Abstract
Uncoupling of nitric oxide synthase (NOS) secondary to redox signaling is a central mechanism in endothelial and macrophage activation. To date studies on the production of nitric oxide (NO) during the development of diabetic complications show paradoxical results. We previously showed that recoupling eNOS by increasing the eNOS cofactor tetrahydrobiopterin (BH4) could restore endothelial function and prevent kidney injury in experimental kidney transplantation. Here, we employed a diabetic mouse model to investigate the effects of diabetes on renal tissue NO bioavailability. For this, we used in vivo NO trapping, followed by electron paramagnetic resonance spectroscopy. In addition, we investigated whether coupling of NOS by supplying the cofactor BH4 could restore glomerular endothelial barrier function. Our data show that overall NO availability at the tissue level is not reduced sixteen weeks after the induction of diabetes in apoE knockout mice, despite the presence of factors that cause endothelial dysfunction, and the presence of the endogenous NOS inhibitor ADMA. Targeting uncoupled NOS with the BH4 precursor sepiapterin further increases NO availability, but did not modify renal glomerular injury. Notably, glomerular heparanase activity as a driver for loss of glomerular barrier function was not reduced, pointing towards NOS-independent mechanisms. This was confirmed by unaltered increased glomerular presence of cathepsin L, the protease that activates heparanase.
Introduction
Endothelial dysfunction is assumed to contribute to kidney disease progression in diabetes [1] with endothelial nitric oxide (NO) production as a key feature of healthy endothelium. Uncoupling of NOS secondary to redox signaling is a central mechanism in endothelial activation. We previously showed that recoupling eNOS by increasing the NOS cofactor tetrahydrobiopterin (BH4) could restore endothelial function and prevent kidney injury in experimental kidney transplantation [2, 3]. BH4 was also shown to modulate the iNOS isoform in macrophages, counterbalancing the redox effector pathway in these cells [4], where macrophage activation has also been implicated in progression of diabetic nephropathy [5, 6].
Conflicting results about the role of NO in diabetic nephropathy have, however, been publish in vitro as well as in vivo. Enhanced NO generation have been reported in studies of cultured hyperglycemic endothelial cells, advanced diabetes and diabetic nephropathy [7–9], whereas NO availability has also been reported to be diminished in the setting of human [10–12] and rodent [13–15] diabetic nephropathy. Also a time dependent effect has been reported: NO generation during the early stages of nephropathy was increased while it decreased in later stages [16, 17].
While eNOS knockout mice have proved to be invaluable for studies assessing the consequences of reduced NO availability in diabetic nephropathy [18], the absence of NO make these mice unsuited for our studies. Therefore, we treated apolipoprotein E knockout mice (apoE KO) with streptozotocin to induce stable and reproducible diabetic nephropathy [19, 20]. We used in vivo NO spin-trapping with iron-dithiocarbamate complexes, a highly-sensitive quantitative technique that enables one to detect localized concentrations of trapped NO in vivo [21–24]. Also, we assessed glomerular endothelial function, by determining glycocalyx integrity and barrier function.
Surprisingly, we observed enhanced renal NO bioavailability in the setting of diabetic nephropathy. We also found that sepiapterin, a BH4 precursor, while further augmenting local NO levels, did not modify renal glomerular injury. This suggests that diabetes-induced endothelial dysfunction is not directly associated with renal NO deficiency. Notably, glomerular heparanase activity as a driver for loss of glomerular barrier function was not reduced, pointing towards NOS-independent mechanisms.
Materials and Methods
Diabetic ApoE KO mouse model
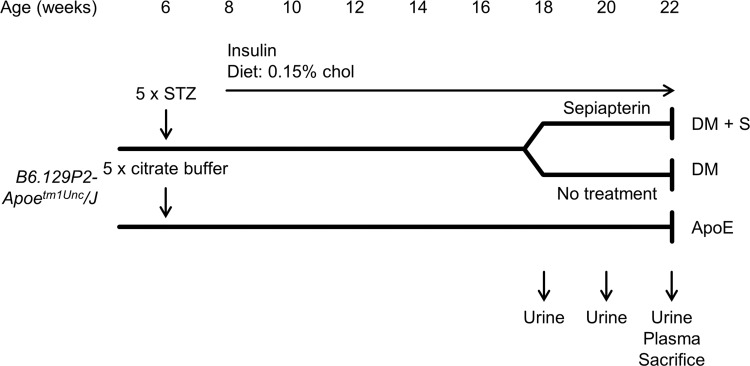
All animal experiments were approved by the ethical committee on animal care and experimentation of the Leiden University Medical Centre. All animal work was performed in compliance with the Dutch governmental guidelines. For our experiments, we used three groups of six weeks old male ApoE KO mice (Jackson Laboratory, Bar Harbor, ME). Diabetes was induced in two groups by intraperitoneal injections with streptozotocin (STZ, 60 mg/kg; Sigma-Aldrich, St. Louis, MO) in citrate buffer (0.1 mol/L, pH = 4.5) for five consecutive days, according to the DiaComp protocol (Fig 1). The non-diabetic control group was injected with citrate buffer alone. All mice were housed under normal day-night cycle with free access to drinking water and chow. Non-diabetic apoE KO mice received standard chow, whereas diabetic apoE KO mice received cholesterol-enriched chow (0.15 wt%) (Technilab-BMI, Someren, The Netherlands). Twelve weeks after the induction of diabetes, these mice were divided in two groups: one group was treated with sepiapterin (10 mg/kg daily) in drinking water for four consecutive weeks, to increase NO levels. The other group received normal drinking water. Data obtained from non-diabetic apoE KO mice as well as control diabetic apoE KO mice are previously published [19].
Fig 1. Experimental set-up for assessment of NO bioavailability.
(A) male ApoE KO mice (B6.129P2- Apoetm1Unc/J) were injected with citrate buffer ± STZ. Diabetic mice received cholesterol enriched diet and insulin from week 8 onwards. At 18 week of age, diabetic mice were treated with sepiapterin or received normal drinking water for 4 weeks. Urine was collected upon commencing with the experimental procedure and after 2 and 4 weeks of treatment. At 22 weeks, plasma was collected and the mice were sacrificed.
Blood glucose concentrations were measured with an Accu-check glucose meter (Roche, Basel, Switzerland). When glucose concentrations exceeded 25 mmol/L, mice were treated with 1–2 units insulin (Lantus, Aventis Pharmaceuticals, Bridgewater, NJ, US) up to three times per week. Sixteen weeks after STZ injections, at 22 weeks of age, mice were sacrificed to perform NO-trapping in kidney, liver and heart.
Urine collection and analyses
A 24-hour urine sample was collected at 12, 14 and 16 weeks after STZ injections. Urinary albumin and creatinine concentrations were determined as previously described [19]. In brief, albumin concentrations were quantified with Rocket immunoelectrophoresis [25] and creatinine concentrations were quantified by the Jaffé method using 0.13% picric acid and a creatinine standard set (Sigma-Aldrich).
Immunohistochemistry
Following sacrificing by CO2 asphyxiation, one half of a kidney was used for histological assessment of classical markers of glomerular damage and morphometric changes to confirm diabetic nephropathy. For this, capillary and mesangial matrix area were quantified in Periodic acid-Schiff (PAS) or Trichrome-stained paraffin embedded 4 μm sections, respectively. We used a semiautomatic image analyzing system (Leica Q600 Qwin; Leica Microsystems, Cambridge, UK) to determine the fraction of glomerular surface area by the point-counting method.
Glomerular heparanase expression was quantified after overnight incubation with primary antibody (Polyclonal rabbit anti-heparanase 1.5 μg/mL, InSight Biopharmaceuticals, Rehovot, Israel), followed by goat anti-rabbit IgG-Alexa 594 (1/1000), for 1 hour, both in blocking buffer. Sections were counterstained with Hoechst (1/1000) and embedded in Vectashield mounting medium (Vector Laboratories Inc., Burlingame, CA). Cathepsin L polyclonal antibody (R&D Systems) was incubated overnight, followed by horseradish peroxidase–conjugated secondary antibody and 3,3’-diaminobenzidine and counterstained with hematoxylin. Staining area was quantified as the percentage of stained area divided by the glomerular area.
Determination of nitric oxide
In all three groups, endogenous NO bioavailability was measured by in vivo spin trapping with iron-diethyldithiocarbamates (Fe2+-DETC) complexes as previously described [19]. After 30 minutes of spin-trapping, mice were sacrificed using CO2 asphyxiation. Subsequently, ~350 mg sections of kidney, liver and heart tissues were submerged in HEPES buffer (450 μl, 150 mmol/L, pH 7.4) and snap frozen with liquid nitrogen. The yield of paramagnetic ferrous mononitrosyl-iron complexes (MNIC) was determined with electron paramagnetic resonance (EPR) spectroscopy. For this, frozen tissue samples were measured at 77 K with an X-band EMX-Plus spectrometer (Bruker BioSpin, Rheinstetten, Germany) equipped with a Bruker liquid finger Dewar flask filled with liquid nitrogen. Spectrometer settings were microwave power, 20 mW; time constant, 82 ms; analog-to-digital conversion time, 82 ms; and detector gain, 104. The magnetic field was modulated with 5-G amplitude at a frequency of 100 kHz. With these settings, a single field sweep provided adequate sensitivity. During the experiments, the inside of the EPR cavity (ER 4119 HS-W1, cylindrical TE011 mode; Bruker) was continuously flushed with dry nitrogen to prevent condensation of ambient humidity on the cool Dewar flask.
Determination of plasma ADMA concentrations
Asymmetric NG, NG-dimethyl-L-arginine (ADMA) is a endogenous inhibitor of nitric oxide synthases. Therefore, we measured plasma ADMA concentrations with a commercially available enzyme-linked immunosorbent assay kit (DLD Diagnostika GmbH, Hamburg, Germany) according to the manufacturer's protocol.
Determination of glomerular endothelial glycocalyx coverage
For electron microscopic visualization of the glycocalyx, three mice per group were anesthetized (intraperitoneal). Left kidneys were perfused with 0.5% bovine serum albumin (BSA) and 5 U/mL heparin in 5 mL Hepes-buffered salt solution (HBSS), followed by 2 mL of cationic ferritin (horse spleen, 2.5 mg/mL, Electron Microscopy Sciences, Fort Washington, PA) in HBSS alone at 2 mL/minute, as described before [19]. Kidneys were excised, the capsules removed and stored in fixative (1.5% glutaraldehyde + 1% paraformaldehyde in 0.1 mol/L sodium-cacodylate buffered solution, pH 7.4) overnight at 4°C and further prepared for transmission electron microscopy (TEM). Data was collected into large virtual slides (stitches) that provide an overview of the glomeruli, allowing for high-detail assessment and quantitative analysis of glomerular (patho)physiology [26]. Images (2Kx2K) were acquired at an acceleration voltage of 120 kV, on FEI Tecnai T12 microscopes (FEI, Eindhoven, the Netherlands), equipped with FEI Eagle 4Kx4K CCD cameras. The polyanionic glycocalyx on the surface of endothelial cells can be visualized using TEM by binding of electron-dense cationic substances, such as cationic ferritin [27]. Within the stitches, individual capillary loops were captured and glycocalyx coverage was quantified in 6–11 capillary loops in 9 glomeruli. The percentage of cationic ferritin positive endothelial surface coverage was determined using an automatic grid overlay in the public domain NIH ImageJ version 1.46. For every capillary, a minimum of 80 crosshairs was at the intersection of the endothelium. This resulted in a percentage glycocalyx positive area.
Statistical analysis
Data are presented as mean ± SD, unless stated otherwise. Changes in ACR during treatment were analyzed using a linear-mixed model regression analysis, since samples were collected over time and were therefore animal dependent (SPSS Statistics, version 20, IBM). Differences in other experiments with continuous variables were determined using one-way ANOVA and post hoc analysis with Tukey’s multiple comparison test. P<0.05 was considered statistically significant.
Results
Diabetes increases renal NO levels
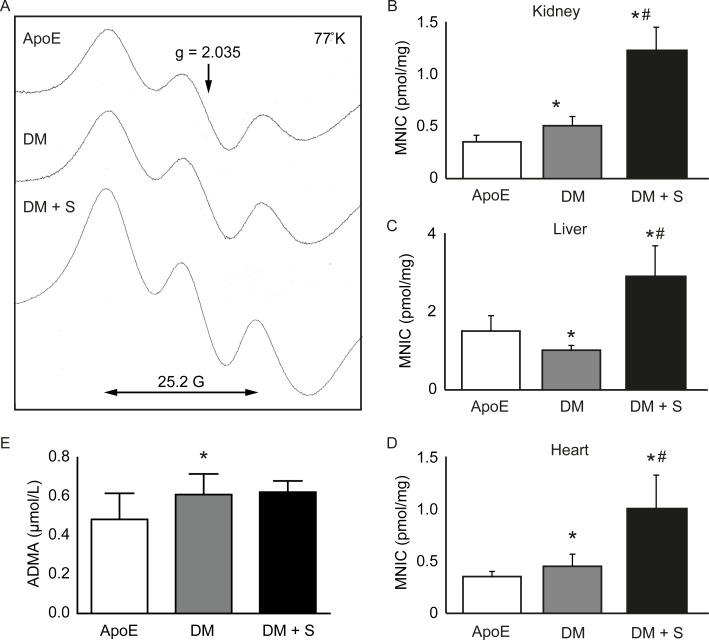
NO levels were determined as MNIC yields in various tissues (Fig 2), showing considerable variation between the tissue types. In particular, the liver was found to produce higher quantities of NO compared to renal and cardiac tissue. The findings in non-diabetic apoE KO mice are in qualitative concordance with previous findings in other rodent models such as rats [28] and C57Bl/6 mice (the latter revealing 0.34 ± 0.1 pmol MNIC / mg in kidney; 2.1 ± 1.0 in liver and 0.52 ± 0.1 pmol MNIC / mg heart). Upon STZ-induced diabetes, apoE KO mice revealed increased renal NO (0.51 ± 0.16 in diabetic apoE KO mice vs 0.29 ± 0.12 pmol MNIC / mg tissue in non-diabetic apoE KO, P<0.01 [19]). Of note, the effect of diabetes on NO bioavailability in cardiac tissue was considerably smaller (P<0.02), whereas STZ induced a reduction in NO bioavailability in hepatic tissue (P<0.01). Clearly, STZ-induced diabetes affects NO homeostasis in a tissue-dependent fashion, although prolonged treatment of diabetic mice with sepiapterin, a tetrahydrobiopterin (BH4) precursor, was found to induce a 2- to 3-fold increase of bioavailable NO in kidney, heart and liver tissue (Fig 2).
Fig 2. Tissue-dependent variation in NO free radical induction.
(A) EPR spectrum of frozen kidney samples. The characteristic triplet structure of the mononitrosyl-iron complex (MNIC, double-headed arrow) centers around g = 2.035 and represents the formation of local nitric oxide in 334–370 mg tissue. (B-D) Quantification of nitric oxide formation in kidney, liver and heart tissue, shown as mean pmol MNIC / mg wet tissue ± SD, n = 7–9. E) Plasma ADMA concentrations, shows as mean ± SD, n = 8. *P<0.05, compared with ApoE; #P<0.05 compared with DM. ApoE = ApoE KO mice, DM = diabetic apoE KO mice, DM + S = diabetic apoE KO mice + sepiapterin.
To see how these different observations at the tissue level reflect those observed in plasma, we also determined the formation of asymmetric dimethyl arginine (ADMA), a known endogenous inhibitor of eNOS. These studies uncovered a small increase in plasma ADMA concentrations upon STZ-induced diabetes (0.48 ± 0.13 μmol/L vs. 0.61 ±0.11 μmol/L, P<0.05; Fig 2E). Treatment with sepiapterin for four weeks did not affect plasma ADMA concentrations.
Increased NO levels do not improve diabetic nephropathy in diabetic apoE KO mice
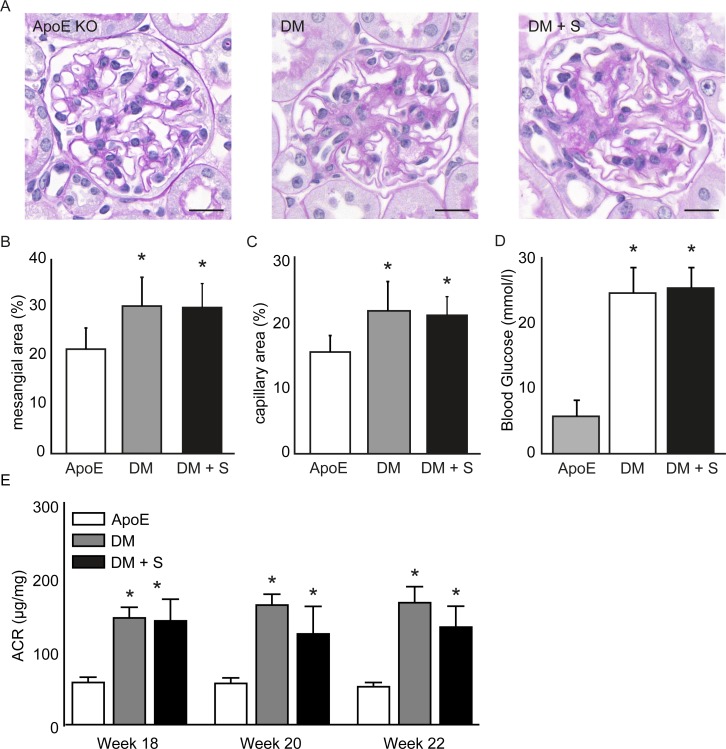
Fourteen weeks after inducing diabetes, we observed common characteristics of diabetic nephropathy, including mesangial expansion, mesangiolysis, and, glomerular hypertrophy, as we described elsewhere [19]. An ensuing treatment with sepiapterin for four weeks did not prevent the development of diabetic renal lesions as quantified in PAS- and Trichrome-stained glomeruli (Fig 3A–3C). Importantly, for these studies we verified that sepiapterin did not affect blood glucose concentrations at the selected dose (Fig 3D).
Fig 3. Sepiapterin does not reduce albuminuria in diabetic apoE KO mice.
(A) PAS-stained glomeruli of apoE KO mice (apoE), diabetic apoE KO mice (DM) and diabetic apoE KO mice treated with sepiapterin (DM + S), showing heterogeneous diabetic lesions 14 weeks after induction of diabetes with STZ (20). Scale bars: 20 μm. Sepiapterin did not affect mesangial area (B,C), nor blood glucose concentrations (D). Data are shown as mean ± SD, *P<0.05 compared with apoE, n = 8. (E) Albumin-creatinine ratios (ACR) at baseline, 2- and 4 weeks after treatment, as indicated by mean ± SEM, *P<0.05 compared with apoE, n = 14–23.
To assess the effect of sepiapterin on kidney function, we collected 24-hours urine samples prior to treatment with sepiapterin, as well as at 2 and 4 weeks after treatment. Diabetic apoE KO mice were characterized by progressive albuminuria (Fig 3E [19]), which is in keeping with a parallel increase in urine production and urinary albumin excretion [19]. Multiple comparisons reveal that a 4-week sepiapterin treatment regimen is insufficient to reduce albuminuria as compared to non-treated mice (-11.8 ± 7.1%, p = 0.46).
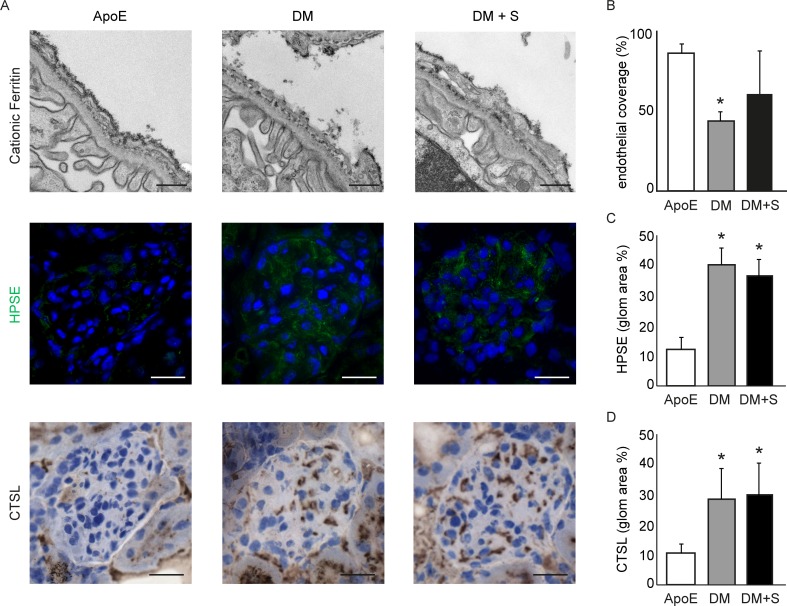
To visualize the consequences of sepiapterin treatment on the endothelial glycocalyx, we quantified the binding of cationic ferritin to the negatively-charged glycocalyx. As shown in Fig 4, diabetic mice displayed decreased endothelial coverage (40.7 ± 7.5%), as compared to non-diabetic apoE KO mice (83.6 ± 8.3%, P<0.05; Fig 4A and 4B [19]). Interestingly, treatment with sepiapterin seems to lead to a partial restoration of the glycocalyx, based on the observation that some glomeruli in non-diabetic mice display cationic ferritin coverage, whereas other glomeruli do not. This phenomenon resulted in high variability in glycocalyx-coverage data (61.6 ± 25.8%, P<0.1).
Fig 4. Increased NO levels affect endothelial glycocalyx non-uniformly.
(A) Representative microscopic images of cationic ferritin (TEM; top), heparanase (HPSE, immunofluorescence; middle) and cathepsin L (CTSL; bottom) in glomeruli of apoE KO mice (apoE), diabetic apoE KO mice (DM) and diabetic apoE KO mice treated with sepiapterin (DM + S). (B) Quantification of endothelial cationic ferritin coverage in 6–8 capillary loops in 9 glomeruli of 3 mice, shown as mean percentage of total capillary length ± SD, (C) Quantification of glomerular heparanase expression, shown as mean area percentage ± SD. (D) Quantification of glomerular cathepsin L expression, shown as mean area percentage ± SD. *P<0.05 compared with ApoE, n = 6–8. Scale bars: 500 nm in TEM images; 20 μm in fluorescent and light microscopic images.
In diabetes, various factors can lead to disruption of the glycocalyx [29]. Heparanase-mediated degradation of heparan sulphate represents one of the most widely recognized causes of glycocalyx perturbation. In particular locally active heparanase plays a critical role in the development of diabetic nephropathy in mice [30]. We previously confirmed enhanced glomerular heparanase protein expression in diabetic apoE mice, compared to non-diabetic apoE KO mice (39.3 ± 10.8% vs. 13.1 ± 9.2%, P<0.01; Fig 4A and 4C). A notable observation is that sepiapterin treatment did not lead to a reduction in the expression levels of this enzyme (36.1 ± 7.4.8%), nor in the heparanase activator cathepsin L (30.2 ± 12.5% vs. 27.3 ± 11.3 for non-treated diabetic mice). This suggests that heparanase is continuously activated by immunocyte derived cathepsin L despite treatment with sepiapterin, which could potentially explain that restoration of glycocalyx coverage by sepiapterin in diabetic nephropathy was only partial.
Despite the observed partial restoration of the luminal glycocalyx and possible regulatory effect of eNOS on heparanase [31], increasing NO bioavailability with sepiapterin was not capable to restore endothelial function.
Discussion
Endothelial dysfunction plays a critical role in the pathogenesis of diabetes. Diabetes is associated with loss of glomerular barrier function. Activation of extracellular heparanase by inflammatory cells or injured epithelial cells was shown to be of critical importance to cause loss of the glomerular endothelial glycocalyx and proteinuria [30, 32]. Nitric oxide (NO) through intracellular s-nitrosylation is a key modifier of cellular function and to ensure cellular quiescence and tissue homeostasis. The three NO synthases (eNOS, iNOS and nNOS) are however complex oxidoreductases with the potential to produce NO as well as reactive oxygen species [33]. Specifically, for the production of NO they require the cofactor tetrahydrobiopterin (BH4). Diabetes has been shown to result in loss of BH4 availability and uncoupling of NOS [7, 34].
The present study shows that improving the NO producing capacity at the tissue level in the kidney does not result in reduction of glomerular heparanase activity or improvement of glomerular barrier function. Our observations add several new insights about 1] NO measurements, 2] NO sources in different diabetes models, and 3] the glomerular endothelial glycocalyx:
When measuring NO availability at the tissue level using state-of-the-art spin trapping technique (EPR) that allows for quantitative NO measurements, diabetes perse is not associated with overall reduction in NO. In fact, NO availability was increased in the kidney and heart upon diabetes, while liver NO was decreased, despite elevated levels of the endogenous NOS inhibitor asymmetric dimethylarginine (ADMA). Reports on NO availability in diabetes have varied from decreased to increased, pointing to the variation in models and the often indirect measurements of NO activity. Given the complex nature of in vivo NO radical detection, surrogate markers for free NO levels have oftentimes been utilized as a readout, including plasma NOx levels, nitrate, nitrosothiols or nitrosylated heme. In this context, we note that previous reports of enhanced NO production in diabetes [8] were based on the quantification of downstream metabolites of NO, such as NOx or nitrate. Furthermore, previous reports on decreased renal NO production in rat 7–10 days after induction of diabetes are based on decreased urinary nitrite/nitrate excretion [35, 36] or decreased plasma nitrite/nitrate levels at 8 weeks after induction of diabetes [15]. Importantly, these NO metabolites serve as poor indicators of the actual NO free radical levels. This is largely in part due to the fact that enhanced synthesis of NO augments NOx generation, whereas a concomitant increase in oxidative stress or reactive oxygen species (ROS) has been established to trigger a rapid depletion of local NO levels. As such, the detection of MNIC, the formation of which is specific for NO free radicals in biological tissues, represents a more suitable experimental and diagnostic approach for detecting local NO levels [21, 22, 37].
Renal NO production in diabetes will be dependent upon different sources including endothelium, macrophages [38] and the tubular system [39]. Chronic uncoupling or inhibition of eNOS has been shown to accelerate kidney disease [40, 41]. Conditions commonly observed in patients with kidney disease, such as hyperglycemia, are characterized by an increase in the generation of advanced glycation end products (AGEs) [42]. AGEs actively promote NO insufficiency by scavenging NO free radicals [13] via pro-inflammatory AGE-specific receptors [43–45], or modification of plasma proteins such as albumin [46]. We recently described that the streptozotocin induced diabetic apoE mouse model, which faithfully recapitulates the renal changes in diabetes, is characterized by macrophage activation, which may explain the increased NO availability. McNeill et al showed in a series of elegant studies that macrophage function is modulated by NOS coupling and BH4 availability [4], as was previously also shown by us for endothelial function [3]. The current data show that it is possible to increase NO production further in diabetes by coupling through BH4 availability, indicating that uncoupling of NOS enzymes was present in the model. In contrast to the current study, sepiapterin showed renal protective effect in diabetic db/db mice [47] and ZSF1 rats, which can possibly be explained by the fact that these models are primarily characterized by insulin resistance. Insulin signaling is coupled to NOS activation and impaired insulin signaling may thus have altered the coupling state of NOS in these models. In our model, mice received small amounts of insulin to keep blood glucose concentrations within reasonable range. Given that insulin stimulates NO release by endothelial cells [48, 49], this could serve as an explanation for the fact that we did not observe overall NO deficiency in our diabetic apoE KO mice.
Endothelial cells are covered with a dense layer of proteoglycans and glycosaminoglycans, the endothelial glycocalyx. It is the first barrier of the vascular wall and is vasculoprotective [50] by acting as a permeability barrier [51–53], a mechanosensor [50, 54–57], and by regulating inflammation [58–61]. Loss of endothelial glycocalyx occurs, amongst others, during oxidative stress and inflammation, both present in diabetes. This is related to the induction of glycocalyx degrading enzymes, such as hyaluronidase, heparanase and chondroitinase [62, 63]. Of these, heparanase activity has been suggested to be affected by NO bioavailability [31]. Heparanase-induced glycocalyx degradation leads to albuminuria [1] and increased glomerular heparanase expression was shown to be associated with the development of diabetic nephropathy in humans and mice [30, 64].
Immunocytes have been implicated in activation of extracellular heparanase and degradation of heparan sulfate in the glycocalyx [1]. Whereas multiple cells can secrete heparanase upon activation, including endothelium, podocytes and immunocytes [30], its activation requires the cleavage of a linker protein by the protease cathepsin L [30, 65]. While sepiapterin successfully increased the modulating potential of NO in this model of diabetic nephropathy, immunocytes were not affected, hence glomerular heparanase and cathepsin L were not reduced and glycocalyx properties not restored.
Conclusion
In conclusion, our data show that overall NO availability at the tissue level is not reduced in diabetes, despite the presence of factors causing endothelial dysfunction, and the presence of increased levels of the endogenous NOS inhibitor ADMA. Targeting uncoupled NOS with the BH4 precursor sepiapterin increases NO availability, but does not modify renal glomerular endothelial barrier function.
Acknowledgments
We thank Prof E. Bouwman (Inorganic Chemistry, Leiden University) for the use of the electron paramagnetic resonance facilities. We thank Prof A.J. Koster (Molecular Cell Biology, Leiden University Medical Center) for the use of electron microscope facilities. We thank Dr. E.P. van der Veer (Department of Internal Medicine, Leiden University Medical Center) for help in the preparation of this manuscript.
Data Availability
All relevant data are within the paper.
Funding Statement
This study was supported by the Glycoren consortium grant of the Dutch Kidney Foundation (CP09.03) and an AbbVie study grant (REN-11-0026).
References
- 1.Rabelink TJ, de Zeeuw D. The glycocalyx—linking albuminuria with renal and cardiovascular disease. Nature reviews Nephrology. 2015;11(11):667–76. 10.1038/nrneph.2015.162 [DOI] [PubMed] [Google Scholar]
- 2.Vos IH, Rabelink TJ, Dorland B, Loos R, Van Middelaar B, Grone HJ, et al. L-arginine supplementation improves function and reduces inflammation in renal allografts. Journal of the American Society of Nephrology: JASN. 2001;12(2):361–7. [DOI] [PubMed] [Google Scholar]
- 3.Huisman A, Vos I, van Faassen EE, Joles JA, Grone HJ, Martasek P, et al. Anti-inflammatory effects of tetrahydrobiopterin on early rejection in renal allografts: modulation of inducible nitric oxide synthase. FASEB journal: official publication of the Federation of American Societies for Experimental Biology. 2002;16(9):1135–7. [DOI] [PubMed] [Google Scholar]
- 4.McNeill E, Crabtree MJ, Sahgal N, Patel J, Chuaiphichai S, Iqbal AJ, et al. Regulation of iNOS function and cellular redox state by macrophage Gch1 reveals specific requirements for tetrahydrobiopterin in NRF2 activation. Free radical biology & medicine. 2015;79:206–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nguyen D, Ping F, Mu W, Hill P, Atkins RC, Chadban SJ. Macrophage accumulation in human progressive diabetic nephropathy. Nephrology. 2006;11(3):226–31. 10.1111/j.1440-1797.2006.00576.x [DOI] [PubMed] [Google Scholar]
- 6.Klessens CQ, Zandbergen M, Wolterbeek R, Bruijn JA, Rabelink TJ, Bajema IM, et al. Macrophages in diabetic nephropathy in patients with type 2 diabetes. Nephrology, dialysis, transplantation: official publication of the European Dialysis and Transplant Association—European Renal Association. 2016. [DOI] [PubMed] [Google Scholar]
- 7.Cai S, Khoo J, Channon KM. Augmented BH4 by gene transfer restores nitric oxide synthase function in hyperglycemic human endothelial cells. Cardiovascular research. 2005;65(4):823–31. 10.1016/j.cardiores.2004.10.040 [DOI] [PubMed] [Google Scholar]
- 8.Adela R, Nethi SK, Bagul PK, Barui AK, Mattapally S, Kuncha M, et al. Hyperglycaemia enhances nitric oxide production in diabetes: a study from South Indian patients. PloS one. 2015;10(4):e0125270 10.1371/journal.pone.0125270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hohenstein B, Hugo CP, Hausknecht B, Boehmer KP, Riess RH, Schmieder RE. Analysis of NO-synthase expression and clinical risk factors in human diabetic nephropathy. Nephrology, dialysis, transplantation: official publication of the European Dialysis and Transplant Association—European Renal Association. 2008;23(4):1346–54. [DOI] [PubMed] [Google Scholar]
- 10.Tessari P, Cecchet D, Cosma A, Vettore M, Coracina A, Millioni R, et al. Nitric oxide synthesis is reduced in subjects with type 2 diabetes and nephropathy. Diabetes. 2010;59(9):2152–9. 10.2337/db09-1772 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tessari P, Cecchet D, Artusi C, Vettore M, Millioni R, Plebani M, et al. Roles of insulin, age, and asymmetric dimethylarginine on nitric oxide synthesis in vivo. Diabetes. 2013;62(8):2699–708. 10.2337/db12-1127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Shiekh GA, Ayub T, Khan SN, Dar R, Andrabi KI. Reduced nitrate level in individuals with hypertension and diabetes. J Cardiovasc Dis Res. 2011;2(3):172–6. 10.4103/0975-3583.85264 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bucala R, Tracey KJ, Cerami A. Advanced glycosylation products quench nitric oxide and mediate defective endothelium-dependent vasodilatation in experimental diabetes. The Journal of clinical investigation. 1991;87(2):432–8. 10.1172/JCI115014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Prabhakar S, Starnes J, Shi S, Lonis B, Tran R. Diabetic nephropathy is associated with oxidative stress and decreased renal nitric oxide production. Journal of the American Society of Nephrology: JASN. 2007;18(11):2945–52. 10.1681/ASN.2006080895 [DOI] [PubMed] [Google Scholar]
- 15.Arya A, Yadav HN, Sharma PL. Involvement of vascular endothelial nitric oxide synthase in development of experimental diabetic nephropathy in rats. Molecular and cellular biochemistry. 2011;354(1–2):57–66. 10.1007/s11010-011-0805-6 [DOI] [PubMed] [Google Scholar]
- 16.Komers R, Anderson S. Paradoxes of nitric oxide in the diabetic kidney. American journal of physiology Renal physiology. 2003;284(6):F1121–37. 10.1152/ajprenal.00265.2002 [DOI] [PubMed] [Google Scholar]
- 17.Dellamea BS, Leitao CB, Friedman R, Canani LH. Nitric oxide system and diabetic nephropathy. Diabetol Metab Syndr. 2014;6(1):17 10.1186/1758-5996-6-17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kanetsuna Y, Takahashi K, Nagata M, Gannon MA, Breyer MD, Harris RC, et al. Deficiency of endothelial nitric-oxide synthase confers susceptibility to diabetic nephropathy in nephropathy-resistant inbred mice. The American journal of pathology. 2007;170(5):1473–84. 10.2353/ajpath.2007.060481 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Boels MG, Avramut MC, Koudijs A, Dane MJ, Lee DH, van der Vlag J, et al. Atrasentan Reduces Albuminuria by Restoring the Glomerular Endothelial Glycocalyx Barrier in Diabetic Nephropathy. Diabetes. 2016;65(8):2429–39. 10.2337/db15-1413 [DOI] [PubMed] [Google Scholar]
- 20.Lassila M, Seah KK, Allen TJ, Thallas V, Thomas MC, Candido R, et al. Accelerated nephropathy in diabetic apolipoprotein e-knockout mouse: role of advanced glycation end products. Journal of the American Society of Nephrology. 2004;15(8):2125–38. 10.1097/01.ASN.0000133025.23732.46 [DOI] [PubMed] [Google Scholar]
- 21.Vanin AF, Huisman A, Van Faassen EE. Iron dithiocarbamate as spin trap for nitric oxide detection: pitfalls and successes. Methods in enzymology. 2002;359:27–42. [DOI] [PubMed] [Google Scholar]
- 22.Vanin A, van Faassen E Mononitrosyl-iron complexes with dithiocarbamate ligands: Physico-chemical properties In: Vanin EvFaA, editor. Radicals for life: The various forms of nitric oxide: Elsevier; 2007. p. 383–406. [Google Scholar]
- 23.van Faassen EE, Koeners MP, Joles JA, Vanin AF. Detection of basal NO production in rat tissues using iron–dithiocarbamate complexes. Nitric Oxide. 2008;18(4):279–86. 10.1016/j.niox.2008.02.003 [DOI] [PubMed] [Google Scholar]
- 24.Ohnishi ST. Measurement of NO using electron paramagnetic resonance. Methods in molecular biology. 1998;100:129–54. [DOI] [PubMed] [Google Scholar]
- 25.Tran TN, Eubanks SK, Schaffer KJ, Zhou CY, Linder MC. Secretion of ferritin by rat hepatoma cells and its regulation by inflammatory cytokines and iron. Blood. 1997;90(12):4979–86. [PubMed] [Google Scholar]
- 26.Faas FG, Avramut MC, van den Berg BM, Mommaas AM, Koster AJ, Ravelli RB. Virtual nanoscopy: generation of ultra-large high resolution electron microscopy maps. The Journal of cell biology. 2012;198(3):457–69. 10.1083/jcb.201201140 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Avasthi PS, Koshy V. The anionic matrix at the rat glomerular endothelial surface. The Anatomical record. 1988;220(3):258–66. 10.1002/ar.1092200306 [DOI] [PubMed] [Google Scholar]
- 28.Koeners MP, van Faassen EE, Wesseling S, de Sain-van der Velden M, Koomans HA, Braam B, et al. Maternal supplementation with citrulline increases renal nitric oxide in young spontaneously hypertensive rats and has long-term antihypertensive effects. Hypertension. 2007;50(6):1077–84. 10.1161/HYPERTENSIONAHA.107.095794 [DOI] [PubMed] [Google Scholar]
- 29.Schott U, Solomon C, Fries D, Bentzer P. The endothelial glycocalyx and its disruption, protection and regeneration: a narrative review. Scand J Trauma Resusc Emerg Med. 2016;24(1):48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gil N, Goldberg R, Neuman T, Garsen M, Zcharia E, Rubinstein AM, et al. Heparanase is essential for the development of diabetic nephropathy in mice. Diabetes. 2012;61(1):208–16. 10.2337/db11-1024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Garsen M, Rops AL, Li J, van Beneden K, van den Branden C, Berden JH, et al. Endothelial Nitric Oxide Synthase Prevents Heparanase Induction and the Development of Proteinuria. PloS one. 2016;11(8):e0160894 10.1371/journal.pone.0160894 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Goldberg R, Rubinstein AM, Gil N, Hermano E, Li JP, van der Vlag J, et al. Role of heparanase-driven inflammatory cascade in pathogenesis of diabetic nephropathy. Diabetes. 2014;63(12):4302–13. 10.2337/db14-0001 [DOI] [PubMed] [Google Scholar]
- 33.Rabelink TJ, Luscher TF. Endothelial nitric oxide synthase: host defense enzyme of the endothelium? Arteriosclerosis, thrombosis, and vascular biology. 2006;26(2):267–71. 10.1161/01.ATV.0000196554.85799.77 [DOI] [PubMed] [Google Scholar]
- 34.Lucotti P, Setola E, Monti LD, Galluccio E, Costa S, Sandoli EP, et al. Beneficial effects of a long-term oral L-arginine treatment added to a hypocaloric diet and exercise training program in obese, insulin-resistant type 2 diabetic patients. Am J Physiol Endocrinol Metab. 2006;291(5):E906–12. 10.1152/ajpendo.00002.2006 [DOI] [PubMed] [Google Scholar]
- 35.Pflueger AC, Larson TS, Hagl S, Knox FG. Role of nitric oxide in intrarenal hemodynamics in experimental diabetes mellitus in rats. The American journal of physiology. 1999;277(3 Pt 2):R725–33. [DOI] [PubMed] [Google Scholar]
- 36.Keynan S, Hirshberg B, Levin-Iaina N, Wexler ID, Dahan R, Reinhartz E, et al. Renal nitric oxide production during the early phase of experimental diabetes mellitus. Kidney international. 2000;58(2):740–7. 10.1046/j.1523-1755.2000.00220.x [DOI] [PubMed] [Google Scholar]
- 37.Vanin AF, Poltorakov AP, Mikoyan VD, Kubrina LN, van Faassen E. Why iron-dithiocarbamates ensure detection of nitric oxide in cells and tissues. Nitric Oxide. 2006;15(4):295–311. 10.1016/j.niox.2005.11.007 [DOI] [PubMed] [Google Scholar]
- 38.Kashem A, Endoh M, Yano N, Yamauchi F, Nomoto Y, Sakai H. Expression of inducible-NOS in human glomerulonephritis: the possible source is infiltrating monocytes/macrophages. Kidney international. 1996;50(2):392–9. [DOI] [PubMed] [Google Scholar]
- 39.Mohaupt MG, Elzie JL, Ahn KY, Clapp WL, Wilcox CS, Kone BC. Differential expression and induction of mRNAs encoding two inducible nitric oxide synthases in rat kidney. Kidney international. 1994;46(3):653–65. [DOI] [PubMed] [Google Scholar]
- 40.Jover B, Mimran A. Nitric oxide inhibition and renal alterations. Journal of cardiovascular pharmacology. 2001;38 Suppl 2:S65–70. [DOI] [PubMed] [Google Scholar]
- 41.Nakagawa T. Uncoupling of the VEGF-endothelial nitric oxide axis in diabetic nephropathy: an explanation for the paradoxical effects of VEGF in renal disease. American journal of physiology Renal physiology. 2007;292(6):F1665–72. 10.1152/ajprenal.00495.2006 [DOI] [PubMed] [Google Scholar]
- 42.Noordzij MJ, Lefrandt JD, Smit AJ. Advanced glycation end products in renal failure: an overview. J Ren Care. 2008;34(4):207–12. 10.1111/j.1755-6686.2008.00038.x [DOI] [PubMed] [Google Scholar]
- 43.Yan SF, Ramasamy R, Schmidt AM. Mechanisms of disease: advanced glycation end-products and their receptor in inflammation and diabetes complications. Nature clinical practice Endocrinology & metabolism. 2008;4(5):285–93. [DOI] [PubMed] [Google Scholar]
- 44.Vlassara H, Uribarri J, Ferrucci L, Cai W, Torreggiani M, Post JB, et al. Identifying advanced glycation end products as a major source of oxidants in aging: implications for the management and/or prevention of reduced renal function in elderly persons. Seminars in nephrology. 2009;29(6):594–603. 10.1016/j.semnephrol.2009.07.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Vlassara H, Striker GE. AGE restriction in diabetes mellitus: a paradigm shift. Nat Rev Endocrinol. 2011;7(9):526–39. 10.1038/nrendo.2011.74 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Xu B, Ji Y, Yao K, Cao YX, Ferro A. Inhibition of human endothelial cell nitric oxide synthesis by advanced glycation end-products but not glucose: relevance to diabetes. Clinical science. 2005;109(5):439–46. 10.1042/CS20050183 [DOI] [PubMed] [Google Scholar]
- 47.Cheng H, Wang H, Fan X, Paueksakon P, Harris RC. Improvement of endothelial nitric oxide synthase activity retards the progression of diabetic nephropathy in db/db mice. Kidney international. 2012;82(11):1176–83. 10.1038/ki.2012.248 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zeng G, Nystrom FH, Ravichandran LV, Cong LN, Kirby M, Mostowski H, et al. Roles for insulin receptor, PI3-kinase, and Akt in insulin-signaling pathways related to production of nitric oxide in human vascular endothelial cells. Circulation. 2000;101(13):1539–45. [DOI] [PubMed] [Google Scholar]
- 49.Dimmeler S, Fleming I, Fisslthaler B, Hermann C, Busse R, Zeiher AM. Activation of nitric oxide synthase in endothelial cells by Akt-dependent phosphorylation. Nature. 1999;399(6736):601–5. 10.1038/21224 [DOI] [PubMed] [Google Scholar]
- 50.Gouverneur M, Berg B, Nieuwdorp M, Stroes E, Vink H. Vasculoprotective properties of the endothelial glycocalyx: effects of fluid shear stress. Journal of internal medicine. 2006;259(4):393–400. 10.1111/j.1365-2796.2006.01625.x [DOI] [PubMed] [Google Scholar]
- 51.Singh A, Satchell SC, Neal CR, McKenzie EA, Tooke JE, Mathieson PW. Glomerular endothelial glycocalyx constitutes a barrier to protein permeability. Journal of the American Society of Nephrology: JASN. 2007;18(11):2885–93. Epub 2007/10/19. 10.1681/ASN.2007010119 [DOI] [PubMed] [Google Scholar]
- 52.van den Berg BM, Vink H, Spaan JA. The endothelial glycocalyx protects against myocardial edema. Circulation research. 2003;92(6):592–4. 10.1161/01.RES.0000065917.53950.75 [DOI] [PubMed] [Google Scholar]
- 53.van den Berg BM, Spaan JA, Vink H. Impaired glycocalyx barrier properties contribute to enhanced intimal low-density lipoprotein accumulation at the carotid artery bifurcation in mice. Pflugers Archiv: European journal of physiology. 2009;457(6):1199–206. Epub 2008/10/08. 10.1007/s00424-008-0590-6 [DOI] [PubMed] [Google Scholar]
- 54.Florian JA, Kosky JR, Ainslie K, Pang Z, Dull RO, Tarbell JM. Heparan sulfate proteoglycan is a mechanosensor on endothelial cells. Circulation research. 2003;93(10):e136–42. 10.1161/01.RES.0000101744.47866.D5 [DOI] [PubMed] [Google Scholar]
- 55.Weinbaum S, Zhang X, Han Y, Vink H, Cowin SC. Mechanotransduction and flow across the endothelial glycocalyx. Proceedings of the National Academy of Sciences of the United States of America. 2003;100(13):7988–95. 10.1073/pnas.1332808100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Pahakis MY, Kosky JR, Dull RO, Tarbell JM. The role of endothelial glycocalyx components in mechanotransduction of fluid shear stress. Biochemical and biophysical research communications. 2007;355(1):228–33. 10.1016/j.bbrc.2007.01.137 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Tarbell JM, Ebong EE. The endothelial glycocalyx: a mechano-sensor and -transducer. Science signaling. 2008;1(40):pt8 10.1126/scisignal.140pt8 [DOI] [PubMed] [Google Scholar]
- 58.Constantinescu AA, Vink H, Spaan JA. Endothelial cell glycocalyx modulates immobilization of leukocytes at the endothelial surface. Arteriosclerosis, thrombosis, and vascular biology. 2003;23(9):1541–7. Epub 2003/07/12. 10.1161/01.ATV.0000085630.24353.3D [DOI] [PubMed] [Google Scholar]
- 59.Rops AL, van den Hoven MJ, Baselmans MM, Lensen JF, Wijnhoven TJ, van den Heuvel LP, et al. Heparan sulfate domains on cultured activated glomerular endothelial cells mediate leukocyte trafficking. Kidney international. 2008;73(1):52–62. 10.1038/sj.ki.5002573 [DOI] [PubMed] [Google Scholar]
- 60.Mulivor AW, Lipowsky HH. Role of glycocalyx in leukocyte-endothelial cell adhesion. American journal of physiology Heart and circulatory physiology. 2002;283(4):H1282–91. Epub 2002/09/18. 10.1152/ajpheart.00117.2002 [DOI] [PubMed] [Google Scholar]
- 61.Chappell D, Dorfler N, Jacob M, Rehm M, Welsch U, Conzen P, et al. Glycocalyx protection reduces leukocyte adhesion after ischemia/reperfusion. Shock. 2010;34(2):133–9. Epub 2010/07/17. 10.1097/SHK.0b013e3181cdc363 [DOI] [PubMed] [Google Scholar]
- 62.Nieuwdorp M, Holleman F, de Groot E, Vink H, Gort J, Kontush A, et al. Perturbation of hyaluronan metabolism predisposes patients with type 1 diabetes mellitus to atherosclerosis. Diabetologia. 2007;50(6):1288–93. 10.1007/s00125-007-0666-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Shafat I, Ilan N, Zoabi S, Vlodavsky I, Nakhoul F. Heparanase levels are elevated in the urine and plasma of type 2 diabetes patients and associate with blood glucose levels. PloS one. 2011;6(2):e17312 10.1371/journal.pone.0017312 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.van den Hoven MJ, Rops AL, Bakker MA, Aten J, Rutjes N, Roestenberg P, et al. Increased expression of heparanase in overt diabetic nephropathy. Kidney international. 2006;70(12):2100–8. 10.1038/sj.ki.5001985 [DOI] [PubMed] [Google Scholar]
- 65.Garsen M, Rops AL, Dijkman H, Willemsen B, van Kuppevelt TH, Russel FG, et al. Cathepsin L is crucial for the development of early experimental diabetic nephropathy. Kidney international. 2016. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All relevant data are within the paper.