Abstract
Flowering in Arabidopsis thaliana is controlled by multiple pathways, including the photoperiod pathway and the FLOWERING LOCUS C (FLC)-dependent pathway. Here, we report that a pair of related jumonji-class transcription factors, EARLY FLOWERING 6 (ELF6) and RELATIVE OF EARLY FLOWERING 6 (REF6), play divergent roles in the regulation of Arabidopsis flowering. ELF6 acts as a repressor in the photoperiod pathway, whereas REF6, which has the highest similarity to ELF6, is an FLC repressor. Ectopic expression studies and expression pattern analyses show that ELF6 and REF6 have different cellular roles and are also regulated differentially despite their sequence similarities. Repression of FLC expression by REF6 accompanies histone modifications in FLC chromatin, indicating that the transcriptional regulatory activity of this class of proteins includes chromatin remodeling. This report demonstrates the in vivo functions of this class of proteins in higher eukaryotes.
INTRODUCTION
Flowering is a major developmental switch in plants and involves a transition from vegetative to reproductive growth. Because reproductive success of plants could be significantly affected by flowering time, each plant species has developed optimum strategies for its flowering time regulation.
Molecular genetic studies have identified four major floral regulatory pathways in Arabidopsis thaliana (for reviews, see Mouradov et al., 2002; Simpson and Dean, 2002). Two of these pathways that are involved in the interpretation of environmental cues are the photoperiod and vernalization pathways. The photoperiod pathway regulates flowering in response to daylength. In Arabidopsis, long days promote flowering (Koornneef et al., 1998). Thus, mutations in photoperiod pathway genes (e.g., gigantea [gi], constans [co], fd, fe, cryptochrome 2/fha [cry2], flowering locus t [ft], and fwa) blind Arabidopsis such that it does not recognize long days as an inductive photoperiod, resulting in delayed flowering in long days but not in short days. GI and CRY2 act upstream of CO, which mediates between the circadian clock and the control of flowering time (Suarez-Lopez et al., 2001). FT is an early target gene of CO (Samach et al., 2000), and FT mRNA levels correlate with CO protein levels (Valverde et al., 2004).
Vernalization confers competence to flowers after an extended cold treatment, thus preventing premature flowering in the fall (Michaels and Amasino, 2000; Henderson et al., 2003). The winter-annual habit is conferred by dominant alleles of FRIGIDA (FRI) and FLOWERING LOCUS C (FLC). FLC is a MADS box transcription factor that represses flowering (Michaels and Amasino, 1999; Sheldon et al., 1999). FLC expression is increased to levels that delay flowering by FRI, a novel protein with two coiled-coil domains (Michaels and Amasino, 1999; Sheldon et al., 1999; Johanson et al., 2000). A large portion of the promotion of flowering by vernalization is achieved through the downregulation of FLC mRNA levels. However, some portion of the vernalization effect is also achieved through FLC-independent pathways as evidenced from the vernalization responsiveness of an flc null mutant (Michaels and Amasino, 2001).
A major role of the autonomous pathway is to repress FLC expression. Arabidopsis plants with mutations in autonomous- pathway genes (fca, fld, fpa, fve, fy, and ld) have elevated levels of FLC expression and mimic the late-flowering behavior of FRI-containing accessions (Michaels and Amasino, 1999, 2001; Sheldon et al., 1999). Like FRI-containing accessions, the elevated expression of FLC and the late flowering of the autonomous-pathway mutants are rapidly repressed by vernalization. Unlike photoperiod pathway late-flowering mutants, autonomous-pathway mutants are delayed in flowering under all daylengths but still respond to photoperiod by flowering earlier in long days than in short days (e.g., Mouradov et al., 2002; Simpson and Dean, 2002).
The gibberellin (GA) pathway acts as a default floral promotion pathway in noninductive short days. Mutations that reduce GA levels or GA perception (e.g., ga1 or gai) cause an extreme delay in flowering in short days but have a minor effect in long days (Wilson et al., 1992). GA-induced floral promotion is mediated by LEAFY (LFY) and SUPPRESSOR OF OVEREXPRESSION OF CO 1 (SOC1) (Blázquez and Weigel, 2000; Moon et al., 2003).
Floral regulatory signals generated in the photoperiod, FLC-dependent, and GA-dependent pathways are integrated by a group of genes called floral integrators. So far, three genes that function as floral integrators have been identified: FT, SOC1, and LFY (reviewed in Simpson and Dean, 2002). FT and SOC1 are able to integrate floral regulatory signals generated in the photoperiod and FLC-dependent pathways. SOC1 is also involved in the integration of GA-dependent floral promotion signals (Moon et al., 2003). LFY integrates photoperiodic and GA-dependent signals through discrete cis elements in the promoter (Blázquez and Weigel, 2000).
Current understanding of the floral regulation pathways mentioned above has largely come from studies on mutations that cause late flowering (Mouradov et al., 2002; Simpson and Dean, 2002). However, many mutations that cause early flowering have also been described (for review, see Sung et al., 2003). Many of these mutations have been placed in the defined floral regulatory pathways, whereas others have not. One class of early-flowering mutations (e.g., elf3, elf4, and toc1) affects the photoperiodic floral regulatory pathway by disrupting circadian rhythms. Another class of early-flowering mutations (e.g., elf5, pie1, esd4, and efs) causes a decrease in FLC expression, especially in genetic backgrounds in which FLC is highly expressed. However, these mutations also appear to affect FLC-independent floral regulation as the mutants flower earlier than an flc null mutant (Soppe et al., 1999; Reeves et al., 2002; Noh and Amasino, 2003; Noh et al., 2004).
In fact, many other early-flowering mutations do not fall into any of the defined floral regulatory pathways and appear to affect multiple flowering and developmental pathways (Sung et al., 2003). The genes affected by many of these mutations encode chromatin remodeling factors (e.g., LHP1/TFL2, EMF1, EMF2, FIE, CLF, and EBS) (Sung et al., 2003). Lesions in these genes often lead to the premature derepression of genes far downstream in floral regulatory pathways such as FT or floral meristem or floral organ identity genes such as AP1 and AG. The early flowering caused derepression of such downstream genes is expected to be epistatic to lesions that delay flowering in multiple flowering pathways.
Here, we report the identification of a pair of homologous genes, EARLY FLOWERING 6 (ELF6) and RELATIVE OF EARLY FLOWERING 6 (REF6) that are involved in the regulation of flowering time in Arabidopsis. ELF6 and REF6 encode nuclear proteins with jumonji (Jmj) and zinc-finger (ZnF) domains and are most homologous to each other. Despite their structural similarities, ELF6 and REF6 have divergent roles in floral regulations: ELF6 acts as an upstream repressor in the photoperiodic flowering pathway, whereas REF6 is an FLC repressor. Spatial expression patterns of ELF6 and REF6 are also different and reflect their roles as repressors of the photoperiodic and the FLC-dependent floral regulatory pathways, respectively. Repression of FLC mRNA expression by REF6 involves histone modifications in FLC chromatin, suggesting a role of Jmj-domain proteins as chromatin remodelers. Therefore, our results demonstrate the in vivo functions of Jmj-domain proteins in higher eukaryotes as well as the identification and characterization of novel floral regulators in Arabidopsis.
RESULTS
Mutations in ELF6 Cause Early Flowering, whereas Mutations in REF6 Cause Late Flowering
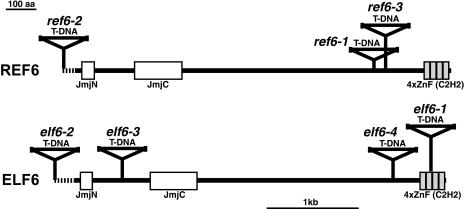
The elf6 mutant was identified in a screen for early-flowering mutants as described previously (Noh and Amasino, 2003; Noh et al., 2004). elf6-1 displayed an earlier floral transition compared with the wild-type Wassilewskija (Ws) in long days (LD; 16 h of light and 8 h of dark) as well as in noninductive short days (SD; 8 h of light and 16 h of dark; Figures 1A and 1C). The early-flowering phenotype of elf6-1 segregated in a recessive manner (data not shown). Except for the early-flowering phenotypes, elf6 mutants displayed normal development of all organs throughout the entire life cycle, suggesting that ELF6 might play a specific role in flowering time regulations. Because elf6-1 was isolated from a T-DNA insertion population, an effort to obtain the sequence flanking T-DNA insertion sites was made (see Methods) and resulted in the identification of a T-DNA insertion at the 3′ end of At5g04240 (Figure 2). To confirm that the loss of At5g04240 leads to the early-flowering phenotype of elf6, three more T-DNA insertion alleles of At5g04240 in the Columbia (Col) background (elf6-2, -3, and -4; Figure 2) were obtained and analyzed for flowering time. Homozygous mutants of all three Col alleles also showed an earlier floral transition compared with the wild-type Col both in LD and SD (Figure 1), confirming that At5g04240 is indeed ELF6. The difference in flowering time in LD between elf6 mutants and the wild type was not large but was statistically significant (Table 1).
Figure 1.
Early Flowering of elf6 and Late Flowering of ref6 Mutants Compared with the Wild Type.
(A) Wild-type (Ws) and elf6-1 plants grown for 49 d under SD.
(B) Wild-type (Col) and ref6-1 plants grown for 32 d under LD.
(C) Flowering time of elf6 mutants. Wild-type and elf6 mutants were grown under LD or SD, and their flowering times were measured as the number of rosette and cauline leaves formed by the primary meristem. Data shown are means ± sd of at least 12 plants for each genotype and treatment (see [C] and [D]).
(D) Flowering time of ref6 mutants. Flowering time was measured as described in (C). Col and ref6-1 mutants were treated with vernalization as previously described (Noh and Amasino, 2003) for 40 (+40D V) or 70 d (+70D V).
Figure 2.
Domains of REF6 and ELF6 and the T-DNA Insertion Sites in ref6 and elf6 Mutants.
Domains predicted by the SMART (http://smart.embl-heidelberg.de/) program are indicated. Lines indicate interdomain regions. T-DNA insertion sites on the genomic sequences of REF6 and ELF6 are marked on the corresponding positions of their translated protein products with their allele numbers. Intergenic or 5′ untranslated regions are indicated as dotted lines. aa, amino acids.
Table 1.
Student's t Test for the Flowering Time of elf6 and ref6 Mutants in LD
| Statistical Significance | Ws versus elf6-1 | Col versus elf6-2 | Col versus elf6-3 | Col versus elf6-4 | ref6-1 versus Col | ref6-3 versus Col |
|---|---|---|---|---|---|---|
| t Value | 4.44780 | 7.98659 | 7.52869 | 6.98178 | 9.10342 | 9.74121 |
| P Value | 0.00008 | 0.00000 | 0.00000 | 0.00000 | 0.00000 | 0.00000 |
Similarity searches using ELF6 against Arabidopsis genome databases showed that At3g48430 is the gene having the highest similarity to ELF6. Thus, we named At3g48430 RELATIVE OF EARLY FLOWERING 6 (Figure 3). To address the function of REF6, three T-DNA insertion alleles of REF6 in the Col background (ref6-1, -2, and -3; Figure 2) were obtained and analyzed for phenotypes. Unlike elf6 mutants, all three ref6 mutants showed recessive late-flowering phenotypes both in LD and SD (Figures 1B and 1D; Col and ref6-2 flowered with 11.8 ± 1.0 and 15.1 ± 1.1 rosette leaves, respectively, in LD). The flowering time difference between ref6 mutants and the wild type in LD was not large but statistically significant (Table 1). Late-flowering mutants in the photoperiodic floral regulatory pathway show late-flowering phenotypes in LD but not in SD, and their late-flowering phenotypes are not effectively suppressed by vernalization (e.g., Koornneef et al., 1998). However, late-flowering mutants in the FLC-dependent pathways display late-flowering phenotypes both in LD and SD, and these late-flowering phenotypes are suppressed effectively by vernalization (e.g., Michaels and Amasino, 2000). Therefore, the photoperiod-independent late-flowering phenotypes of ref6 mutants suggested a role of REF6 in the FLC-dependent pathways. Consistent with this, late-flowering phenotypes of ref6 mutants were also effectively suppressed by vernalization (Figure 1D). The difference in flowering time between wild-type and ref6 mutants was reduced significantly after 40 d of vernalization, and ref6 mutants flowered at a similar time as the wild type after 70 d of vernalization.
Figure 3.
Sequence Comparison of REF6 with ELF6 and Their Three Rice Homologs (OsELF6A, OsELF6B, and OsELF6C).
JmjN and JmjC domains are marked by the dotted and solid lines, respectively. Four ZnF domains are marked by boxes. REF6, ELF6, OsELF6A, and OsELF6B sequences were derived from full-length cDNA sequences of their transcripts. OsELF6C sequence was derived from a protein sequence predicted by the gene prediction program GENSCAN (http://genes.mit.edu/GENSCAN.html).
The rate of leaf initiation was identical in elf6-2, ref6-1, and Col in SD (Table 2), indicating that the altered flowering phenotype of elf6 or ref6 mutants was caused by the earlier or delayed transition of the shoot apical meristem from vegetative to reproductive phase.
Table 2.
Leaf Initiation Rate of elf6 and ref6 Mutants Compared with the Wild Type
| DAP | Genotype | LN |
|---|---|---|
| 20 | Col | 6.0 ± 0.6 |
| elf6-2 | 6.2 ± 0.7 | |
| ref6-1 | 6.1 ± 0.7 | |
| 27 | Col | 11.1 ± 0.9 |
| elf6-2 | 11.0 ± 1.0 | |
| ref6-1 | 11.2 ± 0.9 | |
| 34 | Col | 17.1 ± 1.0 |
| elf6-2 | 17.3 ± 1.1 | |
| ref6-1 | 17.0 ± 1.0 | |
| 41 | Col | 24.2 ± 1.5 |
| elf6-2 | 24.3 ± 1.4 | |
| ref6-1 | 24.4 ± 1.4 |
Values shown are means ± sd of rosette leaf numbers (LN) of wild-type (Col), elf6-2, or ref6-1 plants grown in SD. Visible leaves were scored at 20, 27, 34, and 41 d after planting (DAP). Twelve plants were scored for each genotype.
ELF6 and REF6 Encode Jmj/ZnF-Class Proteins That Are Most Homologous to Each Other
To identify the open reading frames of ELF6 and REF6, ELF6 and REF6 full-length cDNAs were cloned by RT-PCR. One large open reading frame was found within each cDNA that was predicted to encode 1340 amino acids of ELF6 or 1360 amino acids of REF6. The cDNA and predicted protein sequences of ELF6 and REF6 were different from the annotated sequences because of the misprediction of splicing sites, and the correct cDNA and protein sequences were deposited in GenBank.
Both ELF6 and REF6 contain jumonji N (JmjN) and jumonji C (JmjC) domains and four copies of C2H2-type ZnF domains (Figures 2 and 3). Significant sequence similarities between ELF6 and REF6 or between these two proteins and other proteins in the database are detected only in these domains. Clissold and Ponting (2001) categorized numerous eukaryotic proteins containing JmjC domains into seven groups based on sequence similarities. ELF6 and REF6 belong to the Group 1 proteins in which JmjC domains accompany JmjN domains and various classes of DNA binding or protein–protein interaction motifs such as PHD, ARID/BRIGHT, and ZnF domains. Because of such domain organizations, the Group 1 proteins are also called the Jmj family of transcription factors and have been implicated in gene transcription or chromatin remodeling (Balciunas and Ronne, 2000), although the biochemical activities of JmjN and JmjC domains have not yet been demonstrated. The Group 1 type domain organization of ELF6 and REF6 suggests that ELF6 and REF6 might also be involved in transcriptional gene regulation.
In addition to ELF6 and REF6, Arabidopsis has seven additional putative JmjN/JmjC-containing proteins that belong to Group 1 (At5g46910, At2g34880, At1g08620, At4g20400, At1g63490, At1g30810, and At2g38950). ELF6 and REF6 are the most similar to each other among these nine members. Sequence similarity among these nine proteins is confined only to the JmjN/JmjC domains. The seven proteins other than ELF6 and REF6 contain different types of DNA binding or protein–protein interaction motifs from C2H2-type ZnF at their C termini, suggesting divergent biological roles between ELF6/REF6 and the other seven members.
Despite of the phylogenic distance between Arabidopsis and rice (Oryza sativa), it has been reported that these two plant species share key components in their flowering time regulation (Hayama et al., 2003; Izawa et al., 2003; Tadege et al., 2003; Lee et al., 2004). This prompted us to examine if the rice genome has genes homologous to ELF6 or REF6 by similarity searches against the databases of rice full-length cDNAs and rice genome sequences at the National Institute of Agrobiological Sciences (http://www.dna.affrc.go.jp/). Three rice genes were identified to have significant and comparable sequence similarities to ELF6 or REF6 and are named OsELF6A, OsELF6B, and OsELF6C (Figure 3). The predicted protein sequences of the OsELF6 proteins show that the domain organization of ELF6 and REF6 is also conserved in these rice proteins. Whether these OsELF6 genes play similar roles in rice flowering time regulation either to those of ELF6 or REF6 in Arabidopsis remains to be determined.
REF6 Acts as an FLC Repressor, whereas ELF6 Is Involved in the Regulation of Photoperiodic Flowering
Photoperiod-independent late flowering and the effective suppression of the late-flowering phenotype by vernalization of ref6 mutants (Figure 1) suggested a role of REF6 in the regulation of FLC. To address this at the molecular level, we examined FLC expression in ref6 mutants. FLC mRNA level was increased in ref6-1 and ref6-3; however, there was no detectable difference in CO mRNA level between the mutants and the wild type (Figure 4A). To address the role of REF6 in FLC regulation more directly, a double mutant between ref6-1 and an FLC null mutant (flc-3; Michaels and Amasino, 1999) was generated and evaluated for flowering time. ref6-1 flc-3 double mutants flowered at a similar time to either wild-type Col or flc-3 single mutants (Figure 4B), demonstrating that REF6 indeed acts as an FLC repressor. Similarly, the late-flowering phenotype of ref6 was also largely suppressed by a mutation in ELF5, a gene required for high levels of FLC expression (Noh et al., 2004; Figure 4B).
Figure 4.
REF6 Acts as an FLC Repressor, whereas ELF6 Plays a Role in the Photoperiodic Floral Regulatory Pathway.
(A) Increased FLC expression in ref6 mutants. RNA was isolated from 10-d-old seedlings of Col, ref6-1, and ref6-2 grown under continuous light. The blot was probed first with FLC and then reprobed with 18S ribosomal DNA (18S) as a loading control. CO expression was studied by RT-PCR analyses. The RNA used for the RNA gel blot analyses was also used for the RT-PCR. Ubiquitin (UBQ) expression was analyzed as a control for the RT-PCR.
(B) Suppression of ref6-mediated late flowering by flc and elf5 mutations. Wild-type (Col) and each single or double mutant was grown under LD, and their flowering times were measured as the number of rosette and cauline leaves. Data shown are means ± sd of at least 12 plants per genotype (see [C] and [D]).
(C) Earlier flowering of elf6 elf5 double mutants compared with elf6 and elf5 single mutants. Flowering time was measured under SD as described in (B).
(D) Double mutant analyses between elf6 and a variety of late-flowering mutants. Flowering time was measured under LD.
(E) FLC transcript level is not affected by a mutation in elf6. RNA was isolated from 10-d-old seedlings of each genotype grown under LD. The blot was probed first with FLC and then reprobed with 18S ribosomal DNA (18S) as a loading control.
However, double mutants between elf6 and elf5 flowered earlier than either single mutant (Figure 4C). This result showed that ELF6 does not act in a linear pathway to ELF5 and suggested the possibility that ELF6 functions in an FLC-independent pathway. To evaluate the role of ELF6, a series of double mutants were generated between elf6-2 and a variety of late-flowering mutants. elf6 partially suppressed the late-flowering phenotypes induced by mutations in the FLC-dependent pathway genes (ref6, ld, or fpa) or by the presence of FRI, resulting in intermediate flowering phenotypes in the double mutants (Figure 4D). FLC transcript level in elf6 ld or elf6 fpa double mutants was not reduced compared with that in ld or fpa single mutants (Figure 4E), demonstrating that the partial suppression of the late-flowering phenotypes of these single mutants by the elf6 mutation did not result from the downregulation of FLC. However, double mutants between elf6 and the late-flowering mutants in the photoperiodic flowering pathway (co and gi) flowered at the same time as the single photoperiodic late-flowering mutants (i.e., the co and gi mutations are epistatic to the elf6 mutation) (Figure 4D). Taken together, these results indicate that ELF6 does not function in the same pathway as the FLC-dependent pathway but rather functions as an upstream repressor of the photoperiodic flowering pathway. The late-flowering phenotype of soc1 but not ft mutants was partially suppressed by the elf6 mutation (Figure 4D). These results suggest the possibility that the two floral integrators are regulated differentially by the FLC-dependent and the photoperiodic pathways and that the regulation of FT is more dependent on the photoperiodic pathway than on the FLC-dependent pathway.
Overexpression of REF6 Activates Floral Integrators, FT and SOC1, in an FLC-Independent Manner
Genetic analyses using the loss-of-function mutants of REF6 and ELF6 showed they act as an FLC repressor and a negative regulator in the photoperiodic flowering pathway, respectively (Figure 4). We were also interested in testing the effects of the overexpression of these genes. Genomic clones of REF6 and ELF6 were fused to the strong 35S promoter of Cauliflower mosaic virus and introduced into wild-type Ws. 35S:ELF6 did not cause detectable changes in flowering time in wild-type Ws, although it could rescue the early-flowering phenotype of elf6 mutants (data not shown). However, 11 out of 18 transgenic lines containing the 35S:REF6 showed a strong early-flowering phenotype both in LD and SD compared with wild-type Ws (Figures 5A and 5B). No other notable developmental alterations were observed in the transgenic plants except for the fact that a few of these transgenic lines showed growth condition–dependent upward-curling phenotypes in their first and second rosette leaves. REF6 mRNA level was increased dramatically in the two early-flowering REF6 overexpression lines (REF6OE6 and REF6OE7) tested (Figure 5C). The early-flowering phenotypes of the 35S:REF6 plants were more obvious in SD than in LD. However, a photoperiod response was retained in the 35S:REF6 plants as evidenced by earlier flowering in LD than in SD.
Figure 5.
REF6 Overexpression Causes Early Flowering by Increasing the Expression of FT and SOC1.
(A) Early flowering of three REF6 overexpression lines (REF6OE6, REF6OE7, and REF6OE11). Representative plants of Ws and the three REF6 overexpression lines grown in LD at the time of initiation of Ws flowering are shown.
(B) Flowering time of a REF6 overexpression line in LD and SD. Ws and REF6OE6 plants were grown under LD and SD. Data shown are means ± sd of at least 12 plants for each genotype and treatment.
(C) REF6 mRNA expression in Ws and REF6 overexpression lines. RNA was isolated from 10-d-old seedlings of Ws, REF6OE6, and REF6OE7 grown under continuous light. The blot was probed first with REF6 and then reprobed with 18S ribosomal DNA (18S) as a loading control.
(D) Increased expression of FT and SOC1 but not CO in REF6 overexpression lines. RT-PCR analyses of FT, SOC1, and CO in Ws, flc-3 in Ws, REF6OE6, and REF6OE7. RNA was isolated from 10-d-old seedlings grown under continuous light. Ubiquitin (UBQ) expression was analyzed as a control.
One hypothesis to explain the strong early-flowering phenotype of REF6 overexpressors could be that REF6 overexpression might reduce the FLC expression, which in turn increases the expression of downstream floral integrators, FT and SOC1. However, this hypothesis is not likely because an flc null mutant (flc-3) in Ws flowers at a similar time as wild-type Ws (Noh et al., 2004). Therefore, the early-flowering phenotype of the REF6 overexpressors is likely to be caused by an FLC-independent mechanism. To examine the effect of REF6 overexpression on the photoperiodic flowering pathway and downstream floral regulators, we studied the expression of FT, SOC1, and CO by RT-PCR in two REF6 overexpressors in comparison with that in wild-type Ws and flc-3 in Ws. There was no detectable difference in FT, SOC1, and CO mRNA levels between wild-type Ws and flc-3 in Ws (Figure 5D), consistent with their flowering time behaviors. However, the two REF6 overexpressors showed increased expressions of FT and SOC1 but not CO compared with wild-type Ws and flc-3 in Ws. In conclusion, overexpression of REF6 causes increases in FT and SOC1 mRNA levels in an FLC-independent manner that leads to the early-flowering phenotype.
Distinctive Expression Patterns of REF6 and ELF6
Structural similarity but difference in biological role between REF6 and ELF6 raises interesting questions on their molecular functions. These two proteins may have different cellular functions despite their structural similarity. Overexpression studies described above indicate that the differences in biological functions between REF6 and ELF6 come at least partially from differences in their cellular activities. In addition, REF6 and ELF6 may have different expression patterns that reflect their different in vivo roles. The expression patterns of REF6 and ELF6 were evaluated by analysis of fusions of the REF6 and ELF6 upstream and coding regions to the reporter gene β-glucuronidase (GUS; Figure 6A). Because a transcriptional terminator from nopaline synthase (NOS-T in Figure 6A) instead of the genes' own 3′ cognate sequences was used for the fusion constructs, possibilities of additional regulation by the 3′ untranslated region of REF6 or ELF6 could not be tested in our experiments. Nevertheless, these constructs were capable of complementing ref6 and elf6 mutant phenotypes, respectively, indicating that the fusion proteins are functional (data not shown).
Figure 6.
Expression Pattern of REF6 and ELF6.
(A) Schematic representation of the REF6:GUS and ELF6:GUS translational fusion constructs. The translated REF6 and ELF6 exons are indicated by gray boxes. Lines indicate introns or intergenic sequences. The linker sequence between the last amino acid of REF6 or ELF6 and the first amino acid of GUS is shown. Numbers indicate amino acid positions in REF6, ELF6, or GUS.
(B) to (F) Histochemical GUS staining of transgenic Arabidopsis containing the REF6:GUS fusion.
(B) Seventeen-day-old whole seedling grown under continuous light.
(C) to (E) Shoot apical meristem region (C), primary root tip (D), and developing secondary root tips (E) of a 10-d-old seedling grown under continuous light.
(F) Nuclear localization of the REF6:GUS in the cells of the root distal elongation region of a 10-d-old seedling grown under continuous light. Arrows in (F) and (G) indicate cells with clear nuclear localization of REF6:GUS.
(G) Staining of the root tissue shown in (F) with 4′,6′-diamidino-2-phenylindole hydrochloride to visualize nuclei.
(H) and (I) Histochemical GUS staining of transgenic Arabidopsis containing the ELF6:GUS fusion.
(H) Ten-day-old whole seedling grown under continuous light. Arrow indicates a root tip.
(I) Magnification of the shoot apical meristem region of the seedling shown in (H).
In seedlings, REF6 expression was highest in the shoot apical meristem region and primary and secondary root tips (Figures 6B to 6E), but lower expression was also observed in the cotyledons, leaves, and root axis, especially along vascular tissues (Figures 6B and 6E). This expression pattern of REF6 is similar to that of FLC and other genes affecting FLC expression (e.g., Aukerman et al., 1999; Michaels and Amasino, 2000; Schomburg et al., 2001; Macknight et al., 2002; He et al., 2003; Noh and Amasino, 2003; Noh et al., 2004), consistent with the function of REF6 as an FLC repressor. Nuclear localization of the REF6:GUS fusion protein was observed in the cells of the root distal elongation region (Figures 6F and 6G), and this is in agreement with the possible role of REF6 as a transcriptional regulator.
By contrast, the ELF6 expression pattern in seedlings was quite different from that of REF6. Overall, ELF6 was expressed at very low levels, but its highest expression was observed in cotyledons and leaves (Figures 6H and 6I). There was no detectable ELF6 expression in the shoot apical meristem or in root tips. This expression pattern of ELF6 is similar to the expression pattern of photoperiodic flowering genes, CO and FT (Takada and Goto, 2003), consistent with its role as an upstream repressor of the photoperiodic flowering pathway.
Therefore, in addition to the difference in cellular functions between REF6 and ELF6, different spatial expression patterns can also be ascribed to the divergence in their biological roles.
REF6 Represses FLC Transcription through Chromatin Modifications
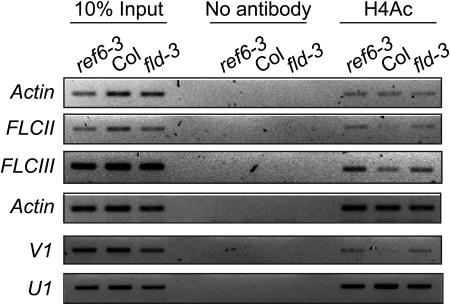
Recently, it has been shown that transcription of FLC is regulated by multiple proteins through chromatin remodeling (He et al., 2003; Ausín et al., 2004; Bastow et al., 2004; Sung and Amasino, 2004). Because REF6 acts as an FLC repressor (Figure 4) and has domains suggesting a role in chromatin remodeling (Figure 2), we tested if the increased FLC mRNA expression in ref6 mutants accompanies alterations in FLC chromatin structure by examining the modification of histone tails. Chromatin immunoprecipitation (ChIP) with antibodies specific to the penta-acetylated histone H4 tail followed by quantitative PCR showed that the amount of histone H4 acetylation in FLC chromatin is increased in ref6-3 mutants as well as in fld-3 mutants in which FLC chromatin is known to be hyperacetylated (He et al., 2003) compared with in wild-type Col (Figure 7). Hyperacetylation of histone H4 in ref6-3 mutants was observed in the promoter region close to the translation start site (FLCII) and in the first intron (FLCIII and V1) but not in the 3′ untranslated region (U1) of FLC. The same type of histone H4 tail modification was observed in fld-3 mutants (Figure 7) as reported previously (He et al., 2003). Thus, our ChIP studies show that there is a positive correlation between the level of H4 acetylation and the expression of FLC mRNA. This is in agreement with the general idea that H4 acetylation is a marker commonly associated with transcriptionally active loci (for review, see Eberharter and Becker, 2002). In summary, the above results indicate that the repression of FLC transcription by REF6, a Jmj/ZnF protein, is accompanied by histone modifications in the FLC chromatin.
Figure 7.
ChIP Using Antibody against Hyperacetylated H4.
ref6 and fld mutations increase the precipitation of regions II, III (He et al., 2003), and V1 (Sung and Amasino, 2004) of FLC. Association of U1 region (Sung and Amasino, 2004) with antibody against hyperacetylated H4 was not increased in ref6 and fld mutants compared with in wild-type Col. Representative images of two independent experiments are shown.
DISCUSSION
In this study, we report the identification of two novel floral regulators, ELF6 and REF6. Interestingly, ELF6 and REF6 both encode nuclear proteins containing JmjN/JmjC and four copies of C2H2-type ZnF domains that are most homologous to each other in Arabidopsis despite the difference in their role in the regulation of flowering time; ELF6 acts as a repressor in the photoperiodic flowering pathway, whereas REF6 is an FLC repressor. The role of REF6 as an FLC repressor was demonstrated in multiple ways. First, the late-flowering phenotype of ref6 mutants was effectively suppressed by vernalization (Figure 1D), a floral inductive process mediated mainly by the repression of FLC expression. Second, mutations in REF6 caused increases in FLC mRNA level without decreases in CO mRNA level (Figure 4A). Third, the late-flowering phenotype of ref6 mutants was almost fully suppressed by an flc null mutation (Figure 4B).
The late-flowering phenotype of photoperiod pathway mutants (gi, co, and ft) are epistatic to the early-flowering phenotype of elf6 mutants (Figure 4D). Furthermore, double mutants between elf6 and FLC-dependent late-flowering mutants (ref6, ld, fpa, and FRI) show intermediate flowering times without reduction of FLC mRNA expression compared with the single late-flowering mutants (Figures 4D and 4E). These results indicate that ELF6 acts as a repressor upstream of the photoperiodic floral regulatory pathway. However, a molecular description of the function of ELF6 in the regulation of photoperiodic flowering has not yet been made. Because the early-flowering phenotype of elf6 mutants is weak and the mRNA expression of photoperiod pathway marker genes, such as GI, CO, and FT, is detected only by RT-PCR but not by the more quantitative method of RNA gel blot analysis, attempts to evaluate if there are differences in mRNA levels of these photoperiod pathway marker genes between wild-type and elf6 mutants have not yet been successful (data not shown). One possibility is that ELF6 may regulate the circadian rhythm of the genes in the photoperiodic floral regulatory pathway. Our double mutant analyses show that ELF6 is upstream of GI, a gene with roles both in the circadian clock and the photoperiodic floral regulation (Fowler et al., 1999; Park et al., 1999). Therefore, it is possible that the mutations in ELF6 may affect flowering time through differential regulation of the circadian clock. It will be of interest to test if elf6 mutants have defective circadian rhythms and if their early-flowering phenotype is derived from circadian defects.
JmjC-domain proteins with additional domains known to be involved in protein–protein interactions or DNA binding have been implicated in the regulation of gene transcription or chromatin remodeling, although the role of the JmjC domain itself still remains enigmatic (Balciunas and Ronne, 2000; Clissold and Ponting, 2001). Lately, it was reported that Epe1, a JmjC-domain protein without a notable DNA binding or protein–protein interaction domain from Saccharomyces pombe, modulates heterochromatin formation, and its JmjC domain is necessary for activity (Ayoub et al., 2003). Dynamic changes in the modification pattern of histones, such as acetylation, phosphorylation, methylation, and ubiquitination, are thought to control transcriptional activity by affecting chromatin structure and accessibility of nonhistone regulatory factors to chromatin (Strahl and Allis, 2000). Growing evidence suggests that remodeling of chromatin structure controls FLC transcription. The chromatin within the FLC locus is structurally modified during the vernalization process, and this modification results in the reduction of FLC transcription (Bastow et al., 2004; Sung and Amasino, 2004). It was also reported that the mutations in FLD and FVE among the autonomous-pathway floral regulatory genes cause hyperacetylation in FLC chromatin, leading to increased levels of FLC mRNA in the mutants (He et al., 2003; Ausín et al., 2004). Similarly, our ChIP studies show that the disruption of REF6 activity changes modification patterns of histone H4 tails in the FLC locus (Figure 7). Therefore, our results further demonstrate the chromatin remodeling-related function of a Jmj-class protein (REF6) at the target gene (FLC) level. FLC-like spatial expression pattern and nuclear localization of REF6 (Figure 6) also indirectly support its role as a component of chromatin-related transcriptional machinery involved in FLC regulation.
Along with the late-flowering phenotypes, ref6 mutants also displayed darker green color in old leaves and stems compared with the wild type (data not shown). The darker green phenotype was observed only in ref6 mutants but not in elf6 mutants and was more obvious in older tissues than in younger tissues. Anthocyanin but not chlorophyll levels were significantly increased in old leaves of ref6 mutants, suggesting that in addition to its role in floral regulations, REF6 also acts as a repressor of anthocyanin biosynthesis or accumulation. Thus, REF6 might have multiple target genes, which is in agreement with general characteristics of chromatin modifiers.
At this moment, the biochemical roles of REF6 in histone modification are not clear. FLD and FVE are homologous to the components of HDAC complexes in human and other organisms; however, no HDAC component has been found to be homologous to REF6. REF6 might be involved in the recruitment or in the stabilization of the binding of a HDAC complex to FLC chromatin through interactions with one of the components in the HDAC complex. Hyperacetylation of histone H4 in ref6 mutants might increase the accessibility of nonhistone regulatory proteins to FLC chromatin that are required for orchestrating highly complex processes of transcription leading to increased levels of FLC expression followed by late flowering.
Difference in biological role between REF6 and ELF6 is indicated by two lines of evidence. First, ectopic expression of REF6 but not ELF6 causes early flowering by an FLC-independent induction of downstream floral activators, FT and SOC1 (Figure 5), indicating that REF6 and ELF6 exert different cellular functions. Second, the spatial expression patterns of these two genes are also quite different (Figure 6); REF6 and ELF6 resemble those of FLC and photoperiod pathway genes (CO and FT), respectively. The expression pattern of REF6 or ELF6 is consistent with the genetic role each plays as an FLC or photoperiodic flowering-pathway repressor. Therefore, both different cellular functions and differential expression patterns of REF6 and ELF6 might contribute to the different biological roles of these two proteins. Pairs of closely related protein having opposite effects on flowering time have been reported. FT and TFL are similar to each other. However, one acts as a floral activator, whereas the other is a floral repressor (Kobayashi et al., 1999). In another case, SVP and AGL24 act as a floral repressor and activator, respectively, despite a high level of sequence similarity between them (Michaels et al., 2003). It would be of interest to find a domain or domains that are responsible for the different cellular roles between REF6 and ELF6 as well as to find a regulatory element responsible for their differential expression patterns.
The FT and SOC1 activation-mediated early flowering of REF6 overexpressors (Figure 5) was surprising. A similar observation was made when VRN1, which allows stable FLC repression after vernalization, was overexpressed (Levy et al., 2002). Similar to the FLC-independent activation of FT and SOC1 in REF6 overexpressors, VRN1 overexpression also caused the activation FT and SOC1 in an FLC-independent manner. Despite the activation of downstream floral integrators, both REF6 and VRN1 overexpressors maintained photoperiod sensitivity as evidenced by their earlier flowering in LD than in SD. The retention of photoperiod sensitivity of REF6 and VRN1 overexpressors combined with the CO-independent activation of FT and SOC1 in REF6 overexpressors (Figure 5D) suggest that REF6 and VRN1 do not promote flowering through the photoperiod pathway. FT and SOC1 integrate flowering signals generated through FLC and CO. GA mainly exerts its floral regulatory effects in SD through the activation of SOC1 and LFY but not FT (Blázquez and Weigel, 2000; Moon et al., 2003). Vernalization induces flowering mainly through the repression of FLC transcription, although it also has a marginal activation effect on SOC1 and FT in an flc null mutant background (Moon et al., 2003). Therefore, FLC- and CO-independent induction of FT and SOC1 in REF6 overexpressors might be mediated through a mechanism yet to be discovered. Recently, floral regulatory functions of a few FLC homologs (FLM, MAF2, etc.) have been reported (Scortecci et al., 2001, 2003; Ratcliffe et al., 2003). Therefore, it would be of interest to test if the strong induction of FT and SOC1 in REF6 overexpressors is mediated through these FLC homologs.
METHODS
Plant Materials and Growth Conditions
elf6-1 in the Ws background was isolated from the BASTA population of the Arabidopsis Knockout Facility (http://www.biotech.wisc.edu/Arabidopsis/). elf6 and ref6 T-DNA insertion lines in the Col background were obtained either from the SALK collection (http://signal.salk.edu/; elf6-2, elf6-3, ref6-1, and ref6-2, which are SALK_018830, SALK_074694, SALK_001018, and SALK_122006, respectively) or from the Torrey Mesa Research Institute (elf6-4 and ref6-3, which are SAIL371D8 and SAIL747A7). The following mutants are in the Col background and described previously: flc-3 (Michaels and Amasino, 1999), elf5-4 (Noh et al., 2004), ld-1 (Lee et al., 1994a), fpa-7 (Michaels and Amasino, 2001), FRI (Lee et al., 1994b), co-101 (Takada and Goto, 2003), gi-2 (Park et al., 1999), soc1-2 (Moon et al., 2003), and ft-1 (Lee et al., 2000). All plants were grown under ∼100 μE m−2 s−1 cool white fluorescent light at 22°C. Plants were vernalized as described previously (Noh and Amasino, 2003).
T-DNA Flanking Sequence Analyses
The sequence flanking the T-DNA of elf6-1 was obtained by thermal asymmetric interlaced PCR (Liu et al., 1995) as described by Schomburg et al. (2003).The T-DNA borders of elf6 and ref6 alleles were defined by sequencing PCR products obtained using a T-DNA border primer and a gene-specific primer. SALKLB1 (5′-GCAAACCAGCGTGGACCGCTTGCTGCAACT-3′) or SAILLB3 (5′-TAGCATCTGAATTTCATAACCAATCTCGATACAC-3′) was used as a T-DNA border primer for the mutants obtained from the SALK collection or from the Torrey Mesa Research Institute, respectively.
ChIP
ChIP was performed as described by Gendrel et al. (2002) using ∼10 to 12-d-old seedlings. Briefly, seedlings were vacuum infiltrated with 1% formaldehyde for cross-linking and ground in liquid nitrogen after quenching cross-linking. Chromatin was isolated and sonicated to ∼0.5 to 1 kb. Antihyperacetylated histone H4 (Upstate Biotechnology, Lake Placid, NY) was added to the chromatin solution precleared with salmon sperm DNA/Protein A agarose beads (Upstate Biotechnology). After subsequent incubation with salmon sperm DNA/Protein A agarose beads, immunocomplexes were precipitated and eluted from the beads. Cross-links were reversed, and residual proteins in the immunocomplexes were removed by the incubation with proteinase K followed by phenol/chloroform extraction. DNA was recovered by ethanol precipitation. The amount of immunoprecipitated FLC chromatin was determined by quantitative PCR on four different regions of FLC locus as previously reported (FLCII and FLCIII [He et al., 2003]; V1 and U1 [Sung and Amasino, 2004]). The sequences of primer pairs for each PCR reaction are as follows: FLCII, CH1 (5′-CTGCGACCATGATAGATACATGAGA-3′) and HIS2 (5′-TTCACTCAACAACATCATCGAGCACG-3′); FLCIII, CH2 (5′-GTTCTCAATTCGCTTGATTTCTAGTTTTT-3′) and CH3 (5′-GGCCCGACGAAGAAAAAGGTAGATAGGC-3′); V1, V1F (5′-ATAGATTTGCCTCATATTTATGTGATTG-3′) and V1R (5′-TATTTCTTACATGAAGACAAGTGTTGTGG-3′); U1, U1F (5′-ACAAAAGGTTGATGAACTTTGTACCT-3′) and U1R (5′-CCGACATAGGCCCAAAAAACCCATGC-3′).
Histochemical GUS Assays
For the construction of the ELF6:GUS translational fusion construct, 7.4-kb genomic fragment of ELF6 containing 2.3-kb 5′ upstream region and the entire coding region was generated by PCR amplification using ELF6GUS-1 (5′-agcggatccAAACTGTTCATCACCCTCTTCATCCACAAG-3′) and ELF6GUS-2 (5′-gccggatccTGTGACATAGTGCATGGTTTTACGTCTGT-3′) as primers. The resulting PCR product was digested with BamHI and ligated to pPZP211G (Noh et al., 2001) at the BamHI site. The REF6:GUS translational fusion construct was generated by PCR amplification of 6 kb of the REF6 genomic DNA fragment containing 1.2-kb 5′ upstream region and the entire coding region using REF6GUS-1 (5′-gaccccgggAGTGATCATTGTCCGGTTACACTCGAGCTT-3′) and REF6GUS-2 (5′-gccactagtCCTTTTGTTGGTCTTCTTAACCGAATGACC-3′) as primers. After restriction digestion with SmaI-SpeI, the PCR product was ligated to pPZP211-GUS (Noh and Amasino, 2003) and digested with PstI-XbaI (PstI site was blunted with T4 DNA polymerase [New England Biolabs, Beverly, MA] after the digestion). In primer sequences, restriction sites for cloning are underlined, and sequences corresponding to the ELF6 or REF6 are in capital letters. elf6-4 or ref6-3 mutant plants were transformed using Agrobacterium tumefaciens strain ABI containing the ELF6:GUS or the REF6:GUS construct, respectively, by infiltration (Clough and Bent, 1998). Histochemical GUS staining was performed as described by Schomburg et al. (2001) using transgenic lines selected as previously described (Noh and Amasino, 2003).
RNA Gel Blot Analyses
Total RNA was isolated from 10-d-old seedlings by TRI Reagent (Sigma, St. Louis, MO) according to the manufacturer's instructions. For RNA gel blot analyses, 40 μg of total RNA was separated by denaturing formaldehyde-agarose gel electrophoresis as described by Sambrook et al. (1989). The FLC probe was a cDNA fragment lacking the conserved MADS domain sequences. The REF6 probe was a 2.5-kb REF6 cDNA fragment generated by PCR using REF6CS-3 (5′-CCTGAAGTATTTGTTAAAGC-3′) and REF6-1 (5′-CCTCCATGTTACATTGGTATGCTGCACATT-3′) as primers. As a control for the quantity of RNA loaded, blots were also probed with 18S rDNA.
RT-PCR Analyses
Reverse transcription was performed with Superscript II (Invitrogen Life Technologies, Carlsbad, CA) according to the manufacturer's instructions using 2.5 μg of total RNA isolated as described above. Quantitative PCR was performed on first strand DNA with ExTaq polymerase (TaKaRa Bio, Otsu, Shiga, Japan) using the following primers as previously described by Cerdan and Chory (2003) and Michaels et al. (2003): FT (5′-GCTACAACTGGAACAACCTTTGGCAAT-3′ and 5′-TATAGGCATCATCACCGTTCGTTACTC-3′), CO (5′-AAACTCTTTCAGCTCCATGACCACTACT-3′ and 5′-CCATGGATGAAATGTATGCGTTATGGTTA-3′), SOC1 (5′-TGAGGCATACTAAGGATCGAGTCAG-3′ and 5′-GCGTCTCTACTTCAGAACTTGGGC-3′), and UBIQUITIN (5′-GATCTTTGCCGGAAAACAATTGGAGGATGGT-3′ and 5′-CGACTTGTCATTAGAAAGAAAGAGATAACAGG-3′).
Overexpression of ELF6 and REF6
For the overexpression of ELF6 or REF6, 5.89- or 5.81-kb genomic DNA containing the entire coding region and the 3′ untranslated region of ELF6 or REF6 was generated by PCR amplification using ELF6OE-1 (5′-cgggatccATGGGTAATGTTGAAATTCCGAATTGGCT-3′) and ELF6OE-2 (5′-cgctcgagCTCAGCAACAGTGCGAATCCATGGTCAAGG-3′) for ELF6 or REF6OE-1 (5′-ggactagtATGGCGGTTTCAGAGCAGAGTCAAGATGTG-3′) and REF6OE-2 (5′-agggggcccCCTCCATTAACTTCTTCTTCTAGTTTCCCA-3′) for REF6; restriction sites for cloning are underlined, and sequences corresponding to the ELF6 or REF6 genomic DNA are in capital letters. The resulting PCR product was digested with BamHI-SacI or SpeI-SmaI and ligated to pPZP211-GUS (Noh and Amasino, 2003) digested with BamHI-SacI or XbaI-SacI (SacI site was blunted with T4 DNA polymerase [New England Biolabs] after the digestion), respectively, generating the transcriptional fusion between the 35S promoter of Cauliflower mosaic virus and either ELF6 or REF6. Transformation of elf6-4 and ref6-3 with A. tumefaciens carrying the corresponding fusion construct and selection of the transgenic lines were performed as described above.
Sequence Analyses
Genes were predicted with GenScan (Burge and Karlin, 1997). Protein sequences were analyzed with SMART (Schultz et al., 2000), PSORT (Nakai and Kanehisa, 1992), and ψ-BLAST (Altschul et al., 1997). Protein sequence alignments were generated using ClustalW (Thompson et al., 1994). cDNA was synthesized, cloned, and sequenced as described (Noh and Amasino, 2003). For the ELF6 or REF6 cDNA cloning, ELF6OE-1 (5′-cgggatccATGGGTAATGTTGAAATTCCGAATTGGCT-3′) and ELF6GUS-2 (5′-gccggatccTGTGACATAGTGCATGGTTTTACGTCTGT-3′) or REF6F (5′-ggctcgagATGGCGGTTTCAGAGCAGAGTCAA-3′) and REF6R (5′-cgactagtCCTTTTGTTGGTCTTCTTAACCG-3′) were used as gene-specific primers for cDNA amplification, respectively; restriction sites for cloning are underlined, and sequences corresponding to the ELF6 or REF6 genomic DNA are in capital letters. OsELF6A and OsELF6B cDNA sequences correspond to the rice full-length cDNA clone J023001N18 (accession number AK068952) and J013002J08 (accession number AK065251) of KOME (http://cdna01.dna.affrc.go.jp/cDNA/).
Sequence data from this article have been deposited with the EMBL/GenBank data libraries under accession numbers AY664499 (REF6) and AY664500 (ELF6).
Acknowledgments
We thank Pill-Soon Song for careful reading of the manuscript. We are grateful to the Salk Institute Genome Analysis Laboratory and Torrey Mesa Research Institute of Syngenta for providing knockout pools containing elf6 and ref6 alleles. This work was supported by a grant from the Molecular and Cellular BioDiscovery Research Program (M1-0311-09-0000) from the Ministry of Science and Technology of Korea to Y.-S.N. and by a grant from the Korea Science and Engineering Foundation to the Environmental Biotechnology National Core Research Center (R15-2003-012-01002-0) to B.N. and Y.-S.N. Work in R.M.A.'s lab was supported by the College of Agricultural and Life Sciences and the Graduate School of the University of Wisconsin and by Grant 0209786 from the National Science Foundation. This is Kumho Life and Environmental Science Laboratory publication 72.
The authors responsible for distribution of materials integral to the findings presented in this article in accordance with the policy described in the Instructions for Authors (www.plantcell.org) are: Bosl Noh (bnoh@kkpc.com) and Yoo-Sun Noh (noh@kkpc.com).
Article, publication date, and citation information can be found at www.plantcell.org/cgi/doi/10.1105/tpc.104.025353.
References
- Altschul, S.F., Madden, T.L., Schaffer, A.A., Zhang, J., Zhang, Z., Miller, W., and Lipman, D.J. (1997). Gapped BLAST and PSI-BLAST: New generation of protein database search programs. Nucleic Acids Res. 25, 3389–3402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aukerman, M.J., Lee, I., Weigel, D., and Amasino, R.M. (1999). The Arabidopsis flowering-time gene LUMINIDEPENDENS is expressed primarily in regions of cell proliferation and encodes a nuclear protein that regulates LEAFY expression. Plant J. 18, 195–203. [DOI] [PubMed] [Google Scholar]
- Ausín, I., Alonso-Blanco, C., Jarillo, J., Ruiz-García, L., and Martínez-Zapater, J.M. (2004). Regulation of flowering time by FVE, a retinoblastoma-associated protein. Nat. Genet. 36, 162–166. [DOI] [PubMed] [Google Scholar]
- Ayoub, N.A., Noma, K., Isaac, S., Kajan, T., Grewal, S.I.S., and Cohen, A. (2003). A novel jmjC domain protein modulates heterochromatization in fission yeast. Mol. Cell. Biol. 23, 4356–4370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balciunas, D., and Ronne, H. (2000). Evidence of domain swapping within the jumonji family of transcription factors. Trends Biochem. Sci. 25, 274–276. [DOI] [PubMed] [Google Scholar]
- Bastow, R., Mylne, J.S., Lister, C., Lippman, Z., Martienssen, R.A., and Dean, C. (2004). Vernalization requires epigenetic silencing of FLC by histone methylation. Nature 427, 164–167. [DOI] [PubMed] [Google Scholar]
- Blázquez, M.A., and Weigel, D. (2000). Integration of floral inductive signals in Arabidopsis. Nature 404, 889–892. [DOI] [PubMed] [Google Scholar]
- Burge, C., and Karlin, S. (1997). Prediction of complete gene structures in human genomic DNA. J. Mol. Biol. 268, 78–94. [DOI] [PubMed] [Google Scholar]
- Cerdan, P.D., and Chory, J. (2003). Regulation of flowering time by light quality. Nature 423, 881–885. [DOI] [PubMed] [Google Scholar]
- Clissold, P.M., and Ponting, C. (2001). JmjC: Cupin metalloenzyme-like domains in jumonji, hairless and phospholipase A2β. Trends Biochem. Sci. 26, 7–9. [DOI] [PubMed] [Google Scholar]
- Clough, S.J., and Bent, A.F. (1998). Floral dip: A simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 16, 735–743. [DOI] [PubMed] [Google Scholar]
- Eberharter, A., and Becker, P.B. (2002). Histone acetylation: A switch between repressive and permissive chromatin. EMBO Rep. 3, 224–229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fowler, S., Lee, K., Onouchi, H., Samach, A., Richardson, K., Coupland, G., and Putterill, J. (1999). GIGANTEA: A circadian clock-controlled gene that regulates photoperiodic flowering in Arabidopsis and encodes a protein with several possible membrane-spanning domains. EMBO J. 18, 4679–4688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gendrel, A.V., Lippman, Z., Yordan, C., Colot, V., and Martienssen, R.A. (2002). Dependence of heterochromatic histone H3 methylation patterns on the Arabidopsis gene DDM1. Science 297, 1871–1873. [DOI] [PubMed] [Google Scholar]
- Hayama, R., Yokoi, S., Tamaki, S., Yano, M., and Shimamoto, K. (2003). Adaptation of photoperiodic control pathways produces short-day flowering in rice. Nature 422, 719–722. [DOI] [PubMed] [Google Scholar]
- He, Y., Michaels, S.D., and Amasino, R.M. (2003). Regulation of flowering time by histone acetylation in Arabidopsis. Science 302, 1751–1754. [DOI] [PubMed] [Google Scholar]
- Henderson, I.R., Shindo, C., and Dean, C. (2003). The need for winter in the switch to flowering. Annu. Rev. Genet. 37, 371–392. [DOI] [PubMed] [Google Scholar]
- Izawa, T., Takahashi, Y., and Yano, M. (2003). Comparative biology comes into bloom: Genomic and genetic comparison of flowering pathways in rice and Arabidopsis. Curr. Opin. Plant Biol. 6, 113–120. [DOI] [PubMed] [Google Scholar]
- Johanson, U., West, J., Lister, C., Michaels, S., Amasino, R., and Dean, C. (2000). Molecular analysis of FRIGIDA, a major determinant of natural variation in Arabidopsis flowering time. Science 290, 344–347. [DOI] [PubMed] [Google Scholar]
- Kobayashi, Y., Kaya, H., Goto, K., Iwabuchi, M., and Araki, T. (1999). A pair of related genes with antagonistic roles in mediating flowering signals. Science 286, 1960–1962. [DOI] [PubMed] [Google Scholar]
- Koornneef, M., Alonso-Blanco, C., Peeters, A.J.M., and Soppe, W. (1998). Genetic control of flowering time in Arabidopsis. Annu. Rev. Plant Physiol. Plant Mol. Biol. 49, 345–370. [DOI] [PubMed] [Google Scholar]
- Lee, H., Suh, S.-S., Park, E., Cho, E., Ahn, J.H., Kim, S.-G., Lee, J.S., Kwon, Y.M., and Lee, I. (2000). The AGAMOUS-LIKE 20 MADS domain protein integrates floral inductive pathways in Arabidopsis. Genes Dev. 14, 2366–2376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee, I., Aukerman, M.J., Gore, S.L., Lohman, K.N., Michaels, S.D., Weaver, L.M., John, M.C., Feldmann, K.A., and Amasino, R.M. (1994. a). Isolation of LUMINIDEPENDENS: A gene involved in the control of flowering time in Arabidopsis. Plant Cell 6, 75–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee, I., Michaels, S.D., Masshardt, A.S., and Amasino, R.M. (1994. b). The late-flowering phenotype of FRIGIDA and mutations in LUMINIDEPENDENS is suppressed in the Landsberg erecta strain of Arabidopsis. Plant J. 6, 903–909. [Google Scholar]
- Lee, S., Kim, J., Han, J.-J., Han, M.-J., and An, G. (2004). Functional analyses of the flowering time gene OsMADS50, the putative SUPPRESSOR OF OVEREXPRESSION OF CO 1/AGAMOUS-LIKE 20 (SOC1/AGL20) ortholog in rice. Plant J. 38, 754–764. [DOI] [PubMed] [Google Scholar]
- Levy, Y.Y., Mesnage, S., Mylne, J.S., Gendall, A.R., and Dean, C. (2002). Multiple roles of Arabidopsis VRN1 in vernalization and flowering time control. Science 297, 243–246. [DOI] [PubMed] [Google Scholar]
- Liu, Y.G., Mitsukawa, N., Oosumi, T., and Whittier, R.F. (1995). Efficient isolation and mapping of Arabidopsis thaliana T-DNA insert junctions by thermal asymmetric interlaced PCR. Plant J. 8, 457–463. [DOI] [PubMed] [Google Scholar]
- Macknight, R., Duroux, M., Laurie, R., Dijkwel, P., Simpson, G., and Dean, C. (2002). Functional significance of the alternative transcript processing of the Arabidopsis floral promoter FCA. Plant Cell 14, 877–888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michaels, S.D., and Amasino, R.M. (1999). FLOWERING LOCUS C encodes a novel MADS domain protein that acts as a repressor of flowering. Plant Cell 11, 949–956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michaels, S.D., and Amasino, R.M. (2000). Memories of winter: Vernalization and the competence to flower. Plant Cell Environ. 23, 1145–1153. [Google Scholar]
- Michaels, S.D., and Amasino, R.M. (2001). Loss of FLOWERING LOCUS C activity eliminates the late-flowering phenotype of FRIGIDA and autonomous pathway mutations but not responsiveness to vernalization. Plant Cell 13, 935–941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michaels, S.D., Ditta, G., Gustafson-Brown, C., Pelaz, S., Yanofsky, M., and Amasino, R.M. (2003). AGL24 acts as a promoter of flowering in Arabidopsis and is positively regulated by vernalization. Plant J. 33, 867–874. [DOI] [PubMed] [Google Scholar]
- Moon, J., Suh, S.-S., Lee, H., Choi, K.-R., Hong, C.B., Paek, N.-C., Kim, S.-G., and Lee, I. (2003). The SOC1 MADS-box gene integrates vernalization and gibberellin signals for flowering in Arabidopsis. Plant J. 35, 613–623. [DOI] [PubMed] [Google Scholar]
- Mouradov, A., Cremer, F., and Coupland, G. (2002). Control of flowering time: Interacting pathways as a basis for diversity. Plant Cell 14 (suppl.), S111–S130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakai, K., and Kanehisa, M. (1992). A knowledge base for predicting protein localization sites in eukaryotic cells. Genomics 14, 897–911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noh, B., Angus, M., and Spalding, E.P. (2001). Multidrug Resistance-like genes of Arabidopsis required for auxin transport and auxin-mediated development. Plant Cell 13, 2441–2454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noh, Y.S., and Amasino, R.M. (2003). PIE1, an ISWI family gene, is required for FLC activation and floral repression in Arabidopsis. Plant Cell 15, 1671–1682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noh, Y.-S., Bizzell, C.M., Noh, B., Schomburg, F.M., and Amasino, R.M. (2004). EARLY FLOWERING 5 acts as a floral repressor in Arabidopsis. Plant J. 38, 664–672. [DOI] [PubMed] [Google Scholar]
- Park, D.H., Somers, D.E., Kim, Y.S., Choy, Y.H., Lim, H.K., Soh, M.S., Kim, H.J., Kay, S.A., and Nam, H.G. (1999). Control of circadian rhythms and photoperiodic flowering by the Arabidopsis GIGANTEA gene. Science 285, 1579–1582. [DOI] [PubMed] [Google Scholar]
- Ratcliffe, O.J., Kumimoto, R.W., Wong, B.J., and Riechmann, J.L. (2003). Analysis of the Arabidopsis MADS AFFECTING FLOWERING gene family: MAF2 prevents vernalization by short periods of cold. Plant Cell 15, 1159–1169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reeves, P.H., Murtas, G., Dash, S., and Coupland, G. (2002). early in short days 4, a mutation in Arabidopsis that causes early flowering and reduces the mRNA abundance of the floral repressor FLC. Development 129, 5349–5361. [DOI] [PubMed] [Google Scholar]
- Samach, A., Onouchi, H., Gold, S.E., Ditta, G.S., Schwarz-Sommer, Z., Yanofsky, M.F., and Coupland, G. (2000). Distinct roles of CONSTANS target genes in reproductive development of Arabidopsis. Science 288, 1613–1616. [DOI] [PubMed] [Google Scholar]
- Sambrook, J., Fritsch, E.F., and Maniatis, T. (1989). Molecular Cloning: A Laboratory Manual. (Cold Spring Harbor, NY: Cold Spring Harbor Laboratory Press).
- Schomburg, F.M., Bizzell, C.M., Lee, D.J., Zeevaart, J.A.D., and Amasino, R.M. (2003). Overexpression of a novel class of gibberellin 2-oxidases decreases gibberellin levels and creates dwarf plants. Plant Cell 15, 151–163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schomburg, F.M., Patton, D.A., Meinke, D.W., and Amasino, R.M. (2001). FPA, a gene involved in floral induction in Arabidopsis, encodes a protein containing RNA-recognition motifs. Plant Cell 13, 1427–1436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schultz, J., Copley, R.R., Doerks, T., Ponting, C.P., and Bork, P. (2000). SMART: A Web-based tool for the study of genetically mobile domains. Nucleic Acids Res. 28, 231–234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scortecci, K., Michaels, S.D., and Amasino, R.M. (2003). Genetic interactions between FLM and other flowering-time genes in Arabidopsis thaliana. Plant Mol. Biol. 52, 915–922. [DOI] [PubMed] [Google Scholar]
- Scortecci, K.C., Michaels, S.D., and Amasino, R.M. (2001). Identification of a MADS-box gene, FLOWERING LOCUS M, that represses flowering. Plant J. 26, 229–236. [DOI] [PubMed] [Google Scholar]
- Sheldon, C.C., Burn, J.E., Perez, P.P., Metzger, J., Edwards, J.A., Peacock, W.J., and Dennis, E.S. (1999). The FLF MADS box gene: A repressor of flowering in Arabidopsis regulated by vernalization and methylation. Plant Cell 11, 445–458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simpson, G.G., and Dean, C. (2002). Arabidopsis, the Rosetta stone of flowering time? Science 296, 285–289. [DOI] [PubMed] [Google Scholar]
- Soppe, W.J.J., Bentsink, L., and Koornneef, M. (1999). The early-flowering mutant efs is involved in the autonomous promotion pathway of Arabidopsis thaliana. Development 126, 4763–4770. [DOI] [PubMed] [Google Scholar]
- Strahl, B.D., and Allis, D. (2000). The language of covalent histone modifications. Nature 403, 41–45. [DOI] [PubMed] [Google Scholar]
- Suarez-Lopez, P., Wheatley, K., Robson, F., Onouchi, H., Valverde, F., and Coupland, G. (2001). CONSTANS mediates between the circadian clock and the control of flowering in Arabidopsis. Nature 410, 1116–1120. [DOI] [PubMed] [Google Scholar]
- Sung, S., and Amasino, R.M. (2004). Vernalization in Arabidopsis thaliana is mediated by the PHD finger protein VIN3. Nature 427, 159–164. [DOI] [PubMed] [Google Scholar]
- Sung, Z.R., Chen, L., Moon, Y.H., and Lertpiriyapong, K. (2003). Mechanisms of floral repression in Arabidopsis. Curr. Opin. Plant Biol. 6, 29–35. [DOI] [PubMed] [Google Scholar]
- Tadege, E., Sheldon, C.C., Helliwell, C.A., Upadhyaya, N.M., Dennis, E.S., and Peacock, W.J. (2003). Reciprocal control of flowering time by OsSOC1 in transgenic Arabidopsis and by FLC in transgenic rice. Plant Biotechnol. J. 1, 361–369. [DOI] [PubMed] [Google Scholar]
- Takada, S., and Goto, K. (2003). TERMINAL FLOWER2, an Arabidopsis homolog of HETEROCHROMATIN PROTEIN1, counteracts the activation of FLOWERING LOCUS T by CONSTANS in the vascular tissues of leaves to regulate flowering time. Plant Cell 15, 2856–2865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson, J.D., Higgins, D.G., and Gibson, T.J. (1994). CLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, positions-specific gap penalties and weight matrix choice. Nucleic Acids Res. 22, 4673–4680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valverde, F., Mouradov, A., Soppe, W., Ravenscroft, D., Samach, A., and Coupland, G. (2004). Photoreceptor regulation of CONSTANS protein in photoperiodic flowering. Science 303, 1003–1006. [DOI] [PubMed] [Google Scholar]
- Wilson, R.N., Heckman, J.W., and Somerville, C.R. (1992). Gibberellin is required for flowering in Arabidopsis thaliana under short days. Plant Physiol. 100, 403–408. [DOI] [PMC free article] [PubMed] [Google Scholar]