/humrep/des101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Manno M, Cervi M, Zadro D, Fuggetta G, Adamo V, Tomei F. Different ART outcomes at increasing peak estradiol levels with long and antagonist protocols: retrospective insights from ten year experience. J Assist Reprod Genet. 2011;28:693–698. doi: 10.1007/s10815-011-9570-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lainas TG, Sfontouris IA, Zorzovilis IZ, Petsas GK, Lainas GT, Alexopoulou E. Flexible GnRH antagonist protocol versus GnRH agonist long protocol in patients with polycystic ovary syndrome treated for IVF: a prospective randomised controlled trial (RCT) Hum Reprod. 2010;25:683–689. doi: 10.1093/humrep/dep436. [DOI] [PubMed] [Google Scholar]