Abstract
This paper assesses hypothesis that sufficient sleep duration and proper circadian rhythms during the early stages of life are indispensable to a successful life. Successful life was defined according to the famous cohort studies of Mischel's and Dunedin. To assess the hypothesis, neuronal elements presumably affecting early daily habits and successful life are reviewed. The effect of sufficient sleep duration and proper circadian rhythms during early stages of life on the development of the prefrontal cortex has been found to be the key issue to verify the hypothesis. Socioeconomic status is found to be another issue to be studied.
Keywords: Serotonin, Oxytocin, Prefrontal cortex, Marshmallow test, The dorsal striatum, Socioeconomic status
1. Introduction
Insufficient sleep duration affects various brain functions [1] and is associated with behavioral, cognitive, physical, and developmental problems [2], [3], [4], [5]. Physical, somatic and mental disturbances are also associated with circadian rhythm disturbances, such as shift work [6], jet lag [6] and social jet lag [7]. Short sleep duration during childhood has been reported to be associated with obesity in adults [8]. However, the long-term effects of unfavorable early daily habits (insufficient sleep duration and disrupted circadian rhythms during the early stages of life) on brain functions are not fully understood [9].
Poor daily habits of modern society (loss of physical contact, decreased physical activity, and short sleep duration with late bed and waking times) during the early stages of life are associated with truancy or dropping out of school, quitting employment, and committing suicide during subsequent years [10]. It has been suggested that deactivation of the serotonergic (5-hydroxytryptamine, 5-HT) system, the dorsal striatum, and the prefrontal cortex (PFC) are the candidate neuronal elements which relate the poor early daily habits with the subsequent unfavorable behaviors [9].
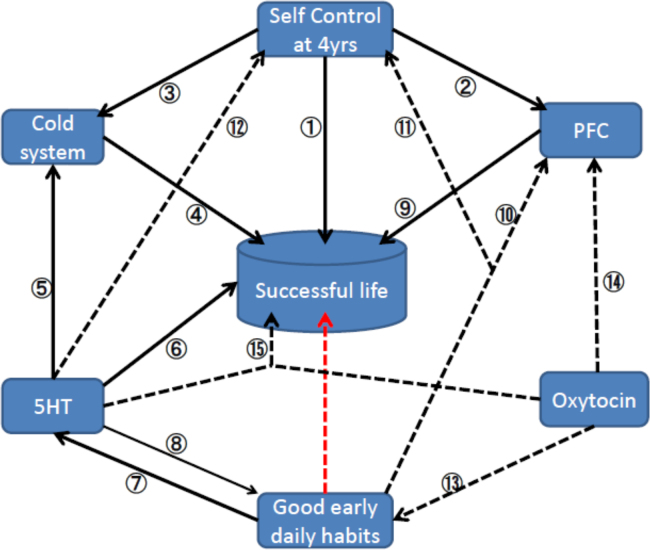
Self-control is known as a significant determinant of success throughout life, although it might be harmful to health by speeding the process of aging, especially for persons who were born into lower social classes [11]. Thus, socioeconomic status (SES) is assumed to affect early development through various elements. Although the famous longitudinal studies by the Mischel group [12], [13], [14] and the Dunedin study [15] revealed the association between early self-control and subsequent lives, they did not mention about SES. Anyway, the former [12], [13], [14] identified factors to build better lives, such as higher academic scores, better social cognitive and emotional coping, higher educational achievement, higher sense of self-worth, better ability to cope with stress, less risky drug use, and lower body mass. The latter [15] identified good health, good wealth, and low crime outcomes. The current brief paper assesses hypothesis that sufficient sleep duration and proper circadian rhythms during the early stages of life (good early daily habits) are indispensable to having a subsequent adequate social life (successful life) (Fig. 1). However, it should be noted that large inter-individual differences are observed in preferred time of day for completion of distinct cognitive tasks, sleep timing, sleep duration and sleep structure [16], although humans are known to be diurnal animals [17]. Similarly, it is hard to define proper circadian rhythm, however, we now know that pre-industrial people awake just before sunrise and go to sleep 3.3 h after sunset [18]. An adequate social life (successful life) was defined according to the famous cohort studies of Mischel's and Dunedin [12], [13], [14], [15] as a situation with presumable factors to build better lives. As mentioned before, these factors includes social cognitive and emotional coping, higher academic scores, higher educational achievement, higher sense of self-worth, better ability to cope with stress, lower body mass, good health, less risky drug use, low crime outcomes and good wealth..
Fig. 1.
Hypothesis and supported data. The red dotted arrow represents the current hypothesis that sufficient sleep duration and proper circadian rhythms during early stages of life are indispensable for an adequate social life. The numbered arrows from (1) to (15) represent the available data to support the hypothesis. Broad lines have evidence, whereas a thin arrow (8) has only weak evidence. Dotted arrows (10–15) need more studies to be confirmed. 5-HT; serotonin, PFC; prefrontal cortex.
To assess the hypothesis, neuronal elements presumably affecting early daily habits and successful life are reviewed. As such neuronal elements, 5-HT, oxytocin, the PFC and the ventral/dorsal striatum were selected with reference to previous studies [9]. Relationships among these elements are shown as numbered arrows in Fig. 1 and are evaluated in the following sections in numerical order. Through these analyses, the further and new study directions on the neuronal bases between the early stage of life and the subsequent one are expected to be found. It should also be noted that arrows do mean necessary requirements but not mean sufficient ones.
1.1. Arrow 1. Self-control at age four and successful life
The marshmallow test that Mischel and colleagues [14] developed in the 1960s assesses the ability of 4-year-old children to delay gratification. The children were given a choice between one reward (one marshmallow) that they could have immediately and a larger reward (two marshmallows) for which they would have to wait alone for up to 15 min [19]. Subsequently, it has been shown that this test has a significant predictive validity for social, cognitive, and mental health later in life [20], including higher educational achievement, higher sense of self-worth, better ability to cope with stress and less risky drug use [14]. Another large cohort study also showed that poor childhood self-control significantly predicted negative adult outcomes: worse health, more financial troubles, and more crimes committed [15]. It could be said that we have evidence in arrow 1 [14], [15], [19], [20].
1.2. Arrows 2–5. PFC, the ventral/dorsal striatum (hot/cold system), 5-HT system and self-control at four years of aged
Casey et al. [21] revealed that children who were less able to delay gratification showed lower self-control in their forties (low delayers). They also showed higher activation in the PFC of high delayers when resisting temptation. In addition, they reported [21] higher activation of the ventral striatum in low delayers than in high delayers. Mischel termed the ventral and dorsal striatum as the hot and cold systems, respectively [14]. According to Caseys’ report [21], arrows 2 and 3 could be said to be supported by evidence [21]. Interestingly, a recent study revealed that patients with attention-deficit/hyperactivity disorder exhibited an increased response to reward receipt in the ventral striatum [22], although the underlying neuromechanisms of attention-deficit/hyperactivity disorder have not yet been determined [23].
Activity in the ventral striatum (hot system) correlates with short-term reward predictions, whereas activity in the dorsal striatum (cold system) correlates with long-term reward predictions [24]. Long-term reward is expected to bring good wealth and thus could be interpreted as producing an adequate social life (successful life). Then, dominancy of the cold system in comparison to the hot system could be interpreted as being advantageous for reward (good wealth), which could contribute to a successful life. It could be said that we have evidence in arrow 4 [24].
These correlations have been reported to be altered by 5-HT levels. An increase in small reward choices and the rate of discounting of delayed rewards have been reported when 5-HT was low [25], and the correlation between ventral striatum activity and short-term reward seeking was enhanced when 5-HT was low (achieved by dietary tryptophan depletion) [24]. Contrarily, the correlation between dorsal striatum activity and long-term reward seeking increased when 5-HT was high (achieved by dietary tryptophan loading) [24]. Arrow 5 could be construed as being supported by evidence [24], [25].
1.3. Arrows 6–8. Good early daily habits, 5-HT system and a successful life
According to Arita [26], 5-HT neurons play a coordinating role for establishing a state that tends toward excitation, without directly acting on the muscles or the heart. In contrast, the person who has a 5-HT-deficient brain has poor awakening, poor standby of the autonomic nerves, a loose facial expression, weak posture, and a feeling of fatigue. Arita [26] also reported that these symptoms were seen in patients who have depressive disorders, poor anger management or with a loss of mental balance. Moreover, Arita [26] indicated that decreased activity of 5-HT neurons seems to underlie the pathologic condition of suicide [26]. Indeed, decreased 5-HT activity, especially in the PFC, is considered to play a significant role in suicidal behaviors [27], [28]. In addition, an elevation of 5-HT is considered to be effective on several mental and/or psychiatric disorders [29]. Therefore, a 5-HT sufficient brain is considered to be necessary for a successful life, although direct evidence in arrow 6 has yet to be obtained. However, as mentioned earlier, the correlation between dorsal striatum activity and long-term reward seeking was greater when 5-HT was high [24]. Since long-term reward could be construed as meaning a successful life, it could be said that we have evidence in arrow 6 [24], albeit indirect.
The non-pharmacological ways to activate the 5-HT system that Young [30] proposed are: food (tryptophan-enriched diet), a positive (happy) mood, exposure to bright light and exercise. The 5-HT system is known to be activated by exposure to morning light [31] and rhythmic movements, such as one's gait, chewing, and respiration [26], [30], [32]. According to the international classification of sleep disorders, version 3 [6], insufficient sleep leads to fatigue and malaise, which naturally results in poor physical activity. Insufficient sleep is disadvantageous for activation of the 5-HT system. Late bed times are often associated with decreased sleep duration [33] as well as increased opportunity for nocturnal light exposure, and late waking times that often reduce morning light exposure. Nocturnal light exposure inhibits melatonin secretion [34] and disrupts the circadian clock [35]. Reduced light exposure during the day time and in the morning [31] is disadvantageous for 5-HT activation [30]. Sufficient sleep duration and proper circadian rhythms (good daily habits), promoting morning sunlight exposure and sufficient physical activity, are considered to be necessary for activating the 5-HT system. An arrow 7 could be said to be true not only during childhood, but also during one's entire life. An arrow 7 could be said to be supported by evidence [26], [30]. Segawa [36] and Arita [26] emphasized that regular 5-HT activity that is established by age four to five or three to six, respectively, is important to obtain an adequate social life [9]. Taking these together, an activation of the 5-HT system that is achieved by sufficient sleep duration and proper circadian rhythms in these early years of life (good early daily habits) is considered to be important for a successful life. However, this history is represented by a red dotted arrow, which so far is unsupported by any direct evidence.
Factors that promote 5-HT activity, such as sufficient physical activity (exercise) may bring comfortable fatigue, which could increase sleep duration with proper circadian rhythms [35]. Daily sunlight exposure promotes 5-HT activity and also enhances nocturnal melatonin secretion [34]. The latter promotes sufficient sleep duration with proper circadian rhythms [35]. Thus, factors that activate the 5-HT system induce sufficient sleep duration and proper circadian rhythms (good daily habits) [35]. An arrow 8 could be said to have supporting data [35]. It should be noted that the 5-HT system is activated during wakefulness, and its activity is reduced during non-rapid eye movement sleep and ceases during rapid eye movement sleep [32]. The direct contribution of 5-HT system activation to inducing sleep remains to be elucidated.
Regarding arrows 7 and 8, factors that promote 5-HT activity are shown to be important for favorable sleep and circadian habits (good daily habits) - and vice versa [9]. Also, it has been shown that factors that attenuate 5-HT activity are associated with poor sleep and circadian habits - and vice versa [9]. Recent study revealed that 41% among general population were classified into evening type (morning type; 32%, neutral type; 27%) [37]. Though evening type persons are associated with problematic behaviors [see ref 2 for summary], an increasing number of persons are obliged to spent evening type life style in 24/7 society. This section emphasize the importance of good daily habits during the early stages of life, although parents of youngsters are obliged to spent daily habits of evening type.
1.4. Arrow 9. PFC and a successful life
Forbes and Grafman [38] reported that the PFC is essential for successful navigation through a complex social world that is inundated with intricate norms and moral values. Prefrontal structural and functional impairments are confirmed in antisocial populations by a meta-analysis of 43 studies [39]. The PFC, particularly the ventromedial/orbitofrontal region, has been implicated in decision-making in the context of neuroeconomics [40]. The medial PFC has been implicated in social information processing from the early stages of life [41]. The medial PFC (paracingulate cortex) is involved in developing a theory of mind that is often completed by age five [42]. In addition, the ventromedial PFC is sensitive to various types of rewards [43], [44]. Moreover, lesions to the ventromedial PFC seriously disrupt social life by affecting decision-making abilities and emotional processing [45]. Thus, the ventromedial PFC is involved in emotion, social and moral behavior, and decision-making. Based on these studies, it is suggested that an adequately functioning PFC plays a role in implicit and explicit social cognitive and moral judgment processing [38]. This results in successful navigation through a complex social world for a successful life. It could be said that we have evidence in arrow 9 [38], [39], [40], [41], [42], [43], [44], [45].
1.5. Arrow 10. Good early daily habits and PFC
Sleep loss decreases activity of the PFC [46], and sleep deprivation is linked to decreased metabolic activity in the ventromedial PFC [47]. In addition, age-related medial PFC gray-matter atrophy is associated with reduced slow wave activity during non-rapid eye movement sleep in older adults [48]. The medial PFC has been implicated in memory consolidation during sleep [49]. Inactivation of the medial PFC produced by sleep deprivation leads to deficits in retrieval of remote memories [49], [50]. These studies revealed sleep-related PFC functions, but no study has examined these effects during the early stages of life. It has been suggested that the medial PFC is important during development, from the early stages of life for emerging social cognitive abilities during infancy [41]. However, we have no direct evidence that sufficient sleep duration and proper circadian rhythms during early stages of development (good early daily habits) promote the subsequent proper development of the PFC. An arrow 10 remains to be proved and is crucial to verify the currently raised hypothesis.
1.6. Arrows 11 and 12. Good early daily habits, 5-HT and self-control at age four
Experiments are needed to determine differences in sleep duration and circadian rhythms, as well as in daily factors that are related to 5-HT activity, such as rhythmic movements and media use, between children who have chosen a small, immediate reward and those who have chosen a larger, delayed reward. The latter children are presumed to be following a proper favorable life customs with sleep of sufficient duration and proper circadian rhythms (good early daily habits), as indicated by an arrow 11. Furthermore, whether immediate or delayed gratification could be altered by changing the level of 5-HT (achieved by dietary tryptophan depletion or loading) in young children as indicated by arrow 12 might be interesting to study, but would be of ethical concern. We have no evidence of arrows 11 and 12.
1.7. Arrows 13–15. Good early daily habits, PFC, oxytocin a and successful life
Although it has been reported that oxytocin improves the quality of sleep in adult humans [51], insufficient data of the involvement of oxytocin in sleep, PFC functioning and sociality has been obtained from humans. However, it has been reported that Homo sapiens, with their high level of dependence on social behavior and cognition, could not have evolved without oxytocin [52]. In rats, the medial PFC is a site where exogenous oxytocin may act to attenuate anxiety-related behavior [53]. The coordinated activity of oxytocin and 5-HT in the nucleus accumbens is required for rewarding properties of social interaction in mice [54]. In addition, it should be noted that experiences during the early stages of life affect oxytocin receptors, which in turn may have long-term effects on social behaviors [55]. Arrows 13, 14 and 15 have yet to be investigated in humans.
1.8. A red arrow. Good early daily life and a successful life
In the earlier part of this article (on arrows 6 and 7), a history of the good early daily life and the successful life (a red dotted arrow) was described. It considered maturation of the 5-HT system, although there was no direct evidence of it. Herein, this history was seen from a view point of risk-taking behavior (RTB). RTB is an action or decision that has both the potential for danger, loss, or harm and the gain of some form of reward [56]. By reviewing literature, Womack et al. concluded that sleep loss is positively associated with RTB [57]. They suggested that this relationship between sleep loss and RTB was mediated by the reduced functioning of the ventromedial PFC [57]. Indeed, sleep deprivation has been linked to decreased metabolic activity [47] and changes in decision value signals [58] in the ventromedial PFC. Killgore et al. [59] reported that the sleep-deprived participants showed decision-making patterns similar that are to those of patients who have lesions in the ventromedial PFC. In addition, the increase in RTB is suggested to be due to changes in the ventromedial PFC [57]. Sleep loss might adversely affect the ventromedial PFC, in turn, this might produce RTB. If one assumes that RTB is one of the factors for an unsuccessful life, a red arrow could be supported by these reports. However, these data were obtained in adults. No data have been obtained from children to connect good early daily life and a successful life.
1.9. . Other issues needed to be reviewed in relation with good early daily habits and successful life
The evidence to support the currently raised hypothesis that sufficient sleep duration and proper circadian rhythms during early stages of life (good early daily habits) are indispensable for successful life are introduced. The direct red arrow (Fig. 1) would be supported indirectly by arrows 7 and 6 (5 and 4) and also by 10 and 9, with some assistance from the oxytocinergic system (arrows 13, 14, and 15).
With regard to oxytocin, however, differences in its receptor gene might also be involved in social behaviors. Knockout mice that are deficient in the oxytocin receptor gene display pervasive social deficits [60], whereas it is reported that oxytocin receptor gene polymorphism shows different protective effects in an adverse social rearing environment [61]. Similarly, polymorphism in 5-HT receptor 2a gene is a possible predisposal factor for aggressive traits [62]. These findings are compatible with Cicchetti's finding that developmental outcomes are shaped by complex interactions between environmental characteristics and genes across development [63]. Remarkably, an arrow 15 could be affected by polymorphism of both oxytocin and 5-HT receptor genes, whereas arrows 5, 6, 8, 12 could also be influenced by 5-HT receptor gene polymorphism.
On environmental factors, as described in the earlier part of this article, it has been reported that low SES is a barrier for self-control to be a determinant of success throughout life [11]. It has also been reported that children from low SES in the USA [64], Australia [65] and Brazil [66]) show higher rates of sleep problems, such as short sleep duration and late bedtime, although the opposite result was reported for China [67]. It has also been reported that low SES is associated with the occurrence of child sleep disordered breathing [68]. Low SES might be associated closely with biological phenomenon of sleep problems during the early stages of life, probably in various ways such as nutritional, hygienic and educational problems, although not expressed by an arrow in Fig. 1. Of course, nutritional, hygienic and educational problems could affect neuronal elements.
Several links remain to be studied; arrows 8, 10, 11, 12, 13, 14, 15, and the red dotted arrow in Fig. 1. Among these arrows, arrows related with 5-HT (arrows 5, 6, 7, 8, 12 and 15) are difficult to be free from receptor gene polymorphism. Then, the relationship between sufficient sleep duration and circadian rhythms during early stages of life and enhanced PFC activity (an arrow 10) seems to be the key element for verifying the currently raised hypothesis. Although arrow 11 is an indirect datum for verification of the current hypothesis, arrow 11 could be confirmed more easily than arrow 10. After confirming arrow 11, intense studies of arrow 10 should be conducted. If arrow 10 were to be verified, the currently raised hypothesis would be confirmed, at least indirectly. The direct red arrow, with an involvement of unknown neural elements, remains to be confirmed.
2. Conclusions
The current analyses demonstrated numerous indirect data supporting favorable effects of sufficient sleep duration and proper circadian rhythms during early stages of life (good early daily habits) on the proper development of the PFC, although direct evidence has remained to be accumulated. In addition, SES was found to be involved in the current hypothesis. Neuroscience of SES should be another important issue to be investigated.
Conflict of interest statement
None.
Funding
No funding was received for this study.
Footnotes
Peer review under responsibility of Brazilian Association of Sleep.
References
- 1.Mindell J.A., Owens J., Alves R., Bruni O., Goh D.Y., Hiscock H. Give children and adolescents the gift of a good night's sleep: a call to action. Sleep Med. 2011;12(3):203–204. doi: 10.1016/j.sleep.2011.01.003. [DOI] [PubMed] [Google Scholar]
- 2.Yokomaku A., Misao K., Omoto F., Yamagishi R., Tanaka K., Takada K. A study of the association between sleep habits and problematic behaviors in preschool children. Chronobiol Int. 2008;25(4):549–564. doi: 10.1080/07420520802261705. [DOI] [PubMed] [Google Scholar]
- 3.Osonoi Y., Mita T., Osonoi T., Saito M., Tamasawa A., Nakayama S. Morningness-eveningness questionnaire score and metabolic parameters in patients with type 2 diabetes mellitus. Chronobiol Int. 2014;31(9):1017–1023. doi: 10.3109/07420528.2014.943843. [DOI] [PubMed] [Google Scholar]
- 4.Schlarb A.A., Sopp R., Ambiel D., Grünwald J. Chronotype-related differences in childhood and adolescent aggression and antisocial behavior—A review of the literature. Chronobiol Int. 2014;31(1):1–16. doi: 10.3109/07420528.2013.829846. [DOI] [PubMed] [Google Scholar]
- 5.Iemura A., Iwasaki M., Yamakawa N., Tomiwa K., Anji Y., Sakakihara Y. Influence of sleep-onset time on the development of 18-month-old infants: japan Children's cohort study. Brain Dev. 2016;38(4):364–372. doi: 10.1016/j.braindev.2015.10.003. [DOI] [PubMed] [Google Scholar]
- 6.American Academy of Sleep Medicine . 3rd ed. American Academy of Sleep Medicine; Westchester, NY, USA: 2014. The International Classification of Sleep Disorder. [Google Scholar]
- 7.Wittmann M., Dinich J., Merrow M., Roenneberg T. Social jetlag: misalignment of biological and social time. Chronobiol Int. 2006;23(1–2):497–509. doi: 10.1080/07420520500545979. [DOI] [PubMed] [Google Scholar]
- 8.Landhuis C.E., Poulton R., Welch D., Hancox R.J. Childhood sleep time and long-term risk for obesity: a 32-year prospective birth cohort study. Pediatrics. 2008;122(5):955–960. doi: 10.1542/peds.2007-3521. [DOI] [PubMed] [Google Scholar]
- 9.Kohyama J. Neural basis of brain dysfunctionp produced by early sleep problems. Brain Sci. 2016;6(1):5. doi: 10.3390/brainsci6010005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kohyama J. The possible long-term effects of early-life circadian rhythm disturbance on social behavior. Expert Rev Neurother. 2014;14(7):745–755. doi: 10.1586/14737175.2014.927735. [DOI] [PubMed] [Google Scholar]
- 11.Miller G.E., Yu T., Chen E., Brody G.H. Self-control forecasts better psychological outcomes but faster epigenetic aging in low-SES youth. Proc Natl Acad Sci USA. 2015;112(33):10325–10330. doi: 10.1073/pnas.1505063112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Mischel W., Shoda Y., Peake P.K. The nature of adolescent competencies predicted by preschool delay of gratification. J Pers Soc Psychol. 1988;54(4):687–696. doi: 10.1037//0022-3514.54.4.687. [DOI] [PubMed] [Google Scholar]
- 13.Ayduk O., Mendoza-Denton R., Mischel W., Downey G., Peake P.K., Rodriguez M. Regulating the interpersonal self: strategic self-regulation for coping with rejection sensitivity. J Pers Soc Psychol. 2000;79(5):776–792. doi: 10.1037//0022-3514.79.5.776. [DOI] [PubMed] [Google Scholar]
- 14.Mischel W. The Marshmallow Test. Little, Brown and Company; New York, NY, USA: 2014. [Google Scholar]
- 15.Moffitt T.E., Arseneault L., Belsky D., Dickson N., Hancox R.J., Harrington H. A gradient of childhood self-control predicts health, wealth, and public safety. Proc Natl Acad Sci USA. 2011;108(7):2693–2698. doi: 10.1073/pnas.1010076108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Landolt H.P., Dijk D.J. Genetic basis of sleep in healthy humans. In: Kryger M.H., Roth T., Dement W.C., editors. Principles Principles and practice of sleep medicine. 5th edition. Elsevier Saunders; 2011. pp. 175–183. [Google Scholar]
- 17.Balkin T.J. Performance deficits during sleep loss; effects of time awake, time of day, and time on task. In: Kryger M.H., Roth T., Dement W.C., editors. Principles and practice of sleep medicine. 5th edition. Elsevier Saunders; 2011. pp. 738–744. [Google Scholar]
- 18.Yetish G., Kaplan H., Gurven M. Natural sleep and its seasonal variations in three pre-industrial societies. Curr Biol. 2015;25(21):2862–2868. doi: 10.1016/j.cub.2015.09.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.MischelW Father-absence and delay of gratification: crosscultural comparisons. J Abnorm Soc Psychol. 1961;63(1):116–124. doi: 10.1037/h0046877. [DOI] [PubMed] [Google Scholar]
- 20.MischelW, Ayduk O., Berman M.G., Casey B.J., Gotlib I.H., Jonides J. “Willpower” over the life span: decomposing self-regulation. Soc Cogn Affect Neurosci. 2011;6(2):252–256. doi: 10.1093/scan/nsq081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Casey B.J., Somerville L.H., Gotlib I.H., Ayduk O., Franklin N.T., Askren M.K. Behavioral and neural correlates of delay of gratification 40 years later. Proc Natl Acad Sci USA. 2011;108(36):14998–15003. doi: 10.1073/pnas.1108561108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.von Rhein D., Cools R., Zwiers M.P., van der Schaaf M., Franke B., Luman M. Increased neural responses to reward in adolescents and young adults with attention-deficit/hyperactivity disorder and their unaffected siblings. J Am Acad Child Adolesc Psychiatry. 2015;54(5):394–402. doi: 10.1016/j.jaac.2015.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sonuga-Barke E.J., Cortese S., Fairchild G., Stringaris A. Annual research review: transdiagnostic neuroscience of child and adolescent mental disorders - differentiating decision, making in attention-deficit/hypersctivity disorder, conduct disorder, depression, and anxiety. J Child Psychol Psychiatry. 2016;57(3):321–349. doi: 10.1111/jcpp.12496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tanaka S.C., Schweighofer N., Asahi S., Shishida K., Okamoto Y., Yamawaki S. Serotonin differentially regulates short- and long-term prediction of rewards in the ventral and dorsal striatum. PLoS One. 2007;2(12):e1333. doi: 10.1371/journal.pone.0001333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Schweighofer N., Bertin M., Shishida K., Okamoto Y., Tanaka S.C., Yamawaki S. Low-serotonin levels increase delayed reward discounting in humans. J Neurosci. 2008;28(17):4528–4532. doi: 10.1523/JNEUROSCI.4982-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Arita H. Brain mechanisms of poor anger management. JMAJ. 2009;52(3):184–190. [Google Scholar]
- 27.Leyton M., Paquette V., Gravel P., Rosa-Neto P., Weston F., Diksic M. Alpha-[11C]methyl-L- tryptophan trapping in the orbital and ventral medial prefrontal cortex of suicide attempters. Eur Neuropsychopharmacol. 2006;16(3):220–223. doi: 10.1016/j.euroneuro.2005.09.006. [DOI] [PubMed] [Google Scholar]
- 28.Kohyama J. Sleep, serotonin, and suicide. J Behav Brain Sci. 2012;2(4):471–478. [Google Scholar]
- 29.American Psychiatric Association . Practice Guideline for the treatment of patients with major depressive disorder. 3rd edn. American Psychiatric Association; 2010. [PubMed] [Google Scholar]
- 30.Young S.N. How to increase serotonin in the human brain without drugs. J Psychiatry Neurosci. 2007;32(6):394–399. [PMC free article] [PubMed] [Google Scholar]
- 31.Cagampang F.R., Yamazaki S., Otori Y., Inouye S.I. Serotonin in the raphe nuclei: regulation by light and an endogenous pacemaker. Neuroreport. 1993;5(1):49–52. doi: 10.1097/00001756-199310000-00012. [DOI] [PubMed] [Google Scholar]
- 32.Jacobs B.L., Azmitia E.C. Structure and function of the brain serotonin system. Physiol Rev. 1992;72(1):165–229. doi: 10.1152/physrev.1992.72.1.165. [DOI] [PubMed] [Google Scholar]
- 33.Kohyama J., Shiiki T., Ohinata-Sugimoto J., Hasegawa T. Potentially harmful sleep habits of 3-year-old children in Japan. J Dev Behav Pedia. 2002;23(2):67–70. doi: 10.1097/00004703-200204000-00001. [DOI] [PubMed] [Google Scholar]
- 34.Mishima K., Okawa M., Shimizu T., Hishikawa Y. Diminished melatonin secretion in the elderly caused by insufficient environmental illumination. J Clin Endocrinol Metab. 2001;86(1):129–134. doi: 10.1210/jcem.86.1.7097. [DOI] [PubMed] [Google Scholar]
- 35.Kohyama J. Neurochemical and neuropharmacological aspects of circadian disruptions: an introduction to asynchronization. Curr Neuropharmacol. 2011;9(2):330–341. doi: 10.2174/157015911795596522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Segawa M. Epochs of development of the sleep-wake cycle reflect the modulation of the higher cortical function particular for each epoch. Sleep Biol Rhythms. 2006;4(1):4–15. [Google Scholar]
- 37.Hu Y., Shmygelska A., Tran D., Eriksson N., Tung J.Y., Hinds D.A. GWAS of 89,283 individuals identifies genetic variants associated with self-reporting of being a morning person. Nat Commun. 2016;7:10448. doi: 10.1038/ncomms10448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Forbes C.E., Grafman J. The role of the human prefrontal cortex in social cognition and moral judgment. Annu Rev Neurosci. 2010;33:299–324. doi: 10.1146/annurev-neuro-060909-153230. [DOI] [PubMed] [Google Scholar]
- 39.Yang Y., Raine A. Prefrontal structural and functional brain imaging findings in antisocial, violent, and psychopathic individuals: ameta-analysis. Psychiatry Res. 2009;174(2):81–88. doi: 10.1016/j.pscychresns.2009.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Pearson J.M., Watson K.K., Platt M.L. Decision making: the neuroethological turn. Neuron. 2014;82(5):950–965. doi: 10.1016/j.neuron.2014.04.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Grossmann T. The role of medial prefrontal cortex in early social cognition. Front Hum Neurosci. 2013;7:340. doi: 10.3389/fnhum.2013.00340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Frith C., Frith U. Theory of mind. Curr Biol. 2005;15(4):R644–R646. doi: 10.1016/j.cub.2005.08.041. [DOI] [PubMed] [Google Scholar]
- 43.Hikosaka O., Bromberg-Martin E., Hong S., Matsumoto M. New insights on the subcortical representation of reward. Curr Opin Neurobiol. 2008;18(2):203–208. doi: 10.1016/j.conb.2008.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sescousse G., Caldú X., Segura B., Dreher J.C. Processing of primary and secondary rewards: a quantitative meta-analysis and review of human functional neuroimaging studies. Neurosci Biobehav Rev. 2013;37(4):681–696. doi: 10.1016/j.neubiorev.2013.02.002. [DOI] [PubMed] [Google Scholar]
- 45.van den Bos W., Güroglu B. The role of the ventral medial prefrontal cortex in social decision making. J Neurosci. 2009;29(24):7631–7632. doi: 10.1523/JNEUROSCI.1821-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ma N., Dinges D.F., Basner M., Rao H. How acute total sleep loss affects the attending brain: a meta-analysis of neuroimaging studies. Sleep. 2015;38(2):233–240. doi: 10.5665/sleep.4404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Thomas M., Sing H., Belenky G. Neural basis of alertness and cognitive performance impairments during sleepiness. II. Effects of 48 and 72h of sleep deprivation on waking human regional brain activity. Thalamus Relat Syst. 2003;2(3):199–229. doi: 10.1046/j.1365-2869.2000.00225.x. [DOI] [PubMed] [Google Scholar]
- 48.Mander B.A., Rao V., Lu B., Saletin J.M., Lindquist J.R., Ancoli-Israel S. Prefrontal atrophy, disrupted NREM slow waves and impaired hippocampal-dependent memory in aging. Nat Neurosci. 2013;16(3):357–364. doi: 10.1038/nn.3324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Euston D.R., Gruber A.J., McNaughton B.L. The role of medial prefrontal cortex in memory and decision making. Neuron. 2012;76(6):1057–1070. doi: 10.1016/j.neuron.2012.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Gais S., Albouy G., Boly M., Dang-Vu T.T., Darsaud A., Desseilles M. Sleep transforms the cerebral trace of declarative memories. Proc Natl Acad Sci USA. 2007;104(47):18778–18783. doi: 10.1073/pnas.0705454104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Mameli S., Pisanu G.M., Sardo S., Marchi A., Pili A., Carboni M. Oxytocin nasal spray in fibromyalgic patients. Rheuma Int. 2014;34(8):1047–1052. doi: 10.1007/s00296-014-2953-y. [DOI] [PubMed] [Google Scholar]
- 52.Carter C.S. Oxytocin pathways and the evolution of human behavior. Annu Rev Psychol. 2014;65:17–39. doi: 10.1146/annurev-psych-010213-115110. [DOI] [PubMed] [Google Scholar]
- 53.Sabihi S., Durosko N.E., Dong S.M., Leuner B. Oxytocin in the prelimbic medial prefrontal cortex reduces anxiety-like behavior in female and male rats. Psychoneuroendocrinology. 2014;45:31–42. doi: 10.1016/j.psyneuen.2014.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Dölen G., Darvishzadeh A., Huang K.W., Malenka R.C. Social reward requires coordinated activity of nucleus accumbens oxytocin and serotonin. Nature. 2013;501(7466):179–184. doi: 10.1038/nature12518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Bales K.L., Perkeybile A.M. Developmental experiences and the oxytocin receptor system. Horm Behav. 2012;61(3):313–319. doi: 10.1016/j.yhbeh.2011.12.013. [DOI] [PubMed] [Google Scholar]
- 56.Leigh B.C. Peril, chance, adventure: concepts of risk, alcohol use and risky behavior in young adults. Addiction. 1999;94(3):371–383. doi: 10.1046/j.1360-0443.1999.9433717.x. [DOI] [PubMed] [Google Scholar]
- 57.Womack S.D., Hook J.N., Reyna S.H., Ramos M. Sleep loss and risk-taking behavior: a review of the literature. Behav Sleep Med. 2013;11(5):343–359. doi: 10.1080/15402002.2012.703628. [DOI] [PubMed] [Google Scholar]
- 58.Libedinsky C., Smith D.V., Teng C.S., Namburi P., Chen V.W., Huettel S.A. Sleep deprivation alters valuation signals in the ventromedial prefrontal cortex. Front Behav Neurosci. 2011;5:70. doi: 10.3389/fnbeh.2011.00070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Killgore W.D., Balkin T.J., Wesensten N.J. Impaired decision making following 49h of sleep deprivation. J Sleep Res. 2006;15(1):7–13. doi: 10.1111/j.1365-2869.2006.00487.x. [DOI] [PubMed] [Google Scholar]
- 60.Takayanagi Y., Yoshida M., Bielsky I.F., Ross H.E., Kawamata M., Onaka T. Pervasive social deficits, but normal parturition, in oxytocin receptor-deficient mice. Proc Natl Acad Sci USA. 2005;102(44):16096–16101. doi: 10.1073/pnas.0505312102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Hostinar C.E., Cicchetti D., Rogosch F.A. Oxytocin receptor gene polymorphism, perceived social support, and psychological symptoms in maltreated adolescents. Dev Psychopathol. 2014;26(2):465–477. doi: 10.1017/S0954579414000066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Banlaki Z., Elek Z., Nanasi T., Szekely A., Nemoda Z., Sasvari-Szekely M. Polymorphism in the serotonin receptor 2a (HTR2A) gene as possible predisposal factor for aggressive traits. PLoS One. 2015;10(2):e0117792. doi: 10.1371/journal.pone.0117792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Cicchetti D. Resilience under conditions of extreme stress: a multilevel perspective. World Psychiatry. 2010;9(1):1–10. doi: 10.1002/j.2051-5545.2010.tb00297.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Hale L., Berger L.M., LeBourgeois M.K., Brooks-Gunn J. Social and demographic predictors of preschoolers’ bedtime routines. J Dev Behav Pedia. 2009;30(5):394–402. doi: 10.1097/DBP.0b013e3181ba0e64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Biggs S.N., Lushington K., James Martin A., van den Heuvel C., Declan Kennedy J. Gender, socioeconomic, and ethnic differences in sleep patterns in school-aged children. Sleep Med. 2013;14(12):1304–1309. doi: 10.1016/j.sleep.2013.06.014. [DOI] [PubMed] [Google Scholar]
- 66.Felden É.P., Leite C.R., Rebelatto C.F., Andrade R.D., Beltrame T.S. Sleep in adolescents of different socioeconomic status: a systematic review. Rev Paul Pedia. 2015;33(4):467–473. doi: 10.1016/j.rpped.2015.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Zhang J., Li A.M., Fok T.F., Wing Y.K. Roles of parental sleep/wake patterns, socioeconomic status, and daytime activities in the sleep/wake patterns of children. J Pedia. 2010;156(4) doi: 10.1016/j.jpeds.2009.10.036. [606-12.e5] [DOI] [PubMed] [Google Scholar]
- 68.Friberg D., Lundkvist K., Li X., Sundquist K. Parental poverty and occupation as risk factors for pediatric sleep-disordered breathing. Sleep Med. 2015;16(9) doi: 10.1016/j.sleep.2015.05.012. [DOI] [PubMed] [Google Scholar]