Abstract
We have previously demonstrated that Mycobacterium bovis BCG-specific immunoglobulin G antibodies in lymphocyte secretions (ALS) can be employed as a marker for active tuberculosis (TB). We aimed to determine whether the ALS method allows detection of subclinical TB infection in asymptomatic individuals. A prospective study of family contacts (FCs) of patients with active TB and healthy controls was performed. Thirteen of 42 FCs had high ALS responses, including 6 FCs who subsequently developed active TB. No correlation was observed between the tuberculin skin test and the ALS responses in the FCs (r = 0.1, P = 0.23). Among patients with active TB, BCG-specific ALS responses steadily declined from the time of diagnosis through 6 months following antimycobacterial chemotherapy (P = 0.001). The ALS assay enabled detection of infection in exposed symptom-free contacts, who are at greater risk for developing active TB. The method may also allow discrimination between effective treatment of active infection and suboptimal response to therapy.
With the emergence of multidrug-resistant Mycobacterium tuberculosis and the pandemic of human immunodeficiency virus, tuberculosis (TB) is a reemerging infectious disease concern worldwide. About one-third of the world's population is believed to be latently infected with M. tuberculosis (7). Immunocompetent individuals with M. tuberculosis infection have a 10% risk during their lifetimes of developing active disease, becoming new foci for transmission, with half of this risk being in the first 2 years after exposure (5, 25). It is therefore important to identify people at risk for recent infection with M. tuberculosis, i.e., close contacts of patients with infectious pulmonary TB, persons having clinical conditions associated with an increased risk for progression to active TB (for example, human immunodeficiency virus infection, injection drug use, and fibrotic lesions on chest radiograms), children, and those who are in an early active phase of their infection (1, 4).
Determination of the prevalence of latent infection is helpful for better understanding the epidemiology of TB and for designing and evaluating TB control strategies (1). The accurate diagnosis of latent or subclinical TB infection is an important component of many TB control programs and depends largely on tuberculin skin testing (TST). In developed countries, in the absence of a better method, the TST using M. tuberculosis purified protein derivative (PPD) is currently used nearly exclusively. However, in developing countries with high rates of M. bovis BCG vaccine use, TST is unreliable because of the broad antigenic cross-reactivity of PPD with environmental nontuberculous strains and the BCG vaccine. About 10 to 20% of patients with proven TB and without any apparent immunosuppression have negative TST results (13, 20). The sensitivity of TST is low, especially in critically ill and immunosuppressed patients with disseminated TB (14). Therefore, there is substantial need for methods to accurately discriminate between latent and active TB infections in regions where the disease is endemic.
Recently we developed a rapid diagnostic method for the identification of patients with active TB (22). The technique allowed differentiation between BCG-vaccinated healthy subjects and patients with symptomatic M. tuberculosis infection. Our aim in the present study was to further investigate the potential use of the method involving antibodies in lymphocyte secretions (ALS) for detection of subclinical TB infection in exposed asymptomatic contacts.
MATERIALS AND METHODS
Study subjects and sampling.
Adult patients with suspected pulmonary TB (with <4 weeks of antimycobacterial drug treatment) cared for at the National Institute of Diseases of the Chest and Hospital (NIDCH) in Dhaka, Bangladesh, were prospectively studied as index cases at the time of enrollment (day 1) and 2 and 6 months later (these patients had been involved in a previous study; see reference 22). The diagnosis of TB in the index cases was established by clinical presentation, chest radiograph examination, and positive sputum smear results as well as the ALS assay (22). The clinical evaluation signs of TB included lung opacity, pyrexia, weight loss, and a high erythrocyte sedimentation rate (ESR). All patients received standard therapy that included rifampin, isoniazid, pyrazinamide, and ethambutol. Signed informed consent was obtained from all patients and their household contacts and healthy controls in accordance with the guidelines of the Ethical Review Committee of ICDDR,B and the Swedish Heart and Lung Foundation. The study was approved by the Ethical Review Committee of ICDDR,B.
One or two close family contacts (FCs) per patient (usually the spouse and/or family members living in the same household) were enrolled in the study and monitored prospectively at 2 and 6 months. At the time of entry into the study, FCs with active TB were excluded by clinical examination, sputum microscopy, and chest radiographs. Twenty-five healthy laboratory personnel (with no known exposure to M. tuberculosis) from ICDDR,B were selected as healthy controls from Bangladesh (BHCs), an area where TB is endemic. Additionally, 10 healthy laboratory personnel were enrolled as healthy controls from the Karolinska Institutet in Sweden (SHCs), an area where TB is not endemic. A TST (0.1 ml, 5 U of tuberculin; Sigma Chemical Co., St. Louis, Mo.) was performed on the FCs and BHCs only. To determine if a patient had been vaccinated for TB, a medical history was obtained from each patient; each patient was also inspected for a scar that typically forms after previous vaccination with BCG. Since some of the FCs developed active disease at later times, all FCs (excluding dropouts) were monitored for another 4 months for further evaluation (day 300). Active TB was defined in FCs by the presence of a chronic cough for >3 weeks, with chest radiographic abnormalities, fever, high ESR, and positive sputum microscopy (acid-fast bacilli [AFB]).
Blood was collected from the index cases and FCs at the day of enrollment (day 1) and at 2 and 6 months after enrollment. In addition, single blood specimens were obtained from BHCs and SHCs. Sputum specimens were collected from all TB patients and from FCs when TB was suspected, and specimens were subjected to sputum microscopy (AFB smear) and culture for M. tuberculosis on Lowenstein-Jensen medium by use of standard culture techniques. The ESR was measured in blood samples from patients and FCs only by the Westergren technique (15).
BCG-specific immunoglobulin G (IgG) ALS.
The ALS assay was performed as described earlier (22). In brief, peripheral blood mononuclear cells were separated from whole blood by differential centrifugation and suspended in 24-well tissue culture plates (Costar, Cambridge, Mass.) in RPMI 1640 medium (GIBCO BRL, Grand Island, N.Y.) containing 10% fetal bovine serum (GIBCO), 2 mM l-glutamine, and a 1% concentration of an amphotericin B-penicillin-streptomycin mixture (Sigma). Cells (106 cells/ml) were incubated at 37°C with 5% CO2, and the culture supernatant was collected at 72 h postincubation. A cocktail of protease inhibitors (0.2 μM 4-aminoethyl benzenesulfonyl fluoride, 1 μg of aprotinin per ml, 10 μM leupeptin, 1 mg of sodium azide per ml in phosphate-buffered saline) was added to the supernatant, and samples were stored at −70°C until used for assay.
With BCG vaccine used as the antigen (freeze-dried glutamate-BCG vaccine for intradermal use, lot no. 1861 [Japan BCG Laboratories, Tokyo, Japan]; no preservatives added, saline used as the diluent), IgG titers were measured in supernatants by an enzyme-linked immunosorbent assay as described previously (22). Microtiter plates (Nunc Maxisorp) were coated with BCG vaccine (1 μg/well in carbonate buffer, pH 9.8) and after overnight incubation at 4°C were blocked with 10% fetal bovine serum in phosphate-buffered saline (pH 7.2) at 37°C for 60 min. Following a wash, lymphocyte supernatants of appropriate dilutions were added and incubated for 2 h at 37°C. After another wash, the plates were incubated with rabbit anti-human IgG-horseradish peroxidase conjugate for 2 h at room temperature. Finally, the plates were developed with the substrate O-phenylenediamine, and the optical density was measured at 492 nm after the enzyme reaction was stopped. Antigen-specific responses were expressed as relative titers, which were defined as the optical density multiplied by the dilution factor of the sample.
In a previous study of verified TB cases, the cutoff for a positive ALS response was determined by constructing a receiver-operator characteristic curve from the BCG- and PPD-specific ALS responses, comparing patients with TB and healthy controls (22). Selection of the best cutoff point was based on the level at which the accuracy was maximum. The best cutoff was found to be 0.42 for BCG-specific responses at a sensitivity of 93% and a specificity of 80%, which was also used in the present study.
Statistical analysis.
Statistical analyses were carried out using SigmaStat software 2.03 (Jandel Scientific, San Rafael, Calif.). Comparisons within groups were made with the one-way analysis of variance (ANOVA) or ANOVA of ranks as appropriate. Comparisons between groups were done using a t test when applicable. Correlation of data was analyzed by Spearman's rank order test. P values of ≤0.05 were considered significant.
RESULTS
Demography of subjects.
Forty patients (geometric mean [GM] age, 32 years; range, 30.36 to 33 years) with suspected pulmonary TB were recruited from the NIDCH. The day of enrollment was considered day 1. Among TB index cases, 5 were lost to follow-up at the 2-month time point (leaving 35 patients), and 10 more were lost at 6 months (leaving 25). A total of 42 close FCs (GM age, 34.3 years; range, 32.7 to 35.9 years) of the TB patients were recruited in the study, of which 17 were lost to follow-up at 2 months (leaving 25 FCs), and 5 more were lost at 6 months (leaving 20). The high attrition rate was due to the fact that most patients and their FCs came from areas of Bangladesh that are quite remote from the national TB hospital situated in Dhaka. A total of 35 healthy controls (25 BHCs and 10 SHCs; GM age, 30.7 years; range, 29.8 to 31.56 years) were enrolled in the study. Twenty-four of 42 FCs, 24 of 25 BHCs, and 5 of 10 SHCs were BCG vaccinated in childhood.
ALS responses in FCs.
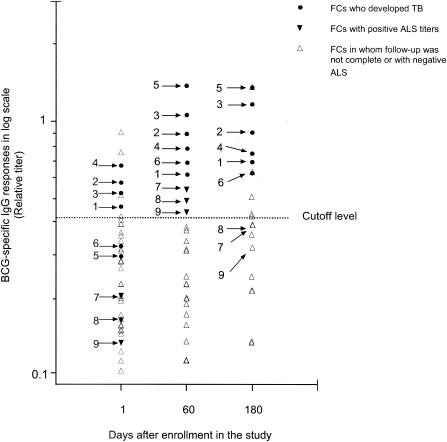
Out of 42 FCs at day 1, 8 had high BCG-specific ALS responses, though chest radiographs were clear for all (Fig. 1). Five of these FCs returned for the 2-month follow-up. They provided sputum specimens for smear examinations and cultures, but none of the specimens were positive (Table 1). However, within the next 4 months, four of these FCs developed early symptoms suggestive of TB (FCs 1 to 4) and returned to the hospital for evaluation. AFB were seen, and M. tuberculosis was isolated from the sputum of FCs 1 and 2, while culture and smear results for the other two FCs remained negative at 6 months (Table 1). However, within the next two months, the sputum smear microscopy results for FCs 3 and 4 were found to be positive, and these FCs were thus classified as having pulmonary TB. The fifth person did not return for further evaluation.
FIG. 1.
BCG-specific IgG ALS responses (relative titers) in lymphocyte secretions in FCs of patients with active TB (index cases) on day 1 and at 2 and 6 months after enrollment. Nine FCs (arrows labeled 1 to 9) who had specific IgG ALS responses above the cutoff level at the different time intervals are identified. FCs 1 to 6 later developed clinical symptoms and were given antimycobacterial treatment. FCs 7 to 9 were sputum culture negative and did not develop TB; their ALS responses were below the cutoff at 6 months. Three additional contacts had high ALS responses at day 180 and were not monitored thereafter.
TABLE 1.
Clinical features of nine family contacts with BCG-specific IgG titers above the cutoff level (0.42)a
| FC no. | TST result (mm) | Presence or duration of chronic cough at:
|
Sputum smear/culture results at:
|
Radiological sign at 6 mo | BCG-specific IgG (relative titer) at:
|
||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Day 1 | 6 mo | Day 1 | 2 mo | 6 mo | 8 to 10 mo | Day 1 | 2 mo | 6 mo | |||
| 1 | 17 | Absent | ≥5 wk | −/− | −/− | +/+ | ND | Infiltration | 0.44 | 0.63 | 0.72 |
| 2 | 20 | Absent | 7 wk | −/− | −/− | +/+ | ND | Infiltration | 0.57 | 0.91 | 0.9 |
| 3 | 15 | Absent | ≥4 wk | −/− | −/− | −/− | +/+ | Infiltration | 0.53 | 1.21 | 1.03 |
| 4 | 18 | Absent | 4-5 wk | −/− | −/− | −/− | +/+ | Doubtful | 0.69 | 0.78 | 0.74 |
| 5 | 14 | Absent | 5-6 wk | −/− | −/− | +/+ | ND | Infiltration | 0.30 | 1.38 | 1.1 |
| 6 | 12 | Absent | ≥5 wk | −/− | −/− | −/+ | ND | Infiltration | 0.33 | 0.69 | 0.65 |
| 7 | 18 | Absent | Absent | −/− | −/− | −/− | −/− | Clear | 0.22 | 0.55 | 0.39 |
| 8 | 16 | Absent | Absent | −/− | −/− | −/− | −/− | Clear | 0.17 | 0.48 | 0.39 |
| 9 | 12 | Absent | Absent | −/− | −/− | −/− | −/− | Clear | 0.14 | 0.43 | 0.33 |
FCs 1 to 6 actually developed TB with sputum smear conversion. FCs 7 to 9 had high BCG-specific IgG titers at 2 months that declined by the follow-up at 6 months. ND, not done.
Five additional FCs who were negative by the ALS assay at day 1 became positive at 2 months. At 6 months, among these five FCs (FCs 5 to 9), FCs 5 and 6 showed symptoms of TB, and their sputum was positive for AFB. FCs 1 to 6 were provided antimycobacterial therapy. FCs 7, 8, and 9 did not become ill (Fig. 1). Instead, at the follow-up visit at 6 months, the ALS responses of these FCs decreased to below the cutoff level, and examinations of their sputum obtained at 2 and 6 months remained negative for AFB. Two additional FCs who had negative ALS responses at the earlier times were ALS response positive at day 180. However, they were not monitored after day 180, and the health status of these FCs is not known.
None of the 11 FCs with an ALS response lower than the cutoff value was found to develop active TB within the study period. None of the healthy controls (neither BHCs nor SHCs) were found to be ALS response positive, and the responses in the two groups were comparable (P = 0.4).
ALS responses in index cases after 6 months.
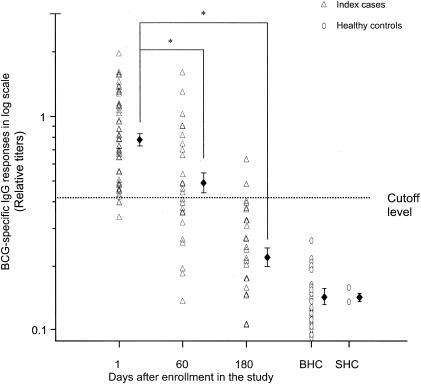
As shown previously (22), ALS responses in index cases were significantly higher at day 1 than those in BHCs (GM relative titer, 0.15; range, 0.13 to 0.15) or SHCs (GM relative titer, 0.09; range, 0.07 to 0.11) (P = 0.001) (Fig. 2). A gradual decline in relative titers of BCG-specific antibodies was found in index cases from day 1 (GM, 0.77; range, 0.73 to 0.83) to 2 months, and by 6 months, ALS antibody titers had declined significantly (GM, 0.22; range 0.2 to 0.24; P = 0.001) (Fig. 2), although the titers at 6 months remained significantly higher than those of controls. Among the seven patients who were infected with multidrug-resistant strains of M. tuberculosis, two patients did not improve clinically after 5 months of antimycobacterial therapy (Table 2), and their ALS responses remained high at 6 months. Therapy was modified after 5 months. When ALS responses were measured at 10 months, they were found to have decreased substantially, concomitant with clinical improvement (ALS titers of 0.237 for index case 00650 and 0.217 for index case 00530).
FIG. 2.
Comparison of BCG-specific IgG ALS responses (relative titers) in lymphocyte secretions from index cases at different time points during the course of the disease. Short bars represent GM titers of specific IgG for the groups. Specific IgG ALS responses at 2 and 6 months were significantly lower than those on day 1 (marked by asterisks). Patients had significantly higher ALS responses at all time points than BHC (P = 0.001) and SHC (P = 0.009). Statistical comparison between groups was performed using the one-way ANOVA or ANOVA on ranks.
TABLE 2.
ALS responses in index cases infected with multidrug-resistant strains of M. tuberculosis during the course of the study period
| Index case no. | Sputum smear/culture result at:
|
ALS responsea at:
|
||||
|---|---|---|---|---|---|---|
| Day 1 | 2 mo | 6 mo | Day 1 | 2 mo | 6 mo | |
| 00810 | +/+ | −/− | −/− | 0.86 | 0.7 | 0.35 |
| 00324 | +/+ | +/+ | −/− | 0.81 | 1.11 | 0.21 |
| 00889 | +/+ | −/− | −/− | 0.46 | 0.48 | 0.37 |
| 00530 | +/+ | +/+ | +/+ | 0.45 | 1.59 | 0.63 |
| 00546 | +/+ | −/− | −/− | 1.54 | 0.65 | 0.15 |
| 00864 | +/+ | −/− | −/− | 0.77 | 0.51 | 0.35 |
| 00650 | +/+ | +/+ | −/+ | 0.85 | 0.73 | 0.48 |
ALS responses are expressed as BCG-specific IgG titers.
When the ALS responses of FCs at day 60 were compared with those of patients at day 1 (before treatment was initiated), the difference was not significant (P = 0.058).
TST results for FCs and healthy controls.
The diameters of induration resulting from TST were measured after 72 h, and the TST results were expressed in millimeters of induration. In accordance with the guidelines in Bangladesh, a TST response was interpreted as positive for TB infection in the absence of any signs or symptoms of active TB when an induration diameter of ≥10 mm was found (19). Based on the above definition, 55.5% of FCs (25 of 42) were positive by TST (≥10 mm). Among 25 TST-positive FCs, 9 had high ALS responses, while among the 17 TST-negative FCs, 4 had high ALS responses. Among the 11 BCG-nonvaccinated FCs, 6 were TST positive compared with 19 of 31 BCG-vaccinated FCs. The GM diameter of the TST induration in HBCs was 11.25 mm with a range of 10.6 to 12 mm. Out of 25 BHCs, 5 were negative by TST. The GM ALS titer for BHCs was 0.15 (range, 0.14 to 0.16).
TST responses in FCs and healthy controls were compared with the ALS responses. No significant correlation was seen between the TST and ALS responses in the FCs (r = 0.1, P = 0.23) or the BHCs (r = 0.02, P = 0.24).
DISCUSSION
We have recently reported a novel technique to detect broad anti-TB-specific antibody responses in order to rapidly identify patients with active TB. This technique allows discrimination of active TB from non-TB illness in patients in whom TB is part of the differential diagnosis, as well as from BCG vaccine-induced immune responses (22). In the present study, we report the potential use of this method for identifying subclinical TB infection in asymptomatic contacts of TB index cases who later progressed to active TB.
Household FCs are at high risk for developing active TB. It is important to evaluate prospectively whether FCs with high anti-BCG-specific IgG ALS responses are at even higher risk of developing active TB and are likely to benefit from preventive therapy. Thirteen FCs had high ALS responses but were asymptomatic at the first and/or second presentations. Though there was a substantial decline in the number of FCs in subsequent follow-up visits, at least six of these FCs became symptomatic and developed illnesses compatible with active TB (with AFB smear or culture positivity). High ALS responses in asymptomatic FCs may indicate recent subclinical infection with M. tuberculosis that may progress to active TB. Unfortunately, due to a high dropout rate, it was not possible to determine if there were individuals with negative ALS responses who developed disease later or individuals with positive ALS responses who did not develop active TB. Thus, data in the present study do not allow for extracting sensitivity and specificity figures. Long-term systematic follow-up (1 to 2 years) of a larger cohort of patients and FCs would be needed to evaluate the specificity, sensitivity, and predictive value of the ALS assay for development of active TB.
The finding of positive TST readings in some FCs who did not exhibit high ALS responses was not unexpected. This could occur due to exposure to environmental or atypical mycobacteria or to BCG vaccination (16). Earlier studies have shown that about 50% of household FCs were positive by TST, and some contacts did not become ill (10). On the other hand, among patients with culture-confirmed TB and no apparent immunosuppression, 10 to 20% have negative TST results (14, 20). The relationship between the size of the TST induration after M. tuberculosis infection and the risk of active TB is not well defined (1, 9). Many studies have shown that the protection conferred by BCG vaccination does not depend on the degree of tuberculin sensitivity induced by the vaccine (3, 6, 9, 11). A study using BCG-vaccinated mice showed that delayed-type hypersensitivity responses and protective immunity were dissociated phenomena (21). Many close contacts of TB index cases do not develop active TB, possibly because they possess protective immunity against TB (25). It is thus conceivable that after frequent exposure to index cases and despite having a transient increase in ALS responses at 2 months, FCs 7 to 9 did not develop active TB. However, the present state of knowledge does not permit discrimination between exposed persons who are at risk for developing active TB and those who have protective immunity. In vitro correlates of protective immunity to mycobacteria have yet to be identified (25).
T-cell-specific gamma interferon (IFN-γ) responses to M. tuberculosis-specific antigens (ESAT-6 and CFP-10) have been used to detect latent TB infection and were found to be highly specific when tested in both BCG-vaccinated and -nonvaccinated subjects (2). However, about one-third of these TST-positive subjects did not have detectable responses to ESAT-6 and CFP-10; this result was similar to the proportion of TST-positive subjects in the present study that did not have a positive ALS response. Since a positive TST response can occur after exposure to nontuberculous mycobacteria, the use of a positive TST response as the “gold standard” for indicating latent TB could be one reason for the discrepancy. In regions where TB is endemic, FCs of TB patients have higher ESAT-6-specific T-cell responses than the patients with active disease themselves (17, 27), thereby limiting the use of the method to countries where the disease is not endemic. However, many studies have reported that this defect in IFN-γ production is a feature of severe TB when patients are immunocompromised (12, 26). Neither the TST nor the IFN-γ responses reliably differentiated among active TB, remission from TB, and latent TB. The innate sensitivity of the TST for detecting latent TB is not known because of a lack of a gold standard test for latent infection. However, the modified ESAT-6-specific IFN-γ spot-forming cell method enabled the identification of patients with active TB (24) as well as subclinical or latently infected TST-positive healthy FCs of the patients (8, 18).
Patients with active TB had significantly higher IgG ALS responses than their FCs, and successful anti-TB chemotherapy resulted in a progressive decline in the ALS responses and normalization within 6 months. The findings of this study suggest that the ALS method possibly allows for discrimination between effectively treated active infection and suboptimal response to therapy and thus may be applicable for monitoring therapeutic responses. Earlier studies have used antibody-secreting cell responses to TB-specific antigens that also allowed monitoring of therapeutic responses in TB patients (23). However, there are conflicting data regarding the frequency of IFN-γ-producing cells reacting to ESAT-6 in relation to disease monitoring, with both high (24) or low (17) levels reported for patients during anti-TB chemotherapy. Moreover, there is a long-term persistence of ESAT-6-specific responses in patients in remission from pulmonary TB in areas of endemicity, making it difficult to discriminate between latent TB and remission from TB (28).
In conclusion, our data support the prospects of using the ALS method for detecting subclinical infection in exposed contacts and for monitoring disease activity in TB patients. The technique may be useful in countries where latent TB infection and BCG vaccination use are prevalent, as BCG vaccination is not a limiting factor. Furthermore, the testing of various antigens of M. tuberculosis for recognition by ALS is in progress, which may allow the identification of suitable antigens for increasing the sensitivity and specificity of this method. The potential applications of the ALS assay would include the evaluation of recent TB contacts in countries with high TB rates and in industrialized countries for contact tracing as well as for screening immigrants from countries where TB is endemic. Further studies will be needed to evaluate these possibilities as well as the potential usefulness of this test in clinical management of symptom-free subjects with positive TST results to determine whether prophylactic therapy is needed.
Acknowledgments
We are grateful to all patients, their FCs, and the healthy subjects who participated in the study. The help of the medical staff of the NIDCH is gratefully acknowledged.
The study was supported by the Gates-Government of Bangladesh Award, 2001, grants from the Swedish Agency for Research Cooperation with Developing Countries (Sida/SAREC Agreement support; grants 2002 to 2004), and the USAID (grant HRN-A-00-96-90005-00).
REFERENCES
- 1.Al Zahrani, K., H. Al Jahdali, and D. Menzies. 2000. Does size matter? Utility of size of tuberculin reactions for the diagnosis of mycobacterial disease. Am. J. Respir. Crit. Care Med. 162:1419-1422. [DOI] [PubMed] [Google Scholar]
- 2.Arend, S. M., A. C. F. Engelhard, G. Groot, K. de Boer, P. Andersen, T. H. M. Ottenhoff, and J. T. van Dissel. 2001. Tuberculin skin testing compared with T-cell responses to Mycobacterium tuberculosis-specific and nonspecific antigens for detection of latent infection in persons with recent tuberculosis contact. Clin. Diagn. Lab. Immunol. 8:1089-1096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Behr, M. A., and P. M. Small. 1997. Has BCG attenuated to impotence? Nature 389:133-134. [DOI] [PubMed] [Google Scholar]
- 4.Cohn, D. L. 2000. Treatment of latent tuberculosis infection: renewed opportunity for tuberculosis control. Clin. Infect. Dis. 31:120-124. [DOI] [PubMed] [Google Scholar]
- 5.Comstock, G. W. 1975. Frost revisited: the modern epidemiology of tuberculosis. Am. J. Epidemiol. 101:363-382. [DOI] [PubMed] [Google Scholar]
- 6.Comstock, G. W. 1988. Identification of an effective vaccine against tuberculosis. Am. Rev. Respir. Dis. 138:479-480. [DOI] [PubMed] [Google Scholar]
- 7.Dye, C., S. Scheele, P. Dolin, V. Pathania, and M. C. Raviglione. 1999. Consensus statement. Global burden of tuberculosis: estimated incidence, prevalence, and mortality by country. W.H.O. Global Surveillance and Monitoring Project. JAMA 282:677-686. [DOI] [PubMed] [Google Scholar]
- 8.Ewer, K., J. Deeks, L. Alvarez, G. Bryant, S. Waller, P. Andersen, P. Monk, and A. Lalvani. 2003. Comparison of T-cell-based assay with tuberculin skin test for diagnosis of Mycobacterium tuberculosis infection in a school tuberculosis outbreak. Lancet 361:1168-1173. [DOI] [PubMed] [Google Scholar]
- 9.Fine, P. E., J. A. Sterne, J. M. Ponnighaus, and R. J. Rees. 1994. Delayed-type hypersensitivity, mycobacterial vaccines and protective immunity. Lancet 344:1245-1249. [DOI] [PubMed] [Google Scholar]
- 10.Grzybowski, S., G. D. Barnett, and K. Styblo. 1975. Contacts of cases of active pulmonary tuberculosis. Bull. Int. Union Tuberc. 50:90-106. [PubMed] [Google Scholar]
- 11.Hart, P. D., I. Sutherland, and J. Thomas. 1967. The immunity conferred by effective BCG and vole bacillus vaccines, in relation to individual variations in induced tuberculin sensitivity and to technical variations in the vaccines. Tubercle 48:201-210. [Google Scholar]
- 12.Hirsch, C. S., Z. Toossi, C. Othieno, J. L. Johnson, S. K. Schwander, S. Robertson, R. S. Wallis, K. Edmonds, A. Okwera, R. Mugerwa, P. Peters, and J. J. Ellner. 1999. Depressed T-cell interferon-gamma responses in pulmonary tuberculosis: analysis of underlying mechanisms and modulation with therapy. J. Infect. Dis. 180:2069-2073. [DOI] [PubMed] [Google Scholar]
- 13.Holden, M., M. R. Dubin, and P. H. Diamond. 1971. Frequency of negative intermediate-strength tuberculin sensitivity in patients with active tuberculosis. N. Engl. J. Med. 285:1506-1509. [DOI] [PubMed] [Google Scholar]
- 14.Huebner, R. E., M. F. Schein, and J. B. Bass. 1993. The tuberculin skin test. Clin. Infect. Dis. 17:968-975. [DOI] [PubMed] [Google Scholar]
- 15.International Council for Standardization in Haematology, Expert Panel on Blood Rheology. 1993. ICSH recommendations for measurement of erythrocyte sedimentation rate. J. Clin. Pathol. 46:198-203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Judson, F. N., and R. A. Feldman. 1974. Mycobacterial skin tests in humans 12 years after infection with Mycobacterium marinum. Am. Rev. Respir. Dis. 109:544-547. [DOI] [PubMed] [Google Scholar]
- 17.Lalvani, A., P. Nagvenkar, Z. Udwadia, A. A. Pathan, K. A. Wilkinson, J. S. Shastri, K. Ewer, A. V. S. Hill, A. Mehta, and C. Rodrigues. 2001. Enumeration of T cells specific for RD1-encoded antigens suggests a high prevalence of latent Mycobacterium tuberculosis infection in healthy urban Indians. J. Infect. Dis. 183:469-477. [DOI] [PubMed] [Google Scholar]
- 18.Lalvani, A., A. A. Pathan, H. Durkan, K. A. Wilkinson, A. Whelan, J. J. Deeks, W. H. Reece, M. Latif, G. Pasvol, and A. V. Hill. 2001. Enhanced contact tracing and spatial tracking of Mycobacterium tuberculosis infection by enumeration of antigen-specific T cells. Lancet 357:2017-2021. [DOI] [PubMed] [Google Scholar]
- 19.Ministry of Health and Family Welfare, Government of Bangladesh. 1995. National guidelines for tuberculosis control. Government of Bangladesh, Dhaka, Bangladesh.
- 20.Nash, D. R., and J. E. Douglass. 1980. Anergy in active pulmonary tuberculosis. A comparison between positive and negative reactors and an evaluation of 5 TU and 250 TU skin test doses. Chest 77:32-37. [DOI] [PubMed] [Google Scholar]
- 21.Orme, I. M., and F. M. Collins. 1984. Adoptive protection of the Mycobacterium tuberculosis-infected lung. Dissociation between cells that passively transfer protective immunity and those that transfer delayed-type hypersensitivity to tuberculin. Cell. Immunol. 84:113-120. [DOI] [PubMed] [Google Scholar]
- 22.Raqib, R., J. Rahman, A. K. Kamaluddin, S. M. Kamal, F. A. Banu, S. Ahmed, Z. Rahim, P. K. Bardhan, J. Andersson, and D. A. Sack. 2003. Rapid diagnosis of active tuberculosis by detecting antibodies from lymphocyte secretions. J. Infect. Dis. 188:364-370. [DOI] [PubMed] [Google Scholar]
- 23.Sousa, A. O., A. Wargnier, Y. Poinsignon, N. Simonney, F. Gerber, F. Lavergne, J. L. Herrmann, and P. H. Lagrange. 2000. Kinetics of circulating antibodies, immune complex and specific antibody-secreting cells in tuberculosis patients during 6 months of antimicrobial therapy. Tuber. Lung Dis. 80:27-33. [DOI] [PubMed] [Google Scholar]
- 24.Ulrichs, T., P. Anding, S. Porcelli, S. H. E. Kaufmann, and M. E. Munk. 2000. Increased numbers of ESAT-6- and purified protein derivative-specific gamma interferon-producing cells in subclinical and active tuberculosis infection. Infect. Immun. 68:6073-6076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ulrichs, T., and S. H. Kaufmann. 2002. Mycobacterial persistence and immunity. Front. Biosci. 7:d458-d469. [DOI] [PubMed] [Google Scholar]
- 26.Ulrichs, T., M. E. Munk, H. Mollenkopf, S. Behr-Perst, R. Colangeli, M. L. Gennaro, and S. H. Kaufmann. 1998. Differential T cell responses to Mycobacterium tuberculosis ESAT6 in tuberculosis patients and healthy donors. Eur. J. Immunol. 28:3949-3958. [DOI] [PubMed] [Google Scholar]
- 27.Vekemans, J., C. Lienhardt, J. S. Sillah, J. G. Wheeler, G. P. Lahai, M. T. Doherty, T. Corrah, P. Andersen, K. P. W. J. McAdam, and A. Marchant. 2001. Tuberculosis contacts but not patients have higher gamma interferon responses to ESAT-6 than do community controls in The Gambia. Infect. Immun. 69:6554-6557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wu-Hsieh, B. A., C.-K. Chen, J.-H. Chang, S.-Y. Lai, C. H. H. Wu, W.-C. Cheng, P. Andersen, and T. M. Doherty. 2001. Long-lived immune response to early secretory antigenic target 6 in individuals who had recovered from tuberculosis. Clin. Infect. Dis. 33:1336-1340. [DOI] [PubMed] [Google Scholar]