Abstract
Recent therapeutic advances in oncology have been driven by the identification of tumour genotype variations between patients, called interpatient heterogeneity, that predict the response of patients to targeted treatments. Subpopulations of cancer cells with unique genomes in the same patient may exist across different geographical regions of a tumour or evolve over time, called intratumour heterogeneity. Sequencing technologies can be used to characterize intratumour heterogeneity at diagnosis, monitor clonal dynamics during treatment and identify the emergence of clinical resistance during disease progression. Genetic interpatient and intratumour heterogeneity can pose challenges for the design of clinical trials that use these data.
There is great promise that knowledge of the biological drivers of cancer will lead to personalized cancer treatment. Oncologists increasingly use molecular characterization of a sample of primary or metastatic tumour to guide their selection of treatments for an individual patient. However, they usually rely on a limited sample of cancer tissue that cannot represent heterogeneity between and within patients.
Cancer genomics studies, including large-scale collaborative sequencing projects such as The Cancer Genome Atlas (TCGA) and the International Cancer Genome Consortium (ICGC), have catalogued genetic interpatient tumour heterogeneity for cancers of the same histological subtype. Non-genetic phenotypic and functional heterogeneity is also well recognized (see the Review by Meacham and Morrison on page 328), as is heterogeneity of the tumour microenvironment (see the Review by Junttila and de Sauvage on page 346). Comprehensive characterization of multiple tumour specimens obtained from the same patient illustrates that remarkable intratumour heterogeneity might exist between geographical regions in the same tumour (spatial heterogeneity), as well as between the primary tumour and a subsequent local or distant recurrence in the same patient (temporal heterogeneity). Inter- and intratumour heterogeneity pose a challenge to personalized cancer medicine because a single needle biopsy or surgical excision is unlikely to accurately capture the complete genomic landscape of a patient’s cancer. Genomic characterization of cell-free circulating tumour DNA (ctDNA) or circulating tumour cells (CTCs) may offer an opportunity to assess clonal dynamics throughout the course of a patient’s illness and identify drivers of therapeutic resistance. Here, we review the clinical implications of interpatient and intratumour heterogeneity for cancer diagnosis, making a prognosis, treatment selection and resistance. We discuss how clinical trials that are restricted to molecular subtypes of cancer could incorporate studies of tumour heterogeneity so that we can better understand the clinical impact of heterogeneity on therapeutic effectiveness and the emergence of treatment resistance.
Current models for diagnosis and treatment
Modern cancer treatment is based on accurate tissue diagnosis of samples obtained from needle biopsy or surgical excision. Cancerous tissues are analysed under a light microscope to evaluate histopathology, and immunostaining and selected molecular tests are used to establish a specific cancer diagnosis. Treatment is based on the anatomical location and tissue of origin of the primary tumour when cancer is localized to an organ site or when cancer has metastasized and the primary site can be identified by imaging or pathological examination. When solid tumours recur after treatment for localized disease or progress after systemic treatment for metastatic disease, taking another biopsy to guide treatment decisions is not routine1. Instead, further systemic treatment of patients with progressive metastatic disease is typically based on the identification of predictive biomarkers in archived primary specimens, which may no longer represent the current disease such as BRAF mutation in melanoma, HER2 (also known as ERBB2) amplification or overexpression in breast cancer, KRAS mutation in colorectal cancer and EGFR mutation in non-small-cell lung cancer2–6.
Intratumour heterogeneity and clonal evolution
The current approach to molecular biomarker testing to inform cancer treatment focuses on interpatient tumour heterogeneity. However, there is a growing recognition that intratumour heterogeneity within the same patient is clinically relevant because the status of predictive biomarkers that are used for making clinical decisions may evolve during tumour progression, in particular metastatic dissemination of the primary tumour to a distant organ or for established metastatic disease under the selection pressure of treatment. Nowell’s theory of clonal evolution states that cancers arise from a single cell of origin, develop genomic instability during replication and then undergo enrichment for the most aggressive clones through the processes of metastasis and the eradication of sensitive clones with cancer treatment7 (see the Reviews by Burrell et al. on page 338 and Klein on page 365). For example, discordance between oestrogen receptor (ER) expression in a primary breast cancer and subsequent distant metastases that may appear many years after completion of primary treatment is observed in 7–25% of patients8–11 (Table 1). Change in ER status may have important implications for treatment because patients with tumours that lack ER expression do not benefit from treatment with endocrine therapy such as tamoxifen or aromatase inhibitors12. Loss of ER or HER2 expression in breast cancer during metastasis is associated with a poorer outcome13,14. Although data from other tumour types are more limited, discordance of prognostic or predictive biomarker testing results between the primary tumour and metastases has been reported in other settings15–24 (Table 1).
Table 1.
Selected single parameter biomarker tests that are routinely used to inform clinical decision-making for advanced solid tumours, and reported frequencies of discordance between primary tumours and metastases
| Tumour type | Biomarker | Prognostic or predictive biomarker | Evidence of discordance |
|---|---|---|---|
| Oligodendroglioma | 1p and19q co-deletion MGMT promoter methylation |
Prognostic/predictive Prognostic/predictive |
Not applicable |
| Medullary thyroid | RET mutation | Prognostic102 | Unknown |
| Breast | ER expression PR expression HER2 amplification |
Prognostic/predictive Prognostic Prognostic/predictive |
7–25%8,11,14 16–49%8,11,12,14 3–24%13,24 |
| Lung |
EGFR mutation EML4-ALK translocation |
Prognostic/predictive Prognostic/predictive |
0–38%103,104 1–2%18,105 |
| Gastric | HER2 amplification | Prognostic106/predictive107 | 1–3%20,21 |
| Colorectal | KRAS mutation | Predictive | 0–10%22,108 |
| Melanoma | BRAF mutation | Prognostic/predictive | 4–25%109 |
| Gastrointestinal stromal |
KIT mutation PDGFRA mutation |
Predictive Predictive |
Acquired mutations evolve during tyrosine kinase inhibitor treatment110,111 |
ER, oestrogen receptor; PR, progesterone receptor
Before metastases are clinically apparent, clonal heterogeneity can be identified within the primary tumour25. For example, complex patterns of HER2 gene amplification detected by fluorescence in situ hybridization are seen in breast26 and gastro-oesophageal cancers27. Similar patterns of regional intratumour heterogeneity have been observed with mutation testing in other tumour types, including KRAS in colorectal cancer, BRAF in melanoma and EGFR in non-small-cell lung cancer23,28–31. Intratumour heterogeneity may account for resistance despite the matching of targeted treatment to the mutation, such as trastuzumab for HER2 amplified breast cancer32, EGFR monoclonal antibody treatment for KRAS-wildtype-colorectal cancer33 and EGFR tyrosine kinase inhibitor treatment for EGFR mutant non-small-cell lung cancer34, through the selection of clonal subpopulations with mutations that confer treatment resistance.
Strategies to measure intratumour heterogeneity
Recognition of intratumour heterogeneity to inform treatment decisions requires test methods that can be applied to clinical tumour samples. Genome-scale technologies provide an unbiased characterization of clonal heterogeneity within tumours beyond a specific genetic locus or a set of loci (see the Review by Burrell et al. on page 338). Studies with karyotype analysis and comparative genomic hybridization allow for detection of clonal subpopulations within the same tumour that can be differentiated on the basis of DNA content and chromosomal imbalances35. Newer techniques such as single nucleotide polymorphism arrays provide greater resolution and can identify smaller-scale allelic imbalances in specific genetic loci. Next-generation sequencing technology allows for the systematic enumeration of single nucleotide mutations and the identification of rare clonal subpopulations that are present in a small fraction of tumour cells. Sequencing studies of normal tissue, early pre-malignant precursors and malignant lesions derived from the same patient have been performed in secondary acute myeloid leukaemia derived from myelodysplastic syndrome36 and invasive breast cancer with adjacent pre-invasive neoplasia37. Clonal lineage has been reconstructed with the identification of antecedent founding clones in a pre-malignant precursor from which malignant disease evolved with the outgrowth of subclones with additional genomic alterations37.
In metastatic disease, recent studies have characterized the emergence of treatment-resistant subclones that were present at a minor frequency in the primary tumour38–44. This raises the tantalizing possibility that the model of cancer diagnosis and treatment in the future could involve characterization of subpopulations within the primary tumour45, monitoring of clonal dynamics during treatment and eradication of treatment-emergent clones. Clinical sequencing using less invasive sampling methods such as cytology specimens, CTC analysis and ctDNA would greatly facilitate this approach42,43,46–52. A recent study52 demonstrated that ctDNA detected using targeted gene sequencing for PIK3CA and TP53 mutations was associated with survival in patients with metastatic breast cancer. Levels of ctDNA were more closely correlated with response to treatment than CTCs or levels of the circulating cancer antigen CA15-3 detected in serum53. A further study involving serial ctDNA exome sequencing of six patients with advanced solid tumours demonstrated an increased representation of certain mutant alleles with the emergence of treatment resistance54.
Challenges of clinical assessment
Beyond initial proof-of-concept studies, larger clinical efforts are required to evaluate whether in-depth genomic characterization and serial monitoring of clonal dynamics leads to better patient care. The falling cost of next-generation sequencing has made high-coverage DNA sequencing of clinically relevant cancer genes accessible at the point of care55,56. Genomic assessment of interpatient and intratumour heterogeneity in the clinical environment57 has several practical challenges.
Surgical resections of primary tumours or metastatic lesions provide large volumes of tumour tissue that are required for assessment of regional heterogeneity and clonal diversity. Tumour specimens are routinely formalin fixed and paraffin embedded (FFPE) after surgical excision to preserve histology. Although tumour nucleic acids can degrade with formalin fixation and this can limit researchers’ ability to perform genome-scale analyses, particularly for RNA sequencing, advances in technology mean that the analysis is becoming more feasible. In addition, deciphering the precise spatial orientation of stored FFPE tumour blocks using the routine clinical annotation that is included in surgical pathology reports to reconstruct intratumour heterogeneity can be difficult. Serial characterization of metastatic lesions through core needle biopsy could be used to identify clonal evolution, but sampling bias may occur because only a limited geographical region of a tumour is analysed. ctDNA is more amenable to serial sampling and presumably represents cancer genomes from multiple metastatic sites. However, ctDNA analysis is in its infancy and is not yet routinely established in the clinical environment. Furthermore, whether there are important mutations that are unique to non-circulating populations of tumour cells is not yet known.
In the United States, clinical laboratories that test human specimens for the purpose of providing information on diagnosis, prevention or treatment of disease to the supervising physician must adhere to Clinical Laboratory Improvement Amendments (CLIA) standards and be accredited by the College of American Pathologists (CAP) for reimbursement58, and similar regulatory standards exist in other countries. Genome-scale sequencing was previously outside the purview of a clinical laboratory owing to the cost of massively parallel sequencing platforms, high-performance computing capacity and the sophisticated bioinformatics expertise that was required for sequence alignment and mutation calling. The recent development of bench-top next-generation sequencing instruments that offer high coverage (≥250 × read depth) of a large targeted panel of clinically relevant cancer genes is well suited to the work flow of a clinical laboratory59–61. The Next Generation Sequencing Standardization of Clinical Testing (Nex-StoCT) workgroup recommends that all clinically actionable mutations should be confirmed by independent analysis using an alternative method before reporting to the treating clinician62. This poses a problem when high-coverage next-generation sequencing identifies a low-frequency mutation that cannot be confirmed by Sanger or PCR sequencing owing to the limitations of sensitivity of direct sequencing methods.
Mutation verification can delay the reporting of results to the oncologist if multiple clinically actionable variants are detected by next-generation sequencing. Patients with metastatic cancer and their oncologists may not be willing to wait for these results before initiating a new treatment63. Deciding which mutation or mutations are clinically relevant, prioritizing mutations for treatment matching when multiple mutations are detected and developing a framework to report results to clinicians that can be easily interpreted are complex tasks. Few mutations have been validated with a high level of evidence for the prediction of treatment response64. Specific mutations may have different clinical implications depending on a cancer’s tissue of origin, such as BRAF(V600E) mutation in patients with melanoma or colorectal cancer65,66 and their response to vemurafenib monotherapy. For mutations in tumour-specific contexts for which there are no clinical studies available, preclinical drug sensitivity encyclopaedias can be mined to infer potential clinical relevance67,68. However, there are concerns about validating predictive genomic biomarkers across cell-line screening data sets69 and the lack of reproducibility of preclinical experiments70.
Trial designs that assess tumour heterogeneity
Despite the challenges associated with genomic assessment in the clinical environment, molecular characterization — from genotyping to targeted genome sequencing — through the use of stored FFPE samples or serially procured fresh tumour biopsies is increasingly used to complement histopathological diagnosis. Clinical-trial design frameworks for cancer diagnostics and therapeutics must be developed to efficiently and dynamically incorporate such genomic data and assess the value of matching profiled patients to specific interventions or targeted therapies.
There are several premises on which clinical-trial design frameworks in the cancer genome era are based. First, genetic aberrations exist in human malignancies with a subset that are present in different cancer types at variable frequencies. Aberrations with functional relevance that lead to cancer initiation, growth and metastasis are the targets of greatest clinical interest because they could potentially be used for diagnosis, prognosis and predicting response to therapy. Second, there are specific interventions or tolerable medicinal agents that may effectively modulate such targets. Last, intratumour heterogeneity and clonal evolution occur and there are feasible technologies to measure these phenomena in the clinical setting. The reliable quantification of both spatial and temporal variations in the molecular landscape of cancers would enable the development of therapeutic strategies to interrogate them. Although, currently, most approved targeted therapies and clinical trials focus on interpatient heterogeneity, considering intratumour heterogeneity will increasingly become important in the future.
Trial designs for interpatient heterogeneity
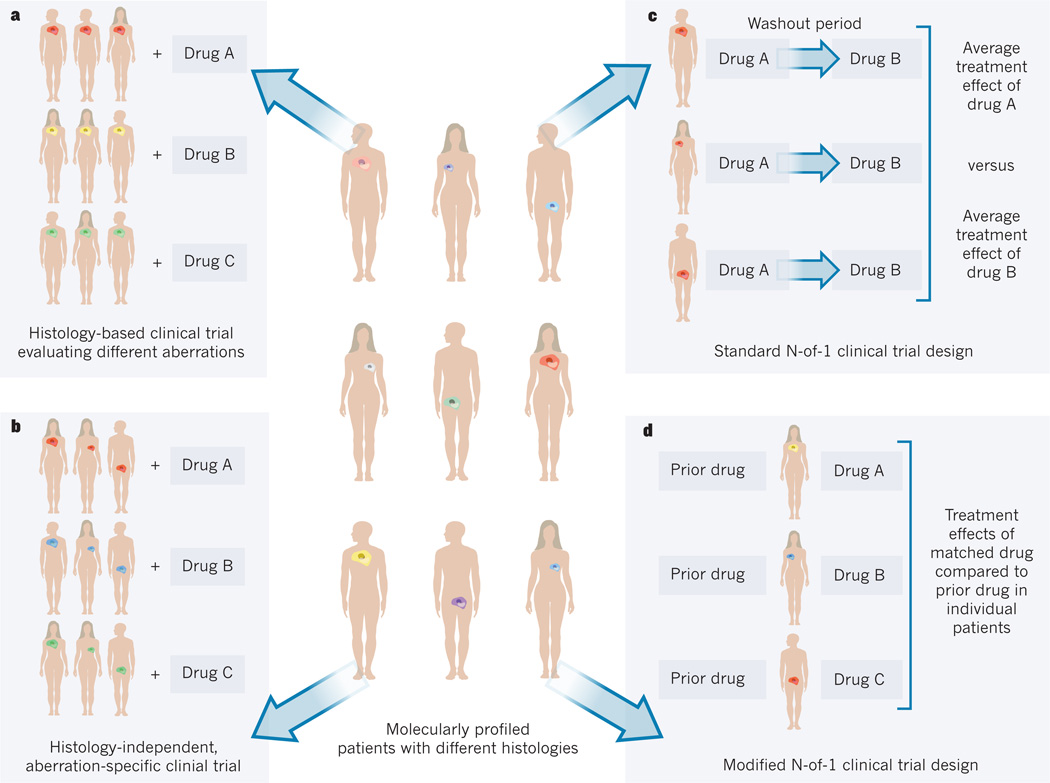
Clinical-trial design frameworks that focus on interpatient tumour heterogeneity are possible, assuming that detailed genomic characterization is feasible71 (Fig. 1).
Figure 1. Clinical-trial design frameworks.
In a population of molecularly profiled patients who have tumours of different histologies (shown by position of tumour) and molecular aberrations (shown as different colours), the framework for a clinical trial can take a number of forms. a, Histology-based clinical trials evaluate different molecular aberrations by enrolling patients with the same tumour histology but who harbour different aberrations, and match groups of patients to different drugs. b, Histology-independent, aberration-specific clinical trials, or ‘basket’ trials, enrol patients with different tumour histologies but who harbour the same or related molecular aberrations, and match drugs to the aberration specific or related groups. c, Standard N-of-1 trials randomly assign patients to different drugs in different sequential orders, with washout periods between drugs to minimize crossover effects. At completion, the individual effect of each drug and the average effects of each drug across individuals can be analysed. d, Modified N-of-1 trials use each patient as his or her own control and compare the treatment effect of the current matched drug with that of the most recent earlier drug.
Longitudinal cohort with nested trials
One framework currently used by many large cancer institutions and national cancer cooperative groups is to prospectively profile a large number of patients to establish a longitudinal cohort with clinical annotation such as demographics, histopathological diagnosis, earlier therapies and outcome (Table 2). Thus far, most clinical molecular profiling programmes worldwide have focused on the genomic characterization of limited but presumably representative specimens obtained at a single time point, typically in patients with metastatic disease who are suitable for systemic therapy. It is logical that a current sample would more accurately reflect the patient’s current disease than an archived sample, although it is unknown whether a small current specimen (for example, from a needle biopsy) is preferable to a larger historical sample (for example, from surgical resection). In instances in which archived tumour tissue is too scant to yield sufficient DNA, or has been exhausted owing to serial evaluations of single markers, then a fresh tumour biopsy would be necessary for genomic profiling. Although there is great enthusiasm for molecular characterization of tumour samples, the clinical use of this approach is still unproven. Some clinical trials of targeted drugs limit enrolment to patients with specific molecular perturbations; however, the effectiveness of such drugs is usually unconfirmed. Nonetheless, the coupling of a molecular characterization strategy with a drug development programme has been widely embraced, despite disparate results from different retrospective series and the lack of definitive supportive data72,73. In this context, patients with specific molecular aberrations are often ‘opportunistically’ enrolled into clinical trials of matching targeted agents. This framework is attractive to those running large programmes who have access to a robust panel of early phase clinical trials that test different molecular targets60,63,73–75. The panel of clinical trials can be ‘nested’ or embedded as distinct research activities under the auspices of an overarching platform of molecular profiling and target–drug matching.
Table 2.
Selected worldwide large-scale clinical molecular profiling programmes by institution or consortium
| Trial or programme name | Platforms or techniques | Genes and mutations | Cancer types | Tumour sample |
|---|---|---|---|---|
| Cancer Research UK, London | ||||
| Stratified Medicine Programme112 | PCR FISH |
9 genes 3 genes |
Melanoma, NSCLC, CRC and breast, prostate and ovarian cancer |
Archival |
| Dana-Farber Cancer Institute, Boston, Massachusetts | ||||
| PROFILE113 | Sequenom | OncoMap: 41 genes, 471 mutations |
All solid tumours | Archival |
| Curie Institute, Paris; French National Cancer Institute | ||||
| SHIVA (NCT01771458) | Ion Torrent PGM CytoScan HD |
AmpliSeq: 46 genes 29 genes |
All solid tumours | Fresh biopsy |
| Gustave Roussy Institute, France (non-paediatric trials) | ||||
| MOSCATO75 (NCT01566019) | aCGH PCR |
NA 96 mutations |
Solid tumour phase I patients | Fresh Biopsy |
| SAFIR01 (NCT01414933) | aCGH PCR |
NA 2 genes |
Breast cancer | Fresh Biopsy |
| MSN | PCR FISH |
Seqcan: 30 genes 5 genes |
Melanoma, SCLC and NSCLC | Fresh Biopsy |
| Massachusetts General Hospital, Boston | ||||
| NS114 | SNaPshot | 14 genes, >50 mutations | NSCLC, CRC, melanoma and breast cancer |
Archival |
| MD Anderson Cancer Center, Houston, Texas | ||||
| T9 Program115 | Sequenom | >40 genes | All solid tumours | Archival |
| IMPACT73 (NCT00851032) | PCR FISH |
10 genes 1 gene |
All solid tumours | Archival |
| Clearing House protocol116 | PCR Illumina NS, Ion Torrent NS NS |
~100 genes T200: 200 genes Whole genome |
All solid tumours | Archival or fresh biopsy |
| Memorial Sloan-Kettering Cancer Center, New York | ||||
| IMPACT (NCT01775072) | Illumina HiSeq Sequenom or MiSeq |
275 genes (Research assays) NS (Clinical assays) |
All solid tumours | Archival |
| Netherlands | ||||
| Centre for Personalized Cancer Treatment117 |
Ion Torrent PGM 5500xl SOLiD |
~150 genes >2,000 genes |
Solid tumours | Fresh biopsy |
| Norwegian Cancer Genomics Consortium | ||||
| Nationwide programme118 | NS | Whole exome | 9 tumour types, both solid and haematopoietic |
Archival or fresh biopsy |
| Princess Margaret Cancer Centre, Toronto, Canada | ||||
| IMPACT60 (NCT01505400) | MiSeq Sequenom |
TSACP: 48 genes, >700 mutations. Customized panel: 23 genes, 279 mutations |
Selected solid tumours | Archival |
| Vall d’Hebron Institute of Oncology, Barcelona, Spain | ||||
| NS72, 119 | Sequenom llumina GAIIx |
OncoCarta, 19 genes, 238 mutations NS |
Breast cancer, solid tumour phase I patients |
Archival |
| Vanderbilt-Ingram Cancer Center, Nashville, Tennessee | ||||
| PCMI120 | SNaPshot | 6–8 genes and >40 mutations | Melanoma, NSCLC, CRC and breast cancer |
Archival |
| WIN Consortium | ||||
| WINTHER83 (NCT01856296) | NGS CNV CGH |
NS NS NA |
Solid tumours | Fresh biopsy (tumour and matched normal) |
aCGH, array comparative genomic hybridization; CGH, comparative genomic hybridization; CNV, copy number variation; CRC, colorectal cancer; FISH, fluorescence in-situ hybridization; GAIIx, genome analyzer IIx; NA, not applicable; NGS, next-generation sequencing; NS, not stipulated; NSCLC, non-small-cell lung cancer; PCR, polymerase chain reaction; PCMI, personalized cancer medicine initiative; PGM, personal genome machine; SCLC, small-cell lung cancer; TSACP, TruSeq amplicon Cancer Panel.
Histology-based design
Other frameworks involve the evaluation of the target–agent matching strategy in large, prospectively conducted clinical trials (Table 2). For instance, histology-based and biomarker-integrated multicentre clinical trials aim to assess a variety of targeted agents matched to specific molecular profiles within a single tumour type (Fig. 1a). The FOCUS 4 trial supported by the UK Medical Research Council, for example, will enrol patients with advanced colorectal cancer who have responsive or stable disease after 16 weeks of chemotherapy76. On molecular profiling, patients with tumours that harbour commonly mutated oncogenes such as KRAS, BRAF or PIK3CA will be given targeted agents or a placebo. Other histology-based clinical trials include the US-based BATTLE-2 trial (NCT01248247) in lung cancer and the I-SPY 2 trial in breast cancer (NCT01042379)77–79 (Table 3).
Table 3.
Prospective and retrospective programmes that evaluate tumour heterogeneity by institution or consortium
| Trial or programme REFERENCES |
Platforms or techniques |
Coverage and depth | Cancer types | Fresh tumour acquisition |
Additional specimen collection |
Drugs | Additional procedures |
Type of heterogeneity |
|---|---|---|---|---|---|---|---|---|
| Dana-Farber Cancer Institute, Boston, Massachusetts; Broad Institute, Cambridge, Massachusetts; and Brigham and Women’s Hospital, Boston | ||||||||
| CanSeq121 | Sequenom NS |
OncoMap Whole exome, depth NS |
NSCLC, CRC, MBC and prostate cancer |
Biopsy at set time point depending on tumour type |
No | No | No | Interpatient |
| Heinrich Heine University, Düesseldorf | ||||||||
| DETECT III101 (NCT01619111) |
NS | NS | HER-2 MBC with HER2+ CTCs |
No | CTCs (CellSearch) |
SOC chemotherapy or endocrine therapy +/− lapatinib |
No | Intratumour |
| Massachusetts General Hospital, Boston | ||||||||
| Biopsies of Cancer Patients for Tumor Molecular Characterization (NCT01061944) |
NS | Genes NS, depth NS | All solid tumours | Biopsy of metastasis (SOC) |
NS | No | No | Interpatient |
| Mayo Clinic, Scottsdale, Arizona | ||||||||
| BEAUTY122 | NS | Whole genome, depth NS |
Non-metastatic breast cancer |
Biopsy of primary pre- and post-neoadjuvant chemotherapy. Primary resection |
NS | Paclitaxel +/− trastuzumab AC or FEC |
Xenografts | Intratumour Interpatient |
| MRC Clinical Trials Unit, London | ||||||||
| FOCUS 4 (ref. 76) |
PCR assays |
3 genes | CRC | Diagnostic, on treatment and PD biopsy (lesion NS) |
No | 5 treatment arms | No | Interpatient |
| MD Anderson Cancer Center, Houston, Texas | ||||||||
| BATTLE-2 (NCT01248247) |
PCR FISH |
11 biomarkers | NSCLC, PD on chemotherapy |
Biopsy (lesion NS) |
NS | Erlotinib, MK2206, AZD6224, |
No | Interpatient |
| BATTLE- Front line (NCT01263782) |
NSCLC, treatment naive |
sorafenib | Interpatient | |||||
| National Cancer Institute, Bethesda, MD | ||||||||
| I-SPY 2 (NCT01042379) |
TargetPrint HER2, Mamma- |
71 genes | Stage 3 breast cancer |
Pre-specified serial primary biopsies. Primary resection |
Blood | Experimental drugs with SOC chemotherapy |
Breast MRI | Interpatient |
| MPACT123 | NS | 22 genes for treatment, 80kb sequenced, 383 amplicons with ≥80% covered >450× |
All solid tumours | Biopsy of metastasis |
NS | MEK, mTOR, PARP, WEE1 inhibitors |
No | Interpatient |
| MATCH124 | NS | Genes and depth NS | All solid tumours and lymphoma. PD on 1 SOC treatment |
Pretreatment and PD biopsy (lesion NS) |
NS | Multiple targeted therapies on clinical trials |
RNA-Seq | Intratumour Interpatient |
| PREDICT Consortium | ||||||||
| E-PREDICT S-PREDICT (ISRCTN 22979604) |
GAIIx HiSeq |
Whole exome, transcriptome, average depth 30× |
Renal cell cancer | Biopsy of primary and metastasis. Nephrectomy |
NS | Everolimus or sunitinib |
Functional RN interference |
Intratumour Interpatient |
| Princess Margaret Cancer Centre, Toronto, Canada | ||||||||
| MATCH (NCT01703585) |
MiSeq | 48 genes, 212 amplicons average depth ~1,000× |
CRC, MBC, gynaecological cancers |
Serial biopsies of metastases at study start and on PD |
Blood: CTCs, ctDNA. Archival tumour |
No | No | Intratumour Interpatient |
| Sequenom | 23 genes, 279 hotspots | |||||||
| University College London | ||||||||
| TRACERx (NCT01888601) |
NS | Whole genome, whole exome, depth NS |
NSCLC | Biopsy of primary and metastasis. Primary resection |
Blood: ctDNA | SOC chemotherapy |
Functional imaging |
Intratumour Interpatient |
AC, adriamycin and cyclophosphamide; CRC, colorectal cancer; CTCs, circulating tumour cells; ctDNA, circulating tumour DNA; FEC, 5-fluorouracil, epirubicin, cyclophosphamide; FISH, fluorescence in situ hybridization; GAIIx, genome analyzer IIx; kb, kilobases; MBC, metastatic breast cancer; MRI, magnetic resonance imaging; NS, not stipulated; NSCLC, non-small-cell lung cancer; PCR, polymerase chain reaction; PD, progressive disease; SOC, standard of care.
Histology-agnostic, aberration-specific design
An alternative framework employs a histology-agnostic, aberration-specific design in which patients whose tumours harbour identical or related molecular profiles are treated in the same ‘basket’ with the same therapeutic regimen (Fig. 1b). An example is the inclusion of different tumour types that harbour PIK3CA mutations or amplifications into a basket trial that evaluates a PI(3)K α-isoform specific inhibitor (NCT01219699). This strategy may be adapted to increase enrolment of patients with tumour types that demonstrate early signals of antitumour activity while excluding those who lack preliminary response. Although this framework will not directly lead to regulatory approval, given its exploratory nature, it does provide a platform to determine the differences in functionality of the same molecular alteration across multiple cancer types.
N-of-1 clinical trial design
The N-of-1 clinical-trial design framework has been pursued for non-oncology diseases, most frequently in neuropsychiatric, pulmonary and musculoskeletal conditions80,81. In their standard context, N-of-1 trials involve individual patients who are typically blinded and randomly assigned to different treatment regimens or to a placebo in different sequential orders, with washout periods, in which patients receive no treatment, between regimen alterations to minimize crossover effects (Fig. 1c). There are limitations to the application of this framework in oncology. For instance, the switch from one regimen to another may occur before there is sufficient time for antitumour activity to be manifested, such that there may be an underestimation of therapeutic efficacy while increasing the risk of inducing drug resistance. Modified N-of-1 designs have been used to investigate the value of individualized therapy. The concept of using each individual patient as his or her own control, for example, to assess the growth modulation index by comparing the time to progression or progression-free survival (PFS) on a current regimen with that attained on the most recent prior treatment, represents such a modification of the N-of-1 design82 (Fig. 1d). This framework may become increasingly relevant for subsets of patients with rare molecular alterations, for which large randomized trials may never be feasible. The WINTHER trial83 (NCT01856296), led by the Worldwide Innovative Networking (WIN) Consortium, is an example of a modified N-of-1 design that is using a variety of advanced profiling technologies to comprehensively characterize oncogenic events in 200 patients with different cancers. The trial compares patients’ PFS on therapy guided by profiling results with that achieved on the regimen immediately preceding trial enrolment. However, the validity of this approach is unknown, given the uncertain correlation in PFS between sequential inactive therapies82.
Trial designs for intratumour heterogeneity
Establishing clinical-trial design frameworks in the context of intrapatient tumour heterogeneity and clonal evolution is challenging because dimensions of both space and time must be incorporated to reflect the dynamic nature of tumour biological characteristics within individuals.
Geographical heterogeneity
The execution of the aforementioned frameworks is typically based on molecular profiling of tumour specimens obtained from one geographical location. These samples, in addition to other biorepositories such as tissue banks and autopsy programmes, provide a means to build knowledge bases that help us to gain insight into complex molecular events such as intratumour heterogeneity40,84,85. One such initiative to build this type of knowledge base is the REACT study (NCT01505400). The aim of this study is to genomically evaluate all archived tumour samples from a cohort of molecularly profiled patients to assess heterogeneity and clonal evolution.
To prospectively assess geographical or spatial heterogeneity, profiling of multiregional tumour samples would be indicated. Although this is feasible (but rarely performed in surgical resections), it is impractical and potentially risky to take biopsies from multiple deep-seated metastatic lesions in every patient to examine the genotypes of different tumour cell clones. If tumour biopsies using fine-gauge needles (23-gauge or smaller) could yield sufficient quantities of tumour nucleic acids for molecular profiling, these would be an attractive alternative to large-bore needles owing to the lower risk of procedure-related complications. An ongoing prospective study called MATCH (NCT01703585) evaluating the quality and quantity of DNA obtained using different sizes of biopsy needles, could determine whether the use of fine-gauge needles is feasible for targeted sequencing. In addition, there are prospective and retrospective tumour-specific programmes that explore heterogeneity and evolution in relation to drug therapy (Table 3). The PREDICT programme for patients with renal cell cancer who are treated with neoadjuvant everolimus or sunitinib is an example of an explicitly designed study to evaluate heterogeneity in the primary tumour through multiregional sampling86,87. Ultimately, the development of non-invasive visualization techniques, such as molecular imaging using radionuclide-based methods that can quantify the expression of tumoral targets with high sensitivity and specificity, would be ideal88.
Temporal heterogeneity
Serial tumour sampling, especially at crucial time points in the disease course such as the development of metastatic disease or progression after initial response to systemic therapy, may reveal the emergence of dominant clones. This type of dynamic examination of clonal evolution is being conducted by programmes such as PREDICT86,87. Until less invasive techniques such as characterization of CTCs or ctDNA are validated to yield sufficient sensitivity and specificity to be representative of clonal distribution and evolutionary pattern, fresh tumour biopsies will probably be used to monitor these events, although limited biopsies may also not reflect the full genomic landscape89,90. Sensitivity of detection of somatic mutations is related to their frequencies in the analysed segments of cancer-related genes, and can be increased by using new techniques such as amplification and deep sequencing of selected genomic regions. ctDNA has already been used as a tracking tool for distinct existent clones51, as well as an early predictor of treatment response or resistance42,43,52 . Optimization of these methods to transition them from research to diagnostic laboratories would enable their applications in clinical trials and eventually in routine cancer care. Advances in molecular imaging that would make longitudinal surveillance possible would be desirable, although it is uncertain whether imaging can ever provide resolution at the level of target expression in tumour cells and be able to reflect changes in the clonal milieu.
Trial designs
The evaluation of geographical and temporal variations in tumour molecular profiles is complex. Their integration into the aforementioned clinical-trial design frameworks that focus on interpatient tumour heterogeneity is possible but would necessitate that the frameworks become dynamic models that consider changes across space and time within individuals. By using these frameworks, geographical heterogeneity and clonal evolution in tumour samples can be prospectively measured, but must first be correlated with clinical outcome to determine whether they portend a prognostic and/or predictive role. Interventions to modulate these phenomena would only be planned if they are demonstrated to have an important link to clinical outcome.
An example of intratumour heterogeneity in the clinic
Given the complexity of intratumour heterogeneity and clonal evolution, it is impossible to provide approaches that are universally applicable. As such, in reality, it is expected that adaptations of clinical trial designs for individuals will be tailored to the unique features of specific malignancies. By using breast cancer as a example, practical applications of these frameworks for prognosis and therapy (Table 4) are discussed below.
Table 4.
The different clinical-trial design frameworks and tumour-sampling strategies that can be used to evaluate intratumour heterogeneity and clonal evolution from pre-malignancy to the development of resistant metastases, using breast cancer as an example.
| Ductal carcinoma in situ | Localized cancer | Micrometastases | Macrometastases | Resistant disease |
|---|---|---|---|---|
| Clinical evaluation | ||||
| Surveillance of pre- malignancy to malignancy |
Forming a prognosis of metastatic potential |
Monitoring response to adjuvant therapy |
Targeting treatment to match driver clones |
Targeting treatment to match resistant clones |
| Evaluation strategies | ||||
| Multiregional sampling, if feasible |
Multiregional sampling Monitoring using CTCs or ctDNA |
Monitoring using CTCs or ctDNA |
Multiregional sampling Molecular imaging Serial sampling CTCs or ctDNA |
Multiregional sampling Serial sampling for CTCs or ctDNA Molecular imaging |
| Clinical-trial design frameworks | ||||
| Longitudinal cohort | Longitudinal cohort Histology-based design |
Longitudinal cohort Histology-based design |
Histology-based design Histology-agnostic basket design |
Histology-based design Histology-agnostic basket design N-of-1 design |
CTC, circulating tumour cell; ctDNA, circulating tumour DNA.
Pre-malignancy to malignancy
The establishment of a longitudinal cohort would enable long-term follow-up of patients with pre-malignant lesions, such as ductal carcinoma in situ (DCIS), for whom the disease might progress to invasive breast cancer. Retrospective analyses of cases with synchronous DCIS and invasive ductal carcinoma have shown that this progression is associated with the appearance of subclones that harbour specific genetic aberrations, such as amplifications of MYC, CCND1 and FGFR1 (refs 91–93). The prospective quantification of geographical and temporal heterogeneity can be achieved by multiregional sampling of DCIS in surgical specimens, and by serial sampling in cases of DCIS recurrence. The identification of biomarkers of progression that may predict the transition from pre-malignancy to malignancy would be relevant. A comparison of surveillance strategies with or without molecular assessment of such biomarkers in different geographical locations and in serially collected samples of pre-malignant lesions can be undertaken to validate their prognostic role.
Metastatic potential of localized cancer
Both the longitudinal cohort strategy and the histology-based design to evaluate multiple aberrations would be reasonable frameworks to consider for metastatic potential of localized cancer. Comprehensive molecular portraits of the four main primary breast cancer subtypes (luminal A, luminal B, basal-like and HER2-enriched) have recently been published94. Multiregional sampling and molecular profiling of primary tumour and regional lymph nodes can be carried out in patients who have undergone curative resections. In addition, depending on the sensitivity of detection, CTCs can be enumerated and profiled, and ctDNA can be extracted and analysed for the presence of somatic genomic alterations. Patients can then be monitored prospectively to determine if the detection of specific biomarkers in multiple locations within the primary surgical specimen or in the circulatory system can help to identify those tumours with biologically aggressive behaviour beyond the prognosis given by standard clinicopathologi-cal factors.
Monitoring for early micrometastases
After definitive local therapy and systemic adjuvant therapy, serial enumeration of CTCs or prospective sequential profiling of ctDNA can be performed, either as a longitudinal cohort or in a histology-based design to evaluate different molecular aberrations51. Single-cell exome sequencing to detect single nucleotide mutations is being developed95–97, such that molecular characterization using captured CTCs could eventually be possible98. These samples can be used as a tracking tool for distinct existent clones that can be assessed to monitor response to adjuvant therapy and to predict disease relapse.
Targeting oncogenic driver clones
In patients who develop macroscopic metastases from breast cancer, current systemic therapy consists mainly of hormonal therapy, cytotoxic chemotherapy and a limited number of targeted agents, such as HER2 inhibitors for HER2-positive tumours, or mTOR inhibitors in hormone-receptor-positive tumours99. At present, other than HER2-targeting, selecting treatment based on a molecular profile is not proven to be superior to standard algorithms in metastatic breast cancer. As such, the design of therapeutic clinical trials that are either histology-based or histology-agnostic to evaluate the benefit of target–drug matching compared with conventional approaches, would be considered investigational. Exploring the impact of intratumour heterogeneity in a therapeutic context adds a further layer of complexity. Even if current technologies such as minimally invasive multiregional sampling of metastases or molecular imaging are able to identify functional tumour subpopulations that are geographically distinct, the design of clinical trials to interrogate these subpopulations is challenging. For instance, if two potentially important clones, one with PIK3CA mutations and the other with FGFR1 amplification coexist, then hypothetical therapeutic possibilities can include either concurrent combination or sequential treatment with PI(3)K and FGFR1 inhibitors (ideally distinguished using carefully designed randomized trials). The accessibility to approved or experimental agents in such scenarios may be limited. Furthermore, the most optimal approach to combine or sequence two or more agents to yield sufficient biological target modulation with tolerable toxicity is often undefined and requires dose-finding studies. Finally, even if appropriate drug combination strategies are determined and can effectively suppress clonal evolution, thus ameliorating or delaying the onset of resistance, a previously undetected or new driver clone may ultimately arise. In contrast to the uncertainty of ‘drugging’ intratumour heterogeneity successfully, the use of CTCs or ctDNA as early biomarkers of treatment response of metastatic breast cancer seems to be more readily tangible52.
Emergence of resistant clones
Intratumour heterogeneity is a key factor that may lead to primary drug resistance because the extent of genomic assessment and molecular characterization determines our ability to identify potentially important subclones100. In patients who have clearly responded to treatment but in whom disease subsequently progresses, a repeat tumour biopsy to detect the expansion of pre-existent resistant subclones or the emergence of newly acquired resistant clones may be highly informative. An important caveat is that clonal population size and architecture cannot be assessed through biopsy sampling of a single metastatic site. If a change in genotype is observed when another biopsy is taken at the onset of progression after systemic treatment, this may be due to either clonal evolution or as a result of an earlier false negative due to sampling bias. To circumvent such limitations of tumour biopsies, characterization of CTC or ctDNA in plasma could be an attractive alternative if they are demonstrated to be more reflective of the global molecular status. Furthermore, these circulating ‘liquid tumours’ may also precede radiological evidence of tumour growth42,43,54. These strategies to identify and tackle primary or acquired resistance can be integrated into clinical trials using histology-based or histology-agnostic frameworks. For instance, DETECT III (NCT01619111)101 is a multicentre, histology-based, randomized phase III study that compares lapatinib (as a HER2-targeted therapy) combined with standard therapy with standard therapy alone in patients with HER2-negative breast cancer who have had HER2-positive CTCs detected in their blood. When the sample size is small, an N-of-1 trial design may be used to sequentially assess, in the same patient, the effects of different agents that may have antitumour activity against the resistant clones. It would seem logical to interrogate an emerging resistant clone as early as possible, using the combination or sequential therapeutic strategies previously described, although the timing for pharmacological counteraction of clonal evolution may also require full assessment through well-conceived clinical trials.
Future directions
The occurrence of intratumour heterogeneity and clonal evolution in cancers, resulting in malignant growth, invasion, metastasis and resistance acquisition has long been recognized. The availability of molecular profiling technologies such as next-generation sequencing coupled with advances in bioinformatics has enabled these previously elusive phenomena to be assayed in the clinical setting. The challenges ahead are immense, and include the reliable and accurate elucidation of geographical and temporal variations in patient samples and the subsequent correlation with both prognosis and treatment response. Current efforts are focused on gathering evidence to support the idea that intratumour heterogeneity substantially affects disease outcome, although the relationship is probably context dependent. Clinical trial strategies to interrogate intratumour heterogeneity are challenging, and for researchers to gain a deeper understanding into these molecular complexities would require not only the active participation of patients who are willing to undergo repeated investigations, but also the collaborative engagement of clinicians and scientists. Without a full understanding of the spectrum of a patient’s mutations, we may risk expending large resources on the development of fundamentally flawed approaches to biomarker-directed therapeutics.
The knowledge that significant intratumour heterogeneity is present in most patients has important implications for predictive biomarker development in the context of early clinical trials. First, quantitative biomarkers (for example, RNA expression) may be misleading, as they are based on the average expression across a heterogeneous tumour. Second, sequencing approaches may be misleading, unless careful attention is paid to detecting minor clones of clinical significance. Last, phenotypic and functional heterogeneity that results from events other than genomic alterations, for instance due to epigenetic alterations or plasticity, is likely to have an important effect on treatment response (see Review by Meacham and Morrison on page 328). Although we do not yet have the knowledge base to successfully individualize treatment by accounting for both interpatient and intrapatient heterogeneity, we believe that the delivery of comprehensive personalized cancer medicine will eventually be possible.
Acknowledgments
Supported in part by the Cancer Care Ontario Applied Cancer Research Units Grant (P.L.B, L.L.S) and by the US National Institute of Health Grant U01 GM61393 (M.J.R).
Footnotes
The authors declare competing financial interests: details accompany the full-text HTML version of this paper at go.nature.com/jofa4v.
Readers are welcome to comment on the online version of this article at go.nature.com/jofa4v.
References
- 1.Tran B, et al. Cancer genomics: technology, discovery, and translation. J. Clin. Oncol. 2012;30:647–660. doi: 10.1200/JCO.2011.39.2316. [DOI] [PubMed] [Google Scholar]
- 2.Mok TS, et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N. Engl. J. Med. 2009;361:947–957. doi: 10.1056/NEJMoa0810699. [DOI] [PubMed] [Google Scholar]
- 3.De Roock W, et al. KRAS wild-type state predicts survival and is associated to early radiological response in metastatic colorectal cancer treated with cetuximab. Ann. Oncol. 2008;19:508–515. doi: 10.1093/annonc/mdm496. [DOI] [PubMed] [Google Scholar]
- 4.Chapman PB, et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N. Engl. J. Med. 2011;364:2507–2516. doi: 10.1056/NEJMoa1103782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Slamon DJ, et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N. Engl. J. Med. 2001;344:783–792. doi: 10.1056/NEJM200103153441101. [DOI] [PubMed] [Google Scholar]
- 6.Karapetis CS, et al. K-ras mutations and benefit from cetuximab in advanced colorectal cancer. N. Engl. J. Med. 2008;359:1757–1765. doi: 10.1056/NEJMoa0804385. [DOI] [PubMed] [Google Scholar]
- 7.Nowell PC. The clonal evolution of tumor cell populations. Science. 1976;194:23–28. doi: 10.1126/science.959840. [DOI] [PubMed] [Google Scholar]
- 8.Amir E, et al. Prospective study evaluating the impact of tissue confirmation of metastatic disease in patients with breast cancer. J. Clin. Oncol. 2012;30:587–592. doi: 10.1200/JCO.2010.33.5232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gong Y, Han EY, Guo M, Pusztai L, Sneige N. Stability of estrogen receptor status in breast carcinoma. Cancer. 2011;117:705–713. doi: 10.1002/cncr.25506. [DOI] [PubMed] [Google Scholar]
- 10.Thompson AM, et al. Prospective comparison of switches in biomarker status between primary and recurrent breast cancer: the Breast Recurrence In Tissues Study (BRITS) Breast Cancer Res. 2010;12:R92. doi: 10.1186/bcr2771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chang HJ, et al. Discordant human epidermal growth factor receptor 2 and hormone receptor status in primary and metastatic breast cancer and response to trastuzumab. Jpn. J. Clin. Oncol. 2011;41:593–599. doi: 10.1093/jjco/hyr020. [DOI] [PubMed] [Google Scholar]
- 12.Early Breast Cancer Trialists’ Collaborative Group. Relevance of breast cancer hormone receptors and other factors to the efficacy of adjuvant tamoxifen: patient-level meta-analysis of randomised trials. Lancet. 2011;378:771–784. doi: 10.1016/S0140-6736(11)60993-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lindström LS, et al. Clinically used breast cancer markers such as estrogen receptor, progesterone receptor, and human epidermal growth factor receptor 2 are unstable throughout tumor progression. J. Clin. Oncol. 2012;30:2601–2608. doi: 10.1200/JCO.2011.37.2482. [DOI] [PubMed] [Google Scholar]
- 14.Liedtke C, et al. Prognostic impact of discordance between triple-receptor measurements in primary and recurrent breast cancer. Ann. Oncol. 2009;20:1953–1958. doi: 10.1093/annonc/mdp263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tapia C, et al. HER2 gene status in primary breast cancers and matched distant metastases. Breast Cancer Res. 2007;9:R31. doi: 10.1186/bcr1676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Artale S, et al. Mutations of KRAS and BRAF in primary and matched metastatic sites of colorectal cancer. J. Clin. Oncol. 2008;26:4217–4219. doi: 10.1200/JCO.2008.18.7286. [DOI] [PubMed] [Google Scholar]
- 17.Kalikaki A, et al. Comparison of EGFR and K-RAS gene status between primary tumours and corresponding metastases in NSCLC. Br. J. Cancer. 2008;99:923–929. doi: 10.1038/sj.bjc.6604629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kim H, et al. Discordance between anaplastic lymphoma kinase status in primary non-small-cell lung cancers and their corresponding metastases. Histopathology. 2013;62:305–314. doi: 10.1111/j.1365-2559.2012.04356.x. [DOI] [PubMed] [Google Scholar]
- 19.Weller M, et al. MGMT promoter methylation in malignant gliomas: ready for personalized medicine? Nature Rev. Neurol. 2010;6:39–51. doi: 10.1038/nrneurol.2009.197. [DOI] [PubMed] [Google Scholar]
- 20.Bozzetti C, et al. Comparison of HER2 status in primary and paired metastatic sites of gastric carcinoma. Br. J. Cancer. 2011;104:1372–1376. doi: 10.1038/bjc.2011.121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kim MA, Lee HJ, Yang HK, Bang YJ, Kim WH. Heterogeneous amplification of ERBB2 in primary lesions is responsible for the discordant ERBB2 status of primary and metastatic lesions in gastric carcinoma. Histopathology. 2011;59:822–831. doi: 10.1111/j.1365-2559.2011.04012.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Knijn N, et al. KRAS mutation analysis: a comparison between primary tumours and matched liver metastases in 305 colorectal cancer patients. Br. J. Cancer. 2011;104:1020–1026. doi: 10.1038/bjc.2011.26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yancovitz M, et al. Intra-and inter-tumor heterogeneity of BRAFV600E mutations in primary and metastatic melanoma. PLoS ONE. 2012;7:e29336. doi: 10.1371/journal.pone.0029336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Niikura N, et al. Loss of human epidermal growth factor receptor 2 (HER2) expression in metastatic sites of HER2-overexpressing primary breast tumors. J. Clin. Oncol. 2012;30:593–599. doi: 10.1200/JCO.2010.33.8889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Fidler IJ, Hart IR. Biological diversity in metastatic neoplasms: origins and implications. Science. 1982;217:998–1003. doi: 10.1126/science.7112116. [DOI] [PubMed] [Google Scholar]
- 26.Starczynski J, et al. HER2 gene amplification in breast cancer: a rogues’ gallery of challenging diagnostic cases: UKNEQAS interpretation guidelines and research recommendations. Am. J. Clin. Pathol. 2012;137:595–605. doi: 10.1309/AJCPATBZ2JFN1QQC. [DOI] [PubMed] [Google Scholar]
- 27.Yoon HH, et al. Adverse prognostic impact of intratumor heterogeneous HER2 gene amplification in patients with esophageal adenocarcinoma. J. Clin. Oncol. 2012;30:3932–3938. doi: 10.1200/JCO.2012.43.1890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Perez K, et al. Heterogeneity of colorectal cancer (CRC) in reference to KRAS proto-oncogene utilizing WAVE technology. Exp. Mol. Pathol. 2013;95:74–82. doi: 10.1016/j.yexmp.2013.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wilmott JS, et al. Intratumoral molecular heterogeneity in a BRAF-mutant, BRAF inhibitor-resistant melanoma: a case illustrating the challenges for personalized medicine. Mol. Cancer Ther. 2012;11:2704–2708. doi: 10.1158/1535-7163.MCT-12-0530. [DOI] [PubMed] [Google Scholar]
- 30.Taniguchi K, Okami J, Kodama K, Higashiyama M, Kato K. Intratumor heterogeneity of epidermal growth factor receptor mutations in lung cancer and its correlation to the response to gefitinib. Cancer Sci. 2008;99:929–935. doi: 10.1111/j.1349-7006.2008.00782.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sakurada A, Lara-Guerra H, Liu N, Shepherd FA, Tsao M-S. Tissue heterogeneity of EGFR mutation in lung adenocarcinoma. J. Thorac. Oncol. 2008;3:527–529. doi: 10.1097/JTO.0b013e318168be93. [DOI] [PubMed] [Google Scholar]
- 32.Rye I, et al. Intra-tumor heterogeneity as a predictor of therapy response in HER2 positive breast cancer. Cancer Res. 2012;72(24 suppl) P3-05-04. [Google Scholar]
- 33.Tougeron D, et al. Effect of low-frequency KRAS mutations on the response to anti-EGFR therapy in metastatic colorectal cancer. Ann. Oncol. 2013;24:1267–1273. doi: 10.1093/annonc/mds620. [DOI] [PubMed] [Google Scholar]
- 34.Bai H, et al. Detection and clinical significance of intratumoral EGFR mutational heterogeneity in Chinese patients with advanced non-small cell lung cancer. PLoS ONE. 2013;8:e54170. doi: 10.1371/journal.pone.0054170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Coons SW, Johnson PC, Shapiro JR. Cytogenetic and flow cytometry DNA analysis of regional heterogeneity in a low grade human glioma. Cancer Res. 1995;55:1569–1577. [PubMed] [Google Scholar]
- 36. Walter MJ, et al. Clonal architecture of secondary acute myeloid leukemia. N. Engl. J. Med. 2012;366:1090–1098. doi: 10.1056/NEJMoa1106968. This study reconstructs the clonal evolution of secondary acute myeloid leukaemia from myelodysplastic syndrome, demonstrating that a single population of myelodysplastic syndrome cells underwent multiple rounds of mutation and selection.
- 37.Newburger DE, et al. Genome evolution during progression to breast cancer. Genome Res. 2013;23:1097–1108. doi: 10.1101/gr.151670.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Shah SP, et al. Mutational evolution in a lobular breast tumour profiled at single nucleotide resolution. Nature. 2009;461:809–813. doi: 10.1038/nature08489. This was the first study to demonstrate how genome sequencing can be used to follow the evolution of a solid tumour from early to advanced stage
- 39.Yachida S, et al. Distant metastasis occurs late during the genetic evolution of pancreatic cancer. Nature. 2010;467:1114–1117. doi: 10.1038/nature09515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Campbell PJ, et al. The patterns and dynamics of genomic instability in metastatic pancreatic cancer. Nature. 2010;467:1109–1113. doi: 10.1038/nature09460. References 39 and 40 describe how genomic instability drives clonal evolution in metastatic pancreatic cancers
- 41.Ding L, et al. Genome remodelling in a basal-like breast cancer metastasis and xenograft. Nature. 2010;464:999–1005. doi: 10.1038/nature08989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Diaz LA, Jr, et al. The molecular evolution of acquired resistance to targeted EGFR blockade in colorectal cancers. Nature. 2012;486:537–540. doi: 10.1038/nature11219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Misale S, et al. Emergence of KRAS mutations and acquired resistance to anti-EGFR therapy in colorectal cancer. Nature. 2012;486:532–536. doi: 10.1038/nature11156. References 42 and 43 demonstrate how KRAS mutant subclones in colorectal cancer can emerge under the selection pressure of EGFR antibody therapy to confer treatment resistance.
- 44. Shah SP, et al. The clonal and mutational evolution spectrum of primary triple-negative breast cancers. Nature. 2012;486:395–399. doi: 10.1038/nature10933. This study demonstrates wide variety of clonal and mutational composition in 104 primary triple-negative breast cancers.
- 45.Aparicio S, Caldas C. The implications of clonal genome evolution for cancer medicine. N. Engl. J. Med. 2013;368:842–851. doi: 10.1056/NEJMra1204892. [DOI] [PubMed] [Google Scholar]
- 46.Buttitta F, et al. Effective assessment of egfr mutation status in bronchoalveolar lavage and pleural fluids by next-generation sequencing. Clin. Cancer Res. 2013;19:691–698. doi: 10.1158/1078-0432.CCR-12-1958. [DOI] [PubMed] [Google Scholar]
- 47.Kinde I, et al. Evaluation of DNA from the papanicolaou test to detect ovarian and endometrial cancers. Sci. Transl. Med. 2013;5 doi: 10.1126/scitranslmed.3004952. 167ra164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Maheswaran S, et al. Detection of Mutations in EGFR in Circulating Lung-Cancer Cells. N. Engl. J. Med. 2008;359:366–377. doi: 10.1056/NEJMoa0800668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Leary RJ, et al. Detection of chromosomal alterations in the circulation of cancer patients with whole-genome sequencing. Sci. Transl. Med. 2012;4 doi: 10.1126/scitranslmed.3004742. 162ra154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Chan KCA, et al. Cancer genome scanning in plasma: detection of tumor-associated copy number aberrations, single-nucleotide variants, and tumoral heterogeneity by massively parallel sequencing. Clin. Chem. 2013;59:211–224. doi: 10.1373/clinchem.2012.196014. [DOI] [PubMed] [Google Scholar]
- 51.Forshew T, et al. Noninvasive identification and monitoring of cancer mutations by targeted deep sequencing of plasma DNA. Sci. Transl. Med. 2012;4 doi: 10.1126/scitranslmed.3003726. 136ra168. [DOI] [PubMed] [Google Scholar]
- 52. Dawson S-J, et al. Analysis of circulating tumor DNA to monitor metastatic breast cancer. N. Engl. J. Med. 2013;368:1199–1209. doi: 10.1056/NEJMoa1213261. References 51 and 52 used targeted sequencing of circulating tumour DNA to monitor tumour dynamics in patients with advanced solid tumours.
- 53.Cristofanilli M, et al. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N. Engl. J. Med. 2004;351:781–791. doi: 10.1056/NEJMoa040766. [DOI] [PubMed] [Google Scholar]
- 54. Murtaza M, et al. Non-invasive analysis of acquired resistance to cancer therapy by sequencing of plasma DNA. Nature. 2013;497:108–112. doi: 10.1038/nature12065. This study shows that exome sequencing of circulating tumour DNA can be used to track genomic evolution of metastatic cancers in response to therapy.
- 55.Lipson D, et al. Identification of new ALK and RET gene fusions from colorectal and lung cancer biopsies. Nature Med. 2012;18:382–384. doi: 10.1038/nm.2673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Beltran H, et al. Targeted next-generation sequencing of advanced prostate cancer identifies potential therapeutic targets and disease heterogeneity. Eur. Urol. 2013;63:920–926. doi: 10.1016/j.eururo.2012.08.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Swanton C. Intratumor heterogeneity: evolution through space and time. Cancer Res. 2012;72:4875–4882. doi: 10.1158/0008-5472.CAN-12-2217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.CLIA. Medicare, Medicaid and CLIA programs: regulations implementing the clinical laboratory improvement amendments of 1988. Fed. Regist. 1992;57:7002–7186. [PubMed] [Google Scholar]
- 59.Wagle N, et al. High-throughput detection of actionable genomic alterations in clinical tumor samples by targeted, massively parallel sequencing. Cancer Discov. 2012;2:82–93. doi: 10.1158/2159-8290.CD-11-0184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Bedard P, et al. Princess Margaret Cancer Centre (PMCC) Integrated Molecular Profiling in Advanced Cancers Trial (IMPACT) using genotyping and targeted next-generation sequencing (NGS) J. Clin. Oncol. 2013;31:11002. [Google Scholar]
- 61. Miller V, et al. Use of next-generation sequencing (NGS) to identify actionable genomic alterations (GA) in diverse solid tumor types: The Foundation Medicine (FMI) experience with 2,200+ clinical samples. J. Clin. Oncol. 2013;31:11020. References 59–61 show that next-generation sequencing technology can be applied to stored formalin-fixed, paraffin-embedded tumour samples in a clinical setting.
- 62.Gargis AS, et al. Assuring the quality of next-generation sequencing in clinical laboratory practice. Nature Biotechnol. 2012;30:1033–1036. doi: 10.1038/nbt.2403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Tran B, et al. Feasibility of real time next generation sequencing of cancer genes linked to drug response: results from a clinical trial. Int. J. Cancer. 2013;132:1547–1555. doi: 10.1002/ijc.27817. [DOI] [PubMed] [Google Scholar]
- 64.Dancey JE, Bedard PL, Onetto N, Hudson TJ. The genetic basis for cancer treatment decisions. Cell. 2012;148:409–420. doi: 10.1016/j.cell.2012.01.014. [DOI] [PubMed] [Google Scholar]
- 65.Chapman PB, et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N. Engl. J. Med. 2011;364:2507–2516. doi: 10.1056/NEJMoa1103782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Kopetz S, et al. PLX4032 in metastatic colorectal cancer patients with mutant BRAF tumors. J. Clin. Oncol. 2010;28:3534. [Google Scholar]
- 67.Barretina J, et al. The cancer cell line encyclopedia enables predictive modelling of anticancer drug sensitivity. Nature. 2012;483:603–607. doi: 10.1038/nature11003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Garnett MJ, et al. Systematic identification of genomic markers of drug sensitivity in cancer cells. Nature. 2012;483:570–575. doi: 10.1038/nature11005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Papillon-Cavanagh S, et al. Comparison and validation of genomic predictors for anticancer drug sensitivity. J. Am. Med. Inform. Assoc. 2013;20:597–602. doi: 10.1136/amiajnl-2012-001442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Begley CG, Ellis LM. Drug development: raise standards for preclinical cancer research. Nature. 2012;483:531–533. doi: 10.1038/483531a. [DOI] [PubMed] [Google Scholar]
- 71.Sleijfer S, Bogaerts J, Siu LL. Designing transformative clinical trials in the cancer genome era. J. Clin. Oncol. 2013;31:1834–1841. doi: 10.1200/JCO.2012.45.3639. [DOI] [PubMed] [Google Scholar]
- 72.Dienstmann R, et al. Molecular profiling of patients with colorectal cancer and matched targeted therapy in phase I clinical trials. Mol. Cancer Ther. 2012;11:2062–2071. doi: 10.1158/1535-7163.MCT-12-0290. [DOI] [PubMed] [Google Scholar]
- 73. Tsimberidou A-M, et al. Personalized medicine in a phase I clinical trials program: the MD Anderson Cancer Center Initiative. Clin. Cancer Res. 2012;18:6373–6383. doi: 10.1158/1078-0432.CCR-12-1627. This study demonstrates that matching investigational treatment to genotype results in better response and survival
- 74.Rodón J, et al. Molecular prescreening to select patient population in early clinical trials. Nature Rev. Clin. Oncol. 2012;9:359–366. doi: 10.1038/nrclinonc.2012.48. [DOI] [PubMed] [Google Scholar]
- 75.Hollebecque A, et al. Molecular screening for cancer treatment optimization (MOSCATO 01): a prospective molecular triage trial — interim results. J. Clin. Oncol. 2013;31:2512. [Google Scholar]
- 76.Kaplan R, Brown L. MRC FOCUS 4: a New Trial Biomarker-Stratified Phase 2/3 Trial Design. Medical Research Council; 2013. http://www.methodologyhubs.mrc.ac.uk/annual_htmr_network_meetings/2013_network_meeting.aspx. [Google Scholar]
- 77.Kim ES, et al. The BATTLE Trial: personalizing therapy for lung cancer. Cancer Discov. 2011;1:44–53. doi: 10.1158/2159-8274.CD-10-0010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Barker AD, et al. I-SPY 2: an adaptive breast cancer trial design in the setting of neoadjuvant chemotherapy. Clin. Pharmacol. Ther. 2009;86:97–100. doi: 10.1038/clpt.2009.68. [DOI] [PubMed] [Google Scholar]
- 79.Berry DA, Herbst RS, Rubin EH. Reports from the 2010 Clinical and Translational Cancer Research Think Tank Meeting: design strategies for personalized therapy trials. Clin. Cancer Res. 2012;18:638–644. doi: 10.1158/1078-0432.CCR-11-2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Lillie EO, et al. The n-of-1 clinical trial: the ultimate strategy for individualizing medicine? Per. Med. 2011;8:161–173. doi: 10.2217/pme.11.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Gabler NB, Duan N, Vohra S, Kravitz RL. N-of-1 trials in the medical literature: a systematic review. Med. Care. 2011;49:761–768. doi: 10.1097/MLR.0b013e318215d90d. [DOI] [PubMed] [Google Scholar]
- 82.Mick R, Crowley JJ, Carroll RJ. Phase II clinical trial design for noncytotoxic anticancer agents for which time to disease progression is the primary endpoint. Control. Clin. Trials. 2000;21:343–359. doi: 10.1016/s0197-2456(00)00058-1. [DOI] [PubMed] [Google Scholar]
- 83. Kurzrock R, Tabernero J. WINTHER — WIN Therapeutics Clinical Trial. WIN Consortium; 2012. http://www.winconsortium.org. This describes an ongoing prospective clinical trial that selects targeted treatment for patients with advanced solid tumours based upon DNA, RNA and microRNA profiling.
- 84.Wu X, et al. Clonal selection drives genetic divergence of metastatic medulloblastoma. Nature. 2012;482:529–533. doi: 10.1038/nature10825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Tao Y, et al. Rapid growth of a hepatocellular carcinoma and the driving mutations revealed by cell-population genetic analysis of whole-genome data. Proc. Natl Acad. Sci. USA. 2011;108:12042–12047. doi: 10.1073/pnas.1108715108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Albiges L, Gerlinger M& PREDICT consortium. The PREDICT (Personalised RNA Interference to Enhance the Delivery of Individualised Cytotoxic and Targeted Therapeutics) approach to biomarker discovery in renal cell carcinoma. Cancer Res. 2012;72:S1746. [Google Scholar]
- 87. Gerlinger M, et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N. Engl. J. Med. 2012;366:883–892. doi: 10.1056/NEJMoa1113205. This study demonstrates mutational intratumour heterogeneity within spatially separated regions of the same tumour and matched primary and metastatic lesions separated by time.
- 88.Tolmachev V, Stone-Elander S, Orlova A. Radiolabelled receptor-tyrosine-kinase targeting drugs for patient stratification and monitoring of therapy response: prospects and pitfalls. Lancet Oncol. 2010;11:992–1000. doi: 10.1016/S1470-2045(10)70088-7. [DOI] [PubMed] [Google Scholar]
- 89.Gasch C, et al. Heterogeneity of epidermal growth factor receptor status and mutations of KRAS/PIK3CA in circulating tumor cells of patients with colorectal cancer. Clin. Chem. 2013;59:252–260. doi: 10.1373/clinchem.2012.188557. [DOI] [PubMed] [Google Scholar]
- 90.Schwarzenbach H, Hoon DS, Pantel K. Cell-free nucleic acids as biomarkers in cancer patients. Nature Rev. Cancer. 2011;11:426–437. doi: 10.1038/nrc3066. [DOI] [PubMed] [Google Scholar]
- 91.Jang MH, et al. FGFR1 is amplified during the progression of in situ to invasive breast carcinoma. Breast Cancer Res. 2012;14:R115. doi: 10.1186/bcr3239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Johnson CE, et al. Identification of copy number alterations associated with the progression of DCIS to invasive ductal carcinoma. Breast Cancer Res. Treat. 2012;133:889–898. doi: 10.1007/s10549-011-1835-1. [DOI] [PubMed] [Google Scholar]
- 93.Heselmeyer-Haddad K, et al. Single-cell genetic analysis of ductal carcinoma in situ and invasive breast cancer reveals enormous tumor heterogeneity yet conserved genomic imbalances and gain of MYC during progression. Am. J. Pathol. 2012;181:1807–1822. doi: 10.1016/j.ajpath.2012.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.The Cancer Genome Atlas Network. Comprehensive molecular portraits of human breast tumours. Nature. 2012;490:61–70. doi: 10.1038/nature11412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Xu X, et al. Single-cell exome sequencing reveals single-nucleotide mutation characteristics of a kidney tumor. Cell. 2012;148:886–895. doi: 10.1016/j.cell.2012.02.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Hou Y, et al. Single-cell exome sequencing and monoclonal evolution of a JAK2-negative myeloproliferative neoplasm. Cell. 2012;148:873–885. doi: 10.1016/j.cell.2012.02.028. [DOI] [PubMed] [Google Scholar]
- 97.Navin N, et al. Tumour evolution inferred by single-cell sequencing. Nature. 2011;472:90–94. doi: 10.1038/nature09807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Xu X, et al. Mutational analysis of circulating tumor cells in breast cancer patients by targeted clonal sequencing. J. Clin. Oncol. 2012;30:10516. [Google Scholar]
- 99.Baselga J, et al. Everolimus in postmenopausal hormone-receptor–positive advanced breast cancer. N. Engl. J. Med. 2012;366:520–529. doi: 10.1056/NEJMoa1109653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Turner NC, Reis-Filho JS. Genetic heterogeneity and cancer drug resistance. Lancet Oncol. 2012;13:e178–e185. doi: 10.1016/S1470-2045(11)70335-7. [DOI] [PubMed] [Google Scholar]
- 101.Melcher C, et al. DETECT III — a multicenter, randomized, phase III study to compare standard therapy alone versus standard therapy plus lapatinib in patients with initially HER2-negative metastatic breast cancer but with HER2-positive circulating tumor cells. Cancer Res. 2012;72 SOT1-1-10. [Google Scholar]
- 102.Elisei R, et al. Prognostic significance of somatic RET oncogene mutations in sporadic medullary thyroid cancer: a 10-year follow-up study. J. Clin. Endocrinol. Metab. 2008;93:682–687. doi: 10.1210/jc.2007-1714. [DOI] [PubMed] [Google Scholar]
- 103.Park S, et al. Discordance of molecular biomarkers associated with epidermal growth factor receptor pathway between primary tumors and lymph node metastasis in non-small cell lung cancer. J. Thorac. Oncol. 2009;4:809–815. doi: 10.1097/JTO.0b013e3181a94af4. [DOI] [PubMed] [Google Scholar]
- 104.Schmid K, et al. EGFR/KRAS/BRAF mutations in primary lung adenocarcinomas and corresponding locoregional lymph node metastases. Clin. Cancer Res. 2009;15:4554–4560. doi: 10.1158/1078-0432.CCR-09-0089. [DOI] [PubMed] [Google Scholar]
- 105.Kim H, et al. Detection of ALK gene rearrangement in non-small cell lung cancer: a comparison of fluorescence in situ hybridization and chromogenic in situ hybridization with correlation of ALK protein expression. J. Thorac. Oncol. 2011;6:1359–1366. doi: 10.1097/JTO.0b013e31821cfc73. [DOI] [PubMed] [Google Scholar]
- 106.Janjigian YY, et al. Prognosis of metastatic gastric and gastroesophageal junction cancer by HER2 status: a European and USA International collaborative analysis. Ann. Oncol. 2012;23:2656–2662. doi: 10.1093/annonc/mds104. [DOI] [PubMed] [Google Scholar]
- 107.Bang Y-J, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet. 2010;376:687–697. doi: 10.1016/S0140-6736(10)61121-X. [DOI] [PubMed] [Google Scholar]
- 108.Vakiani E, et al. Comparative genomic analysis of primary versus metastatic colorectal carcinomas. J. Clin. Oncol. 2012;30:2956–2962. doi: 10.1200/JCO.2011.38.2994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Colombino M, et al. BRAF/NRAS mutation frequencies among primary tumors and metastases in patients with melanoma. J. Clin. Oncol. 2012;30:2522–2529. doi: 10.1200/JCO.2011.41.2452. [DOI] [PubMed] [Google Scholar]
- 110.Antonescu CR, et al. Acquired resistance to imatinib in gastrointestinal stromal tumor occurs through secondary gene mutation. Clin. Cancer Res. 2005;11:4182–4190. doi: 10.1158/1078-0432.CCR-04-2245. [DOI] [PubMed] [Google Scholar]
- 111.Heinrich MC, et al. Molecular correlates of imatinib resistance in gastrointestinal stromal tumors. J. Clin. Oncol. 2006;24:4764–4774. doi: 10.1200/JCO.2006.06.2265. [DOI] [PubMed] [Google Scholar]
- 112.Walker I, et al. Utilizing a collaborative working model to optimize molecular analysis of solid tumors in the Cancer Research UK’s stratified medicine programme. J. Clin. Oncol. 2013;31:11094. [Google Scholar]
- 113.Rollins B, et al. PROFILE: Broadly based genomic testing for all patients at a major cancer center. J. Clin. Oncol. 2013;31:1531. [Google Scholar]
- 114.Sequist LV, et al. Implementing multiplexed genotyping of non-small-cell lung cancers into routine clinical practice. Ann. Oncol. 2011;22:2616–2624. doi: 10.1093/annonc/mdr489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.MD Anderson Cancer Center. The Sheik Khalifa Bin Zayed Al Nayan Institute for Personalized Cancer Therapy. MD Anderson Cancer Center; 2012. http://www.mdanderson.org/education-and-research/research-at-md-anderson/personalized-advanced-therapy/institute-for-personalized-cancer-therapy/index.html. [Google Scholar]
- 116.Mendelson J. Personalizing oncology: perspectives and prospects. J. Clin. Oncol. 2013;31:1904–1911. doi: 10.1200/JCO.2012.45.3605. [DOI] [PubMed] [Google Scholar]
- 117.Voest E. Delivering Stratified Medicine: a Strategic Overview. EACR; 2012. http://www.eacr.org/mpathcourse2012/speaker_presentations.php. [Google Scholar]
- 118.Callaway E. Norway to bring cancer gene tests to the clinic. Nature. 2012 Feb 2; http://dx.doi.org/10.1038/nature.2012.9949.
- 119.Vall d’Hebron Institute of Oncology. Cancer Genomics Group. Vall d’Hebron Institute of Oncology; 2010. http://www.vhio.net/research/core-technologies/cancer-genomics-group/en_index.php. [Google Scholar]
- 120.Vanderbilt-Ingram Cancer Center. Personalized Cancer Therapy at Vanderbilt-Ingram Cancer Center. Vanderbilt-Ingram Cancer Center; 2011. http://www.vicc.org/personalized. [Google Scholar]
- 121.Garraway LA. Genomics-Driven Clinical Trials in Oncology: principles and practice. Institute of Medicine; 2013. http://www.iom.edu/activities/disease/NCPF/2013-Feb-11.aspx. [Google Scholar]
- 122.Getz M, Boughey J. Breast Cancer Genome Guided Therapy Study (BEAUTY) Mayo Clinic; 2012. http://mayoresearch.mayo.edu/center-for-individualized-medicine/breast-cancer-study.asp. [Google Scholar]
- 123.Kummar S. Challenges in Incorporating Integral NGS into Early Clinical Trials. National Cancer Institute; 2012. http://cdp.cancer.gov/next-generation_agenda.htm. [Google Scholar]
- 124.Williams P. Request for Information (RFI): Efficient Implementation of a Sequencing Network for the Proposed NCI-MATCH Clinical Trial. Fort Detrick Business Development Office; 2013. http://www.fdbdo.com/rfi13-151/ [Google Scholar]