Abstract
Background
Acute promyelocytic leukemia (APL) is a disease characterized by expression of Promyelocytic Leukemia–Retinoic Acid Receptor α (PML-RARα) chimeric mRNA. Although APL is curable, early death due to hemorrhage is a major problem. Here, we report the development of a simple and rapid diagnostic method for APL based on reverse transcription loop-mediated isothermal amplification (RT-LAMP).
Methods
An RT-LAMP primer set was designed to detect three types of PML-RARα mRNA in a single reaction. Serial dilutions of plasmid DNA containing bcr1, bcr2, or bcr3 PML-RARα sequences and RNA extracted from bone marrow aspirates of 6 patients with APL were used to compare the results of RT-LAMP and nested PCR assays.
Results
Plasmid DNA was amplified by RT-LAMP, for which the reaction time was > 4 h shorter and the lower detection limit was higher than for nested RT-PCR. Six of 7 samples tested positive by both methods.
Conclusion
We developed an RT-LAMP assay for simple and rapid PML-RARα mRNA detection that may be clinically useful for point-of-care testing and APL diagnosis.
Keywords: acute promyelocytic leukemia, polymerase chain reaction, Promyelocytic Leukemia–Retinoic Acid Receptor α, reverse transcription loop-mediated isothermal amplification, t(15;17) chromosome translocation
Loop-mediated isothermal amplification (LAMP) is an established nucleic acid amplification method that requires a set of 4 specially designed, highly specific inner and outer primers.1 Additional loop primers can further accelerate the reaction.2 The LAMP product is detected by turbidity arising from magnesium pyrophosphate formed during the reaction.3 This method is very rapid and directly amplifies the target region from an RNA sample, and has been used to detect various pathogens, including bacteria and viruses.4 In addition, LAMP has now been developed as commercial kits, some of which have been adopted as officially recommended methods for routine pathogen examination and surveillance in Japan.5 However, there have been few applications of LAMP to the field of hematological malignancy, although one notable exception is the detection of Wilms’ tumor-1 gene mRNA.6
Acute promyelocytic leukemia (APL) is a distinct subtype of acute myeloid leukemia (AML) accounting for approximately 10% of cases. APL is cytogenetically characterized by balanced reciprocal translocation between chromosomes 15 and 17, which results in the fusion of the promyelocytic leukemia (PML) and retinoic acid receptor α (RARα) genes.7 The PML-RARα chimeric gene is detected in nearly all APL patients and is used as a marker for clinical diagnosis. In addition, the mRNA is detected by reverse transcription polymerase chain reaction (RT-PCR) and/or real time RT-PCR for minimal residual disease monitoring of APL.8–10 Three different types of PML-RARα mRNA exist owing to different breakpoints in the PML gene, although these do not result in different treatment outcomes: the long-, variable-, and short-form types are derived from breakpoints at bcr1 (intron 5), bcr2 (exon 6), and bcr3 (intron 3), respectively, and occur at relative frequencies of 55%, 5%, and 40%, respectively.11
APL is the most curable form of AML because of an established molecular targeted therapy combining all-trans retinoic acid (ATRA) and arsenic trioxide.12 Before starting the treatment, it is necessary to verify that the patient has the PML-RARα fusion gene.13 However, early death—mostly due to severe hemorrhage—remains the main cause of APL treatment failure according to recent population-based studies carried out in developed countries.14 Some reports recommend early and timely initiation of ATRA and provision of aggressive supportive care at the first suspicion of the disease, without awaiting molecular confirmation, in order to reduce hemorrhagic deaths in APL.14 An improved PML-RARα mRNA detection test is also needed.
We report here the development of a simple reverse transcription loop-mediated isothermal amplification (RT-LAMP) method for rapid PML-RARα mRNA detection. The advantages of this method with respect to RT-PCR are also discussed.
MATERIALS AND METHODS
Samples
Three different plasmids—containing bcr1, bcr2, and bcr3 PML-RARα mRNA sequences—were purchased from Eurofins Genomics (Louisville, KY) for use as positive controls. Vector plasmid was pTAKN-2 and chimeric sequences were made of sequences derived from the Genbank sequence database (Accession numbers are M73778, NM_000964, and AB067754). IVS-0035 RNA (Invivoscribe Technologies, San Diego, CA) was used as a negative control. Clinical samples used in this study were archived specimens from APL patients diagnosed by detection of APL cells and PML-RARα mRNA by nested RT-PCR at our hospital. RNA was extracted from patient bone marrow aspirates using the RNeasy Plus Mini kit (Qiagen, Valencia, CA) according to the manufacturer’s instructions; these were used for diagnosis, and then frozen at −80 °C until the study. RNA concentrations were measured and 260/280 nm absorbance ratios were determined with a Nanodrop 2000 spectrophotometer (Thermo Fisher Scientific, Waltham, MA) prior to RT-LAMP and nested RT-PCR assays. The study protocol was approved by the Tottori University Hospital ethics committee (number 2326).
Primer design
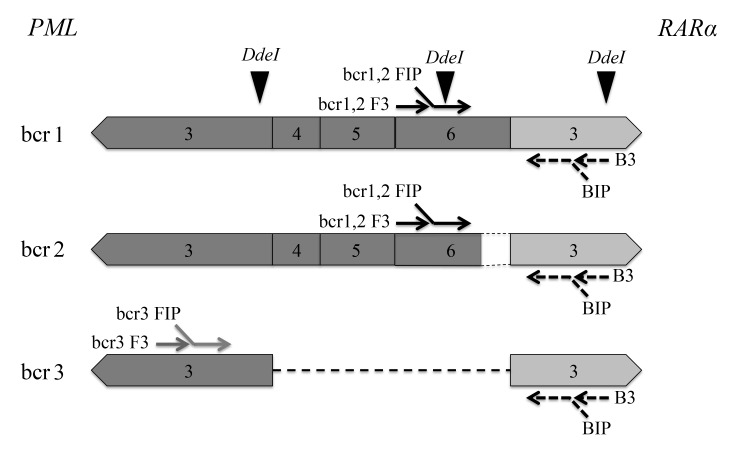
Primer sets for detection of PML-RARα mRNA by RT-LAMP (Table 1) were designed using PrimerExplorer ver. 4 software (Eiken Chemical, Tokyo, Japan). The primer sets were modified to detect the three types of PML-RARα mRNA in a single tube—i.e., bcr1 and bcr2 were amplified with the same primer sets and an additional set consisting of FIP, F3, and LF were included in one reaction to amplify bcr3 (Fig. 1).
Table 1.
Primers for detection of PML-RARα mRNA by RT-LAMP
| Primers | Sequence (5’ → 3’) |
| bcr1,2 FIP | TCCCCTCCTCAGACTCCATCTTGAAGAGGAAGTGCAGCCAGA |
| bcr1,2 F3 | CTCCAATACAACGACAGCC |
| bcr3 FIP | TTGAACTCGTCGAAGCCATCGGCTGGACATGCACGGTT |
| bcr3 F3 | AAGTGCTACGCCTCGGA |
| BIP | TGAGACCCAGAGCAGCAGTTGTAGATGCGGGGTAGA |
| B3 | TGACAGACAAAGCAAGGC |
| bcr1,2 LF | GATGACCTTCCTGGGGCA |
| bcr3 LF | ACGGCAGCTTGCAGGCT |
| LB | TGAAGAGATAGTGCCCAGCCCT |
PML-RARα, Promyelocytic Leukemia–Retinoic Acid Receptor α; RT-LAMP, reverse transcription loop-mediated isothermal amplification.
Fig. 1.
Schematic representation of primer design for detection of PML-RARα mRNA by RT-LAMP and cutting sites of DdeI restriction enzyme assay. Exon numbers of the PML gene (dark) and the RARα gene (light) are shown. PML, promyelocytic leukemia; RARα, retinoic acid receptor α; RT-LAMP, reverse transcription loop-mediated isothermal amplification.
RT-LAMP
The RT-LAMP assay was carried out using the Loopamp RNA Amplification kit (Eiken Chemical, Tokyo, Japan) under the following conditions: 1 μL sample (RNA or DNA) was mixed with 20 pmol each of FIP and BIP primers, 10 pmol each of LF and LB primers, 2.5 pmol each of F3 and B3 primers, 1 μL Enzyme Mix, and 12.5 μL of 2 × Reaction Mix, with distilled water added to obtain a final volume of 25 μL. For real-time turbidity monitoring, the reaction mixture was incubated at 68 °C for 60 min in an RT-160C Loopamp Realtime Turbidimeter (Eiken Chemical). The specificity of LAMP was confirmed by restriction enzyme digestion using DdeI (Takara Bio, Otsu, Japan) under the following conditions: 5 μL LAMP product was mixed with 2 μL of 10 × K buffer (Takara Bio), 1 μL DdeI, with distilled water added to obtain a final volume of 20 μL, followed by incubation at 37 °C for 120 min. DdeI was inactivated at 70 °C for 15 min and the digestion products (10-μL aliquots) were mixed with loading buffer (Nippon Gene, Toyama, Japan) and separated by 1.5% agarose gel electrophoresis (100 V, 35 min) with 0.005% ethidium bromide using a Mupid mini gel electrophoresis system (Advance, Tokyo, Japan), with 5 μL Gene Ladder 100 (Nippon Gene) used as a molecular size marker.
Nested RT-PCR
The RT reaction was carried out using 1 μL RNA sample and the ThermoScript RT-PCR system (Thermo Fisher Scientific) according to the manufacturers’ protocol. Three types of PML-RARα mRNA were detected using two forward primer sets designed according to published sequences.10 Specifically, bcr1 and bcr2 types were amplified with the same primer set. The PCR reaction mixture was prepared using AmpliTaq Gold with 10 × PCR Gold Buffer and MgCl2 (Thermo Fisher Scientific) and the GeneAmp dNTP mix (Thermo Fisher Scientific) under the following conditions: 1 μL plasmid DNA, 2 μL cDNA, or 1 μL first PCR product was mixed with 2.5 μL 10 × PCR Gold buffer, 2.25 nmol MgCl2 solution, 500 pmol dNTP mix, 0.2 μL AmpliTaq Gold DNA polymerase, and 0.25 pmol each forward and reverse primers, with distilled water added to obtain a final volume of 25 μL. The forward primers were as follows: 5’-AGTCAGTGCCCGGGGCACAC-3’ for bcr1 and bcr2 and 5’-AGCTGCTGGAGGCTGTGGACG-3’ for bcr3 in the first PCR; and 5’-AGTGTACGCCTTCTCCATCAAAG-3’ for bcr1 and bcr2 and 5’-TGTGCTGCAGCGACTCCGCA-3’ for bcr3 in the second PCR. The reverse primers were as follows: 5’-AGGGCTGGGCACTATCTCTTC-3’ for bcr1, bcr2, and bcr3 in the first PCR; and 5’-CAGAACTGCTGCTCTGGGTCTCAAT-3’ for bcr1, bcr2, and bcr3 in the second PCR. Reaction conditions on the MultiGene Mini Personal Thermal Cycler (Labnet International, Edison, NJ) were as follows: 94 °C for 5 min; 35 cycles at 94 °C for 30 s, 62 °C for 1 min, and 72 °C for 1 min; and 72 °C for 7 min. The nested PCR products (10-μL aliquots) were mixed with loading buffer and separated along with 5 μL of molecular size marker by 1.5% agarose gel electrophoresis (100 V, 35 min) with 0.005% ethidium bromide.
RNA integrity number (RIN) analysis
The quality of clinical RNA samples (expect for S-4, for which the amount was insufficient) was evaluated by RIN as previously described15 on an Agilent 2100 Bioanalyzer (Agilent Technologies, Santa Clara, CA).
RESULTS
Specificity of amplification by RT-LAMP
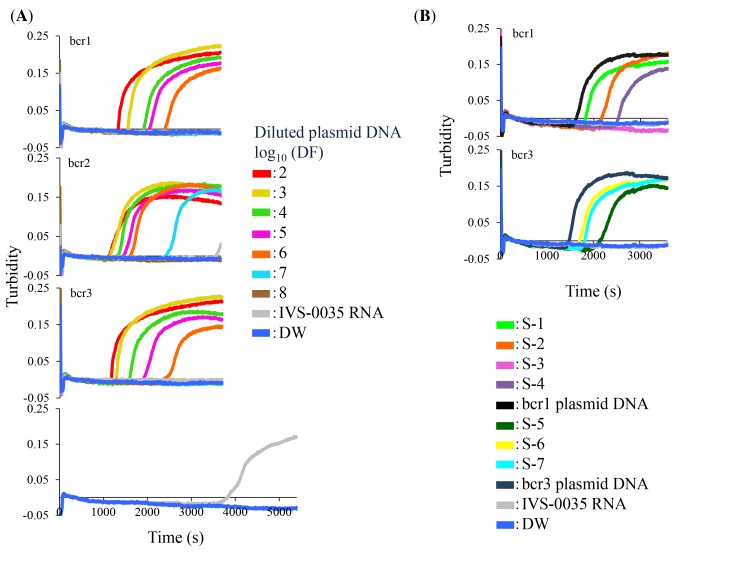
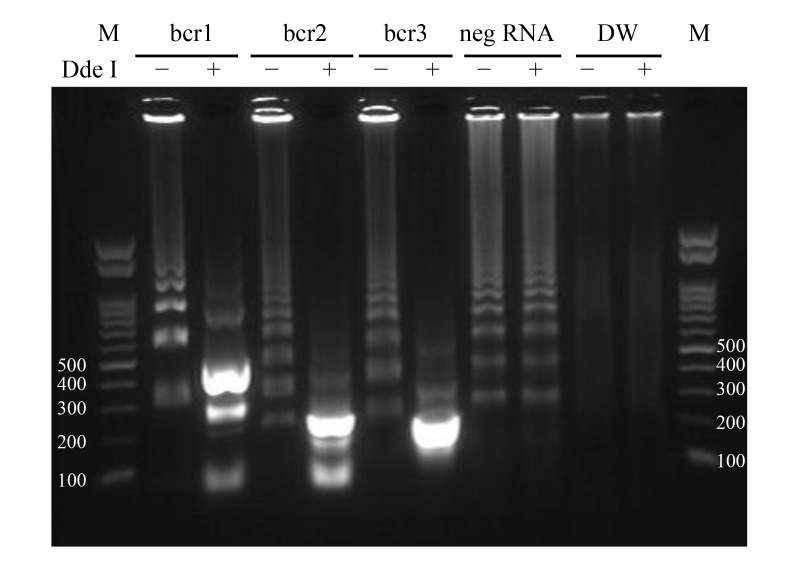
We performed the RT-LAMP assay with two concentrations of plasmid DNA followed by DdeI restriction enzyme analysis. All plasmid DNAs were detected by RT-LAMP in 60 min (data not shown, Fig. 2A) and yielded the expected products after digestion with DdeI (Fig. 3), which differed for each breakpoint. Non-specific amplification products obtained after 60 min from PML-RARα-negative RNA were not digestible by DdeI (Fig. 3).
Fig. 2.
Detection of PML-RARα in plasmid DNA and clinical RNA samples by RT-LAMP. (A) Ten-fold serial dilutions of bcr1, bcr2, and bcr3 plasmid DNAs were used as template; the starting amounts were 8.8, 7.8, and 8.0 × 108 copies/tube, respectively. DF of the indicated plasmid DNA is shown as a logarithmic value. (B) Clinical samples from APL patients (S-1 through -7) were analyzed by RT-LAMP. IVS-0035 RNA and distilled water were used as negative controls. A 100-fold dilution of corresponding plasmid DNAs served as a positive control. The amplification reaction was carried out at 68 °C for indicated times. APL, acute promyelocytic leukemia; DF, dilution factor; PML-RARα, Promyelocytic Leukemia–Retinoic Acid Receptor α; RT-LAMP, reverse transcription loop-mediated isothermal amplification.
Fig. 3.
DdeI restriction enzyme digestion of RT-LAMP products. Products of bcr1, bcr2, and bcr3 plasmid DNA amplification were incubated with (+) or without (−) DdeI at 37 °C for 120 min, followed by deactivation at 70 °C for 15 min and separation by 1.5% agarose gel electrophoresis (100 V, 35 min). The amounts of bcr1, bcr2 and bcr3 plasmid DNAs were 8.8, 7.8, and 8.0 × 106 copies/tube, respectively. The expected sizes of digested RT-LAMP products are the followings: 468 bp, about 234 bp (stem and loop structure) and 83 bp for bcr1; 232 bp, about 116 bp (stem and loop structure) and 83 bp for bcr2; and 139 bp and 121 bp (merged in Figure) for bcr3. DW, distilled water (negative control); M, 100-bp ladder size marker; neg RNA, IVS-0035 RNA (negative control); RT-LAMP, reverse transcription loop-mediated isothermal amplification.
Sensitivity of the RT-LAMP assay
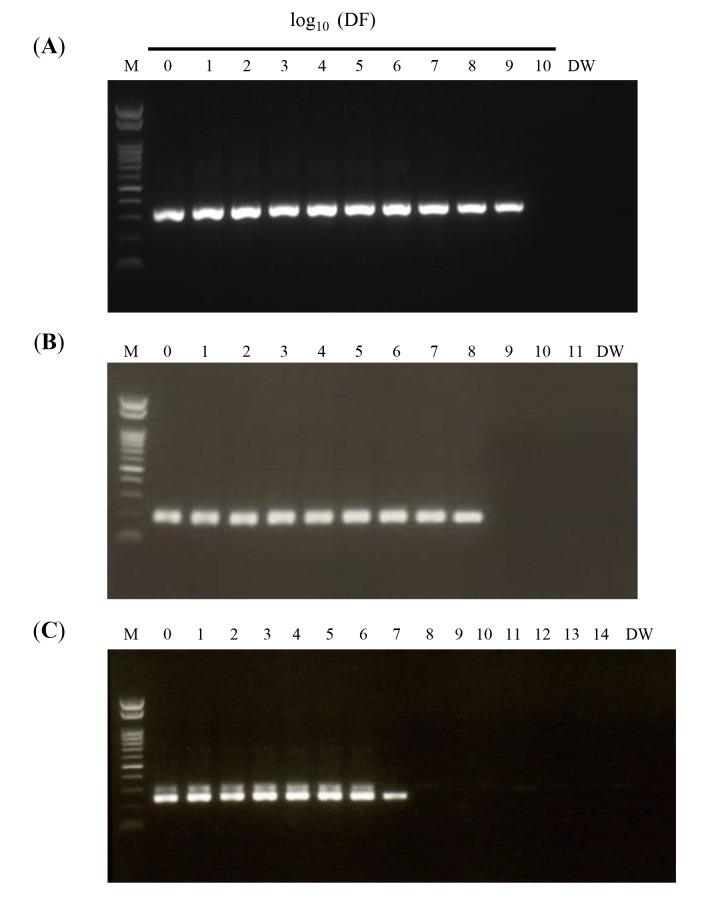
The lower detection limit of the RT-LAMP assay was determined using10-fold serial dilutions of bcr1, bcr2, and bcr3 plasmid DNA; the result was compared to that obtained by nested RT-PCR. RT-LAMP was able to detect 880, 78, and 800 copies/tube of bcr1, bcr2, and bcr3 plasmid, respectively (Fig. 2A), as compared to 0.88, 7.8, and 80 copies/tube, respectively, by nested RT-PCR (Figs. 4A–C). The specificity of all RT-LAMP products was confirmed by DdeI restriction enzyme digestion.
Fig. 4.
Nested RT-PCR amplification using 10-fold serial dilutions of plasmid DNA. Plasmid DNAs containing (A) bcr1, (B) bcr2, and (C) bcr3 sequences (starting amounts: 8.8 × 108, 7.8 × 108, and 8.0 × 108 copies/tube, respectively) were diluted by the indicated DF and amplified by nested RT-PCR. Amplification products were separated by 1.5% agarose gel electrophoresis (100 V, 35 min). DF, dilution factor; DW, distilled water; M, 100-bp ladder size marker; RT-PCR, reverse transcription polymerase chain reaction.
RT-LAMP of clinical samples
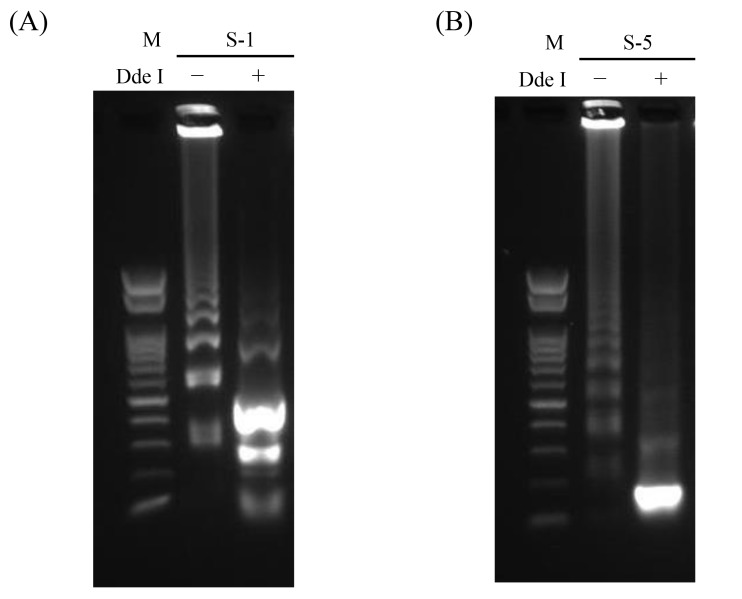
Clinical samples were analyzed by RT-LAMP and nested RT-PCR; these included four bcr1- and three bcr3-type PML-RARα RNAs. The plasmid DNAs were used as positive controls, and IVS-0035 RNA and distilled water were used as negative controls. Three bcr1- and three bcr3-type RNAs were detected by RT-LAMP (Fig. 2B); the products were confirmed by DdeI restriction enzyme digestion (Fig. 5), and the results were the same as those obtained by nested RT-PCR (Table 2). One sample yielded no product with both assays; this RNA had been stored for the longest period (about 9 years) and was likely degraded, as indicated by RIN analysis of RNA quality.15, 16
Fig. 5.
Confirmation of bcr1 and bcr3 of clinical samples after RT-LAMP assay. The RT-LAMP products of clinical sample RNAs of S-1 (A) and S-5 (B) were incubated with (+) or without (−) DdeI at 37 °C for 120 min, followed by deactivation at 70 °C for 15 min and separation by 1.5% agarose gel electrophoresis (100 V, 35 min). M, 100-bp ladder size marker; RT-LAMP, reverse transcription loop-mediated isothermal amplification.
Table 2.
Comparison between RT-LAMP and nested RT-PCR using clinical samples
| Sample number | Breakpoint type | RNA concentration (ng/μL) | Absorbance ratio (260/280 mm) | RIN value | RT-LAMP | Nested RT-PCR |
| S-1 | bcr1 | 250.8 | 2.04 | 9.60 | + | + |
| S-2 | bcr1 | 535.0 | 2.04 | 10 | + | + |
| S-3 | bcr1 | 361.9 | 1.96 | NM | – | – |
| S-4 | bcr1 | 1141.0 | 2.06 | NA | + | + |
| S-5 | bcr3 | 389.8 | 2.06 | 9.50 | + | + |
| S-6 | bcr3 | 309.2 | 2.05 | 9.90 | + | + |
| S-7 | bcr3 | 59.9 | 2.04 | 8.50 | + | + |
NA, not available; NM, not measured due to pattern deformation; RIN, RNA integrity number; RT-LAMP, reverse transcription loop-mediated isothermal amplification; RT-PCR, reverse transcription polymerase chain reaction; Samples 5 and 6 were derived from the same patient: S-5, at the initial visit and S-6, at the time of relapse.
DISCUSSION
Each type of PML-RARα sequence was specifically amplified by RT-LAMP; moreover, each PML-RARα breakpoints as well as non-specific amplification could be distinguished by digesting the products with DdeI. The sensitivity of the RT-LAMP assay was inferior to that of nested RT-PCR by 10 fold for bcr2 and bcr3 and by 1000 fold for bcr1. Some RT-LAMP assays have higher sensitivity than nested RT-PCR,17–20 while others have lower sensitivity.21, 22 This depends on the primers used, which determine the amplification efficiency and has a significant effect on the sensitivity of gene amplification. In this study, we selected 5 primer sets from more than 1000 candidates listed by PrimerExplorer ver. 4. It is virtually impossible to test all candidates, although a primer set with better amplification efficiency may exist.
RT-LAMP and nested RT-PCR yielded similar results in the analysis of clinical samples. This suggests that RT-LAMP has clinical utility as a diagnostic test, especially at initial presentation when a sufficient amount of PML-RARα mRNA is available.
The nested RT-PCR assay requires about 6 h from RNA extraction to target mRNA detection. In contrast, PML-RARα mRNA was amplified in 30 min by RT-LAMP, and is therefore more useful for early diagnosis and determination of an appropriate intervention. In addition, the amount of sample that is lost with RT-LAMP is minimal because the extracted RNA is used directly in the amplification reaction even in one tube. This simplifies the procedure, thereby reducing the labor and cost of laboratory testing.
Spinelli et al. recently reported another assay using RT-LAMP to detect PML-RARα chimeric gene.23 Using fluorescence, the assay appeared to be more rapid and sensitive than our assay. However, the detection of fluorescence requires a dedicated devise and their assay needs 2 reaction tubes to detect 3 PML-RARα mRNAs. In addition, primer sequences were not described. Therefore, our assay has advantages of simple procedure and low cost, and could readily be applied in developing countries. Such simplicity can lead to the development of an automated system that can be used for point-of-care testing and diagnosis of APL.
In conclusion, we developed an RT-LAMP assay for detection of PML-RARα mRNA in clinical samples from APL patients. This assay is more simple and rapid than nested RT-PCR, and can contribute to early diagnosis of APL.
Acknowledgments
Ackonwlegments: The authors would like to thank Mr. Yasuharu Sasaki for technical advice and Editage (www.editage.jp) for English editing.
This study was supported by a grant from Seeds Research Project at Tottori University Hospital.
The authors declare no conflict of interest.
REFERENCES
- 1. Notomi T, Okayama H, Masubuchi H, Yonekawa T, Watanabe K, Amino N, et al. Loop-mediated isothermal amplification of DNA. Nucleic Acids Res. 2000;28:E63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Nagamine K, Hase T, Notomi T. Accelerated reaction by loop-mediated isothermal amplification using loop primers. Mol Cell Probes. 2002;16:223-9. [DOI] [PubMed] [Google Scholar]
- 3. Mori Y, Nagamine K, Tomita N, Notomi T. Detection of loop-mediated isothermal amplification reaction by turbidity derived from magnesium pyrophosphate formation. Biochem Biophys Res Commun. 2001;289:150-4. [DOI] [PubMed] [Google Scholar]
- 4. Dhama K, Karthik K, Chakraborty S, Tiwari R, Kapoor S, Kumar A, et al. Loop-mediated isothermal amplification of DNA (LAMP): a new diagnostic tool lights the world of diagnosis of animal and human pathogens: a review. Pak JBiol Sci. 2014;17:151-66. [DOI] [PubMed] [Google Scholar]
- 5. Mori Y, Notomi T. Loop-mediated isothermal amplification (LAMP): a rapid, accurate, and cost-effective diagnostic method for infectious diseases. J Infect Chemother. 2009;15:62-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Morishita S, Tani H, Kurata S, Nakamura K, Tsuneda S, Sekiguchi Y, et al. Real-time reverse transcription loop-mediated isothermal amplification for rapid and simple quantification of WT1 mRNA. Clin Biochem. 2009;42:515-20. [DOI] [PubMed] [Google Scholar]
- 7. Kakizuka A, Miller WH, Umesono K, Warrell RP, Frankel SR, Murty VV, et al. Chromosomal translocation t(15;17) in human acute promyelocytic leukemia fuses RAR alpha with a novel putative transcription factor, PML. Cell. 1991;66:663-74. [DOI] [PubMed] [Google Scholar]
- 8. Miller WH, Kakizuka A, Frankel SR, Warrell RP, DeBlasio A, Levine K, et al. Reverse transcription polymerase chain reaction for the rearranged retinoic acid receptor alpha clarifies diagnosis and detects minimal residual disease in acute promyelocytic leukemia. Proc Natl Acad Sci USA. 1992;89:2694-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. van Dongen JJ, Macintyre EA, Gabert JA, Delabesse E, Rossi V, Saglio G, et al. Standardized RT-PCR analysis of fusion gene transcripts from chromosome aberrations in acute leukemia for detection of minimal residual disease. Report of the BIOMED-1 Concerted Action: investigation of minimal residual disease in acute leukemia. Leukemia. 1999;13:1901-28. [DOI] [PubMed] [Google Scholar]
- 10. Tobal K, Liu Yin JA. RT-PCR method with increased sensitivity shows persistence of PML-RARA fusion transcripts in patients in long-term remission of APL. Leukemia. 1998;12:1349-54. [DOI] [PubMed] [Google Scholar]
- 11. Gallagher RE, Willman CL, Slack JL, Andersen JW, Li YP, Viswanatha D, et al. Association of PML-RAR alpha fusion mRNA type with pretreatment hematologic characteristics but not treatment outcome in acute promyelocytic leukemia: an intergroup molecular study. Blood. 1997;90:1656-63. [PubMed] [Google Scholar]
- 12. Lo-Coco F, Avvisati G, Vignetti M, Thiede C, Orlando SM, Iacobelli S, et al. Retinoic acid and arsenic trioxide for acute promyelocytic leukemia. New Engl JMed. 2013;369:111-21. [DOI] [PubMed] [Google Scholar]
- 13. Wang ZY, Chen Z. Acute promyelocytic leukemia: from highly fatal to highly curable. Blood. 2008;111:2505-15. [DOI] [PubMed] [Google Scholar]
- 14. Breccia M, Coco FL. Thrombo-hemorrhagic deaths in acute promyelocytic leukemia. Thromb Res. 2014;133:S112-S6. [DOI] [PubMed] [Google Scholar]
- 15. Schroeder A, Mueller O, Stocker S, Salowsky R, Leiber M, Gassmann M, et al. The RIN: an RNA integrity number for assigning integrity values to RNA measurements. BMC Mol Biol. 2006;7:3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Imbeaud S, Graudens E, Boulanger V, Barlet X, Zaborski P, Eveno E, et al. Towards standardization of RNA quality assessment using user-independent classifiers of microcapillary electrophoresis traces. NucleicAcids Res. 2005;33:e56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Lee S, Kim JH, Choi JY, Jang WC. Loop-mediated isothermal amplification assay to rapidly detect wheat streak mosaic virus in quarantined plants. Plant Pathol J. 2015;31:438-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Zhang Q, Liu S, Yang H, Zhu L, Wan X, Li X, et al. Reverse transcription loop-mediated isothermal amplification for rapid and quantitative assay of covert mortality nodavirus in shrimp. J Invertebr Pathol. 2015;September 9. doi: 10.1016/j.jip.2015.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Liu DF, Liu CG, Tian J, Jiang YT, Zhang XZ, Chai HL, et al. Establishment of reverse transcription loop-mediated isothermal amplification for rapid detection and differentiation of canine distemper virus infected and vaccinated animals. Infect Genet Evol. 2015;32:102-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Arunrut N, Suebsing R, Withyachumnarnkul B, Kiatpathomchai W. Demonstration of a very inexpensive, turbidimetric, real-time, RT-LAMP detection platform using shrimp Laem-Singh virus (LSNV) as a model. PLoS ONE. 2014;9:e108047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Hanaki K, Ike F, Hatakeyama R, Hirano N. Reverse transcription-loop-mediated isothermal amplification for the detection of rodent coronaviruses. J Virol Methods. 2013;187:222-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Kiatpathomchai W, Jareonram W, Jitrapakdee S, Flegel TW. Rapid and sensitive detection of Taura syndrome virus by reverse transcription loop-mediated isothermal amplification. J Virol Methods. 2007;146:125-8. [DOI] [PubMed] [Google Scholar]
- 23. Spinelli O, Rambaldi A, Rigo F, Zanghì P, D’Agostini E, Amicarelli G, et al. Simple, rapid and accurate molecular diagnosis of acute promyelocytic leukemia by loop mediated amplification technology. Oncoscience. 2015;2:50-8. [DOI] [PMC free article] [PubMed] [Google Scholar]