Abstract
Plants can perceive environmental changes and respond to external stressors. Here, we show that OsNAC2, a member of the NAC transcription factor family, was strongly induced by ABA and osmotic stressors such as drought and high salt. With reduced yields under drought conditions at the flowering stage, OsNAC2 overexpression lines had lower resistance to high salt and drought conditions. RNAi plants showed enhanced tolerance to high salinity and drought stress at both the vegetative and flowering stages. Furthermore, RNAi plants had improved yields after drought stress. A microarray assay indicated that many ABA-dependent stress-related genes were down-regulated in OsNAC2 overexpression lines. We further confirmed that OsNAC2 directly binds the promoters of LATE EMBRYOGENESIS ABUNDANT 3 (OsLEA3) and Stress-Activated Protein Kinases 1 (OsSAPK1), two marker genes in the abiotic stress and ABA response pathways, respectively. Our results suggest that in rice OsNAC2 regulates both abiotic stress responses and ABA-mediated responses, and acts at the junction between the ABA and abiotic stress pathways.
Drought, high salinity, and low temperature are major stress factors affecting plant growth1. When plants are exposed to variable environments, cells perceive stress signals from the outside world. This process occurs through a series of complex signalling pathways including the ABA-dependent and ABA-independent pathways2,3. The stress signals are sent to transcription factors (TFs) involved in stress responses that then trigger the expression of downstream stress response genes. Through this mechanism, plants reduce the negative influence of abiotic stressors by activating stress tolerance reactions4.
Members of the NAC TF family, which is specific to higher plants, can bind to promoter DNA as a dimer and induce gene expression. The name is derived from the first letters of three genes, NAM from a petunia hybrid and ATAF1/2 and CUC2 from Arabidopsis thaliana, that were initially found by Souer and Aida5,6. To date, there are 117 and 151 NAC TFs in Arabidopsis and Oryza sativa (rice), respectively7. Structural and functional analyses indicated that the NAC family is paralogous to the plant WRKY TF family8. Further evolutionary analysis showed that ancient eukaryotic WRKY proteins may be the common ancestors of plant NAC and WRKY TFs and animal GCM TFs9. NAC proteins have a highly conserved N-terminal domain called the NAC domain that is responsible for DNA binding and a variable C-terminal domain that is a transcriptional activation domain10.
NAC family TFs are mainly involved in plant growth, development, and biotic or abiotic stress responses. The petunia NAM gene is required for shoot tip meristem formation5 while Arabidopsis CUC111, CUC26, CUC12, and rice OsNAC213 can inhibit the growth of certain cells, promote the production of axillary meristems, and promote the development of organ boundaries. NAC114 and AtNAC215 are induced by IAA and promote lateral root growth and NST1 and NST2 co-regulate Arabidopsis secondary cell wall synthesis16,17,18. SND1 stimulates the expression of secondary cell wall synthesis genes19 while VND6/7 are key genes regulating Arabidopsis xylem development20. NAC family members also play important roles in cell division and extension21, floral development and flowering22, senescence23,24,25, and seed germination26,27.
Studies have shown that abiotic stressors can induce the expression of many rice NAC TF genes. SNAC1 can greatly increase rice drought tolerance in both the vegetative and flowering stages28. OsNAC529 and OsNAC630,31 proteins can bind to the promoter of OsLEA3 and significantly increase high salt and drought tolerance. Overexpression of OsNAC10 increased rice yield under drought conditions32. ANAC019, ANAC055 and ANAC072 are induced by drought, high temperatures, and ABA and are required for drought tolerance33. Transgenic Arabidopsis plants expressing the rice gene OsNAC063 had enhanced high salt and drought tolerance34 while ATAF1 may negatively control functional genes in drought stress35. Transgenic Arabidopsis plants expressing LOV1 had enhanced resistance to low temperatures36 while the overexpression of ANAC102 in Arabidopsis confers resistance to hypoxic stress37. No reports have, however, been published outlining the role of OsNAC2 in abiotic stress responses.
In an earlier study, overexpression of OsNAC2 in rice was reported to increase the tiller number13. Our lab previously found that OsNAC2 was also involved in the regulation of plant height through the GA pathway38. Here, we have shown that OsNAC2 is induced by ABA and osmotic stressors like drought and high salt. Rice OsNAC2 overexpression lines had lower drought and high salinity tolerance in both the vegetative and flowering stages compared with wild-type (WT) plants, while in RNAi lines have higher drought and high salinity resistance performance. Additionally, RNAi plants maintained high yields under drought conditions. Gene expression analysis showed that OsNAC2 overexpression down-regulated ABA-dependent stress-related marker genes, suggesting that OsNAC2 is a negative regulator of the high salinity and drought response pathways. We further explored OsNAC2 target genes using ChIP and yeast-one-hybrid analyses. Our data suggests that OsNAC2 has a new function and regulation mechanism in abiotic stress responses by directly regulating OsLEA3 (LATE EMBRYOGENESIS ABUNDANT 3) and OsSAPK1 (Stress-Activated Protein Kinases 1). It may, therefore, play an important role in linking the ABA and abiotic stress response pathways together.
Results
OsNAC2 expression is induced by osmotic stress and ABA
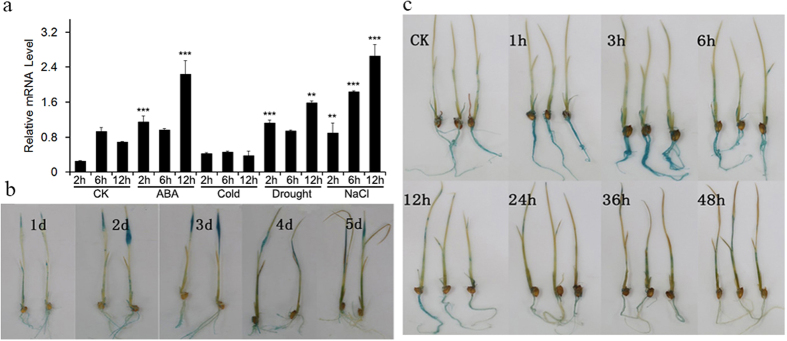
Expression of OsNAC2 in response to ABA, low temperature, dehydration, and NaCl was analysed using qRT-PCR to show time-dependent induction patterns. The OsNAC2 transcript accumulated within 2 hours under ABA, dehydration, and NaCl treatments, with peak expression reached after 12 hours. Conversely, expression of OsNAC2 decreased slightly during 12 hours of low-temperature treatment (Fig. 1a). There was no apparent pattern in OsNAC2 mRNA accumulation in plants treated with water only.
Figure 1. Expression induction of OsNAC2 under different stress and hormone treatments.
(a) Quantitative polymerase chain reaction (PCR) analysis of OsNAC2 expression in response to ABA (100 mM), cold, drought (20% PEG8000) and NaCl (200 mM) for 2 h, 6 h and 12 h. RNA was extracted from whole seedlings. Data were means ± SE with at least three replicates. Asterisks represent statistically significant differences between CK and treated lines. **P < 0.01, ***P < 0.001. (b) GUS staining of ProOsNAC2:GUS seedlings under NaCl (150 mM) treatment for 1d, 2d, 3d, 4d and 5d. (c) GUS staining of ProOsNAC2:GUS seedlings under air dry treatment from 0 h to 2d.
To study the effect of the promoter region on the expression of OsNAC2 in seedlings under abiotic stress, we generated transgenic rice plants containing a 1500-bp OsNAC2 promoter fragment (Fig. S1) fused with a GUS reporter gene to visualise is localisation (Fig. S2). Histochemical GUS staining indicated that the blue colouration was deepest in the leaves of seedlings treated with NaCl for 2d (Fig. 1b) and in the roots of seedlings air-dried for 3 h (Fig. 1c). With both treatments, the expression subsequently decreased as the treatment time increased. The results indicated that OsNAC2 will be induced by NaCl and mainly expressed in leaf. However, OsNAC2 was induced by drought treatment and expressed mostly in root. This suggests that OsNAC2 might play a role in different responses to various abiotic stressors.
Changes in OsNAC2 expression affects salt sensitivity
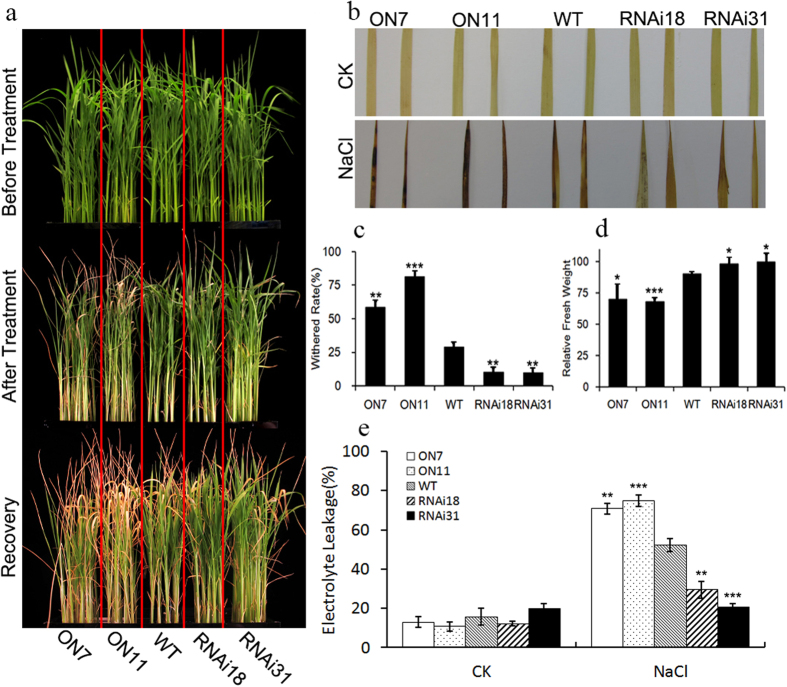
Given that OsNAC2 expression was induced by salt and drought stress, we designed a set of experiments to test its function in abiotic stress tolerance. Therefore, ON7 (OsNAC2 overexpression line no. 7), ON11 (OsNAC2 overexpression line no. 11), RNAi18 (OsNAC2 RNAi line no. 18), RNAi31 (OsNAC2 RNAi line no. 31), and WT plants were evaluated for high salinity tolerance and drought stress. In OsNAC2 overexpression lines, OsNAC2 is promoted by 35 S, and fused with GFP-tag. Two-week-old rice seedlings were treated with 150 mM NaCl for 2.5 d. After high salt treatment, plants were transferred to recover in normal water for 2 d. All the plants grew well under normal conditions. After high salt treatment and recovery, ON7 and ON11 plants were severely withered and had damaged leaves (Fig. 2a). Compared with the WT, significantly more seedlings of the ON7 and ON11 overexpression lines withered (58.5% and 81.3%. respectively). Of the RNAi18 and RNAi31 seedlings, 10.4% and 9.8% withered, respectively, whereas 29.0% of the WT seedlings withered (Fig. 2c). We used diaminobenzidine (DAB) staining to test reactive oxygen species in all the plant lines. Under normal conditions (CK), there was no DAB staining in WT and transgenic plants. After high salt treatment, DAB staining accumulated in ON7 and ON11 leaves and, to a lesser degree, in the leaves of RNAi and WT seedlings (Fig. 2b). These results indicated greater ROS accumulation in overexpression lines after high salinity stress. We also evaluated the effect of salt stress by examining electrolyte leakage and relative fresh weight. All the plants had low electrolyte leakage under normal growth conditions. Overexpression plants exhibited a significantly lower relative fresh weight than WT plants while RNAi18 and RNAi31 had significantly higher fresh weights compared with WT (Fig. 2d). At the same time, a greater increase in electrolyte leakage was found in ON7 and ON11 plants than in WT plants after 150 mM NaCl treatment, while RNAi18 and RNAi31 plants had markedly smaller increases in electrolyte leakage than WT (Fig. 2e). These results indicated that overexpression of OsNAC2 suppresses salt resistance in transgenic plant.
Figure 2. Phenotype analysis of OsNAC2 transgenic plants and WT in response to 150 mM NaCl treatment.
The seedlings were cultivated in basal nutrient solution and normal conditions, 28 °C, 16 h light and 8 h dark. Then two-week-old seedlings were transferred into nutrient solution containing 150 mM NaCl for 2.5d and recovery for 2d. (a) Phenotype of WT and OsNAC2 transgenic plants before and after high salt (150 mM NaCl) treatment for 2.5d and recovery for 2d. (b) DAB staining of WT and transgenic seedlings leaves after recovery. (c) Withered rate of WT and transgenic seedlings after recovery. (d) Relative fresh weight of WT and transgenic seedlings after recovery. (e) Electrolyte leakage of WT and transgenic seedlings after recovery. Data were means ± SE with at least five replicates. Asterisks represent statistically significant differences between WT and transgenic plants. *P < 0.05, **P < 0.01, ***P < 0.001.
Compared with hydroponics, growing plants in pots of soil more closely resembles the real growing environment of rice. All the rice seedlings were, therefore, grown hydroponically until they were 4 w old before being transferred into pots for soil experiments. After 14 d of 150 mM NaCl treatment, the leaves of ON7 and ON11 plants were yellow and wilting while the leaves of RNAi31 plants remained green and upright (Fig. 3a). Approximately 50% of ON7 and ON11 plants had died by this point while approximately 60% of the corresponding WT plants and over 70% of the RNAi31 plants remained alive (Fig. 3a). We also evaluated the viability of all the plants by measuring fresh weights. Before NaCl treatment, there were no significant differences in the fresh weight of WT and transgenic rice seedlings under normal soil conditions. After high salt stress, ON11 lines lost more fresh weight than the corresponding WT plants while RNAi31 lost less fresh weight (Fig. 3c). Chlorophyll plays an important role in plant growth and development. After salt stress, more chlorophyll was degraded in ON11 than in the WT plants while more chlorophyll was conserved in the RNAi31 plants compared with WT (Fig. 3d). These data demonstrated that overexpression of OsNAC2 could make rice more sensitive to high salt stress whether in hydroponic or soil culture conditions.
Figure 3. Soil experiment analysis of high salt stress in control and transgenic rice plants.
Rice seedlings were grown under normal conditions for four weeks, and then irrigated with 150 mM NaCl solution for 14d with 7d recovery. (a) Phenotype of WT and OsNAC2 transgenic plants before and after high salt (150 mM NaCl) stress for 14d in pot. (b) Survival rate of WT and transgenic seedlings after stress. (c) Fresh weight of WT and transgenic seedlings after high stress. (d) Chlorophyll content of WT and transgenic seedlings after high stress. The results are averages of three independent experiments with 10 plants per experiment. Data were means ± SE. Asterisks represent statistically significant differences between WT and transgenic plants. *P < 0.05, **P < 0.01, ***P < 0.001.
Changes in OsNAC2 expression affect drought sensitivity
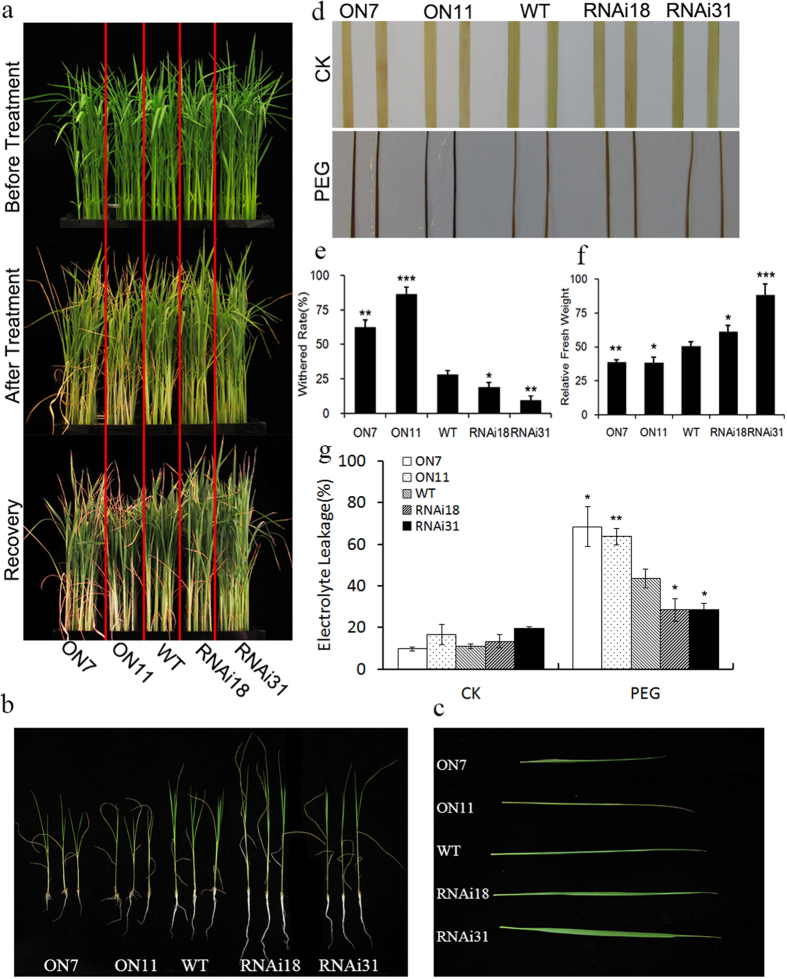
As with salinity stress, OsNAC2 transcription was also induced by drought treatment (Fig. 1a), suggesting that OsNAC2 might also play an important role in the response to drought stress. We used PEG8000 to mimic natural drought conditions. Two-week-old transgenic and WT rice seedlings were transferred into nutrient solution with 20% PEG8000. After 5d treatment and 3d recovery, more leaves of the overexpression lines ON7 and ON11 (62.5% and 86.5%, respectively) had rolled into a needle-like shape than the WT (28.1%), while less RNAi18 and RNAi31 plants had turned yellow (18.8% and 9.4%, respectively) but no needle-like shape leaves (Fig. 4a,b,c,e). DAB staining and analysis of the relative fresh weight and electrolyte leakage were performed to evaluate the drought resistant phenotype. Greater ROS accumulation was observed in ON7 and ON11 lines (Fig. 4d) and both lost more fresh weight than the WT plants during PEG treatment and had significantly more electrolyte leakage (Fig. 4f,g).
Figure 4. Phenotype analysis of OsNAC2 transgenic plants and WT response to 20% PEG8000 treatment.
The seedlings were cultivated in basal nutrient solution and normal conditions, 28 °C, 16 h light and 8 h dark. Then two-week-old seedlings were transferred into nutrient solution containing 20% PEG8000 for 5d and recovery for 3d. (a) Phenotype of WT and OsNAC2 transgenic plants before and after PEG8000 (20%) stress for 5d and recovery for 3d. (b,c) Phenotype of ON7, ON11, WT, RNAi18 and RNAi31 after salt treatment. Leaves turned yellow and rolled into a needle-like shape. (d) DAB staining of WT and transgenic seedlings leaves after recovery. (e) Withered rate of WT and transgenic seedlings after recovery. We sampled 96 individuals of each line. Then we measured the length of all leaves and the length of withered parts in each line, and calculated the result by withered parts’ length/Total leaves length. The assay was repeated three times. (f) Relative fresh weight of WT and transgenic seedlings after recovery. (g) Electrolyte leakage of WT and transgenic seedlings after recovery. Data were means ± SE with at least three biological replicates. Asterisks represent statistically significant differences between WT and transgenic plants. *P < 0.05, **P < 0.001, ***P < 0.001.
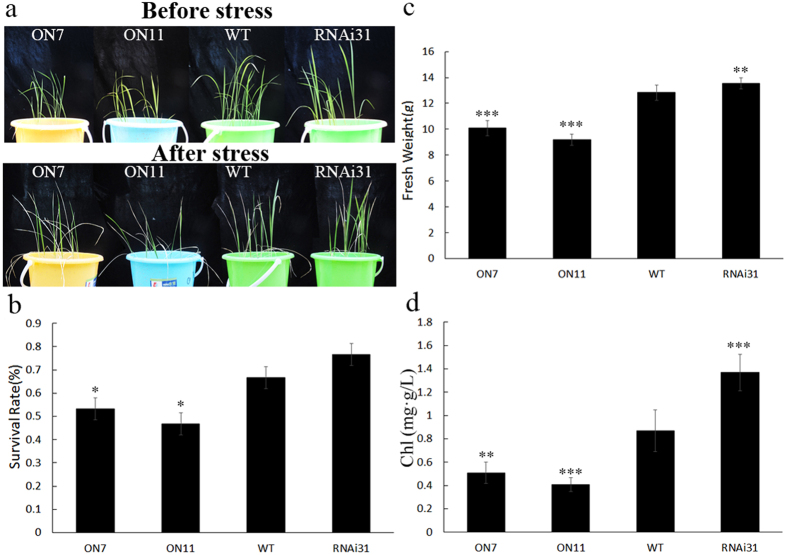
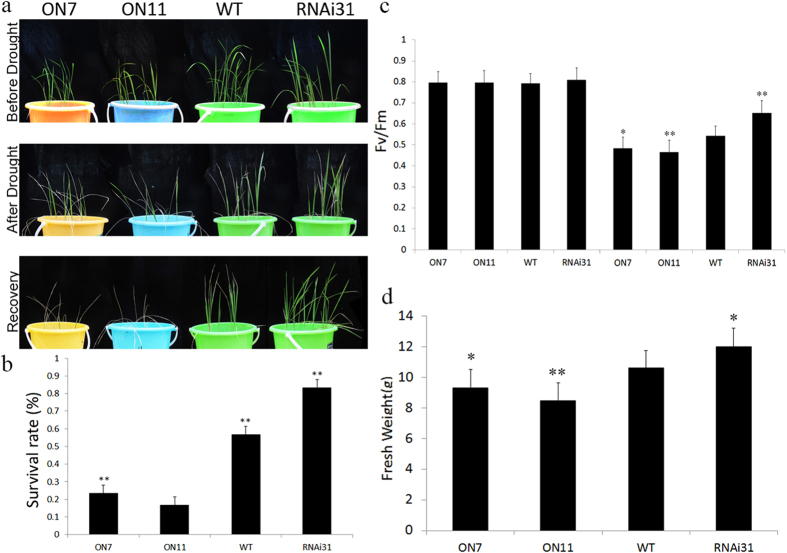
Our results confirmed that overexpression of OsNAC2 decreased the tolerance of rice to the PEG solution leading us to assume that changes in OsNAC2 expression would affect the drought sensitivity of rice. To evaluate the drought tolerance of transgenic and WT rice seedlings we used four-week-old transgenic seedlings that were grown in pots alongside corresponding WT plants. In normal conditions, no phenotypic differences were observed and each seedling was healthy. After 14 d without watering, ON7 and ON11 seedlings turned yellow and wilted while RNAi31 plants remained green. These differences became more obvious after 7 d of re-watering (Fig. 5a). The survival rate of ON7 and ON11 significantly decreased compared with the WT, while the survival rate of RNAi31 increased significantly (Fig. 5b). The Fv/Fm value, which represents the activity of PSII, was used to evaluate the extent of rice plant damage, with more damaged plants having lower values. The Fv/Fm values were lower in ON11 than in the WT, while the values were higher in RNAi31 (Fig. 5c). The post-treatment fresh weights correlated with the OsNAC2 transcript level; ON7 and ON11 had lower fresh weights than the WT while RNAi31 plants had higher values (Fig. 5d). These data indicated that overexpression of OsNAC2 could make rice plants more sensitive to drought stress at the vegetative stage.
Figure 5. Phenotype analysis of OsNAC2 transgenic plants and WT in response to drought treatment for 14 days in the soil.
Rice seedlings were grown under normal conditions for four weeks, gradually reduced water supply and cut out water for 14d with 7d recovery. (a) Phenotype of WT and OsNAC2 transgenic plants before and after drought stress, and after 7d recovery. (b) Survival rate of WT and transgenic seedlings after recovery. (c) Changes in Chl fluorescence (Fv/Fm) under normal conditions and drought treatment. (d) Overground fresh weight of WT and transgenic seedlings after recovery. The results are averages of three independent experiments with 10 plants per experiment. Data were means ± SE with at least three biological replicates. Asterisks represent statistically significant differences between WT and transgenic plants. *P < 0.05, **P < 0.01, ***P < 0.001.
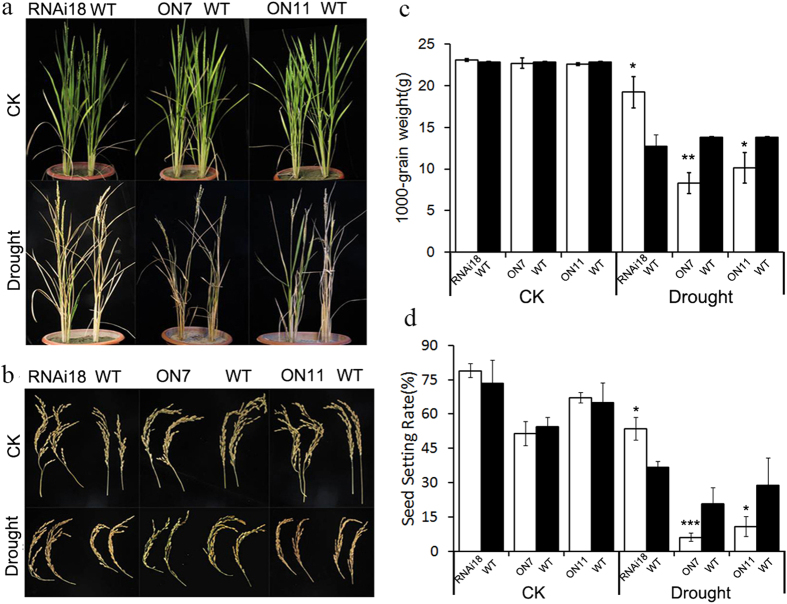
As rice yields and phenotypes are hypersensitive to drought stress during flower development39, we tested the drought tolerance of transgenic rice at this stage. All the seedlings were grown in pots under normal growth conditions before they reached the heading stage. Control pots were watered regularly while those of drought treatment groups were not irrigated for 12 d until harvest. Phenotypic analysis of the different lines after water deficient and recovery shown that ON7 and ON11 suffered severe growth retardation and high senescence (Fig. 6a). We measured several agronomic traits to evaluate the drought damage to rice. After drought treatment, ON7 and ON11 plants had the lowest 1000-grain weight, while RNAi18 plants had the highest (Fig. 6b,c). The seed setting rate of ON7 and ON11 was also visibly lower than in WT while RNAi18 plants had a significantly higher seed setting rate (Fig. 6d). These results further confirmed that overexpression of OsNAC2 made rice plants hypersensitive to drought tolerance and decreased their yield under drought stress.
Figure 6. Drought tolerance of OsNAC2 transgenic rice at the flowering stage.
(a) Phenotype of reproductive rice under normal and drought conditions. (b) The spikelet of rice plants under normal and drought conditions. (c) 1000-grain weight of rice plants under normal and drought conditions. (d) Seed setting rate of rice plants under normal and drought conditions. We counted full seeds and empty seeds proportion in 300 grains of each line, and calculated by full seeds/300. The assay was repeated three times.
Stress-related marker genes are all down-regulated in OsNAC2 overexpression plants
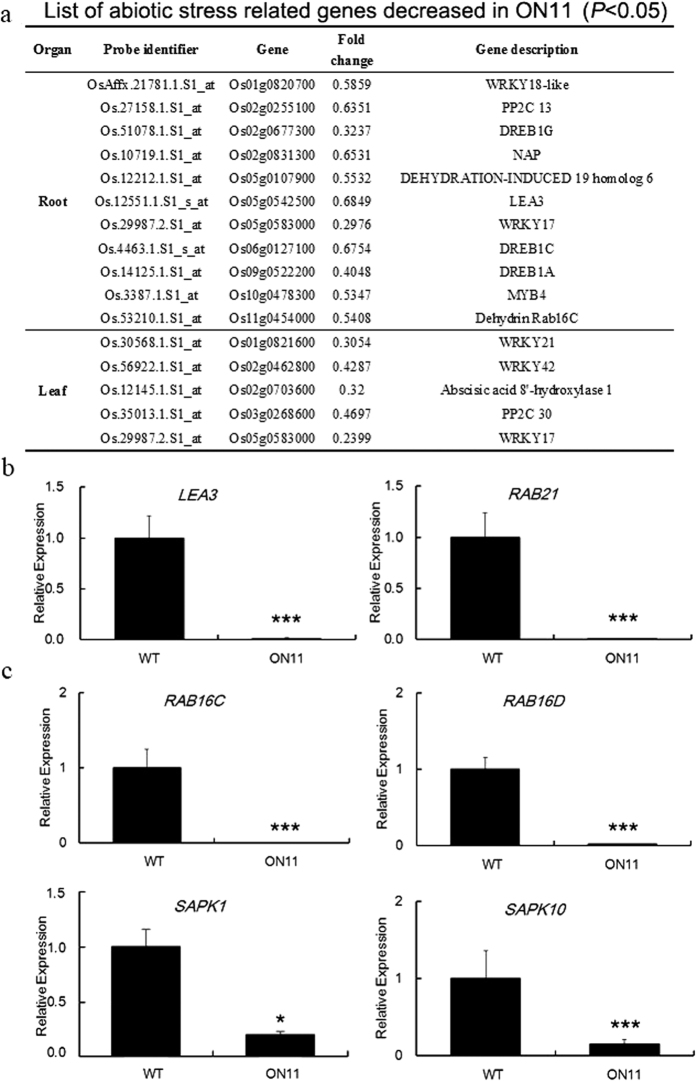
The above results showed that overexpression of OsNAC2 reduced plant tolerances to drought and high salinity stress. To further explore the regulation mechanism of OsNAC2 in the abiotic stress response pathway, we performed gene expression profiling analysis compared WT and OsNAC2-overexpressing plants using the existing rice microarray data in our lab. Compared with WT, many stress response genes were expressed in different patterns in the ON lines (Fig. 7a). According to the gene descriptions, we discovered that many of these differentially expressed genes are involved in drought and high salinity stress responses and may interact with the ABA signalling pathway. Some genes, including LEA3, some GTPases (RAB21, RAB16C, RAB16D), and sucrose nonfermenting1–related protein kinase2 (SnRK2) protein kinase genes (SAPK1 and SAPK10), that function in ABA signalling and stress tolerance were significantly down-regulated in ON11 plants compared with the WT in the result of real-time PCR (Fig. 7b,c). These results verified the microarray data and revealed a potential relationship between OsNAC2 and the ABA-dependent stress signalling pathway.
Figure 7. Microarray analysis and real time PCR confirmation of abiotic stress related genes.
(a) List of abiotic stress related genes which decreased in ON11. (b,c) Expression of stress related genes in leaf and root respectively. Data were means ± SE with at least three biological replicates. Asterisks represent statistically significant differences between WT and transgenic plants. *P < 0.05, **P < 0.01, ***P < 0.001.
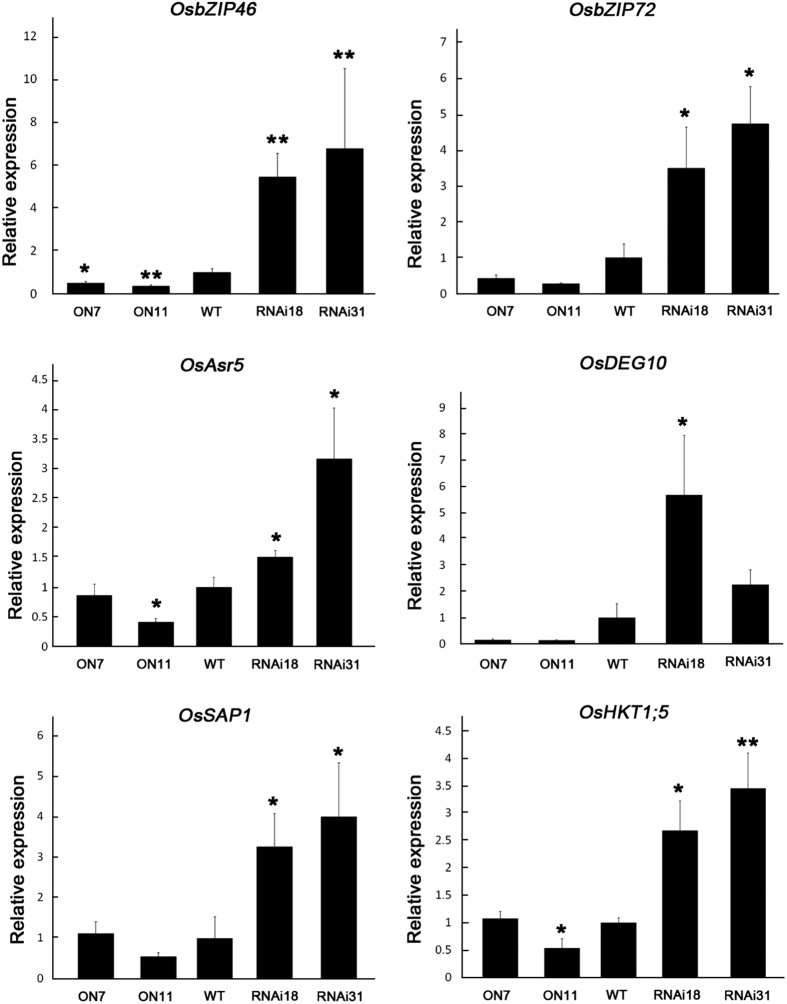
Based on previous reports and our data40,41, we examined two other ABA signalling pathway genes to confirm the relationship between OsNAC2 and ABA signalling (Fig. 8). OsbZIP46 and OsbZIP72 are two members of the third subfamily of bZIP transcription factors in rice, which belong to the bZIP TF family and act downstream of ABA42. OsbZIP46 and OsbZIP72 are positive regulators of ABA signaling and drought stress tolerance of rice40,41,43. The higher transcription of these two genes in RNAi lines matches our previous result that RNAi lines had higher stress resistance. Besides, more stress-related genes are checked. OsAsr5, OsDEG10, OsSAP1 and HKT1;5 are stress marker genes, which responds to ABA44,45, and participates in stress signaling46, stress tolerance47 and Na+ exclusion48. Those genes all showed up-regulated transcription in RNAi lines and might confer stress tolerance in RNAi lines. In conclusion, overexpression of OsNAC2 brings two major differences, one is altered ABA-signaling pathway, and the other is more severe stress phenotypes.
Figure 8. Expression analysis of stress-related and ABA signaling pathway genes in WT, OsNAC2-overexpressing plants and RNAi plants.
The materials grew under normal conditions, 28 °C, 16 h light and 8 h dark. RNA was extracted from whole seedlings. Data were means ± SE with at least three biological replicates. Asterisks represent statistically significant differences between WT and transgenic plants. *P < 0.05, **P < 0.001, ***P < 0.001.
Expression of the stress marker gene OsLEA3 and SnRK2 protein kinase gene OsSAPK1 are regulated by direct binding of OsNAC2 to their promoters
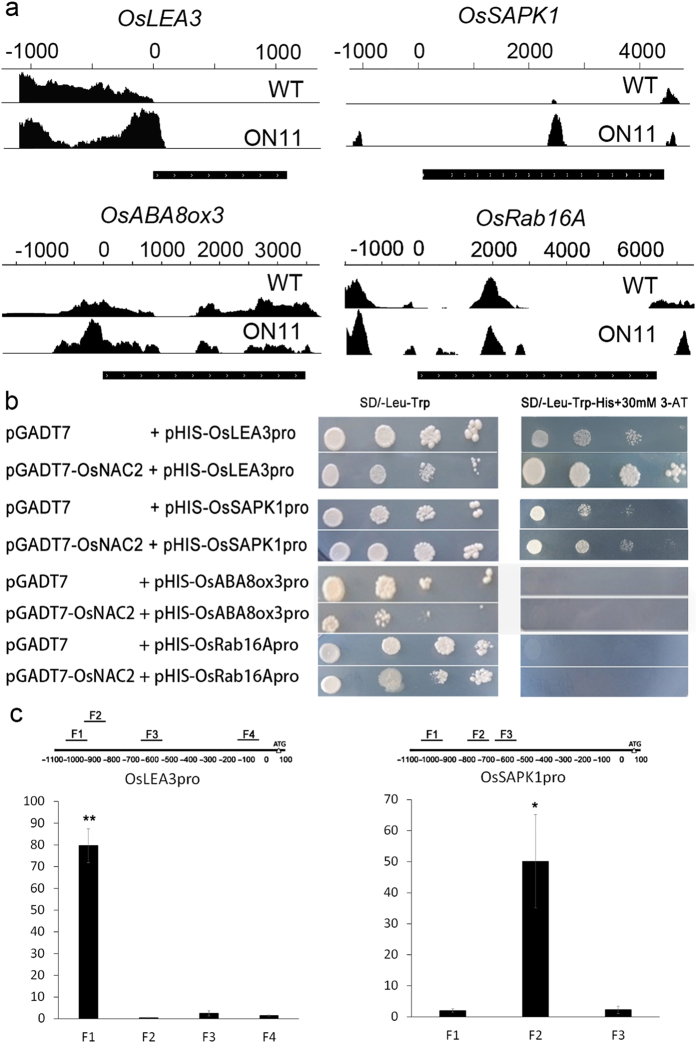
We have shown that overexpression of OsNAC2 reduced plant tolerances to drought and high salinity stress. To identify the target genes of OsNAC2, we performed ChIP-seq where we scanned the promoter region of several stress-related and ABA-pathway genes. We found binding peaks in four of the candidate genes: OsLEA3 (0 bp~−200bp and −900bp~−1100bp), OsSAPK1 (−900bp~−1100bp), OsABA8ox3 (0 bp~−200bp), and OsRAB16A (−100~−250bp) (Fig. 9a). To further confirm whether these genes are direct targets of OsNAC2 we performed a yeast one-hybrid assay and ChIP-qPCR. Here we use OsNAC2 overexpression line 11 (ON11) and WT (as negative control) for this assay. In transgenic line ON11, a GFP-tag was fused with OsNAC2 CDS, which was promoted by 35 S. In the yeast one-hybrid assay, the coding region of the full-length OsNAC2 cDNA was fused in frame to the GAL4 activation domain of the pGADT7 vector. Promoter sequence regions of OsLEA3 (1063 bp), OsSAPK1 (1080 bp), OsABA8OX3 (1090 bp) and OsRab16A (1090 bp) were ligated into the pHIS vector. The yeast one-hybrid assays suggested that OsNAC2 directly interacts with the promoter sequences of OsLEA3 and OsSAPK1 (Fig. 9b).
Figure 9. OsLEA3 and OsSAPK1 are the direct target genes of OsNAC2.
(a) Binding peaks of OsLEA3, OsSAPK1, OsABA8ox3 and OsRab16A in ChIP-seq assay. Black peaks represent for sequence hits on DNA of each gene regions. Higher the peak is, more binding in this region. The bars above the peak show the distance from ATG start codon of each gene. The black bar under the peak represents for the coding area of each gene, arrows on the bar show the coding direction of the gene. (b) Yeast one-hybrid assays showed that OsNAC2 Only binded to the promoter of OsLEA3 and OsSAPK1. (c) ChIP-PCR assays. Total protein extracts from 35 S:OsNAC2–mGFP transgenic plants grown on MS-agar for 2 weeks were immunoprecipitated with an anti-GFP antibody. Fragmented genomic DNA was eluted from the protein–DNA complexes and subjected to qPCR analysis. The long black bars represent for promoter region which we designed primers for. The numbers under the bar show the distance from ATG start codon. Short bars represents for the corresponding region of each pair of primers on the promoter. Error bars are the standard error (SE) for three biological repeats, *P < 0.05, **P < 0.001.
To test whether OsNAC2 specifically bound to its target genes, we performed ChIP-PCR experiments using anti-green fluorescent protein antibodies. We analysed the chromatin immunoprecipitated DNA for specific enrichment of the OsNAC2 target genes mentioned above. The specificity of the ChIP data was demonstrated using negative control DNA derived from WT plants (Fig. 9c). Together, these three data confirmed that OsNAC2 associates specifically with its target genes OsLEA3 (−950bp~−1100bp) and OsSAPK1 (−700~−850bp).
Discussion
NAC family members function widely in plant growth and development processes, especially in relation to abiotic and biotic stress49,50,51. Most NAC genes have been reported to work as positive stress response TFs. For instance, SNAC1 can greatly increase the drought and high salinity tolerance of rice by decreasing the transpiration rate28. OsNAC5 and OsNAC6 can directly bind to the promoter of OsLEA3 and significantly promote high salt and drought tolerance29,30,31 while overexpression of OsNAC10 increases rice yield under drought conditions in the flowering stage32.
In our research, OsNAC2 expression was shown to be strongly induced by ABA and several abiotic stressors. The function of OsNAC2 went exactly opposite to other reported NAC members like OsNAC5, OsNAC6 and OsNAC10. We inferred that OsNAC2 might have new features and pathways in the regulation of abiotic stress in rice, which could enrich the biological functions and regulatory mechanisms of NAC family.
The heading stage, which can also be called flowering stage, is the most critical period in rice agricultural production. During this period rice plants are most sensitive to drought and grain yield is more severely affected by drought stress than at any other stage52. We performed drought stress experiments at the flowering stage in WT and transgenic plants to determine whether OsNAC2 influenced grain yield under drought conditions. The grain yield of WT and transgenic plants did not differ under normal cultural conditions. After drought stress at the flowering stage, however, RNAi plants showed a significantly higher seed setting rate. In terms of 1000-grain weight, RNAi lines also showed significantly higher grain yields than WT lines. In summary, various agricultural traits, such as the seed-setting rate and 1000-grain weight of RNAi lines were significantly higher than that in WT lines after drought treatment at the flowering stage. SNAC128, OsNAC529, and OsNAC1032 enhanced drought resistance in transgenic rice plants at the reproductive stage without affecting yield. Consistent with other NAC family members, therefore, we believe that OsNAC2 represents a practical means of guiding rice agricultural production in drought or salt stress conditions.
Compared with WT plants, we found that many TF and stress-related genes were down-regulated in ON11 lines in our rice gene microarray chip. The expression levels of some of these genes were checked using qRT-PCR and this confirmed that all of these genes were down-regulated in ON11 lines. Many assays have reported that NAC family members can regulate stress-related gene expression resulting in transgenic rice plants with altered tolerance under different adverse conditions. ONAC04549 and OsNAC529 can directly bind to the promoter region of OsLEA3. The SNAC1 promoter contained two DREs, and ABA-responsive elements could be identified in the OsNAC6 promoter49. Therefore, we hypothesise that OsNAC2 may down-regulate these stress genes in response to abiotic stress.
Notably, most of the marker genes we tested were ABA-responsive genes. Some are ABA signalling pathway genes, such as OsbZIP4640 and OsbZIP7241. OsbZIP46, reported to be involved in stress resistance40, was phosphorylated by OsSAPK2, OsSAPK6, and OsSAPK9. Other ABA-responsive genes examined here are stress related genes that are up-regulated by ABA including OsASR545, OsDEG1046, OsSAP147 and OsHKT1;548. We believe that OsNAC2 regulates abiotic stress through an ABA-dependent pathway by acting as a negative regulator of ABA and stress responsive genes. Additionally, we found that OsNAC2 can directly bind to the promoters of OsLEA3 and OsSAPK1 (Fig. 9) and down-regulate their expression (Fig. 7b,c). OsLEA3 is a widely known stress marker gene, overexpression of which confers plant stress resistance29. OsSAPK1 belongs to the SnRK2 family that can be upregulated by osmotic stressors such as salt and mannitol53. Here, we found that OsSAPK1 expression can be directly regulated by OsNAC2 in response to abiotic stressors like drought and high salinity in rice.
To further investigate the biological function of OsNAC2, we constructed OsNAC2-overexpressing and RNAi lines. During cultivation we found several different phenotypes in the overexpression lines including shorter roots, shorter shoot lengths38 and premature leaf senescence (data not shown). This is consistent with evidence that plant tolerance to abiotic stress is often closely associated with growth and development. Root-specific overexpression of OsNAC10 enlarges roots, enhancing the drought tolerance of transgenic plants and increasing their grain yield significantly under field drought conditions32. Conversely, OsNAC5 can improve the stress tolerance of rice without affecting its growth29. Our results show that OsNAC2 functions through the ABA-dependent pathway and may down-regulate many ABA-responsive stress marker genes. Previous work in our lab on OsNAC2 has indicated that it functions in plant height development and potentially in root development (data not published), and in senescence (data not published). Combined with the results of this study, OsNAC2 may be a powerful TF that serves as a node to link multiple pathways together.
In conclusion, we found that the expression of OsNAC2 was induced by osmotic stress and ABA. Overexpression of OsNAC2 resulted in transgenic rice plants that were sensitive to high salinity and drought stress at different growth stages. OsNAC2 directly down-regulated the stress-related marker gene OsLEA3 and SnRK2 family gene OsSAPK1 via the ABA-dependent pathway. Future work is required to determine the specific position of OsNAC2 in the underlying regulatory network in more detail.
Methods
Plants, strain and plasmid
Wild type rice (Oryza sativa) which have an ecotype of Nipponbare, were saved in our lab. We constructed a vector in which 1500 bp full length promoter of OsNAC2 is connected with GUS (Supplementary data Fig. 1). Overexpression and RNAi transgenic lines were constructed before and the details is in Chen et al.38. pCR-Blunt, pCAMBIA-1304 (containing a CaMV 35 S promoter), pCAMBIA1300 (fused with GUS reporter gene), RNAi vector, Escherichia coli strain DH5αand Agrobacterium strain EHA105 were all saved in our lab.
Quantitative RT-PCR analysis
Total RNAs were extracted with TRIzol (TAKARA) from rice seedlings. All rice seedlings were grown under normal conditions of a 16 h light/8 h dark cycle at 28 °C for two weeks. The total RNAs were reverse-transcribed into first-strand cDNA using reverse-transcription enzyme. qRT-PCR was performed in a 10 μL reaction with SYBR (Perfect Real Time code: DRR041 TaKaRa) 5 μL, 10 mmol/L PCR forward primer 0.4 μL, 10 mmol/L PCR reverse primer 0.4 μL, template cDNA 4.2 μL. The reaction used iCycleriQTM real-time quantitative PCR detection system (Bio-RAD). Each sample was repeated 3 times for qRT -PCR detection. We determined the linear range of the target gene and actin gene by detecting standard curve in each experiment. Osactin was used as an inner control. The relative expression levels of the gene were calculated by 2−ΔΔCt analysis. Sequences of primer pairs are listed in supplemental data.
Detection of histochemical GUS activity
Two-week-old rice seedlings were treated with 150 mM NaCl for 1, 2, 3, 4 and 5 days and with air dry condition for 0 h to 2 days. Tissues were then immersed in X-gluc solution in 37 °C overnight. Then the materials were soaked with 70% ethanol to remove chlorophyll. Wild type rice seedlings and tissues were used as normal control.
Stress treatments of rice seedlings
For vegetative tolerance experiment, all rice seedlings were grown in basal nutrient solution and normal conditions, such as 28 °C, 16 h light and 8 h dark and then two-week-old seedlings were transferred into nutrient solution containing 150 mM NaCl for 2.5d or 20% PEG8000 for 5d and recovery for 2d and 3d respectively. For soil experiments, rice seedlings were grown under normal conditions for four weeks, and then irrigated with 150 mM NaCl solution for 14d or no water for 14d with 7d recovery.
Physiological index measurement in high salt and drought tolerance experiments
The withered or survival rate, fresh weight, chlorophyll content and plant root or shoot length we mentioned were measured after stress or recovery. In withered or survival rate statistics, seedlings with all leaves yellow and wilted were calculated. DAB staining were performed after NaCl and PEG stresses. Rice leaves were immersed in DAB staining solution in 25 °C for 24 h, and then soaked with 95% ethanol to remove chlorophyll. Wild type rice seedlings and tissues were used as normal control. Electrolyte leakage was measured as described54. The Fv/Fm values were determined using LI-6400XT portable photosynthesis system following with instructions. Rice leaves were immersed in the extract solution (70% acetone + 20% ethanol + 10% water) at 4 °C until the leaves were bleached. Chlorophyll content was detected using 721 visible light spectrophotometer. Agronomic traits were not counted until rice seeds turn mature.
Yeast one-hybrid
For yeast one-hybrid assays, the coding sequence of OsNAC2 was inserted into EcoR I-Xho I site of pGADT7 vector to generate a construct with activation domain and OsNAC2. The promoter sequence of OsLEA3 (1063 bp), OsSAPK1 (1080 bp), OsABA8OX3 (1090 bp) and OsRab16A (1090 bp) genes was inserted into pHIS2.1 vector through EcoR I-Sma I, EcoR I-Mlu I, EcoR I- Mlu I, EcoR I- Mlu I sites to generate an in-frame fusion with minHIS3. All primers used for cloning these constructs are listed in Table S2. These vectors and empty vector were transformed into yeast strain AH109 by the PEG/LiAc method. Yeast cells were plated onto SD/-His/-Trp/-Leu + 30 mM 3AT medium for stringent screening of the possible interactions, according to the protocol of Matchmaker GAL4 One-Hybrid System (Clontech, www.clontech.com).
ChIP(Chromatin immunoprecipitation)-PCR and ChIP-seq
ChIP assay was performed based on the previous report55 with two-week ON11 transgenic seedlings, in which a mGFP coding sequence was fused in frame to the 3′ end of the OsNAC2 gene in transgenic line, and the expression is driven by 35 S promoter. Because WT doesn’t have GFP, we use it as negative control to erase the background noise. Two-week-old seedlings grown in basal nutrient solution were treated for extraction of total proteins. The OsNAC2 protein was immunoprecipitated using an antibody against GFP. The DNA fragments of the ChIP were then used for quantitative PCR or sequencing. The ChIP-PCR experiments were repeated three times with the similar data. Primer pairs for qRT-PCR were listed in Table S2. For ChIP-seq, libraries were generated using Ovation Ultralow Library System 2 (Nugene) following manufacturer’s standard protocols. Sequencing was performed on a HiSeq2500 (Illumina) using pair end 100 bp mode.
Additional Information
How to cite this article: Shen, J. et al. The NAC-type transcription factor OsNAC2 regulates ABA-dependent genes and abiotic stress tolerance in rice. Sci. Rep. 7, 40641; doi: 10.1038/srep40641 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Material
Acknowledgments
This work was supported by the Ministry of Agriculture of China [grant No. 2016ZX08009-001-008]; The National Natural Science Foundation of China [grant No. 31471152]. The Research Fund of the State Key Laboratory of Genetic Engineering, Fudan University.
Footnotes
Author Contributions B.L., J.S. and F.M. designed the experiments and wrote the manuscript. L.L. and J.H. performed OsNAC2 expression profile assays. B.L. and L.L. performed stress assays and Microarray analysis. J.S. performed stress genes expression level analysis. J.S. and J.H. performed ChIP assays. J.H. and C.M. performed Yeast-one-hybrid assays. D.X. provided advice on the experiments.
References
- Umezawa T., Fujita M., Fujita Y., Yamaguchi-Shinozaki K. & Shinozaki K. Engineering drought tolerance in plants: discovering and tailoring genes to unlock the future. Current opinion in biotechnology 17, 113–122, doi: 10.1016/j.copbio.2006.02.002 (2006). [DOI] [PubMed] [Google Scholar]
- Bu Q. et al. Role of the Arabidopsis thaliana NAC transcription factors ANAC019 and ANAC055 in regulating jasmonic acid-signaled defense responses. Cell research 18, 756–767, doi: 10.1038/cr.2008.53 (2008). [DOI] [PubMed] [Google Scholar]
- Jiang H., Li H., Bu Q. & Li C. The RHA2a-interacting proteins ANAC019 and ANAC055 may play a dual role in regulating ABA response and jasmonate response. Plant signaling & behavior 4, 464–466, doi: 10.1104/pp.109.135269 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Puranik S., Sahu P. P., Srivastava P. S. & Prasad M. NAC proteins: regulation and role in stress tolerance. Trends in plant science 17, 369–381, doi: 10.1016/j.tplants.2012.02.004 (2012). [DOI] [PubMed] [Google Scholar]
- Souer E., van Houwelingen A., Kloos D., Mol J. & Koes R. The no apical meristem gene of Petunia is required for pattern formation in embryos and flowers and is expressed at meristem and primordia boundaries. Cell 85, 159–170 (1996). [DOI] [PubMed] [Google Scholar]
- Aida M., Ishida T., Fukaki H., Fujisawa H. & Tasaka M. Genes involved in organ separation in Arabidopsis: an analysis of the cup-shaped cotyledon mutant. The Plant cell 9, 841–857, doi: 10.1105/tpc.9.6.841 (1997). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nuruzzaman M. et al. Genome-wide analysis of NAC transcription factor family in rice. Gene 465, 30–44, doi: 10.1016/j.gene.2010.06.008 (2010). [DOI] [PubMed] [Google Scholar]
- Yamasaki K. et al. Solution structure of an Arabidopsis WRKY DNA binding domain. The Plant cell 17, 944–956, doi: 10.1105/tpc.104.026435 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamasaki K. et al. Structures and evolutionary origins of plant-specific transcription factor DNA-binding domains. Plant physiology and biochemistry: PPB/Societe francaise de physiologie vegetale 46, 394–401, doi: 10.1016/j.plaphy.2007.12.015 (2008). [DOI] [PubMed] [Google Scholar]
- Ooka H. et al. Comprehensive analysis of NAC family genes in Oryza sativa and Arabidopsis thaliana. DNA research: an international journal for rapid publication of reports on genes and genomes 10, 239–247 (2003). [DOI] [PubMed] [Google Scholar]
- Takada S., Hibara K., Ishida T. & Tasaka M. The Cup-Shaped Cotyledon1 gene of Arabidopsis regulates shoot apical meristem formation. Development 128, 1127–1135 (2001). [DOI] [PubMed] [Google Scholar]
- Raman S. et al. Interplay of miR164, Cup-Shaped Cotyledon genes and Lateral Suppressor controls axillary meristem formation in Arabidopsis thaliana. The Plant journal: for cell and molecular biology 55, 65–76, doi: 10.1111/j.1365-313X.2008.03483.x (2008). [DOI] [PubMed] [Google Scholar]
- Mao C. et al. Overexpression of a NAC-domain protein promotes shoot branching in rice. The New phytologist 176, 288–298, doi: 10.1111/j.1469-8137.2007.02177.x (2007). [DOI] [PubMed] [Google Scholar]
- Xie Q., Frugis G., Colgan D. & Chua N. H. Arabidopsis NAC1 transduces auxin signal downstream of TIR1 to promote lateral root development. Genes & development 14, 3024–3036 (2000). [DOI] [PMC free article] [PubMed] [Google Scholar]
- He X. J. et al. AtNAC2, a transcription factor downstream of ethylene and auxin signaling pathways, is involved in salt stress response and lateral root development. The Plant journal: for cell and molecular biology 44, 903–916, doi: 10.1111/j.1365-313X.2005.02575.x (2005). [DOI] [PubMed] [Google Scholar]
- Mitsuda N., Seki M., Shinozaki K. & Ohme-Takagi M. The NAC transcription factors NST1 and NST2 of Arabidopsis regulate secondary wall thickenings and are required for anther dehiscence. The Plant cell 17, 2993–3006, doi: 10.1105/tpc.105.036004 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhong R., Richardson E. A. & Ye Z. H. The MYB46 transcription factor is a direct target of SND1 and regulates secondary wall biosynthesis in Arabidopsis. The Plant cell 19, 2776–2792, doi: 10.1105/tpc.107.053678 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitsuda N. & Ohme-Takagi M. NAC transcription factors NST1 and NST3 regulate pod shattering in a partially redundant manner by promoting secondary wall formation after the establishment of tissue identity. The Plant journal: for cell and molecular biology 56, 768–778, doi: 10.1111/j.1365-313X.2008.03633.x (2008). [DOI] [PubMed] [Google Scholar]
- Zhong R., Demura T. & Ye Z. H. SND1, a NAC domain transcription factor, is a key regulator of secondary wall synthesis in fibers of Arabidopsis. The Plant cell 18, 3158–3170, doi: 10.1105/tpc.106.047399 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kubo M. et al. Transcription switches for protoxylem and metaxylem vessel formation. Genes & development 19, 1855–1860, doi: 10.1101/gad.1331305 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim Y. S. et al. A membrane-bound NAC transcription factor regulates cell division in Arabidopsis. The Plant cell 18, 3132–3144, doi: 10.1105/tpc.106.043018 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sablowski R. W. & Meyerowitz E. M. Temperature-sensitive splicing in the floral homeotic mutant apetala3-1. The Plant cell 10, 1453–1463 (1998). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo Y. & Gan S. AtNAP, a NAC family transcription factor, has an important role in leaf senescence. The Plant journal: for cell and molecular biology 46, 601–612, doi: 10.1111/j.1365-313X.2006.02723.x (2006). [DOI] [PubMed] [Google Scholar]
- Kim J. H. et al. Trifurcate feed-forward regulation of age-dependent cell death involving miR164 in Arabidopsis. Science 323, 1053–1057, doi: 10.1126/science.1166386 (2009). [DOI] [PubMed] [Google Scholar]
- Balazadeh S. et al. ORS1, an H(2)O(2)-responsive NAC transcription factor, controls senescence in Arabidopsis thaliana. Molecular plant 4, 346–360, doi: 10.1093/mp/ssq080 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoon H. K., Kim S. G., Kim S. Y. & Park C. M. Regulation of leaf senescence by NTL9-mediated osmotic stress signaling in Arabidopsis. Molecules and cells 25, 438–445 (2008). [PubMed] [Google Scholar]
- Gao J. P., Chao D. Y. & Lin H. X. Toward Understanding Molecular Mechanisms of Abiotic Stress Responses in Rice. Rice 1, 36–51 (2008). [Google Scholar]
- Hu H. et al. Overexpressing a NAM, ATAF, and CUC (NAC) transcription factor enhances drought resistance and salt tolerance in rice. Proceedings of the National Academy of Sciences of the United States of America 103, 12987–12992, doi: 10.1073/pnas.0604882103 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takasaki H. et al. The abiotic stress-responsive NAC-type transcription factor OsNAC5 regulates stress-inducible genes and stress tolerance in rice. Molecular genetics and genomics: MGG 284, 173–183, doi: 10.1007/s00438-010-0557-0 (2010). [DOI] [PubMed] [Google Scholar]
- Nakashima K. et al. Functional analysis of a NAC-type transcription factor OsNAC6 involved in abiotic and biotic stress-responsive gene expression in rice. The Plant journal: for cell and molecular biology 51, 617–630, doi: 10.1111/j.1365-313X.2007.03168.x (2007). [DOI] [PubMed] [Google Scholar]
- Hu H. et al. Characterization of transcription factor gene SNAC2 conferring cold and salt tolerance in rice. Plant molecular biology 67, 169–181, doi: 10.1007/s11103-008-9309-5 (2008). [DOI] [PubMed] [Google Scholar]
- Jin S. J. & Kim J. K. Root-Specific Expression of OsNAC10 Improves Drought Tolerance and Grain Yield in Rice under Field Drought Conditions. Plant physiology 153, 185–197 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tran L. S. et al. Isolation and functional analysis of Arabidopsis stress-inducible NAC transcription factors that bind to a drought-responsive cis-element in the early responsive to dehydration stress 1 promoter. The Plant cell 16, 2481–2498, doi: 10.1105/tpc.104.022699 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yokotani N. et al. Tolerance to various environmental stresses conferred by the salt-responsive rice gene ONAC063 in transgenic Arabidopsis. Planta 229, 1065–1075, doi: 10.1007/s00425-009-0895-5 (2009). [DOI] [PubMed] [Google Scholar]
- Lu P. L. et al. A novel drought-inducible gene, ATAF1, encodes a NAC family protein that negatively regulates the expression of stress-responsive genes in Arabidopsis. Plant molecular biology 63, 289–305, doi: 10.1007/s11103-006-9089-8 (2007). [DOI] [PubMed] [Google Scholar]
- Yoo S. Y., Kim Y., Kim S. Y., Lee J. S. & Ahn J. H. Control of flowering time and cold response by a NAC-domain protein in Arabidopsis. PloS one 2, e642, doi: 10.1371/journal.pone.0000642 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christianson J. A., Wilson I. W., Llewellyn D. J. & Dennis E. S. The low-oxygen-induced NAC domain transcription factor ANAC102 affects viability of Arabidopsis seeds following low-oxygen treatment. Plant physiology 149, 1724–1738, doi: 10.1104/pp.108.131912 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen X. et al. OsNAC2 encoding a NAC transcription factor that affects plant height through mediating the gibberellic acid pathway in rice. The Plant journal: for cell and molecular biology 82, 302–314, doi: 10.1111/tpj.12819 (2015). [DOI] [PubMed] [Google Scholar]
- Hakim M. A. et al. Effect of salt stress on morpho-physiology, vegetative growth and yield of rice. Journal of environmental biology 35, 317–326 (2014). [PubMed] [Google Scholar]
- Tang N., Zhang H., Li X., Xiao J. & Xiong L. Constitutive activation of transcription factor OsbZIP46 improves drought tolerance in rice. Plant physiology 158, 1755–1768, doi: 10.1104/pp.111.190389 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu G., Gao C., Zheng X. & Han B. Identification of OsbZIP72 as a positive regulator of ABA response and drought tolerance in rice. Planta 229, 605–615, doi: 10.1007/s00425-008-0857-3 (2009). [DOI] [PubMed] [Google Scholar]
- Hossain M. A. et al. The ABRE-binding bZIP transcription factor OsABF2 is a positive regulator of abiotic stress and ABA signaling in rice. Journal of plant physiology 167, 1512–1520, doi: 10.1016/j.jplph.2010.05.008 (2010). [DOI] [PubMed] [Google Scholar]
- Xiang Y., Tang N., Du H., Ye H. & Xiong L. Characterization of OsbZIP23 as a key player of the basic leucine zipper transcription factor family for conferring abscisic acid sensitivity and salinity and drought tolerance in rice. Plant physiology 148, 1938–1952, doi: 10.1104/pp.108.128199 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takasaki H., Mahmood T., Matsuoka M., Matsumoto H. & Komatsu S. Identification and characterization of a gibberellin-regulated protein, which is ASR5, in the basal region of rice leaf sheaths. Molecular genetics and genomics: MGG 279, 359–370, doi: 10.1007/s00438-007-0317-y (2008). [DOI] [PubMed] [Google Scholar]
- Perez-Diaz J. et al. Organ- and stress-specific expression of the ASR genes in rice. Plant cell reports 33, 61–73, doi: 10.1007/s00299-013-1512-4 (2014). [DOI] [PubMed] [Google Scholar]
- Park H. Y. et al. OsDEG10 encoding a small RNA-binding protein is involved in abiotic stress signaling. Biochemical and biophysical research communications 380, 597–602, doi: 10.1016/j.bbrc.2009.01.131 (2009). [DOI] [PubMed] [Google Scholar]
- Giri J., Vij S., Dansana P. K. & Tyagi A. K. Rice A20/AN1 zinc-finger containing stress-associated proteins (SAP1/11) and a receptor-like cytoplasmic kinase (OsRLCK253) interact via A20 zinc-finger and confer abiotic stress tolerance in transgenic Arabidopsis plants. The New phytologist 191, 721–732, doi: 10.1111/j.1469-8137.2011.03740.x (2011). [DOI] [PubMed] [Google Scholar]
- Ren Z. H. et al. A rice quantitative trait locus for salt tolerance encodes a sodium transporter. Nature genetics 37, 1141–1146, doi: 10.1038/ng1643 (2005). [DOI] [PubMed] [Google Scholar]
- Nakashima K., Takasaki H., Mizoi J., Shinozaki K. & Yamaguchi-Shinozaki K. NAC transcription factors in plant abiotic stress responses. Biochimica et biophysica acta 1819, 97–103, doi: 10.1016/j.bbagrm.2011.10.005 (2012). [DOI] [PubMed] [Google Scholar]
- Wang X. et al. The Arabidopsis ATAF1, a NAC transcription factor, is a negative regulator of defense responses against necrotrophic fungal and bacterial pathogens. Molecular plant-microbe interactions: MPMI 22, 1227–1238, doi: 10.1094/MPMI-22-10-1227 (2009). [DOI] [PubMed] [Google Scholar]
- Zheng X., Chen B., Lu G. & Han B. Overexpression of a NAC transcription factor enhances rice drought and salt tolerance. Biochemical and biophysical research communications 379, 985–989, doi: 10.1016/j.bbrc.2008.12.163 (2009). [DOI] [PubMed] [Google Scholar]
- Lafitte H. R. The Regional Institute - Abiotic stress tolerance in rice for Asia: progress and the future. Molecular Breeding (2004).
- Kobayashi Y., Yamamoto S., Minami H., Kagaya Y. & Hattori T. Differential activation of the rice sucrose nonfermenting1-related protein kinase2 family by hyperosmotic stress and abscisic acid. The Plant cell 16, 1163–1177, doi: 10.1105/tpc.019943 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song S. Y., Chen Y., Chen J., Dai X. Y. & Zhang W. H. Physiological mechanisms underlying OsNAC5-dependent tolerance of rice plants to abiotic stress. Planta 234, 331–345, doi: 10.1007/s00425-011-1403-2 (2011). [DOI] [PubMed] [Google Scholar]
- Gendrel A. V., Lippman Z., Martienssen R. & Colot V. Profiling histone modification patterns in plants using genomic tiling microarrays. Nature Methods 2, 213–218 (2005). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.