Abstract
The plant hormone ethylene regulates many aspects of growth and development. Loss-of-function mutations in ETHYLENE INSENSITIVE2 (EIN2) result in ethylene insensitivity in Arabidopsis, indicating an essential role of EIN2 in ethylene signaling. However, little is known about the role of EIN2 in species other than Arabidopsis. To gain a better understanding of EIN2, a petunia (Petunia × hybrida cv Mitchell Diploid [MD]) homolog of the Arabidopsis EIN2 gene (PhEIN2) was isolated, and the role of PhEIN2 was analyzed in a wide range of plant responses to ethylene, many that do not occur in Arabidopsis. PhEIN2 mRNA was present at varying levels in tissues examined, and the PhEIN2 expression decreased after ethylene treatment in petals. These results indicate that expression of PhEIN2 mRNA is spatially and temporally regulated in petunia during plant development. Transgenic petunia plants with reduced PhEIN2 expression were compared to wild-type MD and ethylene-insensitive petunia plants expressing the Arabidopsis etr1-1 gene for several physiological processes. Both PhEIN2 and etr1-1 transgenic plants exhibited significant delays in flower senescence and fruit ripening, inhibited adventitious root and seedling root hair formation, premature death, and increased hypocotyl length in seedling ethylene response assays compared to MD. Moderate or strong levels of reduction in ethylene sensitivity were achieved with expression of both etr1-1 and PhEIN2 transgenes, as measured by downstream expression of PhEIL1. These results demonstrate that PhEIN2 mediates ethylene signals in a wide range of physiological processes and also indicate the central role of EIN2 in ethylene signal transduction.
The plant hormone ethylene is involved in a wide range of developmental processes in many plant species (Abeles et al., 1992). In Arabidopsis, ethylene signaling is mediated by a complex multicomponent pathway (Kieber, 1997; Guo and Ecker, 2004). ETR1, which encodes a His kinase with homology to bacterial two-component regulators, has been identified as an ethylene receptor (Chang et al., 1993). A total of five ethylene receptor genes have been cloned from Arabidopsis; ETR1, ETR2, ERS1, ERS2, and EIN4 (Hua et al., 1995, 1998; Sakai et al., 1998). Analysis of loss-of-function mutations in multiple receptors has shown that the receptors are negative regulators of ethylene responses (Hua and Meyerowitz, 1998). The receptors are believed to act through CTR1, which is homologous to the Raf family of Ser/Thr kinases and negatively regulates ethylene signaling (Kieber et al., 1993; Huang et al., 2003). Multiple EIN3/EIL (EIN3-like) transcription factors have also been identified in Arabidopsis (Chao et al., 1997). The EIN3 family contains several proteins that bind an ethylene response element in the promoter of a downstream transcription factor, ERF1 (Solano et al., 1998).
An additional component of the ethylene-signaling pathway, EIN2, was isolated from Arabidopsis (Alonso et al., 1999). EIN2 is a single-copy gene and is the only gene known in which loss-of-function mutations result in complete loss of ethylene responsiveness (Chen and Bleecker, 1995; Roman et al., 1995). Genetic epistasis analysis is consistent with EIN2 acting downstream of CTR1 and upstream of EIN3 in Arabidopsis. EIN2 is a novel integral membrane protein with 12 membrane-spanning regions. The amino-terminal hydrophobic domain shows similarity to the Nramp metal-ion transport proteins. By contrast, the carboxyl-terminal hydrophilic region does not show homology to any known protein. Expression of the carboxyl-terminal portion of the protein (EIN2 CEND) is sufficient to constitutively activate some ethylene responses in an ein2 null background. It also restores the ability of the mutant to respond to jasmonic acid and paraquat, a superoxide anion-producing compound, but not ethylene (Alonso et al., 1999). This carboxyl-terminal sequence contains several highly conserved regions in comparison with rice (Oryza sativa) EIN2 (OsEIN2; Jun et al., 2004). These results suggest that the amino-terminal region of the EIN2 protein is necessary for sensing the ethylene signal from upstream components and the carboxyl-terminal region is required for transducing the signal to the downstream components in the ethylene-signaling pathway (Alonso et al., 1999).
Recently, Hall and Bleecker (2003) showed that introduction of the ein2 loss-of function mutation into an ers1 etr1 double mutant, which shows severe developmental defects, resulted in plants that phenocopy the ein2 single mutant. This result indicates that these developmental defects are mediated through EIN2 in Arabidopsis (Hall and Bleecker, 2003). In addition, transgenic rice plants with reduced OsEIN2 expression showed a reduced-shoot elongation phenotype likely resulting from reduced response to ethylene (Jun et al., 2004). Although Arabidopsis is ideal for many genetic and molecular analyses, the many developmental processes mediated by ethylene are difficult to analyze in this plant. There are other model systems that are more suitable to analyze these responses. For example, the ethylene signaling pathway controls flower senescence, fruit ripening, and adventitious root formation in petunia (Petunia × hybrida) and tomato (Lycopersicon esculentum). Transgenic petunias constitutively expressing the mutant Arabidopsis ethylene receptor gene etr1-1 exhibit decreased ethylene sensitivity and delayed flower senescence (Wilkinson et al., 1997). These plants also have delayed fruit ripening (Gubrium et al., 2000) and reduced adventitious root formation (Clark et al., 1999). Never ripe (Nr) tomato plants that contain a mutation in an ethylene receptor produce fruit that do not ripen even when treated with exogenous ethylene (Wilkinson et al., 1995). Similar effects can be observed in plants with reduced expression of the tomato EIN3 genes LeEIL1, LeEIL2, and LeEIL3. These plants have greatly reduced ethylene sensitivity as well as delayed flower senescence and fruit ripening (Tieman et al., 2001). Unfortunately, the role of EIN2 in many of these responses has not been investigated.
To gain a better understanding of the role of EIN2 in ethylene signaling and responses, we isolated a homolog of the Arabidopsis EIN2 gene from petunia (cv Mitchell Diploid [MD]) and produced transgenic plants with reduced expression of PhEIN2 mRNA. Comparison of PhEIN2 transgenic plants and transgenic plants expressing etr1-1 indicates that PhEIN2 mediates ethylene signals involved in a wide range of physiological processes, including flower senescence, fruit ripening, and adventitious root and root hair formation. Characterization of plants displaying moderate and strong reductions in ethylene sensitivity indicates that both the ethylene receptors and EIN2 share central roles in a wide range of physiological responses.
RESULTS
Isolation of EIN2 cDNA in Petunia
A petunia homolog of the Arabidopsis EIN2 gene was isolated through a combination of reverse transcription (RT)-PCR and RACE. The cDNA, designated PhEIN2 (accession no. AY353249), shares 56% nucleotide identity and 46% amino acid sequence identity (62% similarity) with Arabidopsis EIN2 (Fig. 1). The deduced PhEIN2 peptide shares 38% identity (53% similarity) with rice EIN2 (OsEIN2; Jun et al., 2004) and 37% identity (53% similarity) with maize (Zea mays) EIN2 (ZmEIN2; Gallie and Young, 2004). The deduced PhEIN2 protein contains structural features characterized in the Arabidopsis EIN2 protein, including 12 putative transmembrane domains in the amino terminus. There are highly conserved regions among EIN2 proteins in the carboxyl terminus (Fig. 1). These amino acid sequences did not show significant similarity with any known protein. The results from DNA gel-blot analysis implied that EIN2 is likely a single-copy gene in petunia (data not shown).
Figure 1.
Predicted amino acid sequence alignments of petunia (PhEIN2, AY353249>), Arabidopsis (AtEIN2, AF141202), rice (OsEIN2, AY396568), and maize (ZmEIN2, AY359584) EIN2.
PhEIN2 Transgenic Plants
To determine the function of PhEIN2 in ethylene signal transduction, we produced transgenic petunia plants with reduced PhEIN2 expression. Two independent approaches to reduce expression were employed; expression of a sense RNA (PhEIN2-sense) to achieve cosuppression and RNA interference (PhEIN2-RNAi). Seventy-six and 68 independent transgenic lines were produced for the PhEIN2-sense and the PhEIN2-RNAi construct, respectively. Eight lines (11%) of the PhEIN2-sense construct displayed increased flower longevity (more than 2-fold) after ethylene treatment and pollination compared to wild-type (MD) plants in the T0 generation. For PhEIN2-RNAi, 15 lines (22%) displayed increased flower longevity and an additional 8 lines (12%) died prematurely during plant culture. None of the MD or PhEIN2-sense lines displayed premature plant death during culture.
Two homozygous lines, EIN2s-182 and EIN2r-12, which exhibited the greatest flower longevity after ethylene treatment and pollination in the T1 generation, were selected from the PhEIN2-sense and PhEIN2-RNAi lines, respectively. The EIN2r-12 plants showed greater flower longevity compared to the EIN2s-182 plants. Phenotypes of these PhEIN2 transgenic lines were compared to those of two additional transgenic lines, etr-44568 (Wilkinson et al., 1997) and etr-56 (Clevenger, 2000), constitutively expressing the Arabidopsis etr1-1. These etr1-1 transgenic lines have reduced ethylene sensitivity due to expression of a dominant mutant form of the ethylene receptor. The etr-44568 plants show greater flower longevity compared to the etr-56 plants (Clevenger, 2000). All subsequent experiments were performed on plants from these lines.
PhEIN2 mRNA Levels in Wild-Type and Transgenic Petunia Plants
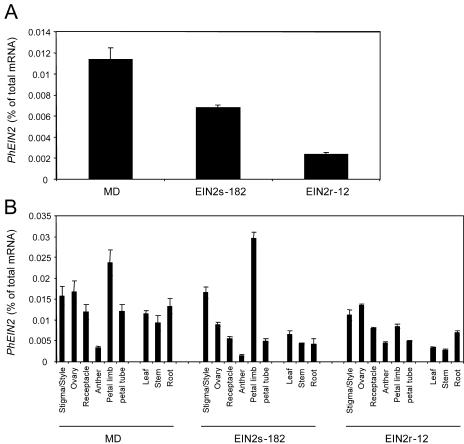
PhEIN2 mRNA levels in whole flowers of EIN2s-182 and EIN2r-12 plants were determined by quantitative real-time RT-PCR. The level of PhEIN2 mRNA was decreased to 60% relative to control (MD) in EIN2s-182 flowers, and the level was decreased to 20% relative to control in EINr-12 flowers (Fig. 2A). Furthermore, to analyze whether PhEIN2 expression was greatly reduced in particular tissues, levels of PhEIN2 mRNA in separate flower parts and vegetative tissues were determined. Levels of PhEIN2 mRNA were decreased in most tissues of the transgenic plants, but the degree of reduction varied in different tissues (Fig. 2B). In the EIN2s-182 plants, the PhEIN2 mRNA level was decreased to 40% of control in petal tubes and 46% of control in receptacles, but no reduction was observed in petal limbs. PhEIN2 mRNA was also decreased in ovaries (53% of control) and anthers (46% of control) of EIN2s-182 flowers compared to controls. Levels of the PhEIN2 mRNA were 57%, 47%, and 31% of control in leaves, stems, and roots, respectively, in the EIN2s-182 plants (Fig. 2B). In the EIN2r-12 plants, the PhEIN2 mRNA level was decreased to 36% of control in petal limbs, 41% of control in petal tubes, and 66% of control in receptacles. Levels of the PhEIN2 mRNA were 28%, 30%, and 52% of control in leaves, stems, and roots, respectively, in the EIN2r-12 plants (Fig. 2B). These results indicate that the suppression level of PhEIN2 expression is not uniform in tissues of these transgenic plants, even though transgene expression was driven by the constitutive cauliflower mosaic virus (CaMV) 35S promoter.
Figure 2.
PhEIN2 mRNA expression in whole flowers (A) and flower parts and vegetative tissues (B) of MD and transgenic plants. Total RNA was isolated from vegetative tissues and from whole flowers and flower parts on the day before anthesis. Levels of PhEIN2 mRNA were determined by quantitative real-time RT-PCR.
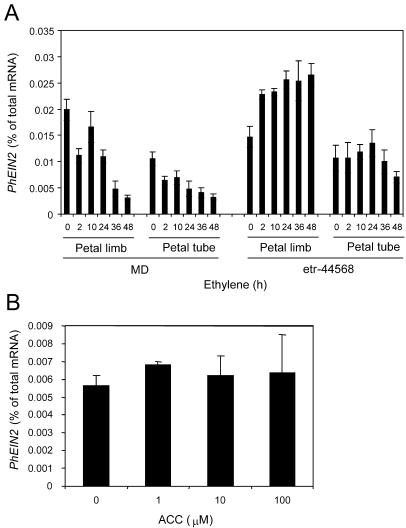
In order to determine whether PhEIN2 is regulated by ethylene, detached flowers were treated with ethylene, and mRNA levels in petal limbs and tubes from MD and etr-44568 flowers were determined. Expression of PhEIN2 mRNA was decreased after ethylene treatment in both petal limbs and petal tubes in MD. Two hours of ethylene treatment was sufficient to reduce PhEIN2 expression in both petal limbs and petal tubes (Fig. 3A). However, at least 9 h of ethylene treatment is required to induce irreversible flower senescence in petunia (Underwood, 2003). Since levels of PhEIN2 expression were restored after 10 h of treatment, the initial reduction of PhEIN2 expression after 2 h of treatment is likely a quick and temporal response of exposure to a high concentration of ethylene. The levels of PhEIN2 mRNA were 15% and 32% of control in the petal limb and tube, respectively, at 48 h after treatment, when senescence of the petal limb started (Fig. 3A). By contrast, levels of PhEIN2 mRNA were not decreased by ethylene treatment in petals of ethylene-insensitive etr-44568 plants during the course of the experiment (Fig. 3A). These results indicate that PhEIN2 mRNA expression is decreased by exogenous ethylene treatment in petals. To determine whether PhEIN2 expression was affected by ethylene in other tissues, levels of PhEIN2 mRNA were determined in seedlings grown in the presence of 1-aminocyclopropane-1-carboxylic acid (ACC), an ethylene precursor. In contrast to the petal, the expression level of PhEIN2 mRNA was not affected by ACC treatment in seedlings (Fig. 3B). These results suggest that PhEIN2 expression is regulated by ethylene in a tissue-specific manner. However, whether this ethylene-associated decrease in petals is a direct effect of the hormone or simply associated with petal senescence remains to be determined.
Figure 3.
A, PhEIN2 mRNA expression after ethylene treatment in petal limbs and petal tubes. MD and etr-44568 flowers were treated with ethylene for 2, 10, 24, 36, and 48 h. B, PhEIN2 mRNA expression in seedlings. MD seeds were plated on media containing 0, 1, 10, and 100 mm ACC and incubated for 7 d. Levels of PhEIN2 mRNA were determined by quantitative real-time RT-PCR.
Phenotypes of PhEIN2 Transgenic Plants
Induction of PhEIL1 Expression by Exogenous Ethylene in Flowers
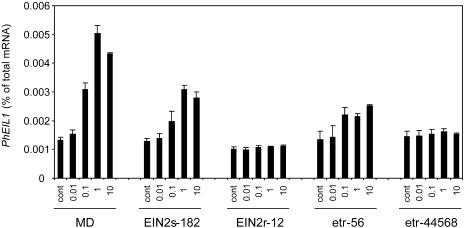
To determine the level of ethylene sensitivity in flowers of transgenic plants at the molecular level, induction of PhEIL1 mRNA by ethylene treatment was analyzed in all transgenic PhEIN2 and etr1-1 lines. PhEIL1 (accession no. AY353248) is a petunia homolog of the Arabidopsis EIN3 gene, which is downstream of EIN2 in the ethylene-signaling pathway (Chao et al., 1997). We have isolated three homologs of Arabidopsis EIN3 in petunia (J. Ciardi and D. Clark, unpublished data) and found that expression of PhEIL1 is induced by ethylene whereas the others are not. Detached flowers were treated with several concentrations of ethylene for 4 h, and mRNA levels for PhEIL1 were determined using real-time RT-PCR. In MD plants, expression of PhEIL1 was induced by high concentrations of exogenous ethylene by up to 370% of the untreated control. In the EIN2s-182 plants, the maximum induction by ethylene was 225% of the untreated control, whereas the EIN2r-12 plants exhibited no induction of the PhEIL1 expression (Fig. 4). The PhEIL1 expression was not induced by exogenous ethylene in etr-44568 plants, whereas it was induced significantly in etr-56 (up to 187% of untreated control) at high ethylene concentrations (Fig. 4). These results indicate that ethylene sensitivity is greatly reduced in EIN2r-12 and etr-44568 flowers and that ethylene sensitivity is measurably reduced in EIN2s-182 and etr-56 flowers, but to a lesser extent than EIN2r-12 and etr-44568.
Figure 4.
Induction of PhEIL1 mRNA by ethylene in MD and transgenic plants. Flowers were detached on the day before anthesis and treated with 0.01, 0.1, 1, and 10 μL L−1 ethylene for 4 h. Total RNA was isolated from whole flowers. Levels of PhEIL1 mRNA were determined by quantitative real-time RT-PCR.
Flower Longevity and Fruit Ripening
Senescence of petunia flowers is accelerated by exogenous ethylene treatment or pollination through an induction of autocatalytic ethylene biosynthesis (Gilissen and Hoekstra, 1984; Tang and Woodson, 1996). Previously ethylene-insensitive etr-44568 petunia plants were shown to have significant delays in flower senescence (Wilkinson et al., 1997). To determine the role of PhEIN2 in flower senescence, flower longevity of the PhEIN2 transgenic plants after exogenous ethylene treatment or pollination was analyzed. MD flowers senesced 2 d after ethylene treatment, whereas the EIN2s-182 flowers exhibited delayed senescence, showing no visible signs of senescence for up to 8 d (Table I; Fig. 5A). The EIN2s-182 plants also displayed delayed flower senescence after pollination. Flowers from these plants senesced after 9 d of pollination, whereas MD flowers senesced after 2 d (Table I). The EIN2r-12 flowers senesced 12 d after ethylene treatment and 14 d after pollination (Table I). Corollas were still turgid, even after the ovaries were fully expanded in these PhEIN2 transgenic plants (Fig. 5B). These results indicate that PhEIN2 is involved in ethylene signaling leading to flower senescence.
Table I.
Flower longevity after ethylene treatment or pollination, and fruit ripening in MD and transgenic plants
| Line | Flower Longevity (Day)
|
Fruit Ripening (Day) | |
|---|---|---|---|
| Ethylene | Pollination | ||
| MD | 2.09 ± 0.05 a | 2.24 ± 0.07 a | 24.3 ± 0.20 a |
| EIN2s-182 | 8.19 ± 0.33 b | 8.45 ± 0.38 b | 26.8 ± 0.44 b |
| EIN2r-12 | 11.6 ± 0.70 c | 13.7 ± 1.25 cd | 31.1 ± 0.77 d |
| etr-56 | 9.41 ± 0.37 bc | 12.4 ± 0.64 c | 26.4 ± 0.30 b |
| etr-44568 | 11.9 ± 0.54 c | 14.8 ± 0.70 d | 29.0 ± 0.38 c |
Values represent mean ± se. Mean separations were analyzed at the 0.05% level (HSD0.05).
Figure 5.
Phenotypes of PhEIN2 transgenic plants. A, MD (left) and EIN2s-182 (right) flowers 3 d after ethylene treatment. Flowers on the day before anthesis were treated with ethylene for 16 h. After treatment, flowers were placed in a growth room. B, MD flower 2 d after pollination (left) and an EIN2r-12 flower 8 d after pollination (right). Flowers were self-pollinated on the day before anthesis. C, Adventitious root formation in MD and transgenic lines. Cuttings from MD, EIN2s-182, EIN2r-12, etr-56, and etr-44568 were propagated for 2 weeks. D, EIN2r-12 plants 7 weeks after germination showing premature death. E, Adventitious root index.
The etr-44568 plants displayed delayed flower senescence after ethylene treatment and pollination, as reported previously by Wilkinson et al. (1997). The etr-56 plants also exhibited increased flower longevity compared to MD, but to a lesser extent than etr-44568. Flower longevity of the EIN2r-12 plants after ethylene treatment and pollination was equivalent to that of etr-44568 and greater than that of EIN2s-182 and etr-56 (Table I). These results indicate that there are intermediate levels of ethylene sensitivity between genotypes for both PhEIN2 and etr1-1 transgenic plants.
Petunia fruits usually fully expand by 7 d after pollination, then dry down and dehisce when ripe. Both PhEIN2 transgenic lines and both etr1-1 lines exhibited a significant delay in fruit ripening compared to MD. Fruits of MD plants turned completely brown by 25 d after pollination, whereas fruit ripening of the EIN2r-12 and etr-44568 plants took 31 and 29 d, respectively, to reach the same stage. The EIN2s-182 and etr-56 plants also displayed a significant delay in fruit ripening compared to MD but less than EIN2r-12 and etr-44568 (Table I). These results indicate an involvement of PhEIN2 in ethylene signaling leading to fruit ripening.
Adventitious Root Formation
To determine the effects of reduced PhEIN2 expression on adventitious root formation, propagation experiments were conducted on vegetative cuttings from MD and transgenic plants. Adventitious rooting was scored using a qualitative adventitious root index (Fig. 5E), and dry weight of adventitious roots was also measured. Cuttings from the EIN2r-12, etr-56, and etr-44568 plants produced significantly fewer adventitious roots compared to MD, with almost no root production in EIN2r-12 and etr-44568 (Table II; Fig. 5C). Cuttings from EIN2s-182 plants produced significant numbers of adventitious roots, scoring 3.4 in the root index, although dry weights of adventitious roots were almost half compared to MD plants (Table II; Fig. 5C). These results indicate that PhEIN2 is required for adventitious root formation and that EIN2s-182 plants have a greater ability to produce adventitious roots compared to other transgenic lines, most likely because the transgene was ineffective in this tissue (see below).
Table II.
Adventitious root index and dry weight in MD and transgenic plants
| Line | Index | Dry Weight |
|---|---|---|
| g | ||
| MD | 3.83 ± 0.05 b | 1.65 |
| EIN2s-182 | 3.42 ± 0.32 b | 0.88 |
| EIN2r-12 | 1.11 ± 0.06 a | <0.01 |
| etr-56 | 1.23 ± 0.32 a | <0.01 |
| etr-44568 | 1.03 ± 0.02 a | <0.01 |
Values represent mean ± se. Mean separations were analyzed at the 0.05% level (HSD0.05).
Seedling Ethylene Response and Root Hair Formation
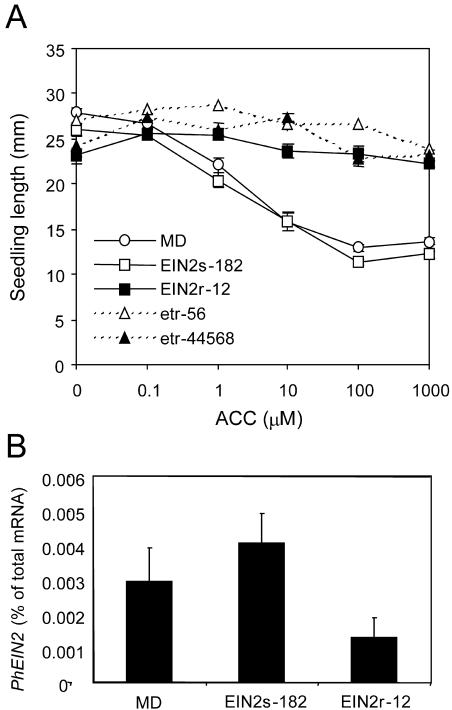
A commonly used assay to measure quantitative ethylene responses is the seedling triple response. Hypocotyl length of dark-grown PhEIN2 and etr1-1 transgenic seedlings was compared to MD in the presence of ACC. Hypocotyl length of MD seedlings was decreased in the presence of increasing concentrations of ACC. In the EIN2r-12, etr-56, and etr-44568 seedlings, hypocotyls were longer compared to MD on higher levels of ACC, indicating reduced sensitivity to ethylene. These three transgenic lines were not significantly different from each other (Fig. 6A), thus indicating that EIN2r-12, etr-56, and etr-44568 seedlings have similar levels of ethylene sensitivity. By contrast, the EIN2s-182 seedlings showed similar responses to MD (Fig. 6A), indicating that the transgene did not reduce ethylene sensitivity at this stage of development. To determine the relationship between the level of ethylene sensitivity and the level of PhEIN2 mRNA, the expression of PhEIN2 mRNA was analyzed in etiolated PhEIN2 seedlings. The level of PhEIN2 mRNA was decreased to 56% of control in EIN2r-12, whereas the level was not decreased significantly in EIN2s-182 (Fig. 6B). These results indicate that there is a correlation between PhEIN2 mRNA levels and ethylene sensitivity in etiolated seedlings. In addition, since even small reduction (50% of control) of PhEIN2 expression altered ethylene sensitivity, the level of PhEIN2 mRNA is likely critical for regulating sensitivity.
Figure 6.
A, Seedling ethylene responses in MD and transgenic plants. Seedlings were grown in the dark for 7 d in the presence of increasing concentrations of ACC, and lengths of hypocotyls were measured. B, PhEIN2 mRNA expression in etiolated seedlings of MD and PhEIN2 transgenic plants. Total RNA was isolated from etiolated seedlings incubated for 7 d. Levels of PhEIN2 mRNA were determined by quantitative real-time RT-PCR.
In the seedling ethylene response assay, we observed differences in root hair formation among MD and transgenic lines. Root hairs of MD and EIN2s-182 seedlings were induced by 1 μm ACC treatment, and the length of primary roots was shortened. By contrast, root hairs were not induced by 1 μm ACC treatment in EIN2r-12, etr-56, and etr-44568 seedlings (Fig. 7). In etr-56, root hairs were slightly induced on the agar plate containing 1.0 mm ACC (data not shown). These results indicate that PhEIN2 also mediates ethylene signals leading to root hair formation.
Figure 7.
Root hair formation of etiolated seedlings in MD and transgenic plants in the presence of ACC. Seedlings were grown vertically on agar plates containing 1 μm ACC in the dark for 7 d.
Premature Death
In the EIN2r-12, etr-44568, and etr-56 lines, 64%, 67%, and 26% of plants, respectively, exhibited premature death during culture of their homozygous progeny. These plants displayed severe wilting of leaves and died during the course of the experiment (Fig. 5 D). Although we observed this premature death at various time points during plant culture, many plants died after transfer of the seedlings to 15-cm pots. We also observed that the percentage of premature death was higher in high temperatures during summertime compared to other seasons (data not shown). Tissue samples from plants that displayed premature death were routinely submitted for pathogen identification to a diagnostic lab and were reported to have either no infection or the gray mold fungus Botrytis cinerea, which is ubiquitous in greenhouses especially on dead or dying tissues. By contrast, none of the MD or EIN2s-182 plants died prematurely during the course of any of these experiments. We have seen that Nr tomato plants also show a significant amount of premature death when they are grown in the field (H. Klee, unpublished data). The premature death observed in the EIN2r-12, etr-56, and etr-44568 lines is likely due to reduced tolerance to abiotic stresses resulting from strong reduction of ethylene sensitivity.
DISCUSSION
EIN2 has been shown to be essential for ethylene signal transduction in Arabidopsis. It is a particularly interesting and important protein, having been identified in screen for multiple hormone responses (Su and Howell, 1992; Fujita and Syono, 1996; Beaudoin et al., 2000). Ethylene is involved in several important developmental processes in many horticultural crop species, but the function of EIN2 has not been examined in these responses. We isolated a petunia homolog of the Arabidopsis EIN2 gene (PhEIN2) and analyzed spatial and temporal regulation of PhEIN2 expression. Furthermore, we produced transgenic lines with reduced PhEIN2 expression and compared these lines to additional transgenic lines expressing Arabidopsis etr1-1 in order to examine the role of PhEIN2 in ethylene signal transduction.
The predicted peptide sequence of PhEIN2 shows high similarity to Arabidopsis EIN2 throughout the protein, with a particularly highly conserved amino acid sequence in the carboxyl terminus (Fig. 1). This region is also conserved in EIN2 proteins of rice (Jun et al., 2004) and maize (Gallie and Young, 2004). Expression of the carboxyl-terminal protein of Arabidopsis EIN2 is sufficient to constitutively activate ethylene response in an ein2 null background (Alonso et al., 1999). This carboxyl-terminal region likely plays an important role in transduction of the ethylene signal to the downstream components.
PhEIN2 mRNA was present at different levels in different tissues (Fig. 2B), indicating that PhEIN2 expression is spatially regulated. In the petal limb and tube of MD plants, PhEIN2 mRNA levels decreased after ethylene treatment (Fig. 3A). This may indicate that feedback inhibition is involved in the regulation of PhEIN2 expression. This result contrasts with results from Arabidopsis and rice, where the expression of EIN2 was not affected by treatment with ethylene (Alonso et al., 1999; Jun et al., 2004). In contrast to the regulated expression in petunia flowers, ACC treatment did not affect levels of PhEIN2 mRNA in seedlings (Fig. 3B). These results indicate that PhEIN2 expression is regulated by ethylene in a tissue-specific manner. The flower senescence process is particularly important for successful pollination and subsequent seed set in plant species that show ethylene-regulated flower senescence. Thus, there may be a regulation system of ethylene sensitivity in flowers through control of PhEIN2 expression. However, it is also possible that the decrease in PhEIN2 mRNA level in a later time course of ethylene treatment is a consequence of the ethylene-induced senescence process in petals.
To determine the function of PhEIN2 in ethylene responses in petunia, transgenic plants with reduced PhEIN2 expression were produced. We used two different constructs to suppress the PhEIN2 mRNA expression, one for expression of a sense RNA to achieve cosuppression and the other for RNAi. The RNAi construct gave a higher percentage of independent transgenic plants showing reduced ethylene sensitivity in flowers compared to the cosuppression construct. These results indicate that RNAi-mediated gene silencing is more efficient than cosuppression, as shown by Wesley et al. (2001). However, neither construct produced a high percentage of plants with greatly reduced ethylene sensitivity. Transgenic plants with strong reduction in ethylene sensitivity showed reduced adventitious root formation (Table II; Fig. 5C). Since rooting of transgenic shoots requires adventitious root formation in petunia, this may be a consequence of selection against highly insensitive plants during the regeneration process.
It has been shown that ethylene induces flower senescence in petunia (Gilissen and Hoekstra, 1984; Tang and Woodson, 1996), and reduced ethylene sensitivity results in a significant delay in flower senescence after ethylene treatment and pollination in etr-44568 petunia (Wilkinson et al., 1997; Table I). The PhEIN2 transgenic plants showed a significant delay in flower senescence in the T0 and T1 progeny after ethylene treatment and pollination. We selected two independent homozygous transgenic lines, EIN2s-182 and EINr-12, which displayed the greatest flower longevity with a PhEIN2-sense and a PhEIN2-RNAi construct, respectively, to examine several physiological responses that have been thought to associate with ethylene signaling. The EIN2s-182 and EIN2r-12 plants showed a significant delay in flower senescence compared to MD after ethylene treatment and pollination, with a greater delay in EIN2r-12 plants (Table I). The level of PhEIN2 mRNA was decreased to 60% relative to the MD in EIN2s-182 flowers, and the level was decreased to 20% relative to MD in EINr-12 flowers (Fig. 2A). Since a reduction of PhEIN2 mRNA expression resulted in delayed flower senescence, these results demonstrate that PhEIN2 positively mediates the ethylene signal leading to flower senescence in petunia.
The decreased ethylene sensitivity of the EIN2s-182 and EIN2r-12 flowers was also evident in the expression of an ethylene-inducible gene, PhEIL1. PhEIL1 is a petunia homolog of the Arabidopsis EIN3 gene that is downstream of EIN2 in the ethylene-signaling pathway (Chao et al., 1997). Expression of PhEIL1 was induced by exogenous ethylene in MD plants (Fig. 4). By contrast, expression of PhEIL1 was not induced by ethylene in EIN2r-12 flowers or in etr-44568 flowers. In EIN2s-182 and etr-56, PhEIL1 expression was induced, but to a lesser degree than in MD (Fig. 4). These results are consistent with the results from flower senescence studies, where EIN2r-12 and etr-44568 plants display a great delay in flower senescence and EIN2s-182 and etr-56 plants display a moderate delay. This induction of PhEIL1 expression by ethylene contrasts with results from Arabidopsis and tomato, where expression of the EIN3 and EIL genes is not regulated by exogenous ethylene (Chao et al., 1997; Tieman et al., 2001). Interestingly, ethylene treatment did not affect expression of the other petunia homologs of EIN3 (PhEIL2 and PhEIL3; K. Shibuya and D. Clark, unpublished data).
Although results on flower longevity and PhEIL1 expression demonstrate a reduction of ethylene sensitivity in flowers of EIN2s-182 plants, the question of whether the 40% reduction of PhEIN2 mRNA relative to MD corresponded to an almost 4-fold increase in flower longevity still remained. To determine whether PhEIN2 mRNA expression was greatly decreased in particular tissues, we analyzed levels of PhEIN2 in separate flower parts. In EIN2s-182, the PhEIN2 mRNA level was decreased 60% compared to MD in the petal tube, whereas the mRNA level was not decreased in the petal limb. Interestingly, the reduction of PhEIN2 mRNA in the petal tube of EIN2r-12, which showed a greater delay in flower senescence than EIN2s-182, was almost the same as that of EIN2s-182. However, in EIN2r-12 plants, the expression level was decreased to 36% relative to MD in the petal limb. These results suggest that a decrease of PhEIN2 mRNA in the petal tube is sufficient to delay flower senescence, and a decrease in the petal limb gives additional longevity to flowers. It has been suggested that the ethylene produced from pistils triggers ethylene production in petals after pollination (Tang and Woodson, 1996). Ethylene perception in the petal tube appears to be essential for the subsequent induction of petal senescence.
Transgenic plants with reduced PhEIN2 expression and etr1-1 transgenic plants also showed a delay in fruit ripening compared to MD, with a greater delay in EIN2r-12 and etr-44568 plants compared to EIN2s-182 and etr-56 plants (Table I). It is well known that ethylene induces and accelerates fruit ripening in many plant species that produce fleshy fruits such as tomato (Abeles et al., 1992; Wilkinson et al., 1995). However, the manner of fruit ripening in petunia is quite different from that of tomato. Petunia fruits ripen by a process of dry down and dehiscence, whereas tomato fruits ripen by a process of softening. Gubrium et al. (2000) reported that etr-44568 plants show a delay in fruit ripening. Our results in PhEIN2 transgenic plants confirm that ethylene is involved in fruit ripening not only in fleshy fruits but also in dry dehiscent fruits.
Ethylene-insensitive phenotypes were also observed in vegetative tissues of the transgenic lines. Adventitious root formation of vegetative cuttings was inhibited in EIN2r-12 plants as strongly as in etr-44568. It has been previously shown that the ethylene-insensitive etr-44568 petunia and Nr tomato plants produce fewer adventitious roots than wild-type plants (Clark et al., 1999). EIN2s-182 cuttings produced adventitious roots but fewer than MD (Table II; Fig. 5C). The PhEIN2 mRNA level was lower in the stem of EIN2r-12 (30% of control) compared to EIN2s-182 (47% of control; Fig. 2B); thus, the level of ethylene sensitivity in the stem likely determines the potential for adventitious root formation.
In the seedling ethylene response assay, EIN2r-12 seedlings exhibited strong ethylene insensitivity equivalent to etr-44568 (Fig. 6A). Although EIN2s-182 plants showed a significant reduction of ethylene sensitivity in flowers and fruits, their seedlings did not exhibit a reduction of ethylene sensitivity (Fig. 6A). However, in the EIN2s-182 seedlings, the PhEIN2 mRNA level was not decreased compared to MD (Fig. 6B). This nonuniform suppression of PhEIN2 expression in different tissues is likely due to a positional effect of the transgene.
Overall, EIN2r-12 plants displayed a strong reduction of ethylene sensitivity equivalent to etr-44568 plants in flower senescence, fruit ripening, adventitious root and root hair formation, and seedling ethylene response assay. Since the transgenic plants with reduced PhEIN2 expression exhibited similar phenotypes to the transgenic plants with manipulated ethylene signaling at the level of the receptor, all signals from ethylene receptors regulating these responses are likely mediated through EIN2 in petunia. The EIN2s-182 and etr-56 plants also displayed phenotypes associated with reduced ethylene sensitivity. However, the severity of phenotypes varied in different responses. For example, these two lines showed a similar delay in fruit ripening, but EIN2s-182 plants produced adventitious roots and etr-56 plants did not. These results indicate that the level of ethylene sensitivity required for a certain response may be different or there may be a threshold of ethylene sensitivity that lies between EIN2s-182 and etr-56 for some of the responses measured here. It is also possible that the difference in these ethylene responses is due to the different levels of transgene expression in different tissues. Partial reductions (60% reduction from control) in PhEIN2 levels caused a great reduction of ethylene sensitivity in flower senescence and seedling ethylene response assay. This suggests that EIN2 must be limiting to signal transduction and also indicates that temporal and spatial regulation of PhEIN2 expression in flowers is responsible for regulation of ethylene sensitivity during senescence. Guo and Ecker (2003) have shown that a major effect of EIN2 is to regulate the accumulation of EIN3 and EIL1 proteins. The differential effect might be due to different levels of EIL mRNA in each of these tissues.
Manipulation of ethylene responses may be useful for production of high-quality crops with long-lasting flowers or fruits with long shelf life. The Arabidopsis etr1-1 gene has been used to alter ethylene sensitivity in petunia and tomato (Wilkinson et al., 1997) and carnation (Dianthus caryophyllus; Bovy et al., 1999). Here, we show that manipulation of EIN2 expression is also effective in producing transgenic plants with long-lasting flowers in petunia. However, ethylene-insensitive plants display several undesirable phenotypes. For example, EIN2r-12 and etr-44568 petunia plants are inhibited in adventitious root formation. The ability to form adventitious roots is critical for crops that are propagated through vegetative cuttings. Furthermore, ethylene-insensitive tobacco (Nicotiana tabacum) plants have been shown to have increased susceptibility to fungal pathogens (Knoester et al., 1998). In this study, we observed a great deal of premature death in EIN2r-12 and etr-44568 petunia plants. These lines showed the greatest reductions in ethylene responses in all tissues examined. The undesirable phenotypes are linked to this strong, constitutive reduction in ethylene responses and are independent of the mechanism used to block ethylene signaling. Although the EIN2s-182 plants showed a significant delay in flower senescence, this line did not exhibit premature death and did produce a significant number of adventitious roots. This phenotype is likely associated with the nonuniform suppression of PhEIN2 mRNA expression, resulting in different levels of ethylene sensitivity in different tissues. The results presented here demonstrate that, through selection of appropriate transgenic events, it is possible to identify plants that have the desired levels of ethylene insensitivity in target tissues while avoiding deleterious, unwanted effects of reduced ethylene sensitivity.
Transgenic plants with reduced PhEIN2 mRNA expression displayed reduced ethylene sensitivity in a wide range of physiological processes, indicating an essential role for PhEIN2 in ethylene signal transduction throughout plant development in petunia. Guo and Ecker (2003) have shown that EIN2 regulates the accumulation of EIN3 protein in Arabidopsis. However, the biochemical functions of EIN2 in terms of ethylene signal transduction are still unknown. We showed that the deduced EIN2 protein contains a highly conserved region in its carboxyl terminus, implying an important role of this region in signal transduction. Further biochemical analysis of EIN2 protein will be needed to understand how this key component of the pathway controls ethylene-regulated responses in plants.
MATERIALS AND METHODS
Plant Material and Treatment
Petunia × hybrida cv Mitchell Diploid (MD; wild type) and transgenic plants were grown under standard greenhouse conditions in commercial potting medium (Fafard 2B; Fafard, Apopka, FL) in 15-cm pots and fertilized at each irrigation with 150 mg L−1 Scott's Excel 15-5-15 (Scotts, Marysville, OH). Flower and vegetative tissues were harvested on the day before anthesis, frozen in liquid nitrogen, and stored at −80°C. For the ethylene treatment, detached flowers were placed in vials of water and then sealed in a glass container containing 2 μL L−1 ethylene for 2, 10, 24, 36, and 48 h under continuous fluorescent light at 20°C to 23°C. For the ACC (Sigma, St. Louis) treatment, MD seeds were surface sterilized with 1.2% sodium hypochlorite for 10 min, rinsed three times in sterile water, and then plated on 0.5× Murashige and Skoog media, pH 5.6, containing 0.8% agar with 0, 1, 10, and 100 mm ACC in a Phytatray II (Sigma). Seeds were incubated under fluorescent light (16-h light/8-h dark) at 24°C for 7 d.
Cloning of the Petunia EIN2 Genes
For cloning of the petunia EIN2 cDNA (PhEIN2) by RT-PCR, RNA was extracted from leaves, according to the method of Ciardi et al. (2000). First-strand cDNA synthesis was performed using a Superscript preamplification system in accordance with the manufacturer's instructions (Life Technologies, Rockville, MD). Degenerate primers were designed based on conserved sequences in EIN2 cDNAs. Using first-strand cDNA as a template, the petunia EIN2 cDNA was amplified using the Expand High-Fidelity PCR system (Roche, Indianapolis). The degenerate primers YTNGAYGARTTYTGGGG (forward) and GCCTGAANGAYTGAAGAAGCT (reverse) were used to generate a 1.1-kb PCR product from petunia cDNA. The remaining 5′ and 3′ cDNA sequences were isolated by RACE. The Gene Racer amplification system (Invitrogen, Carlsbad, CA) and EXPAND Taq polymerase (Perkin-Elmer, Wellesley, MA) were used for all RACE experiments. Once the sequence of the 5′ and 3′ ends of each cDNA had been determined, full-length cDNA for PhEIN2 was isolated by RT-PCR, using the primers ATCCCGGGATGGAATCTGAAACTCAGACT (forward) and ATCCCGGGTACAAGGGAGTGGGTGAATTAACAT (reverse).
PhEIN2 Expression
Total RNA was extracted using an RNeasy plant mini kit (Qiagen, Valencia, CA) and treated with RNase-Free DNase (Qiagen). Quantitative real-time RT-PCR was performed on 250 ng of total RNA in a 25-μL reaction volume using TaqMan Reverse Transcription Reagents and SYBR Green PCR Core Reagents (Applied Biosystems, Foster City, CA) in an Applied Biosystems GeneAmp 5700 sequence-detection system. Reactions were repeated twice with one set of RNAs and once with RNA collected from separate, duplicate tissue. The primers designed with primer express software (Applied Biosystems) for PhEIN2 were: forward primer, TGTGTTTTTCTGGCTGCAACTG; reverse primer, GCACTGCCATTGTCCAAGAGA. The primers for PhEIN2 were designed for the 5′ region of PhEIN2 that the constructs for transformation did not contain. The final concentration of each primer was 300 nm. RT-PCR conditions were 48°C for 30 min, 95°C for 10 min, followed by 40 cycles of 95°C for 15 s and 60°C for 1 min. Sense-strand RNA, synthesized as described in Tieman et al. (2001), was used as a standard to determine absolute levels of PhEIN2 mRNA.
Induction of PhEIL1 mRNA by Ethylene
Two sets of three flowers were detached on the day before anthesis and treated with 0.01, 0.1, 1, and 10 μL L−1 ethylene for 4 h. For control, solid KMnO4 was added to a glass container to remove ethylene emitted by the flowers. Quantitative real-time RT-PCR was performed as described above. The primers for PhEIL1 were: forward primer, GCCTTTTCCATCTCCACTTACTATTG; reverse primer, TGGATATCAAGCCCCAAATTAAA. The primers for PhEIL1 were designed for the 3′ untranslated region of PhEIL1. Specificity of the primer sets was verified by performing PCR eactions with an in vitro transcribed PhEIL2 and PhEIL3 template.
Production of Transgenic Petunia Plants
Transgenic plants containing one of two different constructs, one for expression of a sense RNA (PhEIN2-sense) to achieve cosuppression and the other for RNA interference (PhEIN2-RNAi), were produced. For the PhEIN2-sense construct, a 1.1-kb segment of the PhEIN2 cDNA spanning nucleotides 2,824 to 3,940 was cloned into a vector downstream from a CaMV 35S promoter and upstream of the Agrobacterium nopaline synthase terminator region in the sense orientation. For the PhEIN2-RNAi construct, a 1.0-kb fragment spanning bases 3,250 to 4,250 and a 450-bp fragment spanning bases 3,800 to 4,250 of the PhEIN2 cDNA were cloned into pBluescript SK+ (Stratagene, La Jolla, CA) in sense and antisense orientation, respectively. The RNAi construct was cloned downstream of the CaMV 35S promoter and upstream of the nopaline synthase terminator. These sense expression and RNAi constructs were then cloned into a binary transformation vector containing a gene for kanamycin resistance (NPTII). The transformation vector was transferred to Agrobacterium ABI through triparental mating. Petunia plants (MD) were transformed with these constructs, according to the methods of Jorgensen et al. (1996). Presence of the transgene was confirmed through PCR by amplifying a segment of the NPTII gene. All subsequent experiments were performed on homozygous progeny.
Plants that died during the course of the experiment were scored as exhibiting premature death. Tissue samples from plants that showed premature were death were sent to the Doyle Conner Diagnostic Lab (Gainesville, FL) to identify any potentially lethal pathogens.
Flower Longevity and Fruit Ripening
To measure flower senescence induced by ethylene, three flowers were removed from each of 24 plants (72 flowers in total per genotype) for MD and transgenic lines on the day before anthesis and placed in vials of water. The flowers were then sealed in a glass container and treated with 2 μL L−1 ethylene for 16 h. The flowers were then placed in a growth room under continuous fluorescent light at 20°C to 23°C, and the day on which the flowers completely wilted was recorded. To measure flower longevity after pollination, 3 flowers from each of 24 plants (72 flowers in total per genotype) for MD and each transgenic line were self-pollinated on the plant on the day before anthesis, and the number of days to wilting was recorded.
To determine the period of fruit ripening, 3 flowers each from 24 plants (72 flowers in total per genotype) for MD and each transgenic line were self-pollinated, and the number of days until the seed pods had turned completely brown was recorded.
Data were analyzed using the ANOVA function of SAS 8.02 (Cary, NC) to compare differences among genotypes. Tukey's honestly significant difference mean-separation test was the procedure used for mean separation at the 0.05% level (HSD0.05).
Adventitious Root Formation
Vegetative stem cuttings were taken from approximately 8-week-old MD, EIN2s-182, EIN2r-12, etr-56, and etr-44568 plants. The cuttings were approximately 5 cm in length and had two nodes. Seventy-two cuttings per genotype were propagated, as described by Clark et al. (1999), and rooting index and dry weight were measured after 2 weeks. Representative cuttings that showed various levels of adventitious root formation were used to define an adventitious root index (Fig. 5E). Root index 1 represents no root production. Cuttings scored as root index 2 have a few short adventitious roots. Cuttings for root index 4 showed the most adventitious root formation through the experiment. Root index 3 shows root formation at a level between 2 and 4. For measurement of dry weight, adventitious roots were detached from 50 cuttings of each genotype, combined, and then dried at 70°C for 2 d. Data on root index were statistically analyzed as described above.
Seedling Ethylene Response and Root Hair Formation
MD and transgenic seeds of homozygous progeny were surface sterilized as described above and then plated on 0.5× Murashige and Skoog media, pH 5.6, containing 0.8% agar with 0, 0.1, 1, 10, 100, and 1,000 μm ACC in a Phytatray II (Sigma). Seeds were incubated under fluorescent light (16 h light/8 h dark) at 24°C for 48 h, then incubated at 24°C in the dark for 7 d. Lengths of hypocotyls from 20 seedlings per genotype were measured. Experiments were conducted twice separately with similar results. Data presented are for one experiment.
To analyze root hair formation, seeds were plated on the same medium described above containing 1 μm ACC in petri dishes (15 × 100 mm). The petri dishes were incubated vertically, as described above, for 7 d. Photographs were recorded using a dissecting microscope with a digital camera.
Sequence data from this article have been deposited with the EMBL/GenBank data libraries under accession numbers AY353249 (PhEIN2) and AY353248 (PhEIL1).
Acknowledgments
We thank Dr. Denise Tieman for technical assistance with the real-time RT-PCR analysis and Yvonne Arwood for her assistance with plant growth and maintenance in the greenhouse.
This work was supported by grants from the Fred Gloeckner Foundation, the American Floral Endowment, the U.S. Department of Agriculture Floral and Nursery Crops Initiative, Plantgenix, and the Florida Agriculture Experimental Station (D.G.C.). Florida Agriculture Experimental Station Journal Series No. R–10373.
Article, publication date, and citation information can be found at www.plantphysiol.org/cgi/doi/10.1104/pp.104.046979.
References
- Abeles FB, Morgan PW, Saltveit ME (1992) Ethylene in Plant Biology. Academic Press, San Diego
- Alonso JM, Hirayama T, Roman G, Nourizadeh S, Ecker JR (1999) EIN2, a bifunctional transducer of ethylene and stress responses in Arabidopsis. Science 284: 2148–2152 [DOI] [PubMed] [Google Scholar]
- Beaudoin N, Serizet C, Gosti F, Giraudat J (2000) Interactions between abscisic acid and ethylene signaling cascades. Plant Cell 12: 1103–1115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bovy AG, Angenent GC, Dons HJM, van Altvorst AC (1999) Heterologous expression of the Arabidopsis etr1-1 allele inhibits the senescence of carnation flowers. Mol Breed 5: 301–308 [Google Scholar]
- Chang C, Kwok SF, Bleecker AB, Meyerowitz EM (1993) Arabidopsis ethylene response gene ETR1—similarity of product to two-component regulators. Science 262: 539–544 [DOI] [PubMed] [Google Scholar]
- Chao Q, Rothenberg M, Solano R, Roman G, Terzaghi W, Ecker JR (1997) Activation of the ethylene gas response pathway in Arabidopsis by the nuclear protein ETHYLENE-INSENSITIVE3 and related proteins. Cell 89: 1133–1144 [DOI] [PubMed] [Google Scholar]
- Chen QG, Bleecker AB (1995) Analysis of ethylene signal-transduction kinetics associated with seedling-growth response and chitinase induction in wild-type and mutant Arabidopsis. Plant Physiol 108: 597–607 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ciardi JA, Tieman DM, Lund SD, Jones JB, Stall RE, Klee HJ (2000) Response to Xanthomonas campestris pv. vesicatoria in tomato involves regulation of ethylene receptor gene expression. Plant Physiol 123: 81–92 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark DG, Gubrium EK, Barrett JE, Nell TA, Klee HJ (1999) Root formation in ethylene-insensitive plants. Plant Physiol 121: 53–59 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clevenger D (2000) Horticultural performance of ethylene-insensitive petunias. MS thesis. University of Florida, Gainesville, FL
- Fujita H, Syono K (1996) Genetic analysis of the effects of polar auxin transport inhibitors on root growth in Arabidopsis thaliana. Plant Cell Physiol 37: 1094–1101 [DOI] [PubMed] [Google Scholar]
- Gallie DR, Young TE (2004) The ethylene biosynthetic and perception machinery is differentially expressed during endosperm and embryo development in maize. Mol Genet Genomics 271: 267–281 [DOI] [PubMed] [Google Scholar]
- Gilissen LJW, Hoekstra FA (1984) Pollination-induced corolla wilting in Petunia hybrida rapid transfer through the style of a wilting-inducing substance. Plant Physiol 75: 496–498 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gubrium EK, Clevenger DJ, Clark DG, Barrett JE, Nell TA (2000) Reproduction and horticultural performance of transgenic ethylene-insensitive petunias. J Am Soc Hortic Sci 125: 277–281 [Google Scholar]
- Guo H, Ecker JR (2003) Plant responses to ethylene gas are mediated by SCFEBF1/EBF2-dependent proteolysis of EIN3 transcription factor. Cell 115: 667–677 [DOI] [PubMed] [Google Scholar]
- Guo H, Ecker JR (2004) The ethylene signaling pathway: new insights. Curr Opin Plant Biol 7: 40–49 [DOI] [PubMed] [Google Scholar]
- Hall AE, Bleecker AB (2003) Analysis of combinatorial loss-of-function mutants in the Arabidopsis ethylene receptors reveals that the ers1 etr1 double mutant has severe developmental defects that are EIN2 dependent. Plant Cell 15: 2032–2041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hua J, Chang C, Sun Q, Meyerowitz EM (1995) Ethylene insensitivity conferred by Arabidopsis ERS gene. Science 269: 1712–1714 [DOI] [PubMed] [Google Scholar]
- Hua J, Meyerowitz EM (1998) Ethylene responses are negatively regulated by a receptor gene family in Arabidopsis thaliana. Cell 94: 261–271 [DOI] [PubMed] [Google Scholar]
- Hua J, Sakai H, Nourizadeh S, Chen QG, Bleecker AB, Ecker JR, Meyerowitz EM (1998) EIN4 and ERS2 are members of the putative ethylene receptor gene family in Arabidopsis. Plant Cell 10: 1321–1332 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang Y, Li H, Hutchison CE, Laskey J, Kieber JJ (2003) Biochemical and functional analysis of CTR1, a protein kinase that negatively regulates ethylene signaling in Arabidopsis. Plant J 33: 221–233 [DOI] [PubMed] [Google Scholar]
- Jorgensen RA, Cluster PD, English J, Oue O, Napoli CA (1996) Chalcone synthase cosuppression phenotypes in petunia flowers: comparison of sense vs. antisense constructs and single-copy vs. complex T-DNA sequences. Plant Mol Biol 31: 957–973 [DOI] [PubMed] [Google Scholar]
- Jun SH, Han MJ, Lee S, Seo YS, Kim WT, An G (2004) OsEIN2 is a positive component in ethylene signaling in rice. Plant Cell Physiol 45: 281–289 [DOI] [PubMed] [Google Scholar]
- Kieber JJ (1997) The ethylene response pathway in Arabidopsis. Annu Rev Plant Physiol Plant Mol Biol 48: 277–296 [DOI] [PubMed] [Google Scholar]
- Kieber JJ, Rothenberg M, Roman G, Feldmann K, Ecker JR (1993) CTR1, a negative regulator of the ethylene response pathway in Arabidopsis, encodes a member of the Raf family of protein kinases. Cell 72: 427–441 [DOI] [PubMed] [Google Scholar]
- Knoester M, van Loon LC, Heuvel JPD, Hennig J, Bol JF, Linthorst HJM (1998) Ethylene-insensitive tobacco lacks nonhost resistance against soil-borne fungi. Proc Natl Acad Sci USA 95: 1933–1937 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roman G, Lubarsky B, Kieber JJ, Rothenberg M, Ecker JR (1995) Genetic analysis of ethylene signal transduction in Arabidopsis thaliana: five novel mutant loci integrated into a stress response pathway. Genetics 139: 1393–1409 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sakai H, Hua J, Chen QG, Chang C, Medrano LJ, Bleecker AB, Meyerowitz EM (1998) ETR2 is an ETR1-like gene involved in ethylene signaling in Arabidopsis. Proc Natl Acad Sci USA 95: 5812–5817 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Solano R, Stepanova A, Chao QM, Ecker JR (1998) Nuclear events in ethylene signaling: a transcriptional cascade mediated by ETHYLENE-INSENSITIVE3 and ETHYLENE-RESPONSE-FACTOR1. Genes Dev 12: 3703–3714 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su WP, Howell SH (1992) A single genetic-locus, ckr1, defines Arabidopsis mutants in which root-growth is resistant to low concentrations of cytokinin. Plant Physiol 99: 1569–1574 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang X, Woodson WR (1996) Temporal and spatial expression of 1-aminocyclopropane-1-carboxylate oxidase mRNA following pollination of immature and mature petunia flowers. Plant Physiol 112: 503–511 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tieman DM, Ciardi JA, Taylor MG, Klee HJ (2001) Members of the tomato EIN3 gene family are functionally redundant and regulate ethylene responses throughout plant development. Plant J 26: 47–58 [DOI] [PubMed] [Google Scholar]
- Underwood BA (2003) Effects of ethylene on floral fragrance Petunia × hybrida ‘Mitchell Diploid.’ PhD thesis. University of Florida, Gainesville, FL
- Wesley SV, Helliwell CA, Smith NA, Wang M, Rouse DT, Liu Q, Gooding PS, Singh SP, Abbott D, Stoutjesdijk PA, et al (2001) Construct design for efficient, effective and high throughput gene silencing in plants. Plant J 27: 581–590 [DOI] [PubMed] [Google Scholar]
- Wilkinson JQ, Lanahan MB, Clark DG, Bleecker AB, Chang C, Meyerowitz EM, Klee HJ (1997) A dominant mutant receptor from Arabidopsis confers ethylene insensitivity in heterologous plants. Nat Biotechnol 15: 444–447 [DOI] [PubMed] [Google Scholar]
- Wilkinson JQ, Lanahan MB, Yen HC, Giovannoni JJ, Klee HJ (1995) An ethylene-inducible component of signal transduction encoded by never-ripe. Science 270: 1807–1809 [DOI] [PubMed] [Google Scholar]