Abstract
Werner syndrome (WS) is a rare autosomal recessive disorder characterized by a constellation of adult onset phenotypes consistent with an acceleration of intrinsic biological aging. It is caused by pathogenic variants in the WRN gene, which encodes a multifunctional nuclear protein with exonuclease and helicase activities. WRN protein is thought to be involved in optimization of various aspects of DNA metabolism, including DNA repair, recombination, replication, and transcription. In this update, we summarize a total of 83 different WRN mutations, including eight previously unpublished mutations identified by the International Registry of Werner Syndrome (Seattle, WA) and the Japanese Werner Consortium (Chiba, Japan), as well as 75 mutations already reported in the literature. The Seattle International Registry recruits patients from all over the world to investigate genetic causes of a wide variety of progeroid syndromes in order to contribute to the knowledge of basic mechanisms of human aging. Given the unusually high prevalence of WS patients and heterozygous carriers in Japan, the major goal of the Japanese Consortium is to develop effective therapies and to establish management guidelines for WS patients in Japan and elsewhere. This review will also discuss potential translational approaches to this disorder, including those currently under investigation.
Keywords: Werner syndrome, Progeroid syndrome, WRN, RECQL2, RECQ3, RecQ helicase
Introduction
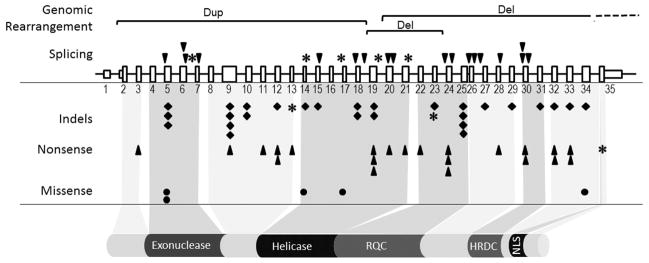
Werner syndrome (WS; MIM# 277700) is a segmental progeroid syndrome whose presentation resembles accelerated aging [Goto, 1997; Oshima et al., 2014]. The WRN gene (RECQL2; MIM# 604611; NM 000553.4, NP 000544.2, NC 000008.11; GRCh38.p7) was positionally cloned in 1996 as the causal gene for WS [Yu et al., 1996]. The WRN locus, located on 8p12, spans approximately 150 kb, including 35 exons, of which 34 are coding (Fig. 1). It encodes a 1,432 amino-acid multifunctional nuclear protein. This is the only recognized product encoded by the wild-type WRN gene; there are no known other alternatively spliced forms that are considered as normal variants, nor are there any other known coding genes within the WRN locus.
Figure 1.
WRN mutation update. The 35 exons of WRN gene locus and WRN protein are shown. Exon 1 and part of 35 are non-coding. The relationship between exons and functional domains is indicated by extending a shaded region from the gene region to the protein functional domains. Known functional domains are indicated by the shaded regions extending from the gene to the protein functional domains. Those domains are the exonuclease and helicase domains, RecQ helicase conserved region (RQC), helicase RNaseD C-terminal conserved region (HRDC), and the nuclear localization signal (NLS). Mutations are grouped by the types shown on the left. Large genomic rearrangements are shown on top using horizontal brackets, labeled “Dup” for duplication or “Del” for deletion. Mutations that affect splicing are indicated with inverted triangles above the WRN locus. Small genomic changes, such as insertions and deletions (Indels), are indicated below the gene locus, using a black-filled diamond shape (◆), whereas stop codons are indicated with a closed triangle (▲). Missense mutations are indicated with black-filled circles (●). Newly identified mutations are indicated for all mutation types by an asterisk (*).
Two major functional domains of the WRN protein are a 3′→5′ exonuclease domain (60–288 aa) in its N-terminal region and an ATP-dependent 3′→5′ helicase domain (551–859 aa) in the central region [Gray et al., 1997; Huang et al., 1998; Perry et al., 2006]. C-terminal to the helicase domain, there are two consensus regions—a RecQ helicase conserved (RQC) region (956–1,064) and a Helicase, RNAse D, C-terminal conserved (HRDC) region (1,142–1,235 aa) [Hu et al., 2005; Kitano et al., 2007; Kitano et al., 2010]. RQC is shared by other RecQ helicases (RECQL1, BLM, RECQL4, and RECQL5), whereas HRDC is shared only by BLM [Croteau et al., 2014]. The RQC domain forms a winged helix-turn-helix structure which interacts with specific substrate DNAs with high affinity, whereas the HRDC domain contains 5 alpha helices thought to mediate interactions with DNA and other proteins [Tadokoro et al., 2012; Kitano, 2014]. A nuclear localization signal (NLS), NKKRRCF, is located near the C-terminal end (1,370–1,375 aa) [Matsumoto et al., 1997b; Suzuki et al., 2001]. An additional nucleolar targeting region was identified within the RQC region [von Kobbe and Bohr, 2002].
The WRN helicase belongs to a DEAH-box containing RecQ family of helicases [Yu et al., 1996; Gray et al., 1997]. Extensive biochemical studies demonstrated a high degree of helicase activity of WRN for the unwinding of unusual DNA structures such as G4 quadruplex sequences and four-way junctions that resemble intermediates of DNA repair and telomere maintenance, as recently reviewed by Croteau et al. (2014). It has been hypothesized that WRN plays a role in the resolution of potentially damaging, complex DNA structures that were accidentally formed during various DNA transactions, including replication, recombination, repair, and transcription [Croteau et al., 2014; Oshima et al., 2016].
In this mutation update, we review the current state of our knowledge of human WRN mutations and document eight previously unpublished mutations. We also discuss ongoing international collaborative efforts toward the development of consensus guidelines for diagnosis and clinical management of WS.
Materials and Methods
Recruitment of Patients
WS patients are referred to the International Registry of Werner Syndrome by physicians who suspect the diagnosis of WS. Biological samples are collected from patients who consent to be enrolled in the study and shipped to the International Registry in Seattle, WA. This study is approved by the Human Subjects Internal Review Board of the University of Washington. Japanese WS patients are anonymously referred to the Japanese Werner Consortium by referring physicians who request a molecular confirmation of a WRN mutation. Consent forms obtained by referring physicians follow local regulations. Isolated specimens of DNA or blood are coded and sent to the Japanese Werner Consortium for the exon sequencing of WRN. This study is approved by the Internal Review Board of Chiba University, Japan.
Sample Processing, Nucleotide Sequencing, and Protein Analysis
Details of blood and skin sample processing have been described previously [Huang et al., 2006]. Briefly, peripheral blood samples from patients and, if available, from members of their nuclear family, are processed immediately upon arrival for cryopreservation of aliquots of primary cells and plasma, isolation of DNA and RNA, and establishment of lymphoblastoid cell lines (LCLs) using Epstein–Barr virus. Punch skin biopsies are used to establish fibroblast cultures. Exon sequencing of WRN utilizes blood DNA, and Western analysis utilizes total protein isolated from LCLs. Detailed protocols for Western analysis and helicase activity assay have also been previously described [Muftuoglu et al., 2008; Friedrich et al., 2010].
Mutations and Polymorphisms of the WRN Gene
The Spectrum of WRN Mutations
To date, a total of 83 different WRN pathogenic variants have been identified across the WRN locus (Fig. 1 and Supp. Table S1). This list of mutations includes 14 reported by other non-registry investigators either in Japan (seven mutations) or outside of Japan (seven mutations) [Goto et al., 1997; Matsumoto et al., 1997a; Uhrhammer et al., 2006; Zhao et al., 2008; Agrelo et al., 2015; Yang et al., 2015]. The mutations include 29 small indels, 23 splicing mutations, 23 nonsense, three genomic rearrangements, and five missense variants. Of these, eight novel mutations identified by the Seattle International Registry of Werner Syndrome (six mutations) or the Japanese Werner Consortium (two mutations) also span across the WRN gene. These newly identified mutations are: c.724G>C, p.V199fs (exon 7 skip); c.1578delC, p.L528fs; c.1720+1G>A, p.V552fs (exon 14 skip); c.1899-3C>G, p.G634fs (exon 17 new splice site); c.2089-1G>C, p.V697fs (exon 19 skip); c.2630+1G>A, p.C817fs (exon 21 skip); c.2772delA, p.A925fs; and c.4216C>T, p.R1406* (“this study” in Supp. Table S1 and * in Fig. 1). Newly reported mutations are added to the Web accessible locus-specific WRN mutation database, http://www.pathology.washington.edu/research/werner/database/.
The majority of WRN mutations are predicted to result in a protein truncation. These are expected to lead to nonsense-mediated decay of mutant mRNAs and/or truncations of C-terminal NLSs, thus resulting in functionally null mutations [Matsumoto et al., 1997b; Yamabe et al., 1997]. There is no existing cellular complementation assay for WS. The interpretations of missense variants follow the general guidelines of the American College of Medical Genetics (ACMG) [Richards et al., 2015]. Pathogenic mechanisms of some of the missense variants have been investigated using recombinant mutant WRN proteins. Two missense variants within the exonuclease domain (p.K125N and p.K135E) were shown to cause protein instability rather than loss of exonuclease activity [Huang et al., 2006]. A missense variant in the helicase domain (p.G574R) was shown to abolish helicase activity [Tadokoro et al., 2013], supporting the notion that loss of enzymatic activities may be pathogenetic mechanism for WS. A second missense variant in the helicase domain (p.R637W), found as a compound heterozygous mutation, was speculated to alter helicase activity, although this has not yet been demonstrated via recombinant protein studies [Uhrhammer et al., 2006]. The pathogenic role of the p.M1350R variant, which was found as the second one for the previously described compound heterozygous case, remains unknown. Clinical phenotypes of compound heterozygotes consisting of one null mutation and one missense variant, however, are indistinguishable from those with two null mutations [Huang et al., 2006; Tadokoro et al., 2013].
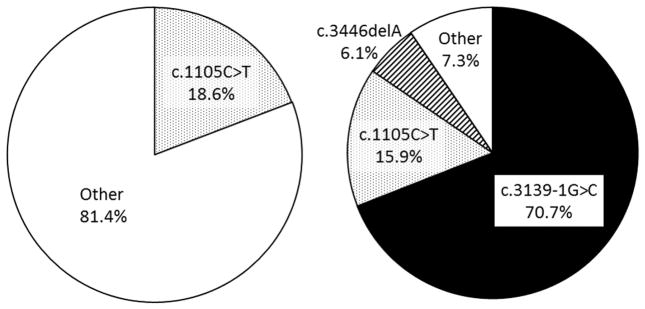
There is a well-established Japanese founder mutation, c.3139-1G>C, r.3139 3233del95 (exon 26 skip). The Japanese carrier frequency of this mutation has been reported to be one out of 167 [Satoh et al., 1999]. The c.3139-1G>C mutation is by far the most frequent WRN disease mutation among genetically confirmed WS cases in the Japanese Werner Consortium—70.7%, or 58 out of 82 mutant alleles (Fig. 2). This is followed by a c.1105C>T, p.R369* mutation, which accounts for 15.9% or 13 out of 82 mutant alleles, and by a c.3446delA, p.E1149fs mutation, which accounts for 6.1% or five out of 82 alleles. These data are essentially in agreement with previous reports from Japan [Goto et al., 1997; Matsumoto et al., 1997a]. The c.3446delA (p.E1149fs) allele has so far been seen only among Japanese patients, raising the possibility of a second founder mutation.
Figure 2.
Frequencies of WRN mutations among non-Japanese and Japanese cases. Frequent mutations of 112 non-Japanese cases in International Registry (Left) and 42 Japanese Consortium (right) are shown.
Interestingly, c.1105C>T, p.R369* is the most common non-Japanese WRN mutation, seen in 18.6% (or 44 out of 236 mutant non-Japanese alleles) in the International Registry cases, indicating that this may be a mutational hot spot across ethnic groups. There have been no haplotype studies for this particular pathogenic variant across various populations. Using publicly available databases, we estimated the disease frequency in the general population. The allele frequencies of c.1105C>T (rs17847577) are 0.0001897 in the ExAC (global) database (Exome Aggregation Consortium, http://exac.broadinstitute.org/) and 0.0003076 in the EVS database (Exome Variant Server, http://evs.gs.washington.edu/EVS/). Estimated WRN mutant allele frequencies were 0.00102 and 0.00165, disease frequencies were calculated to be 1.0 per 106 and 2.7 per 106, and carrier frequencies were one out of 490 and one out of 302, based on ExAC (global) and EVS data, respectively.
Yet another well-established founder mutation, c.2089-3024A>G, r.2088 2089ins106 (resulting in an additional new exon between exons 18 and 19), has been documented among WS patients in the Sardinia region of Italy, where the reported heterozygous frequency is of the order of one out of 120 [Masala et al., 2007].
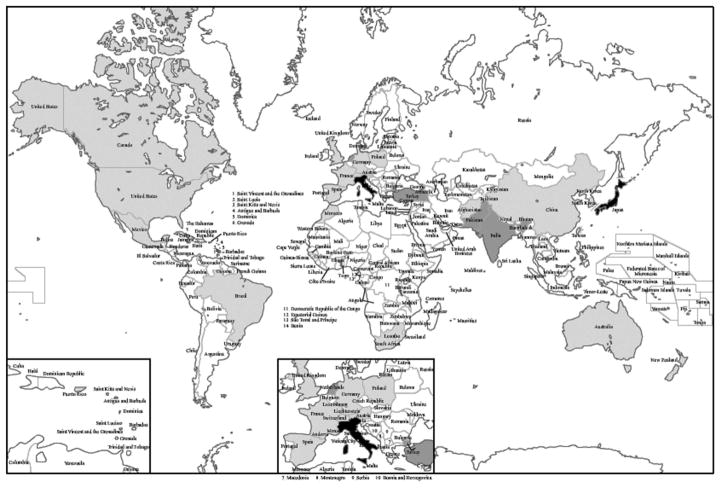
Although less well investigated, apparent ethnically specific mutations (operationally defined as rare unique mutations found in three or more cases from the same region) have been observed in other parts of the world. These include c.561A>G, r.557-654del98 (a new exon 6 splice site) reported in patients originally from India/Pakistan, c.2179dupT, p.C727fs in Morocco, c.3460-2A>G, r.3460 3572del113 (exon 30 skip) in Turkey, and c.3590delA, pN1197fs in the Netherlands [Friedrich et al., 2010; Saha et al., 2013]. Isolated cases have been reported from virtually all continents (Fig. 3).
Figure 3.
Countries of origin of genetically confirmed Werner syndrome patients. In black are countries (Japan and Sardinia, Italy) with known founder mutations with heterozygous frequencies of approximately 1/150. Dark gray indicates countries with possible founder mutations where more than three independent pedigrees with the same mutations have been reported. In light gray are countries with WS patients with documented WRN mutations.
Although no WS cases have yet been reported from Russia and other countries within the former Soviet Union, it is likely that new cases will be identified as our registries and other regional diagnostic facilities become accessible.
Significance of WRN SNPs in Common Age-Related Disorders
WS has long been considered as a model of accelerated aging [Martin, 1978]. Because of the earlier onset of common age-related disorders in WS, a possible involvement of the WRN helicase has been studied in the context of normal aging. Earlier population studies using Finnish centenarian and newborn samples revealed that a common WRN SNP, p.L1074F (rs1800392; MAF 0.447 in ExAC global), may modulate the severity of coronary atherosclerosis and longevity [Castro et al., 2000]. A genome-wide association study (GWAS) of the New England Centenarian Study cohort supported that observation by demonstrating an association between p.L1074F and the age of onset of cardiovascular disease as well as of exceptional longevity, with the leucine being protective [Sebastiani et al., 2012]. Meta-analysis of multiple centenarian cohort studies further confirmed the associations between p.L1074F and exceptional longevity [Sebastiani et al., 2013]. The p.L1074F is located within the RQC domain (amino acid 958–1,052 aa), which interacts with various proteins, including the telomere repeat binding factor 2 (TRF2) crucial for telomere maintenance [Opresko et al., 2002]. It is conceivable that the p.L1074F variation may modulate cardiovascular disease risk and longevity through altering the affinity of this WRN protein for various interacting proteins, although there has not been biological evidence to support this hypothesis.
Based on the publicly available meta-analysis data, Lebel’s group examined p.V114I (rs2230009; MAF 0.060 in ExAC global) and cholesterol metabolism [Berube et al., 2013]. A cell biological study provided evidence that the minor allele coding for isoleucine is associated with less cholesterol efflux as compared with the major allele coding for valine. That observation was in agreement with a population study showing higher cholesterol levels in individuals with Ile at position 114. Although p.V114I is located within the exonuclease domain, there is no evidence that p.V114I alters the enzymatic activity of WRN, but this alteration might be another potential example of an altered protein–protein interaction [Kawabe et al., 2001; Rodriguez-Lopez et al., 2003].
There have been isolated reports of an association between p.C1367R (rs1346044; MAF 0.241 in ExAC global) and the risk of various cancers [Shen et al., 2006; Nakayama et al., 2008; Pinto et al., 2008; Zins et al., 2015]. One study reported that the minor allele coding for Arg increases the risk of breast cancer [Zins et al., 2015], whereas two other studies showed that Arg at position 1,367 is protective against non-Hodgkin lymphoma and soft-tissue sarcomas [Shen et al., 2006; Nakayama et al., 2008]. Another publication reported the absence of an association between this SNP and brain glioma [Pinto et al., 2008]. Given the location of p.C1367R, which is 4 amino acids away from the NLS, functional studies of these two protein variants in relation to the response to DNA damage may prove to be relevant.
Finally, Loeb’s group determined the enzymatic activities of recombinant proteins with various cSNPs in public databases [Kamath-Loeb et al., 2004]. Significant reductions of enzymatic activities were observed in a rare variant, p.R834C, located within the helicase domain, and a modest reduction was noted for another rare variant, p.T172P (rs777437332), located within the exonuclease domain. The biological significance of these observations remains to be determined.
Taken as a whole, the above findings support the proposition that WRN is not only a cause of WS; it may also play a role in the development of specific age-related disorders. The degree to which a given variant may modulate a given age-related disorder likely depends upon the genetic background of the individual subject, as well as his or her cultural background and the “exposome” (http://humanexposomeproject.com/).
Biological Significance and Clinical Relevance of WRN Mutations
WS is the prototypical example of a segmental progeroid syndrome, a group of rare genetic disorders in which patients present with putative accelerated rates of aging and succumb to the complications of aging processes [Martin, 1978]. WS patients usually develop normally until they reach the second decade of life. The first clinical sign is the lack of the pubertal growth spurt during the teen years. Patients begin to suffer from skin atrophy with loss of subcutaneous fat and loss and/or graying of hair in their 20s which, combined with short stature, gives an overall aged appearance. Bilateral ocular cataracts and type 2 diabetes mellitus are generally recognized while patients are in their 20–30s, followed by a series of common age-related disorders including osteoporosis, gonadal atrophy, atherosclerosis, and various malignancies [Goto, 1997; Oshima et al., 2014; Takemoto et al., 2013]. Cataracts are seen in virtually all WS patients, one of the characteristics of WS not seen in some other progeroid syndrome, such as laminopathies [Oshima et al., 2014]. Other symptoms include a high pitched hoarse voice, characteristic facial features (a “inched facial” appearance), thin limbs, truncal obesity, and flat feet. The most common causes of death are from cancers and myocardial infarctions, at a median age of 54 years [Huang et al., 2006; Goto et al., 2013].
One pathognomonic feature is an indolent deep skin ulcer around the ankles associated with soft tissue calcifications, seen in more than 80% of cases [Takemoto et al., 2013]. This is a major quality-of-life issue, as patients often become wheel-chair bound as a result of complications, including amputations.
WS patients are known to have a higher incidence of cancers compared with the general population. In a recent literature review, the most common cancers in WS in Japan were found to be thyroid cancers, malignant melanomas, meningiomas, and soft tissue sarcomas, with a median age at diagnosis of 45 years [Lauper et al., 2013].
The natural history of the disease appears to be remarkably similar across various ethnic groups [Goto, 1997; Huang et al., 2006; Saha et al., 2013]. The median age for the diagnosis of bilateral ocular cataracts was 31 years and the median age of WS diagnosis was 37 for both non-Japanese and Japanese patients [Goto, 1997; Huang et al., 2006]. Ages of death were recently reported to be 54 and 55 years in non-Japanese and Japanese reports, respectively, both of which represent improvements over what had been reported in the previous century [Huang et al., 2006; Goto et al., 2013].
The clinical phenotypes of WS appear to be indistinguishable among WRN mutation types or geographical locations. This is in agreement with the above mentioned molecular studies of WRN mutations showing that all mutations are functionally null, except for a few missense mutations. However, a careful clinical assessment revealed a possible genotype–phenotype correlation of the type of thyroid carcinoma among Japanese WS patients [Ishikawa et al., 1999]. Follicular carcinomas were associated with the c.3139-1G>C mutation (exon 26 skip) and papillary carcinoma with the c.1105C>T, p.R369* mutation [Ishikawa et al., 1999]. Further studies may reveal the phenotypic variation due to either genetic background or environmental variations.
Currently WS is the only known Mendelian disorder associated with the WRN locus. It is inherited exclusively as an autosomal recessive trait, and its penetrance is believed to be 100% [Hisama et al., 2015]. A very unusual case of microcephaly, developmental delay, diabetes mellitus, hypertriglyceridemia, adenomatoid thyroid nodules, and pituitary microadenoma was observed in a patient initially referred to us at age 17 in whom an extensive genetic workup revealed a homozygous p.R369* mutation (Registry# CI1010). This is an isolated case and more likely associated with other genetic or non-genetic causes yet to be discovered.
Although we did not observe clear phenotypic differences among WS patients with different mutations, either null or missense, careful assessment may reveal slight differences in the severity among different disease mutations in the future.
Mouse Models of WS
Initial efforts to develop a WRN knockout mouse model failed to reveal overt WS phenotypes [Lebel and Leder, 1998; Lombard et al., 2000; Wang et al., 2000]. This was thought to be due, in part, to the extremely long telomeres of commonly used laboratory mice such as C57BL6 and 129, as studies of human WS cells suggested that telomere attrition may be required for the phenotypic expression [Crabbe et al., 2007]. Mice and humans differ, of course, in many other aspects of their biochemical genetics. One such potentially significant difference is the gene action which leads to the intracellular localization of the WRN protein. The human WRN protein is localized to nucleoli and is translocated to the nucleoplasm upon DNA damage. The mouse WRN protein shows default localization to the nucleoplasm [Suzuki et al., 2001; Lan et al., 2005].
These difficulties were partially overcome by crossing WRN knockout mice with other knockout lines. The homozygous WRN null mice, Wrn−/−, appeared to live beyond 2 years of age without developing obvious phenotypes [Lombard et al., 2000]. When crossed with p53 null mice, Wrn−/−;p53−/− mice lived slightly shorter than Wrn+/−;p53−/− (149 vs. 129 days; P = 0.016), which suggested an interaction between WRN and p53 [Lombard et al., 2000]. When the same WRN null mice were crossed with mice deficient in the RNA component of telomerase, some, but not all, 4–6th generation offspring of Wrn−/−;mTerc−/− exhibited appearances of accelerated aging, including osteoporosis, type II diabetes and cataracts [Chang et al., 2004]. Cells derived from these Wrn−/−;mTerc−/− offspring showed chromosomal aberrations and telomere abnormalities mimicking human WRN cells [Laud et al., 2005]. There was also an upregulation of the p53/p21 pathway during osteoblast differentiation [Wang et al., 2012]. Subsequently, a triple knockout of Wrn−/−;BlmM3/M3;Terc−/− was generated using a hypomorphic mutant of the BLM helicase [Du et al., 2004], in which 3rd generation offspring showed median lifespans of 7 months compared with greater than 10 months in other genotypes. The extent to which these abnormal phenotypes can be attributed to Wrn−/−, remains uncertain, however, as mTerc−/− alone exhibits considerably more phenotypes than Wrn−/− alone [Rudolph et al., 1999].
Another WRN mouse model carries an in-frame deletion of the helicase domain, WrnΔhel/Δhel, while retaining the other domains of the WRN protein [Lebel and Leder, 1998]. This line was also phenotypically normal, at least during the first year of life [Lebel and Leder, 1998; Labbe et al., 2011]. However, side-by-side comparison with the null mutant, Wrn−/−, revealed that median lifespans were 18.7 months in WrnΔhel/Δhel, 21.4 months in Wrn−/− and 22.8 months in littermate controls [Aumailley et al., 2015b]. Inflammatory cytokines, IL-10 and PAI-I (Plasminogen Activator Inhibitor-1), as well as autophagy influx, were significantly increased in WrnΔhel/Δhel mice compared with Wrn−/− or wild-type controls. Interestingly, stable mutant WRN proteins of WrnΔhel/Δhel were mislocalized to the endoplasmic reticulum and the peroxisomal fractions, which may be responsible for the additional phenotypes in WrnΔhel/Δhel not seen in Wrn−/− [Aumailley et al., 2015b].
WS Patient Registries
The International Registry of Werner Syndrome (www.wernersyndrome.org) was established in 1988 to collect clinically diagnosed WS cases for the positional cloning of the WRN gene [Yu et al., 1996]. Following the successful cloning, the Registry expanded its scope to include progeroid cases with unknown genetic causes as part of the laboratory’s interest in the discovery of a variety of gene actions involved in the biology and pathobiology of aging. For all such cases, however, WRN coding exons are sequenced and, when cells are available, Western blot analysis of WRN protein is performed [Huang et al., 2006; Muftuoglu et al., 2008; Friedrich et al., 2010].
Patients with progeroid features who are referred to our Registry but who do not carry WRN mutations are operationally categorized as “atypical Werner syndrome.” In such cases, we conduct extensive genome-wide analysis, using combinations of array CGH, linkage, and exome sequencing, to identify the putative genetic causes of these patients. This initiative has so far led to the identification of progeroid syndromes caused by mutations in LMNA (nuclear structure and chromatin interaction) [Chen et al., 2003; Hisama et al., 2011], POLD1 (DNA polymerase delta) [Lessel et al., 2015], SPRTN (DNA replication and replication-related G2/M-checkpoint regulation) [Lessel et al., 2014b] and SAMHD1 (regulation of dNTP pools) [Lessel et al., 2014a]. While a canonical C-terminal 50 amino acid deletion within the lamin A protein encoded by the LMNA gene had already been discovered as the basis of almost all cases of the Hutchinson-Gilford syndrome (the classical Progeria of Childhood) [Eriksson et al., 2003], our adult onset cases were associated with mutations within other domains of that gene [Saha et al., 2010; Hisama et al., 2011]. Pedigrees of a BSCL2 mutation known to be responsible for Berardinelli-Seip syndrome as well as a single instance of trisomy 8 mosaicism were also identified among our atypical WS cases [Oshima and Hisama, 2014].
The Seattle International Registry currently has enrolled 155 WRN mutant cases, including 25 cases enrolled since 2010 [Friedrich et al., 2010]. Other currently enrolled cases (and available family members) include 12 with LMNA mutations, eight with POLD1 mutations and 60 other subjects with segmental progeroid phenotypes currently under investigation.
The Japanese Werner Consortium (www.m.chiba-u.jp/class/clin-cellbiol/werner/) was started with the eventual goal of establishing evidence-based management guidelines. A nationwide epidemiological survey of WS patients was conducted between 2009 and 2011 in order to obtain up-to-date natural histories of the disease and to re-evaluate clinical diagnostic guidelines [Okabe et al., 2012; Onishi et al., 2012; Tadokoro et al., 2013]. The first draft of revised diagnostic criteria, published in 2013, most notably included X-rays of ankles showing previously unrecognized characteristic patterns of soft-tissue calcification [Takemoto et al., 2013]. This finding is clinically important because the single most debilitating problem affecting the patients’ quality of life are the indolent deep skin ulcers around ankles and less frequently, around elbows. These lesions will often eventually lead to amputations. Additional clinically significant findings from a nationwide survey included surprising evidence, specifically among diabetic Werner patients (N = 102), of a high incidence of epithelial neoplasms, but not of sarcomas [Onishi et al., 2012], in contrast to their control group of non-diabetic Werner patients (N = 61). Diabetic Werner patients were also shown to have an elevated incidence of hyperlipidemia and cardiovascular disease compared with their non-diabetic cohort of Werner patients. Clinical results of the pharmacological management of diabetic Werner patients have also been favorable [Yokote et al., 2004; Kitamoto et al., 2012; Ide et al., 2016]. The successful control of diabetes led to a reduction of abdominal fat mass and to improvements of vascular function as assessed by flow-mediated vasodilation [Ide et al., 2016]. Efforts to establish management guideline are continuing under the supervision of the Japanese Consortium’s leadership group.
Translational Prospects
To date, there is no cure for WS. The clinical management generally consists of the treatment of specific manifestations and the screening for and prevention of complications such as those associated with diabetes and skin ulcers as described above. Various therapeutic measures have been explored primarily to delay the progression of symptoms. Pharmacological reagents are being tested for their ability to prevent accelerated cellular senescence of WS fibroblasts. One such example is the use of inhibitors of p38 mitogen-activated protein kinase (MAPK). The MAPK pathway mediates Ras stress-signaling to activate a potent tumor suppressor, p53/p21WAF1, and to induce cellular senescence [Xu et al., 2014]. MAPK also stimulates NF-κB transcriptional activity, which in turn induces chronic inflammatory cytokines, part of a phenotype of senescent cells termed senescence-associated secretory phenotype (SASP) [Freund et al., 2011]. Selective inhibitors of p38MAPK, such as SB203580, were shown to prevent accumulation of p21WAF1 and to restore the replicative capacity of WS fibroblasts [Tivey et al., 2013; Davis et al., 2016].
Another example is the use of inhibitors of the mTOR pathway, a key modulator of aging and age-related diseases in diverse organisms [Johnson et al., 2015]. Cells deficient in WRN have elevated basal autophagy, possibly reflecting increased levels of oxidative stress [Talaei et al., 2013; Saha et al., 2014]. Long-term treatment of WS fibroblasts with an mTORC1 inhibitor, rapamycin, resulted in the normalization of autophagy levels, reduction of DNA damage accumulation, and partial restoration of cell growth [Talaei et al., 2013; Saha et al., 2014]. Although not tested in WS, metformin might be another candidate to consider for the treatment of WS. Metformin has been used for the treatment of diabetes for many years, has been claimed to be protective against other age-related disorders and is expected to undergo a clinical trial for amelioration of aging phenotypes via the TAME study (Targeting Aging with Metformin [Barzilai et al., 2016]. Metformin has been shown to have a good safety profile [Barzilai et al., 2016].
Motivated by the free radical theory of aging, an anti-oxidant, vitamin C, has been tested in WS fibroblasts, in WRN-null mesenchymal stem cells (MSC) and in a mouse model of WS, all with favorable results [Kashino et al., 2003; Aumailley et al., 2015a; Li et al., 2016]. In mice carrying helicase deficient WRN (WrnΔhel/Δhel), vitamin C treatment resulted in the normalization of inflammation markers and fatty acid metabolism [Aumailley et al., 2015a]. In WS MSC, vitamin C suppressed induction of major senescent makers, including SASP, GATA4, and p16Ink4a [Li et al., 2016].
There also is a single case report of a Japanese WS patient with a fatty liver who was treated with astaxanthin, a powerful antioxidant [Takemoto et al., 2015]. Administration of this keto-cartinoid antioxidant markedly improved the patient’s fatty liver, possibly via its anti-inflammatory and anti-oxidative left properties.
Correction of nonsense mutations were enabled by the use of pharmacological reagents to read-through premature termination codons (PTC) in WS LCLs carrying either homozygous p.S1256* or homozygous p.R369* mutations [Agrelo et al., 2015]. Both amino-glycosides and PTC-124 partially bypassed PTC to produce full length WRN protein that localized to nuclei, enough to rescue cells from hypersensitivity to a genotoxic agent in culture.
Advances in human induced pluripotent cells (hiPSCs) offer a potentially new approach to disease modeling and to treatment. There are at least two independent reports of the successful establishment of hiPSC derived from WS patient fibroblasts [Cheung et al., 2014; Shimamoto et al., 2014]. Despite the high frequency of karyotypic instability in WS fibroblasts known as variegated translocation mosaicism [Salk et al., 1981], these WS hiPSC were karyotypically stable [Shimamoto et al., 2014]. WS human embryonic stem cells (hESCs) have also been generated via a WRN knockout technique in hESCs [Zhang et al., 2015]. Combined with recent developments of gene editing, these new directions could one day provide the basis for a stem-cell based therapy applicable, for example, for the treatment of the indolent leg ulcers of WS patients.
Future Prospects
For a variety of reasons, WS almost certainly has been under-diagnosed in many regions of the world. Given the demographic trends of aging populations around the world, however [UN, 2002], research on the biochemical genetic basis of biological aging and on diseases of aging are likely to receive much greater attention by governmental and private funding agencies, physicians, and the general public. Such a trend, when coupled with the growth of genomics centers capable of providing rapid, competent, and economic molecular diagnoses, should reveal many more cases of WS and of other segmental progeroid syndromes. As noted above, we also anticipate a welcome acceleration of translational research to address the needs of these patients. It is even conceivable that some of these translational interventions will ultimately be utilized for the retardation of normative aging in the general population, hopefully with major impacts upon healthspans and not just lifespans.
It is also our hope that much more attention will be given to the health status of the much larger numbers of heterozygous carriers of pathogenic WRN mutations. Laboratory research has already documented the vulnerability of cultured somatic cells from such subjects to certain genotoxic agents [Ogburn et al., 1997]. These studies need to be followed up by appropriate epidemiological investigations.
Finally, it should be noted that some of us (FMH, JO, GMM) have called attention to yet another neglected area of research—the search for rare alleles at loci responsible for WS and other segmental progeroid syndromes that may have remarkably enhanced functions; we have referred to such putative alleles as “antiprogeroid” alleles [Hisama et al., 2016].
Table 1.
Potential Therapeutic Approaches Currently Being Investigated
| Mode of action | Stage | References | |
|---|---|---|---|
| p38 MAPK inhibitor | Inhibition of p53 pathway and SASP | In vitro | [Davis et al., 2005; Davis et al., 2016; Tivey et al., 2013] |
| mTOR inhibitor | Stimulation of autophagy | In vitro | [Saha et al., 2014; Talaei et al., 2013] |
| Vitamin C | Reduction of ROS | In vitro | [Aumailley et al., 2015a; Kashino et al., 2003; Li et al., 2016] |
| Astaxanthin | Reduction of ROS | In vivo | [Takemoto et al., 2015] |
| PTC read-through | Bypassing nonsense mutation | In vitro | [Agrelo et al., 2015] |
| Stem cell gene therapy | Gene editing of stem cells | In vivo | [Cheung et al., 2014; Shimamoto et al., 2014; Shimamoto et al., 2015; Zhang et al., 2015] |
Acknowledgments
Contract grant sponsors: NIH/NIA (R24AG42328); NIH/NCI (R01CA210916); Ministry of Health, Labour and Welfare of Japan; Agency for Medical Research and Development (AMED); Ministry of Education, Culture, Sports, Science and Technology of Japan (26115009).
Disclosure statement: The authors declare no conflict of interest.
Footnotes
Additional Supporting Information may be found in the online version of this article.
References
- Agrelo R, Sutz MA, Setien F, Aldunate F, Esteller M, Da Costa V, Achenbach R. A novel Werner Syndrome mutation: pharmacological treatment by read-through of nonsense mutations and epigenetic therapies. Epigenetics. 2015;10:329–341. doi: 10.1080/15592294.2015.1027853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aumailley L, Dubois MJ, Garand C, Marette A, Lebel M. Impact of vitamin C on the cardiometabolic and inflammatory profiles of mice lacking a functional Werner syndrome protein helicase. Exp Gerontol. 2015a;72:192–203. doi: 10.1016/j.exger.2015.10.012. [DOI] [PubMed] [Google Scholar]
- Aumailley L, Garand C, Dubois MJ, Johnson FB, Marette A, Lebel M. Metabolic and phenotypic differences between mice producing a Werner syndrome helicase mutant protein and WRN null mice. PLoS One. 2015b;10:e0140292. doi: 10.1371/journal.pone.0140292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barzilai N, Crandall JP, Kritchevsky SB, Espeland MA. Metformin as a tool to target aging. Cell Metab. 2016;23:1060–1065. doi: 10.1016/j.cmet.2016.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berube J, Garand C, Lettre G, Lebel M. The non-synonymous polymorphism at position 114 of the WRN protein affects cholesterol efflux in vitro and correlates with cholesterol levels in vivo. Exp Gerontol. 2013;48:533–538. doi: 10.1016/j.exger.2013.03.003. [DOI] [PubMed] [Google Scholar]
- Castro E, Edland SD, Lee L, Ogburn CE, Deeb SS, Brown G, Panduro A, Riestra R, Tilvis R, Louhija J, Penttinen R, Erkkola R, et al. Polymorphisms at the Werner locus: II. 1074Leu/Phe, 1367Cys/Arg, longevity, and atherosclerosis. Am J Med Genet. 2000;95:374–380. [PubMed] [Google Scholar]
- Chang S, Multani AS, Cabrera NG, Naylor ML, Laud P, Lombard D, Pathak S, Guarente L, DePinho RA. Essential role of limiting telomeres in the pathogenesis of Werner syndrome. Nat Genet. 2004;36:877–882. doi: 10.1038/ng1389. [DOI] [PubMed] [Google Scholar]
- Chen L, Lee L, Kudlow BA, Dos Santos HG, Sletvold O, Shafeghati Y, Botha EG, Garg A, Hanson NB, Martin GM, et al. LMNA mutations in atypical Werner’s syndrome. Lancet. 2003;362:440–445. doi: 10.1016/S0140-6736(03)14069-X. [DOI] [PubMed] [Google Scholar]
- Cheung HH, Liu X, Canterel-Thouennon L, Li L, Edmonson C, Rennert OM. Telomerase protects werner syndrome lineage-specific stem cells from premature aging. Stem Cell Rep. 2014;2:534–546. doi: 10.1016/j.stemcr.2014.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crabbe L, Jauch A, Naeger CM, Holtgreve-Grez H, Karlseder J. Telomere dysfunction as a cause of genomic instability in Werner syndrome. Proc Natl Acad Sci USA. 2007;104:2205–2210. doi: 10.1073/pnas.0609410104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Croteau DL, Popuri V, Opresko PL, Bohr VA. Human RecQ helicases in DNA repair, recombination, and replication. Annu Rev Biochem. 2014;83:519–552. doi: 10.1146/annurev-biochem-060713-035428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis T, Baird DM, Haughton MF, Jones CJ, Kipling D. Prevention of accelerated cell aging in Werner syndrome using a p38 mitogen-activated protein kinase inhibitor. J Gerontol A Biol Sci Med Sci. 2005;60:1386–1393. doi: 10.1093/gerona/60.11.1386. [DOI] [PubMed] [Google Scholar]
- Davis T, Brook AJ, Rokicki MJ, Bagley MC, Kipling D. Evaluating the role of p38 MAPK in the accelerated cell senescence of Werner syndrome fibroblasts. Pharmaceuticals (Basel) 2016;9:23. doi: 10.3390/ph9020023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Du X, Shen J, Kugan N, Furth EE, Lombard DB, Cheung C, Pak S, Luo G, Pignolo RJ, DePinho RA, et al. Telomere shortening exposes functions for the mouse Werner and Bloom syndrome genes. Mol Cell Biol. 2004;24:8437–8446. doi: 10.1128/MCB.24.19.8437-8446.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eriksson M, Brown WT, Gordon LB, Glynn MW, Singer J, Scott L, Erdos MR, Robbins CM, Moses TY, Berglund P, et al. Recurrent de novo point mutations in lamin A cause Hutchinson-Gilford progeria syndrome. Nature. 2003;423:293–298. doi: 10.1038/nature01629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freund A, Patil CK, Campisi J. p38MAPK is a novel DNA damage response-independent regulator of the senescence-associated secretory phenotype. EMBO J. 2011;30:1536–1548. doi: 10.1038/emboj.2011.69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friedrich K, Lee L, Leistritz DF, Nurnberg G, Saha B, Hisama FM, Eyman DK, Lessel D, Nurnberg P, Li C, et al. WRN mutations in Werner syndrome patients: genomic rearrangements, unusual intronic mutations and ethnic-specific alterations. Hum Genet. 2010;128:103–111. doi: 10.1007/s00439-010-0832-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goto M. Hierarchical deterioration of body systems in Werner’s syndrome: implications for normal ageing. Mech Ageing Dev. 1997;98:239–254. doi: 10.1016/s0047-6374(97)00111-5. [DOI] [PubMed] [Google Scholar]
- Goto M, Imamura O, Kuromitsu J, Matsumoto T, Yamabe Y, Tokutake Y, Suzuki N, Mason B, Drayna D, Sugawara M, et al. Analysis of helicase gene mutations in Japanese Werner’s syndrome patients. Hum Genet. 1997;99:191–193. doi: 10.1007/s004390050336. [DOI] [PubMed] [Google Scholar]
- Goto M, Ishikawa Y, Sugimoto M, Furuichi Y. Werner syndrome: a changing pattern of clinical manifestations in Japan (1917~2008) Biosci Trends. 2013;7:13–22. [PubMed] [Google Scholar]
- Gray MD, Shen JC, Kamath-Loeb AS, Blank A, Sopher BL, Martin GM, Oshima J, Loeb LA. The Werner syndrome protein is a DNA helicase. Nat Genet. 1997;17:100–103. doi: 10.1038/ng0997-100. [DOI] [PubMed] [Google Scholar]
- Hisama FM, Kubisch C, Martin GM, Oshima J. Clinical utility gene card for: Werner Syndrome–Update 2014. Eur J Hum Genet. 2015:23. doi: 10.1038/ejhg.2014.171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hisama FM, Lessel D, Leistritz D, Friedrich K, McBride KL, Pastore MT, Gottesman GS, Saha B, Martin GM, Kubisch C, et al. Coronary artery disease in a Werner syndrome-like form of progeria characterized by low levels of progerin, a splice variant of lamin A. Am J Med Genet A. 2011;155A:3002–3006. doi: 10.1002/ajmg.a.34336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hisama FM, Oshima J, Martin GM. How research on human progeroid and antigeroid syndromes can contribute to the longevity dividend initiative. Cold Spring Harb Perspect Med. 2016:6. doi: 10.1101/cshperspect.a025882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu JS, Feng H, Zeng W, Lin GX, Xi XG. Solution structure of a multifunctional DNA- and protein-binding motif of human Werner syndrome protein. Proc Natl Acad Sci USA. 2005;102:18379–18384. doi: 10.1073/pnas.0509380102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang S, Lee L, Hanson NB, Lenaerts C, Hoehn H, Poot M, Rubin CD, Chen DF, Yang CC, Juch H, et al. The spectrum of WRN mutations in Werner syndrome patients. Hum Mutat. 2006;27:558–567. doi: 10.1002/humu.20337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang S, Li B, Gray MD, Oshima J, Mian IS, Campisi J. The premature ageing syndrome protein, WRN, is a 3′–>5′ exonuclease. Nat Genet. 1998;20:114–116. doi: 10.1038/2410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ide S, Yamamoto M, Takemoto M, Fujimoto M, Ide K, Kobayashi K, Yokote K. Improved glycemic control and vascular function and reduction of abdominal fat accumulation with liraglutide in a case of Werner syndrome with diabetes mellitus. J Am Geriatr Soc. 2016;64:687–688. doi: 10.1111/jgs.13970. [DOI] [PubMed] [Google Scholar]
- Ishikawa Y, Sugano H, Matsumoto T, Furuichi Y, Miller RW, Goto M. Unusual features of thyroid carcinomas in Japanese patients with Werner syndrome and possible genotype-phenotype relations to cell type and race. Cancer. 1999;85:1345–1352. [PubMed] [Google Scholar]
- Johnson SC, Sangesland M, Kaeberlein M, Rabinovitch PS. Modulating mTOR in aging and health. Interdiscip Top Gerontol. 2015;40:107–127. doi: 10.1159/000364974. [DOI] [PubMed] [Google Scholar]
- Kamath-Loeb AS, Welcsh P, Waite M, Adman ET, Loeb LA. The enzymatic activities of the Werner syndrome protein are disabled by the amino acid polymorphism R834C. J Biol Chem. 2004;279:55499–55505. doi: 10.1074/jbc.M407128200. [DOI] [PubMed] [Google Scholar]
- Kashino G, Kodama S, Nakayama Y, Suzuki K, Fukase K, Goto M, Watanabe M. Relief of oxidative stress by ascorbic acid delays cellular senescence of normal human and Werner syndrome fibroblast cells. Free Radic Biol Med. 2003;35:438–443. doi: 10.1016/s0891-5849(03)00326-5. [DOI] [PubMed] [Google Scholar]
- Kawabe Y, Branzei D, Hayashi T, Suzuki H, Masuko T, Onoda F, Heo SJ, Ikeda H, Shimamoto A, Furuichi Y, et al. A novel protein interacts with the Werner’s syndrome gene product physically and functionally. J Biol Chem. 2001;276:20364–20369. doi: 10.1074/jbc.C100035200. [DOI] [PubMed] [Google Scholar]
- Kitamoto T, Takemoto M, Fujimoto M, Ishikawa T, Onishi S, Okabe E, Ishibashi R, Kobayashi K, Kawamura H, Yokote K. Sitagliptin successfully ameliorates glycemic control in Werner syndrome with diabetes. Diabetes Care. 2012;35:e83. doi: 10.2337/dc12-1179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kitano K. Structural mechanisms of human RecQ helicases WRN and BLM. Front Genet. 2014;5:366. doi: 10.3389/fgene.2014.00366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kitano K, Kim SY, Hakoshima T. Structural basis for DNA strand separation by the unconventional winged-helix domain of RecQ helicase WRN. Structure. 2010;18:177–187. doi: 10.1016/j.str.2009.12.011. [DOI] [PubMed] [Google Scholar]
- Kitano K, Yoshihara N, Hakoshima T. Crystal structure of the HRDC domain of human Werner syndrome protein, WRN. J Biol Chem. 2007;282:2717–2728. doi: 10.1074/jbc.M610142200. [DOI] [PubMed] [Google Scholar]
- Labbe A, Garand C, Cogger VC, Paquet ER, Desbiens M, Le Couteur DG, Lebel M. Resveratrol improves insulin resistance hyperglycemia and hepatosteatosis but not hypertriglyceridemia, inflammation, and life span in a mouse model for Werner syndrome. J Gerontol A Biol Sci Med Sci. 2011;66:264–278. doi: 10.1093/gerona/glq184. [DOI] [PubMed] [Google Scholar]
- Lan L, Nakajima S, Komatsu K, Nussenzweig A, Shimamoto A, Oshima J, Yasui A. Accumulation of Werner protein at DNA double-strand breaks in human cells. J Cell Sci. 2005;118:4153–4162. doi: 10.1242/jcs.02544. [DOI] [PubMed] [Google Scholar]
- Laud PR, Multani AS, Bailey SM, Wu L, Ma J, Kingsley C, Lebel M, Pathak S, DePinho RA, Chang S. Elevated telomere-telomere recombination in WRN-deficient, telomere dysfunctional cells promotes escape from senescence and engagement of the ALT pathway. Genes Dev. 2005;19:2560–2570. doi: 10.1101/gad.1321305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lauper JM, Krause A, Vaughan TL, Monnat RJ., Jr Spectrum and risk of neoplasia in Werner syndrome: a systematic review. PLoS One. 2013;8:e59709. doi: 10.1371/journal.pone.0059709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lebel M, Leder P. A deletion within the murine Werner syndrome helicase induces sensitivity to inhibitors of topoisomerase and loss of cellular proliferative capacity. Proc Natl Acad Sci USA. 1998;95:13097–13102. doi: 10.1073/pnas.95.22.13097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lessel D, Hisama FM, Szakszon K, Saha B, Sanjuanelo AB, Salbert BA, Steele PD, Baldwin J, Brown WT, Piussan C, et al. POLD1 Germline Mutations in Patients Initially Diagnosed with Werner Syndrome. Hum Mutat. 2015;36:1070–1079. doi: 10.1002/humu.22833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lessel D, Saha B, Hisama F, Kaymakamzade B, Nurlu G, Gursoy-Ozdemir Y, Thiele H, Nurnberg P, Martin GM, Kubisch C, et al. Atypical Aicardi-Goutieres syndrome: is the WRN locus a modifier? Am J Med Genet A. 2014a;164A:2510–2513. doi: 10.1002/ajmg.a.36664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lessel D, Vaz B, Halder S, Lockhart PJ, Marinovic-Terzic I, Lopez-Mosqueda J, Philipp M, Sim JC, Smith KR, Oehler J, et al. Mutations in SPRTN cause early onset hepatocellular carcinoma, genomic instability and progeroid features. Nat Genet. 2014b;46:1239–1244. doi: 10.1038/ng.3103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y, Zhang W, Chang L, Han Y, Sun L, Gong X, Tang H, Liu Z, Deng H, Ye Y, et al. Vitamin C alleviates aging defects in a stem cell model for Werner syndrome. Protein Cell. 2016;7:478–488. doi: 10.1007/s13238-016-0278-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lombard DB, Beard C, Johnson B, Marciniak RA, Dausman J, Bronson R, Buhlmann JE, Lipman R, Curry R, Sharpe A, et al. Mutations in the WRN gene in mice accelerate mortality in a p53-null background. Mol Cell Biol. 2000;20:3286–3291. doi: 10.1128/mcb.20.9.3286-3291.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin GM. Genetic syndromes in man with potential relevance to the pathobiology of aging. Birth Defects Orig Artic Ser. 1978;14:5–39. [PubMed] [Google Scholar]
- Masala MV, Scapaticci S, Olivieri C, Pirodda C, Montesu MA, Cuccuru MA, Pruneddu S, Danesino C, Cerimele D. Epidemiology and clinical aspects of Werner’s syndrome in North Sardinia: description of a cluster. Eur J Dermatol. 2007;17:213–216. doi: 10.1684/ejd.2007.0155. [DOI] [PubMed] [Google Scholar]
- Matsumoto T, Imamura O, Yamabe Y, Kuromitsu J, Tokutake Y, Shimamoto A, Suzuki N, Satoh M, Kitao S, Ichikawa K, et al. Mutation and haplotype analyses of the Werner’s syndrome gene based on its genomic structure: genetic epidemiology in the Japanese population. Hum Genet. 1997a;100:123–130. doi: 10.1007/s004390050477. [DOI] [PubMed] [Google Scholar]
- Matsumoto T, Shimamoto A, Goto M, Furuichi Y. Impaired nuclear localization of defective DNA helicases in Werner’s syndrome. Nat Genet. 1997b;16:335–336. doi: 10.1038/ng0897-335. [DOI] [PubMed] [Google Scholar]
- Muftuoglu M, Oshima J, von Kobbe C, Cheng WH, Leistritz DF, Bohr VA. The clinical characteristics of Werner syndrome: molecular and biochemical diagnosis. Hum Genet. 2008;124:369–377. doi: 10.1007/s00439-008-0562-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakayama R, Sato Y, Masutani M, Ogino H, Nakatani F, Chuman H, Beppu Y, Morioka H, Yabe H, Hirose H, et al. Association of a missense single nucleotide polymorphism, Cys1367Arg of the WRN gene, with the risk of bone and soft tissue sarcomas in Japan. Cancer Sci. 2008;99:333–339. doi: 10.1111/j.1349-7006.2007.00692.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogburn CE, Oshima J, Poot M, Chen R, Hunt KE, Gollahon KA, Rabinovitch PS, Martin GM. An apoptosis-inducing genotoxin differentiates heterozygotic carriers for Werner helicase mutations from wild-type and homozygous mutants. Hum Genet. 1997;101:121–125. doi: 10.1007/s004390050599. [DOI] [PubMed] [Google Scholar]
- Okabe E, Takemoto M, Onishi S, Ishikawa T, Ishibashi R, He P, Kobayashi K, Fujimoto M, Kawamura H, Yokote K. Incidence and characteristics of metabolic disorders and vascular complications in individuals with Werner syndrome in Japan. J Am Geriatr Soc. 2012;60:997–998. doi: 10.1111/j.1532-5415.2012.03944.x. [DOI] [PubMed] [Google Scholar]
- Onishi S, Takemoto M, Ishikawa T, Okabe E, Ishibashi R, He P, Kobayashi K, Fujimoto M, Kawamura H, Yokote K. Japanese diabetic patients with Werner syndrome exhibit high incidence of cancer. Acta Diabetol. 2012;49(Suppl 1):S259–S260. doi: 10.1007/s00592-012-0424-z. [DOI] [PubMed] [Google Scholar]
- Opresko PL, von Kobbe C, Laine JP, Harrigan J, Hickson ID, Bohr VA. Telomere-binding protein TRF2 binds to and stimulates the Werner and Bloom syndrome helicases. J Biol Chem. 2002;277:41110–41119. doi: 10.1074/jbc.M205396200. [DOI] [PubMed] [Google Scholar]
- Oshima J, Hisama FM. Search and insights into novel genetic alterations leading to classical and atypical Werner syndrome. Gerontology. 2014;60:239–246. doi: 10.1159/000356030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oshima J, Martin GM, Hisama FM. Werner syndrome. In: Pagon RA, Adam MP, Ardinger HH, Wallace SE, Amemiya A, Bean LJH, Bird TD, Fong CT, Mefford HC, Smith RJH, et al., editors. GeneReviews®. Seattle, Washington: University of Washington; 2014. [PubMed] [Google Scholar]
- Oshima J, Sidorova JM, Monnat RJ., Jr Werner syndrome: clinical features, pathogenesis and potential therapeutic interventions. Ageing Res Rev. 2016 doi: 10.1016/j.arr.2016.03.002. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perry JJ, Yannone SM, Holden LG, Hitomi C, Asaithamby A, Han S, Cooper PK, Chen DJ, Tainer JA. WRN exonuclease structure and molecular mechanism imply an editing role in DNA end processing. Nat Struct Mol Biol. 2006;13:414–422. doi: 10.1038/nsmb1088. [DOI] [PubMed] [Google Scholar]
- Pinto GR, Yoshioka FK, Clara CA, Santos MJ, Almeida JR, Burbano RR, Rey JA, Casartelli C. WRN Cys1367Arg SNP is not associated with risk and prognosis of gliomas in Southeast Brazil. J Neurooncol. 2008;90:253–258. doi: 10.1007/s11060-008-9664-8. [DOI] [PubMed] [Google Scholar]
- Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, Grody WW, Hegde M, Lyon E, Spector E, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17:405–424. doi: 10.1038/gim.2015.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodriguez-Lopez AM, Jackson DA, Nehlin JO, Iborra F, Warren AV, Cox LS. Characterisation of the interaction between WRN, the helicase/exonuclease defective in progeroid Werner’s syndrome, and an essential replication factor, PCNA. Mech Ageing Dev. 2003;124:167–174. doi: 10.1016/s0047-6374(02)00131-8. [DOI] [PubMed] [Google Scholar]
- Rudolph KL, Chang S, Lee HW, Blasco M, Gottlieb GJ, Greider C, DePinho RA. Longevity, stress response, and cancer in aging telomerase-deficient mice. Cell. 1999;96:701–712. doi: 10.1016/s0092-8674(00)80580-2. [DOI] [PubMed] [Google Scholar]
- Saha B, Cypro A, Martin GM, Oshima J. Rapamycin decreases DNA damage accumulation and enhances cell growth of WRN-deficient human fibroblasts. Aging Cell. 2014;13:573–575. doi: 10.1111/acel.12190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saha B, Lessel D, Hisama FM, Leistritz DF, Friedrich K, Martin GM, Kubisch C, Oshima J. A novel LMNA mutation causes altered nuclear morphology and symptoms of familial partial lipodystrophy (dunnigan variety) with progeroid features. Mol Syndromol. 2010;1:127–132. doi: 10.1159/000320166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saha B, Lessel D, Nampoothiri S, Rao AS, Hisama FM, Peter D, Bennett C, Nurnberg G, Nurnberg P, Martin GM, et al. Ethnic-specific WRN mutations in South Asian Werner syndrome patients: potential founder effect in patients with Indian or Pakistani ancestry. Mol Genet Genomic Med. 2013;1:7–14. doi: 10.1002/mgg3.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salk D, Au K, Hoehn H, Martin GM. Cytogenetics of Werner’s syndrome cultured skin fibroblasts: variegated translocation mosaicism. Cytogenet Cell Genet. 1981;30:92–107. doi: 10.1159/000131596. [DOI] [PubMed] [Google Scholar]
- Satoh M, Imai M, Sugimoto M, Goto M, Furuichi Y. Prevalence of Werner’s syndrome heterozygotes in Japan. Lancet. 1999;353:1766. doi: 10.1016/S0140-6736(98)05869-3. [DOI] [PubMed] [Google Scholar]
- Sebastiani P, Bae H, Sun FX, Andersen SL, Daw EW, Malovini A, Kojima T, Hirose N, Schupf N, Puca A, et al. Meta-analysis of genetic variants associated with human exceptional longevity. Aging (Albany NY) 2013;5:653–661. doi: 10.18632/aging.100594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sebastiani P, Solovieff N, Dewan AT, Walsh KM, Puca A, Hartley SW, Melista E, Andersen S, Dworkis DA, Wilk JB, et al. Genetic signatures of exceptional longevity in humans. PLoS One. 2012;7:e29848. doi: 10.1371/journal.pone.0029848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen M, Zheng T, Lan Q, Zhang Y, Zahm SH, Wang SS, Holford TR, Leaderer B, Yeager M, Welch R, et al. Polymorphisms in DNA repair genes and risk of non-Hodgkin lymphoma among women in Connecticut. Hum Genet. 2006;119:659–668. doi: 10.1007/s00439-006-0177-2. [DOI] [PubMed] [Google Scholar]
- Shimamoto A, Kagawa H, Zensho K, Sera Y, Kazuki Y, Osaki M, Oshimura M, Ishigaki Y, Hamasaki K, Kodama Y, et al. Reprogramming suppresses premature senescence phenotypes of Werner syndrome cells and maintains chromosomal stability over long-term culture. PLoS One. 2014;9:e112900. doi: 10.1371/journal.pone.0112900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimamoto A, Yokote K, Tahara H. Werner Syndrome-specific induced pluripotent stem cells: recovery of telomere function by reprogramming. Front Genet. 2015;6:10. doi: 10.3389/fgene.2015.00010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki T, Shiratori M, Furuichi Y, Matsumoto T. Diverged nuclear localization of Werner helicase in human and mouse cells. Oncogene. 2001;20:2551–2558. doi: 10.1038/sj.onc.1204344. [DOI] [PubMed] [Google Scholar]
- Tadokoro T, Kulikowicz T, Dawut L, Croteau DL, Bohr VA. DNA binding residues in the RQC domain of Werner protein are critical for its catalytic activities. Aging (Albany NY) 2012;4:417–429. doi: 10.18632/aging.100463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tadokoro T, Rybanska-Spaeder I, Kulikowicz T, Dawut L, Oshima J, Croteau DL, Bohr VA. Functional deficit associated with a missense Werner syndrome mutation. DNA Repair (Amst) 2013;12:414–421. doi: 10.1016/j.dnarep.2013.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takemoto M, Mori S, Kuzuya M, Yoshimoto S, Shimamoto A, Igarashi M, Tanaka Y, Miki T, Yokote K. Diagnostic criteria for Werner syndrome based on Japanese nationwide epidemiological survey. Geriatr Gerontol Int. 2013;13:475–481. doi: 10.1111/j.1447-0594.2012.00913.x. [DOI] [PubMed] [Google Scholar]
- Takemoto M, Yamaga M, Furuichi Y, Yokote K. Astaxanthin improves nonalcoholic fatty liver disease in Werner syndrome with diabetes mellitus. J Am Geriatr Soc. 2015;63:1271–1273. doi: 10.1111/jgs.13505. [DOI] [PubMed] [Google Scholar]
- Talaei F, van Praag VM, Henning RH. Hydrogen sulfide restores a normal morphological phenotype in Werner syndrome fibroblasts, attenuates oxidative damage and modulates mTOR pathway. Pharmacol Res. 2013;74:34–44. doi: 10.1016/j.phrs.2013.04.011. [DOI] [PubMed] [Google Scholar]
- Tivey HS, Brook AJ, Rokicki MJ, Kipling D, Davis T. p38 (MAPK) stress signalling in replicative senescence in fibroblasts from progeroid and genomic instability syndromes. Biogerontology. 2013;14:47–62. doi: 10.1007/s10522-012-9407-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uhrhammer NA, Lafarge L, Dos Santos L, Domaszewska A, Lange M, Yang Y, Aractingi S, Bessis D, Bignon YJ. Werner syndrome and mutations of the WRN and LMNA genes in France. Hum Mutat. 2006;27:718–719. doi: 10.1002/humu.9435. [DOI] [PubMed] [Google Scholar]
- United N. World Population Ageing: 1950–2050. Population Division, Department of Economic and Social Affairs, United Nations; 2002. Available at http://www.un.org/esa/population/publications/worldageing19502050/ [Google Scholar]
- von Kobbe C, Bohr VA. A nucleolar targeting sequence in the Werner syndrome protein resides within residues 949–1092. J Cell Sci. 2002;115(Pt 20):3901–3907. doi: 10.1242/jcs.00076. [DOI] [PubMed] [Google Scholar]
- Wang H, Chen Q, Lee SH, Choi Y, Johnson FB, Pignolo RJ. Impairment of osteoblast differentiation due to proliferation-independent telomere dysfunction in mouse models of accelerated aging. Aging Cell. 2012;11:704–713. doi: 10.1111/j.1474-9726.2012.00838.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang L, Ogburn CE, Ware CB, Ladiges WC, Youssoufian H, Martin GM, Oshima J. Cellular Werner phenotypes in mice expressing a putative dominant-negative human WRN gene. Genetics. 2000;154:357–362. doi: 10.1093/genetics/154.1.357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu Y, Li N, Xiang R, Sun P. Emerging roles of the p38 MAPK and PI3K/AKT/mTOR pathways in oncogene-induced senescence. Trends Biochem Sci. 2014;39:268–276. doi: 10.1016/j.tibs.2014.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamabe Y, Sugimoto M, Satoh M, Suzuki N, Sugawara M, Goto M, Furuichi Y. Down-regulation of the defective transcripts of the Werner’s syndrome gene in the cells of patients. Biochem Biophys Res Commun. 1997;236:151–154. doi: 10.1006/bbrc.1997.6919. [DOI] [PubMed] [Google Scholar]
- Yang L, Wang G, Zhao X, Ye S, Shen P, Wang W, Zheng S. A novel WRN frameshift mutation identified by multiplex genetic testing in a family with multiple cases of cancer. PLoS One. 2015;10:e0133020. doi: 10.1371/journal.pone.0133020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yokote K, Honjo S, Kobayashi K, Fujimoto M, Kawamura H, Mori S, Saito Y. Metabolic improvement and abdominal fat redistribution in Werner syndrome by pioglitazone. J Am Geriatr Soc. 2004;52:1582–1583. doi: 10.1111/j.1532-5415.2004.52430_4.x. [DOI] [PubMed] [Google Scholar]
- Yu CE, Oshima J, Fu YH, Wijsman EM, Hisama F, Alisch R, Matthews S, Nakura J, Miki T, Ouais S, et al. Positional cloning of the Werner’s syndrome gene. Science. 1996;272:258–262. doi: 10.1126/science.272.5259.258. [DOI] [PubMed] [Google Scholar]
- Zhang W, Li J, Suzuki K, Qu J, Wang P, Zhou J, Liu X, Ren R, Xu X, Ocampo A, et al. Aging stem cells. A Werner syndrome stem cell model unveils heterochromatin alterations as a driver of human aging. Science. 2015;348:1160–1163. doi: 10.1126/science.aaa1356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao N, Hao F, Qu T, Zuo YG, Wang BX. A novel mutation of the WRN gene in a Chinese patient with Werner syndrome. Clin Exp Dermatol. 2008;33:278–281. doi: 10.1111/j.1365-2230.2007.02641.x. [DOI] [PubMed] [Google Scholar]
- Zins K, Frech B, Taubenschuss E, Schneeberger C, Abraham D, Schreiber M. Association of the rs1346044 polymorphism of the Werner syndrome gene RECQL2 with increased risk and premature onset of breast cancer. Int J Mol Sci. 2015;16:29643–29653. doi: 10.3390/ijms161226192. [DOI] [PMC free article] [PubMed] [Google Scholar]