Abstract
Glioblastoma multiforme (GBM), or grade IV astrocytoma, is the most common type of primary brain tumor. It has a devastating prognosis with a 2-year-overall survival rate of only 26 % after standard treatment, which includes surgery, radiation, and adjuvant chemotherapy with temozolomide. Also lower grade gliomas are difficult to treat, because they diffusely spread into the brain, where extensive removal of tissue is critical. Better understanding of the cancer’s biology is a key for the development of more effective therapy approaches. The discovery of isocitrate dehydrogenase (IDH) mutations in leukemia and glioma drew attention to specific metabolic aberrations in IDH-mutant gliomas. In the center of the metabolic alterations is α-ketoglutarate (αKG), an intermediate metabolite in the tricarboxylic acid (TCA) cycle, and the associated amino acid glutamate (Glu). This article highlights the role of these metabolites in glioma energy and lipid production and indicates possible weak spots of IDH-mutant and IDH-wt gliomas.
Keywords: Glioma, Glutamate, Alpha-ketoglutarate, Isocitrate dehydrogenase, Branched chain amino acids
Introduction
Glutamic acid is an amino acid that plays an important role in energy metabolism and as excitatory neurotransmitter in the central nervous system. In the context of molecular biology the name of its carboxylate anion glutamate (Glu) is used synonymously, because in the body glutamic acid appears in its dissociated form. A link between Glu and energy metabolism was already described in the 1920s (Thunberg 1920). In 1936—Malherbe showed that the reversible oxidation of Glu to α-ketoglutarate (αKG) could be found in brain tissue. He further hypothesized that in vivo the enzyme “glutamic acid deaminase”, today referred to as glutamate dehydrogenase (GDH), executes the synthesis of Glu more often than its degradation. One part of this article will elucidate under which circumstances Weil-Malherbe’s assumption is true.
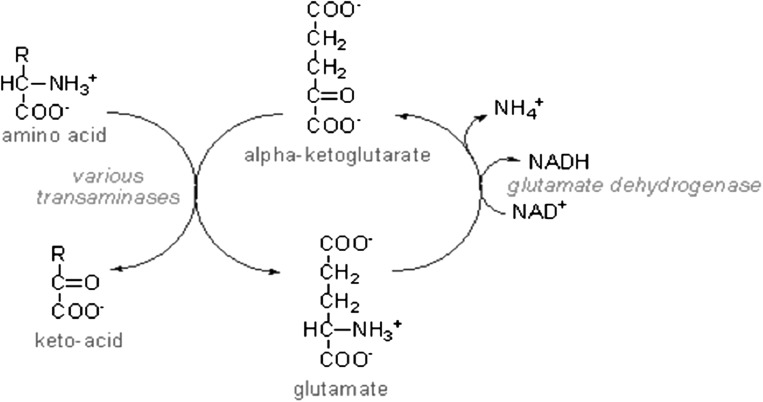
αKG is an intermediate metabolite in the tricarboxylic acid (TCA) cycle (or citric acid cycle, or Krebs cycle). The TCA cycle is an evolutionary conserved pathway. In eukaryotic cells it takes place in the mitochondria and serves mainly the production of ATP by breakdown of macromolecules. The reactions yield the reducing agents NADH and NADPH, which are used in the electron transport (oxidative phosphorylation) pathway for ATP production. In addition the TCA cycle is crucial for providing substrates for lipid and fatty acid synthesis. The transamination of αKG produces Glu and, dependent on the amino acid, a keto-acid (Fig. 1). Amino acids like alanine or aspartate are converted to pyruvate or oxaloacetate, respectively. These products are important metabolites in pathways like the TCA cycle or glycolysis. It became extremely relevant to the field of cancer metabolism when isocitrate dehydrogenase 1/2 (IDH1/2) mutations in glioma and acute myeloid leukemia were discovered (Dang et al. 2010; Reitman and Yan 2010). Mutated IDH enzymes have a shifted functionality, with which αKG becomes the main substrate for 2-hydroxyglutarate (2HG) synthesis.
Fig. 1.
Glutamate dehydrogenase mediates the NADH-producing conversion of glutamate to α-ketoglutarate. From https://www.david-bender.co.uk (assessed 26-02-2016)
Aside from oxidative deamination, Glu is often synthesized from glutamine (Gln). Due to its role as neurotransmitter, Glu is constantly released from neurons. Its synthesis and degradation are part of the glutamate/glutamine cycle. After release Glu is taken up by astrocytes, where it is converted to Gln and released again (Bak et al. 2006; Hertz 1979; Hertz et al. 1999). In the cytoplasm Gln can be converted to Glu by the enzyme glutaminase (GLA).
Gliomas account for 50 % of intracranial tumors and are the most common primary form of cancer in the brain. Glioblastoma multiforme (GBM; WHO grade IV astrocytoma) accounts for most of them. 12 % of GBMs carry a mutation in the IDH1 gene, which encodes for the metabolic enzyme isocitrate dehydrogenase (Parsons et al. 2008). The discovery of IDH mutations in gliomas led the focus on cancer metabolism. A better understanding of tumor genesis could increase the hope of finding effective treatments against it. Better treatment strategies are urgently needed, because the prognosis for gliomas, especially for GBM is abject. Standard treatment for GBM includes resection via surgery, followed by radiation and adjuvant chemotherapy with the alkylating agent temozolomide. Although administration of temozolomide improves overall survival significantly, median survival ranges in between 12 and 15 months and the 2-year survival rate averages 26 % (Stupp et al. 2005).
Glutamate and α-ketoglutarate in glioma metabolism
The Warburg effect
Otto Warburg described a metabolic switch in cancer cells concerning the use of glucose (Warburg et al. 1926; Warburg 1925, 1954). In recent times the shift from pyruvate oxidation to lactic acid fermentation is known as the “Warburg-effect” and is considered a hallmark of cancer (Hanahan and Weinberg 2011). The metabolic change to lactic acid fermentation is baffling at first, because it leads to a smaller net ATP production than pyruvate oxidation and therefore seems like a reduction of the cell’s energy production. Today it is widely believed that the Warburg effect enables the cell to have building blocks like amino acids and nucleosides readily available. They are crucial for the synthesis of macromolecules and organelles, which enable the cancer cell to meet the special needs regarding fast growth and proliferation. The switch is accompanied by an increased glucose influx, possibly to make up for the less efficient ATP production.
Glutaminolysis
Glu plays an important role as intermediate metabolite of glutaminolysis. Glutaminolysis describes the sequence of enzymatic reactions that turn glutamine (Gln) into substrates that enter the TCA cycle. The first step of this sequence is the hydrolysis of the amino group of Gln, which turns it into Glu. The reaction is mediated by glutaminase and can be displayed as: Glutamine + H2O → Glutamate + NH3. In cancer, glycolysis and glutaminolysis are the major mechanisms of ATP production, which means glucose and Gln are crucial nutrients. HIF1α activity can disrupt the function of the pyruvate dehydrogenase (PDH) complex (Kim et al. 2006). This will inhibit the introduction of glucose-derived metabolites into the TCA cycle and therefore renders the cell dependent on glutaminolysis. Glutaminolysis alone can rescue the cell from cell death. It is a key enzymic pathway for cancer metabolism, because it provides nitrogen for nucleotide and amino acid synthesis, it offers an alternative carbon source to supply TCA cycle intermediates, and as a byproduct NADPH is formed for lipid and nucleotide synthesis (reviewed by DeBerardinis and Cheng 2010).
Reductive carboxylation
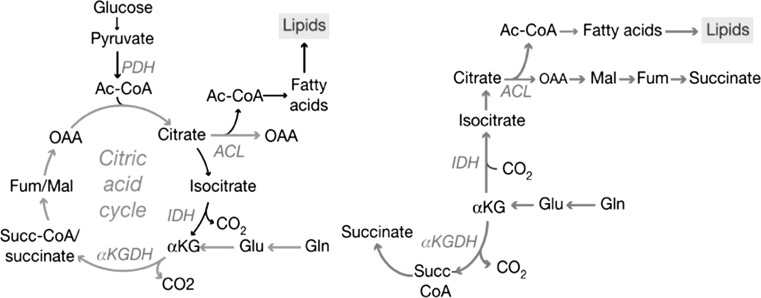
Besides glutaminolysis, Gln can be a substrate for another distinct cytosolic pathway, which includes reversed flux through the TCA cycle. Where glutaminolysis mainly serves the production of energy in the form of ATP, reversed flux through the TCA cycle favors the synthesis of acetyl-CoA (Filipp et al. 2012). In regular cell metabolism glucose-derived pyruvate will enter the TCA cycle and then serves as the main source of acetyl-CoA, a precursor of fatty acids and lipids (Fig. 2). Under conditional aerobic glycolysis or under hypoxic conditions cells convert glucose to lactate. This will reduce the flux of pyruvate into the TCA cycle. Acetyl-CoA is then produced by breakdown of the TCA cycle-metabolite citrate. Hence, a lack of acetyl-CoA could also stem from disabled citrate formation by defective mitochondria, such as disruptions in the TCA cycle or electron transport chain (Mullen et al. 2012). In hypoxia HIF1α activity interferes with glucose carbon use in citrate synthesis by hampering PDH. A study performed with a GBM cell line in hypoxia (0.5 % O2) came to the conclusion that Gln is the major source for carbon under hypoxic conditions (Wise et al. 2011). Additionally, it was shown that reductive carboxylation of αKG (from Gln) is IDH2-dependent (Fig. 2). IDH2 is known to catalyze the oxidative decarboxylation of isocitrate to αKG. Studies indicate that it is also crucial for the reverse reaction (Wise et al. 2011; Mullen et al. 2012). Although the studies showed that IDH2 is crucial for sustained reductive carboxylation under hypoxia, they failed to assess the importance of IDH1 in that matter.
Fig. 2.
Intact TCA cycle (green). When PDH is blocked, or the TCA cycle is disrupted for other reasons, IDH-dependent reductive carboxylation sustains the formation of intermediate metabolites and fatty acids (red). From Mullen et al. (2012) with permission
In contrast to these results it was shown that there is a TCA cycle-independent cytoplasmic pathway of reductive carboxylation of αKG, which is mediated by IDH1 (Metallo et al. 2012). This pathway, too results in the formation of isocitrate, citrate, and finally in acetyl-CoA for lipid synthesis.
Another positive effect of reductive carboxylation for the cancer cells is that it decreases ROS levels and produces NADPH as a byproduct.
Glutaminase isoenzymes can exert contrary functions
As mentioned above, the first step of Gln degradation is catalyzed by glutaminase (GLA). The paralogous GLA isoenzymes are encoded by the Gls and the Gls2 genes (reviewed by Campos-Sandoval et al. 2015). These genes are linked to tumor behavior, because oncogenes and tumor suppressor genes regulate them. Interestingly, differential expression of GLA isoenzymes alters the metabolism of nutrients. In brain tumor settings, however, the exact role of GLA isoenzymes is unclear, because these isoenzymes can exert contrary functions. For example, silencing the Gls gene (GLS) in glioblastoma cells LN229 and SFxL inhibited growth, lowered survival ratios, and induced apoptosis. The growth inhibition was even stronger under oxidative stress. Similar observations were made when the liver-type Gls2 gene was overexpressed in T98G glioma cells (reviewed by Campos-Sandoval et al. 2015).
IDH wt glioma cells release Glu
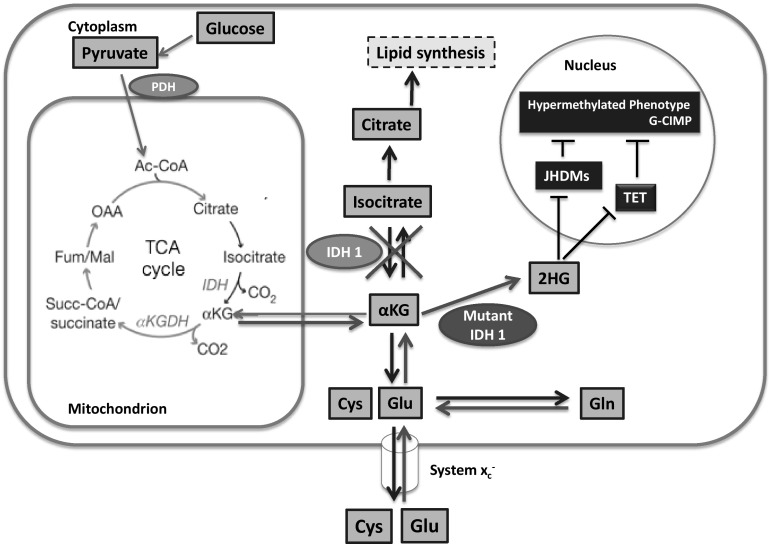
In IDH wild-type (wt) gliomas, Glu synthesis is catalyzed by high levels of branched-chain amino acid transaminase 1 (BCAT1), which convert αKG into Glu (Tönjes et al. 2013). The byproduct of this reaction is ammonia. When intracellular Glu levels rise, excessive Glu is released via the glutamine/cysteine antiporter System x −c (extensively reviewed by Lewerenz et al. 2013) in exchange for cysteine (Cys; Fig. 3). This exchange is favorable for the cancer cells, because Cys is a major component of the antioxidant Glutathione (GSH), which in turn is an antagonist of reactive oxygen species (ROS). Elevated ROS levels trigger apoptosis; hence to antagonize ROS is important for cancer cell survival. There are external and internal causes for elevated ROS levels. The main external cause in GBM, radiotherapy, directly induces ROS through radiation. Internal causes of ROS lie in the altered metabolism itself. In cancer cells large amounts of glucose are oxidized in the TCA cycle, rendering the cell with superoxide anions as side products. These superoxide anions contribute to high ROS levels (Masui et al. 2014). High levels of GSH enable cancer cells to evade the induction of apoptosis through induced ROS.
Fig. 3.
Overview of biochemical reactions for energy and lipid production in the cell. α-ketoglutarate (αKG) and glutamate (Glu) are in the center of pathways like glutaminolysis or reductive carboxylation. Mutant IDH1 changes the dynamics of metabolic processes in the cell and leads to accumulation of 2HG. 2HG in turn inhibits JHDMs and TET, which leads to a hypermethylated phenotype. Ac-CoA, acetyl-CoA; OAA, oxaloacetate; Gln, glutamine; Glu, glutamate; Cys, cysteine; αKG, α-ketoglutarate; Succ-CoA, succinyl-CoA; Fum, fumarate; Mal, malate; PDH, pyruvate dehydrogenase; ACL, ATP-citrate lyase; IDH, isocitrate dehydrogenase; αKGDH, α-ketoglutarate dehydrogenase; JHDMs, Jumonji domain-containing histone demethylases; TET, Ten-Eleven-Translocation
Methionine–Cysteine double deprivation increases ROS levels in glioma cells
Recently it was shown that deprivation of amino acids like methionine (Met) or Cys leads to induction of ROS, too. Met or Cys deprivation alone already resulted in decreased proliferation of cells of the glioma cell lines U87 and U251 (Liu et al. 2015). Double deprivation had a synergistic effect.
When extracellular nutrient supply is scarce, autophagy enables the tumor cells to meet the demand for required amino acids and nucleotides. Also the induction of ROS can induce autophagy in cancer cells (reviewed by Bellot et al. 2013). Liu et al. (2015) studied the effects of double deprivation on autophagy. Met–Cys double deprivation led to an increased abundance of the autophagy-related protein LC3-II and autophagosomes. Furthermore, inhibition of autophagy increased the sensitivity of glioma cells to double deprivation.
In vivo studies showed that glioma proliferation was lower in tumor-bearing mice that received a Met–Cys deprived diet compared to a standard diet. In addition, histologic examination of tumors from Met–Cys deprived mice showed signs of autophagy (Liu et al. 2015).
The induction of ROS in cancer cells through Met–Cys double deprivation offers a potential additional therapy option that would enhance the ROS inducing effects of chemo- and radiotherapy.
High levels of extracellular Glu impair healthy cells in the tumor microenvironment
The exchange of Glu for Cys is favorable for the cancer cell, but not for healthy cells in its vicinity, because Glu causes cell death in the tumor environment (Takano et al. 2001; Ye and Sontheimer 1999). High levels of extracellular Glu lead to an imbalance in the glutamate/glutamine cycle between astrocytes and neurons. The results can be astrocyte swelling, a block of astrocytic Glu uptake and neuronal cell death (Bak et al. 2006; Jayakumar et al. 2006; Albrecht et al. 2006). The challenge for the astrocytes does not seem to be the origin of Glu, but the amount. Yao et al. (2014) showed that astrocytes were able to take up Glu that was released by glioma cells. In a co-culture of astrocytes and glioma cells with the ratio 1:1, Glu released by glioma cells was taken up by astrocytes and no neuronal damage was observed. In these cultures extracellular Glu levels were reduced to 1–5 μM (compared to +30 μM in single glioma culture). Glu uptake by astrocytes reduced glioma cell proliferation, and prevented neuronal death by Ca2+ overload. These conditions might resemble the tumor environment at an early stage of tumor development. In late stage gliomas, the astrocyte to glioma cell ratio is more likely to be around 0.5:1 or smaller. In vitro this ratio led to an increase of extracellular Glu and ammonia levels, which resulted in a block of Glu uptake by astrocytes and cell death (Yao et al. 2014).
Block of system x−c has positive effects on cells in the tumor vicinity
One approach to reduce the damaging effects of high extracellular Glu levels was to reduce the amount of secreted Glu by blocking the System x −c (Savaskan et al. 2008). System x −c is composed of xCT and CD98 in human primary gliomas and tested cell lines. In vitro Glu secretion was successfully reduced by downregulation of xCT via small interfering RNA (siRNA), and with the selective xCT inhibitor S-4-CPG (Savaskan et al. 2008). Reduction of xCT had no impact on morphology, cell cycle progression, ROS formation or proliferation Brain slices challenged with conditioned media from xCT-silenced cells showed significantly less cell death than the control with conditioned media from glioma cells. In vivo studies confirmed that inhibition of xCT through S-4-CPG reduced Glu secretion. That resulted in a lower neuronal damage, later onset of neurological deficits, and prolonged survival compared to the vehicle control group (Savaskan et al. 2008).
Intracellular peaks of acetyl-CoA levels correlate with phases of growth and proliferation in the yeast metabolic cycle. Furthermore, the gene battery in yeast that is differentially acetylated during high acetyl-CoA levels corresponds to target genes of c-Myc in mammalian cells (Ji et al. 2011). It has been concluded that acetyl-CoA levels directly influence epigenetic regulation through differential acetylation, which would describe an evolutionary conservative mechanism to link growth and proliferation to the nutritional state of the cell (Kaelin and McKnight 2013; Masui et al. 2014).
Metabolic compensations to anti-tumor therapy
Key cancer metabolites have been the target of anti-tumor therapies, but most single target approaches have failed, because cancer cells can compensate for disturbed pathways and lacking metabolites. Here, we give some examples of adaptions made by glioma cells to deletion or impairment of metabolic pathways.
Increased synthesis of asparagine halts apoptotic pathway in glutamine-depleted glioma cells
The non-essential amino acid Gln is, beside glucose, the most important energy source for glioma cells. Zhang et al. (2014) reported that Gln withdrawal led to apoptosis in SF188 human glioma cells with MYC amplification. That is somewhat surprising regarding the theoretical ability of the cell to synthesize Gln when sufficient amounts of glucose are available. Since Gln is crucial for asparagine (Asn) synthesis, it was tested if depletion of Asn has an effect on SF188 cells. Asn was depleted through knockdown of the enzyme asparagine synthetase (AS) that catalyzes the biosynthesis of Asn from aspartate and Gln. It has been reported that AS mRNA abundance is negatively correlated with glucose availability (Barbosa-Tessmann et al. 1999). AS depletion led to apoptosis in SF188 cells (Zhang et al. 2014). Addition of extracellular Asn completely restored survival and proliferation. These results are in line with the studies on the evasion of apoptosis through Asn in the case of sarcoma (Hettmer et al. 2015), and human melanoma and epidermoid carcinoma cells (Li et al. 2015). The authors conclude that Asn is crucial for cellular adaption to loss of Gln (Zhang et al. 2014). In Asn-deficient cells, translation of stress response RNAs like CHOP leads to apoptosis. Asn alone was sufficient to stop the apoptotic function of ATF4 through regulation of a pathway that induces translation-dependent apoptosis. Intracellular depletion of Asn alone resulted in apoptosis independent of Glu or glucose availability (Zhang et al. 2014).
Acute lymphoblastic leukemia (ALL) has been successfully treated through extracellular removal of Asn with l-asparaginase (Avramis 2012). Asparaginase (ASNase), an enzyme that hydrolyzes Asn, offers an option to deplete Asn intracellularly. Panosyan et al. (2014) showed that reduction of Asn through ASNase led to growth inhibition in DAOY medulloblastoma cells, GBM-ES cells, U87 cells, and mouse glioma (GL-261) cells in vitro. Gln addition increased Asn synthesis and abrogated the effect. Moreover, ASNase treatment affected the formation of neurospheres negatively.
Despite the promising in vitro results, ASNase-treated mice showed the same DAOY tumor growth as controls. Moreover, co-treatment with ASNase and temozolomide resulted in growth inhibition compared to temozolomide alone.
Regulators of amino acid metabolism
Crucial amino acids can be acquired in various ways. The example of Asn shows that they can derive from breakdown of metabolites like Glu, or they can be taken up from the extracellular matrix. A more common source of free amino acids in the cytosol is the degradation and breakdown of proteins and peptides. This recycling process occurs constantly in healthy and transformed cells and is orchestrated by proteasomes and aminopeptidases (Saric et al. 2004). Aminopetidases such as serine aminopeptidase dipeptidyl peptidase (Busek et al. 2012), leucine aminopeptidase 3 (He et al. 2015) and methionine aminopeptidase 2 (Dasgupta et al. 2005) play a role in glioma. However, no treatment that involves aminopeptidases has been proven to be effective in glioma. However, there is no effective aminopeptidase inhibitor registered, while those currently in clinical development have not yet been tested in glioma (Hitzerd et al. 2014).
There are several approaches to inhibit the protein degradation pathway in cancer, such as E3 ubiquitin ligase inhibitors (Snoek et al. 2013), proteasome inhibitors, and aminopeptidase inhibitors (reviewed by Hitzerd et al. 2014). Some of these have been tested in clinical trials, or are clinically approved like the proteasome inhibitor bortezomib. Insights that were obtained in these trials could help to develop a specific strategy for glioma treatment.
mTOR inhibitors have not been proven effective in glioma therapy
A central regulator of metabolism and cell growth is the kinase mechanistic target of rapamycin (or mammalian target of rapamycin; mTOR). mTOR is a downstream target of EGFR (epidermal growth factor receptor) through the PI3 K-Akt signaling pathway. 40 % of GBM have aberrant EGFR signaling; most carry the EGFRvIII mutant (Ekstrand et al. 1991; Wong et al. 1992). The mutant is characterized by a deletion of exons 2–7 of the EGFR gene, which results in an in-frame deletion of 267 amino acids from the extracellular domain of the receptor. EGFRvIII receptors are unable to bind growth factors, but are constitutively active in downstream signaling.
mTOR is found in two major complexes; mTOR complex 1 (mTORC1) and mTOR complex 2 (mTORC2). Both complexes promote increased c-Myc activity. mTORC1 splices the MYC-interacting protein MAX, which enhances c-Myc action (Babic et al. 2013). mTORC2 controls c-Myc levels in a FOXO-acetylation-dependent manner (Masui et al. 2013). Overexpressed Myc is a strong oncogene and has been explored in multiple types of cancer including glioma and GBM. It has been shown that c-Myc negatively regulates the tumor suppressor gene PTEN (Guo et al. 2013) and is involved in resistance to temozolomide therapy (Luo et al. 2015). Besides these effects it is strongly involved in metabolic reprogramming in glioma. It enhances GLS activity, which leads to increased glutaminolysis, higher Glu production and possibly increased reductive carboxylation.
Targeted therapies with mTOR kinase inhibitors have not proven to be effective in glioma therapy. To elucidate underlying mechanisms, magnetic resonance spectroscopy (MRS) of 12 GBM patients was carried out (Tanaka et al. 2015). The study indicated that Gln was strongly involved in metabolic reprogramming in GBM cells and the authors saw a connection between upregulation of Gln metabolism through increased GLS activity, and resistance to mTOR kinase inhibitors (Tanaka et al. 2015). The results indicated that mTOR-targeted therapy led to increased Gln metabolism and rendered glioma cells Gln-dependent, which could be an approach for effective therapy options. Combinations of mTOR inhibitors with other drugs affecting signaling pathways may have promise for further development.
Another way to inhibit mTOR signaling is the use of aminopeptidase inhibitors. Compounds such as tosedostat or bestatin inhibit aminopeptidase, which results in lowering the free amino acid levels. This in turn decreases mTOR signaling (reviewed by Hitzerd et al. 2014).
Differences between IDH-wt and IDH-mutant gliomas
Although IDH mutations often occur in gliomas, not all gliomas carry IDH mutations. The gain-of-function mutation shifts IDH activity. Its mutant IDH mediates the conversion of αKG into 2HG.
Isocitrate dehydrogenase (IDH)
One of the most exciting discoveries of the past years in the field of cancer metabolism was the one of IDH1/2 mutations in gliomas and acute myeloid leukemia (Dang et al. 2010; Reitman and Yan 2010). Only few mutations in genes that encode for metabolic enzymes are linked to tumor genesis. Examples for homozygous loss-of-function mutations in metabolic tumor suppressors are fumarate hydratase or one of the five subunits comprising the succinate dehydrogenase complex (King et al. 2006). In contrast to these examples, heterozygous IDH mutations lead to a gain-of-function. Therefore, IDH is not a tumor suppressor gene.
Due to the great number of recent reviews on this topic (Guo et al. 2011; van Lith et al. 2014; Molenaar et al. 2014; Bogdanovic 2015; Borodovsky et al. 2015; Parker and Metallo 2015), this report will be limited to a brief summary of main observations on IDH mutations and the consequence for cell metabolism in regard to Glu and αKG.
2-Hydroxyglutarate is an oncometabolite
IDH enzymes catalyze the NADP+/NAD+-dependent conversion of isocitrate to 2HG (also known as 2-oxoglutarate; Zhao et al. 2009). The cytoplasmic version of the enzyme, IDH1, acts in the cytoplasm; IDH2 and IDH3 act in mitochondria, mainly in the TCA cycle. 70–90 % of grades II and III glioma and secondary GBMs carry mutated IDH1 or IDH2 genes (Parsons et al. 2008; Yan et al. 2009). Mutations in the IDH3 gene have not been reported in connection with tumors; therefore, in this article the term IDH mutation refers only to IDH1 and IDH2. The most common amino acid substitution in glioma is the replacement of arginine with histidine in the IDH1 gene (IDH1R132H; Parsons et al. 2008; Yan et al. 2009; Borger and Zhu 2012; Hirata et al. 2015, Table 1). In the case of IDH2, the substrate-binding arginine (Arg) residues Arg 140 and Arg 172 are mutated. In both cases the mutation results in a gain-of-function of the enzyme, which leads to an increased conversion of αKG to 2HG and subsequently to an accumulation of 2HG in the cell (Dang et al. 2009; Gross et al. 2010; Ward et al. 2010). 2HG itself inhibits Ten-Eleven-Translocation (TET) family and Jumonji-C-domain-containing histone demethylases (JHDMs; Chowdhury et al. 2011; Xu et al. 2011; Fig. 3).
Table 1.
Differences between wild-type and mutated IDH
| Wild-type IDH | Mutant IDH | |
|---|---|---|
| Heterozygous point mutations in the catalytic site | None | Arg to His in position 132 (IDH1, R132H) Arg in position 172 and 140 (IDH2) |
| Substrate affinity | Same affinity for isocitrate and α-ketoglutarate | Lower affinity for isocitrate, higher affinity for α-ketoglutarate |
| IDH-mediated reaction | Isocitrate <−> α-ketoglutarate | α-Ketoglutarate—>2-hydroxyglutarate |
| 2-hydroxyglutarate levels | Low | High |
These enzymes are directly involved in demethylating processes in the genome. In that way high 2HG concentrations in the cell lead to global DNA hypermethylation and altered gene expression (Figueroa et al. 2010; Sasaki et al. 2012; Turcan et al. 2012). Unsurprisingly there is a tight correlation between IDH1 mutations and a hypermethylated phenotype named glioma-CpG island methylator phenotype (G-CIMP; Noushmehr et al. 2010). Since these alterations provide the basis for cancer, 2HG is described as ‘oncometabolite’. The exciting fact about IDH mutations is that it seems likely that this metabolic alteration is more of a major contributor to cancer initiation and progression, than a mere side effect. This makes it a potential target for therapy. Inhibitors of mutant IDH1 have been effective in lowering 2HG levels in vivo (Popovici-Muller et al. 2012) and reducing growth of glioma cells in vitro (Rohle et al. 2013). However, there is more to IDH-mutant cancer cells than just a shift in levels of two metabolites.
αKG and Glu in IDH-mutant cancer cells
αKG levels should be low in IDH-mutant cancer cells compared to IDH wt cells, due to increased conversion of αKG to 2HG. In fact they are relatively high, because mitochondrial biosynthesis makes up for missing cytoplasmic αKG (Van Lith et al. 2014). That in turn influences the mitochondrial TCA cycle considerably (Table 2).
Table 2.
Versatile effects of IDH mutations on cell metabolism
| Alterations through IDH mutation | Affected pathway | Negative effect on |
|---|---|---|
| αKG to 2HG-conversion | αKG–2HG | NADPH production |
| Increased 2HG levels | αKG-2HG | DNA methylation ATP synthesis |
| Increased need for cytoplasmic αKG | Glu-αKG | Glu export Cys import |
| Mitochondrial malate–αKG antiporter | αKG levels in mitochondria | |
| Reduced αKG availability | Glutaminolysis | ATP production |
| Distorted conversion of αKG to isocitrate | Reductive carboxylation | Lipid synthesis |
Mutant IDH results in deployed lipid synthesis
Increased synthesis of αKG through mitochondria means decreased levels of αKG substrates like citrate. Citrate, however, is crucial for acetly-CoA synthesis and therefore directly involved in the build-up of fatty acids. It is conceivable that the mutation of IDH leads to an altered ratio of fatty acid products like sphingolipids, or phospholipids. Although no data have been published on this matter, it has been claimed that IDH1 mutations indirectly alter the levels of sphingomyelin in mouse brains (Bogdanovic 2015).
As described earlier, IDH does not only assist the conversion of isocitrate to αKG, but also the reverse reaction during reductive carboxylation. In IDH-mutant tumors one would expect reductive carboxylation to be idle, due to the partial loss of IDH activity. Indeed, in hypoxic conditions heterozygous IDH1-mutant cells were impeded in their reductive carboxylation ability and increased their oxidative TCA metabolism (Table 3). IDH2 mutant cells, however, continued to perform reductive carboxylation (Grassian et al. 2014). It was shown that even an abundant amount of substrate for reductive carboxylation in IDH1 mutant cells did not result in higher citrate levels, which suggest that the process itself is disrupted. IDH1 mutant glioma cells might be sensitive to mitochondrial stress, because the IDH1 mutation silences the cytoplasmic, Gln-dependent pathway for metabolites and fatty acids (Table 3).
Table 3.
Lipids synthesis in cells with wild-type IDH and mutated IDH under different growth conditions
| Wild-type IDH | Mutant IDH | |
|---|---|---|
| IDH-mediated reaction | Isocitrate <−> α-ketoglutarate | α-ketoglutarate—>2-hydroxyglutarate |
| Lipid synthesis under | ||
| Normal conditions | Good | Good |
| Main substrate | Glucose | Glucose |
| Pathway | TCA cycle | TCA cycle |
| Hypoxic conditions | Good | Compromised |
| Main substrate | Glutamine | Glutamine |
| Pathway | IDH-mediated reversed flux | Disturbed reversed flux |
| Inhibited TCA metabolism | Good | Compromised |
| Main substrate | Glutamine | Glutamine |
| Pathway | IDH-mediated reversed flux | Disturbed reversed flux |
IDH-mutant cells are susceptible to ROS-induced damage
In cases of disrupted metabolic pathways, like an impeded TCA cycle, it is crucial for the cell to obtain a TCA cycle-independent source for αKG. Glu can easily be converted into αKG in the cytoplasm. Contrary to IDH wt glioma cells, IDH-mutant glioma cells do not release Glu on a large scale, because it is needed among others as substrate for the TCA cycle. Since IDH-mutant gliomas are “glutamate suckers” (Van Lith et al. 2014), they cannot import much Cys through the system x −c. Therefore, they might have a lack of GSH. Together with low levels of NADPH this can result in a hampered resistance to ROS. Since Glu levels are strongly affiliated with Gln levels, IDH-mutant glioma cells also experience a high demand for Gln.
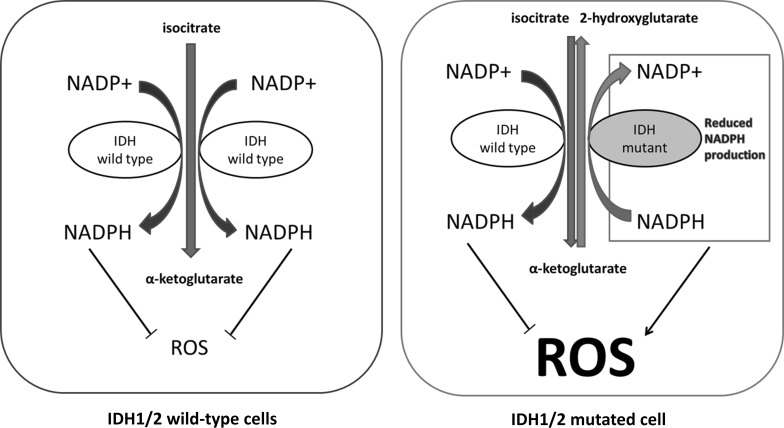
The IDH1-mediated conversion of αKG to isocitrate goes along with the conversion of NADPH to NADP+. The reversed reaction creates NADPH. Mutant IDH1, however, does not mediate reactions with isocitrate anymore. Instead it mediates the unidirectional conversion of αKG into 2HG with NADP+ as abundant side product. Subsequently cytoplasmic production of NADPH or NADH, respectively, is decreased and available NADPH or NADH is consumed (Fig. 4). NADPH is the major metabolite to protect the cell from reactive oxygen species (ROS), because it is necessary to turn oxidized glutathione (GSSG) into reduced glutathione (GSH). GSH in turn directly neutralizes free radicals and ROS.
Fig. 4.
In IDH1/2 wild-type cells, continuous NADPH production ensures low ROS levels (left). Mutant IDH1/2 activity consumes NADPH and lowers NADPH production. That results in increased ROS levels (right). ROS reactive oxygen species
As stated above, it is assumed that increased mitochondrial IDH2 activity makes up for cytoplasmic lack of αKG, which would increase NADPH availability. To what extent mitochondrial NADPH is transported to the cytoplasm remains to be elucidated. Whether the cancers with mutant IDH are more susceptible to ROS-induced stress (e.g., through radiation or temozolomide) is certainly dependent on the effects of the altered metabolism on the pentose phosphate pathway (PPP). The PPP is the major source of NADPH in the cytoplasm (Eggleston and Krebs 1974), and has been linked to cancer metabolism (Tsouko et al. 2014).
2HG directly interferes with ATP synthase
Besides the tumor-initiating effects, both enantiomers of 2HG have another substantial effect on cancer cell metabolism. 2HG and αKG are capable of binding directly to ATP synthase. In this way the enzyme is inhibited and cannot exert ATP synthesis anymore. That has negative effects on mitochondrial respiration and mTOR signaling in ID- mutant cancers (Fu et al. 2015).
Conclusion
In this review we highlight that αKG and Glu are keystones in several crucial metabolic pathways. Next to glucose, Gln and αKG are important energy sources for the cancer cell. Especially in hypoxic conditions glutaminolysis and reductive carboxylation are needed to sustain cancer cell growth and proliferation. Glu and αKG are important links in the sequence of biochemical reactions. Furthermore, released Glu can have an impact on cells in the tumor vicinity, since high Glu levels lead to astrocytic swelling and apoptosis, which is believed to ease tumor expansion. Blockage of the efflux transporter System x −c abrogates the negative effect of glioma cells on their microenvironment. However, it does not hamper the glioma cells’ viability. Similarly, deprivation of amino acids has negative effects on cancer cells, but this is not sufficient to cause apoptosis. For example, Asn alone can block the apoptotic pathway on Glu-deprived glioma cells. Interestingly, when amino acids like Cys or Met are lacking in glioma cells, ROS levels increase. When it would be possible to push ROS levels over a critical limit, glioma proliferation would be inhibited. To achieve this, a combined approach of radiation, temozolomide and deprivation of amino acids and nutrients would be optimal. However, it is questionable if this is applicable in a clinical setting, without severely damaging healthy areas of the brain, regarding the neurotoxic side effects of anti-cancer drugs such as oxaliplatin, bortezomib, or epothilone-B (Ceresa et al. 2014).
Recently a meta-study of 55 observational studies has shown that glioma patients with IDH-mutant tumors have a higher overall survival rate compared to IDH-wt tumors. Furthermore their progression-free survival is significantly increased (Xia et al. 2015). This report is in line with reports of higher degrees of cell death in IDH-mutant gliomas. Regarding the reports about metabolic changes through IDH mutations, it seems natural that tumors with IDH mutations perform worse than IDH-wt tumors. Here we summarize the ways in which IDH mutations impede cellular metabolism:
Mutant IDH1 cannot perform reductive carboxylation to allow macromolecule synthesis in hypoxia.
Mutant IDH enzymes enhance the NADPH-dependent conversion of αKG to 2HG; leaving the cell with low NADPH levels and therefore more susceptible to stress through free radicals and ROS.
2HG directly inhibits ATP synthase by binding to it and leads to unfavorable effects under glucose restriction.
It became apparent that especially hypoxic conditions are highly unfavorable for IDH-mutant cells. IDH-mutant cells might not be able to survive or at least proliferate in hypoxic conditions (Fig. 3). That would explain why IDH mutations are not common in solid tumors and only have been documented in diffuse forms of cancer like glioma or leukemia. In diffuse cancer types cells do not stay in close vicinity and therefore have fewer problems with oxygen supply.
IDH mutations play a large role in tumor onset. Later on, however, the malfunction of the IDH enzyme seems to challenge the cell more than it aids it. The metabolic alterations discussed in this article give a partial explanation at a molecular level for the effects of IDH mutations on overall survival that were reported in clinical studies. With regards to therapy approaches, it might be effective to enhance the problems the cell is confronted with by the mutant IDH enzyme.
Abbreviations
- 2HG
2-Hydroxyglutarate (also known as 2-oxoglutarate)
- ALL
Acute lymphoblastic leukemia
- Arg
Arginine
- AS
Asparagine synthetase
- Asn
Asparagine
- ASNase
Asparaginase
- BCAT1
Branched-chain amino acid transaminase 1
- Cys
Cysteine
- EGFR
Epidermal growth factor receptor
- G-CIMP
Glioma-CpG island methylator phenotype
- GBM
Glioblastoma multiforme
- GDH
Glutamate dehydrogenase
- GLA
Glutaminase
- Gln
Glutamine
- GLS
Gls gene
- Glu
Glutamate
- GSH
(reduced) Glutathione
- GSSG
(oxidized) glutathione
- IDH1
Isocitrate dehydrogenase 1
- IDH2
Isocitrate dehydrogenase 2
- JHDMs
Jumonji-C-domain-containing histone demethylases
- Met
Methionine
- MRS
Magnetic resonance spectroscopy
- mTOR
Mechanistic target of rapamycin (or mammalian target of rapamycin)
- mTORC1
mTOR complex 1
- mTORC2
mTOR complex 2
- PDH
Pyruvate dehydrogenase
- PPP
Pentose phosphate pathway
- ROS
Reactive oxygen species
- siRNA
Small interfering RNA
- TCA
Tricarboxylic acid
- TET
Ten-eleven-translocation
- wt
Wild-type
- αKG
α-Ketoglutarate
Compliance with ethical standards
Conflict of interest
The authors declare that they have no conflict of interest.
This article does not contain any studies with human participants or animals performed by any of the authors. Hence no informed consent was required for any part of this review.
Footnotes
An erratum to this article is available at http://dx.doi.org/10.1007/s00726-017-2414-5.
References
- Albrecht J, Norenberg MD. Glutamine: a trojan horse in ammonia neurotoxicity. Hepatology. 2006;44(4):788–794. doi: 10.1002/hep.21357. [DOI] [PubMed] [Google Scholar]
- Avramis VI. Asparaginases: biochemical pharmacology and modes of drug resistance. Anticancer Res. 2012;2438:2423–2437. [PubMed] [Google Scholar]
- Babic I, Anderson ES, Tanaka K, et al. EGFR mutation-induced alternative splicing of max contributes to growth of glycolytic tumors in brain cancer. Cell Metab. 2013;17(6):1000–1008. doi: 10.1016/j.cmet.2013.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bak LK, Schousboe A, Waagepetersen HS. The glutamate/GABA-glutamine cycle: aspects of transport, neurotransmitter homeostasis and ammonia transfer. J Neurochem. 2006;98(3):641–653. doi: 10.1111/j.1471-4159.2006.03913.x. [DOI] [PubMed] [Google Scholar]
- Barbosa-Tessmann IP, Pineda VL, Nick HS et al (1999) Transcriptional regulation of the human asparagine synthetase gene by carbohydrate availability. Biochem J 339(Pt 1):151–158. doi:10.1042/0264-6021:3390151 [PMC free article] [PubMed]
- Bellot GL, Liu D, Pervaiz S. ROS, autophagy, mitochondria and cancer: Ras, the hidden master? Mitochondrion. 2013;13(3):155–162. doi: 10.1016/j.mito.2012.06.007. [DOI] [PubMed] [Google Scholar]
- Bogdanovic E. IDH1, lipid metabolism and cancer: shedding new light on old ideas. Biochim Biophys Acta Gen Subj. 2015;1850(9):1781–1785. doi: 10.1016/j.bbagen.2015.04.014. [DOI] [PubMed] [Google Scholar]
- Borger DR, Zhu AX. IDH mutations: new genetic signatures in cholangiocarcinoma and therapeutic implications. Expert Rev Anticancer Ther. 2012;12(5):543–546. doi: 10.1586/era.12.32. [DOI] [PubMed] [Google Scholar]
- Borodovsky A, Seltzer MJ, Riggins GJ. Altered cancer cell metabolism in gliomas with mutant IDH1 or IDH2. Curr Opin Oncol. 2012;24(1):83–89. doi: 10.1097/CCO.0b013e32834d816a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Busek P, Stremenova J, Sromova L, et al. Dipeptidyl peptidase-IV inhibits glioma cell growth independent of its enzymatic activity. Int J Biochem Cell Biol. 2012;44(5):738–747. doi: 10.1016/j.biocel.2012.01.011. [DOI] [PubMed] [Google Scholar]
- Campos-Sandoval J, Martín-Rufián M, Cardona C, et al. Glutaminases in brain: multiple isoforms for many purposes. Neurochem Int. 2015;88:1–5. doi: 10.1016/j.neuint.2015.03.006. [DOI] [PubMed] [Google Scholar]
- Ceresa C, Avan A, Giovannetti E, et al. Characterization of and protection from neurotoxicity induced by oxaliplatin, bortezomib and epothilone-B. Anticancer Res. 2014;34(1):517–523. [PubMed] [Google Scholar]
- Chowdhury R, Yeoh KK, Tian Y-M, et al. The oncometabolite 2-hydroxyglutarate inhibits histone lysine demethylases. EMBO Rep. 2011;12(5):463–469. doi: 10.1038/embor.2011.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dang L, White DW, Gross S, et al. Cancer-associated IDH1 mutations produce 2-hydroxyglutarate. Nature. 2009;462(7274):739–744. doi: 10.1038/nature08617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dang L, Jin S, Su SM. IDH mutations in glioma and acute myeloid leukemia. Trends Mol Med. 2010;16(9):387–397. doi: 10.1016/j.molmed.2010.07.002. [DOI] [PubMed] [Google Scholar]
- Dasgupta B, Yi Y, Hegedus B, et al. Cerebrospinal fluid proteomic analysis reveals dysregulation of methionine aminopeptidase-2 expression in human and mouse neurofibromatosis 1-associated glioma. Cancer Res. 2005;65(21):9843–9850. doi: 10.1158/0008-5472.CAN-05-1842. [DOI] [PubMed] [Google Scholar]
- DeBerardinis RJ, Cheng T. Q’s next: the diverse functions of glutamine in metabolism, cell biology and cancer. Oncogene. 2010;29(3):313–324. doi: 10.1038/onc.2009.358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eggleston LV, Krebs H. Regulation of the pentose phosphate cycle. Biochem J. 1974;138(3):425–435. doi: 10.1042/bj1380425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ekstrand AJ, James CD, Cavenee WK, et al. Genes for epidermal growth factor receptor, transforming growth factor alpha, and epidermal growth factor and their expression in human gliomas in vivo. Cancer Res. 1991;51:2164–2172. [PubMed] [Google Scholar]
- Figueroa ME, Abdel-Wahab O, Lu C, et al. Leukemic IDH1 and IDH2 mutations result in a hypermethylation phenotype, disrupt TET2 function, and impair hematopoietic differentiation. Cancer Cell. 2010;18(6):553–567. doi: 10.1016/j.ccr.2010.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Filipp FV, Scott DA, Ronai Z, et al. Reverse TCA cycle flux through isocitrate dehydrogenases 1 and 2 is required for lipogenesis in hypoxic melanoma cells. Pigment Cell Melanoma Res. 2012;25:375–383. doi: 10.1111/j.1755-148X.2012.00989.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fu X, Chin RM, Vergnes L, et al. 2-Hydroxyglutarate inhibits ATP synthase and mTOR signaling. Cell Metab. 2015;22(3):508–515. doi: 10.1016/j.cmet.2015.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grassian AR, Parker SJ, Davidson SM, et al. IDH1 mutations alter citric acid cycle metabolism and increase dependence on oxidative mitochondrial metabolism. Cancer Res. 2014;74(12):3317–3331. doi: 10.1158/0008-5472.CAN-14-0772-T. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gross S, Cairns RA, Minden MD, et al. Cancer-associated metabolite 2-hydroxyglutarate accumulates in acute myelogenous leukemia with isocitrate dehydrogenase 1 and 2 mutations. J Exp Med. 2010;207(2):339–344. doi: 10.1084/jem.20092506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo C, Pirozzi CJ, Lopez GY, et al. Isocitrate dehydrogenase mutations in gliomas. Curr Opin Neurol. 2011;24(6):648–652. doi: 10.1097/WCO.0b013e32834cd415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo P, Nie Q, Lan J, et al. C-Myc negatively controls the tumor suppressor PTEN by upregulating miR-26a in glioblastoma multiforme cells. Biochem Biophys Res Commun. 2013;441(1):186–190. doi: 10.1016/j.bbrc.2013.10.034. [DOI] [PubMed] [Google Scholar]
- Hanahan D, Weinberg R. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- He X, Huang Q, Qiu X, et al. LAP3 promotes glioma progression by regulating proliferation, migration and invasion of glioma cells. Int J Biol Macromol. 2015;72:1081–1089. doi: 10.1016/j.ijbiomac.2014.10.021. [DOI] [PubMed] [Google Scholar]
- Hertz L. Functional interactions between neurons and astrocytes I. Turnover and metabolism of putative amino acid transmitters. Prog Neurobiol. 1979;13:277–323. doi: 10.1016/0301-0082(79)90018-2. [DOI] [PubMed] [Google Scholar]
- Hertz L, Dringen R, Schousboe A, et al. Astrocytes: glutamate producers for neurons. J Neurosci Res. 1999;57(4):417–428. doi: 10.1002/(SICI)1097-4547(19990815)57:4<417::AID-JNR1>3.0.CO;2-N. [DOI] [PubMed] [Google Scholar]
- Hettmer S, Schinzel AC, Tchessalova D, et al. Functional genomic screening reveals asparagine dependence as a metabolic vulnerability in sarcoma. Elife. 2015;4:e09436. doi: 10.7554/eLife.09436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirata M, Sasaki M, Cairns RA et al (2015) Mutant IDH is sufficient to initiate enchondromatosis in mice. Proc Natl Acad Sci USA 112(9):2829–34. doi:10.1073/pnas.1424400112 [DOI] [PMC free article] [PubMed]
- Hitzerd SM, Verbrugge SE, Ossenkoppele G, et al. Positioning of aminopeptidase inhibitors in next generation cancer therapy. Amino Acids. 2014;46(4):793–808. doi: 10.1007/s00726-013-1648-0. [DOI] [PubMed] [Google Scholar]
- Jayakumar AR, Panickar KS, Murthy CRK, et al. Oxidative stress and mitogen-activated protein kinase phosphorylation mediate ammonia-induced cell swelling and glutamate uptake inhibition in cultured astrocytes–Jayakumar et al. 26 (18):4774. J Neurosci. 2006;26(18):4774–4784. doi: 10.1523/JNEUROSCI.0120-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ji H, Wu G, Zhan X et al (2011) Cell-type independent MYC target genes reveal a primordial signature involved in biomass accumulation. PLoS One 6(10). doi:10.1371/journal.pone.0026057 [DOI] [PMC free article] [PubMed]
- Kaelin WG, McKnight SL. Influence of metabolism on epigenetics and disease. Cell. 2013;153(1):56–69. doi: 10.1016/j.cell.2013.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim JW, Tchernyshyov I, Semenza GL, et al. HIF-1-mediated expression of pyruvate dehydrogenase kinase: a metabolic switch required for cellular adaptation to hypoxia. Cell Metab. 2006;3(3):177–185. doi: 10.1016/j.cmet.2006.02.002. [DOI] [PubMed] [Google Scholar]
- King A, Selak M, Gottlieb E. Succinate dehydrogenase and fumarate hydratase: linking mitochondrial dysfunction and cancer. Oncogene. 2006;25(34):4675–4682. doi: 10.1038/sj.onc.1209594. [DOI] [PubMed] [Google Scholar]
- Lewerenz J, Hewett SJ, Huang Y, et al. The cystine/glutamate antiporter system x(c)(-) in health and disease: from molecular mechanisms to novel therapeutic opportunities. Antioxid Redox Signal. 2013;18(5):522–555. doi: 10.1089/ars.2011.4391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H, Zhou F, Du W et al (2015) Knockdown of asparagine synthetase by RNAi suppresses cell growth in human melanoma cells and epidermoid carcinoma cells. Biotechnol Appl Biochem 1–6. doi:10.1002/bab.1383 [DOI] [PubMed]
- Liu H, Zhang W, Wang K, et al. Methionine and cystine double deprivation stress suppresses glioma proliferation via inducing ROS/autophagy. Toxicol Lett. 2015;232(2):349–355. doi: 10.1016/j.toxlet.2014.11.011. [DOI] [PubMed] [Google Scholar]
- Luo H, Chen Z, Wang S, et al. C-Myc-miR-29c-REV3L signalling pathway drives the acquisition of temozolomide resistance in glioblastoma. Brain. 2015;138(12):3654–3672. doi: 10.1093/brain/awv287. [DOI] [PubMed] [Google Scholar]
- Masui K, Tanaka K, Akhavan D, et al. MTOR complex 2 controls glycolytic metabolism in glioblastoma through FoxO acetylation and upregulation of c-Myc. Cell Metab. 2013;18(5):726–739. doi: 10.1016/j.cmet.2013.09.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Masui K, Cavenee WK, Mischel PS. MTORC2 in the center of cancer metabolic reprogramming. Trends Endocrinol Metab. 2014;25(7):364–373. doi: 10.1016/j.tem.2014.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Metallo CM, Gameiro PA, Bell EL et al (2012) Reductive glutamine metabolism by IDH1 mediates lipogenesis under hypoxia. TL - 481. Nature 481VN-(7381):380–384. doi:10.1038/nature10602 [DOI] [PMC free article] [PubMed]
- Molenaar RJ, Radivoyevitch T, Maciejewski JP, et al. The driver and passenger effects of isocitrate dehydrogenase 1 and 2 mutations in oncogenesis and survival prolongation. Biochim Biophys Acta. 2014;1846(2):326–341. doi: 10.1016/j.bbcan.2014.05.004. [DOI] [PubMed] [Google Scholar]
- Mullen AR, Wheaton WW, Jin ES, et al. Reductive carboxylation supports growth in tumour cells with defective mitochondria. Nature. 2012;481(7381):385–388. doi: 10.1038/nature10642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noushmehr H, Weisenberger DJ, Diefes K, et al. Identification of a CpG Island methylator phenotype that defines a distinct subgroup of glioma. Cancer Cell. 2010;17(5):510–522. doi: 10.1016/j.ccr.2010.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Panosyan EH, Wang Y, Xia P, et al. Asparagine depletion potentiates the cytotoxic effect of chemotherapy against brain tumors. Mol Cancer Res. 2014;12(5):694–702. doi: 10.1158/1541-7786.MCR-13-0576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parker SJ, Metallo CM. Metabolic consequences of oncogenic IDH mutations. Pharmacol Ther. 2015;152:54–62. doi: 10.1016/j.pharmthera.2015.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parsons DW, Jones S, Zhang X, et al. An integrated genomic analysis of human glioblastoma multiforme. Science. 2008;2008(1807):1807–1812. doi: 10.1126/science.1164382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Popovici-Muller J, Saunders JO, Salituro FG, et al. Discovery of the first potent inhibitors of mutant IDH1 that lower tumor 2-HG in vivo. ACS Med Chem Lett. 2012;3(10):850–855. doi: 10.1021/ml300225h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reitman ZJ, Yan H. Isocitrate dehydrogenase 1 and 2 mutations in cancer: alterations at a crossroads of cellular metabolism. J Natl Cancer Inst. 2010;102(13):932–941. doi: 10.1093/jnci/djq187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rohle D, Popovici-Muller J, Palaskas N, et al. An inhibitor of mutant IDH1 delays growth and promotes differentiation of glioma cells. Science. 2013;340(6132):626–630. doi: 10.1126/science.1236062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saric T, Graef CI, Goldberg AL. Pathway for degradation of peptides generated by proteasomes: a key role for thimet oligopeptidase and other metallopeptidases. J Biol Chem. 2004;279(45):46723–46732. doi: 10.1074/jbc.M406537200. [DOI] [PubMed] [Google Scholar]
- Sasaki M, Knobbe CB, Munger JC, et al. IDH1(R132H) mutation increases murine haematopoietic progenitors and alters epigenetics. Nature. 2012;488(7413):656–659. doi: 10.1038/nature11323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Savaskan NE, Heckel A, Hahnen E et al (2008) Small interfering RNA—mediated neurodegeneration and alleviates. Nat Mad 14(6):629–632. doi:10.1038/nm1772 [DOI] [PubMed]
- Snoek BC, de Wilt LH, Jansen G, et al. Role of E3 ubiquitin ligases in lung cancer. World J Clin Onco. 2013;4(3):58–69. doi: 10.5306/wjco.v4.i3.58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stupp R, Mason WP, van den Bent MJ, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005;352(10):987–996. doi: 10.1056/NEJMoa043330. [DOI] [PubMed] [Google Scholar]
- Takano T, Lin JH, Arcuino G, et al. Glutamate release promotes growth of malignant gliomas. Nat Med. 2001;7(9):1010–1015. doi: 10.1038/nm0901-1010. [DOI] [PubMed] [Google Scholar]
- Tanaka K, Sasayama T, Irino Y, et al. Compensatory glutamine metabolism promotes glioblastoma resistance to mTOR inhibitor treatment. J Clin Invest. 2015;125(4):1–12. doi: 10.1172/JCI78239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thunberg T. Zur Kenntnis des intermediären Stoffwechsels und der dabei wirksamen Enzyme. Skand Arch Physiol. 1920;40(2):1–91. doi: 10.1111/j.1748-1716.1920.tb01412.x. [DOI] [Google Scholar]
- Tönjes M, Barbus S, Park YJ, et al. BCAT1 promotes cell proliferation through amino acid catabolism in gliomas carrying wild-type IDH1. Nat Med. 2013;19(7):901–908. doi: 10.1038/nm.3217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsouko E, Khan AS, White MA, et al. Regulation of the pentose phosphate pathway by an androgen receptor-mTOR-mediated mechanism and its role in prostate cancer cell growth. Oncogenesis. 2014;3(5):e103. doi: 10.1038/oncsis.2014.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turcan S, Rohle D, Goenka A, et al. IDH1 mutation is sufficient to establish the glioma hypermethylator phenotype. Nature. 2012;483(7390):479–483. doi: 10.1038/nature10866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Lith SM, Navis AC, Verrijp K, et al. Glutamate as chemotactic fuel for diffuse glioma cells: are they glutamate suckers? Biochim Biophys Acta - Rev Cancer. 2014;1846(1):66–74. doi: 10.1016/j.bbcan.2014.04.004. [DOI] [PubMed] [Google Scholar]
- Warburg O. Über den Stoffwechsel der Carcinom-Zelle. Klin Wochenschr. 1925;4(12):534–536. doi: 10.1007/BF01726151. [DOI] [Google Scholar]
- Warburg O. Krebsforschung. Naturwissenschaften. 1954;41(21):485–486. doi: 10.1007/BF00631820. [DOI] [Google Scholar]
- Warburg O, Wind F, Negelein E. Über den Stoffwechsel von Tumoren im Körper. Klin Wochenschr. 1926;5(19):829–832. doi: 10.1007/BF01726240. [DOI] [Google Scholar]
- Ward PS, Thompson CB. Metabolic reprogramming: a cancer hallmark even warburg did not anticipate. Cancer Cell. 2012;21(3):297–308. doi: 10.1016/j.ccr.2012.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ward PS, Patel J, Wise DR, et al. The common feature of leukemia-associated IDH1 and IDH2 mutations is a neomorphic enzyme activity converting α-ketoglutarate to 2-hydroxyglutarate. Cancer Cell. 2010;17(3):225–234. doi: 10.1016/j.ccr.2010.01.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weil-Malherbe H. The metabolism of glutamic acid in brain. Stud brain Metab. 1936;95(1):665–676. doi: 10.1042/bj0300665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wise DR, Ward PS, Shay JES, et al. Hypoxia promotes isocitrate dehydrogenase-dependent carboxylation of α-ketoglutarate to citrate to support cell growth and viability. Proc Natl Acad Sci USA. 2011;108(49):19611–19616. doi: 10.1073/pnas.1117773108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong AJ, Ruppert JM, Bigner SH, et al. Structural alterations of the epidermal growth factor receptor gene in human gliomas. Proc Natl Acad Sci USA. 1992;89(7):2965–2969. doi: 10.1073/pnas.89.7.2965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xia L, Wu B, Fu Z, et al. Prognostic role of IDH mutations in gliomas: a meta-analysis of 55 observational studies. Oncotarget. 2015;6(19):17354–17365. doi: 10.18632/oncotarget.4008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu W, Yang H, Liu Y, et al. Oncometabolite 2-hydroxyglutarate is a competitive inhibitor of α-ketoglutarate-dependent dioxygenases. Cancer Cell. 2011;19(1):17–30. doi: 10.1016/j.ccr.2010.12.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan H, Parsons DW, Jin G, et al. Mutations in Gliomas. N Engl J Med. 2009;360(8):765–773. doi: 10.1056/NEJMoa0808710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yao P-S, Kang D-Z, Lin R-Y, et al. Glutamate/glutamine metabolism coupling between astrocytes and glioma cells: neuroprotection and inhibition of glioma growth. Biochem Biophys Res Commun. 2014;450(1):295–299. doi: 10.1016/j.bbrc.2014.05.120. [DOI] [PubMed] [Google Scholar]
- Ye Z, Sontheimer H. Glioma cells release excitotoxic concentrations of glutamate glioma cells release excitotoxic concentrations of glutamate. Cancer Res. 1999;1:4383–4391. [PubMed] [Google Scholar]
- Zhang J, Fan J, Venneti S, et al. Asparagine plays a critical role in regulating cellular adaptation to glutamine depletion. Mol Cell. 2014;56(2):205–218. doi: 10.1016/j.molcel.2014.08.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao S, Lin Y, Xu W, Jiang W, Zha Z, Wang P, Yu W, Li Z, Gong L, Peng Y, Ding J, Lei Q, Guan KL, Xiong Y (2009) Glioma-derived mutations in IDH1 dominantly inhibit IDH1 catalytic activity and induce HIF-1alpha. Science 324(5924):261–265. doi:10.1126/science.1170944 [DOI] [PMC free article] [PubMed]