Abstract
Background
Antihypertensive treatment mitigates the progression of chronic kidney disease. Here, we comparatively assessed the effects of antihypertensive agents in normotensive and hypertensive diabetic patients with microalbuminuric kidney disease.
Methods
MEDLINE, EMBASE, and the Cochrane Central Register of Controlled Trials were systematically searched for randomized controlled trials (RCTs) comparing oral antihypertensive agents in adult diabetic patients with microalbuminuria. The primary efficacy outcome was reduction in albuminuria, and the primary safety outcomes were dry cough, presyncope, and edema. Random-effects pairwise and Bayesian network meta-analyses were performed to produce outcome estimates for all RCTs, only hypertensive RCTs, or only normotensive RCTs. Surface under the cumulative ranking (SUCRA) probability rankings were calculated for all outcomes. Sensitivity analyses on type 2 diabetes status, age, or follow-up duration were also performed.
Results
A total of 38 RCTs were included in the meta-analyses. The angiotensin-converting enzyme inhibitor-calcium channel blocker (ACEI-CCB) combination therapy of captopril+diltiazem was most efficacious in reducing albuminuria irrespective of blood pressure status. However, the ACEI-angiotensin receptor blocker (ACEI-ARB) combination therapy of trandolapril+candesartan was the most efficacious in reducing albuminuria for normotensive patients, while the ACEI-CCB combination therapy of fosinopril+amlodipine was the most efficacious in reducing albuminuria for hypertensive patients. The foregoing combination therapies displayed inferior safety profiles relative to ACEI monotherapy with respect to dry cough, presyncope, and edema. With respect to type 2 diabetic patients with microalbuminuria, the Chinese herbal medicine Tangshen formula followed by the ACEI ramipril were the most efficacious in reducing albuminuria.
Conclusions
Trandolapril+candesartan appears to be the most efficacious intervention for reducing albuminuria for normotensive patients, while fosinopril+amlodipine appears to be the most efficacious intervention for reducing albuminuria for hypertensive patients. For practitioners opting for monotherapy, our SUCRA analysis supports the use of trandolapril and fosinopril in normotensive and hypertensive adult diabetic patients with microalbuminuria, respectively.
Introduction
Diabetes mellitus affects ~4% of the global adult population with an estimated 382 million affected individuals in 2013 expected to increase to an estimated 592 million affected individuals by the year 2035 [1]. Diabetes is the primary cause of end-stage kidney disease (ESRD), contributing to 40–50% of chronic dialysis patients [2]. As the increasing number of diabetic individuals is projected to have a serious impact on dialysis service and kidney transplant needs, the development of cost-effective therapeutic strategies for individuals with diabetic kidney disease is a crucial public health concern [3].
As it is well-established that blood pressure (BP) control is critical to slowing the decline in the glomerular filtration rate (GFR) [4], antihypertensive treatment has been credited with mitigating the progression of chronic kidney disease (CKD) to ESRD [5]. Specifically, clinical guidelines recommend a 130/80 mm Hg BP target as well as the use of angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) as first-line agents for BP control in CKD patients [5]. Indeed, a recent network meta-analysis consisting of 157 trials by Palmer et al. that comparatively assessed the efficacy and safety of BP-lowering agents in adult diabetic CKD patients found that ESRD risk was significantly reduced after combined treatment with an ACEI and an ARB [6].
However, Palmer et al.’s network meta-analysis specifically noted a limitation to their study: their ESRD outcomes were largely restricted to adult diabetic patients with macroalbuminuric kidney disease [6]. Thus, their results cannot be generalized to adult diabetic patients with microalbuminuric kidney disease. Moreover, although Palmer et al. did conduct a sensitivity analysis restricted to adult diabetic patients with microalbuminuria [6], their group did not comparatively examine normotensive versus hypertensive patients within this cohort. The examination of antihypertensive agents in reducing albuminuria in adult diabetic patients with microalbuminuria is of particular clinical importance for these patients, as (i) microalbuminuria in adult diabetic patients has been identified as a risk factor for adverse cardiovascular events, and (ii) a failure to control increasing albuminuria in these patients (after controlling for other risk factors) heightens the risk of inferior renal outcomes [7]. Moreover, the question is of importance to healthcare practitioners, as optimal long-term management of adult diabetic patients with microalbuminuria requires evidence-based recommendations on the efficacy and safety of various antihypertensive agents that are specifically tailored to this patient population [8].
Therefore, the aim of this network meta-analysis will be to assess the comparative effects of antihypertensive agents in reducing albuminuria in adult diabetic patients with microalbuminuric kidney disease. Moreover, we will specifically examine the comparative effects of antihypertensive agents in reducing albuminuria in normotensive versus hypertensive patients within this microalbuminuric cohort.
Methods
Study Design
This network meta-analysis was conducted according to the Preferred Reporting Items of Systematic Reviews and Meta-Analyses (PRISMA) statement [9]. This network meta-analysis–which integrates direct treatment comparisons within trials as well as indirect treatment comparisons compared against a common comparator across separate randomized controlled trials (RCTs) [10]–was performed using a frequentist analytical approach [11].
Search Strategy
MEDLINE, EMBASE, and the Cochrane Central Register of Controlled Trials were comprehensively searched up to October 2015 for relevant RCTs using the following search terms: (diabetes or diabetic) AND (antihypertensive OR “blood pressure-lower*” OR “blood pressure-reduc*”) AND microalbuminur* AND random* AND control*. An English language restriction was imposed on all searches.
Inclusion and Exclusion Criteria
Two investigators (Rongzhong Huang and Yang Sun) were responsible for independently selecting the studies, with any conflicts resolved through discussion. The inclusion and exclusion criteria were based on Palmer et al.’s previously published criteria with minor modifications [6].
The inclusion criteria for this network meta-analysis were as follows: (i) parallel-group RCT design, (ii) minimal eight-week follow-up period, (iii) adult participants (individuals aged 18 years or older) with diabetes and microalbuminuric kidney disease (i.e., evidenced by a urinary albumin excretion rate (UAER) > 30 mg/day), (iv) comparing an oral antihypertensive agent (alone or in combination) (e.g., ACEI, ARB, calcium-channel blocker (CCB), β-blocker, α-blocker, diuretic, renin inhibitor, aldosterone antagonist, or endothelin inhibitor) against a second antihypertensive agent or combination therapy, placebo, or control, (v) specifically analyzing and reporting on either a hypertensive, normotensive, or a mixed population and (vi) measuring UAER as an outcome. Both fixed-dose and flexible-dose RCTs with dose titration were included. We also included RCTs with a general population of adult diabetes participants when the data for those participants who did have microalbuminuric kidney disease could be extracted separately.
The exclusion criteria for this network meta-analysis were as follows: (i) studies in children and adolescents (defined as individuals aged under 18 years), (ii) studies including patients with microalbuminuric kidney disease secondary to causes other than diabetes (if specified), (iii) studies including patients with an active kidney transplant, undergoing kidney transplantation, or undergoing dialysis (if specified), (iv) studies failing to report the number of patients, (v) studies failing to report the mean value of the primary efficacy outcome, or (vi) studies failing to report the data necessary to estimate the standard deviation (SD) of the primary efficacy outcome.
Outcomes
As albumin excretion has been shown to increase in diabetic patients prior to the development of diabetic nephropathy [12], the primary efficacy outcome was defined as reduction in albuminuria as expressed by the UAER. Specifically, for each included study, the mean percentage (%) reduction in UAER for each normotensive cohort and each hypertensive cohort were calculated in order to enable normotensive versus hypertensive comparisons. The primary safety outcomes were dry cough, presyncope, and peripheral edema.
Data Extraction
Two investigators (Rongzhong Huang and Yang Sun) were responsible for independently extracting the data into a spreadsheet according to a predefined protocol, with any conflicts resolved through discussion. The following parameters were extracted from each included RCT: first author’s name, year of publication, study location, mean age of participants, type of diabetes among participants, definition of microalbuminuria, BP categories of participants, intervention(s) prescribed (with dosage levels), and follow-up period (in months), the primary efficacy outcome of UAER (standardized to μg/min), and the primary safety outcomes of dry cough, presyncope, and peripheral edema.
Risk of Bias Assessment
Risk of bias was assessed using the approach recommended by the Cochrane reviews. The following bias domains were independently assessed by two investigators (Rongzhong Huang and Yang Sun): random sequence generation, allocation concealment, blinding of investigators and/or participants, blinding of outcome assessment, degree of incompleteness of outcome data, and selective reporting of study outcomes. Each bias item was scored as ‘low risk’, ‘unclear risk’, or ‘high risk’.
Statistical Analysis
Standard pairwise and network meta-analyses were performed to estimate the primary efficacy outcome. Prior to performing the meta-analyses, Stata version 13 was first used to estimate the SDs for studies that failed to report SDs using (i) interquartile ranges, (ii) the 95% confidence intervals (CIs) of the primary efficacy outcome, and (iii) the two-sided p-value corresponding to the t-test between the two treatment modalities.
Then, using R. version 3.2.2 (metaphor package and R routines), a random-effects model was used to perform the standard pairwise meta-analysis. Heterogeneity was assessed with the I2 metric. For the primary data analysis, we used the intention-to-treat principle. In studies that only reported a per-protocol analysis, we conservatively assumed that all dropouts were treatment failures for inclusion in the intention-to-treat analysis; this procedure guarded against favoring active drugs that could be the more harmful [13, 14]. The estimates of the primary efficacy outcome were presented as standardized mean differences (SMDs) with 95% CIs.
Next, a random-effects network meta-analysis was performed in ADDIS version 1.16.6 –a software package employing Bayesian Markov chain Monte Carlo methods—assuming a common heterogeneity variable (tau [τ]) for all comparisons. τ is the estimated SD of the underlying effects of treatment across the studies in a meta-analysis. RCTs with drugs that could not be connected to the network were excluded from the network meta-analysis and only drugs directly or indirectly connected to placebo were included in the downstream ranking analysis. Consistency of the RCTs included in the network was assessed through applying inconsistency and node-splitting models. The consistency results were considered insignificant when 95% CIs of inconsistency factors included zero or when the p-value was greater than 0.05 for the comparison between direct and indirect effects in the node splitting analysis. The estimates of the primary efficacy outcome were calculated as SMDs with 95% CIs, while the network meta-analyses estimates of the primary safety outcomes were calculated as odds ratios.
To rank the treatments, we calculated surface under the cumulative ranking (SUCRA) probabilities, which express as a percentage the efficacy or safety of each intervention relative to a hypothetical ideal intervention. Thus, a large SUCRA score indicates a more effective or safer intervention. Rankings were calculated for the primary efficacy outcome and the three primary safety outcomes.
In order to assess the effects of hypertensive status, the foregoing meta-analyses were also re-performed by pooling only hypertensive studies and pooling only normotensive studies. In addition, we performed three separate sensitivity analyses on type 2 diabetes status, age, or follow-up duration by measuring the effects of including only trials with type 2 diabetes participants, trials with participants aged 40 and older, or trials with follow-up durations of 12 months and longer, respectively.
Results
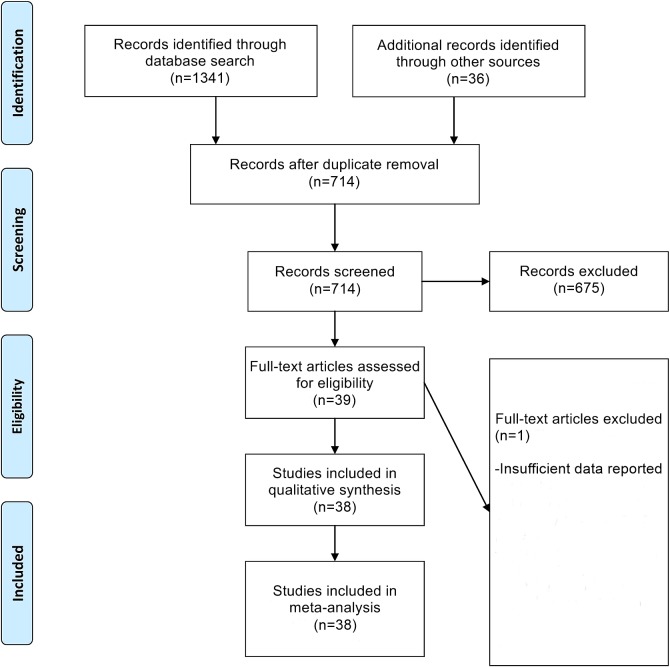
The PRISMA flowchart detailing the study selection process is provided in Fig 1. From an initial set of 714 non-duplicate records, a total of 38 RCTs were finally included in the meta-analysis. The characteristics of these included RCTs are detailed in Table 1. The risk of bias assessment for these included RCTs are detailed in Table 2.
Fig 1. PRISMA Flowchart of Study Selection Process.
Table 1. Characteristics of Included RCTs.
| Study | Location | N | Mean age (range) | Diabetestype | Microalbuminuria definition | BP categories | Intervention(s) | Follow-up (months) |
|---|---|---|---|---|---|---|---|---|
| ABCD-2V (Estacio) 2006 [28] | USA | 129 | 56.1 (40–81) | 2 | UAER 20–200 μg/min | Normotensive | Valsartan 80 mg/d, then valsartan 160 mg/d then HCTZ 12.5 mg/d, 25 mg/d, then metoprolol 50 mg/d, 100 mg twice a day; placebo | 22.8 |
| Atmaca 2006 [29] | Turkey | 26 | 55.1 (36.7–73.5)* | 2 | UAER 30–300 mg/d | Normotensive | Lisinopril 10 mg/d, losartan 50 mg/d, lisinopril 10 mg/d + losartan 50 mg/d | 12 |
| Bojestig 2001 [30] | Sweden | 55 | 39.6 (20.3–58.9)* | 1 | Urine albumin to creatinine ratio 2.5–25 mg/mmol | Normotensive | Ramipril 1.25 mg/d, ramipril 5 mg/d, placebo | 48 |
| CALM (Mogensen) 2000 [31] | Multi-national | 199 | 60.0 (30–75) | 2 | Urine albumin to creatinine ratio 2.5–25 mg/mmol | Hypertensive | Lisinopril 20 mg/d, candesartan 16 mg/d, lisinopril 20 mg/d + candesartan 16 mg/d | 3 |
| ESTIMATE-A (Kojima) 2013 [32] | Japan | 40 | 68.1 (20–75) | 2 | UAE> 30 mg/g creatinine and serum creatinine <1.5 mg/dl for men and <1.2 mg/dl for women | Hypertensive | Telmisartan 40–80 mg/d + TCMZ 1 mg/d, telmisartan 40–80 mg/d + TCMZ 1 mg/d + amlodipine 5 mg/d | 6 |
| EUCLID (Chaturvedi) 1997 [33] | UK | 530 | 33.0 (20–59) | 1 | UAER 20–200 μg/min | Hypertensive | Lisinopril 10–20 mg, placebo | 24 |
| Fogari 1997a [34] | Italy | 50 | 53.9 (51.6–56.1)* | 2 | UAER 30–300 mg/d;serum creatinine<1.4 mg/dl | Hypertensive | Amlodipine 10 mg/d, enalapril 20 mg/d | 12 |
| Fogari 1997b [34] | Italy | 45 | 57.1 (54.7–59.5)* | 2 | UAER 30–300 mg/d;serum creatinine> = 1.3 mg/dl | Hypertensive | Benazepril 10 mg/d, benazepril 10 mg/d + amlodipine 5 mg/d | 6 |
| Fogari 2000 [35] | Italy | 254 | 68.3 (60–75) | 2 | UAER 30–300 mg/d;serum creatinine<1.3 mg/dl | Hypertensive | Amlodipine 5–10 mg/d, fosinopril 10–20 mg/d, placebo | 24 |
| Fogari 2002 [36] | Italy | 453 | 62.5 (44.4–80.7)* | 2 | UAER 30–300 mg/d;serum creatinine<1.5 mg/dl | Hypertensive | Fosinopril 10–30 mg/d, amlodipine 5–15 mg/d, fosinopril 10–30 mg/d + amlodipine 5–15 mg/d | 48 |
| Fogari 2005 [37] | Italy | 121 | 60.3 (47.2–73.3)* | 2 | UAER 30–300 mg/d;serum creatinine<1.4 mg/dl | Hypertensive | Manidipine 10 mg/d, lisinopril 10 mg/d | 24 |
| Fogari 2007 [38] | Italy | 174 | 55.7 (40–65) | 2 | UAER 30–300 mg/d | Hypertensive | Candesartan 16 mg/d + manidipine 10–20 mg/d, candesartan 16 mg/d + HCTZ 12.5–25 mg/d | 6 |
| Fogari 2012 [39] | Italy | 109 | 65.0 (30–75) | 2 | UAER 30–300 mg/d | Hypertensive | Valsartan 160 mg/d + amlodipine 5 mg/d + canrenone 25–50 mg/d, valsartan 160 mg/d + amlodipine 5 mg/d + HCTZ 12.5–25 mg/d | 6 |
| Fogari 2013 [40] | Italy | 176 | 60.8 (25–75) | 2 | UAER 200–300 mg/d | Hypertensive | Imidapril 10–20 mg/d, ramipril 5–10 mg/d | 6 |
| JAPAN-IDDM (Katayama) 2002 [41] | Japan | 79 | 30.9 (20–50) | 1 | UAER>30 mg/d | Hypertensive | Captopril 37.5 mg/d, imidapril 5 mg/d, placebo | 17.8 |
| Jerums 2001 [42] | Australia | 33 | 30.8 (16–65) | 1 | UAER 20–200 μg/min;serum creatinine<200 μmol/l | Normotensive | Perindopril 2–8 mg/d, nifedipine 20–80 mg/d, placebo | 67.2 |
| Josefsberg 1995 [43] | Canada | 21 | 53.0 (37–68) | 2 | UAER 20–200 μg/min | Hypertensive | Nitrendipine 10–40 mg/d, enalapril 5–20 mg/d | 7.5 |
| Kohlmann 2009 [44] | Multi-national | 110 | 63.3 (45–81) | 2 | Urine albumin to creatinine ratio 2.5–25 mg/mmol for men and 3.5–25 mg/mmol for women | Hypertensive | Manidipine 10 mg/d + delapril 30 mg/d, losartan 50 mg/d + HCTZ 12.5 mg/d | 12 |
| Lacourciere 2000 [45] | Canada | 103 | 58.5 (38.8–78.2)* | 2 | UAER 20–350 μg/min;serum creatinine<1.7 mg/dl | Hypertensive | Losartan 50 mg/d, enalapril 5–10 mg/d | 12 |
| Li 2015 [46] | China | 98 | 58.6 (38.8–78.3)* | 2 | UAER 20–200 μg/min, GFR 50–120 ml/min, serum creatinine 50–100 μmol/l | Mixed | Tangshen formula, placebo | 6 |
| MARVAL (Viberti) 2002 [47] | UK | 368 | 58.0 (35–75) | 2 | UAER 20–200 μg/min;normal serum creatinine | Mixed | Valsartan 80 mg/d, amlodipine 5 mg/d | 6 |
| Muirhead 1999 [48] | Canada | 122 | 56.0 (35.9–76.2)* | 2 | UAER 20–300 μg/min;GFR > = 60 ml/min | Mixed | Valsartan 80 mg/d, valsartan 160 mg/d, captopril 75 mg/d, placebo | 12 |
| Melbourne Diabetic Nephropathy Study Group (Doyle) 1991 [49] | Australia | 43 | 50.0 (18–66) | 1 and 2 | UAER 20–200 μg/min;serum creatinine<2.3 mg/dl | Separate normotensive and hypertensive cohorts | Perindopril 2–8 mg/d, nifedipine 10–40 mg twice daily | 12 |
| Nakamura 2002 [50] | Japan | 60 | 56.5 (37.6–75.4)* | 2 | UAER 20–200 μg/min | Normotensive | Trandolapril 2 mg/d, candesartan 8 mg/d, trandolapril 2 mg/d + candesartan 8 mg/d, placebo | 18 |
| Perez-Maraver 2005 [51] | Spain | 36 | 60.4 (45.6–75.2)* | 2 | UAER 30–300 μg/min | Hypertensive | Captopril 25 mg/12 h-50 mg/8 h, captopril 25 mg/12 h-50 mg/8 h + diltiazem 120 mg/d | 24 |
| Poulsen 2001 [52] | Denmark | 21 | 32.4 (18.0–52.2)* | 1 | UAER 20–70 μg/min | Normotensive | Lisinopril 20 mg/d, placebo | 24 |
| PREMIER (Mogensen) 2003 [53] | Multi-national | 481 | 59.6 (40–75) | 2 | UAER 20–500 μg/min, serum creatinine<1.6 mg/dl | Hypertensive | Perindopril 2–8 mg/d + 0.625–2.5 mg/d indapamide, enalapril 10–40 mg/d | 12 |
| Sano 1994 [54] | Japan | 48 | 63.5 (50–76) | 2 | UAER 20–500 μg/min, serum creatinine<140 μmol/l | Normotensive | Enalapril 5 mg/d, placebo | 48 |
| Sato 2003 [55] | Japan | 50 | 63.3 (43.1–83.6)* | 1 and 2 | UAE 30–300 mg/g creatinine,GFR< = 60 ml/min | Separate hypertensive and normotensive cohorts | ACEI (trandolapril 1.5 mg/d or enalapril 7.5 mg/d), candesartan 7.1 mg/d | 11 |
| Schnack 1994 [56] | Austria | 15 | 37.9 (29.9–45.9)* | 1 | UAER 30–300 mg/d;serum creatinine<1.4 mg/dl | Normotensive | Nifedipine 30 mg/d, placebo | 12 |
| Sengul 2006 [57] | Turkey | 145 | 57.2 (40–65) | 2 | UAER 30–300 mg/d;creatinine< = 1.7 mg/dl | Hypertensive | Lisinopril 20 mg/d, telmisartan 80 mg/d, lisinopril 20 mg/d + telmisartan 80 mg/d (following lisinopril monotherapy), lisinopril 20 mg/d + telmisartan 80 mg/d (following telmisartan monotherapy) | 7 |
| Shigihara 2000 [58] | Japan | 30 | 62.9 (56.0–69.7)* | 2 | Microalbuminuria | Hypertensive | ACEI (enalapril 7.0 mg/d, trandolapril 1.6 mg/d, or imidapril 9.2 mg/d), ACEI (enalapril 5.3 mg/d, trandolapril 1.6 mg/d, or imidapril 5 mg/d) + amlodipine 7.0 mg/d | 3 |
| Takebayashi 2006 [59] | Japan | 37 | N/R | 2 | UAE>30 mg/g creatinine | Mixed | Spironolactone 50 mg/d, amlodipine 2.5 mg/d | 3 |
| Tan 2002 [60] | China | 80 | 54.5 (35.6–73.4)* | 2 | UAER 20–200 μg/min | Mixed | Losartan 50 mg/d, placebo | 6 |
| Tutuncu 2001 [61] | Turkey | 34 | 55.6 (38.7–72.5)* | 2 | UAER 30–300 mg/d | Normotensive | Enalapril 5 mg/d, losartan 50 mg/d, enalapril 5 mg/d + losartan 50 mg/d | 12 |
| Weil 2013 [62] | USA | 78 | 42.1 (22.3–61.9)* | 2 | ACR 30–300 mg/g;serum creatinine <1.4 mg/dl | Mixed | Losartan 50–100 mg/d, placebo | 70.8 |
| Viberti 1994 [63] | Multi-national | 92 | 31.5 (18–54) | 1 | UAER 20–200 μg/min;serum creatinine<150 μmol/l | Normotensive | Captopril 100 mg/d, placebo | 24 |
| Zandbergen 2003 [64] | Netherlands | 147 | 57.7 (34.4–81.0)* | 2 | UAER 20–200 μg/min; serum creatinine< = 1.7 mg/dl | Normotensive | Losartan 50–100 mg/d, placebo | 2.5 |
*Age range estimated from the reported standard deviations (SDs) [65].
Abbreviations: UAE, urine albumin excretion; UAER, urine albumin excretion rate; ACR, albumin-to-creatinine ratio; GFR, glomerular filtration rate; ACEI, angiotensin-converting enzyme inhibitor; HCTZ, hydrochlorothiazide; TCMZ, trichlormethiazide
Table 2. Risk of Bias Assessment.
| Study | Random sequence generation | Allocation concealment | Blinding of personnel and participants | Blinding of outcome assessment | Incomplete outcome data | Selective reporting | Other bias | Total score |
|---|---|---|---|---|---|---|---|---|
| ABCD-2V (Estacio) 2006 [28] | L | L | L | L | L | L | L | 7 |
| Atmaca 2006 [29] | L | U | U | U | L | L | H | 3 |
| Bojestig 2001 [30] | L | L | L | L | L | L | H | 6 |
| CALM (Mogensen) 2000 [31] | L | L | L | L | L | L | L | 7 |
| ESTIMATE-A (Kojima) 2013 [32] | L | U | U | U | L | L | h | 3 |
| EUCLID (Chaturvedi) 1997 [33] | L | U | l | U | L | L | L | 5 |
| Fogari 1997a [34] | L | U | U | U | L | L | H | 3 |
| Fogari 1997b [34] | L | U | U | U | L | L | H | 3 |
| Fogari 2000 [35] | L | L | L | L | L | L | L | 7 |
| Fogari 2002 [36] | L | U | U | U | L | L | L | 4 |
| Fogari 2005 [37] | L | U | U | U | L | L | H | 3 |
| Fogari 2007 [38] | L | L | L | L | L | L | L | 7 |
| Fogari 2012 [39] | L | L | L | L | L | L | L | 7 |
| Fogari 2013 [40] | L | L | L | L | L | L | L | 7 |
| JAPAN-IDDM (Katayama) 2002 [41] | L | U | U | U | L | L | H | 3 |
| Jerums 2001 [42] | L | L | L | L | L | L | H | 6 |
| Josefsberg 1995 [43] | L | U | U | U | L | L | H | 3 |
| Kohlmann 2009 [44] | L | L | L | L | L | L | L | 7 |
| Lacourciere 2000 [45] | L | L | L | L | L | L | H | 6 |
| Li 2015 [46] | L | L | L | L | L | L | L | 7 |
| MARVAL (Viberti) 2002 [47] | L | L | L | L | L | L | L | 7 |
| Muirhead 1999 [48] | L | U | l | U | L | L | H | 4 |
| Melbourne Diabetic Nephropathy Study Group (Doyle) 1991 [49] | L | L | L | L | L | L | H | 6 |
| Nakamura 2002 [50] | L | U | U | U | L | L | H | 3 |
| Perez-Maraver 2005 [51] | L | H | H | H | L | L | H | 3 |
| Poulsen 2001 [52] | L | L | L | L | L | L | H | 6 |
| PREMIER (Mogensen) 2003 [53] | L | U | U | U | L | L | H | 3 |
| Sano 1994 [54] | L | U | U | U | L | L | H | 3 |
| Sato 2003 [55] | U | U | U | l | L | L | H | 3 |
| Schnack 1994 [56] | L | L | L | L | L | L | H | 6 |
| Sengul 2006 [57] | L | U | U | U | L | L | L | 4 |
| Shigihara 2000 [58] | L | U | U | U | L | L | H | 3 |
| Takebayashi 2006 [59] | L | U | U | U | L | L | H | 3 |
| Tan 2002 [60] | L | L | L | L | L | L | L | 7 |
| Tutuncu 2001 [61] | L | U | U | U | L | L | H | 3 |
| Weil 2013 [62] | L | L | L | L | L | L | H | 6 |
| Viberti 1994 [63] | L | L | L | L | L | L | H | 6 |
| Zandbergen 2003 [64] | L | L | L | L | L | L | L | 7 |
Abbreviations: L, low risk of bias; U, unclear risk of bias; H, high risk of bias
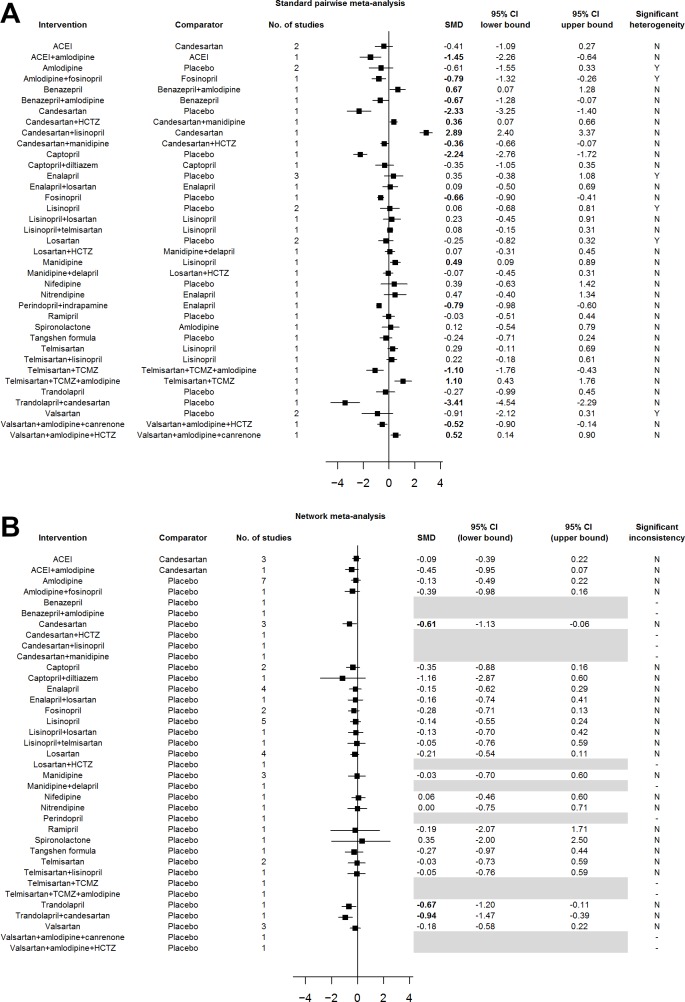
First, standard pairwise meta-analysis (Fig 2A) and network meta-analysis (Fig 2B) were performed on all included RCTs. The network map diagramming the direct comparisons for this overall network meta-analysis is provided in S1 Fig. SUCRA analysis for the primary efficacy outcome of UAER reduction revealed that captopril+diltiazem was the most efficacious intervention, followed by trandolapril+candesartan, followed by trandolapril (Table 3). SUCRA analysis for the primary safety outcomes revealed that: (i) captopril, followed by captopril+diltiazem, followed by ramipril were the safest interventions for dry cough; (ii) ACEI (random selection of enalapril, trandolapril, or imidapril), followed by lisinopril+candesartan, and followed by lisinopril were the safest interventions for presynope, and (iii) lisinopril, followed by candesartan, followed by ACEI (random selection of enalapril, trandolapril, or imidapril) were the safest interventions for peripheral edema (Table 3).
Fig 2. Standard Pairwise and Network Meta-Analyses of All RCTs.
Results from the (A) standard pairwise meta-analysis and (B) network meta-analysis of all RCTs. Standardized mean differences (SMDs) and 95% confidence intervals (CIs) are presented with statistically significant SMDs denoted in bold font. Greyed-out areas represent comparisons that could not be connected into the network. Abbreviations: ACEI, ACE inhibitor (random selection of enalapril, trandolapril, or imidapril); HCTZ, hydrochlorothiazide; TCMZ, trichlormethiazide; SMD, standardized mean difference; 95% CI, 95% confidence interval.
Table 3. SUCRA-Based Primary Efficacy and Safety Outcome Rankings from All RCTs.
| Primary safety outcome rankings | ||||
|---|---|---|---|---|
| Intervention | Primary efficacy | Dry cough | Presyncope | Peripheral edema |
| outcome ranking* | ranking* | ranking* | ranking* | |
| ACEI | - | - | 1 | 4 |
| ACEI+amlodipine | - | - | - | - |
| Amlodipine | 13 | - | - | - |
| Amlodipine+fosinopril | 5 | - | - | - |
| Benazepril | - | - | - | - |
| Benazepril+amlodipine | - | - | - | - |
| Candesartan | 4 | - | 5 | 3 |
| Candesartan+HCTZ | - | - | - | - |
| Candesartan+lisinopril | - | - | 3 | 5 |
| Candesartan+manidipine | - | - | - | - |
| Captopril | 6 | 1 | - | - |
| Captopril+diltiazem | 1 | 2 | - | - |
| Enalapril | 12 | - | - | - |
| Enalapril+losartan | 10 | - | - | - |
| Fosinopril | 8 | - | - | - |
| Lisinopril | 14 | - | 4 | 2 |
| Lisinopril+losartan | 15 | - | - | - |
| Lisinopril+telmisartan† | 18 | - | - | - |
| Losartan | 9 | - | 6 | - |
| Losartan+HCTZ | - | - | - | - |
| Manidipine | 19 | - | - | 6 |
| Manidipine+delapril | - | - | - | - |
| Nifedipine | 22 | - | - | - |
| Nitrendipine | 21 | - | - | - |
| Perindopril | - | - | - | - |
| Perindopril+indapamide | - | - | - | - |
| Ramipril | 23 | 4 | - | - |
| Spironolactone | 24 | - | - | - |
| Tangshen formula | 7 | - | - | - |
| Telmisartan | 20 | - | - | - |
| Telmisartan+lisinopril† | 17 | - | - | - |
| Telmisartan+TCMZ | - | - | - | - |
| Telmisartan+TCMZ+amlodipine | - | - | - | - |
| Trandolapril | 3 | - | - | - |
| Trandolapril+candesartan | 2 | - | - | - |
| Valsartan | 11 | - | - | - |
| Valsartan+amlodipine+canrenone | - | - | - | - |
| Valsartan+amlodipine+HCTZ | - | - | - | - |
*Placebo was ranked 16th in the primary efficacy outcome, 3rd in the cough safety outcome, 2nd in the presyncope safety outcome, and 1st in the peripheral edema safety outcome.
†Lisinopril+telmisartan refers to lisinopril+telmisartan combination therapy following lisinopril monotherapy, while telmisartan+lisinopril refers to lisinopril+telmisartan combination therapy following telmisartan monotherapy.
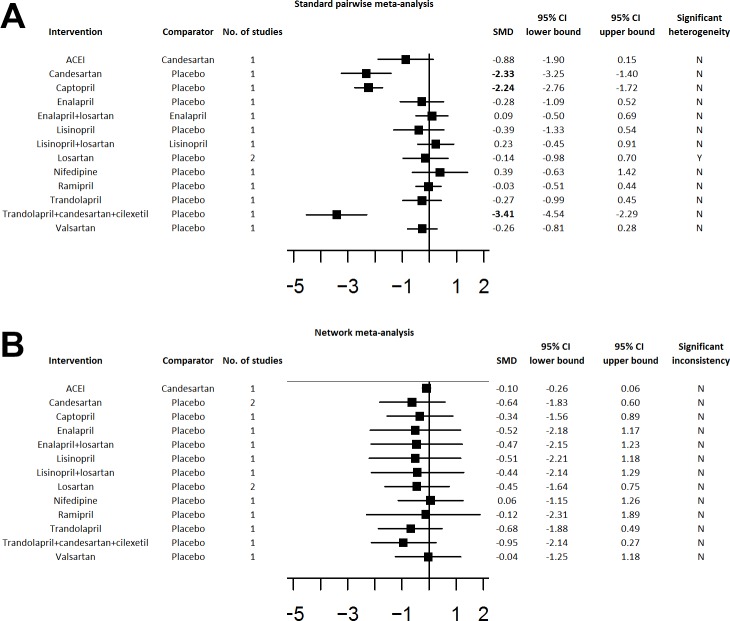
Next, standard pairwise meta-analysis (Fig 3A) and network meta-analysis (Fig 3B) were performed on RCTs with exclusively normotensive participants. The network map diagramming the direct comparisons for this normotensive network meta-analysis is provided in S2 Fig. SUCRA analysis for the primary efficacy outcome of UAER reduction revealed that trandolapril+candesartan was the most efficacious intervention, followed by trandolapril, followed by candesartan (Table 4). SUCRA analysis for the primary safety outcomes revealed that: (i) captopril followed by ramipril were the safest interventions for dry cough; (ii) losartan followed by lisinopril were the safest interventions for presyncope, and (iii) all interventions were equivalently safe to placebo for peripheral edema (Table 4).
Fig 3. Standard Pairwise and Network Meta-Analyses of Normotensive RCTs.
Results from the (A) standard pairwise meta-analysis and (B) network meta-analysis of normotensive RCTs. Standardized mean differences (SMDs) and 95% confidence intervals (CIs) are presented with statistically significant SMDs denoted in bold font. Greyed-out areas represent comparisons that could not be connected into the network. Abbreviations: ACEI, ACE inhibitor (random selection of enalapril, trandolapril, or imidapril); HCTZ, hydrochlorothiazide; TCMZ, trichlormethiazide; SMD, standardized mean difference; 95% CI, 95% confidence interval.
Table 4. SUCRA-Based Primary Efficacy and Safety Outcome Rankings from Normotensive RCTs.
| Primary safety outcome rankings | ||||
|---|---|---|---|---|
| Intervention | Primary efficacy | Dry cough | Presyncope | Peripheral edema |
| outcome ranking* | ranking* | ranking* | ranking* | |
| ACEI | - | - | - | - |
| Candesartan | 3 | - | - | - |
| Captopril | 9 | 1 | - | - |
| Enalapril | 5 | - | - | - |
| Enalapril+losartan | 6 | - | - | - |
| Lisinopril | 4 | - | - | - |
| Lisinopril+losartan | 8 | - | - | - |
| Losartan | 7 | - | 2 | - |
| Nifedipine | 12 | - | - | - |
| Ramipril | 13 | 2 | - | - |
| Trandolapril | 2 | - | - | - |
| Trandolapril+candesartan+cilexetil | 1 | - | - | - |
| Valsartan | 11 | - | - | - |
*Placebo was ranked 10th in the primary efficacy outcome, was ranked 3rd in the cough safety outcome, was ranked 1st in the presyncope safety outcome, and was not ranked in the peripheral edema safety outcome.
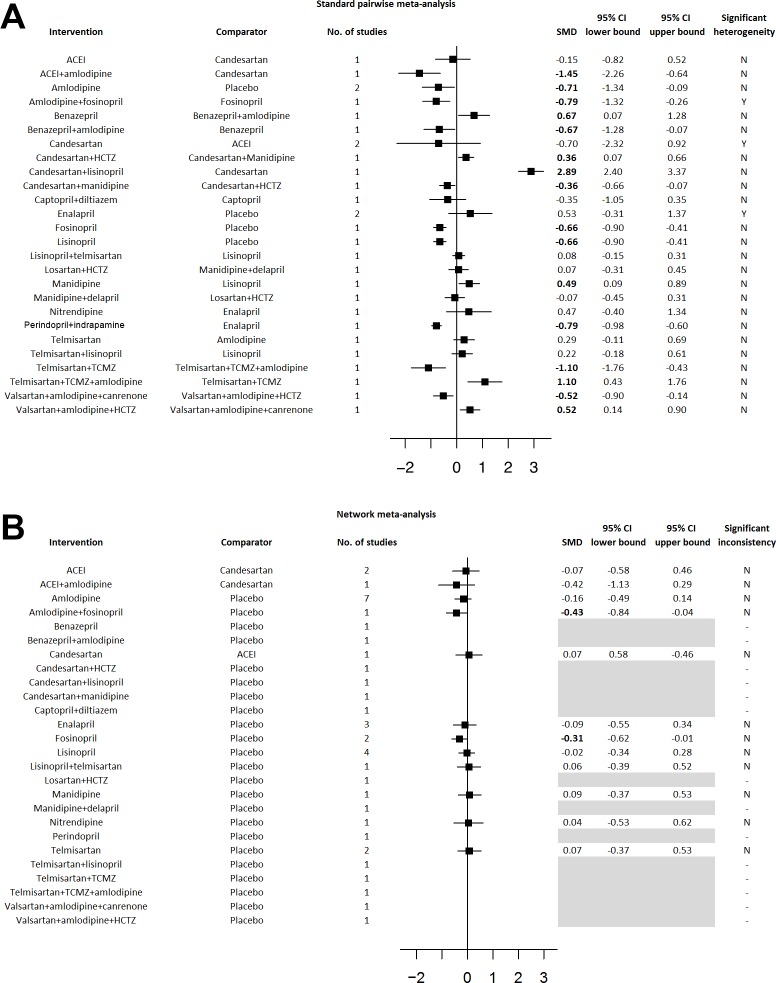
Finally, standard pairwise meta-analysis (Fig 4A) and network meta-analysis (Fig 4B) were performed on RCTs with exclusively hypertensive participants. The network map diagramming the direct comparisons for this hypertensive network meta-analysis is provided in S3 Fig. SUCRA analysis for the primary efficacy outcome of UAER reduction revealed that fosinopril+amlodipine was the most efficacious intervention, followed by fosinopril, followed by amlodipine (Table 5). SUCRA analysis for the primary safety outcomes revealed that: (i) amlodipine, followed by fosinopril+amlodipine, followed by fosinopril were the safest interventions for dry cough; (ii) ACEI (random selection of enalapril, trandolapril, or imidapril), followed by lisinopril+candesartan, and followed by candesartan were the safest interventions for presyncope, and (iii) lisinopril+candesartan followed by lisinopril, followed by ACEI (random selection of enalapril, trandolapril, or imidapril) were the safest interventions for peripheral edema (Table 5).
Fig 4. Standard Pairwise and Network Meta-Analyses of Hypertensive RCTs.
Results from the (A) standard pairwise meta-analysis and (B) network meta-analysis of hypertensive RCTs. Standardized mean differences (SMDs) and 95% confidence intervals (CIs) are presented with statistically significant SMDs denoted in bold font. Greyed-out areas represent comparisons that could not be connected into the network. Abbreviations: ACEI, ACE inhibitor (random selection of enalapril, trandolapril, or imidapril); HCTZ, hydrochlorothiazide; TCMZ, trichlormethiazide; SMD, standardized mean difference; 95% CI, 95% confidence interval.
Table 5. SUCRA-Based Primary Efficacy and Safety Outcome Rankings from Hypertensive RCTs.
| Primary safety outcome rankings | ||||
|---|---|---|---|---|
| Intervention | Primary efficacy | Dry cough | Presyncope | Peripheral edema |
| outcome ranking* | ranking* | ranking* | ranking* | |
| ACEI | - | - | 1 | 4 |
| ACEI+amlodipine | - | - | - | - |
| Amlodipine | 3 | 1 | - | - |
| Amlodipine+fosinopril | 1 | 2 | - | - |
| Benazepril | - | - | - | - |
| Benazepril+amlodipine | - | - | - | - |
| Candesartan | - | - | 4 | 5 |
| Candesartan+HCTZ | - | - | - | - |
| Candesartan+lisinopril | - | - | 3 | 2 |
| Candesartan+manidipine | - | - | - | - |
| Captopril+diltiazem | - | - | - | - |
| Enalapril | 5 | 4 | - | - |
| Fosinopril | 2 | 3 | - | - |
| Lisinopril | 4 | - | 5 | 3 |
| Lisinopril+telmisartan† | 6 | - | - | - |
| Losartan+HCTZ | - | - | - | - |
| Manidipine | 8 | - | - | 6 |
| Manidipine+delapril | - | - | - | - |
| Nitrendipine | 10 | - | - | - |
| Perindopril | - | - | - | - |
| Perindopril+indapamide | - | - | - | - |
| Telmisartan | 9 | - | - | - |
| Telmisartan+lisinopril† | - | - | - | - |
| Telmisartan+TCMZ | - | - | - | - |
| Telmisartan+TCMZ+amlodipine | - | - | - | - |
| Valsartan+amlodipine+canrenone | - | - | - | - |
| Valsartan+amlodipine+HCTZ | - | - | - | - |
*Placebo was ranked 7th in the primary efficacy outcome, was not ranked in the cough safety outcome, was ranked 2nd in the presyncope safety outcome, and was ranked 1st in the peripheral edema safety outcome.
†Lisinopril+telmisartan refers to lisinopril+telmisartan combination therapy following lisinopril monotherapy, while telmisartan+lisinopril refers to lisinopril+telmisartan combination therapy following telmisartan monotherapy.
Finally, three separate sensitivity analyses were performed to account for any putative effects of type 2 diabetes status, age, or follow-up duration upon the SUCRA analysis for the primary efficacy outcome of UAER reduction. First, the network map diagramming the direct comparisons for the type 2 diabetes sensitivity analysis is provided in S4 Fig. For type 2 diabetes patients with microalbuminuric kidney disease, SUCRA analysis for the primary efficacy outcome revealed that the Tangshen formula was the most efficacious intervention, followed by ramipril, followed by spironolactone (Table 6). Second, the network map diagramming the direct comparisons for the age sensitivity analysis is provided in S5 Fig. For diabetic patients with microalbuminuria aged 40 and over, SUCRA analysis for the primary efficacy outcome revealed that benazepril was the most efficacious intervention, followed by ACEI+amlodipine, followed by candesartan+HCTZ (Table 6). Third, the network map diagramming the direct comparisons for the follow-up duration sensitivity analysis is provided in S6 Fig. For studies with a follow-up duration of 12 months and longer, SUCRA analysis for the primary efficacy outcome revealed that captopril was the most efficacious intervention, followed by perindopril+indapamide, followed by benazepril+amlodipine (Table 6).
Table 6. Sensitivity Analyses for the Primary Efficacy Outcome by Diabetes Type, Age, and Follow-Up Duration.
| Intervention | Overall SUCRA rank* | SUCRA rank for type 2 diabetes only* | SUCRA rank for aged 40+ only* | SUCRA rank for 12+ months follow-up only* |
|---|---|---|---|---|
| ACEI | - | 7 | 6 | - |
| ACEI+amlodipine | - | 26 | 2 | - |
| Amlodipine | 13 | 6 | - | - |
| Amlodipine+fosinopril | 5 | 21 | - | 6 |
| Benazepril | - | 20 | 1 | 7 |
| Benazepril+amlodipine | - | 4 | - | 3 |
| Candesartan | 4 | - | 7 | - |
| Candesartan+HCTZ | - | 15 | 3 | - |
| Candesartan+lisinopril | - | 28 | - | - |
| Candesartan+manidipine | - | 5 | 7 | - |
| Captopril | 6 | 10 | 4 | 1 |
| Captopril+diltiazem | 1 | 27 | - | 14 |
| Enalapril | 12 | 24 | 4 | 15 |
| Enalapril+losartan | 10 | 11 | - | 11 |
| Fosinopril | 8 | 8 | 2 | 5 |
| Lisinopril | 14 | 16 | - | 13 |
| Lisinopril+losartan | 15 | 17 | - | 8 |
| Lisinopril+telmisartan† | 18 | 14 | 5 | - |
| Losartan | 9 | 18 | - | 11 |
| Losartan+HCTZ | - | 12 | 8 | 10 |
| Manidipine | 19 | 23 | 6 | 6 |
| Manidipine+delapril | - | 25 | - | 9 |
| Nifedipine | 22 | 12 | - | - |
| Nitrendipine | 21 | 19 | - | - |
| Perindopril | - | 15 | - | 10 |
| Perindopril+indapamide | - | - | - | 2 |
| Ramipril | 23 | 2 | - | 4 |
| Spironolactone | 24 | 3 | - | - |
| Tangshen formula | 7 | 1 | - | - |
| Telmisartan | 20 | 9 | 3 | - |
| Telmisartan+lisinopril† | 17 | 22 | - | - |
| Telmisartan+TCMZ | - | 13 | - | - |
| Telmisartan+TCMZ+amlodipine | - | 29 | - | - |
| Trandolapril | 3 | - | - | - |
| Trandolapril+candesartan | 2 | - | - | - |
| Valsartan | 11 | - | - | 12 |
| Valsartan+amlodipine+canrenone | - | - | - | - |
| Valsartan+amlodipine+HCTZ | - | - | - | - |
*Placebo was ranked 16th in the overall analysis, was ranked 30th in the type 2 diabetes analysis, was ranked 9th in the aged 40+ analysis, and was ranked 16th in the 12+ months follow-up analysis.
†Lisinopril+telmisartan refers to lisinopril+telmisartan combination therapy following lisinopril monotherapy, while telmisartan+lisinopril refers to lisinopril+telmisartan combination therapy following telmisartan monotherapy.
Discussion
Here, employing a Bayesian network meta-analytical approach, we assessed the comparative effects of antihypertensive agents in reducing albuminuria in adult diabetic patients with microalbuminuric kidney disease and also specifically examined the comparative effects of antihypertensive agents in reducing albuminuria in normotensive versus hypertensive patients within this microalbuminuric cohort. We found that the ACEI-CCB combination therapy of captopril+diltiazem was the most efficacious intervention for reducing albuminuria irrespective of BP status. However, the ACEI-ARB combination therapy of trandolapril+candesartan was found to be the most efficacious intervention for reducing albuminuria for normotensive patients, while the ACEI-CCB combination therapy of fosinopril+amlodipine was found to be the most efficacious intervention for reducing albuminuria for hypertensive patients. However, the foregoing combination therapies displayed inferior safety profiles relative to ACEI monotherapy with respect to the key adverse side effects of dry cough, presyncope, and peripheral edema.
Palmer et al.’s previous network meta-analysis on adult diabetic patients with CKD identified four classes of antihypertensive agents that are able to significantly regress albuminuria in this patient population (either alone or in combination) [6]: (i) ACEIs (e.g., captopril, trandolapril, fosinopril) [15], (ii) ARBs (e.g., candesartan, losartan) [16], (iii) CCBs (e.g., diltiazem, amlodipine) [17], and (iv) diuretics (e.g., hydrochlorothiazide, trichlormethiazide) [18]. The consensus of clinical evidence has well-established that blockade of the renin-angiotensin-aldosterone system (RAAS) with either an ACEI or ARB agent reduces the risk of adverse renal events in adult diabetic patients with macroalbuminuria (daily albumin excretion of greater than 300 mg) [8]. As several previous clinical trials have found safety issues arising from ACEI-ARB combination therapy including hyperkalemia and acute kidney injury [19–21], the Eighth Joint National Committee (JNC 8) has recommended against ACEI-ARB combination therapy in these patients [22].
Although the majority of the evidence primarily concerns adult diabetic patients with macroalbumuria (daily albumin excretion of greater than 300 mg), there has been some guidance regarding antihypertensive therapy in adult diabetic patients with microalbuminuria (daily albumin excretion of 30–300 mg). The most recent Kidney Disease: Improving Global Outcomes (KDIGO) guidelines for managing diabetic CKD recommends either ACEI or ARB monotherapy for patients with microalbuminuria unless one of the following factors is present: ACEI or ARB therapy is contraindicated, metastatic cancer, treatment of malignancy in the past six months, or admission to a skilled nursing facility in the past three months [23]. The 2014 (Kidney Disease Outcomes Quality Initiative) KDOQI US commentary on these KDIGO guidelines also supports the use of either ACEI or ARB monotherapy based on evidence demonstrating increased harm with ACEI-ARB combination therapy [19, 24].
Interestingly, our current findings reveal that the ACEI-ARB combination therapy of trandolapril+candesartan appears the most efficacious for reducing albuminuria in normotensive adult diabetic patients with microalbuminuria, while the ACEI-CCB combination therapy of fosinopril+amlodipine appears to be the most efficacious intervention for reducing albuminuria for hypertensive adult diabetic patients with microalbuminuria. Therefore, our findings provide an important clarifying distinction to Palmer et al.’s network meta-analysis, which found that ACEI or ARB therapy combined with CCB therapy produces reductions in albuminuria in adult diabetic patients with kidney disease irrespective of BP or microalbuminuric status [6].
Moreover, our safety findings concord with the foregoing KDIGO and KDOQI guidelines regarding combination therapy, as we found that ACEI monotherapy produces a lower likelihood of key adverse outcomes relative to ACEI-ARB or ACEI-CCB combination therapy. For practitioners opting for monotherapy, our SUCRA analysis supports the use of trandolapril in normotensive adult diabetic patients with microalbuminuria and fosinopril in hypertensive adult diabetic patients with microalbuminuria.
With specific respect to type 2 diabetic patients with microalbuminuria, our sensitivity analysis revealed that the Chinese herbal medicine Tangshen formula followed by the ACEI ramipril were the most efficacious interventions for reducing albuminuria. Multiple clinical studies by Ping Li’s research group have demonstrated the efficacy of the Tangshen formula in attenuating diabetic kidney disease in rodent models and human patients [25]. Notably, a 2015 multicenter RCT in type 2 diabetic patients with diabetic kidney disease has also revealed that the Tangshen formula produces a significant decrease in 24-hour urinary protein as well as significant improvements in the estimated glomerular filtration rate (eGFR) relative to placebo [26]. In addition, our finding regarding the efficacy of ramipril ACEI monotherapy in type 2 diabetes patients with microalbuminuria concords with a previous network meta-analysis by Vejakama et al., which demonstrated a superior reno-protective effect with ACEI monotherapy or ARB monotherapy over CCB monotherapy or placebo in type 2 diabetic patients [27].
There are several limitations to this study. First, the three separate sensitivity analyses on type 2 diabetes status, age, and follow-up duration revealed that each of these three factors had a significant impact upon the findings. That being said, across all sensitivity analyses, ACEI monotherapy was found to be superior to all other antihypertensive medication classes (with the notable exception of the Tangshen formula for type 2 diabetes patients with microalbuminuria). Second, we could not assess the degree of albuminuria reduction attributable to a medication class effect beyond BP reduction. We considered including target/achieved BP as part of our analysis; however, the included RCTs did not report this data to a sufficient degree to enable such an analysis in the current study. Third, as the response to different medication classes may vary with excretory renal function, a subgroup analysis according to the presence or absence of reduced excretory renal function would provide valuable insights. Unfortunately, this analysis was not possible with the available data. Fourth, although we were able to assess the effects of various hypertensive agents upon regressing UAER, the available reported data in the included studies did not enable us to estimate the effects of antihypertensive agents on other clinically relevant renal outcomes. For example, we were unable to examine acute kidney injury as a safety endpoint due to the paucity of reported data on this outcome. Fifth, as most of the included RCTs were from developed Western countries and Japan, there is a scarcity of data from other countries, which may have biased our conclusions. Finally, drug dosing was not controlled for in the meta-analyses; most of the included RCTs allowed the clinical investigators to titrate drug dosing for participants.
In conclusion, the ACEI-ARB combination therapy of trandolapril+candesartan appears the most efficacious for reducing albuminuria in normotensive diabetic patients with microalbuminuria, while the ACEI-CCB combination therapy of fosinopril+amlodipine appears to be the most efficacious intervention for reducing albuminuria for hypertensive diabetic patients with microalbuminuria. However, the foregoing combination therapies displayed inferior safety profiles to ACEI monotherapy with respect to the key adverse side effects of dry cough, presyncope, and peripheral edema. For practitioners opting for monotherapy, our SUCRA analysis supports the use of trandolapril in normotensive adult diabetic patients with microalbuminuria and fosinopril in hypertensive adult diabetic patients with microalbuminuria.
Supporting Information
Networked interventions are placed in green boxes, while non-networked interventions are placed in grey boxes. Blue lines between interventions indicate direct comparisons with the number of studies indicated.
(TIF)
Networked interventions are placed in green boxes, while non-networked interventions are placed in grey boxes. Blue lines between interventions indicate direct comparisons with the number of studies indicated.
(TIF)
Networked interventions are placed in green boxes, while non-networked interventions are placed in grey boxes. Blue lines between interventions indicate direct comparisons with the number of studies indicated.
(TIF)
Networked interventions are placed in blue circles with the circle size reflecting the relative study size. Black lines between interventions indicate direct comparisons with thicker lines indicating a larger number of comparator studies.
(TIF)
Networked interventions are placed in blue circles with the circle size reflecting the relative study size. Black lines between interventions indicate direct comparisons with thicker lines indicating a larger number of comparator studies.
(TIF)
Networked interventions are placed in blue circles with the circle size reflecting the relative study size. Black lines between interventions indicate direct comparisons with thicker lines indicating a larger number of comparator studies.
(TIF)
Data Availability
This is a meta analysis paper and all data is available from online database.
Funding Statement
This work was supported by The Outstanding Young Talent Project of the Second Affiliated Hospital of Chongqing Medical University (grant no. 2014-43) and the Research Projects of the Health Bureau in Chongqing (grant no. 2010-2-153). This work was supported by the National Natural Science Foundation of China (grant nos. 31300137 and 81401423). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Guariguata L, Whiting D, Hambleton I, Beagley J, Linnenkamp U, Shaw J. Global estimates of diabetes prevalence for 2013 and projections for 2035. Diabetes research and clinical practice. 2014;103(2):137–49. 10.1016/j.diabres.2013.11.002 [DOI] [PubMed] [Google Scholar]
- 2.Hong C-S, Chung K-M, Huang P-C, Wang J-J, Yang C-M, Chu C-C, et al. Epidemiology and Mortality of Liver Abscess in End-Stage Renal Disease Dialysis Patients: Taiwan National Cohort Study. PloS one. 2014;9(2):e88078 10.1371/journal.pone.0088078 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Molitch ME, Adler AI, Flyvbjerg A, Nelson RG, So W-Y, Wanner C, et al. Diabetic kidney disease: a clinical update from Kidney Disease: Improving Global Outcomes. Kidney international. 2015;87(1):20–30. 10.1038/ki.2014.128 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bakris GL, Williams M, Dworkin L, Elliott WJ, Epstein M, Toto R, et al. Preserving renal function in adults with hypertension and diabetes: a consensus approach. American journal of kidney diseases. 2000;36(3):646–61. [DOI] [PubMed] [Google Scholar]
- 5.Sarafidis PA, Ruilope LM. Aggressive blood pressure reduction and renin–angiotensin system blockade in chronic kidney disease: time for re-evaluation&quest. Kidney international. 2014;85(3):536–46. 10.1038/ki.2013.355 [DOI] [PubMed] [Google Scholar]
- 6.Palmer SC, Mavridis D, Navarese E, Craig JC, Tonelli M, Salanti G, et al. Comparative efficacy and safety of blood pressure-lowering agents in adults with diabetes and kidney disease: a network meta-analysis. The Lancet. 2015;385(9982):2047–56. [DOI] [PubMed] [Google Scholar]
- 7.Bakris GL, Molitch M. Microalbuminuria as a risk predictor in diabetes: the continuing saga. Diabetes Care. 2014;37(3):867–75. 10.2337/dc13-1870 [DOI] [PubMed] [Google Scholar]
- 8.Tuttle KR, Bakris GL, Bilous RW, Chiang JL, de Boer IH, Goldstein-Fuchs J, et al. Diabetic kidney disease: a report from an ADA consensus conference. American Journal of Kidney Diseases. 2014;64(4):510–33. 10.1053/j.ajkd.2014.08.001 [DOI] [PubMed] [Google Scholar]
- 9.Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Annals of internal medicine. 2009;151(4):264–9. [DOI] [PubMed] [Google Scholar]
- 10.Jansen JP, Fleurence R, Devine B, Itzler R, Barrett A, Hawkins N, et al. Interpreting indirect treatment comparisons and network meta-analysis for health-care decision making: report of the ISPOR Task Force on Indirect Treatment Comparisons Good Research Practices: part 1. Value in Health. 2011;14(4):417–28. 10.1016/j.jval.2011.04.002 [DOI] [PubMed] [Google Scholar]
- 11.Hoaglin DC, Hawkins N, Jansen JP, Scott DA, Itzler R, Cappelleri JC, et al. Conducting indirect-treatment-comparison and network-meta-analysis studies: report of the ISPOR Task Force on Indirect Treatment Comparisons Good Research Practices: part 2. Value in Health. 2011;14(4):429–37. 10.1016/j.jval.2011.01.011 [DOI] [PubMed] [Google Scholar]
- 12.MacIsaac RJ, Jerums G. Diabetic kidney disease with and without albuminuria. Current opinion in nephrology and hypertension. 2011;20(3):246–57. 10.1097/MNH.0b013e3283456546 [DOI] [PubMed] [Google Scholar]
- 13.Li JZ, Winston LG, Moore DH, Bent S. Efficacy of short-course antibiotic regimens for community-acquired pneumonia: a meta-analysis. The American journal of medicine. 2007;120(9):783–90. 10.1016/j.amjmed.2007.04.023 [DOI] [PubMed] [Google Scholar]
- 14.Furukawa TA, McGuire H, Barbui C. Meta-analysis of effects and side effects of low dosage tricyclic antidepressants in depression: systematic review. Bmj. 2002;325(7371):991 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.White CM. Pharmacologic, pharmacokinetic, and therapeutic differences among ACE inhibitors. Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy. 1998;18(3):588–99. [PubMed] [Google Scholar]
- 16.Israili Z. Clinical pharmacokinetics of angiotensin II (AT1) receptor blockers in hypertension. Journal of Human Hypertension. 2000;14:S73–86. [DOI] [PubMed] [Google Scholar]
- 17.Leenen FH, Fourney A. Comparison of the effects of amlodipine and diltiazem on 24-hour blood pressure, plasma catecholamines, and left ventricular mass. The American journal of cardiology. 1996;78(2):203–7. [PubMed] [Google Scholar]
- 18.Hughes AD. How do thiazide and thiazide-like diuretics lower blood pressure? Journal of Renin-angiotensin-aldosterone system. 2004;5(4):155–60. [DOI] [PubMed] [Google Scholar]
- 19.Fried LF, Emanuele N, Zhang JH, Brophy M, Conner TA, Duckworth W, et al. Combined angiotensin inhibition for the treatment of diabetic nephropathy. New England Journal of Medicine. 2013;369(20):1892–903. 10.1056/NEJMoa1303154 [DOI] [PubMed] [Google Scholar]
- 20.Parving H-H, Brenner BM, McMurray JJ, de Zeeuw D, Haffner SM, Solomon SD, et al. Cardiorenal end points in a trial of aliskiren for type 2 diabetes. New England Journal of Medicine. 2012;367(23):2204–13. 10.1056/NEJMoa1208799 [DOI] [PubMed] [Google Scholar]
- 21.Mann JF, Schmieder RE, McQueen M, Dyal L, Schumacher H, Pogue J, et al. Renal outcomes with telmisartan, ramipril, or both, in people at high vascular risk (the ONTARGET study): a multicentre, randomised, double-blind, controlled trial. The Lancet. 2008;372(9638):547–53. [DOI] [PubMed] [Google Scholar]
- 22.James PA, Oparil S, Carter BL, Cushman WC, Dennison-Himmelfarb C, Handler J, et al. 2014 evidence-based guideline for the management of high blood pressure in adults: report from the panel members appointed to the Eighth Joint National Committee (JNC 8). Jama. 2014;311(5):507–20. 10.1001/jama.2013.284427 [DOI] [PubMed] [Google Scholar]
- 23.Qaseem A, Centor R, Dunn A, Baker DW, Centor RM, Cross JT, et al. Screening, Monitoring, and Management of Chronic Kidney Disease: Review of the Performance Measures by the Performance Measurement Committee of the American College of Physicians. 2014. [Google Scholar]
- 24.Inker LA, Astor BC, Fox CH, Isakova T, Lash JP, Peralta CA, et al. KDOQI US commentary on the 2012 KDIGO clinical practice guideline for the evaluation and management of CKD. American Journal of Kidney Diseases. 2014;63(5):713–35. 10.1053/j.ajkd.2014.01.416 [DOI] [PubMed] [Google Scholar]
- 25.Yang X, Zhang B, Lu X, Yan M, Wen Y, Zhao T, et al. Effects of Tangshen Formula on urinary and plasma liver-type fatty acid binding protein levels in patients with type 2 diabetic kidney disease: post-hoc findings from a multi-center, randomized, double-blind, placebo-controlled trial investigating the efficacy and safety of Tangshen Formula in patients with type 2 diabetic kidney disease. BMC Complementary and Alternative Medicine. 2016;16(1):246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Li P, Chen Y, Liu J, Hong J, Deng Y, Yang F, et al. Efficacy and safety of tangshen formula on patients with type 2 diabetic kidney disease: a multicenter double-blinded randomized placebo-controlled trial. PloS one. 2015;10(5):e0126027 10.1371/journal.pone.0126027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Vejakama P, Thakkinstian A, Lertrattananon D, Ingsathit A, Ngarmukos C, Attia J. Reno-protective effects of renin–angiotensin system blockade in type 2 diabetic patients: a systematic review and network meta-analysis. Diabetologia. 2012;55(3):566–78. 10.1007/s00125-011-2398-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Estacio RO, Coll JR, Tran ZV, Schrier RW. Effect of Intensive Blood Pressure Control With Valsartan on Urinary Albumin Excretion in Normotensive Patients With Type 2 Diabetes*. American journal of hypertension. 2006;19(12):1241–8. 10.1016/j.amjhyper.2006.05.011 [DOI] [PubMed] [Google Scholar]
- 29.Atmaca A, Gedik O. Effects of angiotensin-converting enzyme inhibitors, angiotensin II receptor blockers, and their combination on microalbuminuria in normotensive patients with type 2 diabetes. Advances in therapy. 2006;23(4):615–22. [DOI] [PubMed] [Google Scholar]
- 30.Bojestig M, Karlberg BE, Lindström T, Nystrom FH. Reduction of ACE activity is insufficient to decrease microalbuminuria in normotensive patients with type 1 diabetes. Diabetes Care. 2001;24(5):919–24. [DOI] [PubMed] [Google Scholar]
- 31.Mogensen CE, Neldam S, Tikkanen I, Oren S, Viskoper R, Watts RW, et al. Randomised controlled trial of dual blockade of renin-angiotensin system in patients with hypertension, microalbuminuria, and non-insulin dependent diabetes: the candesartan and lisinopril microalbuminuria (CALM) study. Bmj. 2000;321(7274):1440–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kojima M, Ohashi M, Dohi Y, Kimura G. Titration of telmisartan, but not addition of amlodipine, reduces urine albumin in diabetic patients treated with telmisartan–diuretic. Journal of hypertension. 2013;31(1):186–91. 10.1097/HJH.0b013e32835a2724 [DOI] [PubMed] [Google Scholar]
- 33.Chaturvedi N, Group ES. Randomised placebo-controlled trial of lisinopril in normotensive patients with insulin-dependent diabetes and normoalbuminuria or microalbuminuria. The Lancet. 1997;349(9068):1787–92. [PubMed] [Google Scholar]
- 34.Fogari R, Zoppi A, Malamani G, Lusardi P, Destro M, Corradi L. Effects of amlodipine vs enalapril on microalbuminuria in hypertensive patients with type II diabetes. Clinical drug investigation. 1997;13(1):42–9. [Google Scholar]
- 35.Fogari R, Zoppi A, Corradi L, Poletti L, Pasotti M, Fogari E, et al. Long-term effects of amlodipine versus fosinopril on microalbuminuria in elderly hypertensive patients with type 2 diabetes mellitus. Current therapeutic research. 2000;61(3):163–73. [Google Scholar]
- 36.Fogari R, Preti P, Zoppi A, Rinaldi A, Corradi L, Pasotti C, et al. Effects of amlodipine fosinopril combination on microalbuminuria in hypertensive type 2 diabetic patients. American journal of hypertension. 2002;15(12):1042–9. [DOI] [PubMed] [Google Scholar]
- 37.Fogari R, Mugellini A, Zoppi A, Lazzari P, Destro M, Rinaldi A, et al. Effect of successful hypertension control by manidipine or lisinopril on albuminuria and left ventricular mass in diabetic hypertensive patients with microalbuminuria. European journal of clinical pharmacology. 2005;61(7):483–90. 10.1007/s00228-005-0961-2 [DOI] [PubMed] [Google Scholar]
- 38.Fogari R, Corradi L, Zoppi A, Lazzari P, Mugellini A, Preti P, et al. Addition of manidipine improves the antiproteinuric effect of candesartan in hypertensive patients with type II diabetes and microalbuminuria. American journal of hypertension. 2007;20(10):1092–6. 10.1016/j.amjhyper.2007.05.012 [DOI] [PubMed] [Google Scholar]
- 39.Fogari R, Derosa G, Zoppi A, Lazzari P, D'Angelo A, Mugellini A. Comparative effect of canrenone or hydrochlorothiazide addition to valsartan/amlodipine combination on urinary albumin excretion in well-controlled type 2 diabetic hypertensive patients with microalbuminuria. Expert opinion on pharmacotherapy. 2014;15(4):453–9. 10.1517/14656566.2014.874415 [DOI] [PubMed] [Google Scholar]
- 40.Fogari R, Mugellini A, Zoppi A, Gualtierotti R, Lazzari P, Derosa G, et al. Effect of imidapril versus ramipril on urinary albumin excretion in hypertensive patients with type 2 diabetes and microalbuminuria. Expert opinion on pharmacotherapy. 2013;14(18):2463–73. 10.1517/14656566.2013.848195 [DOI] [PubMed] [Google Scholar]
- 41.Katayama S, Kikkawa R, Isogai S, Sasaki N, Matsuura N, Tajima N, et al. Effect of captopril or imidapril on the progression of diabetic nephropathy in Japanese with type 1 diabetes mellitus: a randomized controlled study (JAPAN-IDDM). Diabetes research and clinical practice. 2002;55(2):113–21. [DOI] [PubMed] [Google Scholar]
- 42.Jerums G, Allen TJ, Campbell DJ, Cooper ME, Gilbert RE, Hammond JJ, et al. Long-term comparison between perindopril and nifedipine in normotensive patients with type 1 diabetes and microalbuminuria. American journal of kidney diseases. 2001;37(5):890–9. [DOI] [PubMed] [Google Scholar]
- 43.Josefsberg Z, Ross SA, Lev-Ran A, Hwang DL. Effects of enalapril and nitrendipine on the excretion of epidermal growth factor and albumin in hypertensive NIDDM patients. Diabetes care. 1995;18(5):690–3. [DOI] [PubMed] [Google Scholar]
- 44.Kohlmann O Jr, Roca-Cusachs A, Laurent S, Schmieder RE, Wenzel RR, Fogari R. Fixed-dose manidipine/delapril versus losartan/hydrochlorothiazide in hypertensive patients with type 2 diabetes and microalbuminuria. Advances in therapy. 2009;26(3):313–24. 10.1007/s12325-009-0015-8 [DOI] [PubMed] [Google Scholar]
- 45.Lacourcière Y, Bélanger A, Godin C, Hallé J-P, Ross S, Wright N, et al. Long-term comparison of losartan and enalapril on kidney function in hypertensive type 2 diabetics with early nephropathy. Kidney international. 2000;58(2):762–9. 10.1046/j.1523-1755.2000.00224.x [DOI] [PubMed] [Google Scholar]
- 46.Li P, Chen Y, Liu J, Hong J, Deng Y, Yang F, et al. Efficacy and Safety of Tangshen Formula on Patients with Type 2 Diabetic Kidney Disease: A Multicenter Double-Blinded Randomized Placebo-Controlled Trial. 2015. 10.1371/journal.pone.0126027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Viberti G, Wheeldon NM. Microalbuminuria reduction with valsartan in patients with type 2 diabetes mellitus a blood pressure–independent effect. Circulation. 2002;106(6):672–8. [DOI] [PubMed] [Google Scholar]
- 48.Muirhead N, Feagan BF, Mahon J, Lewanczuk RZ, Rodger NW, Botteri F, et al. The effects of valsartan and captopril on reducing microalbuminuria in patients with type 2 diabetes mellitus: a placebo-controlled trial. Current therapeutic research. 1999;60(12):650–60. [Google Scholar]
- 49.Group MDNS. Comparison between perindopril and nifedipine in hypertensive and normotensive diabetic patients with microalbuminuria. BMJ. 1991;302(6770):210–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Nakamura T, Ushiyama C, Osada S, Takahashi Y, Shimada N, Ebihara I, et al. Combination therapy of trandolapril and candesartan cilexetil reduces microalbuminuria and urinary endothelin-1 excretion in patients with type 2 diabetes. Clinical and experimental nephrology. 2002;6(3):135–9. 10.1007/s101570200023 [DOI] [PubMed] [Google Scholar]
- 51.Pérez-Maraver M, Carrera MJ, Micaló T, Sahun M, Vinzia C, Soler J, et al. Renoprotective effect of diltiazem in hypertensive type 2 diabetic patients with persistent microalbuminuria despite ACE inhibitor treatment. Diabetes research and clinical practice. 2005;70(1):13–9. 10.1016/j.diabres.2005.02.019 [DOI] [PubMed] [Google Scholar]
- 52.Poulsen P, Ebbehøj E, Mogensen CE. Lisinopril reduces albuminuria during exercise in low grade microalbuminuric type 1 diabetic patients: a double blind randomized study. Journal of internal medicine. 2001;249(5):433–40. [DOI] [PubMed] [Google Scholar]
- 53.Mogensen CE, Viberti G, Halimi S, Ritz E, Ruilope L, Jermendy G, et al. Effect of low-dose perindopril/indapamide on albuminuria in diabetes preterax in albuminuria regression: PREMIER. Hypertension. 2003;41(5):1063–71. 10.1161/01.HYP.0000064943.51878.58 [DOI] [PubMed] [Google Scholar]
- 54.Sano T, Kawamura T, Matsumae H, Sasaki H, Nakayama M, Hara T, et al. Effects of long-term enalapril treatment on persistent microalbuminuria in well-controlled hypertensive and normotensive NIDDM patients. Diabetes Care. 1994;17(5):420–4. [DOI] [PubMed] [Google Scholar]
- 55.Sato A, Tabata M, Hayashi K, Saruta T. Effects of the angiotensin II type 1 receptor antagonist candesartan, compared with angiotensin-converting enzyme inhibitors, on the urinary excretion of albumin and type IV collagen in patients with diabetic nephropathy. Journal of Clinical and Experimental Nephrology. 2003;7(3):215–20. [DOI] [PubMed] [Google Scholar]
- 56.Schnack C, Capek M, Banyai M, Kautzky-Willer A, Prager R, Schernthaner G. Long-term treatment with nifedipine reduces urinary albumin excretion and glomerular filtration rate in normotensive type 1 diabetic patients with microalbuminuria. Acta diabetologica. 1994;31(1):14–8. [DOI] [PubMed] [Google Scholar]
- 57.Sengul AM, Altuntas Y, Kürklü A, Aydın L. Beneficial effect of lisinopril plus telmisartan in patients with type 2 diabetes, microalbuminuria and hypertension. Diabetes research and clinical practice. 2006;71(2):210–9. 10.1016/j.diabres.2005.06.010 [DOI] [PubMed] [Google Scholar]
- 58.SHIGIHARA T, SATO A, HAYASHI K, SARUTA T. Effect of Combination Therapy of Angiotensin-Converting Enzyme Inhibitor plus Calcium Channel Blocker on Urinary Albumin Excretion in Hypertensive Microalubuminuric Patients with Type II Diabetes. Hypertension Research. 2000;23(3):219–26. [DOI] [PubMed] [Google Scholar]
- 59.Takebayashi K, Matsumoto S, Aso Y, Inukai T. Aldosterone blockade attenuates urinary monocyte chemoattractant protein-1 and oxidative stress in patients with type 2 diabetes complicated by diabetic nephropathy. The Journal of Clinical Endocrinology & Metabolism. 2006;91(6):2214–7. [DOI] [PubMed] [Google Scholar]
- 60.Tan KC, Chow WS, Ai VH, Lam KS. Effects of angiotensin II receptor antagonist on endothelial vasomotor function and urinary albumin excretion in type 2 diabetic patients with microalbuminuria. Diabetes/metabolism research and reviews. 2002;18(1):71–6. [DOI] [PubMed] [Google Scholar]
- 61.Tütüncü N, Gürlek A, Gedik O. Efficacy of ACE inhibitors and ATII receptor blockers in patients with microalbuminuria: a prospective study. Acta diabetologica. 2001;38(4):157–61. [DOI] [PubMed] [Google Scholar]
- 62.Weil EJ, Fufaa G, Jones LI, Lovato T, Lemley KV, Hanson RL, et al. Effect of losartan on prevention and progression of early diabetic nephropathy in American Indians with type 2 diabetes. Diabetes. 2013;62(9):3224–31. 10.2337/db12-1512 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Viberti G, Mogensen CE, Groop LC, Pauls JF, Boner G, van Dyk D, et al. Effect of captopril on progression to clinical proteinuria in patients with insulin-dependent diabetes mellitus and microalbuminuria. Jama. 1994;271(4):275–9. [PubMed] [Google Scholar]
- 64.Zandbergen AA, Baggen MG, Lamberts SW, Bootsma AH, de Zeeuw D, Rob JT. Effect of losartan on microalbuminuria in normotensive patients with type 2 diabetes mellitus: a randomized clinical trial. Annals of Internal Medicine. 2003;139(2):90–6. [DOI] [PubMed] [Google Scholar]
- 65.Hozo SP, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC medical research methodology. 2005;5(1):1. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Networked interventions are placed in green boxes, while non-networked interventions are placed in grey boxes. Blue lines between interventions indicate direct comparisons with the number of studies indicated.
(TIF)
Networked interventions are placed in green boxes, while non-networked interventions are placed in grey boxes. Blue lines between interventions indicate direct comparisons with the number of studies indicated.
(TIF)
Networked interventions are placed in green boxes, while non-networked interventions are placed in grey boxes. Blue lines between interventions indicate direct comparisons with the number of studies indicated.
(TIF)
Networked interventions are placed in blue circles with the circle size reflecting the relative study size. Black lines between interventions indicate direct comparisons with thicker lines indicating a larger number of comparator studies.
(TIF)
Networked interventions are placed in blue circles with the circle size reflecting the relative study size. Black lines between interventions indicate direct comparisons with thicker lines indicating a larger number of comparator studies.
(TIF)
Networked interventions are placed in blue circles with the circle size reflecting the relative study size. Black lines between interventions indicate direct comparisons with thicker lines indicating a larger number of comparator studies.
(TIF)
Data Availability Statement
This is a meta analysis paper and all data is available from online database.