Abstract
Aim
To predict arrhythmia susceptibility in myocardial infarction (MI) patients with left ventricular ejection fraction (LVEF) >35% using a personalized virtual heart simulation approach.
Methods and results
A total of four contrast enhanced magnetic resonance imaging (MRI) datasets of patient hearts with MI and average LVEF of 44.0 ± 2.6% were used in this study. Because of the preserved LVEF, the patients were not indicated for implantable cardioverter defibrillator (ICD) insertion. One patient had spontaneous ventricular tachycardia (VT) prior to the MRI scan; the others had no arrhythmic events. Simulations of arrhythmia susceptibility were blind to clinical outcome. Models were constructed from patient MRI images segmented to identify myocardium, grey zone, and scar based on pixel intensity. Grey zone was modelled as having altered electrophysiology. Programmed electrical stimulation (PES) was performed to assess VT inducibility from 19 bi-ventricular sites in each heart model. Simulations successfully predicted arrhythmia risk in all four patients. For the patient with arrhythmic event, in-silico PES resulted in VT induction. Simulations correctly predicted that VT was non-inducible for the three patients with no recorded VT events.
Conclusions
Results demonstrate that the personalized virtual heart simulation approach may provide a novel risk stratification modality to non-invasively and effectively identify patients with LVEF >35% who could benefit from ICD implantation.
Keywords: Personalized virtual heart , Ejection fraction >35%, Ventricular tachycardia , Myocardial infarction , Computational modelling
What’s new?
Using computational modelling to non-invasively predict arrhythmia risk in myocardial infarction patients with preserved left ventricular ejection fraction (LVEF).
The methodology used in our simulation is totally automatic, is reproducible and can be applied to image stacks obtained from different centres.
Our model can accurately detect VT arrhythmia susceptibility in patients with LVEF >35% who do not have implantable cardioverter defibrillators (ICDs) implanted. Thus, our technique may help clinicians identify patients that could benefit from ICD implantation.
Introduction
Sudden cardiac death (SCD) is a leading health problem worldwide, accounting for over 300 000 deaths per year in the USA alone.1,2 A large proportion of SCDs result from ventricular arrhythmia, particularly amongst patients with prior heart damage from myocardial infarction (MI). Implantable cardioverter defibrillators (ICDs) are the first line of therapy for preventing SCD in high-risk individuals.3 Optimal deployment of this expensive resource remains a major clinical challenge because of the lack of robust strategies for identifying patients that would and would not benefit from this lifesaving therapy.4 Current clinical criteria for selecting patients for ICDs rely on a ‘one-size-fits-all’ metric representing global reduction in heart pumping function: left ventricular ejection fraction (LVEF) <35%.5 This single metric poorly reflects the mechanisms underlying cardiac arrhythmias in MI and hence, its use as a risk stratifier results in a low rate of appropriate ICD device therapy, only 5%/year.3 Thus, many patients are exposed to ICD risks (e.g. infections, device malfunctions, and inappropriate shocks), without deriving any health benefit.6–8 Importantly, the low LVEF criterion only targets a relatively small subgroup of individuals at risk for SCD, failing to identify the majority of SCD victims. Thus, inadequate SCD risk stratification poses a large socioeconomic burden.9 Despite extensive research on risk prediction strategies, none have proved sufficiently discriminative for use in general clinical practice.10 Given the complex pathophysiology of SCD and MI, a strategy is needed that incorporates the underlying mechanisms, namely the myocardial arrhythmogenic substrate, in an individualized way. A robust, readily available, non-invasive, personalized arrhythmia risk predictor will not only identify individual patients in need of lifesaving therapy, but will reduce unnecessary ICD implantation and the associated complications.
In a recent study in Nature Communications,11 we presented a novel, non-invasive, and accurate test for SCD risk stratification in MI patients with LVEF <35%, which we termed the virtual-heart arrhythmia risk predictor (VARP). The VARP approach is based on personalized magnetic resonance imaging (MRI)-based computational modelling of the patient heart that non-invasively assesses the individual’s susceptibility to lethal ventricular arrhythmias. In that study, we demonstrated VARP’s predictive capability using data from 41 patients with LVEF <35%, and compared it to that of other clinical metrics. The results demonstrated that VARP significantly outperformed clinical metrics in predicting future arrhythmic events.
This feasibility study aims to extend the VARP approach to the arrhythmia risk stratification of patients with MI and LVEF >35%, for whom prophylactic ICD implantation is not clinically indicated. The goal here is to demonstrate that the approach can be applied to such patients, and to compare the VARP prediction for this small cohort with clinical outcomes.
Methods
Study population
For this retrospective study, we used data from four patients who were diagnosed with ischaemic cardiomyopathy between 2008 and 2013 at the University of Pennsylvania Hospital. Table 1 summarizes the baseline characteristics for the four patients. The patients had mean ± standard deviation (SD) LVEF of 44.0 ± 2.6% and were thus not clinically indicated for prophylactic ICD implantation. All patients underwent MRI performed on a 1.5 T scanner using post-gadolinium perfusion and inversion-recovery-prepared delayed enhancement (1.77×1.77×8 mm3 resolution). One of the patients had spontaneous stable monomorphic ventricular tachycardia (VT) prior to the MRI. The other three patients had no spontaneous or inducible arrhythmia pre- or post-MRI.
Table 1.
Baseline characteristics of patients and the normal, grey zone, and scar volumes in the reconstructed heart models
| Patient | Age(y) | Sex | EF (%) | Clinical VT | Tissue classification | Reconstructed volume (cm3) | % of total ventricular volume |
|---|---|---|---|---|---|---|---|
| 1 | 38 | F | 47 | Y | Normal | 149.7 | 92.0 |
| Grey zone | 9.9 | 6.1 | |||||
| Scar | 3.1 | 1.9 | |||||
| 2 | 60 | F | 45 | N | Normal | 88.4 | 90.1 |
| Grey zone | 2.4 | 2.4 | |||||
| Scar | 7.3 | 7.5 | |||||
| 3 | 59 | F | 43 | N | Normal | 125.4 | 91.1 |
| Grey zone | 9.7 | 7.1 | |||||
| Scar | 2.5 | 1.8 | |||||
| 4 | 56 | F | 41 | N | Normal | 105.5 | 91.6 |
| Grey zone | 9.3 | 8.1 | |||||
| Scar | 0.3 | 0.3 | |||||
| Mean ± SD | Normal | 117.3 ± 26.4 | 91.2 ± 0.8 | ||||
| Grey zone | 7.8 ± 3.6 | 5.9 ± 2.5 | |||||
| Scar | 3.3 ± 2.9 | 2.9 ± 3.2 | |||||
| Total | 128.4 ± 28.0 |
Image processing
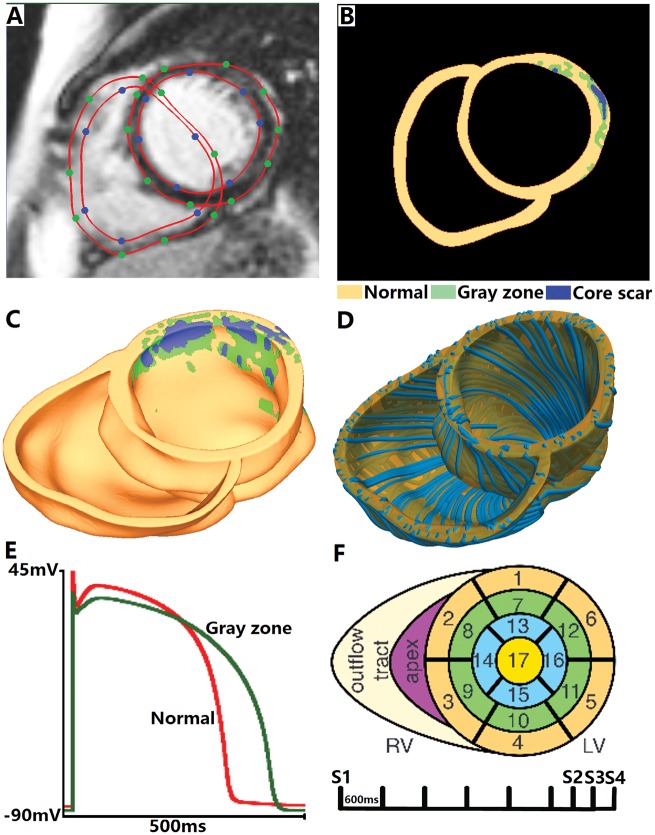
The MRI image stacks were segmented using our recently published segmentation and interpolation method,12,13 an updated methodology in comparison to what was previously used in the VARP approach.11 CardioViz3D (INRIA, Sophia-Antipolis, France) was used to seed points and obtain the surfaces for the epi- and endo-cardial boundaries of the ventricles (Figure 1A). The grey-level thresholding method described in our previous paper13 was used to further segment the myocardium into three tissue types, non-infarcted tissue, border zone (also termed grey zone, because of its intermediate level of signal intensity in the MRI), and scar. The mean and SD of the voxel intensities in the region outside the infarct were calculated. The grey-level value corresponding to mean + 3 SD was assigned as the upper limit for normal tissue (normalmax), and the range of intensities within the infarct tissue (infarctrange) was calculated by obtaining the difference between the maximum intensity within the infarct region (infarctmax) and normalmax. Voxels with intensity between normalmax and (normalmax + 30% of infarctrange) were classified as grey zone. Voxels with intensity more than normalmax + 30% of infarctrange were classified as scar (Figure 1B).
Figure 1.
Virtual-heart arrhythmia risk predictor methodology. Contrast-enhanced cardiac MRI stack with landmark points and splines delineating the endocardial and epicardial surfaces (A), and the resulting ventricular segmentation into normal tissue, grey zone, and core scar (B). High-resolution ventricular structure model (C) with estimated fibre orientations (D). Action potential (E) for non-infarcted tissue (red) and grey zone (green). Virtual-heart arrhythmia risk predictor pacing sites (F).
Discretization and fibre orientation generation
The tetrahedral meshes were constructed directly from the segmented images using a previously described approach, which uses the dual mesh of an octree applied directly to segmented 3D image stacks.14 The procedure preserved the fine geometric details of the ventricles and the different infarct zones (Figure 1C). Finally, fibre orientations were assigned in the mesh using a previously validated rule-based method (Figure 1D).15 It uses the Laplace–Dirichlet method to define transmural and apicobasal directions at every point in the ventricles, and then employs bi-directional spherical linear interpolation to assign fibre orientations based on a set of fibre orientation properties (rules) derived from a large amount of histological and diffusion tensor MRI data.11
Electrophysiological properties
Assigning electrophysiological (EP) properties in the model was done as previously described.11 Briefly, mathematical description of electrical conduction in cardiac tissue was based on the monodomain representation.16 The scar was modelled as passive tissue. The non-infarcted tissue and grey zone were assigned human ventricular cell action potential dynamics.17 Modifications to the ionic model based on experimental recordings were implemented to represent EP remodelling in the grey zone, as we have previously described (Figure 1E),13 namely: peak sodium current, peak L-type calcium current, peak potassium currents IKr, and IKs were decreased to 38%, 31%, 30% and 20% of the original values in the Ten Tusscher model, respectively. As a result, the grey zone action potential was characterized by a longer duration, decreased upstroke velocity, and decreased peak amplitude compared to that in the non-infarcted myocardium (360 vs. 310 ms, 6.7 vs. 11.6 V/s, and 20 vs. 35 mV, respectively), similar to what has been previously reported.18,19
As implemented previously in VARP,11 the values of the non-infarcted tissue conductivities used in this study were 0.255 and 0.0775 S/m in the longitudinal and transverse directions, respectively. Tissue in the grey zone region was characterized with a 90% decrease in transverse conductivity to reflect connexin-43 remodelling in the infarct border zone.20
Stimulation protocol
All simulations were performed using the software package CARP (CardioSolv, LLC) on a parallel computing platform.16,21 To examine the arrhythmogenic propensity of the post-MI ventricular models, programmed electrical stimulation, similar to the protocol used in the standard clinical protocols,22 was simulated; the same protocol was used in the original VARP study.11 Specifically, each patient-derived ventricular model was subjected to pacing from multiple locations in an attempt to elicit reentrant arrhythmias, thus assessing the potential of the disease-remodelled ventricles to cause degeneration of electrical signal propagation into arrhythmic activity following premature beats that originate at different locations in the heart. The stimuli were delivered at 17 locations in the LV, one in each American Heart Association (AHA) segment,23 and at the apex and near the outflow tract of the right ventricle. The rationale for choosing a large number of pacing sites was based on clinical studies, which have shown a positive correlation between the number of pacing sites and inducibility of ventricular arrhythmia.24 The distribution of pacing sites throughout the LV ensured that the protocol covered a large range of possibilities for potential sites at which ectopic foci could emerge.
All models were paced endocardially from each of the 19 locations for 6 beats (S1) at a cycle length of 600 ms followed by a premature stimulus (S2) initially given at 90% of S1 cycle length. The timing between S1 and S2 was progressively shortened until VT was induced. If VT was not induced, a second premature stimulus (S3) was delivered after S2. If VT was not induced either, a third premature stimulus (S4) was delivered after S3 (Figure 1F). Three seconds of VT were simulated.
Results
Table 1 summarizes the volumes of the normal myocardium, grey zone, and scar in the reconstructed hearts. The mean value of ventricular volumes for the four patients is 128.4 cm3. The normal tissue occupies more than 90% of the ventricular volumes in all the patients, but the fractional volume of grey zone within the infarct ranged from 24.7 to 96.9%.
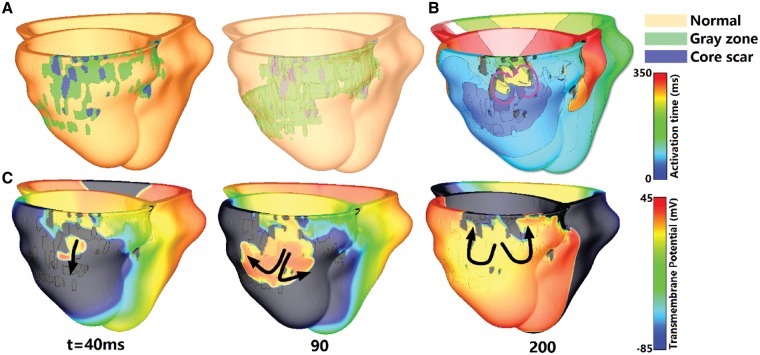
Virtual-heart arrhythmia risk predictor, the execution of which was blinded to clinical outcome, accurately predicted arrhythmia susceptibility in all four patients. Figure 2 summarizes the arrhythmia inducibility simulation results for the only patient that reported clinical VT (Patient 1). In this patient model, the infarcted tissue is located at the inferolateral and posterior area near the base, and the core scar occupies 24% of the infarct volume (Figure 2A). Activation and voltage maps in Figure 2B and 2C, respectively, show that the reentrant circuit in the model is an epicardial figure of eight, with an isthmus located within the inferolateral grey zone at the base. These results are consistent with clinical recordings obtained as part of the EP study performed during the ablation procedure for this patient, which revealed that the reentry conducted through an isthmus located in the lateral and basal scar border zone.
Figure 2.
Geometrical models, which include normal tissue, grey zone, and core scar are presented (A) together with electrical activation isochronal maps (B), with purple arrows representing the direction of propagation of the reentrant arrhythmia. Transmembrane potential maps show the initiation of VT in Patient 1 model (C). Black arrows show direction of propagation. The time instant below each map is counted from the delivery of the last pacing stimulus.
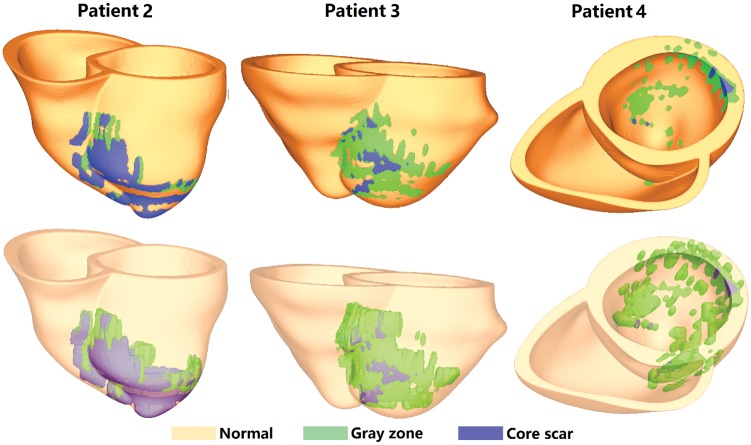
Figure 3 shows the reconstructed geometry for patients 2–4. In the ventricular model of Patient 2, the infarcted tissue is located at the middle anterior and lateral wall of left ventricle, and the core scar occupies 75% of the infarct. In the Patient 3 model, the infarct is located mainly at the arterial and septal wall, with the core scar occupying 20% of it. In Patient 4 model, the infarcted tissue is located at the lateral wall from base to apex, and the core scar occupies only 3% of the infarct. These three patients had no inducible VT as predicted by VARP, which is consistent with the patient’s clinical histories. All of these patients had no recorded case of arrhythmia prior to MRI procedure.
Figure 3.
Geometries of the three personalized heart models in which no arrhythmia was induced.
Discussion
In this article, we present a feasibility study for the use of computational modelling in identifying patients with LVEF >35% who could potentially benefit from prophylactic ICD implantation. Currently, the LVEF <35% metric is used to stratify patient risk for SCD and identify candidates for preventive ICD implantation. However, patients with LVEF <35% account for only one-third of all SCD events.25 Thus a large proportion of patients who are susceptible to SCD are not indicated for prophylactic ICD insertion.
We recently demonstrated that VARP computational simulations could be used to predict arrhythmia susceptibility in patients with MI and LVEF <35%.11 That retrospective study demonstrated that VARP results are superior to other current non-invasive clinical metrics, such as LVEF, grey zone volume,26 scar volume,27 or left ventricular mass,28 as well as to the invasive EP testing. These results point to the translational potential of VARP in being used as an effective patient selection tool for prophylactic ICD implantation.
The case studies presented here demonstrate that VARP can also be used to identify MI patients with preserved LVEF who are nonetheless at risk for arrhythmias and who could benefit from prophylactic ICD implantation. Virtual-heart arrhythmia risk predictor is of low burden to patients since it requires only a single MRI scan and would not significantly alter the current standard of care for these patients. The simulations presented here show that the method is robust for personalized arrhythmia risk stratification in patients with LVEF >35%.
Among the four patients in this article, the grey zone and scar volume did not differ significantly between the VT-inducible and not inducible hearts. This is consistent with the findings from the 41 patients with LVEF <35% examined in our previous study11: in that cohort, neither grey zone nor scar volume were found predictive of arrhythmia events. The reentrant circuit in the inducible model had an isthmus located within the grey zone, which is consistent with our previous findings of VT organizing centres in grey zone,12,13,29,30 and experimental data which showed that the infarcted border served as the isthmus and a centrifugal pathway in different VT morphologies.26,31
The VARP approach entails a complex simulation methodology, validated over a number of studies.13,27,29–31 Accurate reconstruction of ventricular geometry has been validated with ex-vivo and in-vivo imaging datasets.12,32 The approach to construct a model of the infarcted ventricles by thresholding the infarct into only scar and (homogeneous) grey zone, as done in the present study, without accounting for additional small-scale heterogeneities in the grey zone, has been recently validated with experimental data. Specifically, Deng et al.13 used sock epicardial data for infarct-related VT, obtained from in-vivo swine heart, and demonstrated that ventricular models reconstructed from MRI data of the corresponding hearts were able to predict fairly accurately the morphology of each VT reentrant circuit and its organizing centre (e.g. isthmus). This approach to account for the contribution of the grey zone to ventricular arrhythmogenesis has been instrumental in enabling us to perform a large number of simulations of patient hearts.11 Similar personalized virtual heart simulation methodologies are now being implemented to assess arrhythmogenesis and predict optimal ablation strategies for atrial arrhythmias in patients with fibrotic remodelling.33,34
Limitations
A limitation of the current study is the small sample size of only four patients. The predictive capability of VARP for MI patients with preserved LVEF needs to be assessed in large retrospective and ultimately, prospective studies.
Conclusions
This study presents for the first time the use of virtual heart computational modelling to predict the arrhythmia risk in patients with LVEF >35%. The results demonstrate that personalized heart models can accurately detect VT arrhythmia susceptibility in those patients who are usually not candidates for prophylactic ICD implantation. Our proposed methodology has the potential to aid clinicians in preventing SCD in these patients.
Funding
NIH Pioneer Award (DP1-HL123271) to N.T.
Conflict of interest: none declared.
References
- 1. Fishman GI, Chugh SS, Dimarco JP, Albert CM, Anderson ME, Bonow RO. et al. Sudden cardiac death prediction and prevention: report from a National Heart, Lung, and Blood Institute and Heart Rhythm Society Workshop. Circulation 2010;122:2335–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Zipes DP, Camm AJ, Borggrefe M, Buxton AE, Chaitman B, Fromer M. et al. ACC/AHA/ESC 2006 guidelines for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death. Europace 2006;8:746–837. [DOI] [PubMed] [Google Scholar]
- 3. Bardy GH, Lee KL, Mark DB, Poole JE, Packer DL, Boineau R. et al. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. N Engl J Med 2005;352:225–37. [DOI] [PubMed] [Google Scholar]
- 4. Gehi A, Haas D, Fuster V. Primary prophylaxis with the implantable cardioverter-defibrillator: the need for improved risk stratification. JAMA 2005;294:958–60. [DOI] [PubMed] [Google Scholar]
- 5. Epstein AE, DiMarco JP, Ellenbogen KA, Estes NA, 3rd, Freedman RA, Gettes LS. et al. ACC/AHA/HRS 2008 Guidelines for Device-Based Therapy of Cardiac Rhythm Abnormalities: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the ACC/AHA/NASPE 2002 Guideline Update for Implantation of Cardiac Pacemakers and Antiarrhythmia Devices): developed in collaboration with the American Association for Thoracic Surgery and Society of Thoracic Surgeons. Circulation 2008;117:e350–408. [DOI] [PubMed] [Google Scholar]
- 6. Reynolds MR, Cohen DJ, Kugelmass AD, Brown PP, Becker ER, Culler SD. et al. The frequency and incremental cost of major complications among medicare beneficiaries receiving implantable cardioverter-defibrillators. J Am Coll Cardiol 2006;47:2493–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Sohail MR, Henrikson CA, Braid-Forbes MJ, Forbes KF, Lerner DJ. Mortality and cost associated with cardiovascular implantable electronic device infections. Arch Intern Med 2011;171:1821–8. [DOI] [PubMed] [Google Scholar]
- 8. Poole JE, Johnson GW, Hellkamp AS, Anderson J, Callans DJ, Raitt MH. et al. Prognostic importance of defibrillator shocks in patients with heart failure. N Engl J Med 2008;359:1009–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Noyes K, Corona E, Zwanziger J, Hall WJ, Zhao H, Wang H. et al. Health-related quality of life consequences of implantable cardioverter defibrillators: results from MADIT II. Med Care 2007;45:377–85. [DOI] [PubMed] [Google Scholar]
- 10. Alba AC, Agoritsas T, Jankowski M, Courvoisier D, Walter SD, Guyatt GH. et al. Risk prediction models for mortality in ambulatory patients with heart failure: a systematic review. Circ Heart Fail 2013;6:881–9. [DOI] [PubMed] [Google Scholar]
- 11. Arevalo HJ, Vadakkumpadan F, Guallar E, Jebb A, Malamas P, Wu KC. et al. Arrhythmia risk stratification of patients after myocardial infarction using personalized heart models. Nat Commun 2016;7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Prakosa A, Malamas P, Zhang S, Pashakhanloo E, Arevalo H, Herzka DA. et al. Methodology for image-based reconstruction of ventricular geometry for patient-specific modeling of cardiac electrophysiology. Prog Biophys Mol Biol 2014;115:226–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Deng DD, Arevalo H, Pashakhanloo F, Prakosa A, Ashikaga H, McVeigh E. et al. Accuracy of prediction of infarct-related arrhythmic circuits from image-based models reconstructed from low and high resolution MRI. Front Physiol 2015;6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Prassl AJ, Kickinger F, Ahammer H, Grau V, Schneider JE, Hofer E. et al. Automatically generated, anatomically accurate meshes for cardiac electrophysiology problems. IEEE Trans Biomed Eng 2009;56:1318–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Bayer JD, Blake RC, Plank G, Trayanova NA. A novel rule-based algorithm for assigning myocardial fiber orientation to computational heart models. Ann Biomed Eng 2012;40:2243–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Plank G, Zhou L, Greenstein JL, Cortassa S, Winslow RL, O'Rourke B. et al. From mitochondrial ion channels to arrhythmias in the heart: computational techniques to bridge the spatio-temporal scales. Philos Trans A Math Phys Eng Sci 2008;366:3381–409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. ten Tusscher KHWJ, Noble D, Noble PJ, Panfilov AV. A model for human ventricular tissue. Am J Physiol-Heart C 2004;286:H1573–89. [DOI] [PubMed] [Google Scholar]
- 18. Decker KF, Rudy Y. Ionic mechanisms of electrophysiological heterogeneity and conduction block in the infarct border zone. Am J Physiol-Heart C 2010;299:H1588–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Cabo C, Boyden PA. Electrical remodeling of the epicardial border zone in the canine infarcted heart: a computational analysis. Am J Physiol-Heart C 2003;284:H372–84. [DOI] [PubMed] [Google Scholar]
- 20. Yao JA, Hussain W, Patel P, Peters NS, Boyden PA, Wit AL. Remodeling of gap junctional channel function in epicardial border zone of healing canine infarcts. Circ Res 2003;92:437–43. [DOI] [PubMed] [Google Scholar]
- 21. Vigmond EJ, Hughes M, Plank G, Leon LJ. Computational tools for modeling electrical activity in cardiac tissue. J Electrocardiol 2003;3(Suppl 6):69–74. [DOI] [PubMed] [Google Scholar]
- 22. Cheng A, Dalal D, Butcher B, Norgard S, Zhang YY, Dickfeld T. et al. Prospective observational study of implantable cardioverter-defibrillators in primary prevention of sudden cardiac death: study design and cohort description. J Am Heart Assoc 2013;2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Cerqueira MD, Weissman NJ, Dilsizian V, Jacobs AK, Kaul S, Laskey WK. et al. Standardized myocardial segmentation and nomenclature for tomographic imaging of the heart: a statement for healthcare professionals from the Cardiac Imaging Committee of the Council on Clinical Cardiology of the American Heart Association. Circulation 2002;105:539–42. [DOI] [PubMed] [Google Scholar]
- 24. Piccini JP, Hafley GE, Lee KL, Fisher JD, Josephson ME, Prystowsky EN. et al. Mode of induction of ventricular tachycardia and prognosis in patients with coronary disease: the Multicenter UnSustained Tachycardia Trial (MUSTT). J Cardiovasc Electr 2009;20:850–5. [DOI] [PubMed] [Google Scholar]
- 25. Stecker EC, Vickers C, Waltz J, Socoteanu C, John BT, Mariani R. et al. Population-based analysis of sudden cardiac death with and without left ventricular systolic dysfunction: two-year findings from the Oregon sudden unexpected death study. J Am Coll Cardiol 2006;47:1161–6. [DOI] [PubMed] [Google Scholar]
- 26. Arenal A, Perez-David E, Avila P, Fernandez-Portales J, Crisostomo V, Baez C. et al. Noninvasive identification of epicardial ventricular tachycardia substrate by magnetic resonance based signal intensity mapping. Heart Rhythm 2014;11:1456–64. [DOI] [PubMed] [Google Scholar]
- 27. Ashikaga H, Arevalo H, Vadakkumpadan F, Blake RC, Bayer JD, Nazarian S. et al. Feasibility of image-based simulation to estimate ablation target in human ventricular arrhythmia. Heart Rhythm 2013;10:1109–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Vadakkumpadan F, Trayanova N, Wu KC. Image-based left ventricular shape analysis for sudden cardiac death risk stratification. Heart Rhythm 2014;11:1693–700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Arevalo H, Plank G, Helm P, Halperin H, Trayanova N. Tachycardia in post-infarction hearts: insights from 3d image-based ventricular models. Plos One 2013;8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Ukwatta E, Arevalo H, Rajchl M, White J, Pashakhanloo F, Prakosa A. et al. Image-based reconstruction of three-dimensional myocardial infarct geometry for patient-specific modeling of cardiac electrophysiology. Med Phys 2015;42:4579–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Ashikaga H, Sasano T, Dong J, Zviman MM, Evers R, Hopenfeld B. et al. Magnetic resonance-based anatomical analysis of scar-related ventricular tachycardia: implications for catheter ablation. Circ Res 2007;101:939–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Vadakkumpadan F, Arevalo H, Prassl AJ, Chen J, Kickinger F, Kohl P. et al. Image-based models of cardiac structure in health and disease. Wiley Interdiscip Rev Syst Biol Med 2010;2:489–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Chrispin J, Gucuk Ipek E, Zahid S, Prakosa A, Habibi M, Spragg D. et al. Lack of regional association between atrial late gadolinium enhancement on cardiac magnetic resonance and atrial fibrillation rotors. Heart Rhythm 2016;13:654–60. [DOI] [PubMed] [Google Scholar]
- 34. Zahid S, Cochet H, Boyle PM, Schwarz EL, Whyte KN, Vigmond EJ. et al. Patient-derived models link reentrant driver localization in atrial fibrillation to fibrosis spatial pattern. Cardiovasc Res 2016;110(3):43–454. [DOI] [PMC free article] [PubMed] [Google Scholar]