Abstract
Background
Unrecognized myocardial infarction (MI) is a prevalent condition associated with a similar risk of death as recognized MI. It is unknown why some persons experience MI with few or no symptoms; however, one possible explanation is attenuated pain sensitivity. To our knowledge, no previous study has examined the association between pain sensitivity and recognition of MI.
Methods and Results
We conducted a population‐based cross‐sectional study with 4849 included participants who underwent the cold pressor test (a common experimental pain assay) and ECG. Unrecognized MI was present in 387 (8%) and recognized MI in 227 (4.7%) participants. Participants with unrecognized MI endured the cold pressor test significantly longer than participants with recognized MI (hazard ratio for aborting the cold pressor test, 0.64; CI, 0.47–0.88), adjusted for age and sex. The association was attenuated and borderline significant after multivariable adjustment. The association between unrecognized MI and lower pain sensitivity was stronger in women than in men, and statistically significant in women only, but interaction testing was not statistically significant (P for interaction=0.14).
Conclusions
Our findings suggest that persons who experience unrecognized MI have reduced pain sensitivity compared with persons who experience recognized MI. This may partially explain the lack of symptoms associated with unrecognized MI.
Keywords: epidemiology, myocardial infarction, pain tolerance, silent myocardial infarction, unrecognized myocardial infarction
Subject Categories: Cardiovascular Disease, Epidemiology, Women, Myocardial Infarction
Introduction
A large proportion of myocardial infarctions (MIs) are unrecognized,1, 2, 3, 4, 5 often because they are accompanied by few or no symptoms. Unrecognized MI is associated with a similar risk of death and recurrent MI as recognized MI.3, 6, 7, 8, 9, 10 Persons with unrecognized MI may be identified by presence of Q waves on ECG.
It is unknown why some persons experience unrecognized MI. One possible explanation for the absence of chest pain is attenuated pain sensitivity. Small sampled experimental studies have suggested a relationship between attenuated pain sensitivity and silent ischemia.11, 12, 13, 14 To our knowledge, no previous study has examined the relationship between pain sensitivity and recognition of MI.
We examined the cross‐sectional relationship between cold pressor pain tolerance and recognized and unrecognized MI in the Tromsø Study, a large population‐based health study in Tromsø, Norway. We also investigated sex differences in the association between infarct recognition and pain sensitivity. This is of interest because a larger proportion of MIs are unrecognized in women than in men.8, 9, 15
Methods
Study Population
The Tromsø Study is a population‐based cohort study conducted in the municipality of Tromsø, Norway, and was initiated in 1974. The population consists of predominantly white Caucasians. The design of the study includes repeated cross‐sectional health surveys. The sixth survey (Tromsø 6) took place in 2007–2008 and consisted of 2 visits. Total birth cohorts and random samples of birth cohorts were invited to the first visit, and 12 981 attended (attendance rate 66%).16 Those eligible for the second visit were first‐visit participants in the age groups 50 to 62 years and 75 to 84 years, a 20% random sample in the age group 63 to 74 years, and those who had attended the second visit of the fourth survey (Tromsø 4) if aged older than 75 years in 1994.16 A total of 7306 (64%) patients attended the second visit. The first visit included testing of pain tolerance with the cold pressor test.17 The second visit included a standard 12‐lead ECG. A total of 4899 participants were examined with ECG and the cold pressor test. We excluded 50 participants: 18 ECGs had pathologic noninfarct Q waves due to altered conduction (eg, left bundle branch block and Wolff‐Parkinson‐White syndrome) or ventricular enlargement; 14 ECGs were uncodable (eg, pacemaker rhythm or missing leads); and 18 ECGs were not available for manual review (ECG files were missing). The final sample consisted of 4849 participants who had undergone the cold pressor test and had valid ECGs. Figure 1 shows a flow diagram of the participants in the study.
Figure 1.
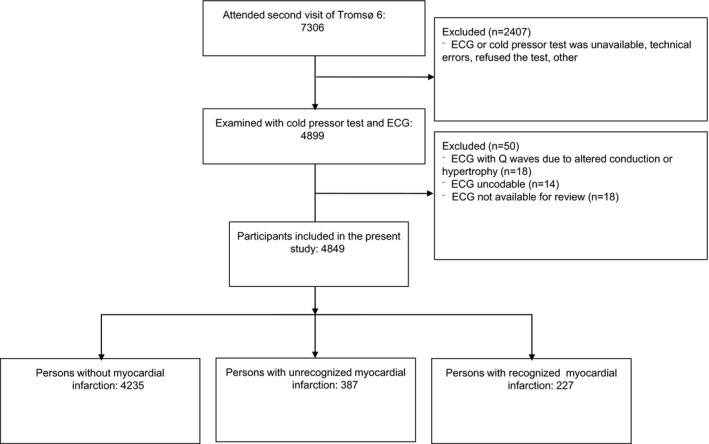
Flow diagram demonstrating inclusion and exclusion of participants in the Tromsø Study, 2007–2008.
All participants gave informed, written consent to research and agreed to linkage with public records of disease and death. The Tromsø Study and projects based on this have approval from the regional ethical committee.
Data Collection
Baseline information on potential confounding variables and use of medication was obtained by self‐reported questionnaires and physical examinations. Data on previous MI are retrospectively registered for all first‐time participants of each survey of the Tromsø Study by linkage to the electronic patient records of the University Hospital of North Norway. Admissions to other hospitals are unlikely as the nearest hospital is more than 200 km from Tromsø. Each event was reviewed and validated by persons with medical expertise based on local hospital records and records from other hospitals.16
Cold Pressor Pain
The cold pressor test is a common pain assay that has been used in experimental pain research for several decades.18 The stimulus consists of submerging the hand or foot in circulating cold water and elicits a deep aching pain thought to originate from activation of venous nociceptors.19 It was historically used as an aid in the diagnosis of angina. Participants had the testing procedures verbally explained and were placed in a comfortable chair. They were asked to insert their dominant hand and wrist into a container with circulating cold water at 3°C and a flow rate of 22 L/min, and sustain the cold immersion for as long as they could endure, up to a maximum of 106 seconds. Cold pressor tolerance was defined as time to withdrawal of the hand from the water.
Electrocardiography
A 12‐lead resting ECG was recorded using a computer‐based electrocardiograph (Cardiovit AT‐104 PC, Schiller AG, Baar, Switzerland). We used a computer‐based algorithm to extract all ECGs with a Q wave of amplitude ≤−0.1 mV and duration ≥0.02 seconds in any lead. Two authors (A.M.Ø. and H.L.) independently assessed the extracted ECGs. Disagreement was resolved after discussion with an expert cardiologist (H.S.). We used the third universal definition of myocardial infarction20 to define prior MI on the ECG as (1) any Q wave in leads V2 to V3 ≥0.02 seconds or QS complex in leads V2 and V3; (2) Q wave ≥0.03 seconds or QS complex in any 2 leads of a contiguous lead grouping (I, aVL; V1–V6; II, III, aVF); or (3) R wave ≥0.04 seconds in V1 to V2 and R/S ≥1 with a concordant positive T wave in absence of conduction defect. We defined a Q wave as a negative deflection on the ECG with amplitude ≤−0.1 mV without any initial positive QRS deflection. We defined a QS wave as a negative deflection on the ECG with amplitude ≤−0.1 mV without any positive deflection in the QRS complex.
Myocardial Infarction
We used the ECGs and the end point registry of hospital admissions for MI to categorize the patients into 3 groups: (1) no MI, (2) unrecognized MI, and (3) recognized MI. We defined participants with unrecognized MI as those with findings of MI on the ECG in Tromsø 6 without any clinical event in the end point registry or a registered silent MI in the end point registry up to the date of examination (diagnosis of MI based on echocardiography, ECG, or radionuclide angiogram). We defined participants with recognized MI as those with a clinical event of definite or probable MI, defined as typical or atypical symptoms with either ECG findings of acute MI or elevated cardiac biomarkers.
Selection of Potential Confounding Variables
We selected age, diabetes mellitus, sex, hypertension, depression, anxiety, physical activity, and smoking as potential confounding variables. Age, diabetes mellitus, and female sex have previously been reported to be associated with increased risk for unrecognized MI6, 7, 10, 21, 22 and are also associated with pain sensitivity.23, 24, 25 Diabetes mellitus was defined as glycated hemoglobin >6.5 or use of antidiabetic medication. Hypertension is a risk factor for unrecognized MI,21 and an association between increasing blood pressure and hypoalgesia has been demonstrated.26, 27, 28 We modeled systolic blood pressure as a continuous variable and also included current use of blood pressure–lowering medication. We modeled hypertension (defined as systolic blood pressure >140 mm Hg, diastolic blood pressure >90 mm Hg or use of antihypertensive medication) as a dichotomous variable for interaction analyses. Depression and anxiety has been reported to be differently associated with unrecognized and recognized MI,29 and is associated with increased risk for pain disorders.30, 31 Depression/anxiety was measured by Hopkin symptom checklist 10‐item version, and modeled as a dichotomous variable (cutoff ≤1.85). Physical activity is considered protective of coronary heart disease32, 33 and was reported to relate to pain sensitivity.34 Physical activity was self‐reported and divided into 3 levels based on the participants’ answer to the question of whether their average physical activity in leisure time was limited to “reading, watching TV, or other sedentary activity,” “walking, cycling, or other forms of exercise at least 4 hours a week” (eg, walking or cycling to place of work, Sunday walking),” or “participation in recreational sports, heavy gardening, etc (note: duration of activity at least 4 hours a week).” It was modeled as a categorical variable. Smoking is an established cardiovascular risk factor that is also linked to pain sensitivity.35 Smoking was self‐reported and defined as “current daily smoker,” “former daily smoker,” or “never daily smoker” and modeled as a categorical variable.
Statistical Analyses and Data Management
We calculated descriptive statistics for 3 groups: participants with recognized MI, participants with unrecognized MI, and participants without MI. We used Pearson's chi‐square test to compare categorical variables and t test to compare continuous variables between unrecognized and recognized MI.
We used the Cox proportional hazard model to compare cold pressor tolerance between unrecognized and recognized MI. Since we could only study the association between cold pressor pain and MI and not causality, and because time to withdrawal of the hand is right‐censored data, we used time to withdrawal as the time to event in the Cox model. Data were right‐censored if the participant endured the cold pressor test to the maximum 106 seconds. Participants with no prior MI were excluded from the main analyses. MI was included as a binary variable (prior recognized MI, prior unrecognized MI). We used participants with recognized MI as the reference group. Hazard ratios (HRs) of aborting the cold pressor test were calculated with 95% CIs. Compared with the reference group, HRs <1 indicated higher tolerance, whereas HRs >1 indicated lower tolerance for pain. Potential confounding factors (listed in Table 1) were included in multivariable models. We examined interactions by adding cross‐product terms of MI group and each of the potential confounding variables to the model. Evaluation of Schoenfeld residuals and inspection of log‐log survival plots did not indicate that the proportional hazards assumption was violated. We performed additional analyses including participants without MI, using this group as a reference group. This was done to describe the relationship of pain sensitivity in the general population without MI with that of persons with unrecognized and recognized MI. All analyses were preplanned and performed in STATA (version 12.0, Stata Corp, College Station, TX).
Table 1.
Characteristics of the Study Population by MI Status––The Tromsø Study, 2007–2008
| No Prior MI (n=4235) | Unrecognized MI (n=387) | Recognized MI (n=227) | P Valuea | |
|---|---|---|---|---|
| Age, y | 62±9 | 64±8 | 68±8 | <0.001 |
| Women | 2482 (59) | 145 (37) | 49 (22) | <0.001 |
| Systolic blood pressure, mm Hg | 139±22 | 144±23 | 139±23 | 0.01 |
| Current use of blood pressure medication | 1032 (24) | 125 (32) | 127 (56) | <0.001 |
| Hypertensionb | 2380 (56) | 255 (66) | 168 (74) | 0.036 |
| Smoking habits | 0.002 | |||
| Current daily smoker | 789 (19) | 63 (17) | 38 (17) | |
| Former daily smoker | 1935 (46) | 195 (52) | 142 (64) | |
| Never daily smoker | 1511 (36) | 129 (33) | 47 (21) | |
| Diabetes mellitusc | 279 (7) | 35 (9) | 28 (12) | 0.20 |
| Physical activity | 0.65 | |||
| Sedentary lifestyle (reading, watching TV) | 698 (18) | 72 (21) | 41 (21) | |
| Walking, cycling, or other forms of exercise >4 h/wk | 2544 (66) | 218 (62) | 123 (63) | |
| Participation in recreational sports, heavy gardening, etc >4 h/wk | 651 (16) | 59 (17) | 30 (16) | |
| Psychological distress (HSCL‐10 score >1.85) | 483 (10) | 32 (8) | 25 (11) | 0.26 |
| Cold pressor tolerance <106 s | 1338 (32) | 95 (25) | 76 (33) | 0.02 |
| Medication use | ||||
| Antiplatelet drugs | 399 (9) | 73 (19) | 188 (83) | <0.01 |
| Anticoagulants | 81 (2) | 13 (3) | 32 (14) | <0.01 |
| Statins | 498 (12) | 82 (21) | 200 (88) | <0.01 |
| β‐Blockers | 418 (10) | 65 (17) | 172 (76) | <0.01 |
| ACEIs | 582 (14) | 79 (20) | 84 (37) | <0.01 |
| Weekly use of painkillers (with or without prescription) | 697 (16) | 42 (11) | 23 (10) | 0.78 |
Values are expressed as mean±SD or number (percentage). ACEIs indicates angiotensin‐converting enzyme inhibitors; HSCL‐10, Hopkin symptom checklist 10‐item version; MI, myocardial infarction.
t or chi‐square tests comparing unrecognized and recognized MI.
Defined as systolic blood pressure >140 mm Hg, diastolic blood pressure >90 mm Hg, or use of antihypertensive medication.
Defined as glycated hemoglobin >6.5 or use of antidiabetic medication.
We also calculated descriptive statistics for participants included (had undergone cold pressor test and had valid ECG) and excluded (not undergone cold pressor test or no valid ECG) from our analyses.
Results
Women had fewer MIs than men (7% versus 19%, P<0.001), but a larger proportion of MIs were unrecognized in women than in men (75% versus 58%, P<0.001). Unrecognized MI was present in 387 (8%) and recognized MI in 227 (4.7%) of the 4849 included participants. Baseline characteristics, by MI, are shown in Table 1.
Pain Tolerance and Presentation of MI
A total of 1509 participants (31%) aborted the cold pressor test before the maximum time of 106 seconds. Fewer participants with unrecognized MI aborted the cold pressor test compared with those with recognized MI (25% versus 33%, P<0.02). Figure 2 shows Kaplan–Meier curves for time to aborting the cold pressor test, by MI and sex. HRs for aborting the cold pressor test, by MI and sex, are shown in Table 2. Participants with unrecognized MI endured the cold pressor test significantly longer than participants with recognized MI (HR for aborting the cold pressor test, 0.64; CI, 0.47–0.88). After adjustment for additional potential confounding factors (mean systolic blood pressure, use of blood pressure–lowering drugs, diabetes mellitus, daily smoking, psychological distress, and physical activity), the association was attenuated and nonsignificant, but the direction of the effect was unaltered.
Figure 2.
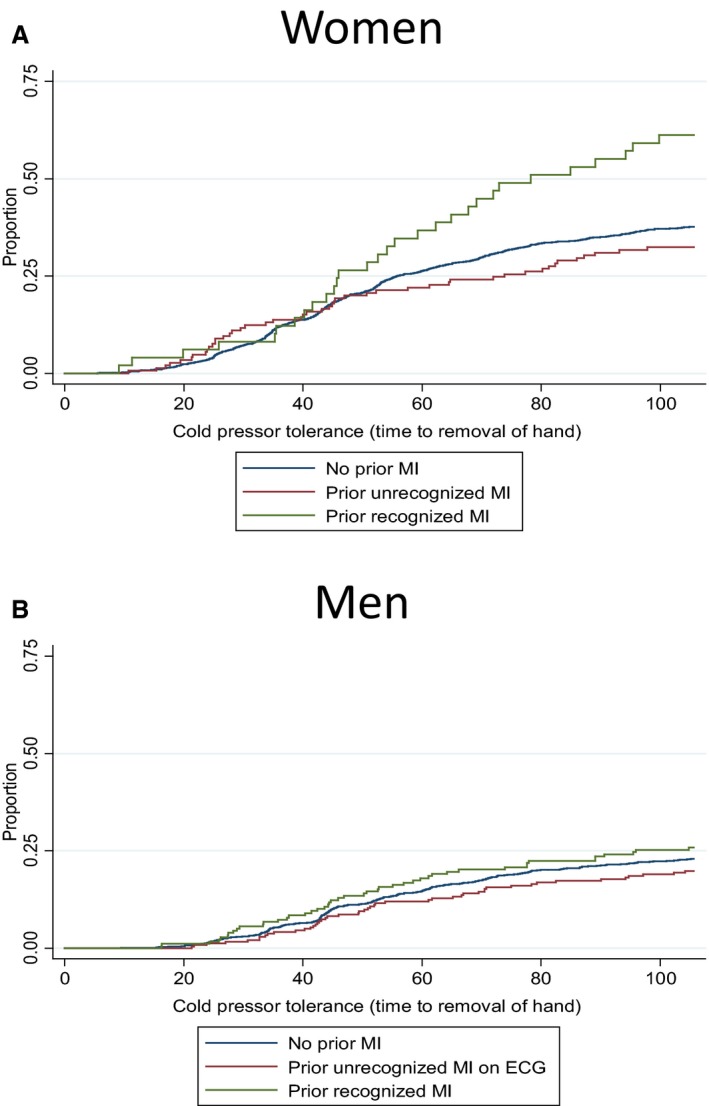
Kaplan–Meier plot for aborting the cold pressor test, by myocardial infarction (MI) status in women (A) and men (B) The Tromsø Study, 2007–2008.
Table 2.
HRs for Cold Pressor Tolerance (Aborted Cold Pressor Test), by MI Status and Sex––The Tromsø Study, 2007–2008
| Adjusted for Age and Sexa | Multivariable Adjustedb | |||
|---|---|---|---|---|
| HR | 95% CI | HR | 95% CI | |
| Women and men | ||||
| Prior recognized MI (n=227) | 1.00 | — | 1.00 | — |
| Prior unrecognized MI (n=387) | 0.64 | 0.47–0.88 | 0.68 | 0.46–1.00 |
| Women only | ||||
| Prior recognized MI (n=49) | 1.00 | — | 1.00 | — |
| Prior unrecognized MI (n=145) | 0.52 | 0.33–0.84 | 0.54 | 0.28–1.03 |
| Men only | ||||
| Prior recognized MI (n=178) | 1.00 | — | 1.00 | — |
| Prior unrecognized MI (n=242) | 0.75 | 0.49–1.12 | 0.81 | 0.50–1.31 |
HRs indicates hazard ratios; MI, myocardial infarction.
The analyses of both women and men were adjusted for sex.
Adjusted for sex, age, mean systolic blood pressure, use of blood pressure–lowering drugs, diabetes mellitus, daily smoking, psychological distress, and physical activity.
Sex Differences
More women aborted the cold pressor test compared with men (38% versus 23%, P<0.0001); however, the association between pain tolerance and infarct recognition was not significantly different in men and women (P for interaction=0.14). We also investigated whether the association between infarct recognition and pain tolerance varied with the potential confounders differently in men and women, and none of these 3‐way interactions were significant (results not shown).
Interaction Analyses
We did not find any statistically significant interaction between groups of MI and systolic blood pressure (P=0.77), hypertension (yes/no) (P=0.32), use of blood pressure–lowering drugs (P=0.10), diabetes mellitus (P=0.66), daily smoking (P=0.32), psychological distress (P=0.48), or physical activity (P=0.58) with regards to pain tolerance.
Additional Analyses
Table S1 shows additional analyses by adding participants without MI and using this group as the reference group. Participants with unrecognized MI did not endure the cold pressor test statistically significantly longer than participants without MI (HR for aborting the cold pressor test, 0.84; 95% CI, 0.68–1.03). Participants with recognized MI endured the cold pressor test significantly shorter than participants without MI (HR for aborting the cold pressor test, 1.30; 95% CI, 1.02–1.65).
Table S2 shows descriptive statistics for participants excluded and included in our analyses. The excluded participants were older (62±9 versus 66±10 years), more often women (55% versus 60%), hypertensive (58% versus 64%), and above the cutoff of 1.85 for the HSCL score for psychological distress (11% versus 14%).
Discussion
The main finding of the present study is that participants with unrecognized MI had higher pain tolerance compared with participants with recognized MI. Our results suggest that differences in pain sensitivity affect the perception of MI.
To our knowledge, no previous study has examined pain tolerance in persons with recognized and unrecognized MI. However, 2 studies have examined pain sensitivity in patients hospitalized in the acute phase of MI. One study of 92 MI patients found that patients with painful acute MI had increased sensitivity to heat pain compared with patients with painless acute MI.36 Another study of 67 persons with ST‐elevation MI found an association between conditioned pain modulation (a test of pain inhibition) and increased patient delay in seeking treatment, suggesting that more efficient pain inhibition reduced pain presentation in acute MI.37 Both studies examined hospitalized patients in the acute phase of MI and are not directly comparable to our study. However, they imply that increased pain tolerance is associated with less symptomatic MI.
Our findings propose that pain tolerance is associated with recognition of MI. It is plausible that ischemic myocardial pain is modulated through similar central processes as other pain modalities, such as cold pressor pain. This can partly explain our results. Experimental studies have shown that asymptomatic ischemia is associated with attenuated response to pain.38, 39, 40 Differences in central modulation of pain have been proposed as part of the explanation for absence of pain in silent ischemia. Activation of the thalamus is seen in both angina and silent ischemia, but activation of the frontal cortex seems to be necessary for the conscious sensation of myocardial pain.13, 41 Comparison of peripheral nerve conduction has shown similar conduction in patients with angina and silent myocardial ischemia, signifying that peripheral nerve transmission is not altered.40
It is possible that the size of MI and differences in pathophysiology also influence symptom severity. A recent study reported that coronary microvascular dysfunction was related to silent positive exercise testing in persons with normal coronary arteries,42 suggesting a different pathophysiology between silent and symptomatic myocardial ischemia. Studies also indicate that unrecognized MIs are smaller43, 44 and manifest less regional wall‐motion abnormalities45, 46 than recognized MIs. However, other studies show no association between pain ratings and the ischemic area at risk or MI size,37, 47, 48 suggesting that MI size cannot be reliably assessed by the patient's symptoms. In the present study, we did not have the opportunity to study these potential influencers on infarct recognition.
Additional analyses shown in Table S1 demonstrate that participants with unrecognized MI do not have a statistically significantly lower pain tolerance than those with no MI. It is, however, possible that persons with no MI have a pain tolerance intermediate to unrecognized and recognized MI, as Figure 2 indicates. However, we cannot exclude the possibility that it is patients with recognized MI who have an increased pain sensitivity compared with those without MI and with unrecognized MI. However, the main purpose of this article was to study differences between recognized and unrecognized MI.
Sex Differences
Women are more likely to present without chest pain or to have atypical symptoms in the setting of an acute MI2, 49 and the proportion of unrecognized MI is larger in women compared with men.8, 15, 50 Most studies of experimental pain show significant sex differences, although sometimes relatively minor and affected by numerous confounding variables.51, 52 When differences are observed, they consistently show that women have higher sensitivity and lower tolerance to pain than men.53, 54 It therefore seems contradictory that women have more unrecognized MIs.
As in previous studies, we found a higher proportion of unrecognized MIs in women and that women were less tolerant to pain than men. We found that the association between unrecognized MI and lower pain tolerance was stronger in women than in men, and statistically significant in women only, but the sex difference was not statistically significant (P for interaction=0.14). It is possible that the larger proportion of unrecognized MIs in women is explained by the fact that they are more likely to have coronary disease misdiagnosed or dismissed because of deficient knowledge and more difficult diagnostics, and not because they do not experience symptoms of the MI. In addition, it might also be that women recognized as having an MI are those most sensitive to pain, presenting with the most severe symptoms, and therefore are more likely to receive a diagnosis. This can potentially explain the stronger association in women, as we have investigated pain sensitivity in those with unrecognized MI relative to those with recognized MI.
Pain Sensitivity and Clinical Implications
This study contributes to increased awareness of unrecognized MI. More specifically, it contributes to the knowledge that pain tolerance affects the presentation of MI. Absence of chest pain should not lower alertness of doctors towards ischemic heart disease. Questions on pain sensitivity or factors that affect this might be important in the assessment of patients at risk for cardiovascular disease. ECG is a low‐cost and widely available investigative method and should be considered in persons with high cardiovascular risk despite no history of chest pain. Pain is a common symptom when seeking medical assistance and is a crucial factor in how health workers recognize and assess the severity of a disease. An increasing body of evidence suggests that pain sensitivity modulates the clinical expression of disease.55, 56 The present study adds to this by showing that pain sensitivity may be of importance in the recognition of MI and contributes with an important aspect to the further research of underlying reasons for unrecognized MI. We encourage researchers of future studies in this field to consider including variables associated with pain sensitivity.
Strengths and Limitations
Strengths of the present study include the population‐based design, large sample size, rigorous validation of previous recognized MI, and that the technicians were blinded to MI status. There are also some limitations. First, as Q‐wave criteria were used to identify those with unrecognized MI, there is probably a larger proportion of non–Q‐wave MIs among participants with recognized compared with unrecognized MI. It is unknown whether this may have affected the results. Second, we do not know whether participants with unrecognized MI experienced symptoms or whether the MIs were truly silent. Third, more women with increased risk factor levels were excluded from the analyses because they had not undergone the cold pressor test or had no valid ECG. Our main results indicate that the ability of this study to detect differences in pain tolerance between persons with unrecognized and recognized MI, and possibly sex differences, were attenuated because of these differences. Fourth, the cross‐sectional design precludes causal inference. Last, the external validity refers to Caucasian middle‐aged and elderly adults and may not be generalizable to other groups.
Conclusions
Our findings suggest that persons who experience unrecognized MI have reduced pain sensitivity compared with persons who experience recognized MI, adjusting for age and sex. This may partially explain the lack of symptoms associated with unrecognized MI.
Sources of Funding
This work was funded by the Norwegian National Advisory Unit on Women's Health, Oslo University Hospital, Oslo, Norway.
Disclosures
None.
Supporting information
Table S1. Hazard Ratios (HRs) for Cold Pressor Tolerance (Aborted Cold Pressor Test) With No Prior Myocardial Infarction (MI) as Reference, by MI Status and Sex––The Tromsø Study, 2007–2008
Table S2. Characteristics of Participants Included and Excluded From Analyses––The Tromsø Study, 2007–2008
Acknowledgments
The authors would like to thank all of the participants and technicians of the Tromsø Study.
(J Am Heart Assoc. 2016;5:e003846 doi: 10.1161/JAHA.116.003846)
References
- 1. Herrick JB. Clinical features of sudden obstruction of the coronary arteries. JAMA. 1983;250:1757–1765. [PubMed] [Google Scholar]
- 2. Khan NA, Daskalopoulou SS, Karp I, Eisenberg MJ, Pelletier R, Tsadok MA, Dasgupta K, Norris CM, Pilote L, Team GP. Sex differences in acute coronary syndrome symptom presentation in young patients. JAMA Intern Med. 2013;173:1863–1871. [DOI] [PubMed] [Google Scholar]
- 3. Sigurdsson E, Thorgeirsson G, Sigvaldason H, Sigfusson N. Unrecognized myocardial infarction: epidemiology, clinical characteristics, and the prognostic role of angina pectoris. The Reykjavik Study. Ann Intern Med. 1995;122:96–102. [DOI] [PubMed] [Google Scholar]
- 4. Canto JG, Shlipak MG, Rogers WJ, Malmgren JA, Frederick PD, Lambrew CT, Ornato JP, Barron HV, Kiefe CI. Prevalence, clinical characteristics, and mortality among patients with myocardial infarction presenting without chest pain. JAMA. 2000;283:3223–3229. [DOI] [PubMed] [Google Scholar]
- 5. Pride YB, Piccirillo BJ, Gibson CM. Prevalence, consequences, and implications for clinical trials of unrecognized myocardial infarction. Am J Cardiol. 2013;111:914–918. [DOI] [PubMed] [Google Scholar]
- 6. Valensi P, Lorgis L, Cottin Y. Prevalence, incidence, predictive factors and prognosis of silent myocardial infarction: a review of the literature. Arch Cardiovasc Dis. 2011;104:178–188. [DOI] [PubMed] [Google Scholar]
- 7. Sheifer SE, Gersh BJ, Yanez ND III, Ades PA, Burke GL, Manolio TA. Prevalence, predisposing factors, and prognosis of clinically unrecognized myocardial infarction in the elderly. J Am Coll Cardiol. 2000;35:119–126. [DOI] [PubMed] [Google Scholar]
- 8. Dehghan A, Leening MJ, Solouki AM, Boersma E, Deckers JW, van Herpen G, Heeringa J, Hofman A, Kors JA, Franco OH, Ikram MA, Witteman JC. Comparison of prognosis in unrecognized versus recognized myocardial infarction in men versus women >55 years of age (from the Rotterdam Study). Am J Cardiol. 2014;113:1–6. [DOI] [PubMed] [Google Scholar]
- 9. Kannel WB, Abbott RD. Incidence and prognosis of unrecognized myocardial infarction. An update on the Framingham Study. N Engl J Med. 1984;311:1144–1147. [DOI] [PubMed] [Google Scholar]
- 10. Kannel WB, Cupples LA, Gagnon DR. Incidence, precursors and prognosis of unrecognized myocardial infarction. Adv Cardiol. 1990;37:202–214. [DOI] [PubMed] [Google Scholar]
- 11. Droste C, Roskamm H. Experimental pain measurement in patients with asymptomatic myocardial ischemia. J Am Coll Cardiol. 1983;1:940–945. [DOI] [PubMed] [Google Scholar]
- 12. Droste C, Greenlee MW, Roskamm H. A defective angina pectoris pain warning system: experimental findings of ischemic and electrical pain test. Pain. 1986;26:199–209. [DOI] [PubMed] [Google Scholar]
- 13. Rosen SD, Paulesu E, Nihoyannopoulos P, Tousoulis D, Frackowiak RS, Frith CD, Jones T, Camici PG. Silent ischemia as a central problem: regional brain activation compared in silent and painful myocardial ischemia. Ann Intern Med. 1996;124:939–949. [DOI] [PubMed] [Google Scholar]
- 14. Di Franco A, Lanza GA, Valeriani M, Villano A, Russo G, Virdis D, Pazzaglia C, Sarullo FM, Rossini PM, Crea F, Vollono C. Impaired pain processing in patients with silent myocardial ischemia. Int J Cardiol. 2015;186:204–206. [DOI] [PubMed] [Google Scholar]
- 15. de Torbal A, Boersma E, Kors JA, van Herpen G, Deckers JW, van der Kuip DAM, Stricker BH, Hofman A, Witteman JCM. Incidence of recognized and unrecognized myocardial infarction in men and women aged 55 and older: the Rotterdam Study. Eur Heart J. 2006;27:729–736. [DOI] [PubMed] [Google Scholar]
- 16. Jacobsen BK, Eggen AE, Mathiesen EB, Wilsgaard T, Njolstad I. Cohort profile: the Tromso Study. Int J Epidemiol. 2012;41:961–967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Johansen A, Schirmer H, Stubhaug A, Nielsen CS. Persistent post‐surgical pain and experimental pain sensitivity in the Tromso study: comorbid pain matters. Pain. 2014;155:341–348. [DOI] [PubMed] [Google Scholar]
- 18. Chen AC, Dworkin SF, Haug J, Gehrig J. Human pain responsivity in a tonic pain model: psychological determinants. Pain. 1989;37:143–160. [DOI] [PubMed] [Google Scholar]
- 19. Klement WAJ. The role of nociceptorsof cutaneous veins in the mediation of cold pain in man. J Physiol. 1992;449:73–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Thygesen K, Alpert JS, Jaffe AS, Simoons ML, Chaitman BR, White HD; Joint ESC/ACCF/AHA/WHF Task Force for Universal Definition of Myocardial Infarction.; Authors/Task Force Members Chairpersons , Thygesen K, Alpert JS, White HD; Biomarker Subcommittee , Jaffe AS, Katus HA, Apple FS, Lindahl B, Morrow DA; ECG Subcommittee , Chaitman BR, Clemmensen PM, Johanson P, Hod H; Imaging Subcommittee , Underwood R, Bax JJ, Bonow JJ, Pinto F, Gibbons RJ; Classification Subcommittee , Fox KA, Atar D, Newby LK, Galvani M, Hamm CW; Intervention Subcommittee , Uretsky BF, Steg PG, Wijns W, Bassand JP, Menasche P, Ravkilde J; Trials and Registries Subcommittee , Ohman EM, Antman EM, Wallentin LC, Armstrong PW, Simoons ML; Trials and Registries Subcommittee , Januzzi JL, Nieminen MS, Gheorghiade M, Filippatos G; Trials and Registries Subcommittee , Luepker RV, Fortmann SP, Rosamond WD, Levy D, Wood D; Trials and Registries Subcommittee , Smith SC, Hu D, Lopez‐Sendon JL, Robertson RM, Weaver D, Tendera M, Bove AA, Parkhomenko AN, Vasilieva EJ, Mendis S; ESC Committee for Practice Guidelines (CPG) , Bax JJ, Baumgartner H, Ceconi C, Dean V, Deaton C, Fagard R, Funck‐Brentano C, Hasdai D, Hoes A, Kirchhof P, Knuuti J, Kolh P, McDonagh T, Moulin C, Popescu BA, Reiner Z, Sechtem U, Sirnes PA, Tendera M, Torbicki A, Vahanian A, Windecker S; Document Reviewers , Morais J, Aguiar C, Almahmeed W, Arnar DO, Barili F, Bloch KD, Bolger AF, Botker HE, Bozkurt B, Bugiardini R, Cannon C, de Lemos J, Eberli FR, Escobar E, Hlatky M, James S, Kern KB, Moliterno DJ, Mueller C, Neskovic AN, Pieske BM, Schulman SP, Storey RF, Taubert KA, Vranckx P, Wagner DR. Third universal definition of myocardial infarction. J Am Coll Cardiol. 2012;60:1581–1598.22958960 [Google Scholar]
- 21. McAreavey D, Vidal JS, Aspelund T, Eiriksdottir G, Schelbert EB, Kjartansson O, Cao JJ, Thorgeirsson G, Sigurdsson S, Garcia M, Harris TB, Launer LJ, Gudnason V, Arai AE. Midlife cardiovascular risk factors and late‐life unrecognized and recognized myocardial infarction detect by cardiac magnetic resonance: ICELAND‐MI, the AGES‐Reykjavik Study. J Am Heart Assoc. 2016;5:e002420 doi: 10.1161/JAHA.115.002420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Nadelmann J, Frishman WH, Ooi WL, Tepper D, Greenberg S, Guzik H, Lazar EJ, Heiman M, Aronson M. Prevalence, incidence and prognosis of recognized and unrecognized myocardial infarction in persons aged 75 years or older: the Bronx Aging Study. Am J Cardiol. 1990;66:533–537. [DOI] [PubMed] [Google Scholar]
- 23. Petrini L, Matthiesen ST, Arendt‐Nielsen L. The effect of age and gender on pressure pain thresholds and suprathreshold stimuli. Perception. 2015;44:587–596. [DOI] [PubMed] [Google Scholar]
- 24. Fillingim RB. Sex, gender, and pain: women and men really are different. Curr Rev Pain. 2000;4:24–30. [DOI] [PubMed] [Google Scholar]
- 25. Bierhaus A, Haslbeck KM, Humpert PM, Liliensiek B, Dehmer T, Morcos M, Sayed AA, Andrassy M, Schiekofer S, Schneider JG, Schulz JB, Heuss D, Neundorfer B, Dierl S, Huber J, Tritschler H, Schmidt AM, Schwaninger M, Haering HU, Schleicher E, Kasper M, Stern DM, Arnold B, Nawroth PP. Loss of pain perception in diabetes is dependent on a receptor of the immunoglobulin superfamily. J Clin Invest. 2004;114:1741–1751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Ghione S, Rosa C, Mezzasalma L, Panattoni E. Arterial hypertension is associated with hypalgesia in humans. Hypertension. 1988;12:491–497. [DOI] [PubMed] [Google Scholar]
- 27. Ghione S. Hypertension‐associated hypalgesia. Evidence in experimental animals and humans, pathophysiological mechanisms, and potential clinical consequences. Hypertension. 1996;28:494–504. [DOI] [PubMed] [Google Scholar]
- 28. Olsen RB, Bruehl S, Nielsen CS, Rosseland LA, Eggen AE, Stubhaug A. Gender differences in blood pressure‐related hypoalgesia in a general population: the Tromso Study. J Pain. 2013;14:699–708. [DOI] [PubMed] [Google Scholar]
- 29. Jovanova O, Luik AI, Leening MJ, Noordam R, Aarts N, Hofman A, Franco OH, Dehghan A, Tiemeier H. The long‐term risk of recognized and unrecognized myocardial infarction for depression in older men. Psychol Med. 2016;46:1951–1960. [DOI] [PubMed] [Google Scholar]
- 30. de Heer EW, Gerrits MM, Beekman AT, Dekker J, van Marwijk HW, de Waal MW, Spinhoven P, Penninx BW, van der Feltz‐Cornelis CM. The association of depression and anxiety with pain: a study from NESDA. PLoS One. 2014;9:e106907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Gerrits MM, van Marwijk HW, van Oppen P, van der Horst H, Penninx BW. Longitudinal association between pain, and depression and anxiety over four years. J Psychosom Res. 2015;78:64–70. [DOI] [PubMed] [Google Scholar]
- 32. Manson JE, Tosteson H, Ridker PM, Satterfield S, Hebert P, O'Connor GT, Buring JE, Hennekens CH. The primary prevention of myocardial infarction. N Engl J Med. 1992;326:1406–1416. [DOI] [PubMed] [Google Scholar]
- 33. Ahmad T, Chasman DI, Buring JE, Lee IM, Ridker PM, Everett BM. Physical activity modifies the effect of LPL, LIPC, and CETP polymorphisms on HDL‐C levels and the risk of myocardial infarction in women of European ancestry. Circ Cardiovasc Genet. 2011;4:74–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Ellingson LD, Colbert LH, Cook DB. Physical activity is related to pain sensitivity in healthy women. Med Sci Sports Exerc. 2012;44:1401–1406. [DOI] [PubMed] [Google Scholar]
- 35. Shi Y, Weingarten TN, Mantilla CB, Hooten WM, Warner DO. Smoking and pain: pathophysiology and clinical implications. Anesthesiology. 2010;113:977–992. [DOI] [PubMed] [Google Scholar]
- 36. Granot M, Khoury R, Berger G, Krivoy N, Braun E, Aronson D, Azzam ZS. Clinical and experimental pain perception is attenuated in patients with painless myocardial infarction. Pain. 2007;133:120–127. [DOI] [PubMed] [Google Scholar]
- 37. Granot M, Dagul P, Darawsha W, Aronson D. Pain modulation efficiency delays seeking medical help in patients with acute myocardial infarction. Pain. 2015;156:192–198. [DOI] [PubMed] [Google Scholar]
- 38. Sheps DS, McMahon RP, Light KC, Maixner W, Pepine CJ, Cohen JD, Goldberg AD, Bonsall R, Carney R, Stone PH, Sheffield D, Kaufmann PG. Low hot pain threshold predicts shorter time to exercise‐induced angina: results from the psychophysiological investigations of myocardial ischemia (PIMI) study. J Am Coll Cardiol. 1999;33:1855–1862. [DOI] [PubMed] [Google Scholar]
- 39. Falcone C, Auguadro C, Sconocchia R, Catalano O, Ochan M, Angoli L, Montemartini C. Susceptibility to pain during coronary angioplasty: usefulness of pulpal test. J Am Coll Cardiol. 1996;28:903–909. [DOI] [PubMed] [Google Scholar]
- 40. Langer A, O'Connor P. Central modulation of pain perception in patients with silent myocardial ischemia. Am J Cardiol. 1994;74:182–184. [DOI] [PubMed] [Google Scholar]
- 41. Rosen SD, Paulesu E, Frith CD, Frackowiak RS, Davies GJ, Jones T, Camici PG. Central nervous pathways mediating angina pectoris. Lancet. 1994;344:147–150. [DOI] [PubMed] [Google Scholar]
- 42. Di Franco A, Lanza GA, Di Monaco A, Sestito A, Lamendola P, Nerla R, Tarzia P, Virdis D, Vollono C, Valeriani M, Crea F. Coronary microvascular function and cortical pain processing in patients with silent positive exercise testing and normal coronary arteries. Am J Cardiol. 2012;109:1705–1710. [DOI] [PubMed] [Google Scholar]
- 43. Cabin HS, Roberts WC. Quantitative comparison of extent of coronary narrowing and size of healed myocardial infarct in 33 necropsy patients with clinically recognized and in 28 with clinically unrecognized (“silent”) previous acute myocardial infarction. Am J Cardiol. 1982;50:677–681. [DOI] [PubMed] [Google Scholar]
- 44. Arenja N, Mueller C, Ehl NF, Brinkert M, Roost K, Reichlin T, Sou SM, Hochgruber T, Osswald S, Zellweger MJ. Prevalence, extent, and independent predictors of silent myocardial infarction. Am J Med. 2013;126:515–522. [DOI] [PubMed] [Google Scholar]
- 45. Ammar KA, Makwana R, Jacobsen SJ, Kors JA, Burnett JC, Redfield MM, Yawn BP, Rodeheffer RJ. Impaired functional status and echocardiographic abnormalities signifying global dysfunction enhance the prognostic significance of previously unrecognized myocardial infarction detected by electrocardiography. Ann Noninvasive Electrocardiol. 2007;12:27–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Ammar KA, Samee S, Makwana R, Urban L, Mahoney DW, Kors JA, Redfield MM, Jacobsen S, Rodeheffer RJ. Echocardiographic characteristics of electrocardiographically unrecognized myocardial infarctions in a community population. Am J Cardiol. 2005;96:1069–1075. [DOI] [PubMed] [Google Scholar]
- 47. Christian TF, Gibbons RJ, Hopfenspirger MR, Gersh BJ. Severity and response of chest pain during thrombolytic therapy for acute myocardial infarction: a useful indicator of myocardial salvage and infarct size. J Am Coll Cardiol. 1993;22:1311–1316. [DOI] [PubMed] [Google Scholar]
- 48. Keele KD. Pain complaint threshold in relation to pain of cardiac infarction. Br Med J. 1968;1:670–673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Goldberg RJ, O'Donnell C, Yarzebski J, Bigelow C, Savageau J, Gore JM. Sex differences in symptom presentation associated with acute myocardial infarction: a population‐based perspective. Am Heart J. 1998;136:189–195. [DOI] [PubMed] [Google Scholar]
- 50. Murabito JM, Evans JC, Larson MG, Levy D. Prognosis after the onset of coronary heart disease. An investigation of differences in outcome between the sexes according to initial coronary disease presentation. Circulation. 1993;88:2548–2555. [DOI] [PubMed] [Google Scholar]
- 51. Unruh AM. Gender variations in clinical pain experience. Pain. 1996;65:123–167. [DOI] [PubMed] [Google Scholar]
- 52. Berkley KJ. Sex differences in pain. Behav Brain Sci. 1997;20:371–380; discussion 435‐513 [DOI] [PubMed] [Google Scholar]
- 53. Mogil JS. Sex differences in pain and pain inhibition: multiple explanations of a controversial phenomenon. Nat Rev Neurosci. 2012;13:859–866. [DOI] [PubMed] [Google Scholar]
- 54. Fillingim RB, King CD, Ribeiro‐Dasilva MC, Rahim‐Williams B, Riley JL III. Sex, gender, and pain: a review of recent clinical and experimental findings. J Pain. 2009;10:447–485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Stabell N, Stubhaug A, Flaegstad T, Mayer E, Naliboff BD, Nielsen CS. Widespread hyperalgesia in adolescents with symptoms of irritable bowel syndrome: results from a large population‐based study. J Pain. 2014;15:898–906. [DOI] [PubMed] [Google Scholar]
- 56. King CD, Sibille KT, Goodin BR, Cruz‐Almeida Y, Glover TL, Bartley E, Riley JL, Herbert MS, Sotolongo A, Schmidt J, Fessler BJ, Redden DT, Staud R, Bradley LA, Fillingim RB. Experimental pain sensitivity differs as a function of clinical pain severity in symptomatic knee osteoarthritis. Osteoarthritis Cartilage. 2013;21:1243–1252. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1. Hazard Ratios (HRs) for Cold Pressor Tolerance (Aborted Cold Pressor Test) With No Prior Myocardial Infarction (MI) as Reference, by MI Status and Sex––The Tromsø Study, 2007–2008
Table S2. Characteristics of Participants Included and Excluded From Analyses––The Tromsø Study, 2007–2008


