Abstract
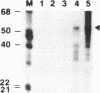
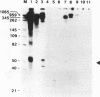
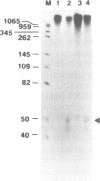
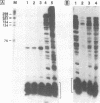
An in vitro RNA synthesis system mimicking replication of genomic influenza virus RNA was developed with nuclear extracts prepared from influenza virus-infected HeLa cells using exogenously added RNA templates. The RNA synthesizing activity was divided into two complementing fractions, i.e. the ribonucleoprotein (RNP) complexes and the fraction free of RNP, which could be replaced with RNP cores isolated from virions and nuclear extracts from uninfected cells, respectively. When nuclear extracts from uninfected cells were fractionated by phosphocellulose column chromatography, the stimulatory activity for RNA synthesis was further separated into two distinct fractions. One of them, tentatively designated RAF (RNA polymerase activating factor), stimulated RNA synthesis with either RNP cores or RNA polymerase and nucleocapsid protein purified from RNP cores as the enzyme source. In contrast, the other, designated PRF (polymerase regulating factor), functioned as an activator only when RNP cores were used as the enzyme source. Biochemical analyses revealed that PRF facilitates dissociation of RNA polymerase from RNP cores. Of interest is that virus-coded non-structural protein 1 (NS1), which has been thought to be involved in regulation of replication, counteracted PRF function. Roles of cellular factors and viral proteins, NS1 in particular, are discussed in terms of regulation of influenza virus RNA genome replication.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barry R. D., Mahy B. W. The influenza virus genome and its replication. Br Med Bull. 1979 Jan;35(1):39–46. doi: 10.1093/oxfordjournals.bmb.a071540. [DOI] [PubMed] [Google Scholar]
- Beaton A. R., Krug R. M. Synthesis of the templates for influenza virion RNA replication in vitro. Proc Natl Acad Sci U S A. 1984 Aug;81(15):4682–4686. doi: 10.1073/pnas.81.15.4682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beaton A. R., Krug R. M. Transcription antitermination during influenza viral template RNA synthesis requires the nucleocapsid protein and the absence of a 5' capped end. Proc Natl Acad Sci U S A. 1986 Sep;83(17):6282–6286. doi: 10.1073/pnas.83.17.6282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braam J., Ulmanen I., Krug R. M. Molecular model of a eucaryotic transcription complex: functions and movements of influenza P proteins during capped RNA-primed transcription. Cell. 1983 Sep;34(2):609–618. doi: 10.1016/0092-8674(83)90393-8. [DOI] [PubMed] [Google Scholar]
- De B. P., Lesoon A., Banerjee A. K. Human parainfluenza virus type 3 transcription in vitro: role of cellular actin in mRNA synthesis. J Virol. 1991 Jun;65(6):3268–3275. doi: 10.1128/jvi.65.6.3268-3275.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dignam J. D., Lebovitz R. M., Roeder R. G. Accurate transcription initiation by RNA polymerase II in a soluble extract from isolated mammalian nuclei. Nucleic Acids Res. 1983 Mar 11;11(5):1475–1489. doi: 10.1093/nar/11.5.1475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fields S., Song O. A novel genetic system to detect protein-protein interactions. Nature. 1989 Jul 20;340(6230):245–246. doi: 10.1038/340245a0. [DOI] [PubMed] [Google Scholar]
- Fodor E., Pritlove D. C., Brownlee G. G. The influenza virus panhandle is involved in the initiation of transcription. J Virol. 1994 Jun;68(6):4092–4096. doi: 10.1128/jvi.68.6.4092-4096.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Francklyn C., Schimmel P. Aminoacylation of RNA minihelices with alanine. Nature. 1989 Feb 2;337(6206):478–481. doi: 10.1038/337478a0. [DOI] [PubMed] [Google Scholar]
- Honda A., Mukaigawa J., Yokoiyama A., Kato A., Ueda S., Nagata K., Krystal M., Nayak D. P., Ishihama A. Purification and molecular structure of RNA polymerase from influenza virus A/PR8. J Biochem. 1990 Apr;107(4):624–628. doi: 10.1093/oxfordjournals.jbchem.a123097. [DOI] [PubMed] [Google Scholar]
- Honda A., Uéda K., Nagata K., Ishihama A. Identification of the RNA polymerase-binding site on genome RNA of influenza virus. J Biochem. 1987 Nov;102(5):1241–1249. doi: 10.1093/oxfordjournals.jbchem.a122163. [DOI] [PubMed] [Google Scholar]
- Honda A., Uéda K., Nagata K., Ishihama A. RNA polymerase of influenza virus: role of NP in RNA chain elongation. J Biochem. 1988 Dec;104(6):1021–1026. doi: 10.1093/oxfordjournals.jbchem.a122569. [DOI] [PubMed] [Google Scholar]
- Hsu M. T., Parvin J. D., Gupta S., Krystal M., Palese P. Genomic RNAs of influenza viruses are held in a circular conformation in virions and in infected cells by a terminal panhandle. Proc Natl Acad Sci U S A. 1987 Nov;84(22):8140–8144. doi: 10.1073/pnas.84.22.8140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang T. S., Palese P., Krystal M. Determination of influenza virus proteins required for genome replication. J Virol. 1990 Nov;64(11):5669–5673. doi: 10.1128/jvi.64.11.5669-5673.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang Y. T., Romito R. R., De B. P., Banerjee A. K. Characterization of the in vitro system for the synthesis of mRNA from human respiratory syncytial virus. Virology. 1993 Apr;193(2):862–867. doi: 10.1006/viro.1993.1195. [DOI] [PubMed] [Google Scholar]
- Ishihama A., Nagata K. Viral RNA polymerases. CRC Crit Rev Biochem. 1988;23(1):27–76. doi: 10.3109/10409238809103119. [DOI] [PubMed] [Google Scholar]
- Kimura N., Fukushima A., Oda K., Nakada S. An in vivo study of the replication origin in the influenza virus complementary RNA. J Biochem. 1993 Jan;113(1):88–92. doi: 10.1093/oxfordjournals.jbchem.a124009. [DOI] [PubMed] [Google Scholar]
- Kimura N., Nishida M., Nagata K., Ishihama A., Oda K., Nakada S. Transcription of a recombinant influenza virus RNA in cells that can express the influenza virus RNA polymerase and nucleoprotein genes. J Gen Virol. 1992 Jun;73(Pt 6):1321–1328. doi: 10.1099/0022-1317-73-6-1321. [DOI] [PubMed] [Google Scholar]
- Li J. J., Kelly T. J. Simian virus 40 DNA replication in vitro. Proc Natl Acad Sci U S A. 1984 Nov;81(22):6973–6977. doi: 10.1073/pnas.81.22.6973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X., Palese P. Mutational analysis of the promoter required for influenza virus virion RNA synthesis. J Virol. 1992 Jul;66(7):4331–4338. doi: 10.1128/jvi.66.7.4331-4338.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo G. X., Luytjes W., Enami M., Palese P. The polyadenylation signal of influenza virus RNA involves a stretch of uridines followed by the RNA duplex of the panhandle structure. J Virol. 1991 Jun;65(6):2861–2867. doi: 10.1128/jvi.65.6.2861-2867.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luytjes W., Krystal M., Enami M., Parvin J. D., Palese P. Amplification, expression, and packaging of foreign gene by influenza virus. Cell. 1989 Dec 22;59(6):1107–1113. doi: 10.1016/0092-8674(89)90766-6. [DOI] [PubMed] [Google Scholar]
- López-Turiso J. A., Martínez C., Tanaka T., Ortín J. The synthesis of influenza virus negative-strand RNA takes place in insoluble complexes present in the nuclear matrix fraction. Virus Res. 1990 Jul;16(3):325–337. doi: 10.1016/0168-1702(90)90056-h. [DOI] [PubMed] [Google Scholar]
- Matsui T., Segall J., Weil P. A., Roeder R. G. Multiple factors required for accurate initiation of transcription by purified RNA polymerase II. J Biol Chem. 1980 Dec 25;255(24):11992–11996. [PubMed] [Google Scholar]
- Moyer S. A., Baker S. C., Lessard J. L. Tubulin: a factor necessary for the synthesis of both Sendai virus and vesicular stomatitis virus RNAs. Proc Natl Acad Sci U S A. 1986 Aug;83(15):5405–5409. doi: 10.1073/pnas.83.15.5405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagata K., Guggenheimer R. A., Enomoto T., Lichy J. H., Hurwitz J. Adenovirus DNA replication in vitro: identification of a host factor that stimulates synthesis of the preterminal protein-dCMP complex. Proc Natl Acad Sci U S A. 1982 Nov;79(21):6438–6442. doi: 10.1073/pnas.79.21.6438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagata K., Takeuchi K., Ishihama A. In vitro synthesis of influenza viral RNA: biochemical complementation assay of factors required for influenza virus replication. J Biochem. 1989 Aug;106(2):205–208. doi: 10.1093/oxfordjournals.jbchem.a122833. [DOI] [PubMed] [Google Scholar]
- Parvin J. D., Palese P., Honda A., Ishihama A., Krystal M. Promoter analysis of influenza virus RNA polymerase. J Virol. 1989 Dec;63(12):5142–5152. doi: 10.1128/jvi.63.12.5142-5152.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piccone M. E., Fernandez-Sesma A., Palese P. Mutational analysis of the influenza virus vRNA promoter. Virus Res. 1993 May;28(2):99–112. doi: 10.1016/0168-1702(93)90129-b. [DOI] [PubMed] [Google Scholar]
- Seong B. L., Brownlee G. G. A new method for reconstituting influenza polymerase and RNA in vitro: a study of the promoter elements for cRNA and vRNA synthesis in vitro and viral rescue in vivo. Virology. 1992 Jan;186(1):247–260. doi: 10.1016/0042-6822(92)90079-5. [DOI] [PubMed] [Google Scholar]
- Shapiro G. I., Krug R. M. Influenza virus RNA replication in vitro: synthesis of viral template RNAs and virion RNAs in the absence of an added primer. J Virol. 1988 Jul;62(7):2285–2290. doi: 10.1128/jvi.62.7.2285-2290.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skorko R., Summers D. F., Galarza J. M. Influenza A virus in vitro transcription: roles of NS1 and NP proteins in regulating RNA synthesis. Virology. 1991 Feb;180(2):668–677. doi: 10.1016/0042-6822(91)90080-u. [DOI] [PubMed] [Google Scholar]
- Szewczyk B., Laver W. G., Summers D. F. Purification, thioredoxin renaturation, and reconstituted activity of the three subunits of the influenza A virus RNA polymerase. Proc Natl Acad Sci U S A. 1988 Nov;85(21):7907–7911. doi: 10.1073/pnas.85.21.7907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takeuchi K., Nagata K., Ishihama A. In vitro synthesis of influenza viral RNA: characterization of an isolated nuclear system that supports transcription of influenza viral RNA. J Biochem. 1987 Apr;101(4):837–845. doi: 10.1093/oxfordjournals.jbchem.a121950. [DOI] [PubMed] [Google Scholar]
- Ulmanen I., Broni B. A., Krug R. M. Role of two of the influenza virus core P proteins in recognizing cap 1 structures (m7GpppNm) on RNAs and in initiating viral RNA transcription. Proc Natl Acad Sci U S A. 1981 Dec;78(12):7355–7359. doi: 10.1073/pnas.78.12.7355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamanaka K., Ishihama A., Nagata K. Reconstitution of influenza virus RNA-nucleoprotein complexes structurally resembling native viral ribonucleoprotein cores. J Biol Chem. 1990 Jul 5;265(19):11151–11155. [PubMed] [Google Scholar]
- Yamanaka K., Ogasawara N., Ueda M., Yoshikawa H., Ishihama A., Nagata K. Characterization of a temperature-sensitive mutant in the RNA polymerase PB2 subunit gene of influenza A/WSN/33 virus. Arch Virol. 1990;114(1-2):65–73. doi: 10.1007/BF01311012. [DOI] [PubMed] [Google Scholar]
- Yamanaka K., Ogasawara N., Yoshikawa H., Ishihama A., Nagata K. In vivo analysis of the promoter structure of the influenza virus RNA genome using a transfection system with an engineered RNA. Proc Natl Acad Sci U S A. 1991 Jun 15;88(12):5369–5373. doi: 10.1073/pnas.88.12.5369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young J. F., Desselberger U., Palese P., Ferguson B., Shatzman A. R., Rosenberg M. Efficient expression of influenza virus NS1 nonstructural proteins in Escherichia coli. Proc Natl Acad Sci U S A. 1983 Oct;80(19):6105–6109. doi: 10.1073/pnas.80.19.6105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de la Luna S., Martín J., Portela A., Ortín J. Influenza virus naked RNA can be expressed upon transfection into cells co-expressing the three subunits of the polymerase and the nucleoprotein from simian virus 40 recombinant viruses. J Gen Virol. 1993 Mar;74(Pt 3):535–539. doi: 10.1099/0022-1317-74-3-535. [DOI] [PubMed] [Google Scholar]
- del Río L., Martínez C., Domingo E., Ortín J. In vitro synthesis of full-length influenza virus complementary RNA. EMBO J. 1985 Jan;4(1):243–247. doi: 10.1002/j.1460-2075.1985.tb02342.x. [DOI] [PMC free article] [PubMed] [Google Scholar]