Abstract
White Matter Disease is increasingly being recognized as an important cause of cognitive decline and dementia. Various investigations have linked chronic diet-related conditions to the development of white matter lesions, which appear as white matter hyperintensities on T2-weighted magnetic resonance imaging (MRI) scans of the brain. Thus, it can be postulated that the metabolic, inflammatory, and microvascular changes accompanying a western diet, hyperlipidemia, hypertension, and diabetes mellitus type II (DMII) are potential mediators in the development and progression of white matter disease, which in turn contributes to the development and progression of cognitive decline. This review will examine evidence for potential metabolic, inflammatory, and microvascular determinants of white matter disease and cognitive decline. Specifically, we will focus on the effects of altered insulin signaling in diabetes, obesity-induced oxidative stress, neuroinflammation, arterial stiffness due to hypertension, ischemia secondary to cerebral small vessel disease, and blood brain barrier disturbances.
Keywords: Alzheimer’s disease, cognitive impairment, dementia, inflammation, neurodegenerative disorders, western diet, white matter disease, vascular biology
Introduction
Alzheimer’s disease is the most common cause of dementia and has be cited as the sixth-leading cause of death in the United States [1]. According to a recent study using the latest data from the 2010 U.S. Census and the Chicago Heath and Aging Project, it is estimated that 5.1 million Americans age 65 and older have Alzheimer’s disease [2]. Given that age is a risk factor for Alzheimer’s disease and other dementias, the number of new and existing cases is predicted to increase as the segment of U.S. elderly (age 65 or over) population increase substantially, especially in the coming years as the baby boom generation ages. However, it is unclear whether the increase in cases of Alzheimer’s disease and other dementias are solely due to increasing age or whether chronic diet-related conditions, such as obesity and diabetes, are also responsible.
The western diet, characterized by high-fat, high-sugar, and excess salt composition, has been linked to major chronic diseases, such as atherosclerosis, hypertension, hyperlipidemia, obesity, and diabetes mellitus type II (DMII). Consumption of the western diet has also been associated with an increased risk for depression and cognitive decline [3-5]. This is consistent with previous studies that have demonstrated that cognitive impairment and decline is more prevalent among patients with metabolic syndrome and that metabolic syndrome is associated with greater risk of dementia later in life [6]. Lower intake of nutrient-dense foods and higher intake of processed “fast foods” have also been found to be independently associated with smaller left hippocampal volume [5], further supporting the concept that consumption of the western diet contributes to the development and progression of cognitive dysfunction.
In the past, most research performed on neurodegenerative diseases primarily focused on deleterious changes in the gray matter of the brain. More recently, diffuse white matter disease has come to be recognized as an important cause of cognitive decline and dementia. On T2-weighted MRI scans, white matter disease is represented as white matter hyperintensities, which are increased signal intensities that are thought to reflect demyelination and axonal loss [7]. These white matter hyperintensities have been shown to be predictive of an increased risk for dementia [8,9]. This is in accordance with numerous studies that have linked white matter hyperintensities to neurodegenerative diseases, such as Alzheimer’s disease and Parkinson’s disease [10,11]. For instance, one study demonstrated that increased white matter hyperintensity volume is predictive of the rate of cognitive decline amongst patients with Alzheimer disease, with larger volume of white matter hyperintensities indicating faster rate of cognitive decline [12,13]. Another study showed that white matter hyperintensities are an important risk factor for Parkinson’s disease with mild cognitive impairment. White matter hyperintensities were shown to be significantly greater in patients with both Parkinson’s disease and cognitive impairment versus patients that had Parkinson’s disease without cognitive impairment [11]. This association between white matter hyperintensities and cognitive decline brings up the question on whether the white matter lesions themselves directly impact cognition or whether their presence is an indication of underlying metabolic, inflammatory, or microvascular pathologies.
Previous investigations have shown strong correlations between chronic diet-related conditions and brain imaging changes [14]. For example, it has been shown that patients with DMII have increased white matter hyperintensity burden [15]. Obesity has been associated with reduced white matter integrity as well [16]. Not surprisingly, the Mediterranean diet, which is believed to be protective against diabetes and cardiovascular disease, has been affiliated with lower white matter hyperintensity volume on MRI scans of the brain [17]. All of these studies suggest that chronic diet-related conditions negatively affect brain white matter, and in turn play a significant role in precipitating white matter disease and cognitive decline. This review will focus on the potential metabolic, inflammatory, and microvascular determinants of white matter disease and cognitive decline in the context of chronic diet-related diseases.
Metabolic determinants
Diabetes
Among patients with DMII, cognitive function was found to have an inverse relationship with hemoglobin A1C levels [18]. This corresponds with another study which showed that diabetic patients had greater white matter hyperintensity volume and brain tissue loss compared to non-diabetic patients. Furthermore, it was shown that long-term weight loss interventions were associated with lower white matter hyperintensitiy volume, suggesting that treating diabetes itself may be the key to preventing neurodegeneration and thus, cognitive decline in diabetic patients [15]. While the mechanism underlying how DMII contributes to the development and progression of cognitive decline remains unclear, the current literature has proposed various theories that may explain why patients with DMII are at higher risk of neurodegenerative diseases. Recent investigations suggest that altered insulin signaling may be a contributory factor in cognitive decline. It is well-known that insulin receptors are highly expressed in cognition-related regions of the brain, as well as the blood brain barrier [19]. One study revealed that reduced brain insulin signaling in mouse models of diabetes increased tau beta phosphorylation and amyloid beta peptide levels, both of which are hallmark characteristics of Alzheimer’s disease [20-22]. Some researchers suggest that insulin resistance at the blood brain barrier reduces the amount of glucose that can reach the brain, resulting in neuronal injury [23]. While others proposed that the diabetic state may lead to a hyperglycemic condition in the brain that would result in the formation of glycated end products, which in turn can induce neuroinflammation [23]. Thus, both hyperglycemic and hypoglycemic conditions in the brain can lead to cognitive dysfunction. On the whole, there is limited information elucidating the mechanistic interactions between diabetes and cognitive decline, and more specifically how the metabolic changes that come with DMII affects white matter tissue in the brain. Although there is strong data showing that diabetic patients have higher prevalence of white matter hyperintensities and cognitive decline, more research is needed to decode how insulin resistance and/or hyperglycemia directly leads to white matter disease and cognitive decline.
Obesity and metabolic syndrome
Central obesity is an independent risk factor of dementia. Larger sagittal abdominal diameter was shown to be associated with an increased risk of developing dementia [24]. This is consistent with a number of studies that have shown that patients with higher body mass index (BMI) were more likely to develop Alzheimer’s disease later in life [25]. Obesity also has been shown to be independently associated with brain imaging changes before the clinical manifestation of cardiovascular or cerebrovascular disease [14]. Obese individuals were more likely to have decreased total cerebral volume and reduced white matter integrity compared to non-obese individuals [14,16]. Likewise, studies indicate that patients with metabolic syndrome have higher brain fatty acid uptake and greater accumulation of fatty acids in the brain in comparison to healthy subjects, with white matter having the highest mean percentage increment [26]. In mouse model studies, it was shown that diet-induced obesity produced higher levels of reactive oxygen species in the brain [27]. Hence, oxidative stress may be responsible for cognitive dysfunction following obesity. This relationship between obesity and cognitive decline suggests that treating obesity itself may lead to better cognitive health. Although there is not sufficient evidence to support the use of bariatric surgery for the prevention and treatment of dementia in obese patients, there has been clinical research showing improved memory function two years following bariatric surgery [28]. Overall, current literature shows that obesity has a deleterious effect on the brain but further investigation is needed to better understand the relationship between obesity, white matter disease, and cognitive dysfunction.
Inflammatory determinants
RAGE signaling, COX-2 expression, and platelet activation
Mounting evidence points to neuroinflammation as a key contributor of cognitive decline [29]. Various studies have linked high serum levels of inflammatory markers, such as C-reactive protein, tumor necrosis factor alpha (TNF-α), and interleukin 6 (IL-6), with cognitive dysfunction [30,31]. One hypothesized mechanism explaining this relationship between neuroinflammation and cognitive decline involves activation of receptors of advanced glycation end-products (RAGE), which are present on microglia and neurons in the brain, including the hippocampus, entorhinal cortex, and superior frontal gyrus [32]. RAGE activation turns on nuclear factor kappa B (NF-κB), which is a transcription factor that controls several pro-inflammatory genes. Some have suggested that RAGE could be involved in the injury of the brain in Alzheimer’s disease through neuroinflammation triggered by activation of RAGE signaling [33]. This is consistent with a study that demonstrated that diabetic patients with mild cognitive impairment had significantly higher serum levels of advanced glycated end-products (AGEs), RAGE, and C-reactive protein compared to diabetic patients without mild cognitive impairment [34]. Furthermore, amyloid-beta peptides activate cyclooxygenase-2 (COX-2) expression in a dose-dependent manner and treatment with COX-2 inhibitors or ibuprofen was shown to reverse this effect, suggesting that non-steroidal anti-inflammatory drugs (NSAIDs) may be used to reduce the risk of Alzheimer’s disease [35]; however, other studies suggest that COX-2 inhibitors may hasten dementia [36]. Hence, it remains controversial whether NSAIDs can be used in the treatment of Alzheimer’s disease. Recent literature has shown that inflammation-related cytokines and growth factors have been inversely associated with integrity of myelin sheath in individuals with bipolar disorder [37]. Activation of platelet function also has been associated with white matter lesions accompanied by cognitive decline [38]. Thus, it can be postulated that neuroinflammation induces white matter damage, which further promotes cognitive dysfunction.
Microvascular determinants
Hypertension: arterial stiffness and ischemia
Even before the manifestation of cardiovascular or cerebrovascular disease, hypertension has been linked to whole-brain volumetric reductions and variable pattern of increased white matter hyperintensities [14]. Although the exact mechanism behind how hypertension exacerbates cognitive dysfunction is still unclear, various studies have suggested that arterial stiffness is responsible for cognitive decline in patients with hypertension [39-41]. Current literature also has shown strong direct correlations between arterial stiffness and white matter hyperintensities [42], reinforcing the link between hypertension and cognitive decline. An additional explanation of how hypertension contributes to cognitive decline is ischemia caused by cerebral small vessel disease. Studies have shown that elevated blood pressure is a risk factor of cerebral small vessel disease [43] and that severity of cerebral small vessel disease is strongly correlated with severity of cognitive impairment [44]. Additionally, perfusion MRI studies have shown that white matter hyperintensities regions are characterized by decreased blood flow [45]. Thus, it makes sense that blunting or reversing the decrease in cerebral blood flow through the use of antihypertensive treatments would be associated with improved cognitive function [46].
Blood brain barrier dysfunction
Recently, it has been observed in multiple studies that rats fed with high energy diets, specifically diets rich in saturated fats and cholesterol, have increased blood brain barrier permeability along with cognitive dysfunction [47-49]. In conjunction with these studies, another investigation demonstrated that anti-inflammatory and lipid-lowering agents could reverse the high fat-induced blood brain barrier damage in rats [47]. Since blood brain barrier injury has been recognized as another contributory factor to the development and progression of cognitive impairment, it would be expected that reversal of blood brain barrier damage will be accompanied with improvement in cognitive function. However, the current level of evidence from several randomized controlled trials have been inconclusive in showing whether lipid-lowering medications, such as statins, can be used for the prevention or treatment of dementia [50].
Conclusions and clinical implications
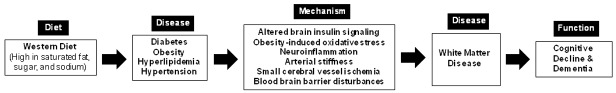
Consumption of the western diet contributes to the development of major chronic diseases, such as diabetes, obesity, hyperlipidemia, and hypertension. In turn, these chronic conditions lead to metabolic, inflammatory, and microvascular changes that affect many parts of our body, including the brain. The western diet is also suspected to directly contribute to cognitive impairment as a result of increases in blood lipids, sugars, and sodium. Our review shows that metabolic, inflammatory, and microvascular changes accompanying chronic diet-related diseases play a significant role in promoting cognitive decline. Abundance evidence shows that there is a strong correlation between these factors and white matter lesions in the brain, allowing us to reasonably speculate that these factors do so by inducing damage to the white matter (Figure 1). Consequently, treating these chronic conditions, such as obesity, diabetes, and hypertension, may be the key to preventing the development and progression of white matter disease and cognitive decline. However, sufficient evidence to recommend anti-inflammatory or lipid-lowering drugs for the prevention and treatment of dementia is not currently available. Further investigation is needed to elucidate the exact mechanistic interactions between these metabolic, inflammatory, and microvascular determinates and white matter disease. A mechanistic understanding of white matter disease is essential to improving our current approach to preventing and treating neurodegenerative diseases, as well as identifying potential targets for further drug development.
Figure 1.
Proposed relationship between western diet, white matter disease, and cognitive decline. Consumption of the western diet contributes to the development of chronic conditions, such as diabetes, obesity, hypertension, and hyperlipidemia, which in turn results in metabolic, inflammatory, and microvascular changes that induce injury to the white matter of the brain. Subsequently, white matter disease promotes the development and progression of cognitive decline and dementia.
Acknowledgements
This work was supported by the UC Davis Medical Student Research Fellowship Fund, the Richard A. and Nora Eccles Harrison Endowed Chair in Diabetes Research Fund, and the National Institutes of Health through the following grants: the National Center for Advancing Translational Sciences UL1 TR001860, National Institute on Aging R01 AG039094, and National Institute of Diabetes and Digestive and Kidney Diseases U24 DK092993-05S1 (to J.C. Rutledge) and U24 DK092993 (to K, Lloyd). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Disclosure of conflict of interest
None.
References
- 1.Xu J, Murphy SL, Kochanek KD, Bastian BA. Deaths: Final Data for 2013. Natl Vital Stat Rep. 2016;64:1–119. [PubMed] [Google Scholar]
- 2.Hebert LE, Weuve J, Scherr PA, Evans DA. Alzheimer disease in the United States (2010-2050) estimated using the 2010 census. Neurology. 2013;80:1778–1783. doi: 10.1212/WNL.0b013e31828726f5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Jacka FN, Pasco JA, Mykletun A, Williams LJ, Hodge AM, O’Reilly SL, Nicholson GC, Kotowicz MA, Berk M. Association of Western and traditional diets with depression and anxiety in women. Am J Psychiatry. 2010;167:305–311. doi: 10.1176/appi.ajp.2009.09060881. [DOI] [PubMed] [Google Scholar]
- 4.Jacka FN, Cherbuin N, Anstey KJ, Butterworth P. Dietary Patterns and Depressive Symptoms over Time: Examining the Relationships with Socioeconomic Position, Health Behaviours and Cardiovascular Risk. PLoS One. 2014;9:e87657. doi: 10.1371/journal.pone.0087657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Jacka FN, Cherbuin N, Anstey KJ, Sachdev P, Butterworth P. Western diet is associated with a smaller hippocampus: a longitudinal investigation. BMC Med. 2015;13:215. doi: 10.1186/s12916-015-0461-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dearborn JL, Knopman D, Sharrett AR, Schneider AL, Jack CR Jr, Coker LH, Alonso A, Selvin E, Mosley TH, Wagenknecht LE, Windham BG, Gottesman RF. The metabolic syndrome and cognitive decline in the Atherosclerosis Risk in Communities study (ARIC) Dement Geriatr Cogn Disord. 2014;38:337–346. doi: 10.1159/000362265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fazekas F, Kleinert R, Offenbacher H, Schmidt R, Kleinert G, Payer F, Radner H, Lechner H. Pathologic correlates of incidental MRI white matter signal hyperintensities. Neurology. 1993;43:1683–1689. doi: 10.1212/wnl.43.9.1683. [DOI] [PubMed] [Google Scholar]
- 8.Debette S, Markus HS. The clinical importance of white matter hyperintensities on brain magnetic resonance imaging: systematic review and meta-analysis. BMJ. 2010;341:c3666. doi: 10.1136/bmj.c3666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fernando MS, Ince PG. Vascular pathologies and cognition in a population-based cohort of elderly people. J Neurol Sci. 2004;226:13–17. doi: 10.1016/j.jns.2004.09.004. [DOI] [PubMed] [Google Scholar]
- 10.Provenzano FA, Muraskin J, Tosto G, Narkhede A, Wasserman BT, Griffith EY, Guzman VA, Meier IB, Zimmerman ME, Brickman AM. White matter hyperintensities and cerebral amyloidosis: necessary and sufficient for clinical expression of Alzheimer disease? JAMA Neurol. 2013;70:455–461. doi: 10.1001/jamaneurol.2013.1321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kandiah N, Mak E, Ng A, Huang S, Au WL, Sitoh YY, Tan LC. Cerebral white matter hyperintensity in Parkinson’s disease: a major risk factor for mild cognitive impairment. Parkinsonism Relat Disord. 2013;19:680–683. doi: 10.1016/j.parkreldis.2013.03.008. [DOI] [PubMed] [Google Scholar]
- 12.Gunning-Dixon FM, Raz N. The cognitive correlates of white matter abnormalities in normal aging: a quantitative review. Neuropsychology. 2000;14:224–232. doi: 10.1037//0894-4105.14.2.224. [DOI] [PubMed] [Google Scholar]
- 13.Brickman AM, Honig LS, Scarmeas N, Tatarina O, Sanders L, Albert MS, Brandt J, Blacker D, Stern Y. Measuring cerebral atrophy and white matter hyperintensity burden to predict the rate of cognitive decline in Alzheimer disease. Arch Neurol. 2008;65:1202–1208. doi: 10.1001/archneur.65.9.1202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Friedman JI, Tang CY, de Haas HJ, Changchien L, Goliasch G, Dabas P, Wang V, Fayad ZA, Fuster V, Narula J. Brain imaging changes associated with risk factors for cardiovascular and cerebrovascular disease in asymptomatic patients. JACC Cardiovasc Imaging. 2014;7:1039–1053. doi: 10.1016/j.jcmg.2014.06.014. [DOI] [PubMed] [Google Scholar]
- 15.Espeland MA, Erickson K, Neiberg RH, Jakicic JM, Wadden TA, Wing RR, Desiderio L, Erus G, Hsieh MK, Davatzikos C, Maschak-Carey BJ, Laurienti PJ, Demos-McDermott K, Bryan RN Action for Health in Diabetes Brain Magnetic Resonance Imaging (Look AHEAD Brain) Ancillary Study Research Group. Brain and White Matter Hyperintensity Volumes After 10 Years of Random Assignment to Lifestyle Intervention. Diabetes Care. 2016;39:764–771. doi: 10.2337/dc15-2230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kullmann S, Schweizer F, Veit R, Fritsche A, Preissl H. Compromised white matter integrity in obesity. Obes Rev. 2015;16:273–281. doi: 10.1111/obr.12248. [DOI] [PubMed] [Google Scholar]
- 17.Gardener H, Scarmeas N, Gu Y, Boden-Albala B, Elkind MS, Sacco RL, DeCarli C, Wright CB. Mediterranean diet and white matter hyperintensity volume in the Northern Manhattan Study. Arch Neurol. 2012;69:251–256. doi: 10.1001/archneurol.2011.548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cukierman-Yaffe T, Gerstein HC, Williamson JD, Lazar RM, Lovato L, Miller ME, Coker LH, Murray A, Sullivan MD, Marcovina SM, Launer LJ Action to Control Cardiovascular Risk in Diabetes-Memory in Diabetes (ACCORD-MIND) Investigators. Relationship Between Baseline Glycemic Control and Cognitive Function in Individuals With Type 2 Diabetes and Other Cardiovascular Risk Factors. The Action to Control Cardiovascular Risk in Diabetes-Memory in Diabetes (ACCORD-MIND) Diabetes Care. 2009;32:221–226. doi: 10.2337/dc08-1153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Stephen CW, Randy JS, Denis GB, Michael WS. Insulin and the Blood-Brain Barrier. Curr Pharm Des. 2003;9:795–800. doi: 10.2174/1381612033455323. [DOI] [PubMed] [Google Scholar]
- 20.Jolivalt CG, Lee CA, Beiswenger KK, Smith JL, Orlov M, Torrance MA, Masliah E. Defective insulin signaling pathway and increased glycogen synthase kinase-3 activity in the brain of diabetic mice: Parallels with Alzheimer’s disease and correction by insulin. J Neurosci Res. 2008;86:3265–3274. doi: 10.1002/jnr.21787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ramos-Rodriguez JJ, Jimenez-Palomares M, Murillo-Carretero MI, Infante-Garcia C, Berrocoso E, Hernandez-Pacho F, Lechuga-Sancho AM, Cozar-Castellano I, Garcia-Alloza M. Central vascular disease and exacerbated pathology in a mixed model of type 2 diabetes and Alzheimer’s disease. Psychoneuroendocrinology. 2015;62:69–79. doi: 10.1016/j.psyneuen.2015.07.606. [DOI] [PubMed] [Google Scholar]
- 22.Ramos-Rodriguez JJ, Spires-Jones T, Pooler AM, Lechuga-Sancho AM, Bacskai BJ, Garcia-Alloza M. Progressive Neuronal Pathology and Synaptic Loss Induced by Prediabetes and Type 2 Diabetes in a Mouse Model of Alzheimer’s Disease. Mol Neurobiol. 2016 doi: 10.1007/s12035-016-9921-3. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 23.Convit A. Links between cognitive impairment in insulin resistance: An explanatory model. Neurobiol Aging. 2005;26(Suppl 1):31–35. doi: 10.1016/j.neurobiolaging.2005.09.018. [DOI] [PubMed] [Google Scholar]
- 24.Whitmer RA, Gustafson DR, Barrett-Connor E, Haan MN, Gunderson EP, Yaffe K. Central obesity and increased risk of dementia more than three decades later. Neurology. 2008;71:1057–1064. doi: 10.1212/01.wnl.0000306313.89165.ef. [DOI] [PubMed] [Google Scholar]
- 25.Xu WL, Atti AR, Gatz M, Pedersen NL, Johansson B, Fratiglioni L. Midlife overweight and obesity increase late-life dementia risk: a population-based twin study. Neurology. 2011;76:1568–1574. doi: 10.1212/WNL.0b013e3182190d09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Karmi A, Iozzo P, Viljanen A, Hirvonen J, Fielding BA, Virtanen K, Oikonen V, Kemppainen J, Viljanen T, Guiducci L, Haaparanta-Solin M, Nagren K, Solin O, Nuutila P. Increased brain fatty acid uptake in metabolic syndrome. Diabetes. 2010;59:2171–2177. doi: 10.2337/db09-0138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Freeman LR, Zhang L, Nair A, Dasuri K, Francis J, Fernandez-Kim SO, Bruce-Keller AJ, Keller JN. Obesity increases cerebrocortical reactive oxygen species and impairs brain function. Free Radic Biol Med. 2013;56:226–233. doi: 10.1016/j.freeradbiomed.2012.08.577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Alosco ML, Spitznagel MB, Strain G, Devlin M, Cohen R, Paul R, Crosby RD, Mitchell JE, Gunstad J. Improved memory function two years after bariatric surgery. Obesity (Silver Spring) 2014;22:32–38. doi: 10.1002/oby.20494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pistell PJ, Morrison CD, Gupta S, Knight AG, Keller JN, Ingram DK, Bruce-Keller AJ. Cognitive impairment following high fat diet consumption is associated with brain inflammation. J Neuroimmunol. 2010;219:25–32. doi: 10.1016/j.jneuroim.2009.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Yaffe K, Lindquist K, Penninx BW, Simonsick EM, Pahor M, Kritchevsky S, Launer L, Kuller L, Rubin S, Harris T. Inflammatory markers and cognition in well-functioning African-American and white elders. Neurology. 2003;61:76–80. doi: 10.1212/01.wnl.0000073620.42047.d7. [DOI] [PubMed] [Google Scholar]
- 31.Schuitemaker A, Dik MG, Veerhuis R, Scheltens P, Schoonenboom NS, Hack CE, Blankenstein MA, Jonker C. Inflammatory markers in AD and MCI patients with different biomarker profiles. Neurobiol Aging. 2009;30:1885–1889. doi: 10.1016/j.neurobiolaging.2008.01.014. [DOI] [PubMed] [Google Scholar]
- 32.Lue LF, Walker DG, Brachova L, Beach TG, Rogers J, Schmidt AM, Stern DM, Yan SD. Involvement of microglial receptor for advanced glycation endproducts (RAGE) in Alzheimer’s disease: identification of a cellular activation mechanism. Exp Neurol. 2001;171:29–45. doi: 10.1006/exnr.2001.7732. [DOI] [PubMed] [Google Scholar]
- 33.Lue LF, Andrade C, Sabbagh M, Walker D. Is There Inflammatory Synergy in Type II Diabetes Mellitus and Alzheimer’s Disease? Int J Alzheimers Dis. 2012;2012:918680. doi: 10.1155/2012/918680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gorska-Ciebiada M, Saryusz-Wolska M, Borkowska A, Ciebiada M, Loba J. C-Reactive Protein, Advanced Glycation End Products, and Their Receptor in Type 2 Diabetic, Elderly Patients with Mild Cognitive Impairment. Front Aging Neurosci. 2015;7:209. doi: 10.3389/fnagi.2015.00209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Woodling NS, Colas D, Wang Q, Minhas P, Panchal M, Liang X, Mhatre SD, Brown H, Ko N, Zagol-Ikapitte I, van der Hart M, Khroyan TV, Chuluun B, Priyam PG, Milne GL, Rassoulpour A, Boutaud O, Manning-Bog AB, Heller HC, Andreasson KI. Cyclooxygenase inhibition targets neurons to prevent early behavioural decline in Alzheimer’s disease model mice. Brain. 2016;139:2063–81. doi: 10.1093/brain/aww117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kukar T, Murphy MP, Eriksen JL, Sagi SA, Weggen S, Smith TE, Ladd T, Khan MA, Kache R, Beard J, Dodson M, Merit S, Ozols VV, Anastasiadis PZ, Das P, Fauq A, Koo EH, Golde TE. Diverse compounds mimic Alzheimer disease-causing mutations by augmenting A[beta] 42 production. Nat Med. 2005;11:545–550. doi: 10.1038/nm1235. [DOI] [PubMed] [Google Scholar]
- 37.Benedetti F, Poletti S, Hoogenboezem TA, Mazza E, Ambree O, de Wit H, Wijkhuijs AJ, Locatelli C, Bollettini I, Colombo C, Arolt V, Drexhage HA. Inflammatory cytokines influence measures of white matter integrity in Bipolar Disorder. J Affect Disord. 2016;202:1–9. doi: 10.1016/j.jad.2016.05.047. [DOI] [PubMed] [Google Scholar]
- 38.Kuriyama N, Mizuno T, Yasuike H, Matsuno H, Kawashita E, Tamura A, Ozaki E, Matsui D, Watanabe I, Koyama T, Miyatani F, Kondo M, Tokuda T, Ohshima Y, Muranishi M, Akazawa K, Takada A, Takeda K, Matsumoto S, Mizuno S, Yamada K, Nakagawa M, Watanabe Y. CD62-mediated activation of platelets in cerebral white matter lesions in patients with cognitive decline. Arch Gerontol Geriatr. 2016;62:118–124. doi: 10.1016/j.archger.2015.09.001. [DOI] [PubMed] [Google Scholar]
- 39.Taniguchi Y, Fujiwara Y, Nofuji Y, Nishi M, Murayama H, Seino S, Tajima R, Matsuyama Y, Shinkai S. Prospective Study of Arterial Stiffness and Subsequent Cognitive Decline Among Community-Dwelling Older Japanese. J Epidemiol. 2015;25:592–599. doi: 10.2188/jea.JE20140250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Gutierrez J, Marshall RS, Lazar RM. Indirect measures of arterial stiffness and cognitive performance in individuals without traditional vascular risk factors or disease. JAMA Neurol. 2015;72:309–315. doi: 10.1001/jamaneurol.2014.3873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Tsao CW, Himali JJ, Beiser AS, Larson MG, DeCarli C, Vasan RS, Mitchell GF, Seshadri S. Association of arterial stiffness with progression of subclinical brain and cognitive disease. Neurology. 2016;86:619–626. doi: 10.1212/WNL.0000000000002368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Saji N, Shimizu H, Kawarai T, Tadano M, Kita Y, Yokono K. Increased brachial-ankle pulse wave velocity is independently associated with white matter hyperintensities. Neuroepidemiology. 2011;36:252–257. doi: 10.1159/000328260. [DOI] [PubMed] [Google Scholar]
- 43.van Dijk EJ, Prins ND, Vrooman HA, Hofman A, Koudstaal PJ, Breteler MM. Progression of cerebral small vessel disease in relation to risk factors and cognitive consequences: Rotterdam Scan study. Stroke. 2008;39:2712–2719. doi: 10.1161/STROKEAHA.107.513176. [DOI] [PubMed] [Google Scholar]
- 44.Smallwood A, Oulhaj A, Joachim C, Christie S, Sloan C, Smith AD, Esiri M. Cerebral subcortical small vessel disease and its relation to cognition in elderly subjects: a pathological study in the Oxford Project to Investigate Memory and Ageing (OPTIMA) cohort. Neuropathol Appl Neurobiol. 2012;38:337–343. doi: 10.1111/j.1365-2990.2011.01221.x. [DOI] [PubMed] [Google Scholar]
- 45.Brickman AM, Zahra A, Muraskin J, Steffener J, Holland CM, Habeck C, Borogovac A, Ramos MA, Brown TR, Asllani I, Stern Y. Reduction in cerebral blood flow in areas appearing as white matter hyperintensities on magnetic resonance imaging. Psychiatry Res. 2009;172:117–120. doi: 10.1016/j.pscychresns.2008.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Efimova IY, Efimova NY, Triss SV, Lishmanov YB. Brain perfusion and cognitive function changes in hypertensive patients. Hypertens Res. 2008;31:673–678. doi: 10.1291/hypres.31.673. [DOI] [PubMed] [Google Scholar]
- 47.Pallebage-Gamarallage M, Lam V, Takechi R, Galloway S, Clark K, Mamo J. Restoration of dietary-fat induced blood-brain barrier dysfunction by anti-inflammatory lipid-modulating agents. Lipids Health Dis. 2012;11:117. doi: 10.1186/1476-511X-11-117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Freeman LR, Granholm AC. Vascular changes in rat hippocampus following a high saturated fat and cholesterol diet. J Cereb Blood Flow Metab. 2012;32:643–653. doi: 10.1038/jcbfm.2011.168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Davidson TL, Monnot A, Neal AU, Martin AA, Horton JJ, Zheng W. The effects of a high-energy diet on hippocampal-dependent discrimination performance and blood-brain barrier integrity differ for diet-induced obese and diet-resistant rats. Physiol Behav. 2012;107:26–33. doi: 10.1016/j.physbeh.2012.05.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Gauthier JM, Massicotte A. Statins and their effect on cognition: Let’s clear up the confusion. Can Pharm J. 2015;148:150–155. doi: 10.1177/1715163515578692. [DOI] [PMC free article] [PubMed] [Google Scholar]