Abstract
The IL-17-family cytokines IL-17A and IL-17C drive the pathogenesis of psoriatic skin inflammation, and anti-IL-17A Abs were recently approved to treat human psoriasis. To date, little is known about mechanisms that restrain IL-17 cytokine-mediated signaling, particularly IL-17C. Here, we show that the endoribonuclease MCPIP1 (also known as Regnase-1) is markedly upregulated in human psoriatic skin lesions. Similarly, MCPIP1 was overexpressed in the imiquimod (IMQ)-driven mouse model of cutaneous inflammation. Mice with an MCPIP1 deficiency (Zc3h12a+/−) displayed no baseline skin inflammation, but they showed exacerbated pathology following IMQ treatment. Pathology in Zc3h12a+/− mice was associated with elevated expression of IL-17A- and IL-17C-dependent genes and also increased accumulation of neutrophils in skin. However, IL-17A and IL-17C expression was unaltered, suggesting that the increased inflammation in Zc3h12a+/− mice was due to enhanced downstream IL-17R signaling. Radiation chimeras demonstrated that MCPIP1 in non-hematopoietic cells is responsible for controlling skin pathology. Moreover, Zc3h12a+/−Il17ra−/− mice given IMQ showed almost no disease. To identify which IL-17RA ligand was essential, Zc3h12a+/−Il17a−/− and Zc3h12a+/−Il17c−/− mice were given IMQ; these mice had reduced but not fully abrogated pathology, indicating that MCPIP1 inhibits both IL-17A and IL-17C signaling. Confirming this hypothesis, Zc3h12a−/− keratinocytes showed increased responsiveness to IL-17A and IL-17C stimulation. Thus, MCPIP1 is a potent negative regulator of psoriatic skin inflammation through IL-17A and IL-17C. Moreover, MCPIP1 is the first described negative regulator of IL-17C signaling.
Introduction
In the past decade, IL-17 family members have emerged as drivers of inflammatory and autoimmune conditions, particularly psoriasis (1). Indeed, the IL-17A-targeting antibodies are approved for treatment of plaque psoriasis, psoriatic arthritis and ankylosing spondylitis, underscoring the importance of IL-17A in these conditions (2, 3). IL-17A can be produced by a variety of immune cells, including Th17 cells, γδ T cells, iNKT cells, ‘natural’ Th17 cells, and ILC3s (4). However, most studies indicate that IL-17 exerts its actions mainly on non-hematopoietic cells (5–9). IL-17A induces expression of a typical gene signature profile in target cells, characterized by proinflammatory cytokines, chemokines and antimicrobial peptides (1). Together, these factors provide important host defense against extracellular microbes, but when dysregulated can promote inflammation in autoimmunity.
In addition to IL-17A, the less studied cytokine IL-17C is implicated in psoriasis. IL-17C is predominantly produced by epithelial cells, and induces a similar set of downstream genes with inflammatory, antibacterial and anti-apoptotic functions. IL-17C appears to act in both hematopoietic and non-hematopoietic cells, including intestinal epithelial cells, keratinocytes and Th17 cells (10–13). IL-17C protein concentrations are ~125-fold higher than IL-17A levels in psoriatic lesions, making it the most abundant IL-17 family member in human psoriasis (14). Additionally, keratinocyte-specific overexpression of IL-17C in mice causes a spontaneous psoriasis-like phenotype (14).
IL-17 family members signal through multimeric receptors composed of a common chain, IL-17RA, and a second chain that varies by ligand. IL-17A signals through IL-17RA and IL-17RC, and IL-17C has been shown to act via an IL-17RA/IL-17RE receptor complex. Both cytokines use the adaptor and E3 ubiquitin ligase Act1 (also known as CIKS) to drive signaling, but to date little else is known about the positive activators of IL-17C downstream signaling (15–17).
Given the prominence of IL-17A in inflammation, numerous mechanisms have evolved to negatively regulate its signaling, which are needed to limit collateral tissue damage during inflammatory processes. IL-17A is a key driver of psoriasis, but little is known about the factors that normally restrain IL-17A-dependent signal transduction in the skin. The endoribonuclease and deubiquitinase MCPIP1 (MCP1-induced protein 1, also known as Regnase-1 and encoded by the ZC3H12A gene) is a vital regulator of inflammation. Its expression is induced by proinflammatory stimuli, including MCP-1, TLR ligands, IL-1β and IL-17A (18–22). MCPIP1 regulates TLR signaling through cleavage of target gene mRNAs, including Il6 (23–25) or deubiquitination of inflammatory mediators (26). In addition, MCPIP1 constitutively restricts TCR signaling, and in T cells MCPIP1 is inducibly degraded following T cell activation. MCPIP1 deficiency in CD4+ cells has been shown to enhance Th17 effector function (27, 28). We have shown that MCPIP1 negatively regulates IL-17-dependent inflammation through the degradation of IL-17A-induced target gene transcripts and IL-17RA mRNA (19). Here, we examined the impact of MCPIP1 on IL-17A and IL-17C signaling in a mouse model of acute psoriatic-like skin inflammation and in human psoriasis clinical samples. Our data identify MCPIP1 as the first recognized inhibitor of IL-17C signaling, and establish this protein as a common regulator of IL-17 family members during skin inflammation.
Methods
Patients
Nine healthy non-psoriatic controls and ten patients with chronic plaque psoriasis were enrolled (PP, PN and NN). Patients were off systemic treatment for at least 4 weeks and off all topical treatments 2 weeks prior to enrollment. Two 6 mm punch biopsies from uninvolved and two biopsies from lesional skin, or two biopsies from normal skin were obtained under local anesthesia. One biopsy was fixed in 4% formaldehyde for immunohistochemistry while the other was snap frozen in liquid nitrogen and stored at −80°C until processing. Informed consent was obtained from all subjects under a protocol approved by the Institutional Review Board of the University of Michigan Medical School (HUM00087890). This study was conducted according to the Declaration of Helsinki Principles.
Mice
C57BL/6 and CD45.1 were from The Jackson Laboratory (Bar Harbor, ME). Zc3h12a−/− mice were provided by P. Kolattukudy (U Central Florida) (29), Il17ra−/− mice were from Amgen (Seattle, WA), Il17a−/− mice were from Y. Iwakura (U of Tokyo, Japan) and Il17c−/− were from Genentech via the MMMRC (Davis, CA). Experimental mice were age-matched, female, between 6–10 weeks and used in accordance with approved University of Pittsburgh IACUC protocols and the NIH Guide for the Care and Use of Laboratory Animals guidelines.
IMQ-driven dermatitis
Dorsal skin was shaved with an electric razor and Nair epilation. Following a 48-hour rest, 6.25 mg of 5% imiquimod (Aldara, 3M) was applied daily to dorsal and ear skin. Contralateral ears or control groups were treated with a control cream (Vanicream, Pharmaceutical Specialties Inc, Rochester MN). Ear thickness was measured by caliper (Mitutoyo). Gross pathology was recorded daily by investigators blinded to the experimental cohorts. Ten 4 mm punch biopsies from random sections of skin were taken after euthanasia, 5 of which were used for flow cytometry and 5 were snap frozen for RNA extraction.
Adoptive Transfers
To generate BM chimeras, mice were irradiated with two 500 Rad doses delivered 4 h apart. 1×107 BM cells mice were delivered i.v. 16 h post-irradiation. Mice were given sulfamethoxazole (960 ug/mL) and trimethoprim (192 ug/mL) in drinking water for 2 weeks to prevent infections. Chimeras were allowed to engraft for 45 d before experimentation.
RNA isolation and qPCR
Human biopsies were homogenized and RNA isolated with RNeasy Plus Mini kit (Qiagen). For qPCR, RNA was reverse transcribed using a High Capacity cDNA Transcription kit (Applied Biosystems). Quantitative qPCR was performed using the 7900HT Fast Real-Time PCR system (Applied Biosystems) with Taqman primers (Applied Biosystems). Expression was normalized to RPLP0. Mouse skin was homogenized in Qiazol (Qiagen), in GentleMACS M tubes (Miltenyi) and total RNA isolated using RNeasy Lipid Mini Kits (Qiagen). Cells were lysed in RLT buffer and RNA extraction was performed using RNeasy Mini Kit (Qiagen). cDNA was prepared using Superscript III First Strand kit (Invitrogen). qPCR was performed with QuantiTect Primer Assays (Qiagen) and SYBR Green Fast Mix ROX (Quanta Biosciences, Gaithersburg, MD). Expression was normalized to Gapdh for mouse skin and Rplp0 for keratinocytes. Samples were analyzed on a 7300 Real Time instrument (Applied Biosystems, CA). Ct values were obtained for target and housekeeping genes and ΔCt values calculated. Expression was normalized to Gapdh for mouse skin and Rplp0 for keratinocytes. Gene induction was then transformed to a linear scale via calculation of 2− ΔCt (relative expression).
Histology and immunohistochemistry
IHC was performed on 5 μm paraffin sections from human skin. Paraffin-embedded sections were heated at 60°C for 30 min, deparaffinized, and rehydrated. Slides were placed in PH6 antigen retrieval buffer and heated at 95°C for 20 min. After cooling, slides were treated with 3% H2O2 (5 min) and blocked with 10% goat serum (30 min). Slides were incubated for 2 h at room temperature with MCPIP1 Abs (Protein Tech Group, Inc. cat. no. 25009-1-AP, 4.8μg/ml, 1:100 dilution). Slides were washed, treated with 2° Ab and peroxidase (30 min) and diaminobenzidine substrate. Mouse skin samples were fixed in 10% formalin, paraffin embedding and H&E staining was performed at the University of Buffalo histology core (Buffalo, NY).
Flow cytometry
Skin biopsies were collected in media + GolgiStop. Samples were lysed in GentleMACS C tubes (Miltenyi) with 1.5 mL digestion mix (Liberase TM, DNAse I and GolgiStop) and incubated at 37°C for 80 min. Homogenates were filtered through 70 μm strainers. Single cell suspensions were Fc-blocked and surface stained with Abs against CD45 (clone 30-F11), Ly6G (1A8), CD11b (M1/70), CD3 (145-2C11) and γδ TCR (GL3). Cells were permeabilized with Cytofix/Cytoperm (BD) and stained with anti-IL-17A Abs (TC11-18H10) or isotype control. Cells were acquired with a BD Fortessa and analyzed by FlowJo (TreeStar).
Primary mouse keratinocytes
KC preparation was performed as described (30, 31). Briefly, skin was excised from postnatal day 2 skin, stretched in a 6 cm culture dish and incubated for 2 h at 4°C. Skins were floated on 0.25% trypsin for 16 h at 4°C. Dermis was separated from epidermis and discarded, and undifferentiated cells were retrieved by vortexing in EMEM with 8% FBS +1.4 mM Ca2+. 4×105 cells per well were plated in 12-well dishes in EMEM with 8% FBS + 0.3 mM Ca2+. After 16 h, media was replaced by EMEM with 8% FBS and 0.05 mM Ca2+ to promote differentiation. KCs were rested for 3 d and stimulated for 16 h with mouse IL-17A (200 ng/mL), IL-17C (200 ng/mL) or TNF-α (2 ng/mL). Supernatants were collected for ELISA and cells were scraped, pelleted and snap frozen for RNA analysis.
Statistics
Data are presented as mean ± SEM. Statistical analysis was performed by ANOVA followed by Bonferroni’s post-hoc test or Mann-Whitney U test, using GraphPad Prism (La Jolla, CA). All experiments were performed a minimum of twice. For qPCR data, statistical analyses were run on ΔCt values. P values <0.05 were considered significant.
Results
MCPIP1 is expressed in psoriasis and limits disease severity in a mouse psoriasis model
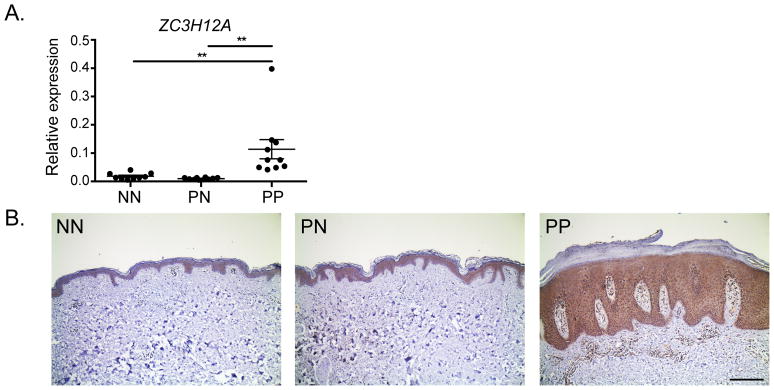
To ascertain whether MCPIP1 is linked with psoriasis, we assessed ZC3H12A expression in lesional (PP) and non-lesional (PN) skin from psoriasis patients or healthy controls (NN). ZC3H12A mRNA expression was elevated ~10-fold in psoriasis lesions compared to non-lesional skin from the same patient or to skin from healthy controls (Fig 1A). Immunohistochemical (IHC) staining of skin samples indicated that MCPIP1 was expressed primarily in the epidermis, but was also detectable in the inflammatory infiltrate (Fig 1B), consistent with the known expression pattern of this protein (32).
Figure 1. MCPIP1 expression is elevated in human psoriasis.
(A) ZC3H12A expression in normal healthy skin (NN), or non-lesional (PN) and paired lesional (PP) skin samples from psoriasis patients (n=9–10) was determined by qPCR. Expression was normalized to RPLP0. (B) MCPIP1 expression was detected by immunohistochemistry in formalin-fixed, paraffin embedded (FFPE) sections from normal healthy skin (NN), uninvolved (PN) and involved (PP) psoriasis skin **p<0.01, Mann-Whitney test. Scale bar = 100 μm.
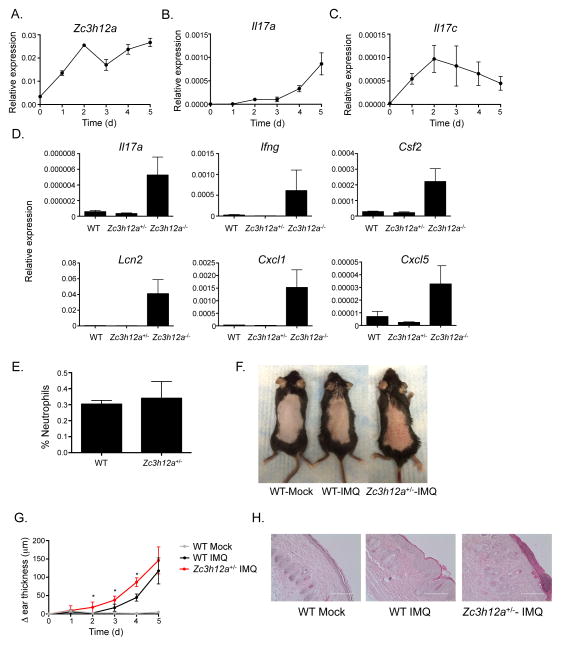
In mice, topical application of imiquimod (IMQ) leads to IL-17A- and IL-23-dependent development of skin lesions with the hallmarks of human psoriasis (33, 34). This system is considered a good model of the early events in psoriatic plaque formation (35). Consistent with the clinical samples, Zc3h12a mRNA expression was induced throughout the course of IMQ-driven psoriasiform dermatitis (Fig 2A). Expression was detectable as early as 1 day following IMQ-treatment. As expected, Il17a and Il17c were also upregulated during this time frame (Fig 2B–C). Il17c expression increased rapidly, peaking by day 2 and remaining elevated. Il17a mRNA was induced somewhat later, with progressively increased levels seen over 5 d.
Figure 2. MCPIP1 expression is elevated in IMQ-driven dermatitis and limits disease severity.
C57BL/6 (WT) mice (n=3/date) were treated topically on dorsal skin with IMQ days 0–4. Expression of (A) Zc3h12a, (B) Il17a and (C) Il17c in skin was determined by qPCR. (D) Expression of the indicated genes was determined by qPCR in untreated Zc3h12a+/+ (WT), Zc3h12a+/− and Zc3h12a−/− skin. (E) Cutaneous neutrophil infiltration was analyzed in skin by flow cytometry and % neutrophils in CD45+ gate is shown. (F) WT or Zc3h12a+/− mice (n=3–5 per day) were treated topically with mock cream or IMQ on days 0–4. Gross skin pathology for a representative animal at day 3 is shown. (G) Change in ear thickness between treated and mock-treated ears was assessed daily in the indicated mice. (H) Representative images of H&E-stained FFPE mouse skin sections on day 3 (x200). Scale bar, 200μm. *p<0.05, Mann-Whitney test.
MCPIP1 negatively regulates several inflammatory stimuli, but its role in psoriasis is poorly defined (19, 36). Zc3h12a−/− mice cannot be used for experimentation as they exhibit severely shortened lifespans as a consequence of unrestricted TLR signaling and widespread inflammation (29, 32). To circumvent these confounding issues, we assessed disease in haploinsufficient mice (Zc3h12a+/−) (19). Importantly, Zc3h12a+/−, unlike Zc3h12a−/− mice, did not present with exacerbated baseline inflammatory levels in skin, as determined by expression of proinflammatory cytokines, chemokines and antimicrobial proteins (Il17a, Ifng, Csf2, Lcn2, Cxcl1, Cxcl5) (Fig 2D). Three was also no difference in cuteaneous neutrophil infiltration in Zc3h12a+/− mice versus Zc3h12a+/+ littermates (hereafter termed “WT”) (Fig 2E). However, after 3 d of IMQ treatment, Zc3h12a+/− mice showed increased disease severity compared to WT, with enhanced erythema, epidermal thickening, skin scaling and ear swelling (Fig 2F, G). H&E staining of dorsal skin from IMQ-treated Zc3h12a+/− mice revealed increased neutrophil microclusters and parakeratosis compared to WT (Fig 2H). Therefore, MCPIP1 restricts inflammation in IMQ-driven dermatitis.
MCPIP1 limits IL-17A and IL-17C-mediated skin inflammation
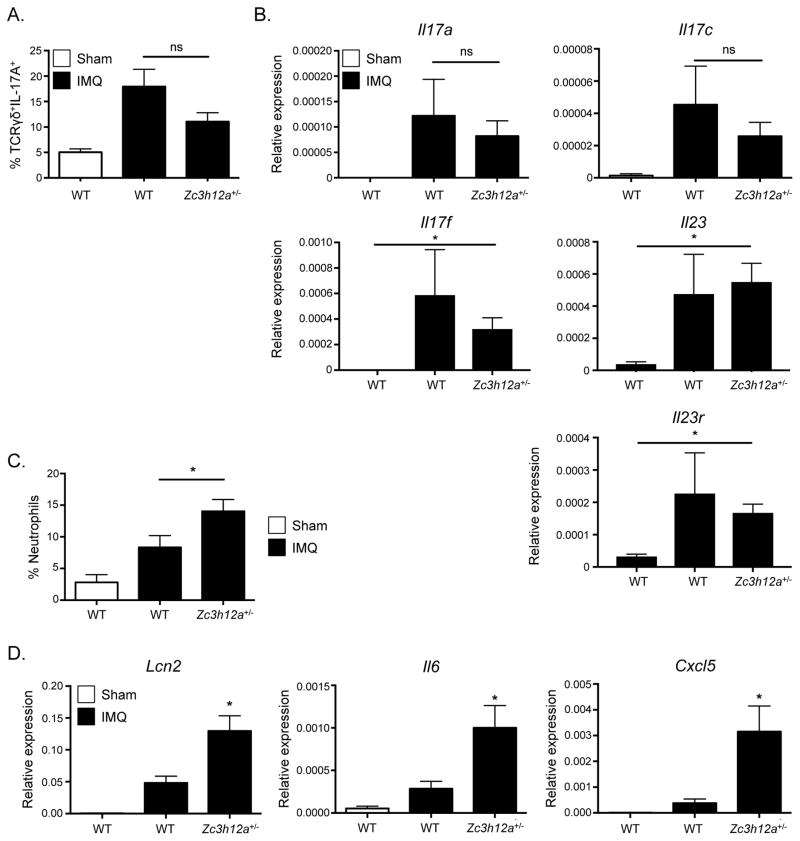
In T cells, MCPIP1-deficiency is associated with increased TCR signaling and amplified Th17 differentiation (28). Thus, an explanation for the increased skin inflammation in Zc3h12a+/− mice could be increased IL-17A production in lymphocytes following IMQ treatment. Vγ4+ γδ-T cells are a major source of IL-17A in IMQ-induced inflammation (37, 38). However, γδ-T-cell-produced IL-17A levels did not differ between IMQ-treated Zc3h12a+/− and WT (Fig 3A). Similarly, Il17a, Il17f and Il17c mRNA levels were not statistically different in IMQ-treated Zc3h12a+/− mice compared to controls, nor were Il23 and its receptor Il23r (Fig 3B).
Figure 3. MCPIP1-deficient mice exhibit increased neutrophil infiltration and expression of IL-17 gene targets upon IMQ treatment.
The indicated mice (n=5–8) were treated topically with IMQ daily. (A) IL-17 production in dermal γδ T cells was determined by flow cytometry. % of IL-17A+ cells within the CD45+γδ-TCRint lymphocyte gate is shown. (B) Expression of the indicated genes in dorsal skin was determined by qPCR. (C) Neutrophil infiltration in skin was evaluated by flow cytometry and % neutrophils in CD45+ gate is shown. (D) Expression of the indicated genes was determined by qPCR. *p<0.05, Mann-Whitney test.
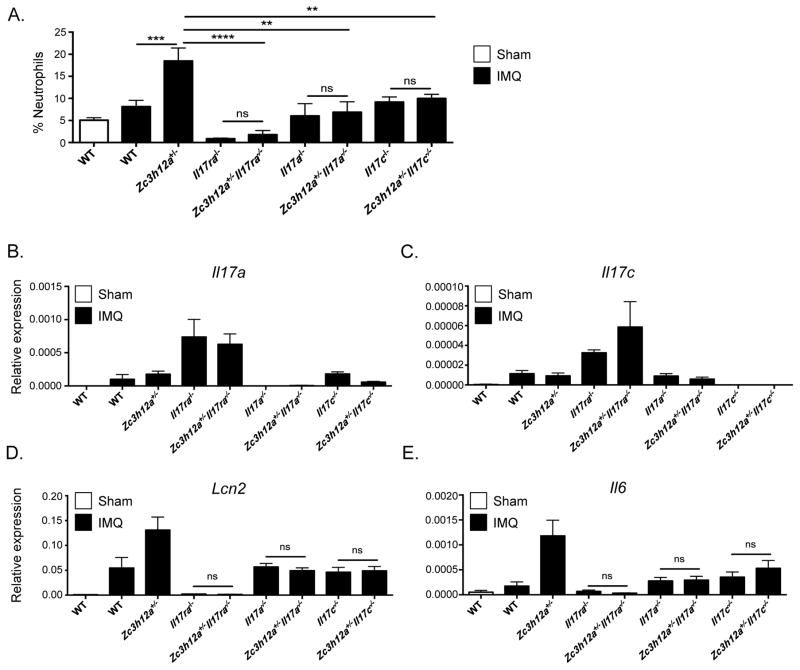
Since the increased IMQ-induced skin inflammation in Zc3h12a+/− mice did not appear to be due to increased IL-17A or IL-17C expression, we hypothesized it might instead be through increased IL-17R signaling. A hallmark of psoriasis is IL-17A-dependent neutrophil infiltration (33). Commensurate with their increased inflammation, Zc3h12a+/− mice showed increased neutrophil frequency, accompanied by increased expression of the neutrophil-attractive chemokine Cxcl5 and IL-17A-associated genes Lcn2 and Il6 (Fig 3C–D). To directly test the hypothesis that the enhanced susceptibility to IMQ-induced dermatitis in Zc3h12a+/− mice was due to enhanced downstream IL-17 signaling, Zc3h12a+/− were crossed to Il17ra−/− mice and treated with IMQ. Zc3h12a+/−Il17ra−/− mice exhibited markedly reduced inflammation compared to Zc3h12a+/− mice, with milder skin pathology, reduced neutrophil infiltration (Fig 4A, data not shown) and abrogated expression of Il17a, Il17c and IL-17-dependent genes associated with psoriasis such as Il6 and Lcn2 (Fig 4B–E). In line with previous reports, Il17ra−/− mice also showed elevated IL-17A and IL-17C expression at baseline (Fig. 4B) (39, 40). These data verify that the MCPIP1-dependent phenotype is due to IL-17 signaling and not other cytokines associated with psoriasis.
Figure 4. Elevated IMQ-induced inflammation in MCPIP1-deficient mice is due to both IL-17A and IL-17C signaling.
The indicated mice were treated topically with control cream or IMQ on days 0, 1 and 2. (A) Skin neutrophil infiltration was determined by flow cytometry and % neutrophils in CD45+ gate is shown. (n=3–18). Data are pooled from 4 independent experiments. (B–E) Expression of the indicated genes was determined by qPCR. (n=3–10). Data are pooled from 2 independent experiments. *p<0.05, **p<0.01, ***p<0.001, Mann-Whitney test. ns-not significant.
IL-17RA is a shared subunit used by several IL-17 family cytokines, so the enhanced inflammation we observed could be due to more than one ligand (11, 41). In particular, IL-17C is reported to signal through IL-17RA paired with IL-17RE, inducing a characteristic subset of inflammatory genes and antimicrobial peptide (AMP) expression in keratinocytes. To determine which ligands were affected by MCPIP1, Zc3h12a+/− mice were crossed to Il17a−/− and Il17c−/− mice. Following IMQ treatment, Il17a and Il17c expression levels did not differ among WT, Zc3h12a+/−, Zc3h12a+/−Il17a−/− and Zc3h12a+/−Il17c−/− mice (Fig 4B,C). Notably, both Zc3h12a+/− Il17a−/− and Zc3h12a+/−Il17c−/− mice exhibited less skin inflammation than Zc3h12a+/− mice, with decreased neutrophil accumulation and reduced Lcn2, Il6 and Defb4 expression (Fig 4A–E). In addition, Il17a−/−, Zc3h12a+/−Il17a−/−, Il17c−/− and Zc3h12a+/−Il17c−/− mice all presented comparable levels of inflammation. These results demonstrate that the increased dermal inflammation seen with MCPIP1-deficiency can be partially rescued through elimination of IL-17A or IL-17C signaling, and more strongly reversed in the absence of the shared subunit IL-17RA.
MCPIP1-driven regulation of IL-17 signaling is restricted to the stromal compartment
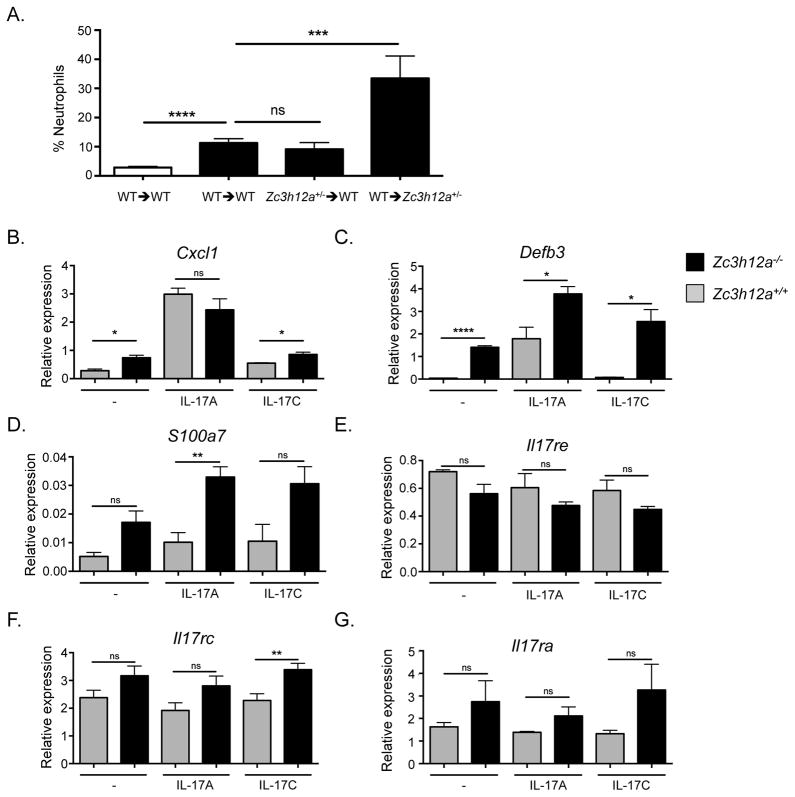
MCPIP1 regulates the effector mechanisms of IL-17A and its production by Th17 cells. To determine the relative contribution of MCPIP1 in the regulation of the immune and stromal compartments, we generated bone marrow (BM) chimeric mice with MCPIP1-haploinsufficiency in hematopoietic or radioresistant cells and subjected them to IMQ-dermatitis. Mice receiving Zc3h12a+/− BM developed gross skin inflammation that was indistinguishable from those receiving WT BM (not shown). Consistently, they showed comparable levels of skin neutrophil accumulation (Fig 5A). In contrast, Zc3h12a+/− mice given WT BM developed exacerbated skin pathology with increased neutrophilia, comparable to those receiving WT BM (Fig 5A). These data indicate that MCPIP1 regulates the skin phenotype specifically in stromal and radioresistant skin cells.
Figure 5. MCPIP1-dependent exacerbation of IMQ-driven dermatitis occurs through resident skin cells and is associated with elevated IL-17A and IL-17C signaling in keratinocytes.
WT or Zc3h12a+/− mice (n=4–9) were lethally irradiated and reconstituted with WT or Zc3h12a+/− bone marrow. After 6 weeks to allow immune reconstitution, mice were treated topically with control cream or IMQ and analyzed on days 0, 1 or 2. (A) Skin neutrophil infiltration was determined by flow cytometry and % neutrophils in CD45+ gate is shown. (B–F) Primary mouse WT (gray) or Zc3h12a−/− (black) neonatal KCs were treated for 16 h with IL-17A or IL-17C, and expression of indicated genes was assessed by qPCR (n=3–5). **p<0.01, ns-not significant, Mann-Whitney test.
Keratinocytes (KCs) are the most abundant cell type in the epidermis, and serve a key role in the initiation and perpetuation of immune responses. Keratinocytes are highly responsive to both IL-17A and IL-17C (14, 42, 43). To determine whether MCPIP1 restricts IL-17R signaling in KCs, primary KC cultures from postnatal day 2 WT and Zc3h12a−/− mice were assessed for expression of various genes in response to IL-17A and IL-17C after 16 h of cytokine treatment. Consistent with a role for MCPIP1 as a negative regulator of signaling, Zc3h12a−/− KCs showed enhanced expression of IL-17A and IL-17C target genes compared to WT (Fig 5B–D), including Cxcl1, Defb3, and S100a7. MCPIP1 was previously shown to induce degradation of mRNA transcripts encoding IL-17RA and IL-17RC (19). Levels of Il17re mRNA were slightly, if not significantly reduced in Zc3h12a−/− KCs (Fig 5E). However, Il17rc and Il17ra mRNA levels were modestly but consistently elevated in Zc3h12a−/− KCs compared to WT (Fig 5F–G), perhaps contributing to increased cytokine signaling.
Discussion
IL-17A and IL-17C play important roles controlling infections, but drive pathology in inflammatory conditions, especially psoriasis. Elevated IL-17A and IL-17C occurs in lesional human psoriasis skin samples (14, 41, 44), and human keratinocytes stimulated with IL-17A upregulate AMPs and neutrophil-attracting chemokines (45, 46). Consistently, genome-wide association studies (GWAS) have identified psoriasis-associated polymorphisms in genes critical for Th17 differentiation and IL-17A signaling, such as IL23R and TRAF3IP2 (encoding the essential IL-17 signaling intermediate Act1) (47–51). Mouse preclinical models of psoriasis have similarly revealed a role for IL-17 family cytokines in mediating disease. In the IMQ dermatitis model, IL-17RA-deficient mice show dramatically reduced disease development (33). IL-17C intradermal application of mice led to enhanced leukocyte recruitment in skin, and IL-17C-deficient mice develop milder skin inflammation upon imiquimod treatment (11). Additionally, a transgenic mouse overexpressing IL-17C in keratinocytes develops spontaneous skin lesions with many features of human psoriasis (14). The importance of IL-17A-mediated inflammation in psoriasis has been more recently highlighted by the clinical success of biologic drugs, including IL-17A-blocking antibodies secukinumab and ixekizumab and the IL-17RA-targeting antibody brodalumab (2, 52–55).
Given its prominent role in inflammation, it is unsurprising that the IL-17A signaling pathway is tightly controlled. Indeed, recent studies have elucidated several mechanisms by which IL-17A signaling is negatively regulated. For example, TRAF3 and TRAF4 interfere with receptor-proximal events by competing with Act1 or TRAF6 for IL-17RA occupancy (56, 57). The deubiquitinase A20 (TNFAIP3) is induced by IL-17A and mediates the removal of K63-linked ubiquitin chains on TRAF6, tempering activation of NF-κB and MAPK pathways (58). Similarly, USP25-mediated deubiquitination of TRAF5 and TRAF6 dampens IL-17A signaling (14). GSK-3β-mediated phosphorylation of the transcription factor C/EBPβ inhibits IL-17-dependent gene expression (59). Finally, MCPIP1 is a feedback inhibitor that degrades IL-17-induced target genes, including Il6, Nfkbiz as well as transcripts encoding IL-17R subunits (19, 22, 60). A new report indicates that the ABIN-1 (Tnip1) controls IL-17-induced pathology in IMQ-induced dermatitis (61). The concept that multiple inhibitors are needed to adequately control inflammatory cytokines is elegantly reviewed in Carpenter et al. (62). Despite its emerging role in inflammatory diseases (14), IL-17C signaling mechanisms are largely undefined, and no studies to date have focused on negative regulation of IL-17C-dependent signal transduction.
Here, we show for the first time that MCPIP1 restricts both IL-17A- and IL-17C-driven skin inflammation. Deficiency of one copy of the Zc3h12a gene was sufficient to render mice hyper-susceptible to IMQ-driven psoriasis. We previously reported that the strong inflammatory phenotype in Zc3h12a+/− heterozygous mice is not due to impaired baseline levels of MCPIP1 in peripheral organs such as kidney and lung; rather, haploinsufficient cells failed to show ligand-inducible upregulation of MCPIP1 (19). We verify here that Zc3h12a+/− mice also do not show exacerbated baseline inflammation in skin (Fig 2). Notably, Zc3h12a+/− mice on an Il17a-deficient or Il17c-deficient background exhibited milder dermatitis than Zc3h12a+/− mice (Fig 4). However, absence of Il17ra was required to render Zc3h12a+/− mice fully resistant to psoriasis, indicating a contribution of both IL-17 family members to skin pathology in this model system.
GWAS analysis of psoriasis has revealed associations with known regulators of immune signaling, including TNFAIP3 (A20), TNIP1 (ABIN-1, NAF1) and NFKBIA (IκBα) (63). To date, 11 non-synonymous SNPs have been reported for the ZC3H12A gene, but so far none are associated with human disease (64). MCPIP1 belongs to a 4-member family of CCCH-type zinc finger proteins. It is intriguing that polymorphisms in ZC3H12C (encoding MCPIP3) were identified in a GWAS study (50, 65). Similar to MCPIP1, MCPIP3 has endoribonuclease activity and curbs inflammatory responses in endothelial cells by suppressing NF-κB signaling (66). In addition, both ZC3H12C and ZC3H12A are induced in LPS-treated macrophages (67), though it is not known whether ZC3H12C is an IL-17 target gene. Future studies are needed to establish how different MCPIP family members regulate inflammation.
We found that ZC3H12A expression was markedly elevated in human psoriatic lesions compared to normal or uninvolved psoriatic skin (Fig 1), consistent with published data (21, 68) and our data in mice (Fig 2). Although this may seem counterintuitive given the role of MCPIP1 in restricting inflammation, it is common for feedback inhibitors like TNFAIP3 and TNIP1 to be associated with inflammation (69). It is likely that the increased ZC3H12A mRNA levels reflect the ongoing inflammatory milieu, and particularly the high IL-17A levels, found in diseased skin (19, 21, 23, 29),
Bone marrow chimeras demonstrated that MCPIP1 haploinsufficiency specifically in resident skin cells, not radiation-sensitive hematopoietic cells is sufficient to exacerbate inflammation in the IMQ model (Fig 5). This agrees with the restricted pattern of tissue expression of the IL-17RC and IL-17RE subunits, whereas IL-17RA is more broadly expressed. Prior studies in a model of autoimmune arthritis similarly demonstrated a role for IL-17RA only in the non-hematopoietic compartment (70). These results also agree with data showing that Act1 drives development of dermal and epidermal pathology dominantly in cells of stromal origin, namely KCs, endothelial cells and skin fibroblasts (34). Previously, MCPIP1 was shown to negatively regulate TCR signaling and IL-17A production by Th17 cells (28). However, we saw that Zc3h12a haploinsufficiency did not cause increased IL-17A production at the mRNA or protein levels during IMQ inflammation (Fig 2). Of note, γδ-T cells are the main IL-17A-producing population in the skin in this model, whereas the frequency of IL-17+ CD4+ T cells was very low in Zc3h12a+/− mice. Accordingly, because this is an acute disease model where IL-17 is made predominantly by γδ-T cells, a role for MCPIP1 in conventional CD4+T cells may not be apparent.
In summary, we have identified a novel role for the endoribonuclease and deubiquitinase MCPIP1 in restricting IL-17A and IL-17C-mediated skin inflammation. This is, to our knowledge, the first report of a negative regulatory element in the IL-17C signaling pathway, though it is not likely to be the last (62). Future studies will lend insight into the degree of conservation of MCPIP1-driven mechanisms of IL-17 cytokine family regulation.
Acknowledgments
We thank P. Kolattukudy for Zc3h12a+/− mice, Y. Iwakura for Il17a−/− mice, Amgen for Il17ra−/− mice, and Genentech for Il17c−/− mice.
Abbreviations
- AMP
antimicrobial peptide
- IMQ
imiquimod
- FFPE
formalin-fixed, paraffin embedded
- KC
keratinocyte
- MCPIP1
MCP1-induced protein 1
Footnotes
Grant support: SLG was supported by NIH grants AI107825, DE022550 and AR062546. NLW was supported by AR062546, AR39750, AR063437, and AR063852. JEG was supported by AR069071 and the A. Alfred Taubman Medical Research Institute Frances and Kenneth Eisenberg Emerging Scholar Award. AM was supported by AR066548 and AR067746. This work is solely the responsibility of the authors and does not necessarily reflect the views of the NIH or other funding bodies.
References
- 1.Gaffen SL, Jain R, Garg AV, Cua DJ. The IL-23-IL-17 immune axis: from mechanisms to therapeutic testing. Nat Rev Immunol. 2014;14:585–600. doi: 10.1038/nri3707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Langley RG, Elewski BE, Lebwohl M, Reich K, Griffiths CE, Papp K, Puig L, Nakagawa H, Spelman L, Sigurgeirsson B, Rivas E, Tsai TF, Wasel N, Tyring S, Salko T, Hampele I, Notter M, Karpov A, Helou S, Papavassilis C, Group ES, Group FS. Secukinumab in plaque psoriasis--results of two phase 3 trials. N Engl J Med. 2014;371:326–338. doi: 10.1056/NEJMoa1314258. [DOI] [PubMed] [Google Scholar]
- 3.Hueber W, Patel DD, Dryja T, Wright AM, Koroleva I, Bruin G, Antoni C, Draelos Z, Gold MH, Durez P, Tak PP, Gomez-Reino JJ, Foster CS, Kim RY, Samson CM, Falk NS, Chu DS, Callanan D, Nguyen QD, Rose K, Haider A, Di Padova F G Psoriasis Study, G Rheumatoid Arthritis Study, G Uveitis Study. Effects of AIN457, a fully human antibody to interleukin-17A, on psoriasis, rheumatoid arthritis, and uveitis. Sci Transl Med. 2010;2:52ra72. doi: 10.1126/scitranslmed.3001107. [DOI] [PubMed] [Google Scholar]
- 4.Sonnenberg GF, Artis D. Innate lymphoid cells in the initiation, regulation and resolution of inflammation. Nat Med. 2015;21:698–708. doi: 10.1038/nm.3892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cua DJ, Tato CM. Innate IL-17-producing cells: the sentinels of the immune system. Nat Rev Immunol. 2010;10:479–489. doi: 10.1038/nri2800. [DOI] [PubMed] [Google Scholar]
- 6.Ivanov II, McKenzie BS, Zhou L, Tadokoro CE, Lepelley A, Lafaille JJ, Cua DJ, Littman DR. The orphan nuclear receptor RORγt directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell. 2006;126:1121–1133. doi: 10.1016/j.cell.2006.07.035. [DOI] [PubMed] [Google Scholar]
- 7.Kronenberg M. Toward an understanding of NKT cell biology: progress and paradoxes. Annu Rev Immunol. 2005;23:877–900. doi: 10.1146/annurev.immunol.23.021704.115742. [DOI] [PubMed] [Google Scholar]
- 8.Marks BR, Nowyhed HN, Choi JY, Poholek AC, Odegard JM, Flavell RA, Craft J. Thymic self-reactivity selects natural interleukin 17-producing T cells that can regulate peripheral inflammation. Nat Immunol. 2009;10:1125–1132. doi: 10.1038/ni.1783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Villanova F, Flutter B, Tosi I, Grys K, Sreeneebus H, Perera GK, Chapman A, Smith CH, Di Meglio P, Nestle FO. Characterization of innate lymphoid cells in human skin and blood demonstrates increase of NKp44+ ILC3 in psoriasis. J Invest Derm. 2014;134:984–991. doi: 10.1038/jid.2013.477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chang SH, Reynolds JM, Pappu BP, Chen G, Martinez GJ, Dong C. Interleukin-17C promotes Th17 cell responses and autoimmune disease via interleukin-17 receptor E. Immunity. 2011;35:611–621. doi: 10.1016/j.immuni.2011.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ramirez-Carrozzi V, Sambandam A, Luis E, Lin Z, Jeet S, Lesch J, Hackney J, Kim J, Zhou M, Lai J, Modrusan Z, Sai T, Lee W, Xu M, Caplazi P, Diehl L, de Voss J, Balazs M, Gonzalez L, Jr, Singh H, Ouyang W, Pappu R. IL-17C regulates the innate immune function of epithelial cells in an autocrine manner. Nat Immunol. 2011;12:1159–1166. doi: 10.1038/ni.2156. [DOI] [PubMed] [Google Scholar]
- 12.Song X, Gao H, Lin Y, Yao Y, Zhu S, Wang J, Liu Y, Yao X, Meng G, Shen N, Shi Y, Iwakura Y, Qian Y. Alterations in the microbiota drive interleukin-17C production from intestinal epithelial cells to promote tumorigenesis. Immunity. 2014;40:140–152. doi: 10.1016/j.immuni.2013.11.018. [DOI] [PubMed] [Google Scholar]
- 13.Song X, Zhu S, Shi P, Liu Y, Shi Y, Levin SD, Qian Y. IL-17RE is the functional receptor for IL-17C and mediates mucosal immunity to infection with intestinal pathogens. Nat Immunol. 2011;12:1151–1158. doi: 10.1038/ni.2155. [DOI] [PubMed] [Google Scholar]
- 14.Johnston A, Fritz Y, Dawes SM, Diaconu D, Al-Attar PM, Guzman AM, Chen CS, Fu W, Gudjonsson JE, McCormick TS, Ward NL. Keratinocyte overexpression of IL-17C promotes psoriasiform skin inflammation. J Immunol. 2013;190:2252–2262. doi: 10.4049/jimmunol.1201505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Li X. Act1 modulates autoimmunity through its dual functions in CD40L/BAFF and IL-17 signaling. Cytokine. 2008;41:105–113. doi: 10.1016/j.cyto.2007.09.015. [DOI] [PubMed] [Google Scholar]
- 16.Song X, Qian Y. The activation and regulation of IL-17 receptor mediated signaling. Cytokine. 2013;62:175–182. doi: 10.1016/j.cyto.2013.03.014. [DOI] [PubMed] [Google Scholar]
- 17.Gaffen SL, Jain R, Garg A, Cua D. IL-23-IL-17 immune axis: From mechanisms to therapeutic testing. Nat Rev Immunol. 2014;14:585–600. doi: 10.1038/nri3707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dhamija S, Winzen R, Doerrie A, Behrens G, Kuehne N, Schauerte C, Neumann E, Dittrich-Breiholz O, Kracht M, Holtmann H. Interleukin-17 (IL-17) and IL-1 activate translation of overlapping sets of mRNAs, including that of the negative regulator of inflammation, MCPIP1. J Biol Chem. 2013;288:19250–19259. doi: 10.1074/jbc.M113.452649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Garg AV, Amatya N, Chen K, Cruz JA, Grover P, Whibley N, Conti HR, Hernandez Mir G, Sirakova T, Childs EC, Smithgall TE, Biswas PS, Kolls JK, McGeachy MJ, Kolattukudy PE, Gaffen SL. MCPIP1 Endoribonuclease Activity Negatively Regulates Interleukin-17-Mediated Signaling and Inflammation. Immunity. 2015;43:475–487. doi: 10.1016/j.immuni.2015.07.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jura J, Skalniak L, Koj A. Monocyte chemotactic protein-1-induced protein-1 (MCPIP1) is a novel multifunctional modulator of inflammatory reactions. Biochim Biophys Acta. 2012;1823:1905–1913. doi: 10.1016/j.bbamcr.2012.06.029. [DOI] [PubMed] [Google Scholar]
- 21.Ruiz-Romeu E, Ferran M, Gimenez-Arnau A, Bugara B, Lipert B, Jura J, Florencia EF, Prens EP, Celada A, Pujol RM, Santamaria-Babi LF. MCPIP1 RNase is aberrantly distributed in psoriatic epidermis and rapidly induced by IL-17A. J Invest Derm. 2016;136:1599–1607. doi: 10.1016/j.jid.2016.04.030. [DOI] [PubMed] [Google Scholar]
- 22.Sonder SU, Saret S, Tang W, Sturdevant DE, Porcella SF, Siebenlist U. IL-17-induced NF-κB activation via CIKS/Act1: physiologic significance and signaling mechanisms. J Biol Chem. 2011;286:12881–12890. doi: 10.1074/jbc.M110.199547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Matsushita K, Takeuchi O, Standley DM, Kumagai Y, Kawagoe T, Miyake T, Satoh T, Kato H, Tsujimura T, Nakamura H, Akira S. Zc3h12a is an RNase essential for controlling immune responses by regulating mRNA decay. Nature. 2009;458:1185–1190. doi: 10.1038/nature07924. [DOI] [PubMed] [Google Scholar]
- 24.Mino T, Murakawa Y, Fukao A, Vandenbon A, Wessels HH, Ori D, Uehata T, Tartey S, Akira S, Suzuki Y, Vinuesa CG, Ohler U, Standley DM, Landthaler M, Fujiwara T, Takeuchi O. Regnase-1 and Roquin Regulate a Common Element in Inflammatory mRNAs by Spatiotemporally Distinct Mechanisms. Cell. 2015;161:1058–1073. doi: 10.1016/j.cell.2015.04.029. [DOI] [PubMed] [Google Scholar]
- 25.Liang J, Wang J, Saad Y, Warble L, Becerra E, Kolattukudy PE. Participation of MCP-induced protein 1 in lipopolysaccharide preconditioning-induced ischemic stroke tolerance by regulating the expression of proinflammatory cytokines. J Neuroinflammation. 2011;8:182. doi: 10.1186/1742-2094-8-182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Niu J, Shi Y, Xue J, Miao R, Huang S, Wang T, Wu J, Fu M, Wu ZH. USP10 inhibits genotoxic NF-kappaB activation by MCPIP1-facilitated deubiquitination of NEMO. EMBO J. 2013;32:3206–3219. doi: 10.1038/emboj.2013.247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Uehata T, Iwasaki H, Vandenbon A, Matsushita K, Hernandez-Cuellar E, Kuniyoshi K, Satoh T, Mino T, Suzuki Y, Standley DM, Tsujimura T, Rakugi H, Isaka Y, Takeuchi O, Akira S. Malt1-Induced Cleavage of Regnase-1 in CD4(+) Helper T Cells Regulates Immune Activation. Cell. 2013;153:1036–1049. doi: 10.1016/j.cell.2013.04.034. [DOI] [PubMed] [Google Scholar]
- 28.Jeltsch KM, Hu D, Brenner S, Zoller J, Heinz GA, Nagel D, Vogel KU, Rehage N, Warth SC, Edelmann SL, Gloury R, Martin N, Lohs C, Lech M, Stehklein JE, Geerlof A, Kremmer E, Weber A, Anders HJ, Schmitz I, Schmidt-Supprian M, Fu M, Holtmann H, Krappmann D, Ruland J, Kallies A, Heikenwalder M, Heissmeyer V. Cleavage of roquin and regnase-1 by the paracaspase MALT1 releases their cooperatively repressed targets to promote T(H)17 differentiation. Nat Immunol. 2014;15:1079–1089. doi: 10.1038/ni.3008. [DOI] [PubMed] [Google Scholar]
- 29.Liang J, Saad Y, Lei T, Wang J, Qi D, Yang Q, Kolattukudy PE, Fu M. MCP-induced protein 1 deubiquitinates TRAF proteins and negatively regulates JNK and NF-kappaB signaling. J Exp Med. 2010;207:2959–2973. doi: 10.1084/jem.20092641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Dlugosz AA, Glick AB, Tennenbaum T, Weinberg WC, Yuspa SH. Isolation and utilization of epidermal keratinocytes for oncogene research. Methods Enzymol. 1995;254:3–20. doi: 10.1016/0076-6879(95)54003-2. [DOI] [PubMed] [Google Scholar]
- 31.Lichti U, Anders J, Yuspa SH. Isolation and short-term culture of primary keratinocytes, hair follicle populations and dermal cells from newborn mice and keratinocytes from adult mice for in vitro analysis and for grafting to immunodeficient mice. Nat Protoc. 2008;3:799–810. doi: 10.1038/nprot.2008.50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Matsushita K, Takeuchi O, Standley DM, Kumagai Y, Kawagoe T, Miyake T, Satoh T, Kato H, Tsujimura T, Nakamura H, Akira S. Zc3h12a is an RNase essential for controlling immune responses by regulating mRNA decay. Nature. 2009;458:1185–1190. doi: 10.1038/nature07924. [DOI] [PubMed] [Google Scholar]
- 33.van der Fits L, Mourits S, Voerman JS, Kant M, Boon L, Laman JD, Cornelissen F, Mus AM, Florencia E, Prens EP, Lubberts E. Imiquimod-induced psoriasis-like skin inflammation in mice is mediated via the IL-23/IL-17 axis. J Immunol. 2009;182:5836–5845. doi: 10.4049/jimmunol.0802999. [DOI] [PubMed] [Google Scholar]
- 34.Ha HL, Wang H, Pisitkun P, Kim JC, Tassi I, Tang W, Morasso MI, Udey MC, Siebenlist U. IL-17 drives psoriatic inflammation via distinct, target cell-specific mechanisms. Proc Natl Acad Sci U S A. 2014;111:E3422–3431. doi: 10.1073/pnas.1400513111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wohn CT, Pantelyushin S, Ober-Blobaum JL, Clausen BE. Aldara-induced psoriasis-like skin inflammation: isolation and characterization of cutaneous dendritic cells and innate lymphocytes. Methods Mol Biol. 2014;1193:171–185. doi: 10.1007/978-1-4939-1212-4_16. [DOI] [PubMed] [Google Scholar]
- 36.Yang C, Huang S, Wang X, Gu Y. Emerging Roles of CCCH-Type Zinc Finger Proteins in Destabilizing mRNA Encoding Inflammatory Factors and Regulating Immune Responses. Crit Rev Eukaryot Gene Expr. 2015;25:77–89. doi: 10.1615/critreveukaryotgeneexpr.2015013022. [DOI] [PubMed] [Google Scholar]
- 37.Pantelyushin S, Haak S, Ingold B, Kulig P, Heppner FL, Navarini AA, Becher B. Rorgammat+ innate lymphocytes and gammadelta T cells initiate psoriasiform plaque formation in mice. J Clin Invest. 2012;122:2252–2256. doi: 10.1172/JCI61862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Cai Y, Shen X, Ding C, Qi C, Li K, Li X, Jala VR, Zhang HG, Wang T, Zheng J, Yan J. Pivotal role of dermal IL-17-producing gammadelta T cells in skin inflammation. Immunity. 2011;35:596–610. doi: 10.1016/j.immuni.2011.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Nagata T, McKinley L, Peschon JJ, Alcorn JF, Aujla SJ, Kolls JK. Requirement of IL-17RA in Con A induced hepatitis and negative regulation of IL-17 production in mouse T cells. J Immunol. 2008;181:7473–7479. doi: 10.4049/jimmunol.181.11.7473. [DOI] [PubMed] [Google Scholar]
- 40.Kumar P, Monin L, Castillo P, Elsegeiny W, Horne W, Eddens T, Vikram A, Good M, Schoenborn AA, Bibby K, Montelaro RC, Metzger DW, Gulati AS, Kolls JK. Intestinal Interleukin-17 Receptor Signaling Mediates Reciprocal Control of the Gut Microbiota and Autoimmune Inflammation. Immunity. 2016;44:659–671. doi: 10.1016/j.immuni.2016.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Johansen C, Usher PA, Kjellerup RB, Lundsgaard D, Iversen L, Kragballe K. Characterization of the interleukin-17 isoforms and receptors in lesional psoriatic skin. Br J Dermatol. 2009;160:319–324. doi: 10.1111/j.1365-2133.2008.08902.x. [DOI] [PubMed] [Google Scholar]
- 42.Teunissen MB, Koomen CW, de Waal Malefyt R, Wierenga EA, Bos JD. Interleukin-17 and interferon-g synergize in the enhancement of proinflammatory cytokine production by human keratinocytes. J Invest Derm. 1998;111:645–649. doi: 10.1046/j.1523-1747.1998.00347.x. [DOI] [PubMed] [Google Scholar]
- 43.Chiricozzi A, Nograles KE, Johnson-Huang LM, Fuentes-Duculan J, Cardinale I, Bonifacio KM, Gulati N, Mitsui H, Guttman-Yassky E, Suarez-Farinas M, Krueger JG. IL-17 induces an expanded range of downstream genes in reconstituted human epidermis model. PLoS One. 2014;9:e90284. doi: 10.1371/journal.pone.0090284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wilson NJ, Boniface K, Chan JR, McKenzie BS, Blumenschein WM, Mattson JD, Basham B, Smith K, Chen T, Morel F, Lecron JC, Kastelein RA, Cua DJ, McClanahan TK, Bowman EP, de Waal Malefyt R. Development, cytokine profile and function of human interleukin 17-producing helper T cells. Nat Immunol. 2007;8:950–957. doi: 10.1038/ni1497. [DOI] [PubMed] [Google Scholar]
- 45.Liang SC, Tan XY, Luxenberg DP, Karim R, Dunussi-Joannopoulos K, Collins M, Fouser LA. Interleukin (IL)-22 and IL-17 are coexpressed by Th17 cells and cooperatively enhance expression of antimicrobial peptides. J Exp Med. 2006;203:2271–2279. doi: 10.1084/jem.20061308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Nograles KE, Zaba LC, Guttman-Yassky E, Fuentes-Duculan J, Suarez-Farinas M, Cardinale I, Khatcherian A, Gonzalez J, Pierson KC, White TR, Pensabene C, Coats I, Novitskaya I, Lowes MA, Krueger JG. Th17 cytokines interleukin (IL)-17 and IL-22 modulate distinct inflammatory and keratinocyte-response pathways. Br J Dermatol. 2008;159:1092–1102. doi: 10.1111/j.1365-2133.2008.08769.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Huffmeier U, Uebe S, Ekici AB, Bowes J, Giardina E, Korendowych E, Juneblad K, Apel M, McManus R, Ho P, Bruce IN, Ryan AW, Behrens F, Lascorz J, Bohm B, Traupe H, Lohmann J, Gieger C, Wichmann HE, Herold C, Steffens M, Klareskog L, Wienker TF, Fitzgerald O, Alenius GM, McHugh NJ, Novelli G, Burkhardt H, Barton A, Reis A. Common variants at TRAF3IP2 are associated with susceptibility to psoriatic arthritis and psoriasis. Nat Genet. 2010;42:996–999. doi: 10.1038/ng.688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ellinghaus E, Ellinghaus D, Stuart PE, Nair RP, Debrus S, Raelson JV, Belouchi M, Fournier H, Reinhard C, Ding J, Li Y, Tejasvi T, Gudjonsson J, Stoll SW, Voorhees JJ, Lambert S, Weidinger S, Eberlein B, Kunz M, Rahman P, Gladman DD, Gieger C, Wichmann HE, Karlsen TH, Mayr G, Albrecht M, Kabelitz D, Mrowietz U, Abecasis GR, Elder JT, Schreiber S, Weichenthal M, Franke A. Genome-wide association study identifies a psoriasis susceptibility locus at TRAF3IP2. Nat Genet. 2010;42:991–995. doi: 10.1038/ng.689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Cargill M, Schrodi SJ, Chang M, Garcia VE, Brandon R, Callis KP, Matsunami N, Ardlie KG, Civello D, Catanese JJ, Leong DU, Panko JM, McAllister LB, Hansen CB, Papenfuss J, Prescott SM, White TJ, Leppert MF, Krueger GG, Begovich AB. A large-scale genetic association study confirms IL12B and leads to the identification of IL23R as psoriasis-risk genes. Am J Hum Genet. 2007;80:273–290. doi: 10.1086/511051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Tsoi LC, Spain SL, Knight J, Ellinghaus E, Stuart PE, Capon F, Ding J, Li Y, Tejasvi T, Gudjonsson JE, Kang HM, Allen MH, McManus R, Novelli G, Samuelsson L, Schalkwijk J, Stahle M, Burden AD, Smith CH, Cork MJ, Estivill X, Bowcock AM, Krueger GG, Weger W, Worthington J, Tazi-Ahnini R, Nestle FO, Hayday A, Hoffmann P, Winkelmann J, Wijmenga C, Langford C, Edkins S, Andrews R, Blackburn H, Strange A, Band G, Pearson RD, Vukcevic D, Spencer CC, Deloukas P, Mrowietz U, Schreiber S, Weidinger S, Koks S, Kingo K, Esko T, Metspalu A, Lim HW, Voorhees JJ, Weichenthal M, Wichmann HE, Chandran V, Rosen CF, Rahman P, Gladman DD, Griffiths CE, Reis A, Kere J, Nair RP, Franke A, Barker JN, Abecasis GR, Elder JT, Trembath RC P Collaborative Association Study of, C. Genetic Analysis of Psoriasis, E. Psoriasis Association Genetics, C. Wellcome Trust Case Control. Identification of 15 new psoriasis susceptibility loci highlights the role of innate immunity. Nat Genet. 2012;44:1341–1348. doi: 10.1038/ng.2467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Sonder SU, Paun A, Ha HL, Johnson PF, Siebenlist U. CIKS/Act1-mediated signaling by IL-17 cytokines in context: implications for how a CIKS gene variant may predispose to psoriasis. J Immunol. 2012;188:5906–5914. doi: 10.4049/jimmunol.1103233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Durham LE, Kirkham BW, Taams LS. Contribution of the IL-17 Pathway to Psoriasis and Psoriatic Arthritis. Curr Rheum Rep. 2015;17:55. doi: 10.1007/s11926-015-0529-9. [DOI] [PubMed] [Google Scholar]
- 53.Sanford M, McKeage K. Secukinumab: first global approval. Drugs. 2015;75:329–338. doi: 10.1007/s40265-015-0359-0. [DOI] [PubMed] [Google Scholar]
- 54.Leonardi C, Matheson R, Zachariae C, Cameron G, Li L, Edson-Heredia E, Braun D, Banerjee S. Anti-interleukin-17 monoclonal antibody ixekizumab in chronic plaque psoriasis. N Engl J Med. 2012;366:1190–1199. doi: 10.1056/NEJMoa1109997. [DOI] [PubMed] [Google Scholar]
- 55.Baeten D, Sieper J, Braun J, Baraliakos X, Dougados M, Emery P, Deodhar A, Porter B, Martin R, Andersson M, Mpofu S, Richards HB, Group MS, Group MS. Secukinumab, an Interleukin-17A Inhibitor, in Ankylosing Spondylitis. N Engl J Med. 2015;373:2534–2548. doi: 10.1056/NEJMoa1505066. [DOI] [PubMed] [Google Scholar]
- 56.Zhu S, Pan W, Shi P, Gao H, Zhao F, Song X, Liu Y, Zhao L, Li X, Shi Y, Qian Y. Modulation of experimental autoimmune encephalomyelitis through TRAF3-mediated suppression of interleukin 17 receptor signaling. J Exp Med. 2010;207:2647–2662. doi: 10.1084/jem.20100703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Zepp JA, Liu C, Qian W, Wu L, Gulen MF, Kang Z, Li X. Cutting edge: TNF receptor-associated factor 4 restricts IL-17-mediated pathology and signaling processes. J Immunol. 2012;189:33–37. doi: 10.4049/jimmunol.1200470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Garg AV, Ahmed M, Vallejo AN, Ma A, Gaffen SL. The deubiquitinase A20 mediates feedback inhibition of interleukin-17 receptor signaling. Sci Signal. 2013;6:ra44. doi: 10.1126/scisignal.2003699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Shen F, Li N, Gade P, Kalvakolanu DV, Weibley T, Doble B, Woodgett JR, Wood TD, Gaffen SL. IL-17 receptor signaling inhibits C/EBPbeta by sequential phosphorylation of the regulatory 2 domain. Sci Signal. 2009;2:ra8. doi: 10.1126/scisignal.2000066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Somma D, Mastrovito P, Grieco M, Lavorgna A, Pignalosa A, Formisano L, Salzano AM, Scaloni A, Pacifico F, Siebenlist U, Leonardi A. CIKS/DDX3X Interaction Controls the Stability of the Zc3h12a mRNA Induced by IL-17. J Immunol. 2015;194:3286–3294. doi: 10.4049/jimmunol.1401589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Ippagunta SK, Gangwar R, Finkelstein D, Vogel P, Pelletier S, Gingras S, Redecke V, Hacker H. Keratinocytes contribute intrinsically to psoriasis upon loss of Tnip1 function. Proc Natl Acad Sci U S A. 2016;113:E6162–E6171. doi: 10.1073/pnas.1606996113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Carpenter S, Ricci EP, Mercier BC, Moore MJ, Fitzgerald KA. Post-transcriptional regulation of gene expression in innate immunity. Nat Rev Immunol. 2014;14:361–376. doi: 10.1038/nri3682. [DOI] [PubMed] [Google Scholar]
- 63.Harden JL, Krueger JG, Bowcock AM. The immunogenetics of Psoriasis: A comprehensive review. J Autoimm. 2015;64:66–73. doi: 10.1016/j.jaut.2015.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Cifuentes RA, Cruz-Tapias P, Rojas-Villarraga A, Anaya JM. ZC3H12A (MCPIP1): molecular characteristics and clinical implications. Clinica chimica acta; Int J Clin Chem. 2010;411:1862–1868. doi: 10.1016/j.cca.2010.08.033. [DOI] [PubMed] [Google Scholar]
- 65.Munir S, ber Rahman S, Rehman S, Saba N, Ahmad W, Nilsson S, Mazhar K, Naluai AT. Association analysis of GWAS and candidate gene loci in a Pakistani population with psoriasis. Molec Immunol. 2015;64:190–194. doi: 10.1016/j.molimm.2014.11.015. [DOI] [PubMed] [Google Scholar]
- 66.Liu L, Zhou Z, Huang S, Guo Y, Fan Y, Zhang J, Zhang J, Fu M, Chen YE. Zc3h12c inhibits vascular inflammation by repressing NF-kappaB activation and pro-inflammatory gene expression in endothelial cells. Biochem J. 2013;451:55–60. doi: 10.1042/BJ20130019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Liang J, Wang J, Azfer A, Song W, Tromp G, Kolattukudy PE, Fu M. A novel CCCH-zinc finger protein family regulates proinflammatory activation of macrophages. J Biol Chem. 2008;283:6337–6346. doi: 10.1074/jbc.M707861200. [DOI] [PubMed] [Google Scholar]
- 68.Tian S, Krueger JG, Li K, Jabbari A, Brodmerkel C, Lowes MA, Suarez-Farinas M. Meta-analysis derived (MAD) transcriptome of psoriasis defines the “core” pathogenesis of disease. PLoS One. 2012;7:e44274. doi: 10.1371/journal.pone.0044274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ma A, Malynn BA. A20: linking a complex regulator of ubiquitylation to immunity and human disease. Nat Rev Immunol. 2012;12:774–785. doi: 10.1038/nri3313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Lubberts E, Schwarzenberger P, Huang W, Schurr JR, Peschon JJ, van den Berg WB, Kolls JK. Requirement of IL-17 receptor signaling in radiation-resistant cells in the joint for full progression of destructive synovitis. J Immunol. 2005;175:3360–3368. doi: 10.4049/jimmunol.175.5.3360. [DOI] [PubMed] [Google Scholar]