Abstract
Vitamin D is an essential fat-soluble vitamin with multiple functions. Vitamin D receptor has been shown to be expressed in several types of immune cells suggesting vitamin D may have immune regulatory roles. Vitamin D insufficiency has been suggested to increase the risk of autoimmune diseases. However, little is known regarding its immunomodulatory effects in the condition of immune suppression. The aim of the present study was to investigate the regulatory effects of vitamin D on immune function in immunosuppressant mice. An immunosuppressant mouse model was induced by intraperitoneal injection with glucocorticiod for 3 days. Immunosuppressant mice were intragastrically administered with 1,25-dihydroxy-vitamin D3 [1,25(OH)2D3; 0,4, 6 or 10 IU/g body weight] for 7 days. On day 8, the mice were decapitated. The body weight and the weights of thymus and spleen were measured. Thymus and spleen indexes were calculated. The ratio of CD4+/CD8+ T lymphocytes in the peripheral blood, proliferation and interleukin-2 (IL-2) production of spleen T lymphocytes was detected. Compared with the mice in the control group, the body weight, thymus and spleen indexes, the ratios of CD4+/CD8+ in peripheral blood and IL-2 production and proliferation of spleen T lymphocytes were decreased in immunosuppressant mice induced by glucocorticiod. However, in vitamin D-treated mice, the thymus indexes, the ratios of CD4+/CD8+, secretion of IL-2 and the proliferation index of spleen T lymphocytes were significantly increased (P<0.05). Among the three doses of 1,25(OH)2D3, 6 IU/g was most effective in improving the immune function. These results indicate that vitamin D supplementation can improve immune recovery in immunosuppressant mice by stimulating T-cell proliferation and elevating IL-2 production.
Keywords: vitamin D, immunity, T lymphocytes, interleukin-2
Introduction
Vitamin D is an essential fat-soluble vitamin with multiple functions. The main source of vitamin D is transformed from 7-dehydrocholesterol after exploration with ultraviolet irradiation and sequential hydroxylation into 25(OH)D and an active hormone, 1,25-dihydroxy-vitamin D3 [1,25(OH)2D3], by hydroxylases in the kidney and liver. It can also be absorbed from dietary intake or oral supplements. It has been shown that vitamin D receptor (VDR) is highly expressed in the intestine, kidney, thyroid and bone (1). Previous findings showed that several types of immune cells such as T lymphocytes, monocytes, macrophages, and dendritic cells express VDR as well. The active form of vitamin D exerts its effects on these tissues by binding to VDR. In addition, some studies present that macrophages, dendritic cells and lymphocytes also express vitamin D activating enzyme, 1-α-hydroxylase (CYP27B1) (2,3). Therefore, except for the classical physiological function of regulation of calcium and bone metabolism, vitamin D may also have immunomodulatory effects.
Epidemiologic evidence indicates that vitamin D deficiency is related to autoimmune diseases including multiple sclerosis, type 1 diabetes, systemic lupus erythematosus (SLE) and inflammatory bowel disease (4). Studies have found that the levels of serum 25(OH)D and 1,25(OH)2D3 are significantly lower in patients with multiple sclerosis than those in healthy controls (5,6). Additionally, the effect of vitamin D on type 1 diabetes mellitus is closely correlated with the serum concentration of 25(OH)D. An epidemiological investigation in Britain by Staples et al proves that the prevalence of type 1 diabetes is positively associated with the increase of residential latitude, and inversely associated with ultraviolet radiation, which is consistent with a previous report for multiple sclerosis in Australia (7). Additionally, the percentage of SLE has a close relationship with the serum concentration of 25(OH)D. In the study of Ruiz-Irastorza et al, the percentage of vitamin D deficiency in patients with SLE was higher than that in the healthy population, and approximately 70% of SLE patients had a serum concentration of 25(OH)D<30 ng/ml (8).
In addition to epidemiological data, animal experiments have also presented relationships between vitamin D and autoimmune diseases. In the study of Ye et al, the percentage of CD4+ and CD8+ T lymphocytes in guinea pigs with experimental allergic encephalomyelitis was decreased and the CD4+/CD8+ ratio was increased. However, when guinea pigs were supplemented with 1,25(OH)2D3, the percentage of CD4+ and CD8+ T lymphocyte were significantly elevated and simultaneously, CD4+/CD8+ was reduced (9). Another study showed that the immunological rejection of corneal allograft was inhibited by 1,25(OH)2D3 (10). Furthermore, 1,25(OH)2D3 also inhibited the expression of inflammatory related cytokines, such as interferon-γ, interleukin-2 (IL-2) and interleukin-17A (10).
However, little is known about the immunomodulatory effects of vitamin D in the condition of immune suppression. Vitamin D deficiency increases the susceptibility and vulnerability to tuberculosis. The immune response of patients with type 2 diabetes and spinal tuberculosis who receive long-term drug therapy can be improved by supplementation with 1,25(OH)2D3 (11,12). Those studies suggested that vitamin D may enhance the immune function under immunocompromised condition. The aim of the present study was to investigate the effects of vitamin D on immune function in immunosuppressant mice induced by glucocorticoid.
Materials and methods
Reagents
Concanavalin A (Con A), 1,25(OH)2D3 and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide (MTT) were purchased from Sigma (St. Louis, MO, USA). A mouse enzyme-linked immunosorbent assay (ELISA) kit for IL-2 was purchased from R&D Systems, Inc., (Minneapolis, MN, USA). Fluorescence-labeled anti-mouse antibody was purchased from eBioscience, Inc. (San Diego, CA, USA).
Animal studies
A total of 40 ICR male mice (6–8 weeks old) were purchased from the Animal Center of the Medical Laboratory in Peking University Health Science Center. Mice were randomly divided into five groups (8 in each group) including the control group (C group), immunosuppression model group (M group) and three different doses of 1,25(OH)2D3 supplemented groups. All the mice except those in the C group were injected intraperitoneally with dexamethasone (DEX, 25 mg/kg) for three days to establish the immunosuppressant mouse model. Mice in the C group were injected with the same volume of normal saline. From the first day of DEX injection, 1,25(OH)2D3 at three different doses of 4 IU/g (+4D group), 6 IU/g (+6D group) or 10 IU/g (+10D group) body weight was given for 7 days by intragastric administration to mice in vitamin D-treated groups. Mice in the C and M groups received the same volume of physiological saline via intragastric administration.
All the mice were weighed and sacrificed by cervical dislocation at day 8. Spleens and thymuses were collected and weighed. Indexes for thymus and spleen were calculated according to the formulae: Spleen index (mg/g) = weight of spleen (mg)/body weight (g) and thymus index = weight of thymuses (mg)/body weight (g). Animal experiments performed in the study were approved by the Ethics Committee (ref no. LUNSHEN 2014012) of the 306th Hospital of PLA (Beijing, China).
Proliferation assay of splenocytes
Cell proliferation was assessed by MTT assay. The splenocyte suspension was obtained by grinding the spleen tissue in RPMI-1640 and passing tissue through a fine-mesh cell strainer. Erythrocytes were removed by hemolytic solution. Cells were washed, centrifuged and suspended in RPMI-1640 with 10% fetal calf serum (FCS). Cells were counted, and resuspended in culture medium at a concentration of 1×107 cells/ml. Cells (100 µl/well) were seeded in 96-well plates. Culture medium (100 µl/well) containing Con A (10 µg/ml) or without Con A (as control) was added to each well and cultured for 48 h. Cell proliferation was detected by MTT assay. MTT reagent (20 µl, 5 mg/ml) was added to each well and cultured for an additional 4 h. Culture medium was discarded and 100 µl dmethyl sulfoxide was added to each well. Optical density (OD) value was read at 570 nm using a spectrophotometer (Bio-Rad Laboratories, Hercules, CA, USA). Stimulated proliferation index was calculated according to the formula: Proliferation index = ODConA/ODcontrol.
Quantification for IL-2 production
The concentration of IL-2 in cultures of splenocytes was determined using a mouse ELISA kit. Cells from spleen were prepared as described in the proliferation assay. Cells (107/ml; 350 µl/well) were plated on 24-well plates and 350 µl/well Con A (10 µg/ml) was added to each well and cultured for 48 h. The cell culture medium was collected. The concentration of IL-2 in cell culture medium was detected by ELISA following the manufacturer's instructions.
Flow cytometric analysis of cell markers of CD4 and CD8
To determine the ratio of CD4+ and CD8+ T lymphocytes (CD4+/CD8+) in peripheral blood, the anticoagulated blood was collected from all the mice and treated with haemolysin. Cells were washed and cell suspension was prepared for immunofluorescent staining. Fluorescein-labeled rat anti-mouse monoclonal antibodies (1:200 diluted anti-CD4 FITC, cat. no. 11-0041; and 1:200 diluted anti-CD8 PE, cat. no. 12-0081) were added to the cells and incubated for 30 min in dark. The cells were washed and analyzed by FACSCalibur and Cell Quest software (BD Biosciences, Franklin Lakes, NJ, USA).
Statistical analysis
Statistical analysis was performed using Statistical Product and Service Solutions (SPSS, Armonk, NY, USA) software version 16.0. Data were expressed as mean ± standard deviation (mean ± SD). One-way ANOVA analysis was carried out to compare means variability among all the groups. P<0.05 was considered to indicate a statistically significant difference.
Results
1,25(OH)2D3 improves immune organ recovery
As shown in Table I, the thymus and spleen indexes in mice in the M group were significantly lower than those in mice in the C group. When mice were given 1,25(OH)2D3 for 7 days, the thymus indexes increased compared with the mice in the M group (P<0.05). Among the three vitamin D-treated groups, the recovery of the thymus index in mice of the +6D group was the most significant. However, for the spleen indexes, there was no difference between vitamin D-treated mice and the mice in M group (Table I).
Table I.
The effect of 1,25(OH)2D3 on thymus index and spleen indexes (mean ± SD).
| Groups | Thymus (mg) | Spleen (mg) | Thymus index (mg/g) | Spleen index (mg/g) |
|---|---|---|---|---|
| C | 73.38±11.53 | 109.09±22.42 | 2.83±0.42 | 4.20±0.73 |
| M | 32.43±8.07a | 79.08±17.09 | 1.23±0.30a | 3.01±0.62a |
| +4D | 41.79±6.27a | 78.48±17.75 | 1.77±0.21a,b | 3.3±0.72 |
| +6D | 66.99±15.36b | 78.06±13.41 | 2.58±0.62b | 2.99±0.46a |
| +10D | 55.75±15.95b | 76.99±10.3a | 2.11±0.53a,b | 2.93±0.37a |
P<0.05 vs. the C group
P<0.05 vs. the M group. SD, standard deviation.
1,25(OH)2D3 promotes proliferation of lymphocytes in spleen
We evaluated the effects of 1,25(OH)2D3 on the proliferation of T lymphocytes in spleen. As shown in Fig. 1, the proliferation index of lymphocytes was obviously inhibited by the glucocorticoid hormone. The proliferation index was much lower in the M group (1.5±0.35) compared with the C group (7.49±2.16, P=0.01). However, the proliferation index was higher in the +6D group (2.55±0.66) compared to the M group (1.50±0.35, P=0.021). Differences of proliferation indexes of mice were not significant between M group and the other two 1,25(OH)2D3-treated groups.
Figure 1.
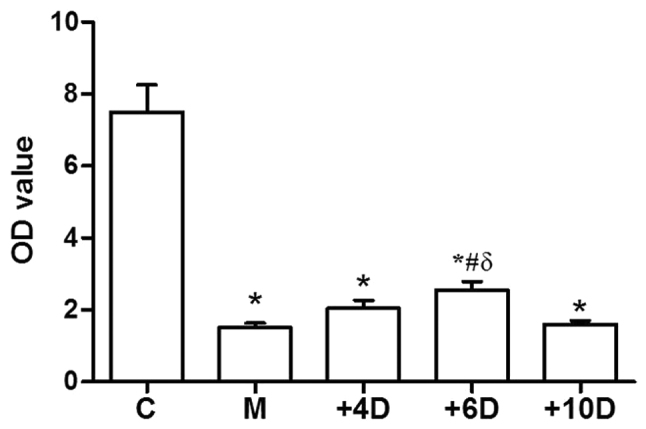
Effect of 1,25(OH)2D3 on proliferation index of spleen lymphocytes. *P<0.05 vs. C group; #P<0.05 vs. M group; δP<0.05 vs. +4D group. 1,25(OH)2D3, 1,25-dihydroxy-vitamin D3.
1,25(OH)2D3 promotes the production of IL-2 in spleen lymphocytes
To investigate the effects of 1,25(OH)2D3 on the function of T lymphocytes in spleen, the production of IL-2 in cultures of lymphocytes was detected. As expected, the secretion of IL-2 in spleen lymphocytes was significantly inhibited by glucocorticoid hormone injection in mice of the M group (P<0.01). However, mice supplemented with 1,25(OH)2D3 (6 IU/g) had much higher levels of IL-2 (0.58±0.22mg/l) compared to the M group (0.35±0.10mg/l, P=0.03). The level of IL-2 in mice of the +6D group was similar to that in the control group. However, IL-2 production of spleen lymphocytes in mice of the +4D and +10D groups was not different from that in mice of the M group (Fig. 2).
Figure 2.
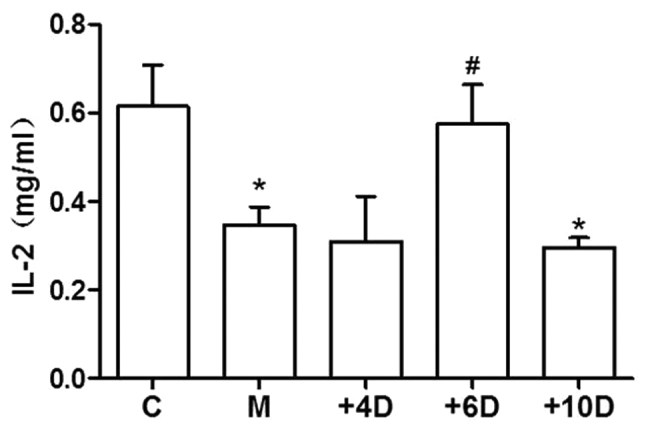
Effect of 1,25(OH)2D3 on IL-2 production in spleen lymphocytes.
1,25(OH)2D3 upregulates the ratio of CD4+ to CD8+ T lymphocytes
To investigate the effects of 1,25(OH)2D3 on the immune functions, we examined the ratio of CD4+ to CD8+ T cells in peripheral blood. As the data show in Fig. 3, the CD4+/CD8+ ratio in the M group (1.20±0.24) was significantly decreased compared to that in the C group (3.18±1.04; P<0.05). The ratios of CD4+/CD8+ T cells were 1.58±0.49, 1.74±0.35 and 1.75±0.57 in the +4D, +6D and +10D groups, respectively. The ratio of CD4+/CD8+ T cells was significantly higher in mice treated with 1,25(OH)2D3 at doses of 6 and 10 IU/g body weights compared with that in mice of the M group (Fig. 3).
Figure 3.
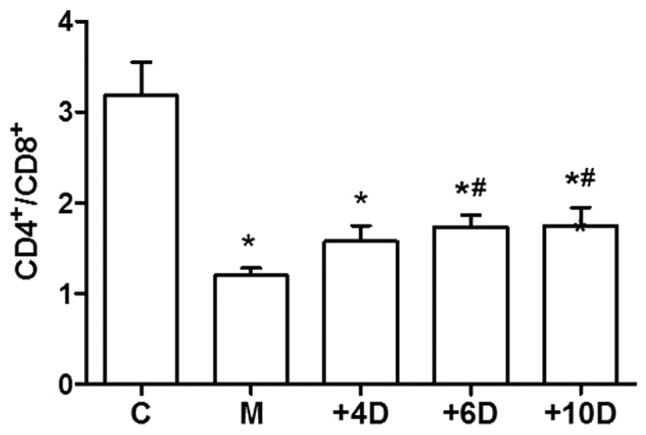
Effect of 1,25(OH)2D3 on the ratio of CD4+ to CD8+ T cells (mean ± SD). *P<0.05 vs. C group; #P<0.05 vs. M group. 1,25(OH)2D3, 1,25-dihydroxy-vitamin D3. SD, standard deviation.
Discussion
Previous reports regarding the effects of 1,25(OH)2D3 on the regulation of immune function were inconsistent. Most evidence of epidemiological studies, animal and in vitro experiments suggest a role for 1,25(OH)2D3 in negatively influencing the T cells in autoimmune disorders (9,13). The results of an in vivo study suggest that 1,25(OH)2D3 supplementation inhibits the proliferation of T lymphocytes, reduces the immune organ indexes and decreases the ratio of CD4+/CD8+ T cells in a model of adjuvant arthritis (14). In mice stimulated with Bacillus Calmette-Guerin, vitamin D deficiency may result in increasing the percentage of CD4+ and CD8+ T cells and reducing the CD4+/CD8+ ratio, while 1,25(OH)2D3 supplementation leads to a reduction of CD4+ and CD8+ T lymphocytes and elevation of CD4+/CD8+ ratio. These results suggest that vitamin D exerts immunosuppressive effects in autoimmune diseases. By contrast, data from a clinical study show that the immunity of patients with long-term type 2 diabetes may be improved by vitamin D supplementation (11). Furthermore, the study of Gao et al suggests that the concentrations of 25(OH)D in patients with tuberculosis is lower than that in healthy adults, and 25(OH)D has a positive correlation with the CD4+/CD8+ ratio (15). A study by Panda et al, an animal model of 1-α hydroxylase deficiency was used to investigate the effects of 1,25(OH)2D3 on immune function. Their result showed that vitamin D deficiency resulted in the inhibition of the proliferation of spleen lymphocyte and a reduction in CD4+ and CD8+ peripheral T cells (16). The results presented in our study showed that 1,25(OH)2D3 may improve the thymus index recovery, promote the IL-2 production and proliferation of T cells and elevate CD4+/CD8+ ratio in glucocorticoid-induced immunosuppressant mice. The data indicates that 1,25(OH)2D3 has a positive influence on immune function under the state of hypoimmunity. The immunomodulatory effects of 1,25(OH)2D3 may be dependent on the immune status.
How vitamin D affects the immune system has not been fully elucidated. Previous studies have demonstrated that T cells express VDR (17). In addition, activated T cells express Cyp27B1 (vitamin D activating enzyme) (18,19). Those studies suggest that T cells are not only the targets of 1,25(OH)2D3 but are also able to produce 1,25(OH)2D3 locally. IL-2, secreted by type 1 helper T cells, mediates the cellular immune response and can induce the proliferation of T, B and natural killer cells (20). IL-2 may participate in the regulatory effects of vitamin D on immune system. A study showed that IL-2 secreted by CD4+ T cells was enhanced by 1,25(OH)2D3 treatment (21). Additionally, in vitro treatment with 1,25(OH)2D3 may upregulate the expression of IL-2 receptor in T lymphocytes and monocytes (22). The current study showed that vitamin D enhanced IL-2 production of T cells in spleen. Thus, the changes of IL-2 production may participate in the immune regulatory effects of vitamin D.
T cells mainly include CD4+ T cells and CD8+ T cells. The CD4+/CD8+ ratio has been recognized as an important indicator for evaluating the state of immunomodulation and response to homeostasis of the intrinsic immune system (23). Previous studies have shown that the increased CD4+/CD8+ ratio has been observed in many autoimmune diseases such as SLE (24), inflammatory bowel disease (25) and multiple sclerosis (26). A low CD4+/CD8+ ratio was considered as a marker of decreased immune function (27), which could result in increasing the risk of HIV-infected, pulmonary tuberculosis and even some tumors (28,29). The current study demonstrated that the CD4/CD8 ratio significantly decreased by glucocorticoid injection and partially recovered by vitamin D supplementation. These results further prove that vitamin D may improve the immune status in immunosuppressant conditions.
The present findings have demonstrated that vitamin D can partially improve the immune recovery in immunosuppressant mice by stimulating T-cell proliferation and elevating IL-2 production.
Acknowledgements
The present study was supported by The Medical Research Grant of The 306th Hospital of PLA, Beijing, China.
Glossary
Abbreviations
- OH
hydroxide
- 1,25(OH)2D3
1,25-dihydroxy vitamin D3
- Con A
concanavalin A
- MTT
3-(4,5-dimethyl thiazol-2-yl)-2,5-diphenyl-tetrazolium bromide
- IL-2
interleukin-2
- ELISA
enzyme-linked immunosorbent assay
- DEX
dexamethasone
- FCS
fetal calf serum
- OD
optical density
References
- 1.Wang Y, Zhu J, DeLuca HF. Where is the vitamin D receptor? Arch Biochem Biophys. 2012 Jul 1;523(1):123–133. doi: 10.1016/j.abb.2012.04.001. [DOI] [PubMed] [Google Scholar]
- 2.Overbergh L, Decallonne B, Valckx D, Verstuyf A, Depovere J, Laureys J, Rutgeerts O, Saint-Arnaud R, Bouillon R, Mathieu C. Identification and immune regulation of 25-hydroxyvitamin D-1-alpha-hydroxylase in murine macrophages. Clin Exp Immunol. 2000;120:139–146. doi: 10.1046/j.1365-2249.2000.01204.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hewison M, Freeman L, Hughes SV, Evans KN, Bland R, Eliopoulos AG, Kilby MD, Moss PA, Chakraverty R. Differential regulation of vitamin D receptor and its ligand in human monocyte-derived dendritic cells. J Immunol. 2003;170:5382–5390. doi: 10.4049/jimmunol.170.11.5382. [DOI] [PubMed] [Google Scholar]
- 4.Prietl B, Treiber G, Pieber TR, Amrein K. Vitamin D and immune function. Nutrients. 2013;5:2502–2521. doi: 10.3390/nu5072502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Soilu-Hänninen M, Airas L, Mononen I, Heikkilä A, Viljanen M, Hänninen A. 25-Hydroxyvitamin D levels in serum at the onset of multiple sclerosis. Mult Scler. 2005;11:266–271. doi: 10.1191/1352458505ms1157oa. [DOI] [PubMed] [Google Scholar]
- 6.Correale J, Ysrraelit MC, Gaitán MI. Immunomodulatory effects of vitamin D in multiple sclerosis. Brain. 2009;132:1146–1160. doi: 10.1093/brain/awp033. [DOI] [PubMed] [Google Scholar]
- 7.Staples JA, Ponsonby AL, Lim LL, McMichael AJ. Ecologic analysis of some immune-related disorders, including type 1 diabetes, in Australia: Latitude, regional ultraviolet radiation, and disease prevalence. Environ Health Perspect. 2003;111:518–523. doi: 10.1289/ehp.5941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ruiz-Irastorza G, Egurbide MV, Olivares N, Martinez-Berriotxoa A, Aguirre C. Vitamin D deficiency in systemic lupus erythematosus: Prevalence, predictors and clinical consequences. Rheumatology (Oxford) 2008;47:920–923. doi: 10.1093/rheumatology/ken121. [DOI] [PubMed] [Google Scholar]
- 9.Ye YL, Li ZX, Ye XM. Influence of 1, 25-dihydroxyvitamin D3 on variation of peripheral blood lymphocyte subsets in guinea pigs with experimental autoimmune encephalomyelitis. J Chin Gen Pract. 2008;11:391–393. (In Chinese) [Google Scholar]
- 10.Wu J, Zhang J, Hou GH, Cui YB, Wang C, Chen J. Effect of 1α,25-dihydroxyvitamin D3 on T helper cell 17 and expression of related cytokines in penetrating keratoplasty in mice. Chin J Pathophysiol. 2014;30:2226–2231. (In Chinese) [Google Scholar]
- 11.Luwen C, Liqing T, Ruiping Y. Study of impact of vitamin D in immune function and infections in patients with type 2 diabetes. Chin J Nosocomiol. 2015;25:1106–1109. [Google Scholar]
- 12.Dezhi L, Zehua Z, Litao L, Zhengqi C. Effection of vitamin D deficiency to T lymphocyte subgroups in spinal tuberculosis Chin. J Orthop. 2012;20:448–450. [Google Scholar]
- 13.Cantorna MT, Snyder L, Lin YD, Yang L. Vitamin D and 1,25(OH)2D regulation of T cells. Nutrients. 2015;7:3011–3021. doi: 10.3390/nu7043011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wang C, Wu J, Hou GH, Chen J, Qi WJ, Cui YB, Zhang J. The immuno-regulation effect of 1,25(OH)2D3 on T lymphocytes. J Guangdong Med. 2013;34:3114–3116. [Google Scholar]
- 15.Gao WW, Wang Y, Zhang XR, Yin CY, Hu CM, Tian M, Wang HW, Zhang X. Levels of 1,25(OH)2D3 for patients with pulmonary tuberculosis and correlations of 1,25(OH)2D3 with the clinical features of TB. J Thorac Dis. 2014;6:760–764. doi: 10.3978/j.issn.2072-1439.2014.05.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Panda DK, Miao D, Tremblay ML, Sirois J, Farookhi R, Hendy GN, Goltzman D. Targeted ablation of the 25-hydroxyvitamin D 1alpha -hydroxylase enzyme: Evidence for skeletal, reproductive, and immune dysfunction. Proc Natl Acad Sci USA. 2001;98:7498–7503. doi: 10.1073/pnas.131029498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Schedel M, Jia Y, Michel S, Takeda K, Domenico J, Joetham A, Ning F, Strand M, Han J, Wang M, et al. 1,25D3 prevents CD8(+)Tc2 skewing and asthma development through VDR binding changes to the Cyp11a1 promoter. Nat Commun. 2016;7:10213. doi: 10.1038/ncomms10213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ooi JH, McDaniel KL, Weaver V, Cantorna MT. Murine CD8+ T cells but not macrophages express the vitamin D 1α-hydroxylase. J Nutr Biochem. 2014;25:58–65. doi: 10.1016/j.jnutbio.2013.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kongsbak M, von Essen MR, Levring TB, Schjerling P, Woetmann A, Ødum N, Bonefeld CM, Geisler C. Vitamin D-binding protein controls T cell responses to vitamin D. BMC Immunol. 2014;15:35. doi: 10.1186/s12865-014-0035-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lu Z, Jin M, Huang M, Wang Y, Wang Y. Bioactivity of selenium-enriched exopolysaccharides produced by Enterobacter cloacae Z0206 in broilers. Carbohydr Polym. 2013;96:131–136. doi: 10.1016/j.carbpol.2013.03.063. [DOI] [PubMed] [Google Scholar]
- 21.Yan G, Xi Y, Xu S, Chen J, Lin Y, Dai H, Cheng P, Xiao H, Liu Z, Qi Z. Inhibiting accelerated rejection mediated by alloreactive CD4(+) memory T cells and prolonging allograft survival by 1α,25-dihydroxyvitamin D(3) in nude mice. Immunol Lett. 2013;149:54–61. doi: 10.1016/j.imlet.2012.11.001. [DOI] [PubMed] [Google Scholar]
- 22.Prehn JL, Jordan SC. Incubation of T cell or monocytic cell lines with 1,25-dihydroxyvitamin D3 before mitogen stimulation potentiates IL-2 and IL-1 beta mRNA levels. Transplant Proc. 1989;21:90–91. [PubMed] [Google Scholar]
- 23.Dhur A, Galan P, Preziosi P, Hercberg S. Lymphocyte subpopulations in the thymus, lymph nodes and spleen of iron-deficient and rehabilitated mice. J Nutr. 1991;121:1418–1424. doi: 10.1093/jn/121.9.1418. [DOI] [PubMed] [Google Scholar]
- 24.Zhao L, Jiang Z, Jiang Y, Ma N, Wang K, Zhang Y. Changes in immune cell frequencies after cyclophosphamide or mycophenolate mofetil treatments in patients with systemic lupus erythematosus. Clin Rheumatol. 2012;31:951–959. doi: 10.1007/s10067-012-1958-8. [DOI] [PubMed] [Google Scholar]
- 25.Wang FY, Su M, Zheng YQ, Wang XG, Kang N, Chen T, Zhu EL, Bian ZX, Tang XD. Herbal prescription Chang'an II repairs intestinal mucosal barrier in rats with post-inflammation irritable bowel syndrome. Acta Pharmacol Sin. 2015;36:708–715. doi: 10.1038/aps.2014.170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Harrer A, Pilz G, Wipfler P, Oppermann K, Sellner J, Hitzl W, Haschke-Becher E, Afazel S, Rispens T, van der Kleij D, et al. High interindividual variability in the CD4/CD8 T cell ratio and natalizumab concentration levels in the cerebrospinal fluid of patients with multiple sclerosis. Clin Exp Immunol. 2015;180:383–392. doi: 10.1111/cei.12590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hadrup SR, Strindhall J, Køllgaard T, Seremet T, Johansson B, Pawelec G, Straten P thor, Wikby A. Longitudinal studies of clonally expanded CD8 T cells reveal a repertoire shrinkage predicting mortality and an increased number of dysfunctional cytomegalovirus-specific T cells in the very elderly. J Immunol. 2006;176:2645–2653. doi: 10.4049/jimmunol.176.4.2645. [DOI] [PubMed] [Google Scholar]
- 28.Yin Y, Qin J, Dai Y, Zeng F, Pei H, Wang J. The CD4+/CD8+ ratio in pulmonary tuberculosis: Systematic and meta-analysis article. Iran J Public Health. 2015;44:185–193. [PMC free article] [PubMed] [Google Scholar]
- 29.Serrano-Villar S, Sainz T, Lee SA, Hunt PW, Sinclair E, Shacklett BL, Ferre AL, Hayes TL, Somsouk M, Hsue PY, et al. HIV-infected individuals with low CD4/CD8 ratio despite effective antiretroviral therapy exhibit altered T cell subsets, heightened CD8+ T cell activation, and increased risk of non-AIDS morbidity and mortality. PLoS Pathog. 2014;10:e1004078. doi: 10.1371/journal.ppat.1004078. [DOI] [PMC free article] [PubMed] [Google Scholar]


