Abstract
Atherosclerosis is the leading cause of morbidity and mortality in the Western world. Peripheral artery disease (PAD) has been less studied then coronary artery disease but is nearly as common. PAD impairs blood flow to the leg(s) and causes functional impairment, leg pain, and amputation. The last drug approved for PAD was in 1999. Blood flow to leg proceeds through one major artery and in PAD total occlusions in the course of that vessel are common. Thus, the extent of new blood vessel growth determines a patients’ clinical course. Promoting the growth of new blood vessels (therapeutic angiogenesis) was a major goal of therapy. Results from studies using cytokine growth factors have shown disappointing results. Using clinical and preclinical studies, our laboratory has identified several novel therapeutic approaches. One, a modulator of innate immunity, will be reviewed as an approach that has the potential to create new therapies for PAD.
INTRODUCTION
Clinical Burden/Epidemiology of Peripheral Artery Disease
Atherosclerosis remains the leading cause of morbidity and mortality in the Western world and is becoming an ever greater health care problem in the developing worlds as well. In the United States, for far too many years, peripheral artery disease (PAD) was viewed as the “poor” stepchild to the more extensively studied coronary artery disease (CAD). A reduction in the ankle brachial blood pressure index (ABI: higher of the two ankle blood pressure values to the highest arm pressure value) is the hallmark for PAD, and when this is used as the diagnostic criteria, the prevalence of PAD in the United States was roughly three-quarters that of CAD a decade ago (1).Evidence continues to mount that CAD rates are declining and that PAD likely will become as common as CAD within the next decade (2−4).
One of the major reasons for the increasing burden of PAD is the fact that the epidemiologic risk factors for PAD are actually slightly different than CAD. For CAD there are multiple independent risk factors, with hypercholesterolemia and hypertension being two of the largest, whereas for PAD the major drivers for PAD are advancing age, smoking, and diabetes (3−5).
There are other reasons that PAD differs from CAD. Total occlusion in the inflow arteries that follow almost a complete singular path from the aorta to one or both leg(s) is extremely common in patients with PAD, and thus the magnitude of distal blood flow becomes dependent on the number and extent of collateral blood vessels that connect to the distal microvasculature (6). Almost uniformly, current medical therapies used to treat PAD were derived from studies designed to treat CAD and prevent acute thrombotic occlusion and include antiplatelet agents, statins to lower cholesterol, antihypertensive therapy with angiotensin converting enzyme inhibitors or receptor blocker and/or beta-blockers, blood glucose control, and smoking cessation (7,8). However, there are no medical therapies available that have shown the ability to improve leg blood flow in patients with PAD and thus no medical therapies are available to directly treat the primary problem of reduced blood flow. New treatment paradigms are needed for PAD (2,3,6).
Spectrum of Clinical Manifestations of PAD
A full review of the clinical manifestations of PAD is beyond the scope of this article but details can be found and additional reference can be located in the most current, and frequently updates Trans-Atlantic Inter-Society Consensus on the Management of PAD (TASC) documents (8).The ABI measures the ratio of systolic blood pressure in the ankle to that of the brachial vessels and an ABI <0.9 mm Hg is considered diagnostic for PAD. Additional testing with duplex ultrasonography, segmental Doppler pressure or volume plethysmography, and magnetic resonance or contrast angiography may be utilized if clinical suspicion remains in patients who have risk factors but a normal ABI or for whom the ABI is >1.30. Evidence remains that even when the ABI is abnormal 50% or more of patients with PAD lack the classic symptoms of PAD and thus are often considered asymptomatic. There are two major clinical manifestations of PAD: intermittent claudication (IC) and critical limb ischemia (CLI). The diagnosis of IC versus CLI is based upon time-tested clinical classification schemes, namely the Rutherford and the Fontaine classifications. In patients with IC and likely those with PAD but no IC symptoms, there is an inability to increase blood flow to the legs to meet the demands of exercise. In the Rutherford classification, IC encompasses categories 1−3 (mild, moderate, and severe claudication, respectively), whereas CLI includes categories 4−6 (ischemic rest pain, minor tissue loss, and ulceration or gangrene, respectively). The Fontaine classification is more commonly used in Europe, with stages IIa and IIb describing IC, whereas stages III−IV are categories of CLI. There is no biomarker or hemodynamic measure that is pathognomonic for either IC or CLI. Patients with IC have amputation and an annual mortality rate of 1% to 2%; those with CLI have a 6-month amputation risk of 25% to 40% and an annual mortality rate of 20%.
Angiogenesis and (Predominant) Failure of Therapeutic Angiogenesis in Humans
For patients with IC, exercise therapy when added to “best” medical therapy remains the treatment of choice. However, supervised exercise is limited in availability and still is not frequently reimbursed despite the intensive and time-demanding aspects of that therapy (5,9). Unfortunately, a large fraction of patients with CLI are poor candidates for surgical or catheter based revascularization and some are not a candidate for either; thus, PAD remains a leading cause of amputation (3,6). Angiogenesis is by definition the growth and proliferation of blood vessels from pre-existing vascular structure (10). Because of the lack of viable medical options for PAD, therapeutic angiogenesis emerged as an experimental approach to attempt to improve tissue perfusion in PAD (11). Most studies used cytokine growth factors including vascular endothelial growth factor, fibroblast-derived growth factor, hepatocyte growth factor, and angiopoietins delivered in protein or gene-based therapy. Recent reports from our group and others have reviewed the outcome of therapeutic angiogenesis when used for IC and CLI (6,12). The largest of the studies for CLI was representative of the results of these types of trials. A phase III study, which was designed to evaluate the safety and efficacy of NV1FGF in 525 CLI patients with skin lesions (due to ulcer or gangrene) who were unsuitable for revascularization (ClinicalTrials.gov Identifier: NCT00566657), was disappointing. At 1 year, no significant difference was observed between the NV1GF group versus the placebo arm in the primary end-point of time to major amputation or death (13).
MATERIALS AND METHODS FOR AN ALTERNATIVE APPROACHE TO DEVELOP NOVEL THERAPEUTICS FOR PAD
Juxtaposition of IC in CLI and Use of Preclinical Models of PAD
The agents used in human clinical trials that failed or were at best marginally successful were based on successful preclinical trials. A comprehensive discussion on the potential reasons for the failures of the human trials can be found in our recent review and will not be repeated here. Our laboratory has taken three complementary approaches (Figure 1) to advancing therapeutic angiogenesis in humans. These approaches include the new ways to assess outcomes in human trials, new ways to deliver gene products, seeking approaches to identify novel agents, and, in particular, selecting factors in a more agnostic manner.
Fig. 1.
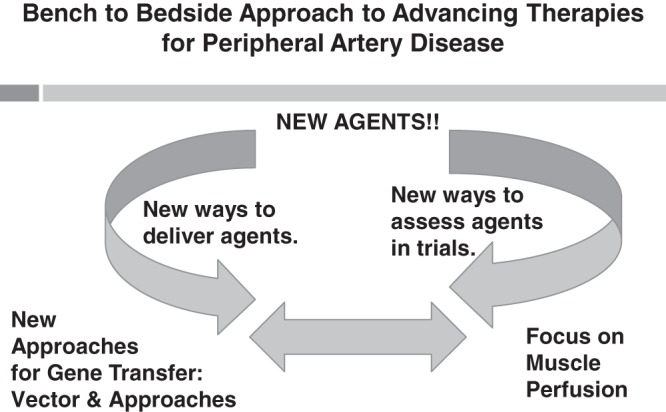
This figure demonstrates how the identification of new agents, new approaches for “gene” delivery, and constant reassessment of outcomes in human and preclinical trials are used to identify new approaches to treat peripheral artery disease.
For the selection of new factors, we built on the fact that the angiogenic response that follows surgically induced hindlimb ischemia (HLI) in mice has features that follow the human disease; that is, factors that are bad for mice after HLI are also bad for humans with PAD (14,15). In addition, whereas PAD is caused by atherosclerosis in humans, it is well established that there is a great deal of patient-to-patient variability, even when adjusting for demographic risk factors and the extent of angiographic disease (16,17). Our lab and others have shown that C57BL/6 mice recover from HLI much better than BALB/c mice (14,18). We described a 31M base pair quantitative trait locus on the short arm of mouse chromosome 7 (termed LSq-1) that was sufficient to determine the extent of perfusion recovery and tissue loss after HLI using the outcome from C57 BL/o, and one additional BALBlc, inbred mouse strain (11).
Interleukin 21 Receptor as a Novel Target for PAD
The interleukin 21receptor (IL-21R) was cloned in by Dr. Warren J. Leonard’s group at National Institutes of Health based on the knowledge that IL-21 plays a key role in innate and adaptive immunity with signaling via the heterodimer of IL-21R and the common cytokine receptor γ chain (19,20). Therefore, in many ways, this finding met the criteria of a factor that would, a priori, be selected as an agent with an angiogenic property. To the best of our knowledge there were no prior reports of IL-21R in the human condition of PAD. Recombinant IL-21 has been studied in at least 12 human clinical trials, and IL-21−specific antibodies are entering human studies to limit transplant rejection (21).
RESULTS
The general utility of clinical/preclinical, claudication/CLI strategy was reported by our laboratory in 2008 (14). Although exciting in concept, actual targets resulting from this approach are now coming to fruition as our group now has four separate reports using this strategy (Figure 2). Complete details from the mouse and the human studies on the IL-21 and the IL-21R, as an example from this strategy, can be found in the original publications (22,23), but the major results are summarized in the paragraphs below.
Fig. 2.
Lists the recent papers from our group where new and unexpected targets developed from the integrated translational research program.
We previously described that it is essential to study the time points when the degree of perfusion recovery is comparable at an early (day 3) time point after HLI to identify factors and pathways involved in regulating perfusion recovery and angiogenesis after HLI (15). Using this approach we found that the Il21r mRNA level was strikingly up-regulated in ischemic hindlimb muscles from C57BL/6 mice (strain with good/excellent perfusion recovery post-HLI) than from BALB/c (strain with poor perfusion recovery post-HLI) mice. We next sought to determine the major cell type(s) that contributed to the IL-21R elevation after HLI. CD31+ cells were isolated from C57BL/6 hindlimb (24). Whereas the overall increase in C57BL/6 versus BALB/c in whole muscle was quite large, within the CD31+ fraction, it was even greater and immunofluorescence staining of the ischemic muscle from C57BL/6 mice at day 3 post-HLI showed numerous examples of co-staining of IL-21R with CD31. The final piece of evidence was that flow cytometry showed that CD31+ fractions from ischemic hindlimb muscle had higher IL-21R protein level than the CD31+ fraction from non-ischemic hindlimb muscle in C57BL/6 mice. In contrast, the same cell fraction from BALB/c mice did not show a difference between ischemic and non-ischemic muscle. All of the original data can be found in Figure 1 from Wang et al (22). In addition, we found that the differences between the C57BL/6 and BALB/c strain was at the level of the IL-21R as IL-21 levels did not differ based on either ischemia or strain and were able to recapitulate the up-regulation of IL21R in in vitro endothelial cells (human umbilical vein endothelial cells) and showed the pro-survival effects of this strategy.
We performed two complementary in vivo loss-of-function studies. First, we compared the time course and extent of perfusion recovery after HLI in Il21r-/- mice compared to age-matched and gender-matched wild-type littermates and found that Il21r-/- mice showed attenuated perfusion recovery, impaired angiogenesis relatively reduced cell survival proteins, and numerically more tissue loss. To eliminate features and factors that may be altered by the global genetic knock-out, we also used a neutralizing IL-21R-Fc fusion protein versus control, (25) via intra-peritoneal injection into wild-type C57BL/6 mice. The results were similar to the results seen with Il21r-/- mice; neutralization of IL-21 resulted in impaired perfusion recovery compared to vehicle-treated mice. All of this data is located in Figure 4 of Wang et al (22).
Of critical importance, our studies were undertaken in the background of a published report showing that IL-21 was angiostatic not angiogenic. In a tumor model, Castermans et al (26) showed that Il-21 versus control protein caused less blood vessel growth and signaling through this IL-21R−activated pathway and was mediated by phosphorylation and activation of signal transducer and activator of transcription (STAT) 1 and its downstream signaling. Although a full description of all the experimental data will not be repeated here, our data convincingly showed that in hypoxic endothelium, treatment with IL-21 versus control protein led to activation of STAT 3 (27); not STAT 1. Thus our data demonstrates the context dependence of the IL21/IL21R signaling in endothelial cells.
Animal models, of course, only recapitulate selected aspects of the human disease condition. With an eye toward the most direct translational approaches, two critical findings had to be present. First, endothelial cells from the distal (i.e., calf) muscle from patients with PAD would need to express IL-21R at higher levels than endothelium from control subjects. Second, whereas circulating levels of IL-21 could be elevated in PAD versus control subjects, the degrees of elevation should be modest such that a systemic protein delivery strategy (if safe in humans) could achieve levels that are far above endogenous levels. A full description of the data can be found in Figure 1 from Wang et al (23). Indeed, by immunofluorescence there was a greater ratio of IL-21R+cells per total nuclei, as well as a much higher percentage of IL-21R+ staining endothelial cells in muscle specimens from PAD patients compared with muscle from the healthy controls limbs. This finding was an important validation of the ability of the model to predict one critical aspect PAD. To the best of our knowledge, no data on IL-21 plasma levels in PAD patients compared to healthy controls exists. Although we found that PAD patients had a significantly higher plasma concentration of IL-21 compared to healthy controls, the mean difference between the groups was less than 2-fold; therefore, therapeutic administration should easily exceed levels seen in patients. The utility of this approach remains questionable if the receptor is not up-regulated and thus not available in hypoxic endothelium to transduce survival signals.
DISCUSSION AND FUTURE DIRECTION
PAD affects millions of patients in the United States alone. PAD, when considered in any single form or in totality, presents a large unmet clinical need. Indeed, PAD was noted by the Institute of Medicine as a high-priority research area that needs to focus on reducing mortality and morbidity (28). PAD is a result of systemic atherosclerosis, but for advances in PAD research and therapy to occur, PAD must be recognized as a distinct subtype of atherosclerotic vascular disease. Although overlap exists, PAD differs from coronary artery and cerebrovascular disease with respect to the associated strength of risk factors for disease generation and in disease pathogenesis. Thrombotic occlusion and embolic event which dominate coronary artery and cerebrovascular disease, respectively, are less relevant in PAD. In the majority of patients with PAD, total occlusions of the arteries dominate and the relative ability of the individual to generate a neovascularization response will be inversely correlated with the patients’ clinical course.
Despite its limitations, the response that follows surgically induced HLI in mice has been widely used to test agents to treat PAD; however, a downside to this approach is that agents with predicted pathways are typically selected for development (29). We hypothesized that a mouse quantitative trait locus that modulated the neovascularization response would allow for the identification of novel therapeutic targets (14). Our data in totality demonstrates that the IL-21R, which lies at or near the geographic peak of the strength of association with preclinical outcomes, is adaptively up-regulated after HLI. Under ischemic conditions, either IL-21 ligand or receptor inhibition contributes to impaired perfusion recovery after experimental HLI (widely used as preclinical PAD model) through the STAT 3 pathway. Our study is the first one to elucidate the role of IL-21/IL-21R in PAD as a novel therapeutic target for PAD therapy (22,23).
Currently, listings at http://www.clinicaltrials.gov show that recombinant IL-21 due to its potent effects in modulating immune system is currently being tested in at least 12 different clinical trials, mostly being used in various malignancies (20). In turn, blocking IL-21R has also been tested in several phase I clinical studies for different disease indications (20,21). We not only found the receptor to be up-regulated in PAD patients versus control calf muscle biopsy specimens, but we also found the increased IL-21R expression to be predominantly in the endothelium from the most symptomatic leg, which parallels what we found in the unilateral mouse HLI model. It is certainly plausible that increased endothelial cell IL-21R expression in ischemic muscle and elevated blood IL-21 ligand levels in PAD patients may be at least partially adaptive. The presence of IL-21R in ischemic muscle may establish systemic IL-21 as a potential strategy to treat PAD.
ACKNOWLEDGMENTS
Dr Annex would like to thank all of my clinical and basic research colleagues that I have had the privilege to work with over the years, as well as his current laboratory members at the University of Virginia for all their hard work and ongoing commitment to the advancement of translational research. This work was supported in part by 1R01 HL116455, 1R01 HL121635, and 2R01 HL101200.
Footnotes
Potential Conflicts of Interest: None disclosed.
DISCUSSION
Feldman, Philadelphia: Brian that was great talk. I saw a patient about a month ago who had peripheral vascular disease and in one leg just incredible wasting. There was a 3-cm difference between the normal leg and the wasted one. When we sent him off for flow studies, the flow studies were actually identical in the two legs, so it made me question whether is it all about flow or is there something intrinsic in the cells that either causes wasting or apoptosis, and if we are not going to get the information from flow, how do we get his information?
Annex, Charlottesville: That’s a great question. I sometimes will actually talk about our human studies where we have actually looked at a combination of calf muscle biopsies and the injury pattern we see in the tissues, as well as what we think are the best methods now for assessing, not flow, but perfusion. There was a fascinating trial completed called the CLEVER (Claudication: Exercise Versus Endoluminal Revascularization) trial which randomized patients to stenting versus exercise for isolated aorto-iliac disease. The bottom line is restoring flow, which you did by stenting, was not as effective as exercise, which affects, we think, the microvascular and muscle perfusion. We’re actually testing that now in an NIH grant to determine if MR-based calf muscle perfusion can be the best measure right now.
Taylor, Atlanta: Brian thanks for a really nice talk. So what about arteriogenesis versus angiogenesis? As you pointed out in your earlier slides with VEGF, you get a lot of dysfunctional leaky vessels, what do you think about the roles there? I am interested in your thoughts about that.
Annex, Charlottesville: Very good question. We have actually begun to look at models where we can parse out the effects of angiogenesis versus atherogenesis. I am just going to use one set of studies as an example. Dan Gary made the original myoglobin knockout mouse but he also made an over-expresser and we got access to those mice. That is a model where there is a selective up-regulation of myoglobin and we showed in a paper several years ago that as you absorb nitric oxide, you have an impaired angiogenic response but no difference in the atherogenic response. In those mice at least, the angiogenesis seems to be the dominant factor with the possibility that differences in atherogenesis will follow. That is obviously theory and one that we are testing in more involved models. That was an excellent question.
Schreiner, Los Altos: Would you comment on the potential systemic effects of interleukin 21, which is a known activator of T and B cells and in fact promotes cytotoxic T cell activation? How do you envision this as a therapy in the context of peripheral artery disease when you also have a very active immune modulator?
Annex, Charlottesville: That is a great question. I am going to state a qualifier, I am a cardiologist and I will seek to get advice on that subject as we go along. Clearly, we are going to have to figure that out. I think our early work to this point is showing that likely the effects of interleukin 21 when administered are dependent on the receptor and its activation. If so, what is critical is the context with which that the receptor is activated. I think I can envision a way [to give interleukin 21] because we know the receptor is up regulated and the pathway can be activated in PAD and muscle ischemia so that we might be able to do that. Again this would be done with the proper test with safety in mind. The flipside of that though is also important: Interleukin 21 receptor blockers are actually going to be given to patients and are entering human trials for mixed connective tissue diseases, I suspect that some of those patients are going to have peripheral arterial disease just by the epidemiologic data and obviously understanding this beforehand will give us way to exclude that as a toxicity.
REFERENCES
- 1.American Heart Association Heart Disease and Stroke Statistics. https://my.americanheart.org/idc/groups/ahamah-public/@wcm/@sop/@smd/documents/downloadable/ucm_459072.pdf.2004 . [Google Scholar]
- 2.Wennberg PW. Approach to the patient with peripheral arterial disease. Circulation. 2013;128:2241–50. doi: 10.1161/CIRCULATIONAHA.113.000502. [DOI] [PubMed] [Google Scholar]
- 3.Peacock JM, Keo HH, Duval S, et al. The incidence and health economic burden of critical limb ischemia and ischemic amputation in Minnesota 2005–2008. Prev Chronic Dis. 2011;8:A141. [PMC free article] [PubMed] [Google Scholar]
- 4.Rosamond WD, Chambless LE, Heiss G, et al. Twenty-two–year trends in the incidence of myocardial infarction, coronary artery disease mortality, and case fatality in 4 US communities 1987–2008. Circulation. 2012;125:1848–57. doi: 10.1161/CIRCULATIONAHA.111.047480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.White C. Intermittent claudication. N Engl J Med. 2007;356:1241–50. doi: 10.1056/NEJMcp064483. [DOI] [PubMed] [Google Scholar]
- 6.Annex BH. Therapeutic angiogenesis for critical limb ischemia. Nat Rev Cardiol. 2013;10:387–96. doi: 10.1038/nrcardio.2013.70. [DOI] [PubMed] [Google Scholar]
- 7.Hirsch AT. Treatment of peripheral arterial disease — extending “intervention” to “therapeutic choice.”. N Engl J Med. 2006;354(18):1944–7. doi: 10.1056/NEJMe068037. [DOI] [PubMed] [Google Scholar]
- 8.Norgren L, Hiatt WR, Dormandy JA, et al. Inter-society consensus for the management of peripheral arterial disease (TASC II) J Vasc Surg. 2007;45:S5–67. doi: 10.1016/j.jvs.2006.12.037. [DOI] [PubMed] [Google Scholar]
- 9.Murphy TP, Cutlip DE, Regensteiner JG, et al. Supervised exercise, stent revascularization, or medical therapy for claudication due to aortoiliac peripheral artery disease: the CLEVER study. J Am Coll Cardiol. 2015;65:2055. doi: 10.1016/j.jacc.2014.12.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Folkman J. Seminars in medicine of the Beth Israel Hospital, Boston. Clinical applications of research on angiogenesis. N Engl J Med. 1995;333:1757–63. doi: 10.1056/NEJM199512283332608. [DOI] [PubMed] [Google Scholar]
- 11.Folkman J. Therapeutic angiogenesis in ischemic limbs. Circulation. 1998;97:1108–10. doi: 10.1161/01.cir.97.12.1108. [DOI] [PubMed] [Google Scholar]
- 12.Jones WS, Annex BH. Growth factors for therapeutic angiogenesis in peripheral arterial disease. Curr Opin Cardiol. 2007;22:458–63. doi: 10.1097/HCO.0b013e328236741b. [DOI] [PubMed] [Google Scholar]
- 13.Lambert MA, Belch JJ. Medical management of critical limb ischemia: where do we stand today? J Intern Med. 2013;274:295–307. doi: 10.1111/joim.12102. [DOI] [PubMed] [Google Scholar]
- 14.Dokun AO, Keum S, Hazarika S, et al. A quantitative trait locus (LSq-1) on mouse chromosome 7 is linked to the absence of tissue loss after surgical hindlimb ischemia. Circulation. 2008;117:1207–15. doi: 10.1161/CIRCULATIONAHA.107.736447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hazarika S, Dokun AO, Li Y, et al. Impaired angiogenesis following hind-limb ischemia in type 2 diabetes mellitus: differential regulation of VEGFR-1 and soluble VEGFR-1. Circ Res. 2007;101:948–56. doi: 10.1161/CIRCRESAHA.107.160630. [DOI] [PubMed] [Google Scholar]
- 16.Murabito JM, Guo CY, Fox CS, et al. Genetic contributions to peripheral arterial disease: heritability of the ankle-brachial blood pressure index in the Framingham Heart Study. Am J Epidemiol. 2006;164:963–8. doi: 10.1093/aje/kwj295. [DOI] [PubMed] [Google Scholar]
- 17.Leeper NJ, Kullo IJ, Cooke JP. Genetics of peripheral artery disease. Circulation. 2012;125:3220–8. doi: 10.1161/CIRCULATIONAHA.111.033878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wang S, Zhang H, Wiltshire T, et al. Genetic dissection of the Canq1 locus governing variation in the extent of the collateral circulation. PLoS One. 2012;7:e31919. doi: 10.1371/journal.pone.0031910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ozaki K, Spolski R, Feng CG, et al. A critical role for IL-21 in regulating immunoglobulin production. Science. 2002;298:1630–4. doi: 10.1126/science.1077002. [DOI] [PubMed] [Google Scholar]
- 20.Spolski R, Leonard WJ. Interleukin-21: a double-edged sword with therapeutic potential. Nat Rev Drug Discov. 2014;13:381–93. doi: 10.1038/nrd4296. [DOI] [PubMed] [Google Scholar]
- 21.Thompson JA, Curti BD, Redman BG, et al. Phase I study of recombinant interleukin-21 in patients with metastatic melanoma and renal cell carcinoma. J Clin Oncol. 2008;26:2034–9. doi: 10.1200/JCO.2007.14.5193. [DOI] [PubMed] [Google Scholar]
- 22.Wang T, Cunningham A, Dokun AO, et al. Loss of interleukin-21 receptor activation in hypoxic endothelial cells impairs perfusion recovery after hindlimb ischemia. Atheroscler Thromb Vasc Biol. 2015;35:1218–25. doi: 10.1161/ATVBAHA.115.305476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wang T, Cunningham A, Houston K, et al. Endothelial interleukin-21 receptor up-regulation in peripheral artery disease. Vasc Med. 2015 doi: 10.1177/1358863X15621798. epub ahead of press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Imoukhuede PI, Dokun AO, Annex BH, Popel AS. Endothelial cell-by-cell profiling reveals temporal dynamics of VEGFR1 and VEGFR2 membrane-localization following murine hindlimb ischemia. Am J Phys Heart Circ Phys. 2013;304:H1085–193. doi: 10.1152/ajpheart.00514.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Spolski R, Wang L, Wan CK, et al. IL-21 promotes the pathologic response to pneumovirus infection. J Immunol. 2012;188:1924–32. doi: 10.4049/jimmunol.1100767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Castermans K, Tabruyn SP, Zeng R, et al. Angiostatic activity of the antitumor cytokine interleukin-21. Blood. 2008;112:4940–7. doi: 10.1182/blood-2007-09-113878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chen Z, Han ZC. STAT3: A critical transcription activator in angiogenesis. Med Res Rev. 2008;28:185–200. doi: 10.1002/med.20101. [DOI] [PubMed] [Google Scholar]
- 28.Iglehart JK. Prioritizing comparative-effectiveness research — IOM recommendations. N Engl J Med. 2009;361:325–328. doi: 10.1056/NEJMp0904133. [DOI] [PubMed] [Google Scholar]
- 29.Waters RE, Terjung RL, Peters KG, et al. Preclinical models of human peripheral arterial occlusive disease: implications for investigation of therapeutic agents. J Appl Physiol. 2004;97:773–80. doi: 10.1152/japplphysiol.00107.2004. [DOI] [PubMed] [Google Scholar]



