Abstract
Due to their size and tailorable physicochemical properties, nanomaterials are an emerging class of structures utilized in biomedical applications. There are now many prominent examples of nanomaterials being used to improve human health, in areas ranging from imaging and diagnostics to therapeutics and regenerative medicine. An overview of these examples reveals several common areas of synergy and future challenges. This Nano Focus discusses the current status and future potential of promising nanomaterials and their translation from the laboratory to the clinic, by highlighting a handful of successful examples.
Advances in medicine in the areas of genomics, proteomics, tissue engineering, and regenerative medicine are occurring at a rate that was previously unthinkable. The development of new materials resulting from these breakthroughs, such as those that can be used to replace blood vessels, to promote tissue growth, to monitor blood glucose levels, or to improve the bioavailability of drugs, has been equally rapid and diverse.1,2 One of the most exciting frontiers is the development of nanomaterials for biomedical applications.3,4 Nanomaterials have size-, shape-, and composition-dependent physical, chemical, optical, and electronic properties, among others, that can be designed and tuned, and they are showing great promise for the diagnosis, treatment, monitoring, and control of disease.5–8 A recent survey found that more than 247 nanomaterial-based medical products have been approved by the Food and Drug Administration (FDA) and are currently in various stages of clinical study.9 Their intended uses range from the treatment of clinically unresectable cancers to the preparation of antibacterial hand gels to the regeneration of heart tissue. At the same time, common themes emerge when discussing nanomaterials in medicine. Indeed, one of the biggest issues is how to translate nanomaterials from the laboratory to the clinic effectively.
At the 2015 Materials Research Society (MRS) Spring Meeting in San Francisco (April 6–10), a special symposium focusing on translational research involving nanomaterials in the biomedical field was held. Researchers from all over the world gathered to exchange ideas and to discuss criteria for the selection, development, synthesis, and utilization of nanomaterials. In this Nano Focus, we highlight these discussions, which fall into three categories: nanotherapeutics, imaging and diagnostics, and tissue regeneration.
Enhancing Efficacy in Nanotherapeutics
Among the numerous nanomaterials explored in therapeutic applications, those often found in clinical trials are gold nanoparticles, polymeric nanoparticles, liposomes, and carbon-based nanomaterials. By sharing expertise across fields, researchers can accelerate the utilization of nanomaterials in addressing the challenges faced by traditional therapeutic agents.
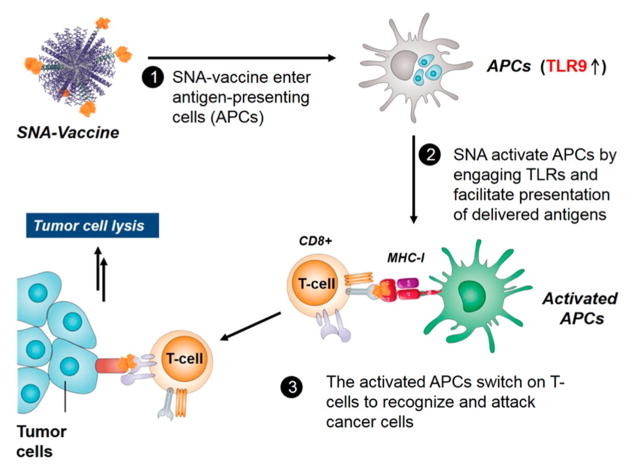
Since the development of gold-nanoparticle-based spherical nucleic acids (SNAs) in 1996,10 the Mirkin group at Northwestern University has exploited the properties of this class of nanostructures in many areas of biomedicine. Spherical nucleic acids typically consist of a nanoparticle core and a highly oriented and densely packed shell of oligonucleotides.11 The properties of SNAs emerge from the orientation and arrangement of oligonucleotides on the surfaces of these particles. For example, SNAs are taken up into many different cell types (over 60 tested to date) at high levels and rates without the need for transfection agents,12 have high affinities for nucleic acid targets (100 times greater than linear DNA of the same sequence),13 and cross both the blood–brain barrier14 and the epidermis15 to reach dificult-to-target tissues in therapeutic applications. Success in the utilization of SNAs for intracellular mRNA detection has led to the commercialization of NanoFlare technology16,17 under the trade name SmartFlares (Merck Millipore in partnership with AuraSense, LLC, Skokie, Illinois), and SNAs have also been applied as agents in gene regulation as therapeutics for a host of cancers, including glioblastoma multiforme (an aggressive form of brain cancer), and skin disorders, among others. The Mirkin group has made progress in using SNAs as immunomodulatory agents.18 Spherical nucleic acids functionalized with oligonucleotides displaying toll-like receptor (TLR)-agonist or TLR-antagonist activity were shown to be capable of either stimulating or modulating the activity of the immune system, respectively. Such structures do so with potencies up to several orders of magnitude higher than the conventional linear nucleic acids from which they are composed. This discovery paves the way for the development of SNAs as therapeutic cancer vaccines. Mirkin’s demonstration of the ability of SNAs to reduce tumor burden and to enhance survival in vivo, in mouse models of lymphoma, proved that immunomodulatory SNAs can be directed to activate the response of the immune system to destroy tumors in an antigen-specific manner (Figure 1).
Figure 1.
In vivo activity of immunomodulatory SNAs as cancer vaccines.
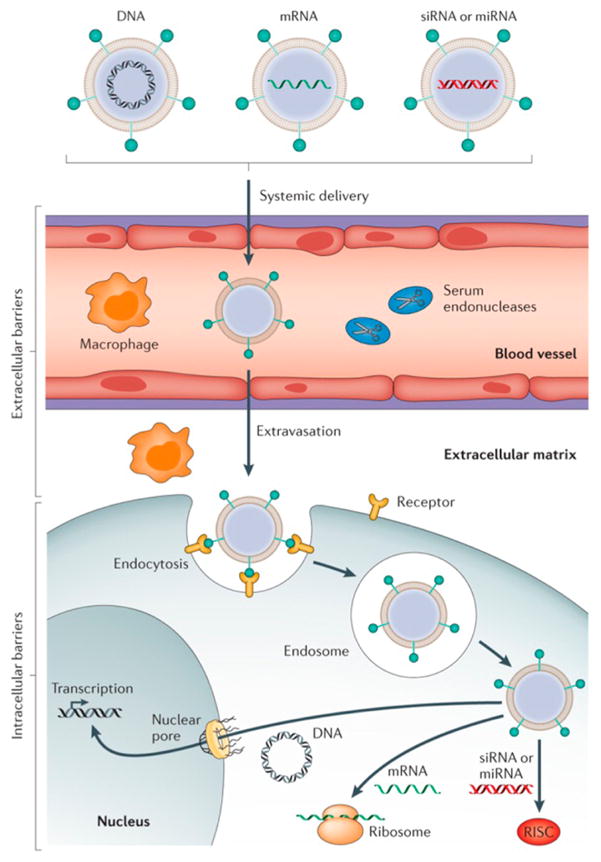
A fundamental challenge is the delivery of therapeutic molecules inside target cells in the body. Nanoparticles have shown immense promise as vehicles for intracellular delivery, with proof-of-principle experiments in humans being completed with small interfering RNA (siRNA).19 However, there are many barriers to achieving safe and effective delivery systems (Figure 2), and potential delivery systems must have multiple functionalities to allow in vivo delivery, making the design criteria dificult to define for nanoparticles capable of accomplishing intracellular delivery. To accelerate the design and discovery process, the Anderson group at the Massachusetts Institute of Technology has pioneered combinatorial methods for nanoparticulate drug delivery. Combinatorial chemical methods have been developed to enable the rapid synthesis and characterization of a range of nanoformulations based on biodegradable polymers, lipid-like materials, and other materials.20–22 These have generated new formulations with particular promise as delivery vehicles for RNA and other nucleic acids;23 these formulations have the potential to be used as therapies for many diseases, including cancer.24
Figure 2.
Barriers to successful in vivo delivery of nucleic acids using nonviral vectors. Reprinted with permission from ref 19. Copyright 2014 Macmillan Publishers Ltd.
Furthermore, Shi at Brigham and Women’s Hospital/Harvard Medical School described the rational design and development of lipid–polymer hybrid nanoparticle platforms to address the bottlenecks faced in the delivery of RNA interference (RNAi) therapeutics, such as siRNA.25–27 Specifically, the clinical applications of RNAi in cancer therapy are currently hindered by the challenge of achieving the effective systemic in vivo delivery of siRNA to tumors. Multiple physiological barriers, such as enzymatic degradation, rapid elimination by renal excretion or the mononuclear phagocyte system, and poor cellular uptake and endosomal escape, must be overcome. By utilizing hybrid nanoparticles, Shi achieved sustained gene silencing, and prolonged circulation of siRNA in the blood for high tumor extravasation and accumulation. The successful application of these RNAi nanoparticles to validate the therapeutic role of Prohibitin1 in non-small cell lung cancer treatment27 indicates the significant potential of this platform for the validation of many other potential cancer targets and for the clinical development of novel cancer therapies.
Kataoka at the University of Tokyo has pioneered the synthesis and development of “polymeric micelle drugs”, which have proven useful for targeting a variety of drugs to tissues and organs and to tumors in particular. There has been significant recent progress in the clinical development of polymeric micelles loaded with a variety of cytotoxic reagents. Notably, five different micellar formulations have already been explored in clinical trials in Asia and the United States.28 A version loaded with paclitaxel is in the final stage of a Phase III clinical trial in Japan for the treatment of recurrent breast cancer, and it is expected to proceed into the application for approval within a year. More recently, Kataoka and colleagues have been active in developing a second generation system of polymeric micelles installed with ligand moieties at their peripheries. Particularly, cRGD-conjugated polymeric micelles were able to cross the blood–brain tumor barrier via a transcytosis mechanism, achieving high efficacy in treating intractable orthotopic glioblastoma in animal experiments.29 Antibody fragments can also be conjugated to polymeric micelles. In this way, higher drug payloads can be achieved without antibody precipitation or impaired binding compared to when drugs are directly conjugated to antibody molecules using more conventional approaches.30
In 1989, the Kabanov group at the University of North Carolina at Chapel Hill investigated the use of polymeric micelles for targeted drug delivery.31 They discovered that Pluronic block copolymers can be used in the sensitization of multi-drug-resistant (MDR) cancer and cancer stem cells and elucidated the mechanisms responsible for these effects.32 This research led to first-in-man polymeric micelle drug candidates for the treatment of cancer (SP1049C) that show high efficacy against advanced esophageal cancer in Phase II trials.33 Recently, they discovered polymeric micelles based on amphiphilic poly(2-oxazoline) blocks with unprecedented high capacities for poorly soluble, uncharged drugs (e.g., taxanes) and drug combinations, enabling increasing therapeutic indices compared to current drug formulations.34,35
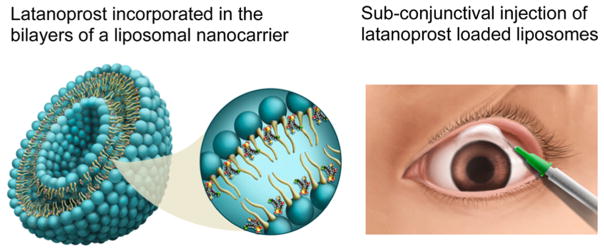
Liposomes are another popular nanomaterial used in preclinical and clinical studies, but it is dificult to sustain release from these structures for more than a few days. The Venkatraman group at Nanyang Technological University has developed a subconjunctivally injected nanoliposome drug delivery system for the long-term (3–5 months) delivery of latanoprost that can be used in glaucoma treatment, which went from concept to clinic in less than 5 years (Figure 3).36,37 Glaucoma is a chronic progressive optic neuropathy that is characterized by optic nerve changes and visual field loss. Elevated intraocular pressure (IOP) is the main modifiable risk factor. The chronic instillation of daily eye drops to lower IOP is the primary treatment of choice, although this regimen requires patient adherence and correct performance. Hence, a sustained-delivery system would be a big boon to patients with glaucoma. Venkatraman’s group also explored the nanoparticle-mediated sustained delivery of siRNA for preventing fibrosis following surgery.38 This method has applications in ocular and other types of surgeries/implantations. They have shown sustained efficacy of action with a siRNA-incorporated nanoparticle. As shown in the above two examples, the premise of their work rests on the ability of nanoparticles to sustain drug/protein/siRNA release.
Figure 3.
Subconjunctival instillation of nanocarriers incorporating latanoprost lowers eye pressure in glaucoma patients for up to 3 months. Reproduced from ref 37. Copyright 2014 American Chemical Society.
The Chow and Ho groups at the National University of Singapore and the University of California, Los Angeles (UCLA), respectively, study nanodiamonds, which are an emerging class of carbon-based nanomaterials, due to their advantageous surface characteristics. Nanodiamond facets mediate electrostatic properties that have resulted in potent and scalable anthracycline drug binding as well as marked enhancements in magnetic resonance imaging (MRI) efficiency.39,40 With regard to drug delivery, nanodiamond–doxorubicin compounds (NDX) were administered to treat drug-resistant breast and liver tumors in mouse models. This study demonstrated the nanodiamond-mediated improvement of drug tolerance; lethal dosages of doxorubicin delivered as NDX resulted in the smallest tumors observed (compared to saline controls and unmodified drug administration), and all of the treated mice survived the full duration of the study.41 The active targeting of triple-negative breast tumors in vivo using nanodiamond–epirubicin complexes functionalized with the epidermal growth factor receptor (EGFR) antibody resulted in complete tumor regression.42 More recently, nanodiamond–anthracycline agents have been used to treat hepatic cancer stem cells.43
Toward the further enhancement of the potency and safety of cancer therapies, a prevalent challenge in the field of nanomedicine is the ability to move beyond monotherapies toward combinatorial cancer treatments. Using phenotypic instead of genotypic profiling to drive combinatorial optimization, Ho and colleagues developed a powerful mechanism-independent engineering optimization platform, termed Feedback System Control.II (FSC.II), to identify globally optimized nanodiamond–anthracycline drug combinations rapidly.44 FSC.II does not require the use of feedback, and instead, it utilizes a selected set of experimental validation assays to formulate phenotypic profiles from which drug combinations can rapidly be pin-pointed.
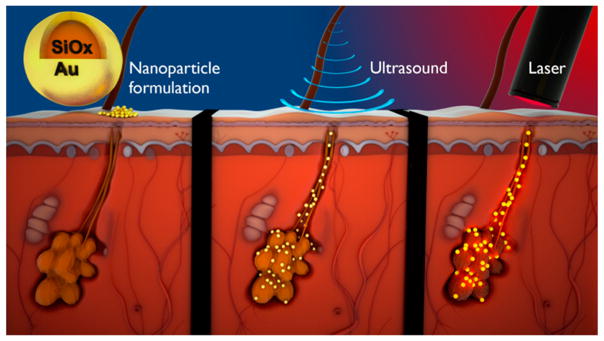
Mitragotri at the University of California, Santa Barbara, also translated nanomaterials to clinical applications (Figure 4). Specifically, he used gold-coated silica nanoparticles for the treatment of acne. These 150 nm, poly(ethylene glycol)-coated, silica–gold nanoparticles are designed to absorb near-infrared light and produce localized heating. The delivery of these nanoparticles into skin is a major hurdle due to the skin’s barrier properties. Mitragotri and colleagues showed that these nanoparticles can be delivered deep into the skin’s sebaceous glands using low-frequency ultrasound.45 Ultrasound induces cavitation on the surface of the skin, which produces microjets and shock waves that open transport pathways into the glands.46 Once delivered deep into the glands, the thermal activation of the nanoparticles using near-infrared light caused thermolysis and the inactivation of overactive sebaceous glands, the underlying pathology of acne. This nanoparticle-based technology provides several advantages over standard treatments for acne; for instance, systemic side effects are avoided with this treatment.
Figure 4.
Schematic for the nanoparticle-based treatment of acne. Silica–gold nanoparticles are delivered into sebaceous glands using low-frequency ultrasound. Nanoparticles are then activated using near-infrared light to induce thermolysis. Reprinted with permission from ref 45. Copyright 2015 Elsevier.
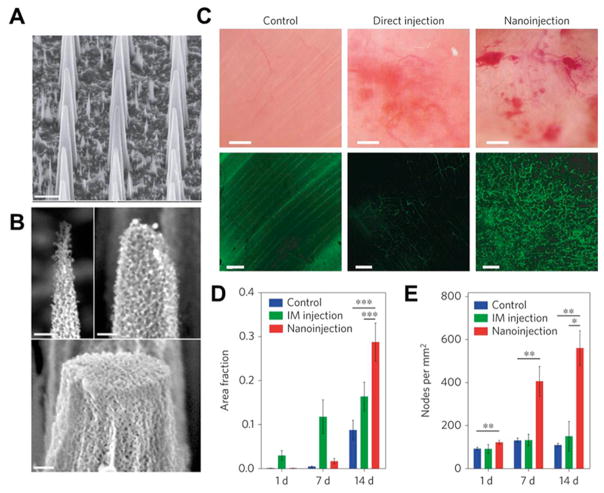
The Stevens group at the Imperial College of London and collaborators at the Houston Methodist Research Institute have recently reported engineering a platform of mesoporous silicon nanoneedles for the delivery of nanoparticle and other therapeutic payloads to cells and tissues (Figure 5A, B).47,48 This technology could prove transformative in the fields of drug delivery, regenerative medicine, and biosensing. The dynamics of the nanoneedle entry to the cell and study of the nanoneedle–cell interface have been elucidated and pave the way for highly controlled delivery of a range of nanoparticle payloads intracellularly.47 Furthermore, the nanoneedle array can simultaneously deliver both DNA and siRNA with high efficiency (over 90%) and in vivo proved successful in upregulating blood vessel formation in muscle by delivery of the VEGF-165 gene (Figure 5C–E).48 The Stevens group has also developed several other notable nanomaterials-based technologies, particularly enzyme-response nanoparticle systems that have a wide range of important impacts in the field of biosensing.8,49,50
Figure 5.
Nanoneedle for the delivery of VEGF-165 to upregulate blood vessel formation in muscle. (A) Scanning electron microscope (SEM) micrographs showing the morphology of porous silicon nanoneedle arrays with pitches of 2 μm. Scale bars, 2 μm. (B) High-resolution SEM micrographs of nanoneedle tips showing the nanoneedles’ porous structure and the tunability of tip diameter from less than 100 nm to over 400 nm. Scale bars, 200 nm. (C) Intravital bright-field (top) and confocal (bottom) microscopy images of the vasculature of untreated (left) and hVEGF-165-treated muscles with either direct injection (center) or nanoinjection (right). The fluorescence signal originates from systemically injected FITC–dextran. Scale bars, bright-field 100 μm; confocal 50 μm. (D, E) Quantification of the fraction of fluorescent signal (dextran) (D) and the number of nodes in the vasculature per mm2 (E) within each field of view acquired for untreated control, intramuscular injection (IM) and nanoinjection. *p = 0.05, **p < 0.01, ***p < 0.001. Error bars represent the SD of the averages of five areas taken from three animals. Reprinted with permission of figure and caption text from ref 48. Copyright 2015 Macmillan Publishers Ltd.
From the above discussion of a variety of nanomaterials as nanotherapeutic agents for enhancing treatment efficacy, we conclude that successful translation of nanomaterials relies on the identification of a clinical problem and innovative ideas to solve it through rational design. In addition, more and more versatile nanomaterials are being exploited in emerging research themes, areas such as cancer vaccines51 and genome editing.19
Imaging and Diagnostics with High Sensitivity and Selectivity
Besides their use as therapeutic agents, nanomaterials are also being used for imaging and diagnostics purposes. The most well-known examples include silica nanoparticles, quantum dots, magnetic nanoparticles, and microbubbles. These nanostructures have been used to detect small molecules (like H2O2), cells including circulating tumor cells (CTCs), and tumor tissues.
The Sun group at Brown University is interested in monitoring cellular H2O2, which is an important reactive oxygen species generated via oxygen metabolism; it is actively involved in cell signaling and cell growth.52 Unfortunately, its uncontrolled overproduction can cause the detrimental oxidation of biomolecules and lead to aging, cancer, and other diseases.53 Sun recently developed dumbbell (Au–Fe3O4 and PdPt–Fe3O4) and core/shell (Au/MnO) magnetic nanoparticles as sensitive probes for H2O2 detection.54,55 Dumbbell magnetic nanoparticles were prepared by the controlled nucleation and growth of Fe3O4 on presynthesized noble metal nanoparticles, while the core/shell Au/MnO nanoparticles were made by the controlled oxidation of AuMn alloy nanoparticles. Both dumbbell and core/shell nanoparticles are active for the electrochemical reduction of H2O2 with detection limits reaching as low as 5 nM. Highly sensitive electrochemical sensors have been used to monitor H2O2 concentration levels released from living cells; tumorigenic cells were found to have higher levels of H2O2 than nontumorigenic ones. These composite nanoparticle probes can be used in high-sensitivity cancer detection schemes and may also help to increase the efficacy of cancer therapies.
The Chen group at the National Institute of Biomedical Imaging and Bioengineering of the National Institutes of Health uses nanomaterials as platforms to provide imaging contrast in positron emission tomography (PET). In medical imaging, PET can provide a direct, highly sensitive, and quantitative readout of organ/tissue targeting efficiency and pharmacokinetics. Compared with radiolabeled antibodies, proteins, peptides, and other biologically relevant molecules, radiolabeled nanoparticles represent a new frontier in molecular imaging probe design because they can combine different imaging modalities and targeting ligands in a single vector, synergistically improving the imaging quality.56 However, the applications of radiolabeled nanoparticles are based on the premise that the radioisotopes are stably attached to the nanomaterials. Chen has developed general rules for selecting appropriate isotopes for given types of nanoparticles as well as adjusting the labeling reaction according to specific applications. The stability (colloidal and radiochemical) of the radiolabeled nanoparticles as well as their biological fate must be assessed; special attention should be paid to labeling strategies as they affect the stability of radiolabeled nanoparticles and might cause discrepancies in the interpretation of PET data (owing to the distribution of nanoparticles).
Wang’s group at the Chinese Academy of Sciences is interested in creating nano–bio interfaces with controllable adhesion properties. The cell-adhesive biointerfaces are based on the cooperative effects of multiscale structural matching and molecular recognition.57 They explore the relationship between cell-specific adhesion and surface structure (with nanowires,21 nanofibers,58 nanofractals,59 and soft nanotubes60). Also, they developed a series of biointerfaces with specific recognition and stimuli-responsive capture and release properties (i.e., temperature,61 electric,59 enzymatic,21 and pH62). They have made progress in the isolation of viable rare CTCs from the blood via their designed cell-adhesive biointerfaces and developed adhesion-based CTC isolation approaches as cancer diagnostics with high efficiencies (>97%).63 In particular, these biointerfaces with controlled cell adhesion are capable of capturing rare viable CTCs for early cancer detection and the monitoring of cancer therapy, single-cell gene analysis,64 and other purposes.
Biomimetic and Bio-Inspired Scaffolds for Tissue Engineering
Nanomaterials for tissue engineering involve a broad spectrum of nanoscale formulations and structures developed to mimic tissue complexity and to modulate cellular function to yield therapeutic benefits.
The Xia group at the Georgia Institute of Technology and Emory University has been developing practical nanomaterials for medical applications. They use electrospun nanofibers in tissue engineering.65 Electrospinning has been widely explored to process polymeric materials into nanofibers with tunable and controllable compositions, diameters, porosities, and surface properties. Owing to its small feature size, high porosity, and large surface area, a nonwoven mat of electrospun fibers can serve as a superb scaffold that mimics the extracellular matrix (ECM), which is critical to cell attachment and spreading. The nanofibers themselves can also be functionalized through the encapsulation or attachment of bioactive species, such as ECM proteins, enzymes, and growth factors. In addition, the nanofibers can be readily assembled into a wide variety of arrays or hierarchical structures by manipulating their alignment, stacking, or folding. All these attributes make electrospinning a powerful tool for generating nanostructured materials for a broad range of biomedical applications, including controlled release, drug delivery, and tissue engineering. Xia highlighted the use of aligned nanofibers to control the differentiation of embryonic stem cells into different types of neural lineages and to guide the outgrowth of neurites for peripheral nerve repair.66 He also pointed out that nanofiber scaffolds could be designed for repairing injuries to the flexor tendon and the tendon-to-bone insertion site;67 they could also be used as wound dressings for brain surgery.68
Karp from Brigham and Women’s Hospital suggests that different approaches are required for solving medical problems versus solving basic science problems. He asserts that one must develop design criteria relevant to solving the problem in animal models, while considering the multiple steps required to bring a technology from the laboratory to the clinic. One must think through scale-up, manufacturing, regulatory issues, and patent strategy and then impose these criteria to advance toward a potential solution. In particular, turning to nature for solutions can aid in the problem-solving process, recognizing that everything living has overcome challenges, and thus we are surrounded by solutions. Through elucidating mechanisms behind these solutions with state-of-the-art tools, he asserts that we can identify ideas to help overcome even the most challenging of problems. Karp has created tissue adhesives using inspiration from slugs and snails,69 spiny-headed worms,70 porcupine quills,71 and spider webs. One of the adhesive technologies led to the formation of a company, Gecko Biomedical (Paris, France), and is on track to be tested for vascular graft applications in humans in late 2015. Of note is that, in these developments, failure should be embraced as part of the problem-solving process and is likely a prerequisite to success. In this work, it was important to build highly functional and multi-disciplinary teams that know what resources are available in their environment and how to access them.
Teoh from Nanyang Technological University is interested in processing biomaterials in an environmentally friendly way.72,73 To this end, his group has unearthed a solvent-free approach known as cryomilling, where biomaterials are processed at near cryogenic temperatures. Polymer particle size reduction has been achieved due to the high-energy collision process, which occurs below the glass transition temperature. Particle sizes may be reduced to the nanoscale, where surface thermodynamics play critical roles in determining their behavior, particularly the interactions between two chemically distinct phases, as in the case of composite biomaterials. Notably, there is also a significant challenge in obtaining reproducible, well-distributed composites. They have demonstrated that a variety of composite biomaterials that incorporate second phases, such as inorganic elements, trace elements, and even drugs, may be processed efficiently via cryomilling.73 This technique also addresses second phase distribution issues that plague many other processing techniques, such as solvent-casting, electrospinning, and melt extrusion.
FUTURE OUTLOOK
Tremendously exciting advances in nanomaterials-based medical treatments, from cancer therapies, diabetes administration, and vaccine development to molecular diagnostics, tissue repair, and regeneration, are both underway and yet to come. The confluence of nanomaterials and emerging biomedical science provides vast commercial opportunities. While fundamental science has been fueled with numerous innovations over the past decade, as evidenced by the number of nanomaterial-associated formulations and devices in clinical development, the number of marketed products is still small, compared to traditional medications. Remaining challenges include effectively improving efficacy, while minimizing potential concerns, through the rational design and thorough evaluation of nanomaterials.
In oncology nanomedicine, for example, the acceleration of the translation process relies profoundly on a thorough understanding of how nanocarriers interact with the physiological environment. In addition to general evaluations based on the enhanced permeability and retention effect, biodistribution, and clearance mechanisms, more precise details should be taken into account, such as how the particles pass through the tumor microenvironment and enter cells to reach active sites.74,75,76 Moreover, in designing stimuli-responsive or programmable nanocarriers,77,78 a current theme in nanomedicine, closer investigations of the dynamic relationships between the phase transitions of materials and the relevant gradients in the biological environment, such as pH, redox, glucose, ATP, and enzyme activity, should enable more precise targeting and release.79–82
Second, a major stumbling block in the translation of nanomaterials for biomedical applications is safety concerns, especially for invasive administration. On one hand, clinical use requires the careful, prolonged evaluation of the local and systemic toxicity of nanomaterials as well as their potential immunogenicity.83 On the other hand, there is an urgent need to invent and to tailor new materials with excellent biocompatibility. Ideas inspired by nature, mimicking the structures and composites of natural particles, including viruses, vesicles, and cells, have attracted increasing interest and brought promising outcomes.
Third, regarding the rational design of nanomaterials with specific physicochemical properties84 for clinical applications, it is important to set uniformity in preclinical trials. Variability in particle size, surface properties, and stability as well as differences in cell lines, tumor properties, therapeutic doses, and pharmacokinetics/pharmacodynamics analysis have prevented the systematic comparison of relevant nanomaterials and have been an impediment to creating design rules for optimizing a specific formulation or device.
Last but not least, the design, development, and ultimately commercialization of clinically used nanomaterials require seamless collaboration and commitment between a broad range of research investigators, investors, and regulatory authorities. Key to these activities are platforms for fusing ideas to shepherd emerging technologies further along safe and effective pipelines.
Overall, given the progress that has been made so far, we are optimistic that nanomaterials-based clinical development will continue to be exciting, with growing numbers of innovations as well as those currently garnering FDA approval entering the clinic soon. We hope the ideas and concepts presented in this Nano Focus will be useful in the development of “ideal” nanomaterials features, the expansion of design criteria, and the enlightenment of research opportunities for evolving the next generations of biomedical materials.85,86,87
Footnotes
Conflict of Interest: The authors declare the following competing financial interest(s): S.M. is a Scientific Advisor to Sebacia Inc. J.M.K. is part owner of and consultant to Gecko Biomedical.
REFERENCES AND NOTES
- 1.Ratner BD, Hoffman AS, Schoen FJ, Lemons JE. Biomaterials Science: An Introduction to Materials in Medicine. Elsevier Academic Press; Amsterdam: 2004. [Google Scholar]
- 2.Lee S, Henthorn D. Materials in Biology and Medicine. CRC Press/Taylor & Francis Group; Boca Raton, FL: 2012. [Google Scholar]
- 3.Langer R. Drug Delivery and Targeting. Nature. 1998;392:5–10. [PubMed] [Google Scholar]
- 4.Farokhzad OC, Langer R. Nanomedicine: Developing Smarter Therapeutic and Diagnostic Modalities. Adv Drug Delivery Rev. 2006;58:1456–1459. doi: 10.1016/j.addr.2006.09.011. [DOI] [PubMed] [Google Scholar]
- 5.Doshi N, Mitragotri S. Designer Biomaterials for Nanomedicine. Adv Funct Mater. 2009;19:3843–3854. [Google Scholar]
- 6.Mitragotri S, Lahann J. Physical Approaches to Biomaterial Design. Nat Mater. 2009;8:15–23. doi: 10.1038/nmat2344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dobrovolskaia MA, McNeil SE. Immunological Properties of Engineered Nanomaterials. Nat Nanotechnol. 2007;2:469–478. doi: 10.1038/nnano.2007.223. [DOI] [PubMed] [Google Scholar]
- 8.Howes PD, Chandrawati R, Stevens MM. Colloidal Nanoparticles as Advanced Biological Sensors. Science. 2014;346:1247390. doi: 10.1126/science.1247390. [DOI] [PubMed] [Google Scholar]
- 9.Etheridge ML, Campbell SA, Erdman AG, Haynes CL, Wolf SM, McCullough J. The Big Picture on Nanomedicine: The State of Investigational and Approved Nanomedicine Products. Nanomedicine. 2013;9:1–14. doi: 10.1016/j.nano.2012.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mirkin CA, Letsinger RL, Mucic RC, Storhoff JJ. A DNA-Based Method for Rationally Assembling Nanoparticles into Macroscopic Materials. Nature. 1996;382:607–609. doi: 10.1038/382607a0. [DOI] [PubMed] [Google Scholar]
- 11.Cutler JI, Auyeung E, Mirkin CA. Spherical Nucleic Acids. J Am Chem Soc. 2012;134:1376–1391. doi: 10.1021/ja209351u. [DOI] [PubMed] [Google Scholar]
- 12.Rosi NL, Giljohann DA, Thaxton CS, Lytton-Jean AK, Han MS, Mirkin CA. Oligonucleotide-Modified Gold Nanoparticles for Intracellular Gene Regulation. Science. 2006;312:1027–1030. doi: 10.1126/science.1125559. [DOI] [PubMed] [Google Scholar]
- 13.Lytton-Jean AK, Mirkin CA. A Thermodynamic Investigation into the Binding Properties of DNA Functionalized Gold Nanoparticle Probes and Molecular Fluorophore Probes. J Am Chem Soc. 2005;127:12754–12755. doi: 10.1021/ja052255o. [DOI] [PubMed] [Google Scholar]
- 14.Jensen SA, Day ES, Ko CH, Hurley LA, Luciano JP, Kouri FM, Merkel TJ, Luthi AJ, Patel PC, Cutler JI. Spherical Nucleic Acid Nanoparticle Conjugates as an RNAi-Based Therapy for Glioblastoma. Sci Transl Med. 2013;5:209ra152. doi: 10.1126/scitranslmed.3006839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zheng D, Giljohann DA, Chen DL, Massich MD, Wang XQ, Iordanov H, Mirkin CA, Paller AS. Topical Delivery of siRNA-Based Spherical Nucleic Acid Nanoparticle Conjugates for Gene Regulation. Proc Natl Acad Sci US A. 2012;109:11975–11980. doi: 10.1073/pnas.1118425109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Seferos DS, Giljohann DA, Hill HD, Prigodich AE, Mirkin CA. Nano-Flares: Probes for Transfection and mRNA Detection in Living Cells. J Am Chem Soc. 2007;129:15477–15479. doi: 10.1021/ja0776529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Halo TL, McMahon KM, Angeloni NL, Xu Y, Wang W, Chinen AB, Malin D, Strekalova E, Cryns VL, Cheng C. NanoFlares for the Detection, Isolation, and Culture of Live Tumor Cells from Human Blood. Proc Natl Acad Sci US A. 2014;111:17104–17109. doi: 10.1073/pnas.1418637111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Radovic-Moreno AF, Chernyak N, Mader CC, Nallagatla S, Kang RS, Hao L, Walker DA, Halo TL, Merkel TJ, Rische CH. Immunomodulatory Spherical Nucleic Acids. Proc Natl Acad Sci US A. 2015;112:3892–3897. doi: 10.1073/pnas.1502850112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yin H, Kanasty RL, Eltoukhy AA, Vegas AJ, Dorkin JR, Anderson DG. Non-viral Vectors for Gene-Based Therapy. Nat Rev Genet. 2014;15:541–555. doi: 10.1038/nrg3763. [DOI] [PubMed] [Google Scholar]
- 20.Sahay G, Querbes W, Alabi C, Eltoukhy A, Sarkar S, Zurenko C, Karagiannis E, Love K, Chen D, Zoncu R. Efficiency of siRNA Delivery by Lipid Nanoparticles Is Limited by Endocytic Recycling. Nat Biotechnol. 2013;31:653–658. doi: 10.1038/nbt.2614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lovell JF, Jin CS, Huynh E, Jin H, Kim C, Rubinstein JL, Chan WC, Cao W, Wang LV, Zheng G. Porphysome Nanovesicles Generated by Porphyrin Bilayers for Use as Multimodal Biophotonic Contrast Agents. Nat Mater. 2011;10:324–332. doi: 10.1038/nmat2986. [DOI] [PubMed] [Google Scholar]
- 22.Dahlman JE, Barnes C, Khan OF, Thiriot A, Jhunjunwala S, Shaw TE, Xing Y, Sager HB, Sahay G, Speciner L. In Vivo Endothelial siRNA Delivery Using Polymeric Nanoparticles with Low Molecular Weight. Nat Nanotechnol. 2014;9:648–655. doi: 10.1038/nnano.2014.84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Dong Y, Love KT, Dorkin JR, Sirirungruang S, Zhang Y, Chen D, Bogorad RL, Yin H, Chen Y, Vegas AJ. Lipopeptide Nanoparticles for Potent and Selective siRNA Delivery in Rodents and Nonhuman Primates. Proc Natl Acad Sci US A. 2014;111:3955–3960. doi: 10.1073/pnas.1322937111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yin H, Xue W, Chen S, Bogorad RL, Benedetti E, Grompe M, Koteliansky V, Sharp PA, Jacks T, Anderson DG. Genome Editing with Cas9 in Adult Mice Corrects a Disease Mutation and Phenotype. Nat Biotechnol. 2014;32:551–553. doi: 10.1038/nbt.2884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Shi J, Xiao Z, Votruba AR, Vilos C, Farokhzad OC. Differentially Charged Hollow Core/Shell Lipid–Polymer– Lipid Hybrid Nanoparticles for Small Interfering RNA Delivery. Angew Chem, Int Ed. 2011;123:7165–7169. doi: 10.1002/anie.201101554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Shi J, Xu Y, Xu X, Zhu X, Pridgen E, Wu J, Votruba AR, Swami A, Zetter BR, Farokhzad OC. Hybrid Lipid–Polymer Nanoparticles for Sustained siRNA Delivery and Gene Silencing. Nanomedicine. 2014;10:897–900. doi: 10.1016/j.nano.2014.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhu X, Xu Y, Solis LM, Tao W, Wang L, Behrens C, Xu X, Zhao L, Liu D, Wu J, et al. Long-Circulating siRNA Nanoparticles for Validating Prohibitin1-Targeted Non-Small Cell Lung Cancer Treatment. Proc Natl Acad Sci USA. 2015 doi: 10.1073/pnas.1505629112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Cabral H, Kataoka K. Progress of Drug-Loaded Polymeric Micelles into Clinical Studies. J Controlled Release. 2014;190:465–476. doi: 10.1016/j.jconrel.2014.06.042. [DOI] [PubMed] [Google Scholar]
- 29.Miura Y, Takenaka T, Toh K, Wu S, Nishihara H, Kano MR, Ino Y, Nomoto T, Matsumoto Y, Koyama H. Cyclic RGD-Linked Polymeric Micelles for Targeted Delivery of Platinum Anticancer Drugs to Glioblastoma through the Blood–Brain Tumor Barrier. ACS Nano. 2013;7:8583–8592. doi: 10.1021/nn402662d. [DOI] [PubMed] [Google Scholar]
- 30.Ahn J, Miura Y, Yamada N, Chida T, Liu X, Kim A, Sato R, Tsumura R, Koga Y, Yasunaga M. Antibody Fragment-Conjugated Polymeric Micelles Incorporating Platinum Drugs for Targeted Therapy of Pancreatic Cancer. Biomaterials. 2015;39:23–30. doi: 10.1016/j.biomaterials.2014.10.069. [DOI] [PubMed] [Google Scholar]
- 31.Kabanov AV, Chekhonin V, Alakhov VY, Batrakova E, Lebedev A, Melik-Nubarov N, Arzhakov S, Levashov A, Morozov G, Severin E. The Neuroleptic Activity of Haloperidol Increases after Its Solubilization in Surfactant Micelles: Micelles as Microcontainers for Drug Targeting. FEBS Lett. 1989;258:343–345. doi: 10.1016/0014-5793(89)81689-8. [DOI] [PubMed] [Google Scholar]
- 32.Alakhova DY, Kabanov AV. Pluronics and MDR Reversal: An Update. Mol Pharmaceutics. 2014;11:2566–2578. doi: 10.1021/mp500298q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Valle JW, Armstrong A, Newman C, Alakhov V, Pietrzynski G, Brewer J, Campbell S, Corrie P, Rowinsky EK, Ranson M. A Phase 2 Study of SP1049C, Doxorubicin in P-Glycoprotein-Targeting Pluronics, in Patients with Advanced Adenocarcinoma of the Esophagus and Gastroesophageal Junction. Invest New Drugs. 2011;29:1029–1037. doi: 10.1007/s10637-010-9399-1. [DOI] [PubMed] [Google Scholar]
- 34.Han Y, He Z, Schulz A, Bronich TK, Jordan R, Luxenhofer R, Kabanov AV. Synergistic Combinations of Multiple Chemotherapeutic Agents in High Capacity Poly(2-oxazoline) Micelles. Mol Pharmaceutics. 2012;9:2302–2313. doi: 10.1021/mp300159u. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.He Z, Schulz A, Wan X, Seitz J, Bludau H, Alakhova DY, Darr DB, Perou CM, Jordan R, Ojima I. Poly(2-oxazoline) Based Micelles with High Capacity for 3rd Generation Taxoids: Preparation, In Vitro and In Vivo Evaluation. J Controlled Release. 2015;208:67–75. doi: 10.1016/j.jconrel.2015.02.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Natarajan JV, Ang M, Darwitan A, Chattopadhyay S, Wong TT, Venkatraman SS. Nanomedicine for Glaucoma: Liposomes Provide Sustained Release of Latanoprost in the Eye. Int J Nanomed. 2012;7:123–131. doi: 10.2147/IJN.S25468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Natarajan JV, Darwitan A, Barathi VA, Ang M, Htoon HM, Boey F, Tam KC, Wong TT, Venkatraman SS. Sustained Drug Release in Nanomedicine: A Long-Acting Nanocarrier-Based Formulation for Glaucoma. ACS Nano. 2014;8:419–429. doi: 10.1021/nn4046024. [DOI] [PubMed] [Google Scholar]
- 38.Tan YF, Mundargi RC, Chen MHA, Lessig J, Neu B, Venkatraman SS, Wong TT. Layer-by-Layer Nanoparticles as an Efficient siRNA Delivery Vehicle for SPARC Silencing. Small. 2014;10:1790–1798. doi: 10.1002/smll.201303201. [DOI] [PubMed] [Google Scholar]
- 39.Huang H, Pierstorff E, Osawa E, Ho D. Active Nanodiamond Hydrogels for Chemotherapeutic Delivery. Nano Lett. 2007;7:3305–3314. doi: 10.1021/nl071521o. [DOI] [PubMed] [Google Scholar]
- 40.Manus LM, Mastarone DJ, Waters EA, Zhang XQ, Schultz-Sikma EA, MacRenaris KW, Ho D, Meade TJ. Gd(III)-Nanodiamond Conjugates for MRI Contrast Enhancement. Nano Lett. 2010;10:484–489. doi: 10.1021/nl903264h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Chow EK, Zhang XQ, Chen M, Lam R, Robinson E, Huang H, Schaffer D, Osawa E, Goga A, Ho D. Nanodiamond Therapeutic Delivery Agents Mediate Enhanced Chemoresistant Tumor Treatment. Sci Transl Med. 2011;3:73ra21. doi: 10.1126/scitranslmed.3001713. [DOI] [PubMed] [Google Scholar]
- 42.Moore L, Chow EKH, Osawa E, Bishop JM, Ho D. Diamond-Lipid Hybrids Enhance Chemotherapeutic Tolerance and Mediate Tumor Regression. Adv Mater. 2013;25:3532–3541. doi: 10.1002/adma.201300343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wang X, Low XC, Hou W, Abdullah LN, Toh TB, Mohd Abdul Rashid M, Ho D, Chow EKH. Epirubicin-Adsorbed Nanodiamonds Kill Chemoresistant Hepatic Cancer Stem Cells. ACS Nano. 2014;8:12151–12166. doi: 10.1021/nn503491e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wang H, Lee DK, Chen KY, Chen JY, Zhang K, Silva A, Ho CM, Ho D. Mechanism-Independent Optimization of Combinatorial Nanodiamond and Unmodified Drug Delivery Using a Phenotypically Driven Platform Technology. ACS Nano. 2015;9:3332–3344. doi: 10.1021/acsnano.5b00638. [DOI] [PubMed] [Google Scholar]
- 45.Paithankar D, Hwang BH, Munavalli G, Kauvar A, Lloyd J, Blomgren R, Faupel L, Meyer T, Mitragotri S. Ultrasonic Delivery of Silica–Gold Nanoshells for Photothermolysis of Sebaceous Glands in Humans: Nanotechnology from the Bench to Clinic. J Controlled Release. 2015;206:30–36. doi: 10.1016/j.jconrel.2015.03.004. [DOI] [PubMed] [Google Scholar]
- 46.Tezel A, Mitragotri S. Interactions of Inertial Cavitation Bubbles with Stratum Corneum Lipid Bilayers during Low-Frequency Sonophoresis. Biophys J. 2003;85:3502–3512. doi: 10.1016/S0006-3495(03)74770-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Chiappini C, Martinez JO, De Rosa E, Almeida CS, Tasciotti E, Stevens MM. Biodegradable Nanoneedles for Localized Delivery of Nanoparticles in Vivo: Exploring the Biointerface. ACS Nano. 2015;9:5500–5509. doi: 10.1021/acsnano.5b01490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Chiappini C, De Rosa E, Martinez J, Liu X, Steele J, Stevens M, Tasciotti E. Biodegradable Silicon Nanoneedles Delivering Nucleic Acids Intracellularly Induce Localized In Vivo Neovascularization. Nat Mater. 2015;14:532–539. doi: 10.1038/nmat4249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.De La Rica R, Stevens MM. Plasmonic ELISA for the Ultrasensitive Detection of Disease Biomarkers with the Naked Eye. Nat Nanotechnol. 2012;7:821–824. doi: 10.1038/nnano.2012.186. [DOI] [PubMed] [Google Scholar]
- 50.Chapman R, Lin Y, Burnapp M, Bentham A, Hillier D, Zabron A, Khan S, Tyreman M, Stevens MM. Multivalent Nanoparticle Networks Enable Point-of-Care Detection of Human Phospholipase-A2 in Serum. ACS Nano. 2015;9:2565–2573. doi: 10.1021/nn5057595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Dewitte H, Verbeke R, Breckpot K, De Smedt SC, Lentacker I. Nanoparticle Design To Induce Tumor Immunity and Challenge the Suppressive Tumor Microenvironment. Nano Today. 2014;9:743–758. [Google Scholar]
- 52.Gough DR, Cotter TG. Hydrogen Peroxide: A Jekyll and Hyde Signalling Molecule. Cell Death Dis. 2011;2:e213. doi: 10.1038/cddis.2011.96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lobo V, Patil A, Phatak A, Chandra N. Free Radicals, Antioxidants and Functional Foods: Impact on Human Health. Pharmacogn Rev. 2010;4:118–126. doi: 10.4103/0973-7847.70902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Sun X, Guo S, Liu Y, Sun S. Dumbbell-like PtPd–Fe3O4 Nanoparticles for Enhanced Electrochemical Detection of H2O2. Nano Lett. 2012;12:4859–4863. doi: 10.1021/nl302358e. [DOI] [PubMed] [Google Scholar]
- 55.Zhu H, Sigdel A, Zhang S, Su D, Xi Z, Li Q, Sun S. Core/Shell Au/MnO Nanoparticles Prepared through Controlled Oxidation of AuMn as an Electrocatalyst for Sensitive H2O2 Detection. Angew Chem, Int Ed. 2014;53:12508–12512. doi: 10.1002/anie.201406281. [DOI] [PubMed] [Google Scholar]
- 56.Sun X, Cai W, Chen X. Positron Emission Tomography Imaging Using Radiolabeled Inorganic Nanomaterials. Acc Chem Res. 2015;48:286–294. doi: 10.1021/ar500362y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Liu X, Wang S. Three-Dimensional Nano-Biointerface as a New Platform for Guiding Cell Fate. Chem Soc Rev. 2014;43:2385–2401. doi: 10.1039/c3cs60419e. [DOI] [PubMed] [Google Scholar]
- 58.Ma L, Yang G, Wang N, Zhang PC, Guo FY, Meng JX, Zhang FL, Hu ZJ, Wang ST, Zhao Y. Trap Effect of Three-Dimensional Fibers Network for High Efficient Cancer-Cell Capture. Adv Healthcare Mater. 2015;4:838–843. doi: 10.1002/adhm.201400650. [DOI] [PubMed] [Google Scholar]
- 59.Zhang P, Chen L, Xu T, Liu H, Liu X, Meng J, Yang G, Jiang L, Wang S. Programmable Fractal Nanostructured Interfaces for Specific Recognition and Electrochemical Release of Cancer Cells. Adv Mater. 2013;25:3566–3570. doi: 10.1002/adma.201300888. [DOI] [PubMed] [Google Scholar]
- 60.Liu X, Chen L, Liu H, Yang G, Zhang P, Han D, Wang S, Jiang L. Bio-Inspired Soft Polystyrene Nanotube Substrate for Rapid and Highly Efficient Breast Cancer Cell Capture. NPG Asia Mater. 2013;5:e63. [Google Scholar]
- 61.Liu H, Liu X, Meng J, Zhang P, Yang G, Su B, Sun K, Chen L, Han D, Wang S, Jiang L. Hydrophobic Interaction-Mediated Capture and Release of Cancer Cells on Thermoresponsive Nanostructured Surfaces. Adv Mater. 2013;25:922–927. doi: 10.1002/adma.201203826. [DOI] [PubMed] [Google Scholar]
- 62.Liu HL, Li YY, Sun K, Fan JB, Zhang PC, Meng JX, Wang ST, Jiang L. Dual-Responsive Surfaces Modified with Phenylboronic Acid-Containing Polymer Brush To Reversibly Capture and Release Cancer Cells. J Am Chem Soc. 2013;135:7603–7609. doi: 10.1021/ja401000m. [DOI] [PubMed] [Google Scholar]
- 63.Wang S, Liu K, Liu J, Yu ZTF, Xu X, Zhao L, Lee T, Lee EK, Reiss J, Lee YK, et al. Highly Efficient Capture of Circulating Tumor Cells by Using Nanostructured Silicon Substrates with Integrated Chaotic Micromixers. Angew Chem, Int Ed. 2011;50:3084–3088. doi: 10.1002/anie.201005853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Duan R, Zuo X, Wang S, Quan X, Chen D, Chen Z, Jiang L, Fan C, Xia F. Lab in a Tube: Ultrasensitive Detection of MicroRNAs at the Single-Cell Level and in Breast Cancer Patients Using Quadratic Isothermal Amplification. J Am Chem Soc. 2013;135:4604–4607. doi: 10.1021/ja311313b. [DOI] [PubMed] [Google Scholar]
- 65.Liu W, Thomopoulos S, Xia Y. Electrospun Nanofibers for Regenerative Medicine. Adv Healthcare Mater. 2012;1:10–25. doi: 10.1002/adhm.201100021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Xie J, Liu W, MacEwan MR, Bridgman PC, Xia Y. Neurite Outgrowth on Electrospun Nanofibers with Uniaxial Alignment: The Effects of Fiber Density, Surface Coating, and Supporting Substrate. ACS Nano. 2014;8:1878–1885. doi: 10.1021/nn406363j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Smith L, Xia Y, Galatz LM, Genin GM, Thomopoulos S. Tissue-Engineering Strategies for the Tendon/Ligament-to-Bone Insertion. Connect Tissue Res. 2012;53:95–105. doi: 10.3109/03008207.2011.650804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Xie J, MacEwan MR, Ray WZ, Liu W, Siewe DY, Xia Y. Radially Aligned, Electrospun Nanofibers as Dural Substitutes for Wound Closure and Tissue Regeneration Applications. ACS Nano. 2010;4:5027–5036. doi: 10.1021/nn101554u. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Lang N, Pereira MJ, Lee Y, Friehs I, Vasilyev NV, Feins EN, Ablasser K, O’Cearbhaill ED, Xu C, Fabozzo A, et al. A Blood-Resistant Surgical Glue for Minimally Invasive Repair of Vessels and Heart Defects. Sci Transl Med. 2014;6:218ra6. doi: 10.1126/scitranslmed.3006557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Yang SY, O’Cearbhaill ED, Sisk GC, Park KM, Cho WK, Villiger M, Bouma BE, Pomahac B, Karp JM. A Bio-Inspired Swellable Microneedle Adhesive for Mechanical Interlocking with Tissue. Nat Commun. 2013;4:1702. doi: 10.1038/ncomms2715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Cho WK, Ankrum JA, Guo D, Chester SA, Yang SY, Kashyap A, Campbell GA, Wood RJ, Rijal RK, Karnik R, et al. Microstructured Barbs on the North American Porcupine Quill Enable Easy Tissue Penetration and Dificult Removal. Proc Natl Acad Sci US A. 2012;109:21289–21294. doi: 10.1073/pnas.1216441109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Liu Y, Lim J, Teoh SH. Review: Development of Clinically Relevant Scaffolds for Vascularised Bone Tissue Engineering. Biotechnol Adv. 2013;31:688–705. doi: 10.1016/j.biotechadv.2012.10.003. [DOI] [PubMed] [Google Scholar]
- 73.Lim J, Chong MSK, Chan JKY, Teoh SH. Polymer Powder Processing of Cryomilled Polycaprolactone for Solvent-Free Generation of Homogeneous Bioactive Tissue Engineering Scaffolds. Small. 2014;10:2495–2502. doi: 10.1002/smll.201302389. [DOI] [PubMed] [Google Scholar]
- 74.Peer D, Karp JM, Hong S, Farokhzad OC, Margalit R, Langer R. Nanocarriers as an Emerging Platform for Cancer Therapy. Nat Nanotechnol. 2007;2:751–760. doi: 10.1038/nnano.2007.387. [DOI] [PubMed] [Google Scholar]
- 75.Chow EKH, Ho D. Cancer Nanomedicine: From Drug Delivery to Imaging. Sci Transl Med. 2013;5:216rv4. doi: 10.1126/scitranslmed.3005872. [DOI] [PubMed] [Google Scholar]
- 76.Godwin H, Nameth C, Avery D, Bergeson LL, Bernard D, Beryt E, Boyes W, Brown S, Clippinger AJ, Cohen Y, et al. Nanomaterial Categorization for Assessing Risk Potential to Facilitate Regulatory Decision-Making. ACS Nano. 2015;9:3409–3417. doi: 10.1021/acsnano.5b00941. [DOI] [PubMed] [Google Scholar]
- 77.Mura S, Nicolas J, Couvreur P. Stimuli-Responsive Nanocarriers for Drug Delivery. Nat Mater. 2013;12:991–1003. doi: 10.1038/nmat3776. [DOI] [PubMed] [Google Scholar]
- 78.Pacardo DB, Ligler FS, Gu Z. Programmable Nanomedicine: Synergistic and Sequential Drug Delivery Systems. Nanoscale. 2015;7:3381–3391. doi: 10.1039/c4nr07677j. [DOI] [PubMed] [Google Scholar]
- 79.Lu Y, Sun W, Gu Z. Stimuli-Responsive Nanomaterials for Therapeutic Protein Delivery. J Controlled Release. 2014;194:1–19. doi: 10.1016/j.jconrel.2014.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Gu Z, Aimetti AA, Wang Q, Dang TT, Zhang Y, Veiseh O, Cheng H, Langer RS, Anderson DG. Injectable Nano-network for Glucose-Mediated Insulin Delivery. ACS Nano. 2013;7:4194–4201. doi: 10.1021/nn400630x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Mo R, Jiang T, DiSanto R, Tai W, Gu Z. ATP-Triggered Anticancer Drug Delivery. Nat Commun. 2014;5:3364. doi: 10.1038/ncomms4364. [DOI] [PubMed] [Google Scholar]
- 82.Gu Z, Dang TT, Ma M, Tang BC, Cheng H, Jiang S, Dong Y, Zhang Y, Anderson DG. Glucose-Responsive Microgels Integrated with Enzyme Nanocapsules for Closed-Loop Insulin Delivery. ACS Nano. 2013;7:6758–6766. doi: 10.1021/nn401617u. [DOI] [PubMed] [Google Scholar]
- 83.Godwin H, Nameth C, Avery D, Bergeson LL, Bernard D, Beryt E, Boyes W, Brown S, Clippinger AJ, Cohen Y, et al. Nanomaterial Categorization for Assessing Risk Potential to Facilitate Regulatory Decision-Making. ACS Nano. 2015;9:3409–3417. doi: 10.1021/acsnano.5b00941. [DOI] [PubMed] [Google Scholar]
- 84.Mitragotri S, Burke PA, Langer R. Overcoming the Challenges in Administering Biopharmaceuticals: Formulation and Delivery Strategies. Nat Rev Drug Discovery. 2014;13:655–672. doi: 10.1038/nrd4363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Chertok B, Webber MJ, Succi MD, Langer R. Drug Delivery Interfaces in the 21st Century: From Science Fiction Ideas to Viable Technologies. Mol Pharmaceutics. 2013;10:3531–3543. doi: 10.1021/mp4003283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Hammond PT. A Growing Place for Nano in Medicine. ACS Nano. 2014;8:7551–7552. doi: 10.1021/nn504577x. [DOI] [PubMed] [Google Scholar]
- 87.Khademhosseini A, Peppas NA. Micro- and Nanoengineering of Biomaterials for Healthcare Applications. Adv Healthc Mater. 2013;2:10–12. doi: 10.1002/adhm.201200444. [DOI] [PubMed] [Google Scholar]