Abstract
Purpose
Persistent pain is a common side effect of breast cancer treatment. The present study aimed to assess the prevalence, associated treatment-related factors, and the type of pain (neuropathic or nociceptive) in patients who had undergone a unilateral mastectomy.
Methods
All women who underwent a unilateral mastectomy at a University Hospital between 2009 and 2013 were eligible for inclusion. Women with breast reconstruction or active cancer were excluded. Participants were mailed a questionnaire evaluating the prevalence, location, intensity, and frequency of surgical site pain. Additionally, the painDETECT®, a validated instrument to evaluate neuropathic pain, was mailed to all participants.
Results
A total of 305 women were included, and of them, 261 (85.6%) completed the study questionnaire. After a median follow-up period of 3.0 years, 100 women (38.3%) reported experiencing pain at the surgical site. Body mass index ≥30 kg/m2, radiation therapy, and axillary lymph node dissection were significantly associated with persistent pain in univariate models. However, only body mass index ≥30 kg/m2 was independently associated with persistent pain (odds ratio, 2.13; 95% confidence interval, 1.06–4.27; p=0.034) in a multivariate analysis. Of the patients reporting pain, 71.0% were unlikely to have a neuropathic pain component. A moderate, but highly significant, positive correlation was observed between the pain intensity and the painDETECT® score (rs=0.47, p<0.001).
Conclusion
Persistent pain after breast cancer treatment continues to have a high prevalence. Our results indicate that the largest proportion of patients experiencing persistent pain after breast cancer treatment do not have a clear neuropathic pain component.
Keywords: Breast neoplasms, Chronic pain, Mastectomy, Neuralgia
INTRODUCTION
Persistent pain after breast cancer treatment (PPBCT) is a common side effect to breast cancer treatment, with a reported prevalence of 24% to 47% [1,2,3,4,5]. With the increased breast cancer survival seen in the recent years, late sequelae to the treatment, such as persistent pain, have become increasingly important to recognize in order to, if possible, administer relevant treatment [6]. Persistent pain may be severely debilitating for the affected patients as it often causes considerable morbidity in terms of declined physical and emotional well-being [4,7,8,9,10,11]. Studies have shown that patients with PPBCT are more likely to report anxiety and depressive symptoms and have a higher level of perceived stress when compared to patients without persistent pain [4]. Additionally, PPBCT has been identified as the most important predictor of a poor health-related quality of life after breast cancer surgery [12]. Even though PPBCT is well described in the literature, the exact mechanism of its development remains uncertain and is likely multifactorial [13]. The persistent pain may result from damage to the nerve fibers during the surgery, or from the adjuvant radiation- or chemotherapy, suggesting a predominantly neuropathic pain character [14,15]; however, evidence for this is currently lacking and few studies have investigated the prevalence of neuropathic pain in this patient group [13,16,17]. Although several studies have investigated the risk factors associated with developing PPBCT [1,2,3,4,5], most studies published to date have investigated populations consisting of both breast-conserving surgery (BCS) and mastectomized patients, which may give rise to multicollinearity in the analysis of risk factors, due to the inherent correlation between type of breast surgery, tumor related variables, and the adjuvant therapy administered.
The primary purpose of our study was to assess the prevalence and associated treatment-related factors for PPBCT in a population consisting exclusively of patients who underwent unilateral mastectomy. We further aimed to investigate the type of pain experienced by the patients, hypothesizing that most patients reporting pain after a mastectomy would have a clear neuropathic pain component.
METHODS
Patients
All women who underwent a unilateral mastectomy at Aarhus University Hospital, Aarhus, Denmark, between 2009 and 2013, and followed the national treatment guidelines, were eligible to participate in the study. Exclusion criteria were as follows: an active breast cancer recurrence, active cancer elsewhere, emigration, death, or breast reconstruction. The breast cancer tumor and the administered treatment data were retrieved from the Danish Breast Cancer Cooperative Group (DBCG) database. Data on the patients' current cancer status, as well as any breast reconstructive procedure performed, were retrieved from the electronic patient files.
Definition of the pain
We used the International Association for the Study of Pain definition of chronic pain, which proposes a 3 months duration as a cutoff, but does not consider the intensity or frequency of the pain [18]. The presence of pain was assessed in five areas: the excised breast area, the mastectomy scar, the ipsilateral thorax, the axilla, and/or the medial upper arm. All patients who reported experiencing persistent pain after mastectomy and reporting a nonzero pain intensity in any of the five aforementioned areas were classified as having PPBCT.
Study questionnaire
A study specific questionnaire was developed and was used to ask the patients whether they were experiencing persistent pain after their mastectomy. If the patient reported experiencing persistent pain, the presence or absence of pain in each of the five assessed areas was evaluated. If pain was present in one/more of the specified areas, the patient was asked to evaluate the intensity of the pain on a numerical rating scale (NRS) of 0 to 10. We chose the NRS as it is recommended by the Initiative on Methods, Measurement, and Pain Assessment in Clinical Trials. Lastly, the pain frequency was evaluated on a three step rating scale with the possible answers being: <1 day per week, 1 to 3 days per week, or >3 days per week. In addition to the study specific questionnaire, the painDETECT® questionnaire (PDQ) was mailed to the enrolled patients [19]. The PDQ is a self-report questionnaire comprised of nine items, and does not require an additional bedside clinical examination. The PDQ was validated in a multicenter study and exhibited an 85% sensitivity, 80% specificity, and an 83% ability to correctly classify patients' pain as neuropathic or nociceptive [19]. The PDQ has been officially translated into several languages, including Danish, as was used in the present study. A summated score of the nine questionnaire items is calculated, and ranges between 0 and 38, with higher scores indicating a higher probability of neuropathic pain. According to the PDQ manual, a score of 0 to 12 indicates nociceptive pain (a neuropathic pain component is unlikely, <15%), a score of 13 to 18 is unclear (ambiguous, a neuropathic pain component may be present), and a score of 19 to 38 indicates a likely neuropathic pain (>90%). Only patients reporting pain in one/more of the five assessed areas completed the PDQ. The questionnaire package was mailed to the enrolled patients at the beginning of October 2014, and a reminder was sent a month later to the nonresponders.
Treatment regimen
All women in the study were treated in accordance with the DBCG treatment guidelines that are described in detail elsewhere [20].
Statistical considerations
Subgroup analysis was performed using chi-square tests for categorical variables and Student t-test for normally distributed continuous variables. The Spearman rank correlation coefficient was used to evaluate the association between the intensity of the pain and the PDQ score. Univariate logistic regression analysis was performed to test the association between PPBCT and treatment-, patient-, and tumor-related variables. Adjusted odds ratios (OR) were calculated in a multivariate logistic regression model, adjusting for variables identified to be significantly associated with PPBCT in the univariate logistic regression analysis (body mass index [BMI], radiotherapy, and axillary dissection procedure). Multicollinearity was evaluated by calculating the variance inflation factors. Data are presented as OR, 95% confidence interval, and p-values, with p-values <0.05 considered statistically significant. Missing values were coded as missing and were omitted from the analysis. The design, verification, and digitalization of the questionnaire data were performed using the Teleform™ software package version 10.5.1 (Cardiff Software, Vista, USA). All statistical analyses were performed using the STATA® software IC 13 (Stata Corp., College Station, USA).
Ethical considerations
The study was performed in accordance with the Declaration of Helsinki and was approved by the Regional Ethics Committee (case number: 1-10-72-76-13), the Danish Data Protection Agency (case number: 1-16-02-214-14), and The National Board of Health (case number: 3-3013-590/1). The enrolled patients did not receive any compensation or incentives for participation.
RESULTS
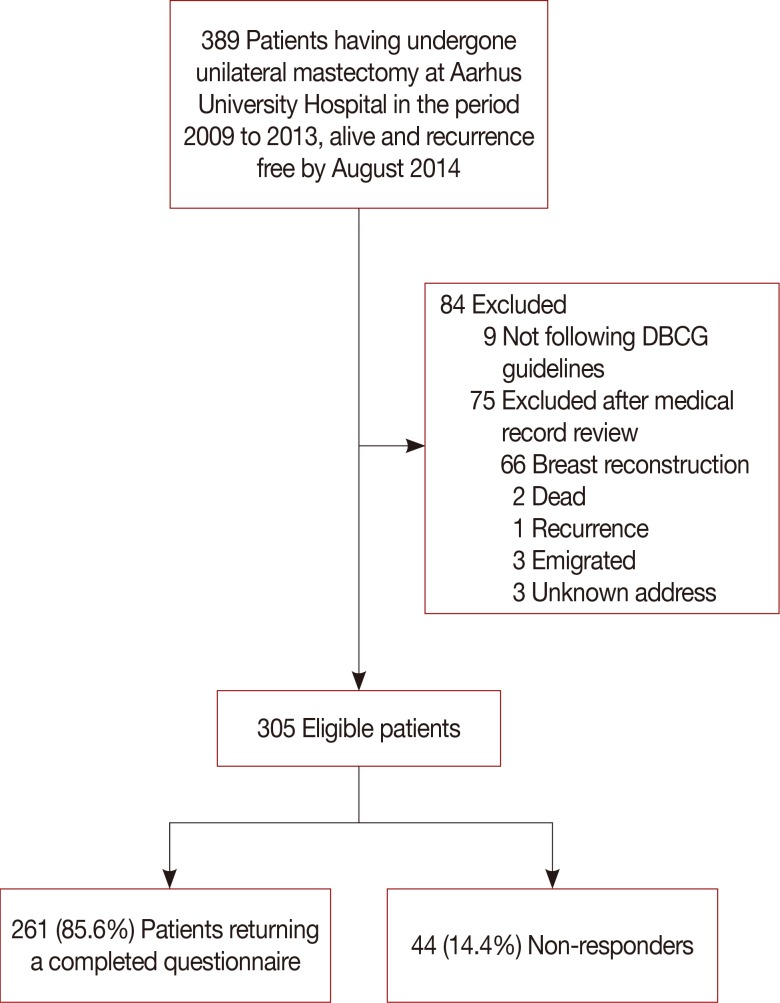
Data from 389 patients, who had undergone a unilateral mastectomy, and were alive and free of recurrence by August 2014, was retrieved from the DBCG database. Exclusion criteria were assessed by reviewing the electronic patient records. Patients who were not treated in accordance with the national DBCG treatment guidelines, had received a breast reconstruction, had emigrated, had died, or had been diagnosed with a recurrence between the time of data extraction and review of electronic records at the end of September 2014, were excluded. In total, 305 patients were eligible for enrolment and were mailed the study questionnaire. Of them, 261 patients (85.6%) returned a completed questionnaire and were included in the study. A flowchart of the inclusion process is depicted in Figure 1, and detailed patient demographics are shown in Table 1. No statistically significant differences were observed between the responders and nonresponders to the mailed questionnaire with respect to age, size, and location of the tumor, tumor-positive lymph nodes, or having undergone an axillary lymph node dissection (ALND). The median follow-up time since the mastectomy was 3.0 years (range, 0.8–5.8 years) and the mean age at the time of the breast cancer surgery was 63.6±11.3 years.
Figure 1. Flowchart of the patient inclusion process.
DBCG=Danish Breast Cancer Cooperative Group.
Table 1. Demographics of participating patients.
| Variable | All patients (n=261) No. (%) |
With PPBCT (n=100) No. (%) |
Without PPBCT (n=161) No. (%) |
| Age (yr)* | 63.6 ± 11.3 | 61.3 ± 11.1 | 65.0 ± 11.2 |
| BMI (kg/m2)* | 25.4 ± 5.26 | 26.6 ± 6.2 | 24.7 ± 4.5 |
| Tumor side† | |||
| Left breast | 119 (46.0) | 48 (48.5) | 71 (44.4) |
| Right breast | 140 (54.1) | 51 (51.5) | 89 (55.6) |
| Tumor size > 20 mm‡ | 141 (54.2) | 59 (59) | 82 (51.3) |
| Tumor in upper lateral quadrant‡ | 62 (23.9) | 24 (24) | 38 (23.8) |
| Positive lymph nodes‡ | 149 (57.3) | 65 (65) | 84 (52.5) |
| Chemotherapy‡ | |||
| Yes | 96 (36.9) | 44 (44) | 52 (32.5) |
| No | 164 (63.1) | 56 (56) | 108 (67.5) |
| Radiation therapy‡ | |||
| Yes | 112 (56.9) | 52 (52) | 60 (37.5) |
| No | 148 (43.1) | 48 (48) | 100 (62.5) |
| Endocrine treatment§ | |||
| Yes | 181 (79.4) | 70 (78.7) | 111 (79.9) |
| No | 47 (20.6) | 19 (21.4) | 28 (20.1) |
PPBCT=persistent pain after breast cancer treatment; BMI=body mass index.
*Mean±SD; †Missing data fo r n=2 (0.8%); ‡Missing data for n=1 (0.4%); §Missing data for n=33 (12.6%).
The prevalence and location of the pain
Of the 261 patients who responded to the questionnaire, 100 (38.3%) reported pain in at least one of the five assessed areas. The patients most frequently reported pain in the axilla (n=64, 24.5%), followed by the excised breast area (n=53, 20.3%), the medial arm (n=49, 18.8%), ipsilateral thorax (n=44, 16.9%), and the mastectomy scar (n=44, 16.9%). Of the women reporting pain, 26% reported pain in only one of the assessed areas, 74% reported pain in more than one area, 45% reported pain in more than two areas, 24% reported pain in more than three areas, and 11% reported pain in all the five assessed areas (Table 2).
Table 2. Distribution and frequency of pain.
| Investigated area | No. of women reporting pain (%) | |||
|---|---|---|---|---|
| Mild | Moderate | Severe | Total | |
| In the area of the missing breast | ||||
| Every day or almost every day | 5 (1.9) | 12 (4.6) | 8 (3.1) | 25 (9.6) |
| 1–3 Times weekly | 4 (1.5) | 10 (3.8) | 0000 | 14 (5.4) |
| Less than once a week | 11 (4.2) | 3 (1.1) | 0000 | 14 (5.4) |
| All frequencies | 20 (7.7) | 25 (9.6) | 8 (3.1) | 53 (20.3) |
| In the axilla | ||||
| Every day or almost every day | 9 (3.4) | 16 (6.1) | 7 (2.7) | 32 (11.3) |
| 1–3 Times weekly | 10 (3.8) | 7 (2.7) | 1 (0.4) | 18 (6.9) |
| Less than once a week | 7 (2.7) | 7 (2.7) | 0000 | 14 (5.4) |
| All frequencies | 26 (10.0) | 30 (11.5) | 8 (3.1) | 64 (24.5) |
| Side of the thorax | ||||
| Every day or almost every day | 10 (3.8) | 11 (4.2) | 4 (1.5) | 25 (9.6) |
| 1–3 Times weekly | 1 (0.4) | 8 (3.1) | 0000 | 9 (3.4) |
| Less than once a week | 6 (2.3) | 4 (1.5) | 0000 | 10 (3.8) |
| All frequencies | 17 (6.5) | 23 (8.8) | 4 (1.5) | 44 (16.9) |
| In the mastectomy scar | ||||
| Every day or almost every day | 4 (1.5) | 10 (3.8) | 5 (1.9) | 19 (7.3) |
| 1–3 Times weekly | 2 (0.8) | 7 (2.7) | 0000 | 9 (3.4) |
| Less than once a week | 10 (3.8) | 6 (2.3) | 0000 | 16 (6.1) |
| All frequencies | 16 (6.1) | 23 (8.8) | 5 (1.9) | 44 (16.9) |
| In the arm | ||||
| Every day or almost every day | 8 (3.1) | 13 (5.0) | 6 (2.3) | 27 (10.3) |
| 1–3 Times weekly | 7 (2.7) | 6 (2.3) | 1 (0.4) | 14 (5.4) |
| Less than once a week | 3 (1.1) | 4 (1.5) | 1 (0.4) | 8 (3.1) |
| All frequencies | 18 (6.9) | 23 (8.8) | 8 (3.1) | 49 (18.8) |
Patients scored their pain on a 0 to 10 numerical rating scale ranging from 0 (no pain) to 10 (worst imaginable pain). NRS scores of 1 to 3, mild pain; 4 to 7, moderate pain; and 8 to 10, severe pain. A total of 100 patients reported pain. Percentages are based on the total investigated population. Patients may report pain in more than one area.
Intensity and frequency of the pain
The average pain intensity was 4.7±2.3 on the NRS for patients reporting persistent pain. Of the patients reporting pain, 34.0% reported a mild pain (NRS, 1–3), 50.0% reported a moderate pain (NRS, 4–7), while 16.0% reported experiencing severe pain (NRS, 8–10) (Table 2). Of all patients reporting persistent pain, 58.0% reported to be experiencing pain every day or nearly every day, 20.0% reported experiencing pain 1 to 3 times a week, and 22.0% reported experiencing pain less than once a week. A moderate, but highly significant, positive correlation between the pain intensity and the pain frequency was observed (rs=0.37, p<0.001).
painDETECT®
The mean PDQ scale score, for the patients who reported to be experiencing persistent pain, was 10.2±6.7. Of the 100 patients who reported experiencing pain, 13.0% (n=13, 5.0% of the total cohort) scored high enough on the PDQ to indicate a “most likely” neuropathic pain, 16.0% (n=16) scored in the “unclear/ambiguous” range, while 71.0% (n=71) scored low on the scale to indicate an “unlikely” neuropathic pain (nociceptive pain). A moderate, but highly significant, positive correlation between reporting higher pain intensity and having a higher score on the PDQ was observed (rs=0.47, p<0.001).
Factors associated with persistent pain
In univariate analysis, a BMI ≥30 kg/m2, receiving radiation therapy, and receiving an ALND procedure were all significantly associated with PPBCT (Table 3). Additionally, receiving chemotherapy was associated with persistent pain, with a borderline statistical significance (p=0.057). In the subsequent multivariate analysis, only a BMI ≥30 kg/m2 demonstrated a statistically significant association with PPBCT (Table 3).
Table 3. Factors associated with persistent pain after breast cancer treatment.
| Variable | Without PPBCT (n=161) No. (%) |
With PPBCT (n=100) No. (%) |
Unadjusted | Adjusted* | ||
|---|---|---|---|---|---|---|
| OR (95% CI) | p-value | OR (95% CI) | p-value | |||
| Age (yr) | ||||||
| < 60 | 110 (42.1) | 59 (22.6) | 1.50 (0.89–2.52) | 0.126 | - | |
| ≥ 60 | 51 (19.5) | 41 (15.7) | 1 (Reference) | - | - | |
| BMI (kg/m2) | ||||||
| < 18.5 | 13 (5.0) | 5 (1.9) | 0.81 (0.27–2.44) | 0.707 | 0.86 (0.28–2.65) | 0.798 |
| 18.5–24.9 | 80 (30.7) | 38 (14.6) | 1 (Reference) | - | 1 (Reference) | - |
| 25–29.9 | 44 (16.9) | 33 (12.6) | 1.58 (0.87–2.86) | 0.132 | 1.69 (0.92–3.10) | 0.090 |
| ≥ 30 | 22 (8.4) | 23 (8.8) | 2.20 (1.09–4.43) | 0.027 | 2.13 (1.06–4.27) | 0.034 |
| Dominant hand same as side of surgery | ||||||
| Yes | 93 (35.6) | 48 (18.4) | 0.67 (0.41–1.11) | 0.125 | - | |
| No | 68 (26.1) | 52 (19.9) | 1 (Reference) | - | - | |
| Radiotherapy | ||||||
| Yes | 60 (23.0) | 52 (19.9) | 1.82 (1.10–3.02) | 0.020 | 1.21 (0.87–1.68) | 0.254 |
| No | 101 (38.7) | 48 (18.4) | 1 (Reference) | - | 1 (Reference) | - |
| Chemotherapy | ||||||
| Yes | 52 (19.9) | 44 (16.9) | 1.65 (0.98–2.76) | 0.057 | - | |
| No | 109 (41.8) | 56 (21.5) | 1 (Reference) | - | - | |
| Endocrine treatment† | ||||||
| Yes | 112 (48.9) | 70 (30.6) | 0.92 (0.48–1.77) | 0.806 | - | |
| No | 28 (12.2) | 19 (8.3) | 1 (Reference) | - | - | |
| Axillary procedure‡ | ||||||
| ALND§ | 88 (33.7) | 69 (26.4) | 1.80 (1.06–3.04) | 0.029 | 1.46 (0.74–2.88) | 0.280 |
| SN | 71 (27.2) | 31 (11.9) | 1 (Reference) | - | 1 (Reference) | - |
| Location of tumor | ||||||
| Upper lateral quadrant | 39 (14.9) | 24 (9.2) | 0.99 (0.55–1.77) | 0.967 | - | |
| All other locations | 122 (46.7) | 76 (29.1) | 1 (Reference) | - | - | |
| Tumor sideII | ||||||
| Right | 90 (34.5) | 53 (20.3) | 0.88 (0.53–1.45) | 0.608 | - | |
| Left | 70 (26.8) | 47 (18.0) | 1 (Reference) | - | - | |
| Tumor size (mm) | ||||||
| > 20 | 83 (31.8) | 59 (22.6) | 1.35 (0.82–2.24) | 0.241 | - | |
| ≤ 20 | 78 (29.9) | 41 (15.7) | 1 (Reference) | - | - | |
PPBCT=persistent pain after breast cancer treatment; OR=odds ratio; CI=confidence interval; BMI=body mass index; ALND=axillary lymph node dissection; SN=sentinel node.
*Adjusted for BMI, radiotherapy and axillary procedure; †Data on 229 patients (12.3% missing data); ‡Data on 259 patients (0.8% missing data); §Axillary lymph node dissection was defined as 10 or more excised lymph nodes; ∥Data on 260 patients (0.4% missing data).
Dysesthesia, phantom breast pain, and pain before surgery
One hundred forty-seven (56.3%) enrolled patients reported experiencing cutaneous dysesthesia at the surgical site. A statistically significant association between dysesthesia and experiencing PPBCT was observed (p<0.001). A total of 42 patients (16.1%) reported experiencing phantom breast sensations, and 13 patients (5.0%) reported phantom breast pain. Having phantom breast sensations was significantly associated with reporting PPBCT (p=0.005). A total of 25 patients (9.3%) reported to have experienced preoperative breast pain; however, this was not significantly associated with PPBCT (p=0.058).
DISCUSSION
In the present study, we investigated the prevalence of PPBCT in 261 women who had undergone a unilateral mastectomy at our institution between 2009 and 2013. With a median follow-up period of 3.0 years, 38.3% of the participating women reported surgical site pain. In a multiple regression analysis, having a BMI ≥30 kg/m2 was the only independent risk factor for developing PPBCT. Of all patients reporting a persistent pain, neuropathic pain was indicated in only 13.0%, based on the PDQ scores.
The prevalence of PPBCT in the present study corresponds well with the 24% to 47% prevalence rate reported in the literature [2,3,4,5]. The large range of the reported prevalence is likely caused by a lack of consistency in the definition of PPBCT used in these studies, especially in terms of the requisite intensity and frequency of the pain. A few studies have used a cutoff value of 3 or 4 on a 0–10 NRS, as this was deemed as clinically relevant pain, whereas other studies have used any nonzero pain value to denote persistent pain [4]. However, most studies use pain persisting for more than three months after surgery as a cutoff. In the present study, having a BMI ≥30 kg/m2 was the only variable independently associated with PPBCT. There is no clear consensus in the literature regarding overweight as a risk factor for PPBCT. In concordance with our results, Smith et al. [21] also observed a correlation between high BMI and persistent pain, while Macdonald et al. [8] reported that a higher body weight, but not a higher BMI, was associated with persistent pain. However, some other studies reported a lack of association between an increased BMI and a higher risk of developing persistent pain [9,11,22]. An association between high BMI and PPBCT may possibly be explained by the more extensive surgery required for overweight women, both in terms of the often larger breasts, and a more difficult axillary dissection. Several previous studies have concluded that ALND is associated with developing persistent pain after breast cancer surgery [3,5,16]. In the present study, in an unadjusted analysis, ALND was significantly associated with persistent pain (p=0.029); however, this association lost statistical significance in an adjusted multivariate analysis (p=0.386). The lack of association may be due to the relatively small sample size of the present study.
The mastectomy itself, as well as the ALND/sentinel node biopsy, may cause damage to the intercostobrachial nerve (ICBN) and the thoracic intercostal nerves. This has given rise to the assumption that the PPBCT is mostly neuropathic in nature [14]. Furthermore, studies often report that some patients describe the pain as burning, shooting, or stabbing, a description consistent with neuropathic pain [8,14,23]. Contrary to these reports, in our study, most women with PPBCT did not have a neuropathic pain component. Only 13.0% of the women experiencing persistent pain exhibited a clear neuropathic pain component, although a neuropathic pain component could not be excluded in another 16.0% of the enrolled women. Thus, the prevalence of neuropathic pain in our study is lower than the 40% prevalence reported by Bruce et al. [16], and 32% prevalence reported by Andersen et al. [17]. This lower prevalence of neuropathic pain may be explained by the difference in the studied population–our study population comprised solely of mastectomized patients, whereas the population studied by Bruce et al. and Andersen et al. consisted predominantly of BCS patients. Furthermore, the length of the follow-up period in our study (mean follow-up period, 3.0 years) was considerably longer than the follow-up period in the studies by Bruce et al. (0.75 years) and Andersen et al. (1.0 year). The longer follow-up period in our study may have increased the risk of a recall bias, which may also have affected the observed neuropathic pain prevalence.
In the present study, we observed a highly significant positive correlation between higher pain intensity and the neuropathic pain score, which is consistent with a previous study by Langford et al. [13]. Additionally, neuropathic pain in the acute postoperative period has been found to be predictive of experiencing pain a year after the surgery [17]. The association between developing neuropathic pain after a mastectomy and reporting a higher pain intensity may have considerable clinical relevance in choosing the optimal treatment regimen for individual patients. The low prevalence of neuropathic pain in our study population prohibited further statistical investigation to identify risk factors for developing neuropathic pain after mastectomy; future studies delving in this area would be interesting and relevant.
A few previous studies have used quantitative sensory testing to evaluate the pain components of PPBCT. Vilholm et al. [24] observed that patients with PPBCT had a higher thermal detection threshold, higher frequency of cold allodynia, and a greater temporal pain summation evoked by repetitive pinprick compared to pain-free patients. Cold allodynia refers to the pain experienced due to mild temperatures that would normally not evoke a pain response in healthy individuals, and temporal pain summation refers to a condition where the patient demonstrates an increase in the perceived pain intensity with repeated stimulation. Furthermore, Gottrup et al. [25] reported that patients with PPBCT experienced a higher frequency of temporal pain summation on the operated side compared to patients without PPBCT. Both studies thus indicate a role of neuropathic pain in the mechanism of developing PPBCT. However, none of the studies evaluated the prevalence of neuropathic symptoms. Moreover, women with PPBCT often have scar tissue with adhesion to the underlying tissue [26], and this type of pain is supposedly nociceptive and not neuropathic. The association between PPBCT and more extensive axillary procedures may also be explained in part by the increased scarring from a more extensive procedure, and may not necessarily be due to more damage to the ICBN. Our observation that neuropathic pain plays a minor role in PPBCT is at least to some extent supported by previous studies investigating intentional sparing of the ICBN, that report divergent and inconclusive results [17,27,28,29]. Additionally, Langford et al. [30] have recently concluded that even though patients with severe persistent breast pain exhibit neuropathic pain symptoms, the pattern of response to the pain quality assessment scale corresponds well to the pattern expected from patients with nonneuropathic pain conditions. Our findings are thus in accordance with the observations of Langford et al. [30], and further indicate that the pain mechanism for PPBCT patients may be of several different etiologies, and may thus require different approaches for optimal treatment.
In the present study, younger age was not associated with PPBCT. This is in contrast to most available literature where younger age is one of the most consistent factors associated with the development of PPBCT [3,5,7,8,11,16]. However, not all studies report a correlation [10]. Several explanations for this association have been proposed, including that younger women have increased anxiety and a lower tolerance for pain [21], and that younger women are frequently offered more aggressive adjuvant treatment for breast cancer. Radiation therapy was significantly associated with PPBCT in the univariate regression model but did not reach statistical significance in the adjusted model. In this aspect, our results do not concur with relevant literature that identifies radiation therapy as an independent risk factor for PPBCT [3]. The lack of significance in the multivariate model may be an effect of the lower power. Chemotherapy, as a risk factor for PPBCT, achieved only borderline statistical significance (p=0.057) in the univariate regression model. This is similar to the results of a previous nationwide study in Denmark, wherein Gärtner et al. [3] showed that chemotherapy was not independently associated with the risk of developing PPBCT.
In our study, 8.9% of the patients reported experiencing preoperative breast pain, with a borderline significant association between experiencing preoperative pain and reporting PPBCT (p=0.058). Preoperative pain has previously been investigated as a risk factor for PPBCT, but with contradictory results [17,30]. Furthermore, higher acute postoperative pain scores have also been associated with the development of PPBCT [9,11]. These results highlight the need for future prospective studies with longer follow-up period, especially as the acute postoperative pain is, to some extent, a modifiable risk factor.
Some limitations to the current study must be acknowledged. First, the cross-sectional nature of our study does not allow any inferences of causality. Second, even though the PDQ has shown a high specificity/sensitivity for detecting neuropathic pain, the gold standard for establishing a neuropathic pain diagnosis is a clinical examination. A higher sensitivity would thus require physical examinations to be conducted by appropriately trained personnel, which was beyond the scope of this study, but nevertheless relevant for future studies. The high response rate adds strength to our study, although, the nonresponders may still induce a bias. Additionally, recall bias may be present in the data regarding the experience of preoperative pain. Furthermore, the results from our adjusted analysis of factors associated with PPBCT indicate that our study may not be sufficiently powered to evaluate independently associated factors.
In conclusion, the results of our study show that PPBCT has a high prevalence after mastectomy. A BMI ≥30 kg/m2, receiving radiotherapy, and receiving ALND were all associated with the development of PPBCT; however, a BMI ≥30 kg/m2 was the only factor independently associated with PPBCT. Furthermore, our results reveal that most women with persistent pain after mastectomy do not have a clear neuropathic pain component, indicating that PPBCT may be caused by several different mechanisms, and may thus need different strategies for optimal pain control.
ACKNOWLEDGMENTS
The authors wish to express their gratitude to the participating patients.
Footnotes
This study was funded by grants from the Novo Nordisk Foundation (NNF13OC0007477) and The Graduate School of Health, Aarhus University, Aarhus, Denmark.
CONFLICT OF INTEREST: The authors declare that they have no conflict of interests.
References
- 1.Andersen KG, Kehlet H. Persistent pain after breast cancer treatment: a critical review of risk factors and strategies for prevention. J Pain. 2011;12:725–746. doi: 10.1016/j.jpain.2010.12.005. [DOI] [PubMed] [Google Scholar]
- 2.Mejdahl MK, Andersen KG, Gärtner R, Kroman N, Kehlet H. Persistent pain and sensory disturbances after treatment for breast cancer: six year nationwide follow-up study. BMJ. 2013;346:f1865. doi: 10.1136/bmj.f1865. [DOI] [PubMed] [Google Scholar]
- 3.Gärtner R, Jensen MB, Nielsen J, Ewertz M, Kroman N, Kehlet H. Prevalence of and factors associated with persistent pain following breast cancer surgery. JAMA. 2009;302:1985–1992. doi: 10.1001/jama.2009.1568. [DOI] [PubMed] [Google Scholar]
- 4.Belfer I, Schreiber KL, Shaffer JR, Shnol H, Blaney K, Morando A, et al. Persistent postmastectomy pain in breast cancer survivors: analysis of clinical, demographic, and psychosocial factors. J Pain. 2013;14:1185–1195. doi: 10.1016/j.jpain.2013.05.002. [DOI] [PubMed] [Google Scholar]
- 5.Vilholm OJ, Cold S, Rasmussen L, Sindrup SH. The postmastectomy pain syndrome: an epidemiological study on the prevalence of chronic pain after surgery for breast cancer. Br J Cancer. 2008;99:604–610. doi: 10.1038/sj.bjc.6604534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Berry DA, Cronin KA, Plevritis SK, Fryback DG, Clarke L, Zelen M, et al. Effect of screening and adjuvant therapy on mortality from breast cancer. N Engl J Med. 2005;353:1784–1792. doi: 10.1056/NEJMoa050518. [DOI] [PubMed] [Google Scholar]
- 7.Caffo O, Amichetti M, Ferro A, Lucenti A, Valduga F, Galligioni E. Pain and quality of life after surgery for breast cancer. Breast Cancer Res Treat. 2003;80:39–48. doi: 10.1023/A:1024435101619. [DOI] [PubMed] [Google Scholar]
- 8.Macdonald L, Bruce J, Scott NW, Smith WC, Chambers WA. Long-term follow-up of breast cancer survivors with post-mastectomy pain syndrome. Br J Cancer. 2005;92:225–230. doi: 10.1038/sj.bjc.6602304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Steegers MA, Wolters B, Evers AW, Strobbe L, Wilder-Smith OH. Effect of axillary lymph node dissection on prevalence and intensity of chronic and phantom pain after breast cancer surgery. J Pain. 2008;9:813–822. doi: 10.1016/j.jpain.2008.04.001. [DOI] [PubMed] [Google Scholar]
- 10.Tasmuth T, Blomqvist C, Kalso E. Chronic post-treatment symptoms in patients with breast cancer operated in different surgical units. Eur J Surg Oncol. 1999;25:38–43. doi: 10.1053/ejso.1998.0597. [DOI] [PubMed] [Google Scholar]
- 11.Miaskowski C, Cooper B, Paul SM, West C, Langford D, Levine JD, et al. Identification of patient subgroups and risk factors for persistent breast pain following breast cancer surgery. J Pain. 2012;13:1172–1187. doi: 10.1016/j.jpain.2012.09.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rietman JS, Dijkstra PU, Debreczeni R, Geertzen JH, Robinson DP, De Vries J. Impairments, disabilities and health related quality of life after treatment for breast cancer: a follow-up study 2.7 years after surgery. Disabil Rehabil. 2004;26:78–84. doi: 10.1080/09638280310001629642. [DOI] [PubMed] [Google Scholar]
- 13.Langford DJ, Paul SM, West C, Levine JD, Hamolsky D, Elboim C, et al. Persistent breast pain following breast cancer surgery is associated with persistent sensory changes, pain interference, and functional impairments. J Pain. 2014;15:1227–1237. doi: 10.1016/j.jpain.2014.08.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jung BF, Ahrendt GM, Oaklander AL, Dworkin RH. Neuropathic pain following breast cancer surgery: proposed classification and research update. Pain. 2003;104:1–13. doi: 10.1016/s0304-3959(03)00241-0. [DOI] [PubMed] [Google Scholar]
- 15.Haroutiunian S, Nikolajsen L, Finnerup NB, Jensen TS. The neuropathic component in persistent postsurgical pain: a systematic literature review. Pain. 2013;154:95–102. doi: 10.1016/j.pain.2012.09.010. [DOI] [PubMed] [Google Scholar]
- 16.Bruce J, Thornton AJ, Powell R, Johnston M, Wells M, Heys SD, et al. Psychological, surgical, and sociodemographic predictors of pain outcomes after breast cancer surgery: a population-based cohort study. Pain. 2014;155:232–243. doi: 10.1016/j.pain.2013.09.028. [DOI] [PubMed] [Google Scholar]
- 17.Andersen KG, Duriaud HM, Jensen HE, Kroman N, Kehlet H. Predictive factors for the development of persistent pain after breast cancer surgery. Pain. 2015;156:2413–2422. doi: 10.1097/j.pain.0000000000000298. [DOI] [PubMed] [Google Scholar]
- 18.International Association for the Study of Pain, Subcommittee on Taxonomy. Classification of chronic pain. Descriptions of chronic pain syndromes and definitions of pain terms: prepared by the International Association for the Study of Pain, Subcommittee on Taxonomy. Pain Suppl. 1986;3:S1–S226. [PubMed] [Google Scholar]
- 19.Freynhagen R, Baron R, Gockel U, Tölle TR. painDETECT: a new screening questionnaire to identify neuropathic components in patients with back pain. Curr Med Res Opin. 2006;22:1911–1920. doi: 10.1185/030079906X132488. [DOI] [PubMed] [Google Scholar]
- 20.Christiansen P, Vejborg I, Kroman N, Holten I, Garne JP, Vedsted P, et al. Position paper: breast cancer screening, diagnosis, and treatment in Denmark. Acta Oncol. 2014;53:433–444. doi: 10.3109/0284186X.2013.874573. [DOI] [PubMed] [Google Scholar]
- 21.Smith WC, Bourne D, Squair J, Phillips DO, Chambers WA. A retrospective cohort study of post mastectomy pain syndrome. Pain. 1999;83:91–95. doi: 10.1016/s0304-3959(99)00076-7. [DOI] [PubMed] [Google Scholar]
- 22.Carpenter JS, Sloan P, Andrykowski MA, McGrath P, Sloan D, Rexford T, et al. Risk factors for pain after mastectomy/lumpectomy. Cancer Pract. 1999;7:66–70. doi: 10.1046/j.1523-5394.1999.07208.x. [DOI] [PubMed] [Google Scholar]
- 23.Carpenter JS, Andrykowski MA, Sloan P, Cunningham L, Cordova MJ, Studts JL, et al. Postmastectomy/postlumpectomy pain in breast cancer survivors. J Clin Epidemiol. 1998;51:1285–1292. doi: 10.1016/s0895-4356(98)00121-8. [DOI] [PubMed] [Google Scholar]
- 24.Vilholm OJ, Cold S, Rasmussen L, Sindrup SH. Sensory function and pain in a population of patients treated for breast cancer. Acta Anaesthesiol Scand. 2009;53:800–806. doi: 10.1111/j.1399-6576.2009.01938.x. [DOI] [PubMed] [Google Scholar]
- 25.Gottrup H, Andersen J, Arendt-Nielsen L, Jensen TS. Psychophysical examination in patients with post-mastectomy pain. Pain. 2000;87:275–284. doi: 10.1016/S0304-3959(00)00291-8. [DOI] [PubMed] [Google Scholar]
- 26.Juhl AA, Karlsson P, Damsgaard TE. Fat grafting for alleviating persistent pain after breast cancer treatment: a randomized controlled trial. J Plast Reconstr Aesthet Surg. 2016;69:1192–1202. doi: 10.1016/j.bjps.2016.07.003. [DOI] [PubMed] [Google Scholar]
- 27.Abdullah TI, Iddon J, Barr L, Baildam AD, Bundred NJ. Prospective randomized controlled trial of preservation of the intercostobrachial nerve during axillary node clearance for breast cancer. Br J Surg. 1998;85:1443–1445. doi: 10.1046/j.1365-2168.1998.00843.x. [DOI] [PubMed] [Google Scholar]
- 28.Johansen J, Overgaard J, Blichert-Toft M, Overgaard M. Treatment of morbidity associated with the management of the axilla in breast-conserving therapy. Acta Oncol. 2000;39:349–354. doi: 10.1080/028418600750013122. [DOI] [PubMed] [Google Scholar]
- 29.Salmon RJ, Ansquer Y, Asselain B. Preservation versus section of intercostal-brachial nerve (IBN) in axillary dissection for breast cancer: a prospective randomized trial. Eur J Surg Oncol. 1998;24:158–161. doi: 10.1016/s0748-7983(98)92793-7. [DOI] [PubMed] [Google Scholar]
- 30.Langford DJ, Schmidt B, Levine JD, Abrams G, Elboim C, Esserman L, et al. Preoperative breast pain predicts persistent breast pain and disability after breast cancer surgery. J Pain Symptom Manage. 2015;49:981–994. doi: 10.1016/j.jpainsymman.2014.11.292. [DOI] [PMC free article] [PubMed] [Google Scholar]