Abstract
Human papillomavirus (HPV) is a recently identified causative agent for a subset of head and neck cancers, primarily in the oropharynx, and is largely responsible for the rising worldwide incidence of oropharyngeal cancer (OPC). Patients with HPV-positive OPC have distinct risk factor profiles and generally have a better prognosis than patients with traditional, HPV-negative, head and neck cancer. Concurrent chemotherapy and radiation is a widely accepted primary treatment modality for many patients with HPV-positive OPC. However, recent advances in surgical modalities, including transoral laser and robotic surgery, have led to the reemergence of primary surgical treatment for HPV-positive patients. Clinical trials are under way to determine optimal treatment strategies for the growing subset of patients with HPV-positive OPC. Similarly, identifying those patients with HPV-positive cancer who are at risk for recurrence and poor survival is critical in order to tailor individual treatment regimens and avoid potential undertreatment.
Keywords: human papillomavirus, oropharyngeal cancer, oropharyngeal squamous cell carcinoma
HUMAN PAPILLOMAVIRUS CHARACTERISTICS
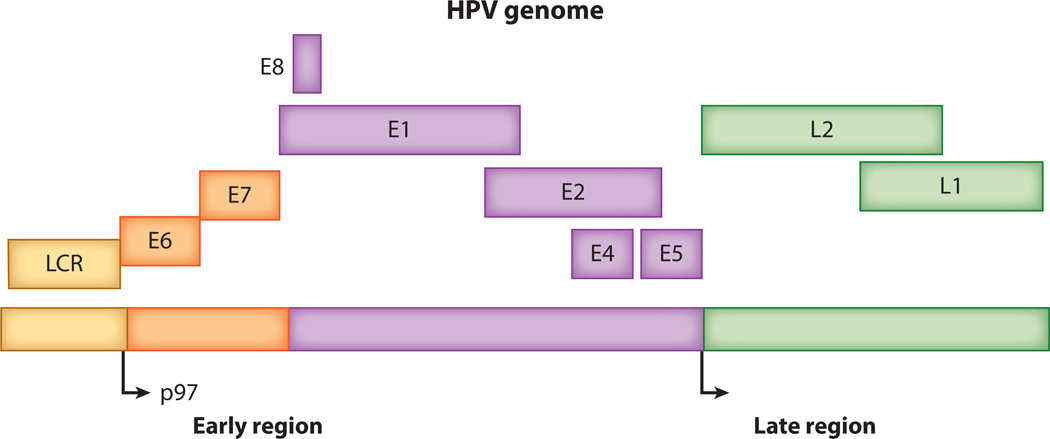
The human papillomaviruses (HPV) are small, double-stranded, circular DNA viruses (Figure 1). More than 100 distinct HPV subtypes can infect squamous epithelia and are responsible for both benign and cancerous lesions (1). Benign HPV subtypes, most commonly 6 and 11, are responsible for condyloma acuminata and respiratory papillomatosis. However, this review focuses on HPV-related cancer, which is caused by high-risk subtypes, notably 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, and 58 (2).
Figure 1.
The human papillomavirus genome. Abbreviation: LCR, long control region.
HPV has long been identified as a necessary cause of cervical cancer, the second leading cause of cancer death among women. It has also been shown to cause some cancers of the anal, rectal, and penile regions. Similarly, HPV has been identified as a distinct causative agent in oropharyngeal cancer (OPC) (3). HPV has also been isolated in nasopharyngeal and oral cancers (4, 5), but its oncogenic link in these nonoropharyngeal head and neck sites remains unclear.
While the incidence of laryngeal and hypopharyngeal squamous cell carcinoma (SCC) in developed countries is declining because of a reduction in the use of tobacco and alcohol (6), OPC incidence continues to rise. This rising incidence of OPC is thought to be related to the increasing proportion of cancers caused by HPV. Investigators have shown an increase in HPV prevalence in OPC from 16.3% during 1984–1989 to 71.7% during 2000–2004 (7). This rising incidence has been corroborated by multiple institutions and dates back to the 1950s (7–9).
It remains unclear why HPV-positive OPC is currently on the rise. Patients with HPV-positive OPC have a distinct demographic profile, tending to be younger, male, less likely to smoke, and of higher socioeconomic status than their HPV-negative counterparts (10–12). Furthermore, investigators have identified specific lifestyle factors that are associated with HPV-positive OPC, including a high lifetime number of vaginal- and oral-sex partners (12). The age of sexual debut has decreased over the last few decades, with a concomitant rise in the number of lifetime sexual partners (13, 14). Therefore, it is possible that changes in social norms and sexual practices may play a role in the rising incidence of HPV-related OPC (15).
NATURAL HISTORY OF HPV-RELATED OROPHARYNGEAL CANCER
From Infection to Malignant Transformation
Much of our current knowledge regarding HPV-related head and neck cancer is extrapolated from cervical carcinogenic models. From these models, it is known that high-risk HPV infects the basal cells of stratified squamous epithelia through microabrasions or epithelial wounds (1). Once the host cell is infected, the episomal viral genome is partitioned into two daughter cells. One daughter cell becomes a basal cell while the other enters the differentiation process and is ultimately shed with desquamated host cells as virions. HPV can live for decades in this latent state, utilizing host machinery to replicate. However, in some cases, typically many years following the initial infection, HPV-infected cells can undergo malignant transformation.
The factors that lead to malignant transformation comprise an area of intense investigation. It is well known that the oncogenes E6 and E7 are largely responsible for malignant transformation. The E6 and E7 oncoproteins inactivate the tumor suppressor proteins p53 and pRb, respectively. The action of E6 and E7 oncoproteins, along with other cellular mechanisms, such as viral integration into the host DNA, are among the integral factors related to carcinogenesis (1, 16).
Role of Immune System Evasion in Malignant Transformation
A firm understanding of the HPV life cycle is paramount to understanding how the virus evades the immune system to persist in a latent state prior to malignant transformation. Once HPV infects the basal layer of the squamous epithelium, it replicates at a low copy number (approximately 50–100 copies per host cell), thereby evading the host immune system. Once the virus reaches the superficial layer of the epithelium and differentiates into a keratinocyte, there is a substantial amount of viral replication and upregulation of genes, including the oncogenic E6 and E7. At this point the viral copy number increases, but the infected keratinocyte is shed with other desquamated cells, thereby evading the adaptive immune response (17, 18). Furthermore, because the virus is shed from the squamous epithelium, it has limited exposure to the host lymphatic system (18). These are some of the key ways in which HPV remains virtually invisible to the host immune system.
HPV also has several mechanisms for directly inhibiting the function of the host innate immune system. Interferons such as IFN-α and -β are a key component of the host response to viral infections. The HPV oncoproteins, E6 and E7, have been shown to directly interfere with multiple specific mediators of the interferon signaling pathways. The HPV E7 protein inhibits the pathway necessary for IFN-α-induced transcription (17–19). The HPV E6 protein has also been shown to decrease the expression of IFN-α, IFN-β, and STAT1 (18). Aside from its effect on interferons, HPV has the ability to alter the function of other immunostimulatory cytokines, including interleukin (IL)-18, while enhancing the expression of immunosuppressive cytokines (1, 19, 20). These are a few of the methods by which HPV evades the immune system in order to become carcinogenic.
Prognosis
The prevalence of HPV 16 in oral rinse samples from healthy adults ranges from 1.0% to 7.6% (21, 22). Only a fraction of those with oral HPV infection will develop HPV-positive OPC after many years. Patients who do develop HPV-positive OPC have a better prognosis than those with HPV-negative OPC. When compared to patients with HPV-negative OPC, patients with HPV-positive OPC have a 50% lower risk of death (23). The superior prognosis of HPV-positive OPC patients is related to the high responsiveness of these tumors to all types of treatment. Furthermore, as mentioned previously, patients who develop HPV-positive cancer are often young nonsmokers with few comorbidities, improving their tolerance to treatment and recovery thereafter. Their greater survival has been demonstrated by both recent institutional and cooperative group prospective trials (10, 11).
The Eastern Cooperative Oncology Group investigators designed the first prospective trial (ECOG 2399) to investigate HPV-related head and neck cancers (11). These investigators utilized an induction chemotherapy regimen of paclitaxel and carboplatin in patients with both HPV-positive and HPV-negative laryngeal and oropharyngeal SCC. After a median follow-up of 39.1 months, patients with HPV-positive tumors demonstrated higher overall survival than patients with HPV-negative tumors (two-year overall survival was 95% versus 62%, respectively). Similar survival differences were observed when the analysis was restricted to patients with OPC. Furthermore, among patients with oropharyngeal primary tumors, the investigators found that patients with HPV-positive tumors had a better response to induction chemotherapy (82% versus 58%) and primary chemoradiation treatment (84% versus 54%) than patients with HPV-negative tumors (11).
Soon after, the Radiation Therapy Oncology Group (RTOG) investigators performed an unplanned subset analysis of patients with advanced head and neck SCC embedded within the RTOG 0129 trial. They demonstrated superior three-year survival (82.4%) among those with HPV-positive SCC compared to patients with HPV-negative tumors (57.1%). Furthermore, using recursive partitioning analysis, they were able to stratify patients into low, intermediate, and high risk of death according to HPV status, smoking history, and tumor size. Being a smoker with a large HPV-negative tumor was associated with increased risk of death and therefore placed into the high-risk category (10).
MANAGEMENT OF HPV-RELATED OROPHARYNGEAL CANCER
Traditional Surgical Approaches to the Oropharynx
The current paradigm for the management of HPV-positive OPC is best understood by exploring the historical treatment of patients with OPC. Prior to the 1990s, advanced OPC was managed by external surgical approaches, often requiring a tracheostomy and feeding tube. Conversely, T1 and small T2 primary tumors involving the upper oropharynx, such as the tonsil, soft palate, or posterior pharyngeal wall, were often managed via a transoral approach. For patients with trismus, deep tumor invasion, or simply larger tumors, a mandibulotomy and/or mandibular swing was often required for the necessary exposure to obtain negative margins. Several external approaches, including transcervical, transpharyngeal, and transmandibular, were frequently employed for the treatment of OPC. Unfortunately, these approaches—aside from the transoral approach for small primaries—carried significant morbidity in the form of functional outcomes and cosmesis.
Nonsurgical Management of the Oropharynx
In the early 1990s, as clinicians sought to find a treatment modality with less morbidity than traditional open approaches to the oropharynx, primary radiation became a promising treatment for OPC. Several large trials in the early 1990s, including the Department of Veterans Affairs Laryngeal Cancer Study and the RTOG 91–11 trial, demonstrated that patients treated with primary radiation as opposed to upfront surgery had similar survival rates with less severe morbidity and lower mortality (24). Early clinical trials on organ preservation did not account for HPV status, as it was not yet discovered as a distinct etiology of OPC.
Definitive radiation therapy regimens were extensively explored for all subsites of the head and neck, including the oropharynx. The underlying principles of radiation therapy rely on the reassortment of cells into the sensitive phase of the cell cycle, repair of normal tissues, reoxygenation, and repopulation of tumor cells. Both conventional-dose radiation treatment and fractionation regimens were investigated thoroughly. Clinical trials have since revealed that hyperfractionation and accelerated fractionation regimens bring about higher local control rates and even improved survival for patients with OPC (25).
The addition of chemotherapy to primary radiation treatment for OPC, referred to as chemoradiation, became increasingly widespread following the initial clinical trials of the 1990s. Adjuvant chemotherapy, often in the form of cisplatin, carboplatin, docetaxel, paclitaxel, or 5-fluorouracil (5-FU), increases the effect of radiation on tumor cells. In nonoropharyngeal subsites of the head and neck, concurrent chemoradiation therapy results in similar if not superior locoregional control rates and survival when compared to radiation alone (26).
No current clinical data directly compare surgery with adjuvant radiation therapy to upfront concurrent chemoradiation for OPC. However, it has been reported that concurrent chemoradiation leads to higher survival rates and locoregional control than radiation alone for advanced-stage OPC (27). Despite the lack of direct evidence that chemoradiation improves outcomes compared to surgery, most clinicians have extrapolated from other head and neck subsites and consider the existing data sufficient to offer chemoradiation as standard primary treatment for OPC. Exceptions include early-stage OPC, which is often treated with either radiation therapy alone or surgery with or without adjuvant radiation.
The emerging role of HPV in head and neck cancer has somewhat clouded the clinical trial data in regard to OPC patients. Patients with HPV-positive OPC are more likely to have greater survival and disease control rates, regardless of treatment modality (11, 28). Furthermore, patients with HPV-positive OPC are more likely to present with smaller primary tumors and more advanced nodal disease (29). The excellent outcomes of patients with HPV-positive OPC fail to reflect the advanced stage of many of these patients on presentation. Furthermore, this discontinuity between staging and outcome in HPV-related disease potentially leads to overtreatment of these patients. Investigators have begun reevaluating the traditional staging systems for HPV-positive OPC (30, 31).
Minimally Invasive Surgical Approaches to the Oropharynx
Technological advancements in laser delivery systems and the DaVinci™ robot have caused a pendulum swing back toward surgical management of OPC in recent years. Transoral laser microsurgery (TLM) was first introduced in 1972 (32) and has become an important tool for minimally invasive head and neck surgery (33). Large multicenter cohort studies and prospective trials of OPC patients have demonstrated similar oncologic results with TLM compared to nonsurgical therapies, with better functional outcomes (33, 34). One of these prospective multicenter trials investigated TLM in the management of 204 advanced-stage OPC patients (33). The investigators demonstrated a two-year overall survival of 89% and a disease-specific survival of 91% for stage III/IV OPC patients treated with TLM. These survival results are comparable to those of similar patients undergoing primary treatment with chemoradiation (35). In regard to treatment-related morbidity, those treated with chemoradiation had a G-tube rate of 35% compared with 3.4% in the TLM study.
More recently, robotic surgery has become a commonly used tool for patients with primarily oropharyngeal (tonsillar and base-of-tongue) SCC (Figures 2 and 3). The first documented transoral robotic surgery (TORS) for OPC was reported in three patients with early-stage base-of-tongue SCC at the University of Pennsylvania (36). Soon after, multiple institutions began describing their experience with TORS for early-stage OPC and even nononcologic surgeries, including the use of TORS for obstructive sleep apnea. Several institutions have documented excellent oncologic outcomes in patients undergoing TORS as primary treatment for OPC (37–39).
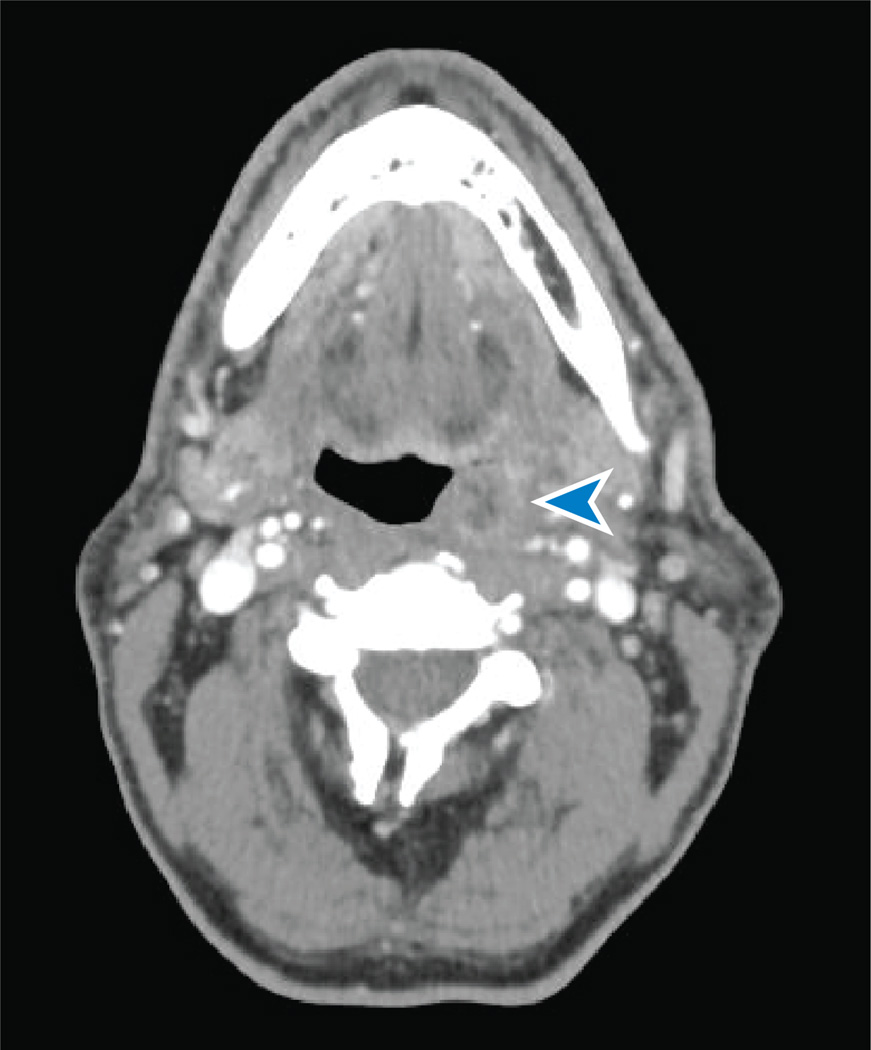
Figure 2.
Computed tomography scan with contrast demonstrating an HPV-positive tonsillar cancer (blue arrowhead) amenable to transoral robotic resection.
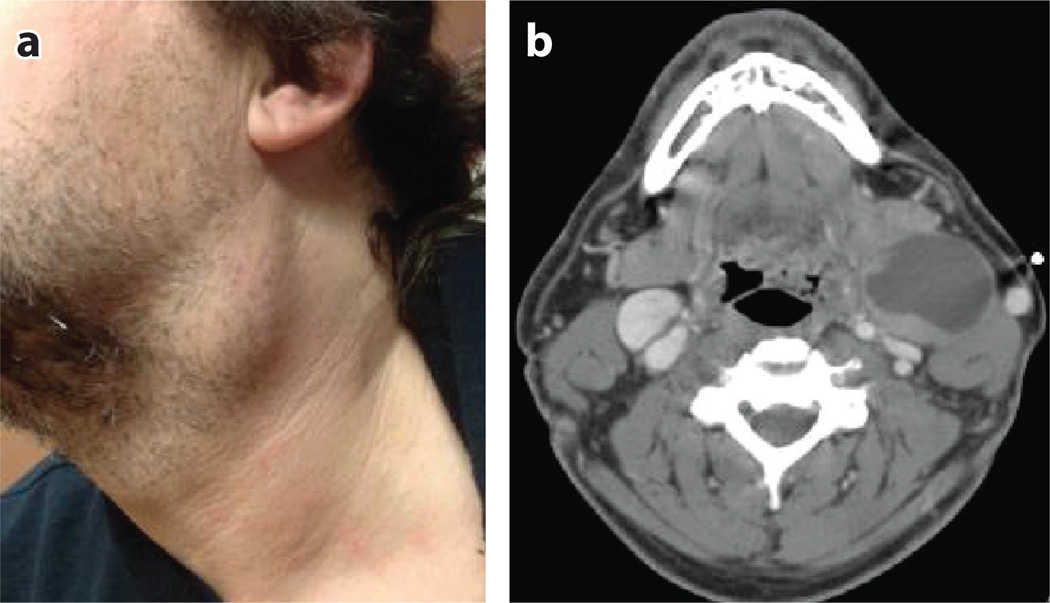
Figure 3.
(a) Left neck mass in a patient diagnosed with HPV-positive oropharyngeal cancer; (b) the computed tomography scan correlate demonstrating the cystic appearance of cervical lymph node metastases commonly seen in patients with HPV-positive oropharyngeal cancer.
Weinstein et al. (37) reported a series of 47 patients with stage III/IV oropharyngeal SCC treated with TORS. They demonstrated a disease-specific survival of 98% at one year and 90% at two years. Interestingly, 38% of these patients avoided adjuvant chemotherapy following TORS, and 11% did not require any adjuvant therapy. The same group of investigators went on to complete a prospective phase I single-arm study of 31 patients with oropharyngeal SCC who underwent TORS with selective neck dissection (37). They reported a 100% local disease control rate, with seven patients avoiding adjuvant therapy and 15 of the remaining 24 (62.5%) receiving radiation alone. None of the patients in this series were G-tube dependent at two years.
Patients with OPC treated with TORS have excellent functional outcomes when compared to primary nonsurgical modalities. Several institutions have demonstrated superior swallowing, diet tolerance, and overall quality of life for patients treated with TORS compared to those treated with chemoradiation (38, 40). Despite the superior outcomes of OPC patients, regardless of treatment modality, there remains a portion of HPV-positive patients at risk for local and regional failure, distant metastases, and poor survival.
Investigators have identified several risk factors for poor prognosis in HPV-positive patients, including smoking history and bulky disease (10, 41). Controlling for potential confounding variables, a recent Surveillance, Epidemiology and End Results (SEER) analysis of early-stage tonsillar cancer patients suggested a higher survival rate in those treated with primary surgery (tonsillectomy) (42). These results suggest that surgical reduction of tumor volume, even with a nononcologic resection, may improve outcomes and could potentially benefit high-risk OPC patients. Recent advances in minimally invasive surgery, including TORS and TLM, provide the opportunity to tailor treatment regimens to individual patients with minimal added morbidity.
RISK STRATIFICATION AMONG PATIENTS WITH OROPHARYNGEAL CANCER
Overall, HPV-positive OPC patients have a better prognosis than HPV-negative head and neck cancer patients. However, a few individuals with HPV-positive OPC develop locoregional recurrences or distant metastases and have poor survival. Clinicians are reluctant to deintensify standard treatment regimens for all HPV-positive OPC patients because some of these patients appear to have aggressive disease. Therefore, identifying which HPV-positive patients are at greater risk of death and recurrence is of utmost importance in treatment planning.
Ang and colleagues (10) stratified patients with OPC according to their risk of death. They identified smoking, HPV-negative status, and large primary tumors as risk factors for poor prognosis. They also demonstrated that half of the deaths (25/50) among HPV-positive patients within three years resulted from the primary cancer. In fact, of OPC patients who failed treatment, 31.8% developed distant metastases. O’Sullivan et al. (43) further investigated which HPV-positive patients were at risk for distant metastases. They retrospectively reviewed 505 OPC patients, the majority of whom were HPV-positive (n = 382). All patients were treated with either concurrent chemoradiation or radiation alone based on current institutional guidelines. They found that compared to HPV-negative patients, HPV-positive patients had better local (94% versus 80%) and regional (95% versus 82%) control; however, distant control was similar (90% versus 86%) (43).
O’Sullivan et al. also segregated OPC patients by risk of distant metastases based on recursive partitioning analysis. They found that HPV-positive patients with T1–3, N0–2c disease had high distant control rates of ~93%. However, HPV-positive patients with T4 or N3 disease had poor distant control rates of 72%, similar to their HPV-negative counterparts. Furthermore, HPV-positive patients with N2c disease who underwent chemotherapy in addition to their radiation regimen had better distant control than those who received radiation alone.
The retrospective stratification of HPV-positive patients into risk categories has led to deintensification trials for HPV-positive patients in the low- to intermediate-risk group, such as ECOG 31–11. In this trial, HPV-positive OPC patients are treated surgically and then stratified by pathological parameters into low-, intermediate-, and high-risk groups. Those stratified to the intermediate group include patients with close margins, 2–4metastatic lymph nodes, less than 1mm of extracapsular spread in the cervical lymph node metastases, perineural invasion, and lymphovascular invasion. This group of intermediate-risk patients are then randomized to standard-dose intensity-modulated radiation therapy (IMRT; 60 Gy over 30 fractions) or lower dose IMRT (50 Gy over 25 fractions). The high-risk group undergoes adjuvant chemoradiation, and the low-risk group is observed following surgery. Patients are monitored for at least two years after treatment and followed for locoregional recurrence and functional outcomes. Other deintensification trials for HPV-positive OPC patients are currently being developed.
HPV VACCINES, IMMUNOTHERAPY, AND BIG DATA
The development of preventative vaccines for HPV infection stemmed from the discovery of HPV as a necessary cause of some anogenital cancers, such as cervical cancer. It has since been discovered that immunization with HPV virus-like particles (VLPs) that mimic the L1 protein produce a humoral antibody response that is significantly greater than that produced by natural HPV infection (18). Currently, there are two commercially available HPV vaccines: a quadrivalent vaccine designed to protect against high-risk subtypes 16 and 18 and low-risk subtypes 6 and 11 (Gardasil®, Merck and Co., Whitehouse Station, NJ), and a bivalent vaccine designed to protect against high-risk subtypes 16 and 18 (Cervarix®, GlaxoSmithKline Biologicals, Rixensart, Belgium).
Several randomized double-blinded controlled trials involving thousands of young women have demonstrated impressive efficacy for the prevention of HPV-related diseases. These trials include the FUTURE I/FUTURE II trials (Gardasil), the PATRICIA trial (Cervarix), and the Costa Rica HPV Vaccine Trial (Cervarix). Both Gardasil and Cervarix were found to have 90.9–100% efficacy for preventing HPV-related benign and premalignant lesions of the cervix in young women who received all three vaccine doses. Similarly, a randomized double-blinded controlled trial of >4,000 young men in 18 countries showed that Gardasil had >90% efficacy for prevention of benign and premalignant HPV-related anogenital lesions when all three doses were given. These trials have also demonstrated excellent safety profiles (44), leading to worldwide public health initiatives to vaccinate preadolescent girls and boys.
Despite the success of preventative vaccines, therapies aimed at clearance of HPV infection or HPV-related lesions remain investigational. Topical toll-like receptor agonists have shown some efficacy for treatment of HPV-related genital lesions, although their use in HPV-related head and neck cancer has not yet been studied (45, 46). Immunotherapy delivery via viral vectors is also being investigated in head and neck cancer patients. A vaccinia virus used to express E6, E7, and human IL-2 (TG4001) has shown efficacy in treating premalignant lesions of the cervix (46). A phase II trial is evaluating the vaccinia virus in conjunction with chemoradiation in HPV-positive oropharyngeal cancer patients (46, 47). Similarly, the Listeria monocytogenes bacteria may also be used as a viral vector. Clinical trials are evaluating the use of Listeria-based HPV vaccines for patients with HPV-positive oropharyngeal and cervical cancer (46, 48).
There has been recent interest in immune checkpoints as the critical pathways by which HPV evades the host immune response. These checkpoints include programmed cell death protein 1 (PD-1) pathways and cytotoxic T lymphocyte antigen 4 (CTLA-4) pathways. PD-1 is expressed on many cell types and suppresses T cell responses to infection (49). HPV-positive OPC tissue has higher expression of PD-1 than benign tonsil tissue (50). Furthermore, Badoual and colleagues (51) found that levels of PD-1 expression on tumor-infiltrating T cells correlated with clinical outcome in HPV-positive OPC patients. These investigators suggested that high PD-1-expressing tumor-infiltrating T cells reflected a greater immune response to the tumor. Although elevated PD-1 expression is not unique to HPV-positive tumors, it remains a possible therapeutic strategy for patients with HPV-positive OPC.
Recent genome-wide association studies using next-generation sequencing techniques have elucidated the genomic landscape of HPV-positive cancers (52–56). Specifically, these studies have highlighted genomic alterations that have the potential to become therapeutic targets in the treatment of HPV-positive OPC. Furthermore, these studies have demonstrated that HPV integration into the host genome can drive carcinogenesis independent of enhanced expression of the viral oncogenes, E6 and E7 (54, 55). Several genetic mutations or alterations have been identified in patients with HPV-positive head and neck SCC. For instance, PIK3CA mutations have been found to be enriched in HPV-positive SCC (52, 53, 57). Other genes found to have recurrent mutations in patients with HPV-positive disease include TRAF3 (TNF receptor–associated factor 3) and TP63 (tumor protein p63) (52, 53). These genetic alterations, and many others, are potential therapeutic biomarkers, and further investigation into their clinical implication is warranted.
CONCLUSIONS
HPV is a recently identified causative agent for a subset of head and neck cancers, primarily in the oropharynx, and is largely responsible for the rising worldwide incidence of OPC. Patients with HPV-positive OPC tend to be younger, male, and less likely to smoke. Patients with HPV positive OPC generally have a better prognosis than patients with traditional, HPV-negative, head and neck cancer. Although the majority of HPV-positive OPC patients have excellent survival, a subset of these patients develop regional recurrences and distant metastases and ultimately fail standard treatment regimens. Risk factors for poor prognosis among HPV-positive OPC patients include a history of smoking, large bulky primary tumors, and N3 disease. However, further risk stratification among HPV-positive OPC patients is warranted, and clinical trials investigating both surgical and nonsurgical modalities are currently under way.
Footnotes
DISCLOSURE STATEMENT
The authors are not aware of any affiliations, memberships, funding, or financial holdings that might be perceived as affecting the objectivity of this review.
LITERATURE CITED
- 1.Bodily J, Laimins LA. Persistence of human papillomavirus infection: keys to malignant progression. Trends Microbiol. 2011;19:33–39. doi: 10.1016/j.tim.2010.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Munoz N, Bosch FX, de Sanjose S, et al. Epidemiologic classification of human papillomavirus types associated with cervical cancer. N. Engl. J. Med. 2003;348:518–527. doi: 10.1056/NEJMoa021641. [DOI] [PubMed] [Google Scholar]
- 3.Gillison ML, Koch WM, Capone RB, et al. Evidence for a causal association between human papillomavirus and a subset of head and neck cancers. J. Natl. Cancer Inst. 2000;92:709–720. doi: 10.1093/jnci/92.9.709. [DOI] [PubMed] [Google Scholar]
- 4.Maxwell JH, Kumar B, Feng FY, et al. HPV-positive/p16-positive/EBV-negative nasopharyngeal carcinoma in white North Americans. Head Neck. 2010;32:562–567. doi: 10.1002/hed.21216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ryerson AB, Peters ES, Coughlin SS, et al. Burden of potentially human papillomavirus-associated cancers of the oropharynx and oral cavity in the US, 1998–2003. Cancer. 2008;113:2901–2909. doi: 10.1002/cncr.23745. [DOI] [PubMed] [Google Scholar]
- 6.Conway DI, Brenner DR, McMahon AD, et al. Estimating and explaining the effect of education and income on head and neck cancer risk: INHANCE consortium pooled analysis of 31 case-control studies from 27 countries. Int. J. Cancer. 2015;136:1125–1139. doi: 10.1002/ijc.29063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chaturvedi AK, Engels EA, Pfeiffer RM, et al. Human papillomavirus and rising oropharyngeal cancer incidence in the United States. J. Clin. Oncol. 2011;29:4294–4301. doi: 10.1200/JCO.2011.36.4596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chenevert J, Chiosea S. Incidence of human papillomavirus in oropharyngeal squamous cell carcinomas: now and 50 years ago. Hum. Pathol. 2012;43:17–22. doi: 10.1016/j.humpath.2011.03.009. [DOI] [PubMed] [Google Scholar]
- 9.Maxwell JH, Ferris RL, Gooding W, et al. Extracapsular spread in head and neck carcinoma: impact of site and human papillomavirus status. Cancer. 2013;119:3302–3308. doi: 10.1002/cncr.28169. [DOI] [PubMed] [Google Scholar]
- 10.Ang KK, Harris J, Wheeler R, et al. Human papillomavirus and survival of patients with oropharyngeal cancer. N. Engl. J. Med. 2010;363:24–35. doi: 10.1056/NEJMoa0912217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fakhry C, Westra WH, Li S, et al. Improved survival of patients with human papillomavirus-positive head and neck squamous cell carcinoma in a prospective clinical trial. J. Natl. Cancer Inst. 2008;100:261–269. doi: 10.1093/jnci/djn011. [DOI] [PubMed] [Google Scholar]
- 12.D’Souza G, Kreimer AR, Viscidi R, et al. Case-control study of human papillomavirus and oropharyngeal cancer. N. Engl. J. Med. 2007;356:1944–1956. doi: 10.1056/NEJMoa065497. [DOI] [PubMed] [Google Scholar]
- 13.Bajos N, Bozon M, Beltzer N, et al. Changes in sexual behaviours: from secular trends to public health policies. AIDS. 2010;24:1185–1191. doi: 10.1097/QAD.0b013e328336ad52. [DOI] [PubMed] [Google Scholar]
- 14.Turner CF, Danella RD, Rogers SM. Sexual behavior in the United States 1930–1990: trends and methodological problems. Sex. Transm. Dis. 1995;22:173–190. doi: 10.1097/00007435-199505000-00009. [DOI] [PubMed] [Google Scholar]
- 15.D’Souza G, Cullen K, Bowie J, et al. Differences in oral sexual behaviors by gender, age, and race explain observed differences in prevalence of oral human papillomavirus infection. PLoS ONE. 2014;9:e86023. doi: 10.1371/journal.pone.0086023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Thorland EC, Myers SL, Gostout BS, Smith DI. Common fragile sites are preferential targets for HPV16 integrations in cervical tumors. Oncogene. 2003;22:1225–1237. doi: 10.1038/sj.onc.1206170. [DOI] [PubMed] [Google Scholar]
- 17.O’Brien PM, Saveria Campo M. Evasion of host immunity directed by papillomavirus-encoded proteins. Virus Res. 2002;88:103–117. doi: 10.1016/s0168-1702(02)00123-5. [DOI] [PubMed] [Google Scholar]
- 18.Stanley MA, Pett MR, Coleman N. HPV: from infection to cancer. Biochem. Soc. Trans. 2007;35:1456–1460. doi: 10.1042/BST0351456. [DOI] [PubMed] [Google Scholar]
- 19.Kanodia S, Fahey LM, Kast WM. Mechanisms used by human papillomaviruses to escape the host immune response. Curr. Cancer Drug Targets. 2007;7:79–89. doi: 10.2174/156800907780006869. [DOI] [PubMed] [Google Scholar]
- 20.Scott M, Nakagawa M, Moscicki AB. Cell-mediated immune response to human papillomavirus infection. Clin. Diagn. Lab. Immunol. 2001;8:209–220. doi: 10.1128/CDLI.8.2.209-220.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Beachler DC, Viscidi R, Sugar EA, et al. A longitudinal study of human papillomavirus 16 L1, E6, and E7 seropositivity and oral human papillomavirus 16 infection. Sex. Transm. Dis. 2015;42:93–97. doi: 10.1097/OLQ.0000000000000236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gillison ML, Broutian T, Pickard RK, et al. Prevalence of oral HPV infection in the United States, 2009–2010. JAMA. 2012;307:693–703. doi: 10.1001/jama.2012.101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ragin CC, Taioli E. Survival of squamous cell carcinoma of the head and neck in relation to human papillomavirus infection: review and meta-analysis. Int. J. Cancer. 2007;121:1813–1820. doi: 10.1002/ijc.22851. [DOI] [PubMed] [Google Scholar]
- 24.Parsons JT, Mendenhall WM, Stringer SP, et al. Squamous cell carcinoma of the oropharynx: surgery, radiation therapy, or both. Cancer. 2002;94:2967–2980. doi: 10.1002/cncr.10567. [DOI] [PubMed] [Google Scholar]
- 25.Bourhis J, Overgaard J, Audry H, et al. Hyperfractionated or accelerated radiotherapy in head and neck cancer: a meta-analysis. Lancet. 2006;368:843–854. doi: 10.1016/S0140-6736(06)69121-6. [DOI] [PubMed] [Google Scholar]
- 26.Forastiere AA, Zhang Q, Weber RS, et al. Long-term results of RTOG91–11: a comparison of three nonsurgical treatment strategies to preserve the larynx in patients with locally advanced larynx cancer. J. Clin. Oncol. 2013;31:845–852. doi: 10.1200/JCO.2012.43.6097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Calais G, Alfonsi M, Bardet E, et al. Randomized trial of radiation therapy versus concomitant chemotherapy and radiation therapy for advanced-stage oropharynx carcinoma. J. Natl. Cancer Inst. 1999;91:2081–2086. doi: 10.1093/jnci/91.24.2081. [DOI] [PubMed] [Google Scholar]
- 28.Fischer CA, Zlobec I, Green E, et al. Is the improved prognosis of p16 positive oropharyngeal squamous cell carcinoma dependent of [sic] the treatment modality? Int. J. Cancer. 2010;126:1256–1262. doi: 10.1002/ijc.24842. [DOI] [PubMed] [Google Scholar]
- 29.Straetmans JM, Olthof N, Mooren JJ, et al. Human papillomavirus reduces the prognostic value of nodal involvement in tonsillar squamous cell carcinomas. Laryngoscope. 2009;119:1951–1957. doi: 10.1002/lary.20593. [DOI] [PubMed] [Google Scholar]
- 30.Ward MJ, Mellows T, Harris S, et al. Staging and treatment of oropharyngeal cancer in the human papillomavirus era. Head Neck. 2015;37:1002–1013. doi: 10.1002/hed.23697. [DOI] [PubMed] [Google Scholar]
- 31.Spector ME, Gallagher KK, Bellile E, et al. Patterns of nodal metastasis and prognosis in human papillomavirus-positive oropharyngeal squamous cell carcinoma. Head Neck. 2014;36:1233–1240. doi: 10.1002/hed.23438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Strong MS, Jako GJ. Laser surgery in the larynx. Early clinical experience with continuous CO2 laser. Ann. Otol. Rhinol. Laryngol. 1972;81:791–798. doi: 10.1177/000348947208100606. [DOI] [PubMed] [Google Scholar]
- 33.Haughey BH, Hinni ML, Salassa JR, et al. Transoral laser microsurgery as primary treatment for advanced-stage oropharyngeal cancer: a United States multicenter study. Head Neck. 2011;33:1683–1694. doi: 10.1002/hed.21669. [DOI] [PubMed] [Google Scholar]
- 34.Grant DG, Hinni ML, Salassa JR, et al. Oropharyngeal cancer: a case for single modality treatment with transoral laser microsurgery. Otolaryngol.–Head Neck Surg. 2009;135:1225–1230. doi: 10.1001/archoto.2009.185. [DOI] [PubMed] [Google Scholar]
- 35.Huang K, Xia P, Chuang C, et al. Intensity-modulated chemoradiation for treatment of stage III and IV oropharyngeal carcinoma: the University of California–San Francisco experience. Cancer. 2008;113:497–507. doi: 10.1002/cncr.23578. [DOI] [PubMed] [Google Scholar]
- 36.O’Malley BW, Jr, Weinstein GS, Snyder W, Hockstein NG. Transoral robotic surgery (TORS) for base of tongue neoplasms. Laryngoscope. 2006;116:1465–1472. doi: 10.1097/01.mlg.0000227184.90514.1a. [DOI] [PubMed] [Google Scholar]
- 37.Weinstein GS, O’Malley BW, Jr, Cohen MA, Quon H. Transoral robotic surgery for advanced oropharyngeal carcinoma. Otolaryngol.–Head Neck Surg. 2010;136:1079–1085. doi: 10.1001/archoto.2010.191. [DOI] [PubMed] [Google Scholar]
- 38.Genden EM, Desai S, Sung CK. Transoral robotic surgery for the management of head and neck cancer: a preliminary experience. Head Neck. 2009;31:283–289. doi: 10.1002/hed.20972. [DOI] [PubMed] [Google Scholar]
- 39.Hurtuk A, Agrawal A, Old M, et al. Outcomes of transoral robotic surgery: a preliminary clinical experience. Otolaryngol.–Head Neck Surg. 2011;145:248–253. doi: 10.1177/0194599811402172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Maxwell JH, Mehta V, Wang H, et al. Quality of life in head and neck cancer patients: impact of HPV and primary treatment modality. Laryngoscope. 2014;124:1592–1597. doi: 10.1002/lary.24508. [DOI] [PubMed] [Google Scholar]
- 41.Maxwell JH, Kumar B, Feng FY, et al. Tobacco use in human papillomavirus-positive advanced oropharynx cancer patients related to increased risk of distant metastases and tumor recurrence. Clin. Cancer Res. 2010;16:1226–1235. doi: 10.1158/1078-0432.CCR-09-2350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Holliday MA, Tavaluc R, Zhuang T, et al. Oncologic benefit of tonsillectomy in stage I and II tonsil cancer: a surveillance epidemiology and end results database review. Otolaryngol.–Head Neck Surg. 2013;139:362–366. doi: 10.1001/jamaoto.2013.107. [DOI] [PubMed] [Google Scholar]
- 43.O’Sullivan B, Huang SH, Siu LL, et al. Deintensification candidate subgroups in human papillomavirus-related oropharyngeal cancer according to minimal risk of distant metastasis. J. Clin. Oncol. 2013;31:543–550. doi: 10.1200/JCO.2012.44.0164. [DOI] [PubMed] [Google Scholar]
- 44.Schiller JT, Castellsague X, Garland SM. A review of clinical trials of human papillomavirus prophylactic vaccines. Vaccine. 2012;30(Suppl. 5):F123–F138. doi: 10.1016/j.vaccine.2012.04.108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Gildener-Leapman N, Lee J, Ferris RL. Tailored immunotherapy for HPV positive head and neck squamous cell cancer. Oral Oncol. 2014;50:780–784. doi: 10.1016/j.oraloncology.2013.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Gildener-Leapman N, Ferris RL, Bauman JE. Promising systemic immunotherapies in head and neck squamous cell carcinoma. Oral Oncol. 2013;49:1089–1096. doi: 10.1016/j.oraloncology.2013.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Brun JL, Dalstein V, Leveque J, et al. Regression of high-grade cervical intraepithelial neoplasia with TG4001 targeted immunotherapy. Am. J. Obstet. Gynecol. 2011;204:169.e1–169.e8. doi: 10.1016/j.ajog.2010.09.020. [DOI] [PubMed] [Google Scholar]
- 48.Sewell DA, Pan ZK, Paterson Y. Listeria-based HPV-16 E7 vaccines limit autochthonous tumor growth in a transgenic mouse model for HPV-16 transformed tumors. Vaccine. 2008;26:5315–5320. doi: 10.1016/j.vaccine.2008.07.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Quezada S, Peggs K. Exploiting CTLA-4, PD-1 and PD-L1 to reactivate the host immune response against cancer. Br. J. Cancer. 2013;108:1560–1565. doi: 10.1038/bjc.2013.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Lyford-Pike S, Peng S, Young G, et al. Evidence for a role of the PD-1:PD-L1 pathway in immune resistance of HPV-associated head and neck squamous cell carcinoma. Cancer Res. 2013;73:1733–1741. doi: 10.1158/0008-5472.CAN-12-2384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Badoual C, Hans S, Merillon N, et al. PD-1-expressing tumor infiltrating T cells are a favorable prognostic biomarker in HPV-associated head and neck cancer. Cancer Res. 2013;73:128–138. doi: 10.1158/0008-5472.CAN-12-2606. [DOI] [PubMed] [Google Scholar]
- 52.Cancer Genome Atlas Network. Comprehensive genomic characterization of head and neck squamous cell carcinomas. Nature. 2015;517:576–582. doi: 10.1038/nature14129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Seiwert TY, Zuo Z, Keck MK, et al. Integrative and comparative genomic analysis of HPV-positive and HPV-negative head and neck squamous cell carcinomas. Clin. Cancer Res. 2015;21:632–641. doi: 10.1158/1078-0432.CCR-13-3310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Parfenov M, Pedamallu CS, Gehlenborg N, et al. Characterization of HPV and host genome interactions in primary head and neck cancers. PNAS. 2014;111:15544–15549. doi: 10.1073/pnas.1416074111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Akagi K, Li J, Broutian TR, et al. Genome-wide analysis of HPV integration in human cancers reveals recurrent, focal genomic instability. Genome Res. 2014;24:185–199. doi: 10.1101/gr.164806.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Ojesina AI, Lichtenstein L, Freeman SS, et al. Landscape of genomic alterations in cervical carcinomas. Nature. 2014;506:371–375. doi: 10.1038/nature12881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Rusan M, Li YY, Hammerman PS. Genomic landscape of human papillomavirus-associated cancers. Clin. Cancer Res. 2015;21:2009–2019. doi: 10.1158/1078-0432.CCR-14-1101. [DOI] [PMC free article] [PubMed] [Google Scholar]