The generation of immunity against viral pathogens is diverse; and immunity is formed from a complex interaction of the virus, host immunity, and the backdrop of the microenvironment in which interactions occur. Although the initial response to different viral pathogens is similar, these diverse interactions drive chronic immunologic responses to different points in the differentiation pathway responsible for the generation of helper and effector responses. It is important to understand why and how viral infections mediate activation and differentiation of the immune response differently, because it is these differences that may explain whether an infection will be controlled or not. In individuals infected with human immunodeficiency virus (HIV) type 1 (HIV-1), the T-cell memory responses that develop are distinctly different from the responses to other viruses that develop. Why this occurs and the importance of these differences are not fully understood. In this minireview the memory phenotypes of CD4 and CD8 T-cell populations in response to HIV-1 infection are discussed in relation to the memory phenotypes of CD4 and CD8 T-cell populations in response to other chronic viral infections. The recovery and persistence of memory cell responses after highly active antiretroviral therapy (HAART) are examined as well.
PHENOTYPIC MARKERS OF MEMORY CELL POPULATIONS
The development of naïve cells into chronic differentiated memory cells follows a series of progressive steps. Various phenotypic markers can delineate the sequence of differentiation events that occur. These markers have been used to study the development of differentiated cells in acute viral infections and chronic viral infections and the changes in the cell population that occur secondary to antiretroviral therapy for those individuals infected with HIV-1. The phenotypic markers most commonly used to define populations in these stages of differentiation are CD45RA, CD45RO, CCR7, CD62L, CD27, and CD28. CD45RA and CD45RO are relatively complementary markers, in that when the amount of CD45RA is elevated, CD45RO production is depressed; and when the amount of CD45RO is elevated, CD45RA is down-regulated. T cells that are CD45RA+ (CD45RO−) are either naïve or terminal effector cells. T cells that are CD45RA− (CD45RO+) are memory cells. When cells are proliferating, they down-regulate expression of CD45RA (1, 14, 25). Alternatively, cells that express CD45RO can revert to a CD45RA+ phenotype (34, 65). The coupling of tyrosine phosphatase activity and CD45 expression during T-cell activation has supported the role of CD45 in T-cell development (12, 37).
The efforts to define memory cell populations by phenotype have developed from attempts to correlate these markers and various functional activities, such as effector potential and proliferative capacity. For the most part, studies of the memory phenotype have been divided into those groups that use CCR7 or CD62L (l-selectin) in conjunction with CD45RA (CD45RO) or those groups that use CD27 in conjunction with CD45RA (CD45RO). The phenotypic descriptors obtained by using CCR7 or CD62L have been driven by early work with nonhuman species. In the rat, cell function could be discerned from the cell's circulation pattern (21). In a later study with sheep, researchers demonstrated that memory cells, as defined by the proliferative response to antigen and CD45 expression, circulated from the blood to peripheral tissues and then to the lymph nodes (32). In distinction, naïve cells were trafficked from blood to lymph nodes by the high endothelial venules (32). In murine CD4 cells (55), this circulation pattern provided a means to study various adhesion markers, such as l-selectin (CD62L), which is capable of distinguishing these populations. In humans, Sallusto and colleagues (52) evaluated the chemokine homing receptor CCR7 and made a case for the use of this marker in conjunction with CD45 isoforms to define memory cell populations. In that work they found that CD4 T lymphocytes that had the effector memory phenotype (CD45RA− CCR7−) expressed high levels of type I and II cytokines, whereas naïve cells (CD45RA+ CCR7+) or central memory cells (CD45RA− CCR7+) expressed interleukin-2 (IL-2) but did not express gamma interferon (IFN-γ) or type II cytokines. In the CD8 population the terminal effector cells (CD45RA+ CCR7−) expressed the largest amounts of perforin. However, work from other groups found significant effector function in T cells with an earlier memory phenotype, suggesting a less stringent linkage between phenotype and function. In one study phorbol myristate acetate and ionomycin were used to compare chemokine receptor expression and cytokine production in CD4+ T cells from blood and synovial fluid (27). Pertinent to the present discussion was the finding that a significant percentage of cells in the CCR7− subset and the CCR7+ subset of CD4+ cells produced IFN-γ (27). In another study CD4+ T cells were evaluated for cytokine production as a function of their expression of CD62L. In that work it was found that all IFN-γ-producing cells were in the CD62L− population (23). These cells were distributed to a greater extent in the CCR7− population but can be found in the CCR7+ population as well (52).
In addition, the description of terminal effectors defined by elaboration of effector molecules and no proliferative responses to antigen, as measured by shortened telomeres, has been brought into question. Recently, it has been demonstrated that CD8+ CD45RA+ CD27− CCR7− cells from cytomegalovirus (CMV)-infected individuals can up-regulate expression of CCR7 in the presence of cognate peptide and various cytokines and can proliferate well. This demonstrated that some cells with a previously defined “terminal” phenotype have proliferative capacity (62).
The other frequently used descriptors of memory cells are CD45RA and CD27 in context with CD28. As a lymphocyte-specific member of the tumor necrosis factor-nerve growth factor receptor family, it was reasonable to study CD27 as a participant in cell differentiation and/or survival, similar to other members in the family. Using this logic, Hintzen and colleagues (25) explored the use of CD27 as a differentiation marker. Importantly, they demonstrated that activation of peripheral blood lymphocytes down-modulated the expression of CD27. Cells that lost the ability to express CD27 did not regain the ability to express it under the culture conditions used. Similarly, it has been shown that isolated CD4+ T cells stimulated with phytohemagglutinin transiently up-regulate CD27 expression over the resting value but after 3 weeks are negative for CD27 expression (54). These data suggest that CD27 is a marker of cell differentiation and that once it is lost, it is not reexpressed. CD28 is a major costimulatory molecule involved in T-cell activation. Upon activation CD28 expression is lost (30, 58) but can be reexpressed (64). It is expressed early and is expressed on central memory cells. As differentiation proceeds, CD28 expression is lost from the cell (18).
CD4+ T-CELL MEMORY IN PBLS FROM HIV-1-INFECTED INDIVIDUALS
Efforts to view the development of immunologic responses to HIV-1 and differences between HIV-1 and other viral infections have been studied by a number of researchers using the CD45 and CCR7 phenotypes. Numerous studies have demonstrated an inverse relationship between viral load and peripheral blood lymphocyte (PBL) proliferative responses to antigen, whereas this relationship is not found with the production of IFN-γ. In order to further evaluate these findings, Palmer and colleagues (44) asked whether CD4+ T-cell responses to the HIV-1 gag antigen differed from CD4+ T-cell responses to CMV antigen in the same HIV-1-infected cohort. By stimulating cells with the relevant antigen and gating on the cytokine-producing cells, they evaluated the memory distribution using CD45RA and CCR7. They found that treatment-naïve infected individuals had a significantly higher proportion of IFN-γ-producing cells in the CD4 population that were CD45RA− CCR7− than did either those who were on suppressive HAART or those who were slow progressors. Interestingly, the percentage of IFN-γ-producing cells in the central memory population had a significant negative correlation as a function of the viral load, whereas cells in the effector population had a significant positive correlation as a function of viral load. The antigen-responsive CD4+ T cells directed against CMV antigen had a more differentiated phenotype than the antigen-responsive CD4+ T cells directed against the HIV-1 antigen.
In another study (67), the phenotypic markers CD27, CD28, and CCR7 were used to compare CD4+ T cells that produced cytokine in the presence of HIV-1 or CMV antigen. In that study the CD4+ T cells that were responsive to gag antigen were mostly early memory cells (CD27+ CD28+) and CCR7+, whereas those that were responsive to CMV antigen were mostly of the late memory phenotype (CD27− CD28−) and CCR7−, consistent with the findings of the previously described study (44). After 6 months on suppressive HAART, there was a general increase in the CD27+ CD28− memory phenotype and a decrease in the CD27− CD28− late memory phenotype (67).
CD8+ T-CELL MEMORY IN PBLS FROM HIV-1-INFECTED INDIVIDUALS
The availability of HIV-1-specific class I tetramers has provided a means to evaluate the memory phenotypes of CD8+ HIV-specific T cells directly. Using the tetramer complexes directed to CD8+ T cells that recognize specific HIV-1 amino acid sequences, Champagne and colleagues (11) determined that CD8+ T cells from HIV-1-infected individuals were found predominantly in the CD45RA− CCR7− population. On the other hand, CMV-specific cells were predominantly in the effector population (CD45RA+ CCR7−), whereas the HIV-specific CD8+ T cells were of the memory phenotype. Therefore, CMV infection is able to drive the differentiation of CD8+ T cells toward a later effector phenotype compared to the phenotype toward which they are driven by HIV-1 infection, in a manner similar to that seen in the CD4+ T-cell responses. In that study little proliferation to antigen was seen in the CCR7− phenotypes. Although consistent with other studies ( 23, 52), this finding has recently been brought into question by a number of laboratories in which the addition of an exogenous cytokine overrides the lack of CD28 costimulation and produces proliferation of CD8+ CD45RA+ CCR7− T cells (62, 66). This suggests that a favorable cytokine environment can drive proliferative responses in this cell population.
Studies similar to those described above have evaluated the lymphocyte differentiation phenotypes by using CD27 and CD28 for different viruses. In acute viral infection, activation markers are increased on CD8+ T cells that are antigen specific and proliferating (7, 49). The response to an acute viral infection seems to be the same whether it is HIV-1, CMV, Epstein-Barr virus (EBV), or hepatitis C virus. Cells stimulated with antigen first down-regulate the CD45RA+ phenotype and express the low-molecular-weight isoform, CD45RO+, while they maintain CD27 and CD28 expression. This is followed by a loss of CD28 expression, which leads to cells with an acute memory phenotype, CD45RO+ CD27+ CD28− (59). The loss of CD28 follows the loss of CCR7, since CD45RA− CD27+ CD28+ subsets contain both CCR7+ and CCR7− cells, whereas CD45RA− CD27+ CD28− or CD45RA− CD27− CD28− subsets contain only CCR7− cells (56).
The development of memory phenotypes in chronic viral infections differs, depending on the virus. In HIV-1-infected individuals the dominant phenotype for CD8+ T cells that recognize HIV antigen is CD45RO+ CD27+ (39). When the ratio of the effector CD45RO+ CD27− cell population to the larger memory CD45RO+ CD27+ cell population is monitored over time, it remains relatively constant or decreases (39, 61). This is in distinction to the same ratio for EBV-specific T cells, which increases over time, suggesting an enhancement in the CD27− population (61). The presence of CD27− cells may be important in HIV-1-infected individuals since it has been found that the greater the percentage of CD8+ CD27− T cells is, the slower the rate of progression to AIDS is (61). Additional support for the importance of fully differentiated T cells in HIV-1 disease comes from a recent study that evaluated the presence of CD45RA+ CCR7− CD8+ T-cell subsets in infected individuals who were able to control their viral loads (24). In that work it was shown that the HIV-specific CD8+ T-cell phenotype was predominantly CD45RA+ CCR7− in those infected individuals who were nonprogressors and was enhanced in those exhibiting viral control after structured treatment interruption (24). Those who were unable to control virus had CD8+ T cells of the CD45RA− CCR7− phenotype (24).
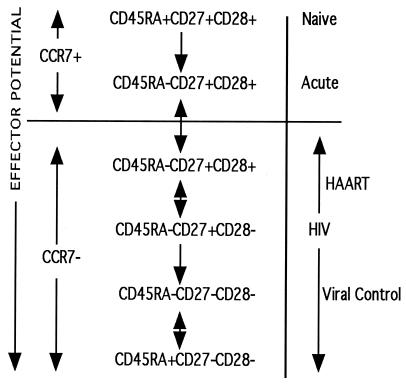
In another study, peptide-loaded tetramers were used to identify the dominant differentiation CD8+ T-cell phenotype for HIV-1, EBV, CMV, and hepatitis C virus in patients with acute and chronic infections (7). Appay and colleagues (7) found that in chronic infection the majority of EBV or hepatitis C virus tetramer-positive cells were enriched in the early, CD27+ CD28+, phenotype. CMV tetramer-positive cells from chronically infected individuals were enriched in the late, CD27− CD28−, phenotype. Antigen-specific CD8+ T cells from chronically HIV-1-infected individuals had a dominant CD27+ CD28− phenotype, similar to that in patients with the acute form of infection. Tussey and colleagues (59) also found similar phenotypic differences for chronic EBV, CMV, and HIV-1 infections. In addition, they demonstrated that the memory phenotype for antigen-specific CD8+ T cells from HIV-1-infected individuals on suppressive HAART tended toward the CD45RA− CD28+ memory phenotype for EBV and influenza viruses, two viruses with low antigenic burdens. Figure 1 summarizes the schema for the memory cell-to-effector cell differentiation pathway for the differentiation phenotypes described above.
FIG. 1.
CD8+ T-cell differentiation pathway. The differentiation of CD8+ T cells is displayed as a linear model with various points where phenotypes can move between differentiation stages (double-headed arrows), as described in the text. A single-headed arrow represents a unidirectional differentiation step. The cells of most individuals with HIV disease have the phenotype displayed and can move to a later or earlier phenotype under the conditions shown. The cellular effector potential is shown to be increasing as the differentiation phenotype moves toward a fully differentiated chronic infection phenotype.
As indicated above, phenotypic designations may not correlate completely with effector function. Additional data supporting this contention were obtained from the study by Appay and colleagues (7), in which moderate levels of expression of perforin and high levels of expression of granzyme A were found in the antigen-specific T cells in both chronic EBV and HIV-1 infections. Other groups have demonstrated that the phenotype does not completely parallel effector function. This may best be seen in acute infection, in which CD27+ T cells can produce a robust lysis of target cells (22, 56). Other factors, such as cell activation, participate in disease progression (20) as well as CD4-cell recovery after HAART (6) and confound the strict use of the memory phenotype as the sole predictor of outcome (7) as well. That said, the importance of driving differentiation to the effector phenotype has implications in controlling HIV-1 infection, as described above (24, 39, 61), suggesting that the fully differentiated cells tend to have a greater effector potential than earlier memory cell populations
T-CELL RECOVERY AFTER HAART
In many clinical trials with HIV-infected patients, it has been demonstrated that T-cell recovery of the memory cell population occurs rapidly in the plasma compartment, followed by a slow increase in the naïve cell population (8, 31, 38). Whether these changes in memory cell populations can be attributed to proliferation or redistribution from lymphoid organs after the initiation of HAART continues to be a point of discussion. Following the seminal studies on the effect of HAART on the viral load (26, 63), a flurry of letters (36, 53, 45) followed and suggested that although proliferation could explain the increase in CD4 counts, the redistribution of sequestered lymphocytes released into the blood after a reduction in the antigenic burden might explain the increase in the CD4 count as well. Although a mathematical argument suggested that this could be true (43), Bucy and colleagues (10) demonstrated this in a definitive manner by sampling lymph nodes before and after HAART. In that work they demonstrated a decrease in the lymph node cellularity following HAART. Before HAART there was a significantly greater number of CD8+ CD45+ CD62L+ cells in the lymph nodes than in the blood. After HAART there was no difference in the percentage of cells displaying the CD8+ CD45+ CD62L+ phenotype between these compartments, pointing to a possible redistribution of these cells. In a later study (29), changes in the distribution of the T-cell receptor Vβ in the blood, which occurred after HAART, demonstrated that both T-cell expansion and T-cell redistribution from lymph node compartments contribute to the changes in CD4 and CD8 T cells. Studies demonstrating that lymphocytes preferentially localize in anatomical sites commensurate with viral distribution preferences and antigenic burden (33) provide indirect evidence that redistribution occurs after HAART. Ellefsen and colleagues (16) showed that CD8+ T cells targeted HIV in lymph nodes, whereas this was not the case with CMV. In that work they evaluated both the phenotypic and the functional characteristics of CD8+ T cells directed against HIV-1 epitopes by tetramer staining. Analysis of CD8+ T cells that were CMV specific revealed that a significant percentage of CCR7− cells were in the blood compartment and that a low percentage were in the lymph nodes, whereas analysis of CD8+ T cells that were HIV-1 specific revealed that a high percentage in both the blood and the lymph node compartments had the CCR7− phenotype, demonstrating a proliferation or recruitment of these cells to the lymph nodes in HIV-1 disease.
Recovery of naïve cells after HAART occurs more slowly than that of the memory cell population. This recovery is in part secondary to cell redistribution as well as increased output by the thymus. By using deletion circles (remnants of T-cell receptor recombination), it has been demonstrated that the thymic output of naïve cells increases (15) upon the institution of HAART in HIV-1-positive individuals. The production of new cells is dependent on age (15) and the pre-HAART thymic volume (51). Changes in T-cell turnover contribute to the changes in the distributions of memory cell and naïve cell subsets after HAART initiation. When the in vivo kinetics of d-glucose-labeled T cells are evaluated, the rate of T-cell turnover for naïve HIV-1-infected individuals is greater than that for uninfected individuals (35). After HAART, T-cell turnover diminishes toward a level seen in infected controls (35).
Although memory cell numbers recover rapidly after the initiation of HAART, there is controversy as to whether memory cells persist while a patient is receiving fully suppressive therapy. This controversy revolves, in part, on whether the presence of antigen is required for the maintenance of immunologic memory. However, over the past decade a significant body of data has demonstrated the presence of long-lived antigen-independent memory cell populations (for a review, see reference 17). Data have demonstrated a decrease in the frequency of memory cells in HIV-1 disease, as determined by the frequency of cells that produce cytokine in the presence of HIV antigen, commensurate with the time of suppression during HAART (46). Results from proliferation experiments have been mixed in regard to demonstrating PBL memory responses to HIV antigens from suppressed donors (2, 5, 9, 13, 47, 48, 50, 57). The differences between these findings may be one of assay sensitivity rather than one of an inability to respond to antigen (28). A number of studies have evaluated cytotoxic T-lymphocyte (CTL) specificity in PBLs from individuals during structured interruption of suppressive HAART in HIV-1-infected individuals (4, 42). These CTLs were shown to have the same specificity as the CTLs studied before treatment with HAART. Such studies demonstrate the persistence of memory cell responses. The persistence of memory CD4+ T cells is important, since the generation of protective CTLs is dependent on a vigorous CD4 response (3, 4, 41). The continued existence of antigen-specific memory CD4+ T cells thereby provides a potential framework by which such a vigorous response is generated and provides a platform for vaccine research.
MAINTAINING MEMORY
It is clear that different viruses generate different chronic memory differentiation profiles. Although this is due in part to the antigenic burden, other variables participate in the development and the persistence of the memory response. In HIV-1 disease, understanding these variables is important in the development of vaccines. It is known that cytokines drive proliferative responses without antigen. In the mid-1990s it was shown that a cocktail of cytokines was able to support CD4-cell proliferation and affected the differentiation phenotype (60). In a later work (19) it was demonstrated that cytokines that bind to the receptors containing the γc chain, such as IL-7 and IL-15, support proliferation in the presence of dendritic cell cytokines or supernatants from lipopolysaccharide-stimulated dendritic cells in the absence of antigen. In that work IL-15 plus IL-7 and the dendritic cell cytokines drove the proliferation of memory cell subsets. In the central memory cell population (CD45RA− CCR7+), use of this cytokine mixture resulted in the development of cells with an effector phenotype (19). Oh and colleagues (40) recently addressed the relevance of IL-15 in maintaining immunity to HIV-1. In that study mice were immunized with recombinant vaccinia viruses that expressed IL-15 or IL-2 and the full-length HIV-IIIB gp160. In that system mice immunized and boosted with the IL-15-expressing vector had a greater number of tetramer-specific CD8+ T cells than mice treated with the HIV vector alone or boosted with IL-2 over time. The transfer of CD8+-specific cells from animals immunized with the IL-15-expressing vector to naïve animals demonstrated that a greater number of cells were maintained and that the cells continued to proliferate. These data, along with the findings of in vitro studies, demonstrate that both cognate and humoral factors are critical in the maintenance of HIV-1 immunity.
CONCLUSIONS
Over the past decade significant advances have been made in evaluating memory cell differentiation pathways for viruses. The reasons why one virus drives a dominant cell differentiation phenotype and another virus drives a different phenotype are areas of great interest. Even with the ability to tease out the potential contributions of cytokines and the interplay of cognate interactions and costimulatory signals in vitro, the task of translating these results into an understanding of viral interactions in the host is significant and difficult. For HIV-1 disease this problem is somewhat amplified owing to the lack of a small-animal model that allows work to be performed in a defined environment. It is hoped that the in vitro work, described above, that has evaluated the development of helper and effector T cells in individuals with HIV-1 disease can be used to target relevant therapeutic interventions that will lead to improved outcomes. This possibility will be tested over the coming years.
REFERENCES
- 1.Akbar, A. N., L. Terry, A. Timms, P. C. Beverley, and G. Janossy. 1988. Loss of CD45R and gain of UCHL1 reactivity is a feature of primed T cells. J. Immunol. 140:2171-2178. [PubMed] [Google Scholar]
- 2.Al-Harthi, L., J. Siegel, J. Spritzler, J. Pottage, M. Agnoli, and A. Landay. 2000. Maximum suppression of HIV replication leads to the restoration of HIV-specific responses in early HIV disease. AIDS 14:761-770. [DOI] [PubMed] [Google Scholar]
- 3.Altfeld, M., E. S. Rosenberg, R. Shankarappa, J. S. Mukherjee, F. M. Hecht, R. L. Eldridge, M. M. Addo, S. H. Poon, M. N. Phillips, G. K. Robbins, P. E. Sax, S. Boswell, J. O. Kahn, C. Brander, P. J. Goulder, J. A. Levy, J. I. Mullins, and B. D. Walker. 2001. Cellular immune responses and viral diversity in individuals treated during acute and early HIV-1 infection. J. Exp. Med. 193:169-180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Altfeld, M., J. van Lunzen, N. Frahm, X. G. Yu, C. Schneider, R. L. Eldridge, M. E. Feeney, D. Meyer-Olson, H. J. Stellbrink, and B. D. Walker. 2002. Expansion of pre-existing, lymph node-localized CD8+ T cells during supervised treatment interruptions in chronic HIV-1 infection. J. Clin. Investig. 109:837-843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Angel, J. B., K. G. Parato, A. Kumar, S. Kravcik, A. D. Badley, C. Fex, D. Ashby, E. Sun, and D. W. Cameron. 2001. Progressive human immunodeficiency virus-specific immune recovery with prolonged viral suppression. J. Infect. Dis. 183:546-554. [DOI] [PubMed] [Google Scholar]
- 6.Anthony, K. B., C. Yoder, J. A. Metcalf, R. DerSimonian, J. M. Orenstein, R. A. Stevens, J. Falloon, M. A. Polis, H. C. Lane, and I. Sereti. 2003. Incomplete CD4 T cell recovery in HIV-1 infection after 12 months of highly active antiretroviral therapy is associated with ongoing increased CD4 T cell activation and turnover. J. Acquir. Immune Defic. Syndr. 33:125-133. [DOI] [PubMed] [Google Scholar]
- 7.Appay, V., P. R. Dunbar, M. Callan, P. Klenerman, G. M. Gillespie, L. Papagno, G. S. Ogg, A. King, F. Lechner, C. A. Spina, S. Little, D. V. Havlir, D. D. Richman, N. Gruener, G. Pape, A. Waters, P. Easterbrook, M. Salio, V. Cerundolo, A. J. McMichael, and S. L. Rowland-Jones. 2002. Memory CD8+ T cells vary in differentiation phenotype in different persistent virus infections. Nat. Med. 8:379-385. [DOI] [PubMed] [Google Scholar]
- 8.Autran, B., G. Carcelain, T. S. Li, C. Blanc, D. Mathez, R. Tubiana, C. Katlama, P. Debre, and J. Leibowitch. 1997. Positive effects of combined antiretroviral therapy on CD4+ T cell homeostasis and function in advanced HIV disease. Science 277:112-116. [DOI] [PubMed] [Google Scholar]
- 9.Bart, P. A., G. P. Rizzardi, G. Tambussi, J. P. Chave, A. G. Chapuis, C. Graziosi, J. M. Corpataux, N. Halkic, J. Y. Meuwly, M. Munoz, P. Meylan, W. Spreen, H. McDade, S. Yerly, L. Perrin, A. Lazzarin, and G. Pantaleo. 2000. Immunological and virological responses in HIV-1-infected adults at early stage of established infection treated with highly active antiretroviral therapy. AIDS 14:1887-1897. [DOI] [PubMed] [Google Scholar]
- 10.Bucy, R. P., R. D. Hockett, C. A. Derdeyn, M. S. Saag, K. Squires, M. Sillers, R. T. Mitsuyasu, and J. M. Kilby. 1999. Initial increase in blood CD4+ lymphocytes after HIV antiretroviral therapy reflects redistribution from lymphoid tissues. J. Clin. Investig. 103:1391-1398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Champagne, P., G. S. Ogg, A. S. King, C. Knabenhans, K. Ellefsen, M. Nobile, V. Appay, G. P. Rizzardi, S. Fleury, M. Lipp, R. Forster, S. Rowland-Jones, R. P. Sekaly, A. J. McMichael, and G. Pantaleo. 2001. Skewed maturation of memory HIV-specific CD8 T lymphocytes. Nature 410:106-111. [DOI] [PubMed] [Google Scholar]
- 12.Charbonneau, H., N. K. Tonks, K. A. Walsh, and E. H. Fischer. 1988. The leukocyte common antigen (CD45): a putative receptor-linked protein tyrosine phosphatase. Proc. Natl. Acad. Sci. USA 85:7182-7186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Connick, E., M. M. Lederman, B. L. Kotzin, J. Spritzler, D. R. Kuritzkes, M. St. Clair, A. D. Sevin, L. Fox, M. H. Chiozzi, J. M. Leonard, F. Rousseau, J. D'Arc Roe, A. Martinez, H. Kessler, and A. Landay. 2000. Immune reconstitution in the first year of potent antiretroviral therapy and its relationship to virologic response. J. Infect. Dis. 181:358-363. [DOI] [PubMed] [Google Scholar]
- 14.De Jong, R., M. Brouwer, B. Hooibrink, T. Van der Pouw-Kraan, F. Miedema, and R. A. Van Lier. 1992. The CD27− subset of peripheral blood memory CD4+ lymphocytes contains functionally differentiated T lymphocytes that develop by persistent antigenic stimulation in vivo. Eur. J. Immunol. 22:993-999. [DOI] [PubMed] [Google Scholar]
- 15.Douek, D. C., R. D. McFarland, P. H. Keiser, E. A. Gage, J. M. Massey, B. F. Haynes, M. A. Polis, A. T. Haase, M. B. Feinberg, J. L. Sullivan, B. D. Jamieson, J. A. Zack, L. J. Picker, and R. A. Koup. 1998. Changes in thymic function with age and during the treatment of HIV infection. Nature 396:690-695. [DOI] [PubMed] [Google Scholar]
- 16.Ellefsen, K., A. Harari, P. Champagne, P. A. Bart, R. P. Sekaly, and G. Pantaleo. 2002. Distribution and functional analysis of memory antiviral CD8 T cell responses in HIV-1 and cytomegalovirus infections. Eur. J. Immunol. 32:3756-3764. [DOI] [PubMed] [Google Scholar]
- 17.Farber, D. L. 2003. Remembrance of antigens past: new insights into memory T cells. Scand. J. Immunol. 58:145-154. [DOI] [PubMed] [Google Scholar]
- 18.Fukada, K., Y. Sobao, H. Tomiyama, S. Oka, and M. Takiguchi. 2002. Functional expression of the chemokine receptor CCR5 on virus epitope-specific memory and effector CD8+ T cells. J. Immunol. 168:2225-2232. [DOI] [PubMed] [Google Scholar]
- 19.Geginat, J., F. Sallusto, and A. Lanzavecchia. 2001. 2001. Cytokine-driven proliferation and differentiation of human naive, central memory, and effector memory CD4+ T cells. J. Exp. Med. 194:1711-1719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Giorgi, J. V., Z. Liu, L. E. Hultin, W. G. Cumberland, K. Hennessey, and R. Detels. 1993. Elevated levels of CD38+ CD8+ T cells in HIV infection add to the prognostic value of low CD4+ T cell levels: results of 6 years of follow-up. The Los Angeles Center, Multicenter AIDS Cohort Study. J. Acquir. Immune Defic. Syndr. 6:904-912. [PubMed] [Google Scholar]
- 21.Gowan, J. L. 1959. The recirculation of lymphocytes from blood to lymph in the rat. J. Physiol. 146:54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hamann, D., P. A. Baars, M. H. Rep, B. Hooibrink, S. R. Kerkhof-Garde, M. R. Klein, and R. A. van Lier. 1997. Phenotypic and functional separation of memory and effector human CD8+ T cells. J. Exp. Med. 186:1407-1418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hengel, R. L., V. Thaker, M. V. Pavlick, J. A. Metcalf, G. Dennis, Jr., J. Yang, R. A. Lempicki, I. Sereti, and H. C. Lane. 2003. Cutting edge: l-selectin (CD62L) expression distinguishes small resting memory CD4+ T cells that preferentially respond to recall antigen. J. Immunol. 170:28-32. [DOI] [PubMed] [Google Scholar]
- 24.Hess, C., M. Altfeld, S. Y. Thomas, M. M. Addo, E. S. Rosenberg, T. M. Allen, R. Draenert, R. L. Eldrige, J. van Lunzen, H. J. Stellbrink, B. D. Walker, and A. D. Luster. 2004. HIV-1 specific CD8+ T cells with an effector phenotype and control of viral replication. Lancet 363:863-866. [DOI] [PubMed] [Google Scholar]
- 25.Hintzen, R. Q., R. de Jong, S. M. Lens, M. Brouwer, P. Baars, and R. A. van Lier. 1993. Regulation of CD27 expression on subsets of mature T-lymphocytes. J. Immunol. 151:2426-2435. [PubMed] [Google Scholar]
- 26.Ho, D. D., A. U. Neumann, A. S. Perelson, W. Chen, J. M. Leonard, and M. Markowitz. 1995. Rapid turnover of plasma virions and CD4 lymphocytes in HIV-1 infection. Nature 373:123-126. [DOI] [PubMed] [Google Scholar]
- 27.Kim, C. H., L. Rott, E. J. Kunkel, M. C. Genovese, D. P. Andrew, L. Wu, and E. C. Butcher. 2001. Rules of chemokine receptor association with T cell polarization in vivo. J. Clin. Investig. 108:1331-1339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kolber, M. A., and M. O. Saenz. 2003. Antigen-presenting cell modulation induces a memory response to p24 in peripheral blood leukocytes from human immunodeficiency virus-infected individuals. Clin. Diagn. Lab. Immunol. 10:757-763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kostense, S., F. M. Raaphorst, J. Joling, D. W. Notermans, J. M. Prins, S. A. Danner, P. Reiss, J. M. Lange, J. M. Teale, and F. Miedema. 2001. T cell expansions in lymph nodes and peripheral blood in HIV-1-infected individuals: effect of antiretroviral therapy. AIDS 15:1097-1107. [DOI] [PubMed] [Google Scholar]
- 30.Lake, R. A., R. E. O'Hehir, A. Verhoef, and J. R. Lamb. 1993. CD28 mRNA rapidly decays when activated T cells are functionally anergized with specific peptide. Int. Immunol. 5:461-466. [DOI] [PubMed] [Google Scholar]
- 31.Li, T. S., R. Tubiana, C. Katlama, V. Calvez, H. Ait Mohand, and B. Autran. 1998. Long-lasting recovery in CD4 T-cell function and viral-load reduction after highly active antiretroviral therapy in advanced HIV-1 disease. Lancet 351:1682-1686. [DOI] [PubMed] [Google Scholar]
- 32.Mackay, C. R., W. L. Marston, and L. Dudler. 1990. Naive and memory T cells show distinct pathways of lymphocyte recirculation. J. Exp. Med. 171:801-817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Masopust, D., V. Vezys, A. L. Marzo, and L. Lefrancois. 2001. Preferential localization of effector memory cells in nonlymphoid tissue. Science 291:2413-2417. [DOI] [PubMed] [Google Scholar]
- 34.McLean, A. R., and C. A. Michie. 1995. In vivo estimates of division and death rates of human T lymphocytes. Proc. Natl. Acad. Sci. USA 92:3707-3711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mohri, H., A. S. Perelson, K. Tung, R. M. Ribeiro, B. Ramratnam, M. Markowitz, R. Kost, A. Hurley, L. Weinberger, D. Cesar, M. K. Hellerstein, and D. D. Ho. 2001. Increased turnover of T lymphocytes in HIV-1 infection and its reduction by antiretroviral therapy. J. Exp. Med. 194:1277-1287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Mosier, D. E. 1995. HIV results in the frame: CD4+ cell turnover. Nature 375:193. (Letter.) [DOI] [PubMed] [Google Scholar]
- 37.Mustelin, T., K. M. Coggeshall, and A. Altman. 1989. Rapid activation of the T-cell tyrosine protein kinase pp56lck by the CD45 phosphotyrosine phosphatase. Proc. Natl. Acad. Sci. USA 86:6302-6306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Notermans, D. W., N. G. Pakker, D. Hamann, N. A. Foudraine, R. H. Kauffmann, P. L. Meenhorst, J. Goudsmit, M. T. Roos, P. T. Schellekens, F. Miedema, and S. A. Danner. 1999. Immune reconstitution after 2 years of successful potent antiretroviral therapy in previously untreated human immunodeficiency virus type 1-infected adults. J. Infect. Dis. 180:1050-1056. [DOI] [PubMed] [Google Scholar]
- 39.Ogg, G. S., S. Kostense, M. R. Klein, S. Jurriaans, D. Hamann, A. J. McMichael, and F. Miedema. 1999. Longitudinal phenotypic analysis of human immunodeficiency virus type 1-specific cytotoxic T lymphocytes: correlation with disease progression. J. Virol. 73:9153-9160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Oh, S., J. A. Berzofsky, D. S. Burke, T. A. Waldmann, and L. P. Perera. 2003. Coadministration of HIV vaccine vectors with vaccinia viruses expressing IL-15 but not IL-2 induces long-lasting cellular immunity. Proc. Natl. Acad. Sci. USA 100:3392-3397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Oxenius, A., D. A. Price, P. J. Easterbrook, C. A. O'Callaghan, A. D. Kelleher, J. A. Whelan, G. Sontag, A. K. Sewell, and R. E. Phillips. 2000. Early highly active antiretroviral therapy for acute HIV-1 infection preserves immune function of CD8+ and CD4+ T lymphocytes. Proc. Natl. Acad. Sci. USA 97:3382-3387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Oxenius, A., D. A. Price, H. F. Gunthard, S. J. Dawson, C. Fagard, L. Perrin, M. Fischer, R. Weber, M. Plana, F. Garcia, B. Hirschel, A. McLean, and R. E. Phillips. 2002. Stimulation of HIV-specific cellular immunity by structured treatment interruption fails to enhance viral control in chronic HIV infection. Proc. Natl. Acad. Sci. USA 99:13747-13752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pakker, N. G., D. W. Notermans, R. J. de Boer, M. T. Roos, F. de Wolf, A. Hill, J. M. Leonard, S. A. Danner, F. Miedema, and P. T. Schellekens. 1998. Biphasic kinetics of peripheral blood T cells after triple combination therapy in HIV-1 infection: a composite of redistribution and proliferation. Nat. Med. 4:208-214. [DOI] [PubMed] [Google Scholar]
- 44.Palmer, B. E., E. Boritz, and C. C. Wilson. 2004. Effects of sustained HIV-1 plasma viremia on HIV-1 gag-specific CD4+ T cell maturation and function. J. Immunol. 172:3337-3347. [DOI] [PubMed] [Google Scholar]
- 45.Phillips, A. N., C. A. Sabin, A. Mocroft, and G. Janossy. 1995. HIV results in the frame: antiviral therapy. Nature 375:195. (Letter.) [DOI] [PubMed] [Google Scholar]
- 46.Pitcher, C. J., C. Quittner, D. M. Peterson, M. Connors, R. A. Koup, V. C. Maino, and L. J. Picker. 1999. HIV-1-specific CD4+ T cells are detectable in most individuals with active HIV-1 infection, but decline with prolonged viral suppression. Nat. Med. 5:518-525. [DOI] [PubMed] [Google Scholar]
- 47.Pontesilli, O., S. Kerkhof-Garde, N. G. Pakker, D. W. Notermans, M. T. Roos, M. R. Klein, S. A. Danner, and F. Miedema. 1999. Antigen-specific T-lymphocyte proliferative responses during highly active antiretroviral therapy (HAART) of HIV-1 infection. Immunol. Lett. 66:213-217. [DOI] [PubMed] [Google Scholar]
- 48.Rizzardi, G. P., G. Tambussi, P. A. Bart, A. G. Chapuis, A. Lazzarin, and G. Pantaleo. 2000. Virological and immunological responses to HAART in asymptomatic therapy-naive HIV-1-infected subjects according to CD4 cell count. AIDS 14:2257-2263. [DOI] [PubMed] [Google Scholar]
- 49.Roos, M. T., R. A. van Lier, D. Hamann, G. J. Knol, I. Verhoofstad, D. van Baarle, F. Miedema, and P. T. Schellekens. 2000. Changes in the composition of circulating CD8+ T cell subsets during acute Epstein-Barr and human immunodeficiency virus infections in humans. J. Infect. Dis. 182:451-458. [DOI] [PubMed] [Google Scholar]
- 50.Rosenberg, E. S., J. M. Billingsley, A. M. Caliendo, S. L. Boswell, P. E. Sax, S. A. Kalams, and B. D. Walker. 1997. Vigorous HIV-1-specific CD4+ T cell responses associated with control of viremia. Science 278:1447-1450. [DOI] [PubMed] [Google Scholar]
- 51.Ruiz-Mateos, E., A. Rubio, A. Vallejo, R. De la Rosa, A. Sanchez-Quijano, E. Lissen, and M. Leal. 2004. Thymic volume is associated independently with the magnitude of short- and long-term repopulation of CD4+ T cells in HIV-infected adults after highly active antiretroviral therapy (HAART). Clin. Exp. Immunol. 136:501-506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Sallusto, F., D. Lenig, R. Forster, M. Lipp, and A. Lanzavecchia. 1999. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 401:708-712. [DOI] [PubMed] [Google Scholar]
- 53.Sprent, J., and D. Tough. 1995. HIV results in the frame: CD4+ cell turnover. Nature 375:194. (Letter.) [DOI] [PubMed] [Google Scholar]
- 54.Sugita, K., T. Hirose, D. M. Rothstein, C. Donahue, S. F. Schlossman, and C. Morimoto. 1992. CD27, a member of the nerve growth factor receptor family, is preferentially expressed on CD45RA+ CD4 T cell clones and involved in distinct immunoregulatory functions. J. Immunol. 149:3208-3216. [PubMed] [Google Scholar]
- 55.Tietz, W., and A. Hamann. 1997. The migratory behavior of murine CD4+ cells of memory phenotype. Eur. J. Immunol. 27:2225-2232. [DOI] [PubMed] [Google Scholar]
- 56.Tomiyama, H., T. Matsuda, and M. Takiguchi. 2002. Differentiation of human CD8+ T cells from a memory to memory/effector phenotype. J. Immunol. 168:5538-5550. [DOI] [PubMed] [Google Scholar]
- 57.Tortajada, C., F. Garcia, M. Plana, T. Gallart, M. J. Maleno, J. M. Miro, and J. M. Gatell. 2000. Comparison of T-cell subsets' reconstitution after 12 months of highly active antiretroviral therapy initiated during early versus advanced states of HIV disease. J. Acquir. Immune Defic. Syndr. 25:296-305. [DOI] [PubMed] [Google Scholar]
- 58.Trimble, L. A., L. W. Kam, R.S. Friedman, Z. Xu, and J. Lieberman. 2000. CD3zeta and CD28 down-modulation on CD8 T cells during viral infection. Blood 96:1021-1029. [PubMed] [Google Scholar]
- 59.Tussey, L. G., U. S. Nair, M. Bachinsky, B. H. Edwards, J. Bakari, K. Grimm, J. Joyce, R. Vessey, R. Steigbigel, M. N. Robertson, J. W. Shiver, and P. A. Goepfert. 2003. Antigen burden is major determinant of human immunodeficiency virus-specific CD8+ T cell maturation state: potential implications for therapeutic immunization. J. Infect. Dis. 187:364-374. [DOI] [PubMed] [Google Scholar]
- 60.Unutmaz, D., F. Baldoni, and S. Abrignani. 1995. Human naive T cells activated by cytokines differentiate into a split phenotype with functional features intermediate between naive and memory T cells. Int. Immunol. 7:1417-1424. [DOI] [PubMed] [Google Scholar]
- 61.van Baarle, D., S. Kostense, E. Hovenkamp, G. Ogg, N. Nanlohy, M. F. Callan, N. H. Dukers, A. J. McMichael, M. H. van Oers, and F. Miedema. 2002. Lack of Epstein-Barr virus- and HIV-specific CD27− CD8+ T cells is associated with progression to viral disease in HIV-infection. AIDS 16:2001-2011. [DOI] [PubMed] [Google Scholar]
- 62.van Leeuwen, E. M., L. E. Gamadia, P. A. Baars, E. B. Remmerswaal, I. J. ten Berge, and R. A. van Lier. 2002. Proliferation requirements of cytomegalovirus-specific, effector-type human CD8+ T cells. J. Immunol. 169:5838-5843. [DOI] [PubMed] [Google Scholar]
- 63.Wei, X., S. K. Ghosh, M. E. Taylor, V. A. Johnson, E. A. Emini, P. Deutsch, J. D. Lifson, S. Bonhoeffer, M. A. Nowak, B. H. Hahn, M. S. Saag, and G. M. Shaw. 1995. Viral dynamics in human immunodeficiency virus type 1 infection. Nature 373:117-122. [DOI] [PubMed] [Google Scholar]
- 64.Wierenga, E. A., M. Snoek, H. M. Jansen, J. D. Bos, R. A. van Lier, and M. L. Kapsenberg. 1991. Human atopen-specific types 1 and 2 T helper cell clones. J. Immunol. 147:2942-2949. [PubMed] [Google Scholar]
- 65.Wills, M. R., A. J. Carmichael, M. P. Weekes, K. Mynard, G. Okecha, R. Hicks, and J. G. Sissons. 1999. Human virus-specific CD8+ CTL clones revert from CD45ROhigh to CD45RAhigh in vivo: CD45RAhighCD8+ T cells comprise both naive and memory cells. J. Immunol. 162:7080-7087. [PubMed] [Google Scholar]
- 66.Wills, M. R., G. Okecha, M. P. Weekes, M. K. Gandhi, P. J. Sissons, and A. J. Carmichael. 2002. Identification of naive or antigen-experienced human CD8+ T cells by expression of costimulation and chemokine receptors: analysis of the human cytomegalovirus-specific CD8+ T cell response. J. Immunol. 168:5455-5464. [DOI] [PubMed] [Google Scholar]
- 67.Yue, F. Y., C. M. Kovacs, R. C. Dimayuga, P. Parks, and M. A. Ostrowski. 2004. HIV-1-specific memory CD4+ T cells are phenotypically less mature than cytomegalovirus-specific memory CD4+ T cells. J. Immunol. 172:2476-2486. [DOI] [PubMed] [Google Scholar]