Abstract
Loss of expression of cadherin-11 protein is correlated with a loss of epithelial phenotype and a gain in tumor cell proliferation and invasion. It has been hypothesized that cadherin-11 may be a molecular marker for a more aggressive subtype of breast cancer. The present study examined the expression of the mesenchymal gene/protein cadherin-11 in malignant, benign and healthy breast cancer samples. A paraffin-embedded tissue microarray of both malignant and benign/healthy breast tumor was used. Clinicopathological parameters, including age, grading, tumor size, hormone receptors and HER2 receptors status were obtained from patient medical records. Expression of cadherin-11 was analyzed using the monoclonal mouse anti cadherin-11 IgG2B clone. Total RNA was extracted from each breast cancer sample and subjected to semi-quantitative RT-PCR analysis for cadherin-11. Cadherin-11 was detected in 80/82 malignant breast cancer samples and in 33/70 non-malignant tissue samples. Cadherin-11 expression was observed to be predominantly localized to the membrane of tumor cells. When compared to healthy breast tissue biopsies, both cadherin-11 mRNA and protein were demonstrated to be significantly overexpressed in breast carcinoma (P=0.040 and P<0.0001, respectively). Within malignant tumors, however, protein expression was not identified to be associated with other clinicopathological parameters. Our results indicate that cadherin-11 expression is upregulated in malignant human breast cancer.
Keywords: cadherin-11, breast cancer, normal breast tissue
Introduction
Cadherins belong to a family of transmembraneous adhesion molecules that are important in maintaining cell polarity and tissue integrity (1). They can mediate calcium-dependent cell-to-cell adhesion by interacting with the cytoplasmatic catenins, α, β, and γ (2,3). The catenins link cadherins to the actin cytoskeleton, but also have signaling functions of their own. Over the years, >20 types of cadherins have been identified and characterized, including the original E-, P- and N-cadherin (Type I), and cadherins 5 to 12 (Type II) (1–4). While the two subgroups share structural similarities, they exhibit surprisingly little sequence homology. Cadherins are involved in normal mammary gland development and function, and they appear to influence breast cancer and its clinical outcome (1,3–6). Berx and van Roy (7) reviewed the role of cadherins in malignant disease, and it has been demonstrated that loss of E-cadherin expression was associated with increased invasiveness and decreased differentiation. Interestingly, re-induction of E-cadherin in invasive breast cancer cells did not result in a less aggressive behavior in vitro, thereby suggesting that E-cadherin is rather an indicator of a more invasive phenotype than a causative factor (3,5,8).
Cadherin-11, also known as OB-cadherin was first identified in mouse osteoblasts and is normally expressed in cells with a mesenchymal phenotype, including the mesenchyme of the kidney and brain during development (8,9). Cadherin-11 is also expressed in cartilage synoviocytes and is an important mediator of the synoviocyte reaction that characterizes rheumatoid arthritis (10). In the adult, cadherin-11 is strongly expressed in bone as well as certain cancers that metastasize to bone (11). While the exact expression profile of cadherin-11 in healthy mammary gland is not known, it has been shown that it interacts with the fibroblast growth factor (FGF) signaling pathway (2,8,12), and thus modulates the response to growth factors. Cadherin-11 is typically expressed in many types of condensing mesenchyme and when expressed in epithelium, EMT is thought to have occurred (12–17). It may also provide the cell with an ability to establish itself into the bone environment (5,11). The majority of patients that succumb to breast (or prostate) cancer have metastases to the skeleton. It is possible that these cadherin-11-expressing tumor cells activate either osteoclasts or osteoblasts, depending on the type of cancer metastasis, leading to bone remodeling (11).
While the precise role of cadherins in cancer remains unclear, they are important in the basic events and processes in breast cancer tumorigenesis (4,12). Several events in tumorigenesis are strongly connected to changes in cadherin expression (14–18). One example is cadherin switching, where cadherins change from those expressed in epithelial cells to those predominant in mesenchymal cells (8). This event is part of a process that is vital to malignant change, the epithelial-mesenchymal transition (EMT). EMT is a key biologic process that was initially identified as a developmental program that enables polarized epithelial cells to acquire a motile mesenchymal phenotype (13–17,19,20). This transition results in a more invasive and metastatic phenotype (15–17,19). Research suggests that cadherin switching is required for increased motility but not for the morphological changes that accompany EMT (13,19). The reverse process of EMT, the mesenchymal-epithelial transition (MET) involves the conversion of mesenchymal cells to their epithelial derivatives (14). In carcinoma progression, reactivation of the EMT program promotes tumor metastasis by driving tumor cell invasion and enhancing tumor cell survival (16,17). These changes are highly dynamic and many intermediate phenotypes exist. Dubois-Marshall et al (16) described two distinct possible mechanisms of EMT arising in breast cancer, one of which is uncoupled from cadherin switching. Some difficulty lies in the fact that it is not yet fully understood how cadherins expression profiles change in EMT. This emanates from the fact that there are multiple ways to regulate cadherin expression, and many, but not all of these overlap (2). Recently, cadherins were demonstrated to regulate stem cell maintenance and differentiation (20). The use of mesenchymal stem cells for tissue repair requires the migration and homing to the site of damaged tissue and it has been shown that both the migratory and proliferation potential of these cells are affected by cadherin-2 and cadherin-11 (20).
Pishvaian et al (6) have demonstrated that cadherin-11 mRNA and protein, as well as a cadherin-11 variant mRNA are expressed in invasive and poorly differentiated breast cancer cell lines. In these cells, cadherin-11 is localized to the cell membrane in a detergent-soluble complex, where it associates with α and β-catenin, and may facilitate tumor cell invasion and metastasis. Assefnia et al and Dakshanamurthy et al (21,22) demonstrated that cadherin-11 is increased in early stages of human breast cancer and in other malignancies. When compared to healthy breast tissue, cadherin-11 was markedly elevated in DCIS and also in the stroma of invasive breast cancers compared to normal stroma. While this seems counter-intuitive at first, it illustrates that the functional diversity of cadherins in physiological cell is also reflected in processes connected to malignant disease.
There is paucity of studies evaluating cadherin-11 expression in human invasive breast cancer. The aim of the present study was to investigate cadherin-11 expression in malignant breast tissue samples and benign and/or healthy breast tissue samples. The expression was then correlated with several clinicopathological parameters.
Materials and methods
Patient samples
Human breast tissue microarray (TMA) slides were obtained from US Biomax Inc. (Rockville, MD, USA). These TMAs consists of malignant and benign breast tumors, and healthy breast tissue adjacent to a malignant tumor or from women undergoing reduction mammoplasty. Clinicopathological information was obtained, including age, tumor grade, tumor size and histology. Hormone receptor status, e.g., estrogen, progesterone and HER2, as well as cadherin-11 expression were analyzed using immunohistochemistry (IHC).
Immunohistochemistry for cadherin-11
Tissue sections of paraffin-embedded formalin-fixed tissue blocks were deparaffinized with xylene for 5 min each, followed by two washes with 100% ethanol for 10 min. The slides were then incubated in 95% ethanol for another 10 min and washed with dH2O twice for 5 min. Antigen retrieval was performed by placing slides in 10 mmol/l citrate buffer (pH 6.0) and microwave treatment for 15 min. Tissue sections were cool down to room temperature (RT), washed in phosphate-buffered saline (PBS) and distilled water. Afterwards, sections were blocked with Ultra V Block (Lab Vision, Westinghouse Drive, Fremont, CA, USA) for 4 min. After a consecutive PBS wash, slides were incubated with the monoclonal Mouse anti cadherin-11 IgG2B Clone # 283416 Catalog Number: MAB1790 (R&D Systems). Negative controls were performed on all tissue sections by replacing primary antibodies with diluted isotype immunoglobulin (ImmunoCruz™ Staining system, Santa Cruz Biotechnology). Then the slides were incubated with goat anti-polyvalent and streptavidin-HRP (both Lab Vision) for 60 min, followed by an incubation with 3-amino-9-ethylcarbazole (AEC). Finally, slides were washed in PBS, counterstained with hematoxylin for 5 sec and cover-slipped.
Semi-quantitative reverse transcription polymerase chain reaction (RT-PCR) for Cadherin-11
A total of 0.5–1 µg of total RNA was extracted from each of the breast cancer samples, subsequently subjected to DNase (RNase-Free DNase Set; Qiagen, Hilden, Germany) treatment and then incubated with 0.5 µg/µl random hexamers (Promega Corp., Madison, WI, USA). The final volume was adjusted to 5 µl with diethyl pyrocarbonate-treated double distilled water (DEPC-treated ddH2O), before being heat-denatured at 70°C for 5 min and chilled on ice. The samples were then added to a reaction mix consisting of 4 µl of 5X RT-buffer (250 mM Tris-HCl, pH 8.3, 375 mM KCl, 15 mM MgCl2), 2 µl dNTP mix stock solution (10 mM each Pharmacia Biotech, Uppsala, Sweden), 1 µl RNase inhibitor (Applied Biosystems, Vienna, Austria), 1 µl dithiothreitol (DTT), and 1 µl MMLV (Moloney murine leukemia virus)-RT (200 U/µl, Amersham Bioscience Ltd.). The reaction mix was vortexed and centrifuged briefly before being incubated at 37°C for 1 h. The reaction was stopped by heating to 80°C for 10 min. The tubes were chilled briefly on ice before they were centrifuged and stored at −20°C.
PCR was performed by adding 20 µl reaction mix to 2.5 µl 10X PCR-buffer, 2 µl dNTP mix (10 mM each; New England Biolabs, Hertfordshire, UK), 0.25 µl primer (100 µM), 5 µl Taq polymerase (5 U/ml) to the cadherin-11 primers (primer sequences: Forward, 5′-ACCAGATGTCTGTGTCAGA-3′ and reverse, 3′-GTCATCCTTGTCATCTGCA-5′. The gene GAPDH (primer sequences: Forward, 5′-GAAGGTGAAGGTCGGAGTC-3′ and reverse, 3′-GAAGATGGTGATGGGATTTC-5′) was used as a reference for normalization. A total volume of 45 µl was reached by adding DEPC-treated ddH2O. Cycling conditions were as follows: Depending on the primers, 25–35 cycles were carried out of 94°C for 1 min, 68°C for 2 min, 72°C for 2 min, with an extension of 5 sec, with each subsequent cycle. ddH2O was used instead of total RNA for negative controls. Agarose gel electrophoresis was performed by adding 20 µl of each of the PCR products and subjecting them to 1.2% NuSieve® (Lonza Ltd., Basel, Switzerland) 3:1 agarose gel electrophoresis in 1X TBE buffer, separated by applying a constant voltage at 80 V for 1–2 h. DNA bands were then visualized by ethidium bromide, using UV transilluminator (Syngene®, Cambridge, UK). Band size was determined by a co-loaded DNA size marker.
Evaluation of immunohistochemical staining and statistical analysis
Immunostained slides were scored under a microscope (Olympus BX51; Olympus, Tokyo, Japan). The staining intensity of hormone receptors was scored according to Remmele et al (23). The HER2 receptor status has been evaluated according to standardized assessment (24). Only slides with a IHC 3+ status of HER2 receptors were categorized as positive. Chi-square and Student's t-test were used to compare cadherin-11 protein expression and age. Associations between cadherin-11 and clinical-pathological parameters were analyzed using Pearson's rho correlation test (2-sided). For all analyses, P<0.05 was considered to indicate a statistically significant difference. Data were analyzed using SAS version 8.1 (SAS Institute Inc., Cary, NC, USA).
Results
A total of 82 malignant tumor samples and 70 healthy breast tissue and benign breast lesions were analyzed by IHC and semi-quantitative RT-PCR. The patient tumor characteristics are shown in Table I. The median age of the patients with malignant tumors was 51 years, and median age of patients with benign or healthy tissue was 48 years. The difference in median age was not statistically significant. Of the malignant tumors, 75% (n=62) were infiltrating ductal carcinomas, and the remaining histological types included infiltrating lobular carcinomas (n=3) and otherwise specified (n=17). Regarding tumor size, 56% of malignant tumors were stage 2 (n=45), 24.4% were stage 1 (n=20), 19.5% were stage T3 (n=16), and 1% were stage 4 (n=1). The majority of tumors were grade 2 (53.7%, n=44), followed by grade 1 (18.3%, n=15) and Grade 3 (17%, n=14). In 9 cases (11%), the tumor grades were unknown. The estrogen receptor status was positive in 71% of the samples (n=58), negative in 19.5% of cases (n=16), with 9.8% (n=8) unknown. The progesterone receptor status was positive in 42 (51.2%) cases, negative in 26.8% (n=22) cases and 22% were unknown. The HER2 receptor status was positive in 22 cases (26.8%) were HER2 receptors evaluated as positive and negative in 60 cases (73.2%).
Table I.
Patients characteristics.
| Characteristic | Malignant tumor (n=82) | Benign tumors and/or normal tissue (n=70) | P-value |
|---|---|---|---|
| Median age (range), years | 51 (42–62) | 48 (41–55) | |
| Cadherin-11a, n (%) | <0.0001 | ||
| 0 | 2 (2.4) | 37 (52.9) | |
| + | 21 (25.6) | 13 (18.6) | |
| ++ | 34 (41.5) | 12 (17.1) | |
| +++ | 25 (30.5) | 8 (11.4) | |
| Histology, n (%) | N/A | ||
| Invasive ductal | 62 (75.6) | N/A | |
| Invasive lobular | 3 (3.7) | ||
| Other | 17 (20.7) | ||
| Tumor grade, n (%) | N/A | ||
| G1 | 15 (18.3) | ||
| G2 | 44 (53.7) | ||
| G3 | 14 (17.0) | ||
| Unknown | 9 (11.0) | N/A | |
| Tumor size, n (%) | N/A | ||
| T1 | 20 (24.4) | ||
| T2 | 45 (56.1) | ||
| T3 | 16 (19.5) | N/A | |
| T4 | 1 (1.2) | ||
| ER, n (%) | N/A | ||
| 0 | 16 (19.5) | ||
| 5% | 10 (12.2) | ||
| 5–10% | 8 (9.8) | ||
| >10% | 40 (48.8) | ||
| Unknown | 8 (9.8) | N/A | |
| PR, n (%) | N/A | ||
| 0 | 22 (26.8) | ||
| 5% | 12 (14.6) | ||
| 5–10% | 16 (19.5) | ||
| >10% | 14 (17.1) | ||
| Unknown | 18 (22.0) | N/A | |
| HER2, n (%) | N/A | ||
| Positiveb | 22 (26.8) | ||
| Negative | 60 (73.2) | N/A |
Cadherin 11 immunohistochemical scoring: Negative (0); low (+); intermediate (++); high positive (+++).
HER2 positive equals a score of IHC 3+. ER, estrogen receptor; PR, progesterone receptor; HER2, human epidermal growth factor receptor 2.
Fig. 1 shows the IHC results for cadherin-11. In malignant tissue samples, 25 cases (30.5%) exhibited strong positivity for cadherin-11, 34 cases (41.5%) had moderate positivity, and weak positivity in another 21 cases (25.6%). Of 82 cases, only two (2.4%) were negative for cadherin-11. As for benign/healthy samples, only 8 cases (11.4%) exhibited strong positivity for cadherin-11, 12 (17.1%) were moderate positive, and 13 (18.6%) were weak positive. However, more benign/normal tissues tested negative for cadherin-11 than malignant tumors (52.9 vs. 2.4%, respectively). This difference was statistically significant (P<0.0001).
Figure 1.
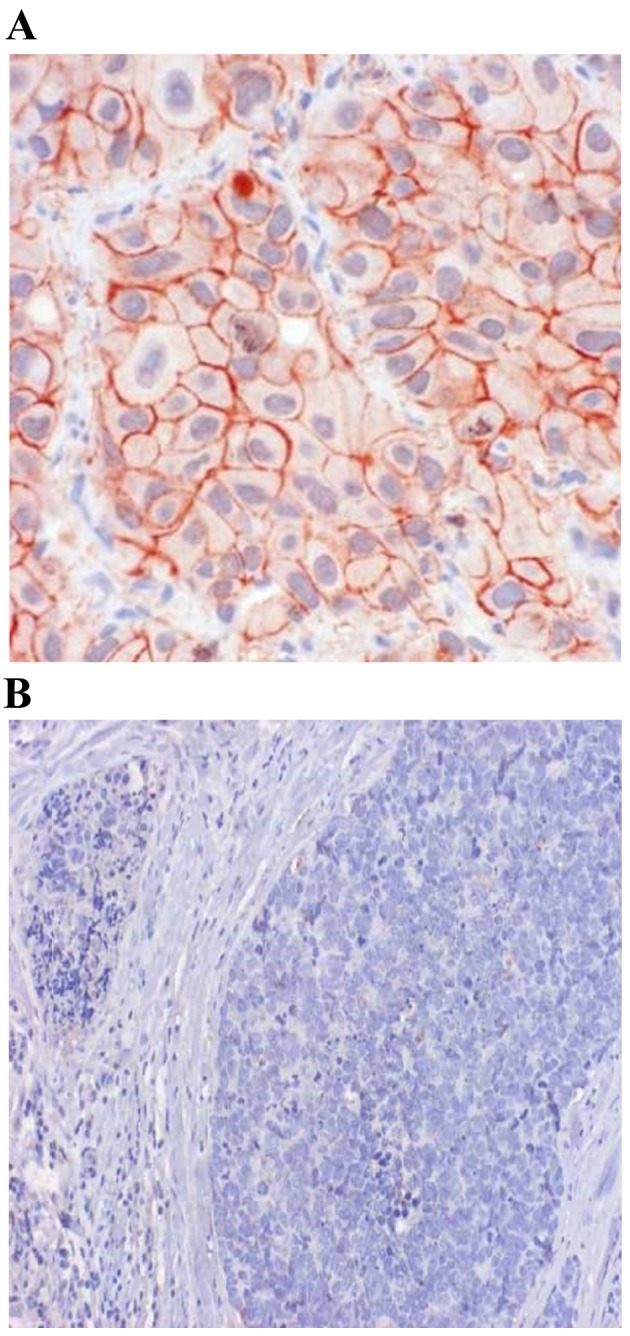
Cadherin-11 protein expression in (A), malignant breast cancer tissue and in (B) normal breast tissue. Immunohistochemical staining with MAB1790. Magnification, (A) ×400 and (B) ×100. Cadherin-11 is (A) localized on the cell membrane of malignant cells (red color), and (B) absent in normal tissue.
Correlations between cadherin-11 protein expression and other clinical-pathological parameters are shown in Table II. The expression of cadherin-11 protein was not correlated with patient age, tumor size, grading, or hormone receptors status.
Table II.
Correlation between cadherin-11 protein expression and clinicopathological parameters.
| Parameter | Age | Grading | Tumor size | ER | PR | HER2 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cadherin-11 | ||||||||||||
| Correlation coeff. | −0.109 | −0.034 | 0.340 | 0.034 | 0.029 | 0.128 | ||||||
| Sig. (2-tailed) | 0.331 | 0.762 | 0.701 | 0.760 | 0.795 | 0.252 | ||||||
| Age | ||||||||||||
| Correlation coeff. | 0.107 | 0.071 | −0.125 | −0.167 | −0.017 | |||||||
| Sig. (2-tailed) | 0.340 | 0.525 | 0.263 | 0.133 | 0.882 | |||||||
| Grading | ||||||||||||
| Correlation coeff. | −0.185 | 0.043 | 0.049 | 0.081 | ||||||||
| Sig. (2-tailed) | 0.097 | 0.703 | 0.664 | 0.470 | ||||||||
| Tumor size | ||||||||||||
| Correlation coeff. | 0.125 | 0.199 | 0.057 | |||||||||
| Sig. (2-tailed) | 0.173 | 0.074 | 0.610 | |||||||||
| ER | ||||||||||||
| Correlation coeff. | −0.169 | 0.036 | ||||||||||
| Sig. (2-tailed) | 0.129 | 0.748 | ||||||||||
| PR | ||||||||||||
| Correlation coeff. | 0.138 | |||||||||||
| Sig. (2-tailed) | 0.215 | |||||||||||
ER, estrogen receptor; PR, progesterone receptor; HER2, human epidermal growth factor receptor 2.
The expression of cadherin-11 mRNA in malignant tissues (black lanes 1–12) vs. benign/healthy tissues (white lanes 13–24) is shown in Fig. 2. The difference in cadherin-11 mRNA levels between malignant, and benign and/or healthy tissue samples was statistically significant (P=0.040).
Figure 2.
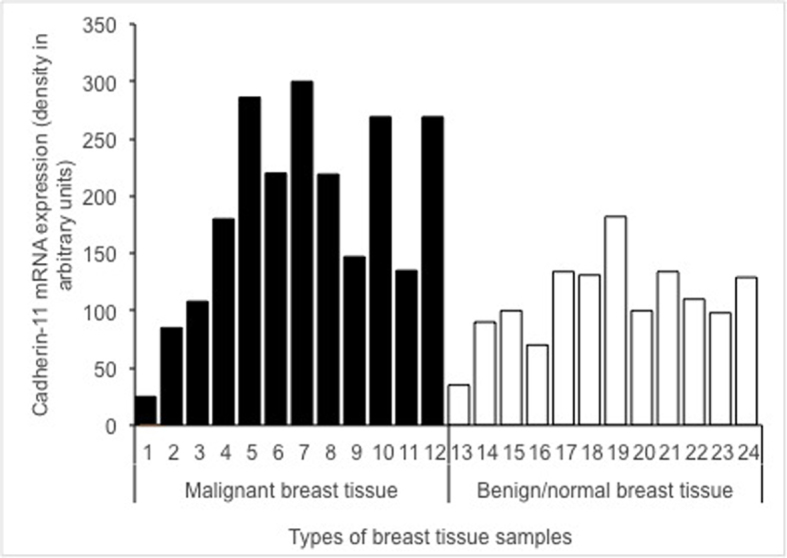
Cadherin-11 mRNA expression in malignant breast tissue samples (black lanes 1–12) and benign/normal breast tissue samples (white lanes 13–24). On the x-axis are each group of breast tissue samples and the y-axis presents the density in arbitrary units. The differences in mRNA expression between malignant, and benign and/or normal tissue samples were statistically significant (P=0.040).
Discussion
The present study demonstrates a significant difference in both mRNA transcription and protein expression of cadherin-11 in malignant breast tissue, when compared to benign and/or healthy tissue. These findings are consistent with past research and further emphasize the role of cadherins in the fundamental mechanics of the disease (3,5,6,13,19). Furthermore, this also points to the suspected role of cadherin-11 in EMT (12–17,19).
Our results are in agreement with the data of Pishvaian et al (6), who examined the expression of cadherin-11 in breast cancer cell lines and demonstrated that cadherin-11 mRNA and protein were expressed in the most invasive cell lines, but not in any of the noninvasive cell lines. Based on these results, it is anticipated that cadherin-11 expression may be well correlated with the invasive phenotype in cancer cells and could serve as a molecular marker for the more aggressive, invasive subset of breast tumors. Pishvaian et al (6) reported that cadherin-11 expression was significantly upregulated in malignant tissue samples and that it was localized on the cell membrane of the malignant cells, which is also in line with the results presented in Fig. 1. Similarly, the difference in mRNA expression in malignant and benign tissue samples was statistically significant (P=0.040) in the present study (Fig. 2). Cadherin-11 was preferentially expressed in basal-like breast cancer (13). The differences between expression of cadherin-11 protein and grading or hormone receptors status were not statistically significant in our study. There was also no correlation between estrogen and progesterone receptors in malignant breast tissue samples (Table II). The lack of correlation may be due to the median age of the sample group (51 years), since older, post-menopausal women are more likely to develop estrogen receptor positive breast cancer. Previous studies have demonstrated that there is no relationship between age and progesterone receptor positivity (25–27).
The present study was limited in several ways. Firstly, the control group consisted of tissue samples with undefined benign pathologies, which may influence the expression profile of cadherins. Secondly, the protein expression of cadherin-11 was performed using immunohistochemistry, which is subjective, and proper evaluation of the score is lacking. We also lacked clinical data on bone metastasis, which could have proven relevant in this study.
The current study succeeded in demonstrating that cadherin-11 expression is upregulated in invasive human breast cancer. We hypothesize that the expression confers a more mesenchymal cellular phenotype, which promotes invasion and metastasis in invasive tumors.
Cadherin-11 is a major therapeutic target in rheumatoid arthritis (10,21). Using a new proteochemometric computational drug repurposing method, it was identified that the drug celecoxib, a United States Food and Drug Administration approved drug, and 2,5-dimethyl-celecoxib, a celecoxib analogue without cyclooxygenase 2 inhibitory activity, had the structural potential to bind cadherin-11 (22). As cadherin-11 may be an important target in cancer progression (21,28), this finding could potentially translate into clinical application in cancer therapy. In conclusion, our results indicate that cadherin-11 expression is upregulated in malignant human breast cancer. Based on the fact that cadherin-11 is typically expressed in cells of mesenchymal origin, this suggests that EMT took place. These data suggest that cadherin-11 is important for malignant progression and is a potential therapeutic target in breast cancer.
References
- 1.Knudsen KA, Wheelock MJ. Cadherins and the mammary gland. J Cell Biochem. 2005;95:488–496. doi: 10.1002/jcb.20419. [DOI] [PubMed] [Google Scholar]
- 2.Andrews JL, Kim AC, Hens JR. The role ad function of cadherins in the mammary gland. Breast Cancer Res. 2012;14:203. doi: 10.1186/bcr3065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Farina AK, Bong YS, Feltes CM, Byers SW. Post-transcriptional regulation of cadherin-11 expression by GSK-3 and beta-catenin in prostate and breast cancer cells. PLoS One. 2009;4:e4797. doi: 10.1371/journal.pone.0004797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Albergaria A, Ribeiro AS, Vieira AF, Sousa B, Nobre AR, Seruca R, Schmitt FC, Paredes J. P-cadherin role in normal breast development and cancer. Int J Dev Biol. 2011;55:811–822. doi: 10.1387/ijdb.113382aa. [DOI] [PubMed] [Google Scholar]
- 5.Tamura D, Hiraga T, Myoui A, Yoshikawa H, Yoneda T. Cadherin-11-mediated interactions with bone marrow stromal/osteoblastic cells support selective colonization of breast cancer cells in bone. Int J Oncol. 2008;33:17–24. [PubMed] [Google Scholar]
- 6.Pishvaian MJ, Feltes CM, Thompson P, Bussemakers MJ, Schalken JA, Byers SW. Cadherin-11 is expressed in invasive breast cancer cell lines. Cancer Res. 1999;59:947–952. [PubMed] [Google Scholar]
- 7.Berx G, van Roy F. Involvement of members of the cadherin superfamily in cancer. Cold Spring Harb Perspect Biol. 2009;1:a003129. doi: 10.1101/cshperspect.a003129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Maeda M, Johnson KR, Wheelock MJ. Cadherin switching: Essential for behavioral but not morphological changes during epithelium-to-mesenchyme transition. J Cell Sci. 2005;118:873–887. doi: 10.1242/jcs.01634. [DOI] [PubMed] [Google Scholar]
- 9.Kalluri R, Weinberg RA. The basics of epithelial-mesenchymal transition. J Clin Invest. 2009;119:1420–1428. doi: 10.1172/JCI39104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lee DM, Kiener HP, Agarwal SK, Noss EH, Watts GF, Chisaka O, Takeichi M, Brenner MB. Cadherin-11 in synovial lining formation and pathology in arthritis. Science. 2007;315:1006–1010. doi: 10.1126/science.1137306. [DOI] [PubMed] [Google Scholar]
- 11.Huang CF, Lira C, Chu K, Bilen MA, Lee YC, Ye X, Kim SM, Ortiz A, Wu FL, Logothetis CJ, et al. Cadherin-11 increases migration and invasion of prostate cancer cells and enhances their interaction with osteoblasts. Cancer Res. 2010;70:4580–4589. doi: 10.1158/0008-5472.CAN-09-3016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Nieto MA. Epithelial-mesenchymal transitions in development and disease: Old views and new perspectives. Int J Dev Biol. 2009;53:1541–1547. doi: 10.1387/ijdb.072410mn. [DOI] [PubMed] [Google Scholar]
- 13.Sarrió D, Rodriguez-Pinilla SM, Hardisson D, Cano A, Moreno-Bueno G, Palacios J. Epithelial-mesenchymal transition in breast cancer relates to the basal-like phenotype. Cancer Res. 2008;68:989–997. doi: 10.1158/0008-5472.CAN-07-2017. [DOI] [PubMed] [Google Scholar]
- 14.Chao YL, Shepard CR, Wells A. Breast carcinoma cells re-express E-cadherin during mesenchymal to epithelial reverting transition. Mol Cancer. 2010;9:179. doi: 10.1186/1476-4598-9-179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Clevers H, Nusse R. Wnt/ß-catenin signaling and disease. Cell. 2012;149:1192–1205. doi: 10.1016/j.cell.2012.05.012. [DOI] [PubMed] [Google Scholar]
- 16.Dubois-Marshall S, Thomas JS, Faratian D, Harrison DJ, Katz E. Two possible mechanisms of epithelial to mesenchymal transition in invasive ductal breast cancer. Clin Exp Metastasis. 2011;28:811–818. doi: 10.1007/s10585-011-9412-x. [DOI] [PubMed] [Google Scholar]
- 17.Aceto N, Toner M, Maheswaran S, Haber D. En route to metastasis: Circulating tumor cell clusters and epithelial-to-mesenchymal transition. Trends in Cancer. 2015;1:44–52. doi: 10.1016/j.trecan.2015.07.006. [DOI] [PubMed] [Google Scholar]
- 18.Satcher RL, Pan T, Cheng CJ, Lee YC, Lin SC, Yu G, Li X, Hoang AG, Tamboli P, Jonasch E, et al. Cadherin-11 in renal cell carcinoma bone metastasis. PLoS One. 2014;9:e89880. doi: 10.1371/journal.pone.0089880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Li Y, Chao F, Huang B, Liu D, Kim J, Huang S. HOXC8 promotes breast tumorigenesis by transcriptionally facilitating cadherin-11 expression. Oncotarget. 2014;5:2596–2607. doi: 10.18632/oncotarget.1841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Alimperti S, Andreadis ST. CDH2 and CDH11 act as regulators of stem cell fate decisions. Stem Cell Res. 2015;14:270–282. doi: 10.1016/j.scr.2015.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Assefnia S, Dakshanamurthy S, Auvil JM Guidry, Hampel C, Anastasiadis PZ, Kallakury B, Uren A, Foley DW, Brown ML, Shapiro L, et al. Cadherin-11 in poor prognosis malignancies and rheumatoid arthritis: Common target, common therapies. Oncotarget. 2014;5:1458–1474. doi: 10.18632/oncotarget.1538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Dakshanamurthy S, Issa NT, Assefnia S, Seshasayee A, Peters OJ, Madhavan S, Uren A, Brown ML, Byers SW. Predicting new indications for approved drugs using a proteochemometric method. J Med Chem. 2012;55:6832–6848. doi: 10.1021/jm300576q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Remmele W, Schicketanz KH. Immunohistochemical determination of estrogen and progesterone receptor content in human breast cancer. Computer-assisted image analysis (QIC score) vs. subjective grading (IRS) Pathol Res Pract. 1993;189:862–866. doi: 10.1016/S0344-0338(11)81095-2. [DOI] [PubMed] [Google Scholar]
- 24.Hicks DG, Schiffhauer L. Standardized assessment of the HER2 status in breast cancer by immunohistochemistry. Lab Med. 2011;42:459–467. doi: 10.1309/LMGZZ58CTS0DBGTW. [DOI] [Google Scholar]
- 25.Savci-Heijink CD, Halfwerk H, Hooijer GK, Horlings HM, Wesseling J, van de Vijver MJ. Retrospective analysis of metastatic behaviour of breast cancer subtypes. Breast Cancer Res Treat. 2015;150:547–557. doi: 10.1007/s10549-015-3352-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Salmen J, Neugebauer J, Fasching PA, Haeberle L, Huober J, Wöckel A, Rauh C, Schuetz F, Weissenbacher T, Kost B, et al. Pooled analysis of the prognostic relevance of progesterone receptor status in five German cohort studies. Breast Cancer Res Treat. 2014;148:143–151. doi: 10.1007/s10549-014-3130-4. [DOI] [PubMed] [Google Scholar]
- 27.Sun JY, Wu SG, Li FY, Lin HX, He ZY. Progesterone receptor loss identifies hormone receptor-positive and HER2-negative breast cancer subgroups at higher risk of relapse: A retrospective cohort study. Onco Targets Ther. 2016;9:1707–1713. doi: 10.2147/OTT.S98666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.van Roy F. Beyond E-cadherin: Roles of other cadherin superfamily members in cancer. Nat Rev Cancer. 2014;14:121–134. doi: 10.1038/nrc3647. [DOI] [PubMed] [Google Scholar]


