Abstract
Noroviruses (previously Norwalk-like viruses) are the most common viral agents associated with food- and waterborne outbreaks of gastroenteritis. In the absence of culture methods for noroviruses, animal caliciviruses were used as model viruses to study inactivation by nonionizing (253.7-nm-wavelength [UV]) and ionizing (gamma) radiation. Here, we studied the respiratory feline calicivirus (FeCV) and the presumed enteric canine calicivirus (CaCV) and compared them with the well-studied bacteriophage MS2. When UV irradiation was used, a 3-log10 reduction was observed at a fluence of 120 J/m2 in the FeCV suspension and at a fluence of 200 J/m2 for CaCV; for the more resistant phage MS2 there was a 3-log10 reduction at a fluence of 650 J/m2. Few or no differences were observed between levels of UV inactivation in high- and low-protein-content virus stocks. In contrast, ionizing radiation could readily inactivate MS2 in water, and there was a 3-log10 reduction at a dose of 100 Gy, although this did not occur when the phage was diluted in high-protein-content stocks of CaCV or FeCV. The low-protein-content stocks showed 3-log10 reductions at a dose of 500 Gy for FeCV and at a dose of 300 for CaCV. The inactivation rates for both caliciviruses with ionizing and nonionizing radiation were comparable but different from the inactivation rates for MS2. Although most FeCV and CaCV characteristics, such as overall particle and genome size and structure, are similar, the capsid sequences differ significantly, making it difficult to predict human norovirus inactivation. Adequate management of UV and gamma radiation processes for virus inactivation should limit public health risks.
Human caliciviruses are the most important enteric agents of gastroenteritis in industrialized countries (13, 17). Many waterborne outbreaks associated with noroviruses have been reported (1, 3, 5, 10, 15). Usually, epidemiological data prove that water was the common source for the outbreak, and in some molecular tracing studies norovirus strains were successfully identified in the source water and identical sequences were found in stools from patients (4, 8, 11, 12, 16). Virus concentrations in source water may peak with sewage overflow due to heavy rainfall or leaking septic tanks or pipes (2, 20). Breakdown or malfunctioning of water treatment plants may lead to insufficient reductions in virus concentrations in drinking water (20). Moreover, European legislation is mainly based on bacteriological quality parameters that determine fecal contamination of food and water. Viruses, however, are more resistant to treatment (9), are generally harder to detect, and exhibit lower dose-response relationships than bacteria. Therefore, water may meet microbiological safety guidelines but still pose a considerable health hazard.
Irradiation treatment is being used more frequently for disinfection of food and water. Bacteriophages have been used as indicators for inactivation of viruses by ionizing and nonionizing radiation since bacteria are generally less resistant to UV than viruses. Since noroviruses cannot be cultured (6), it is difficult to assess their resistance to ionizing and nonionizing radiation. The rate of inactivation by radiation has to be studied by using molecular methods or model viruses. Detection by PCR underestimates the efficiency of radiation for inactivating viruses (23). In this study we determined the inactivation of two cultivable animal caliciviruses, feline calicivirus (FeCV) strain F9 and canine calicivirus (CaCV) strain no. 48, which have highly divergent capsid sequences, and compared the results with inactivation of bacteriophage MS2. In addition to inactivation by nonionizing (253.7-nm-wavelength [UV]) radiation, inactivation by ionizing (gamma) radiation was studied as a process for calicivirus reduction. Virus stocks with high and low protein contents were compared to establish the possible effect of stabilizing solute constituents on inactivation by radiation.
MATERIALS AND METHODS
Viruses and cells.
Crandell Reese feline kidney (CRFK) cells and Madin-Darby canine kidney (MDCK) cells were cultured in Dulbecco's minimal essential medium supplemented with 10% (vol/vol) heat-inactivated fetal calf serum, gentamicin (50 μg/ml), and 1% (vol/vol) nonessential amino acids (Invitrogen, Paisley, Scotland). The medium used for virus propagation was identical except that it contained a low serum concentration (0 to 2%, vol/vol). FeCV strain F9 (kindly provided by H. Egberink, Utrecht University, Utrecht, The Netherlands) was propagated in monolayers of CRFK cells. CaCV strain no. 48 (kindly provided by F. Roerink, Kyoritsu Shoji Corporation, Tokyo, Japan) was propagated in MDCK cells (19). Virus stocks used in the inactivation experiments were obtained by inoculation of young cells (within 36 h after seeding) at a multiplicity of infection of 0.1 50% tissue culture infectious dose (TCID50) per cell. The suspensions were harvested after 24 h by one cycle of freezing and thawing and were clarified by centrifugation (10 min, 1,800 × g). The clarified stocks were diluted 10-fold in tap water. Stocks FeCV1 and CaCV1 contained 1.3 × 106 and 1.6 ×106 TCID50 of FeCV and CaCV, respectively, and the protein contents were 3 and 4 μg/ml, respectively, as measured by the Bio-Rad protein assay with bovine serum albumin as the standard. The 253.7-nm transmittance measured over 1 cm was 68 and 57% of the Viennese tap water transmittance for the FeCV1 and CaCV1 stocks, respectively.
Further purification was done by extraction with chloroform (10%, vol/vol) and subsequent pelleting of the virus by centrifugation (95,000 × g, 3 h), resuspension in Dulbecco's minimal essential medium, and 1,000-fold dilution in tap water. This resulted in stocks FeCV2 and CaCV2. Clarified stocks FeCV2 and CaCV2 contained 2.5 × 105 and 1.6 ×105 TCID50 of FeCV and CaCV, respectively. The protein contents of stocks FeCV2 and CaCV2 were below the detection limit of the Bio-Rad protein assay (<0.2 μg/ml), with 100% 253.7-nm transmittance relative to tap water transmittance measured over 1 cm. The titers of infectious virus were determined by determining TCID50 in cell cultures (10× dilutions in the 96-well format).
Phages.
F-specific RNA bacteriophages were detected by using ISO 10705-1. Briefly, a (diluted) sample was mixed with 2.5 ml of semisolid nutrient medium (tryptone-yeast extract-glucose agar). A culture of the host strain (Salmonella enterica serovar Typhimurium WG49) was added and plated on a solid nutrient medium (tryptone-yeast extract-glucose agar).The plates were incubated at 37°C for 12 to 18 h, and the visible plaques were counted. MS2 stocks were added to the animal calicivirus stocks to establish phage stocks with low and high protein contents.
Radiation experiments.
Experiments were performed at room temperature. The main chemical characteristics of the tap water were pH 7.6, a conductivity of 285 μS/cm2, 3.0 mg of KMnO4 demand per liter, 0.8 mg of total organic carbon per liter, 1.7 mg of chloride per liter, 4.6 mg of nitrate per liter, 8.0 mg of oxygen per liter, 9.0 mg of magnesium per liter, 11.0 mg of sulfate per liter, 44.0 mg of calcium per liter, 159.0 mg of hydrogen carbonate per liter, and no detectable ammonia (<0.02 mg/liter) or nitrite (<0.01 mg/liter). Linear regression analyses were carried out for comparisons of virus inactivation rates.
(i) UV irradiation.
A board on which 10 low-pressure mercury UV lamps (EK 36; length, 500 mm; wavelength, 253.7 nm; ozone free; Katadyn, Wallisellen, Switzerland) were mounted was horizontally suspended over the irradiation vessel. An aperture (100 by 150 mm) was located directly below the UV lamps and provided a bundle of quasi-parallel radiation to the irradiation vessel. The fluence was about 1 J/m2. The UV fluence at 253.7 nm was measured online with a research radiometer (IL 1700 SED 240; International Light), taking into consideration the absorption at 253.7 nm of the test suspension and the reflection at the surface. Experiments were performed at room temperature with continuous stirring.
(ii) Gamma irradiation.
The gamma irradiation experiments were carried out by using an AECL gammacell 220 cobalt 60 source (AECL, Mississauga, Canada) with an average dose rate of 0.96 Gy/s during the experiment. Samples (15 ml) were irradiated in glass vials. Dose rate calibrations have been performed with different chemical dosimeters (7).
Statistics.
Linear regression analyses were performed with each data set obtained with each type of irradiation for inactivation of each virus in each suspension.
RESULTS
Inactivation by UV irradiation.
The levels of inactivation of bacteriophage MS2 and the animal caliciviruses FeCV and CaCV by UV (253.7-nm) radiation were compared in tap water or calicivirus stocks (Fig. 1). A 3-log10 reduction at a fluence of 650 J/m2 was observed for MS2 in tap water (slope, −0.0045; r2 = 95.6%). Dilution of MS2 in the high-protein-content FeCV1 stock (Fig. 2a) (slope, −0.0060; r2 = 97.9%) did not result in a difference in the inactivation of MS2 compared with the inactivation of the low-protein-content FeCV2 stock (slope, −0.0065; r2 = 98.7%) or the high-protein content (slope, −0.0063; r2 = 98.9%) or low-protein-content (slope, −0.0065; r2 = 99.3%) CaCV stock (Fig. 2b). The FeCV data revealed a 3-log10 reduction at a fluence of 120 J/m2 for both the FeCV1 stock (high protein content) and the FeCV2 stock (low protein content), with slopes of −0.013 and −0.020 and r2 values of 90.0 and 82.0%, respectively. For CaCV a 3-log10 reduction required 200 J/m2 in either of the two stocks (high-protein-content CaCV1 stock [slope, −0.013; r2 = 94.8%] and low-protein-content CaCV2 stock [slope, −0.013; r2 = 92.5%]).
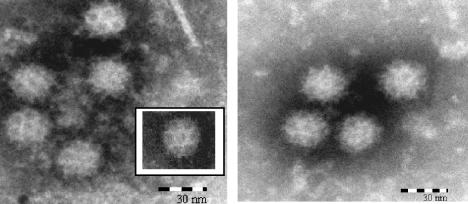
FIG. 1.
Electron micrographs of negatively stained FeCV and CaCV. (Left panel) FeCV in culture suspension of CRFK cells infected with FeCV strain F9. (Inset) Virus from the same suspension displaying characteristic Caliciviridae morphology. (Right panel) CaCV in culture suspension of MDCK cells infected with CaCV strain no. 48.
FIG. 2.
UV (253.7-nm) inactivation of bacteriophage MS2 in tap water (×), of MS2 in a calicivirus suspension (open symbols), or of CaCV (▪) or FeCV (▴) suspensions with high protein contents (a) or low protein contents (b). The regression lines for MS2 are dashed; the solid lines are regression lines for the animal caliciviruses.
Inactivation by gamma irradiation.
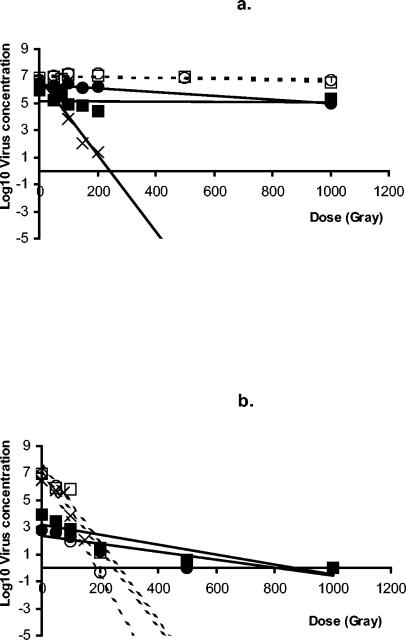
Experiments in which the inactivation of viruses with ionizing irradiation was examined showed that MS2 could be readily inactivated in tap water (slope, −0.028; r2 = 93.7%) and in low-protein-content stocks (FeCV2 [slope, −0.039; r2 = 92.6%] and CaCV2 [slope, −0.028; r2 = 89.3%]) (Fig. 3a). A 3-log10 reduction was observed at a gamma radiation dose of approximately 120 Gy. No inactivation was found when MS2 was irradiated by gamma rays in high-protein-content stocks (FeCV1 [slope, −0.00018; r2 = 8.6%] and CaCV1 [slope, −0.00041; r2 = 57.1%]) (Fig. 3b). As expected, little reduction in the FeCV and CaCV infectivity was observed in the high-protein-content stocks either (FeCV1 [slope, −0.0014; r2 = 85.4%] and CaCV1 [slope, −0.00010; r2 = 0.4%]). Both CaCV and FeCV were inactivated less efficiently (P = 0.03 and P = 0.01) than MS2 in the low-protein-content stocks (FeCV2 [slope, −0.0029; r2 = 77.3%] and CaCV2 [slope, −0.037; r2 = 81.1%]). At a dose of 200 Gy the amount of MS2 was reduced 5 to 7 log10 units, whereas the amount of CaCV was reduced 2.4 log10 units and the amount of FeCV was reduced 1.6 log10 units. Moreover, the inactivation of the animal caliciviruses appeared to follow second-order kinetics, with faster inactivation occurring at doses between 0 and 400 Gy and slower inactivation occurring at doses from 400 to 800 Gy.
FIG. 3.
Inactivation by gamma radiation (cobalt 60) of bacteriophage MS2 in tap water (×), of MS2 in a calicivirus suspension (open symbols), or of CaCV (▪) or FeCV (▴) suspensions with high protein contents (a) or low protein contents (b). The regression lines for MS2 are dashed; the solid lines are regression lines for the animal caliciviruses.
DISCUSSION
Since noroviruses cannot be cultured (6), their resistance to ionizing and nonionizing radiation was assessed by using animal caliciviruses as a model. Here, we studied not only the respiratory virus FeCV strain F9 but also the presumed enteric virus CaCV strain no. 48 and compared them with the well-studied bacteriophage MS2 in tap water.
Inactivation by UV irradiation.
Similar reductions were observed for FeCV and CaCV, although inactivation of MS2 required a UV dose that was three times higher. Previously, Nuanualsuwan et al. compared the levels of UV resistance of different viruses in phosphate-buffered saline (22). A reduction of 90% was obtained with UV fluences of 480, 370, 240, 230, and 160 J/m2 for FeCV, hepatitis A virus, poliovirus type 1, MS2, and φX174, respectively (22). Another recent study, however, showed that the resistance of MS2 was greater than that of FeCV in buffered demand-free water (90% reduction at fluences of 230 and 60 J/m2, respectively) and that the resistance of human adenovirus type 40 was even greater at a fluence of 500 J/m2 (25). The high level of resistance of bacteriophage MS2 to UV radiation has been reported by our groups (9, 24), as well as by other groups (18). The different results obtained in the studies on the efficiency of UV inactivation of FeCV may have resulted from differences in the FeCV strains used or from differences in the compositions of the suspensions. Little or no effect on UV inactivation was observed.
The virus characteristics, such as size, genome, and weight, are very similar for the animal and human caliciviruses (Table 1). FeCV strain F9 and CaCV strain no. 48 are even more closely related, clustering within the vesivirus genogroup (14). However, the capsid sequences are very different, with only 37% amino acid homology. In comparison, in the capsid region there is 85% amino acid homology between different FeCV strains and 16 and 17% homology between Norwalk virus and CaCV and FeCV, respectively. Recently, it was shown that the viral capsid is the primary target for UV inactivation by capsid epitope binding experiments (21). We found that the very high diversity in capsid sequences of FeCV and CaCV did not result in profound differences in sensitivity to UV radiation. This suggests that these animal caliciviruses might indeed be a valuable model for inactivation experiments.
TABLE 1.
Characteristics of caliciviruses
| Virus | Nucleic acid | Length of RNA (kb) | Capsid protein
|
Buoyant density in CsCl (g/cm3) | UV fluence (J/m2)a | Gamma dose (Gy)a | Size (nm) | |
|---|---|---|---|---|---|---|---|---|
| No. of amino acids | Molecular mass (kDa) | |||||||
| FeCV | Single-stranded RNA | 7.7 | 668-671 | 62 | 1.33 | 120 | 500 | 30 |
| CaCV | Single-stranded RNA | 8.5 | 691 | 57 | 1.38 | 200 | 300 | 30 |
| Norovirus | Single-stranded RNA | 7.6-7.7 | 530-580 | 56-58 | 1.36-1.41 | Unknown | Unknown | 30-35 |
Values for a 3-log10 reduction in caliciviruses in low-protein-content stocks.
Inactivation by gamma irradiation.
Ionizing radiation is being used more frequently as a useful and effective means of disinfection. The mechanism of virus inactivation is mainly based on the reaction of OH free radicals with the nucleic acid strands, and the virus coat may also play a role. As previously described, gamma radiation of viruses is less effective in the presence of solutes which react with OH free radicals, the so-called scavengers (24). Therefore, we studied stocks with defined high or low protein contents. MS2 dispersed in the high-protein-content stocks was not inactivated, whereas it was very efficiently inactivated in the low-protein-content stocks and in tap water. Further purification of the FeCV and CaCV stocks that resulted in lower protein, sugar, and fat contents also resulted in a higher rate of inactivation by gamma radiation. These results show the importance of the reaction with OH free radicals in virus inactivation by gamma radiation. The major effects of solutes (for example, proteins) on the effectiveness of gamma radiation for inactivation of viruses raises questions concerning the application of this method for production of virus-safe foods.
The tap water and low-protein-content stocks contained few scavengers, in contrast to the high-protein-content stocks. In highly scavenged systems, gamma radiation may be regarded as the main cause of damage to the genomic material. MS2 and both animal caliciviruses showed high levels of resistance to gamma radiation in the high-protein-content stocks. These results are in agreement with the fact that these are all single-stranded RNA viruses. In addition, we observed that the resistance of FeCV to gamma radiation was somewhat greater than the resistance of CaCV, which may be explained, at least in part, by differences in virus capsid sequences. In light of the reported enteric nature and respiratory nature of CaCV and FeCV, respectively, this result was unexpected since in general, enteric viruses are considered to be more resistant than respiratory viruses belonging to the same family.
Virus reduction by radiation.
Quantitative risk assessment may be a useful tool for protection of public health by exposure to pathogenic microorganisms in water and food. The virus concentration in drinking water is so low that it cannot be measured directly; it can be determined only by measuring the virus concentration in the source water and the subsequent reduction via treatment processes that are applied to reduce the source water virus concentration. Here, we show that two cultivable caliciviruses, which are very comparable in terms of their virus characteristics but differ in their capsid amino acid sequences, display comparable sensitivities to radiation. However, the resistance of CaCV and FeCV differed significantly from that observed for MS2 for inactivation by UV radiation (P = 0.0003 and P = 0.01, respectively) and gamma radiation (P = 0.01 and P = 0.03, respectively). Moreover, CaCV is significantly more resistant to UV inactivation than FeCV (P = 0.008), whereas FeCV shows significantly greater resistance to gamma radiation (P = 0.002). The threefold-higher reduction in the level of MS2 compared with the animal caliciviruses could make MS2 a very conservative indicator for UV inactivation of caliciviruses. Pending a culture method for human caliciviruses, we suggest that the most resistant calicivirus should be used as a surrogate to assess inactivation by radiation, which is different for different types of radiation. Human caliciviruses are more divergent from the feline and canine vesiviruses; however, even though there were significant differences between the inactivation profiles of CaCV and FeCV, we found that in contrast to MS2, the rates of inactivation of the animal caliciviruses after irradiation were the same order of magnitude. Extrapolation from animal to human caliciviruses may be justified. More importantly, the efficiency of gamma rays is hampered by scavengers in high-protein-content substances, as we show here. The gamma radiation doses of 2 to 4 kGy often used for bacterial control in food products may not be sufficient to reduce calicivirus infectivity, but this should be studied. We have shown that scavengers present no problems for the use of the ionizing radiation used to produce water fit for consumption. Adequate management of radiation processes in the food and water industry will be the only way to ensure protection of public health.
Acknowledgments
This work was funded by the Ministry of Housing, Physical Planning and the Environment under project 289202 and by the Environmental Inspectorate under project 330000, Water Microbiology.
We gratefully acknowledge the help of Harry Vennema with the comparison of the calicivirus capsid sequences.
REFERENCES
- 1.Baron, R. C., F. D. Murphy, H. B. Greenberg, C. E. Davis, D. J. Bregman, G. W. Gary, J. M. Hughes, and L. B. Schonberger. 1982. Norwalk gastrointestinal illness: an outbreak associated with swimming in a recreational lake and secondary person-to-person transmission. Am. J. Epidemiol. 115:163-172. [DOI] [PubMed] [Google Scholar]
- 2.Beller, M., A. Ellis, S. H. Lee, M. A. Drebot, S. A. Jenkerson, E. Funk, M. D. Sobsey, O. D. Simmons III, S. S. Monroe, T. Ando, J. Noel, M. Petric, J. P. Middaugh, and J. S. Spika. 1997. Outbreak of viral gastro-enteritis due to a contaminated well. JAMA 278:563-568. [PubMed] [Google Scholar]
- 3.Boccia, D., A. E. Tozzi, B. Cotter, C. Rizzo, T. Russo, G. Buttinelli, A. Caprioli, M. L. Marziano, and F. M. Ruggeri. 2002. Waterborne outbreak of Norwalk-like virus gastro-enteritis at a tourist resort, Italy. Emerg. Infect. Dis. 8:563-568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brown, C. M., J. W. Cann, G. Simons, R. L. Fankhauser, W. Thomas, U. D. Parashar, and M. J. Lewis. 2001. Outbreak of Norwalk virus in a Caribbean island resort: application of molecular diagnostics to ascertain the vehicle of infection. Epidemiol. Infect. 126:425-432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Brugha, R., I. B. Vipond, M. R. Evans, Q. D. Sandifer, R. J. Roberts, R. L. Salmon, E. O. Caul, and A. K. Mukerjee. 1999. A community outbreak of food-borne small round-structured virus gastro-enteritis caused by a contaminated water supply. Epidemiol. Infect. 122:145-154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Duizer, E., K. J. Schwab, F. H. Neill, R. L. Atmar, M. P. G. Koopmans, and M. K. Estes. 2004. Laboratory efforts to cultivate noroviruses. J. Gen. Virol. 85:79-87. [DOI] [PubMed] [Google Scholar]
- 7.Gehringer, P., and H. Eschweiler. 1998. Radiation-induced clean up of water and wastewater, p. 325-340. In W. J. Cooper, R. D. Curry, and K. E. O’Shea (ed.), Environmental applications of ionizing radiation. Wiley, New York, N.Y.
- 8.Hafliger, D., P. Hubner, and J. Luthy. 2000. Outbreak of viral gastro-enteritis due to sewage-contaminated drinking water. Int. J. Food Microbiol. 54:123-126. [DOI] [PubMed] [Google Scholar]
- 9.Havelaar, A. H., T. J. Nieuwstad, C. C. E. Meulemans, and M. van Olphen. 1991. F-specific RNA bacteriophages as model viruses in UV disinfection of wastewater. Water Sci. Technol. 24:347-352. [Google Scholar]
- 10.Hejkal, T. W., B. H. Keswick, R. L. LaBelle, C. P. Gerba, Y. Sanchez, G. Dreesman, B. Hafkin, and J. L. Melnick. 1982. Viruses in a community water supply associated with an outbreak of gastroenteritis and infectious hepatitis. J. Am. Water Works Assoc. 74:318-321. [Google Scholar]
- 11.Hoebe, C. J. P. A., H. Vennema, A. M. de Roda Husman, and Y. T. H. P. van Duynhoven. 2004. Norovirus outbreak among primary school children who had played in a recreational water fountain. J. Infect. Dis. 189:699-705. [DOI] [PubMed] [Google Scholar]
- 12.Kukkula, M., L. Maunula, E. Silvennoinen, and C.-H. von Bonsdorff. 1999. Outbreak of viral gastroenteritis due to drinking water contaminated by Norwalk-like viruses. J. Infect. Dis. 180:1771-1776. [DOI] [PubMed] [Google Scholar]
- 13.Lopman, B. A., D. W. Brown, and M. Koopmans. 2002. Human caliciviruses in Europe. J. Clin. Virol. 24:137-160. [DOI] [PubMed] [Google Scholar]
- 14.Matsuura, Y., Y. Tohya, K. Nakamura, M. Shimojima, F. Roerink, M. Mochizuki, K. Takase, H. Akashi, and T. Sugimura. 2002. Complete nucleotide sequence, genome organization and phylogenetic analysis of the canine calicivirus. Virus Genes 25:67-73. [DOI] [PubMed] [Google Scholar]
- 15.Maurer, A. M., and D. Sturchler. 2000. A waterborne outbreak of small round structured virus, campylobacter and shigella co-infections in La Neuville, Switzerland, 1998. Epidemiol. Infect. 125:325-332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.McCarthy, N., B. de Jong, T. Ziese, R. Sjolund, C. A. Hjalt, and J. Giesecke. 1998. Epidemiological investigation of an outbreak of gastroenteritis in Sweden in the absence of detailed microbiological information. Eur. J. Epidemiol. 14:711-718. [DOI] [PubMed] [Google Scholar]
- 17.Mead, P. S., L. Slutsker, V. Dietz, L. F. McCaig, J. S. Bresee, C. Shapiro, P. M. Griffin, and R. V. Tauxe. 1999. Food-related illness and death in the United States. Emerg. Infect. Dis. 5:607-625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Meng, S., and C. P. Gerba. 1996. Comparative inactivation of enteric adenoviruses, poliovirus and coliphages by ultraviolet irradiation. Water Res. 30:2665-2668. [Google Scholar]
- 19.Mochizuki, M., M. Hashimoto, F. Roerink, Y. Tohya, Y. Matsuura, and N. Sasaki. 2002. Molecular and seroepidemiological evidence of canine calicivirus infections in Japan. J. Clin. Microbiol. 40:2629-2631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Morens, D. M., R. M. Zweighaft, T. M. Vernon, G. W. Gary, J. J. Eslien, B. T. Wood, R. C. Holman, and R. Dolin. 1979. A waterborne outbreak of gastroenteritis with secondary person-to-person spread. Lancet i:964-966. [DOI] [PubMed] [Google Scholar]
- 21.Nuanualsuwan, S., and D. O. Cliver. 2003. Capsid functions of inactivated human picornaviruses and feline calicivirus. Appl. Environ. Microbiol. 69:350-357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nuanualsuwan, S., T. Mariam, S. Himathongkham, and D. O. Cliver. 2002. Ultraviolet inactivation of feline calicivirus, human enteric viruses and coliphages. Photochem. Photobiol. 76:406-410. [DOI] [PubMed] [Google Scholar]
- 23.Richards, G. P. 1999. Limitations of molecular biological techniques for assessing the virological safety of foods. J. Food Prot. 62:691-697. [DOI] [PubMed] [Google Scholar]
- 24.Sommer, R., W. Pribil, S. Appelt, P. Gehringer, H. Eschweiler, H. Leth, A. Cabaj, and T. Haider. 2001. Inactivation of bacteriophages in water by means of non-ionizing (UV-253.7 nm) and ionizing (gamma) radiation: a comparative approach. Water Res. 35:3109-3116. [DOI] [PubMed] [Google Scholar]
- 25.Thurston-Enriquez, J. A., C. N. Haas, J. Jacangelo, K. Riley, and C. P. Gerba. 2003. Inactivation of feline calicivirus and adenovirus type 40 by UV radiation. Appl. Environ. Microbiol. 69:577-582. [DOI] [PMC free article] [PubMed] [Google Scholar]