Abstract
OBJECTIVE
To determine whether inpatient sleep duration and efficiency are associated with a greater risk of hyperglycemia in hospitalized patients with and without diabetes.
RESEARCH DESIGN AND METHODS
In this retrospective analysis of a prospective cohort study, medical inpatients ≥50 years of age were interviewed, and their charts were reviewed to obtain demographic data and diagnosis. Using World Health Organization criteria, patients were categorized as having normal blood glucose, impaired fasting blood glucose, or hyperglycemia based on morning glucose from the electronic health record. Wrist actigraphy measured sleep. Multivariable ordinal logistic regression models, controlling for subject random effects, tested the association between inpatient sleep duration and proportional odds of hyperglycemia versus impaired fasting blood glucose or impaired fasting blood glucose versus normal blood glucose in hospitalized adults.
RESULTS
A total of 212 patients (60% female and 74% African American) were enrolled. Roughly one-third (73, 34%) had diabetes. Objective inpatient sleep measures did not differ between patients with or without diabetes. In ordinal logistic regression models, each additional hour of in-hospital sleep was associated with an 11% (odds ratio 0.89 [95% CI 0.80, 0.99]; P = 0.043) lower proportional odds of a higher glucose category the next morning (hyperglycemia vs. elevated and elevated vs. normal). Every 10% increase in sleep efficiency was associated with an 18% lower proportional odds of a higher glucose category (odds ratio 0.82 [95% CI 0.74, 0.89]; P < 0.001).
CONCLUSIONS
Among medical inpatients, both shorter sleep duration and worse sleep efficiency were independently associated with greater proportional odds of hyperglycemia and impaired fasting glucose.
Introduction
Hyperglycemia is common among hospitalized patients, even in those patients who have not been previously diagnosed with diabetes. Although unrecognized diabetes may account for some of this, there is also the known phenomenon of stress hyperglycemia, which is elevated blood glucose attributed to acute illness. Hyperglycemia during hospitalization is associated with worse patient outcomes, including higher risk of myocardial infarction and stroke (1). The increased risks associated with hyperglycemia are not limited to patients with diabetes. Inpatients with new hyperglycemia in the hospital are more likely to be admitted to the intensive care unit, have longer lengths of stay, and are less likely to be discharged home compared with patients with known diabetes and those without hyperglycemia (2). In addition, hyperglycemia in admitted patients without a diagnosis carries a higher 3-year risk of later being diagnosed with diabetes (3).
Patients with diabetes account for nearly one in five hospitalizations in the U.S. (4). Hospital stays for patients with diabetes accounted for 7.7 million stays and cost $83 billion or roughly one-quarter of total hospital costs in 2008 (5). Unfortunately, the optimal treatment of hyperglycemia of hospitalization is unclear because the exact etiology is unknown (6). Although the general mechanism for hyperglycemia of hospitalization has always been assumed to be the physiologic stress of the acute illness leading to hospitalization, it is possible that sleep loss and/or reduced sleep quality during hospitalization plays a role. Studies show that medical patients not only achieve 2 h less sleep in the hospital than at home, but also suffer from increased sleep fragmentation, compared with sleep at home (7).
There are several physiologic reasons to believe that sleep loss in the hospital could be associated with hyperglycemia of hospitalization. First, laboratory studies demonstrate that a few days of sleep restriction or of experimentally induced sleep fragmentation can result in impaired glucose tolerance (8–12). Second, prospective epidemiological studies also demonstrate a relationship between shorter sleep duration or poorer sleep quality and the risk of incident diabetes in community-dwelling adults (13,14). It has also been shown that chronic insomnia with short sleep duration is strongly associated with a risk for diabetes (15). Recent reviews have proposed mechanisms by which sleep affects metabolism, as it may affect both the brain and peripheral organs playing a pivotal role in metabolism, and thus sleep may be a promising interventional point to treat obesity and type 2 diabetes (16,17). Given these associations, it is worth exploring whether acute sleep loss is associated with increased risk of hyperglycemia in hospitalized adults. The aim of this study was to assess whether sleep duration and sleep efficiency during hospitalization, measured with objective methods, were associated with greater odds of hyperglycemia in both patients with and without diabetes.
Research Design and Methods
We conducted a retrospective observational study using data collected from an existing prospective cohort study of sleep in hospitalized patients ≥50 years of age on a general medicine ward. To avoid including patients with pre-existing sleep disorders or abnormal sleep patterns, we excluded patients with known sleep disorders or individuals who were admitted from another hospital, a nursing facility, or a rehabilitation facility. To improve the accuracy of actigraphy readings, we only enrolled patients who were ambulatory and not on bed rest. Additionally, every patient had a single room at this institution. Any patient who was ≥50 years old, was on a general medicine ward, was ambulatory, and had a morning blood sample drawn to assess a basic metabolic panel (BMP) was eligible for this study. A patient was excluded if he/she was nonambulatory, was in respiratory isolation, had been in the hospital for >72 h, was readmitted to the hospital within the past 2 weeks, or was transferred from another institution. Trained research assistants screened patients using the electronic health record and a brief screening questionnaire. Patients provided written informed consent to participate. The University of Chicago Institutional Review Board approved this study.
Wrist actigraphy was used to measure hospital sleep duration and efficiency (percentage of time in bed spent asleep) (18). Patients were asked to wear the Actiwatch 2 (Phillips Respironics, Murraysville, PA) from study day enrollment to discharge. Time in bed was based on the patient's sleep diary indicating bedtime and wake time. Actual sleep obtained during time in bed was determined by the actigraphy software, and sleep efficiency was determined by dividing actual sleep by the time in bed. Noise was measured using bedside sound meters (Larson Davis, Depew, NY). Patients also completed an inpatient interview that was used to obtain demographic data (race) as well as screen for obstructive sleep apnea (Berlin Questionnaire) (19). Chart reviews were done by trained research assistants to collect patient sex, age, diagnosis, comorbidity burden, and BMI from the medical record (20).
Morning fasting glucose was obtained by extracting routine morning laboratories (drawn between 4 and 6 a.m.) from the Epic electronic health record (Epic Systems, Verona, WI). Blood glucose was obtained each morning as part of a sample collected intravenously to measure a BMP. We created categories of hyperglycemia based on the World Health Organization definition outlined below (21). Morning plasma glucose levels between 60 and 110 mg/dL were considered to be normal, 110 to 126 mg/dL were defined as impaired fasting glucose (IFG), and levels ≥126 mg/dL were defined as hyperglycemia. Because relying on administrative data alone can lead to an underestimation of the prevalence of diabetes in hospitalized patients, we used three different methods to determine whether a patient was carrying a diagnosis (22). A patient was deemed to have a diagnosis of diabetes if they reported having diabetes during an inpatient interview by a trained research assistant, if their chart review by trained research assistants revealed a diagnosis of diabetes, or if their hospitalization was ultimately billed with a code for diabetes (ICD-9 250.00–250.99) by our trained medical coders. Billing data were obtained from our hospital’s billing system. For the purposes of this analysis, we tested the association between morning glucose level and sleep during the preceding night.
Descriptive statistics were used to summarize sleep duration, sleep efficiency, and rates of hyperglycemia and IFG. We compared demographic and sleep characteristics for patients with diabetes and patients without diabetes using t and χ2 tests where appropriate. Ordinal multivariable logistic regression, clustered by subject and controlling for diabetes diagnosis, was used to test the association between sleep duration and sleep efficiency and proportional odds of hyperglycemia versus IFG or normal blood glucose. Models were repeated to control for potential confounders, namely age, sex, race, overweight BMI, and obstructive sleep apnea (OSA) risk as measured by the Berlin Questionnaire. All statistical analyses were performed using Stata 12.0 (StataCorp, College Station, TX), and statistical significance was defined as a P value of <0.05.
Results
From April 2010 to June 2014, 212 patients (60% female and 74% African American) from the University of Chicago inpatient general medicine service were enrolled in the study. The patient population had a vast heterogeneity of principal diagnoses (the reason for admission), which we acquired through ICD-9 codes. Of the 212 patients, an ICD code for principal diagnosis was available for 211 of them. Only 10 diagnoses were seen in more than 4 patients. Of these 10, the most common (diabetes) was seen as a reason for admission in only 13 patients. The top 10 diagnoses were diabetes (6.15%), kidney failure (5.69%), diseases of the pancreas (4.26%), cellulitis and abscesses (3.79%), gastrointestinal hemorrhage (3.31%), complications as a result of procedures (3.31%), asthma (2.84%), diverticula of the intestine (2.84%), disorders of fluid electrolytes and acid–base imbalance (2.37%), and complications of medical care (2.37%). These 10 diagnoses constitute only 37% of the total diagnoses of our patient population. One-third (73; 34%) of participants were determined to have diabetes by self-report, chart review, or diabetes billing code. Average length of stay was 5.7 days, with the majority of patients staying ≤5 days (65.09%). Patients with diabetes were more likely to be African American, be overweight, and have a high risk for OSA as assessed by the Berlin Questionnaire (Table 1).
Table 1.
Demographics and descriptive statistics
| All patients | Without diabetes | With diabetes | ||
|---|---|---|---|---|
| Demographics | n = 212 | n = 139 | n = 73 | |
| Age (years) | 63.9 ± 11.1 | 64.1 ± 11.4 | 63.5 ± 10.5 | |
| Female | 59.9% | 58.3% | 63.0% | |
| African American | 74.1% | 69.1% | 83.6%* | |
| Mean BMI | 27.6 ± 8.3 | 26.4 ± 7.7 | 29.9 ± 8.8* | |
| Overweight (BMI ≥25) | 61.3% | 53.2% | 76.7%* | |
| High OSA risk* | 43.4% | 36.7% | 56.2%* | |
| Descriptive statistics (368 mornings) | n = 368 | n = 246 | n = 122 | |
| Sleep duration (min) | 318.2 ± 138.6 | 319.5 ± 139.3 | 315.5 ± 137.6 | |
| Sleep efficiency (%) | 69.7 ± 22.2 | 69.7 ± 22.3 | 69.8 ± 49.9 | |
| Fasting blood glucose (mg/dL) | 112.5 ± 37.6 | 103.9 ± 25.4 | 129.9 ± 22.1* | |
| Mornings with elevated blood glucose (110–126 mg/dL) | 14.4% | 13.8% | 15.6% | |
| Mornings with hyperglycemia (≥126 mg/dL) | 24.7% | 14.6% | 45.1% | |
| Sleep debt (in hours) | 1.17 ± 2.66 (n = 366) | 1.19 ± 2.64 (n = 244) | 1.13 ± 2.72 | |
Data are means ± SD, unless otherwise indicated. Breakdown of patient demographics including all patients, which was then separated by diabetes diagnosis. Comparison of descriptive measures in hospital mornings.
*Statistically significant difference between patients with and without diabetes (P < 0.05). High OSA risk determined by calculation from Berlin OSA survey.
Based on actigraphy, mean objective sleep duration was slightly >5 h (318 ± 139 min), and mean sleep efficiency was below the normal threshold of 80% (69.7 ± 22.2%). Mean objective sleep duration and mean sleep efficiency did not differ significantly between patients with or without diabetes (Table 1). Although patients with diabetes were more likely to be overweight and have a higher risk of OSA by Berlin Score, objective inpatient sleep measures did not differ between groups (Table 1).
Glucose levels were obtained for 368 mornings for 212 patients (mean glucose 112.5 mg/dL [95% CI 100.7, 107.0]). For 14.4% of mornings, patients had IFG (110–126 mg/dL), and for 24.7% of mornings, patients had hyperglycemia (blood glucose ≥126 mg/dL). Not surprisingly, mean fasting glucose and rates of hyperglycemia were higher among patients with diabetes than patients without diabetes (Table 1).
In ordinal logistic regression models, for each additional hour of sleep in the hospital the night before, the proportional odds of having glucose levels characterized as elevated or hyperglycemic the next morning (hyperglycemia vs. elevated; elevated vs. normal) were 11% lower (odds ratio 0.89 [95% CI 0.80, 0.99]; P = 0.043). Likewise, every 10% increase in sleep efficiency was associated with 18% lower proportional odds of a higher glucose category (odds ratio 0.82 [95% CI 0.74, 0.89]; P < 0.001) (Fig. 1 and Table 2). These results remained significant when controlling for diabetes diagnosis, age, sex, overweight BMI (≥25), African-American race, and risk of OSA. Additionally, these results remained significant when controlling for objective average and maximum noise levels recorded by bedside sound meters (Leq) (Supplementary Tables 1 and 2) in addition to severity of illness as measured by Charlson comorbidity score. Of the 368 nights studied, 27 nights (7.34% of total nights) were the patient’s first night of stay in the hospital. Controlling for either first night of hospital stay or the number of days since admission did not affect the significance of our findings. After chart audits for nighttime insulin use, we found very few cases of patients receiving insulin during the sleep period (23 insulin administrations). Nocturnal insulin administration did not alter the significant relationship between sleep and blood glucose category.
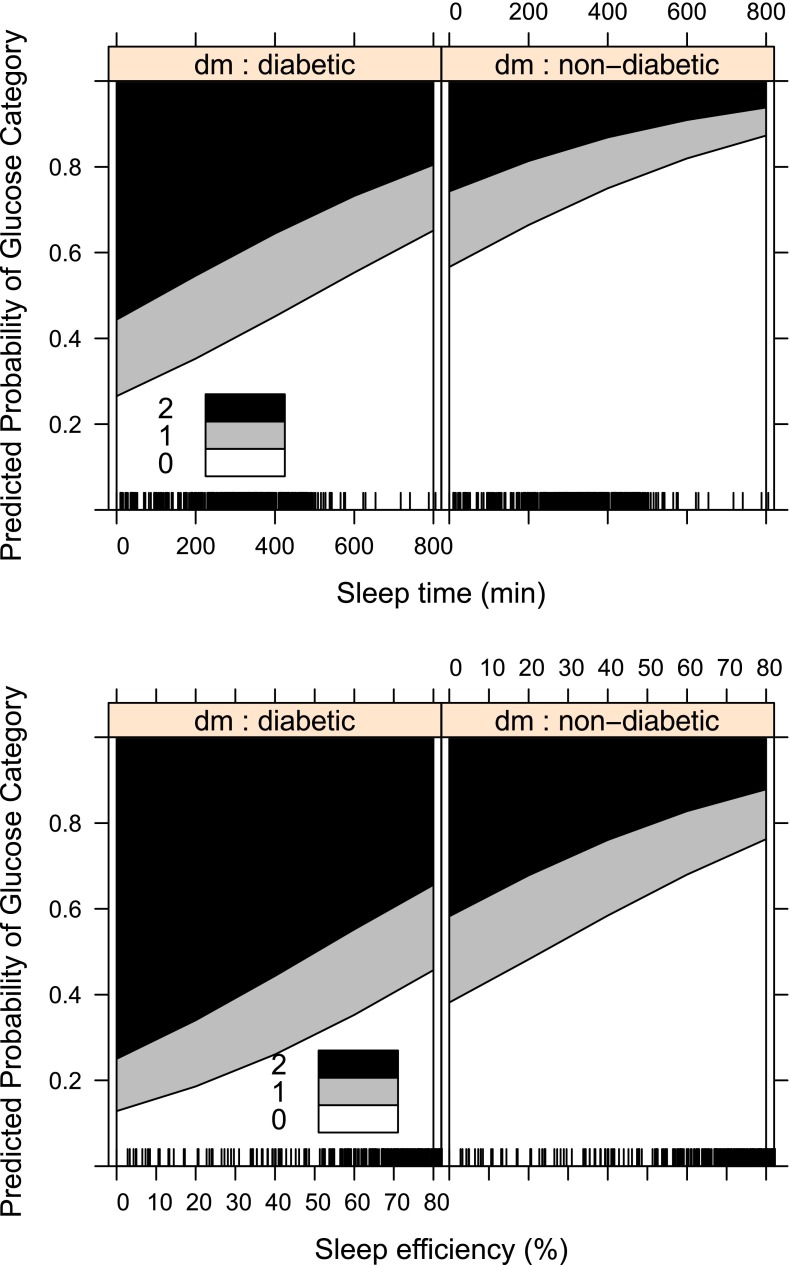
Figure 1.
Ordinal logistic regressions separated by diabetes diagnosis status comparing sleep time (top) and sleep efficiency (bottom). 0 refers to the probability of having glucose within normal limits (60–110 mg/dL), 1 refers to the probability of having elevated glucose (110–126 mg/dL), and 2 refers to the probability of having hyperglycemia (≥126 mg/dL) (using World Health Organization criteria).
Table 2.
Ordinal logistic regressions for comparison of sleep duration and sleep efficiency as measured by actigraphy watch to elevated morning fasting blood glucose (110–126 mg/dL) and hyperglycemia (≥126 mg/dL)
| Association between odds of blood glucose level (normal, elevated, and hyperglycemic) and sleep duration (in hour increments) (n = 368 mornings from 212 subjects) |
|||
|---|---|---|---|
| Covariate | Model 1 | Model 2 | Model 3 |
| Sleep duration (hours) | 0.89* (0.80, 0.99) | 0.89* (0.79, 0.99) | 0.89* (0.80, 0.9966) |
| Diabetes diagnosis | 4.28* (2.55, 7.17) | 4.25* (2.54, 7.12) | 3.56* (2.06, 6.17) |
| Female | — | 1.14 (0.65, 1.99) | 0.99 (0.55, 1.77) |
| Age | — | — | 1.04 (0.84, 1.30) |
| BMI ≥25 | — | — | 1.78* (1.03, 3.06) |
| African American | — | — | 1.42* (0.75, 2.69) |
| OSA risk | — | — | 1.74* (1.04, 2.94) |
| Cutpoint 1 | 0.34 | 0.53 | 1.24 |
| Cutpoint 2 | 1.10 | 1.29 | 2.02 |
| Association between odds of blood glucose levels (normal, elevated, and hyperglycemic) and sleep efficiency (in 10% increments) (n = 368 mornings from 212 subjects) |
|||
| Sleep efficiency (10% increments) | 0.82* (0.75, 0.90) | 0.81* (0.74, 0.89) | 0.81* (0.74, 0.89) |
| Diabetes diagnosis | 4.55* (2.73, 7.58) | 4.51* (2.71, 7.50) | 3.77* (2.19, 6.51) |
| Female | — | 1.26 (0.72, 2.18) | 1.09 (0.61, 1.94) |
| Age | — | — | 1.02 (0.82, 1.27) |
| BMI ≥25 | — | — | 1.77* (1.03, 3.05) |
| African American | — | — | 1.43* (0.75, 2.70) |
| OSA risk | — | — | 1.76* (1.06, 2.91) |
| Cutpoint 1 | −0.40 | −0.11 | 0.55 |
| Cutpoint 2 | 0.38 | 0.68 | 1.35 |
Cutpoints are the estimated point on the latent variable (sleep) used to differentiate normal blood glucose from elevated blood glucose and elevated blood glucose from hyperglycemia when the predictor variables are evaluated at zero.
*P < 0.05.
Figure 1 (created using R; R Foundation, Vienna, Austria) shows that the predicted probability of a higher glucose category increases as sleep time and sleep efficiency decrease. Although patients with diabetes are more likely to have higher odds of a higher glucose category compared with patients without diabetes at any sleep duration, it is important to note that the relationship between sleep duration and glucose category is similar for both patients with and without diabetes. A similar relationship was observed for sleep efficiency and glucose category for both groups of patients.
Conclusions
This study is the first to report that shorter sleep duration among hospitalized general medicine patients was associated with greater proportional odds of hyperglycemia and IFG in both patients with and without diabetes. Furthermore, a similar association was observed for sleep efficiency, with worsening sleep efficiency associated with higher proportional odds of hyperglycemia. This finding is consistent with both prior laboratory and epidemiological studies that show that acute sleep loss can result in impaired glucose tolerance or increased risk of diabetes. Because inpatient sleep duration, and potentially sleep quality, may be modifiable, future studies should examine whether improving sleep among inpatients can reduce the risk of hyperglycemia during hospitalization.
It is worth considering possible mechanisms for these findings. One possibility is that in-hospital sleep loss may contribute to hyperglycemia. Multiple mechanisms may be responsible for the adverse effects of sleep loss on glucose tolerance. These could include abnormalities in several hormones involved in glucose regulation, including cortisol, growth hormone, leptin, and ghrelin. In addition, increases in sympathetic nervous system activity and decreases in cerebral glucose metabolism resulting from the sleep debt may play a role. Another causal pathway could be related to the fact that disruptions or insufficient amounts of deep non–rapid eye movement sleep, also known as slow wave sleep, considered the most “restorative” sleep, can lead to disruptions in glucose homeostasis (23). Studies show that suppression of slow wave sleep is associated with increased risk of diabetes (24). It is also possible that sleep loss is associated with unrecognized confounders that are also associated with hyperglycemia, such as illness severity.
This study suggests that future research is needed to explore whether the sleep disturbances that are typically associated with hospitalization could be an underlying cause of hyperglycemia. Moreover, future research should also examine if interventions to optimize sleep duration and quality result in better glucose control. Health administrators and clinicians should consider investing in improving sleep for hospitalized patients by limiting unnecessary nocturnal care and reducing disruptions, such as noise (25). Toward that end, the American Academy of Nursing has recommended not waking patients unless critical to the patient’s care as part of its Choosing Wisely campaign (26).
This study has several limitations. Because this was an observational study, we cannot assume a direction of causality. It is possible that patients with hyperglycemia have worse sleep for metabolic reasons that are related to the hyperglycemia itself. For example, patients who have inflammation may also have both hyperglycemia and poor sleep because of an inflammatory response. If we assume that sleep the night before is causing the hyperglycemia, a patient with diabetes who is more likely to have morning hyperglycemia may have shorter sleep in the hospital because of frequent checks on their blood glucose. Although this could explain the case for patients with diabetes, it is unlikely that patients without diabetes would have blood glucose checks ordered at night, and so this cannot explain this relationship in patients without diabetes. Similar to frequent mechanical stimuli such as blood glucose checks, noise could be a substantial interruption for our patients, as this has been previously shown to affect insulin sensitivity (27,28). However, all of the patients enrolled in this study were in single-occupancy rooms, and after controlling for objective noise as measured by sound meters, we saw no change in our primary results (Supplementary Tables 1 and 2).
Moreover, the study was performed on a single ward at a single institution, which limits the ability to generalize the findings. Additionally, the reasons for admission were not included in our analysis because the general medicine ward considered in our study has a wide heterogeneity of admitting diagnoses, with the 10 most popular reasons for admission constituting only 37% of the total principal diagnoses. We did not control for diabetic pharmacologic regimen or glucose control prior to hospitalization. We were unable to determine the true incidence of hospital-associated hyperglycemia, because that would require follow-up to ascertain if hyperglycemia normalized. It is possible that a patient with no known diagnosis of diabetes in the hospital, upon further outpatient follow-up and treatment, may have been found to have a diagnosis of diabetes. We also did not perform oral glucose tolerance tests on patients to formally identify the risk of diabetes. Although we abstracted morning blood glucose levels between 4 and 6 a.m. from chart review with the assumption that these were fasting values, it is certainly possible, although extremely unlikely, that a patient ingested calories in the middle of the night. Patients had to have a BMP assessed in order to be eligible for analysis, and although a BMP may be more likely to be assessed for certain diagnoses, it is a routine laboratory test performed on a daily basis for most patients in their stay. Our study did not assess napping behavior, but only measured nocturnal sleep duration and efficiency. Lastly, unlike polysomnography, the use of actigraphy in this study means that we cannot examine sleep architecture, but only sleep duration and efficiency.
Shorter sleep duration during hospitalization was associated with increased odds of elevated blood glucose and hyperglycemia in both patients with and without diabetes. Although there have been many epidemiologic and laboratory studies linking hyperglycemia to sleep deprivation, to our knowledge, this is the first that shows this relationship in the real-world setting of the hospital ward. Along with the epidemiologic and laboratory studies showing a relationship between reduced sleep duration and/or quality and hyperglycemia, the detected association between insufficient and/or poor sleep in the hospital and hyperglycemia highlights the need for further studies to explore whether optimizing inpatient sleep could improve inpatient glycemic control. Future research should test whether improving patient sleep in hospitals could represent a novel nonpharmacological approach to reducing the risk of hyperglycemia during hospitalization and ultimately lead to better health and reduced health care costs.
Supplementary Material
Article Information
Acknowledgments. The authors thank Kristen Wroblewski of the Department of Public Health Sciences, University of Chicago, for valuable assistance.
Funding. This work was supported by National Institute on Aging grant K23AG033763 (Environment and Perceived Control: Improving Sleep in Hospitalized Older Patients) and National Heart, Lung, and Blood Institute grant R25HL116372 (Sleep for Inpatients: Empowering Staff to Act [SIESTA]).
Duality of Interest. No potential conflicts of interest relevant to this article were reported.
Author Contributions. R.H.D. conceived of and designed the study, acquired and analyzed data, and drafted and reviewed the manuscript. K.L.K. conceived of and designed the study, analyzed data, reviewed the manuscript, and supervised the study. L.S. and S.L.A. acquired and analyzed data, reviewed the manuscript, and provided administrative support. D.O.M. was responsible for the conception and design of the study, acquired data, reviewed the manuscript, and supervised the study. E.V.C. conceived of and designed the study, analyzed data, and reviewed the manuscript. V.M.A. conceived of and designed the study, acquired and analyzed data, drafted and reviewed the manuscript, obtained funding, provided administrative support, and supervised the study. V.M.A. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Footnotes
Clinical trial reg. no. NCT01057823, clinicaltrials.gov.
This article contains Supplementary Data online at http://care.diabetesjournals.org/lookup/suppl/doi:10.2337/dc16-1683/-/DC1.
References
- 1.Levetan C. Controlling hyperglycemia in the hospital: A matter of life and death. Clin Diabetes 2000;18:17–25 [Google Scholar]
- 2.Umpierrez GE, Isaacs SD, Bazargan N, You X, Thaler LM, Kitabchi AE. Hyperglycemia: an independent marker of in-hospital mortality in patients with undiagnosed diabetes. J Clin Endocrinol Metab 2002;87:978–982. DOI: 10.1210/jcem.87.3.8341 [DOI] [PubMed] [Google Scholar]
- 3.McAllister DA, Hughes KA, Lone N, et al. Stress hyperglycaemia in hospitalised patients and their 3-year risk of diabetes: a Scottish retrospective cohort study. PLoS Med 2014;11:e1001708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Butala NM, Johnson BK, Dziura JD, et al. Decade-long trends in mortality among patients with and without diabetes mellitus at a major academic medical center. JAMA Intern Med 2014;174:1187–1188 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fraze T, Jiang HJ, Burgess J; Healthcare Cost and Utilization Project. Statistical brief #93. Hospital stays for patients with diabetes, 2008 [article online]. Available from http://www.hcup-us.ahrq.gov/reports/statbriefs/sb93.pdf. Accessed 16 June 2016
- 6.Clement S, Braithwaite SS, Magee MF, et al.; American Diabetes Association Diabetes in Hospitals Writing Committee . Management of diabetes and hyperglycemia in hospitals. Diabetes Care 2004;27:553–591 [DOI] [PubMed] [Google Scholar]
- 7.Arora VM, Chang KL, Fazal AZ, et al. Objective sleep duration and quality in hospitalized older adults: associations with blood pressure and mood. J Am Geriatr Soc 2011;59:2185–2186 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Van Cauter E, Blackman JD, Roland D, Spire JP, Refetoff S, Polonsky KS. Modulation of glucose regulation and insulin secretion by circadian rhythmicity and sleep. J Clin Invest 1991;88:934–942 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Reutrakul S, Van Cauter E. Interactions between sleep, circadian function, and glucose metabolism: implications for risk and severity of diabetes. Ann N Y Acad Sci 2014;1311:151–173 [DOI] [PubMed] [Google Scholar]
- 10.Koren D, Levitt Katz LE, Brar PC, Gallagher PR, Berkowitz RI, Brooks LJ. Sleep architecture and glucose and insulin homeostasis in obese adolescents. Diabetes Care 2011;34:2442–2447 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Cedernaes J, Lampola L, Axelsson EK, et al. A single night of partial sleep loss impairs fasting insulin sensitivity but does not affect cephalic phase insulin release in young men. J Sleep Res 2016;25:5–10 [DOI] [PubMed] [Google Scholar]
- 12.Schmid SM, Hallschmid M, Jauch-Chara K, et al. Disturbed glucoregulatory response to food intake after moderate sleep restriction. Sleep 2011;34:371–377 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Knutson KL, Ryden AM, Mander BA, Van Cauter E. Role of sleep duration and quality in the risk and severity of type 2 diabetes mellitus. Arch Intern Med 2006;166:1768–1774 [DOI] [PubMed] [Google Scholar]
- 14.Anothaisintawee T, Reutrakul S, Van Cauter E, Thakkinstian A. Sleep disturbances compared to traditional risk factors for diabetes development: Systematic review and meta-analysis. Sleep Med Rev 2015;30:11–24 [DOI] [PubMed] [Google Scholar]
- 15.Vgontzas AN, Liao D, Pejovic S, Calhoun S, Karataraki M, Bixler EO. Insomnia with objective short sleep duration is associated with type 2 diabetes: A population-based study. Diabetes Care 2009;32:1980–1985 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cedernaes J, Schiöth HB, Benedict C. Determinants of shortened, disrupted, and mistimed sleep and associated metabolic health consequences in healthy humans. Diabetes 2015;64:1073–1080 [DOI] [PubMed] [Google Scholar]
- 17.Schmid SM, Hallschmid M, Schultes B. The metabolic burden of sleep loss. Lancet Diabetes Endocrinol 2015;3:52–62 [DOI] [PubMed] [Google Scholar]
- 18.Ancoli-Israel S, Cole R, Alessi C, Chambers M, Moorcroft W, Pollak CP. The role of actigraphy in the study of sleep and circadian rhythms. Sleep 2003;26:342–392 [DOI] [PubMed] [Google Scholar]
- 19.Netzer NC, Stoohs RA, Netzer CM, Clark K, Strohl KP. Using the Berlin Questionnaire to identify patients at risk for the sleep apnea syndrome. Ann Intern Med 1999;131:485–491 [DOI] [PubMed] [Google Scholar]
- 20.Meltzer D, Manning WG, Morrison J, et al. Effects of physician experience on costs and outcomes on an academic general medicine service: results of a trial of hospitalists. Ann Intern Med 2002;137:866–874 [DOI] [PubMed] [Google Scholar]
- 21.World Health Organization. Definition, Diagnosis, and Classification of Diabetes Mellitus and its Complications: Report of a WHO Consultation. Part 1: Diagnosis and Classification of Diabetes Mellitus. Geneva, World Health Org., 1999 (WHO/NCD/NCS/99.2)
- 22.Levetan CS, Passaro M, Jablonski K, Kass M, Ratner RE. Unrecognized diabetes among hospitalized patients. Diabetes Care 1998;21:246–249 [DOI] [PubMed] [Google Scholar]
- 23.Van Cauter E, Spiegel K, Tasali E, Leproult R. Metabolic consequences of sleep and sleep loss. Sleep Med 2008;9(Suppl. 1):S23–S28. DOI: 10.1016/S1389-9457(08)70013-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tasali E, Leproult R, Ehrmann DA, Van Cauter E. Slow-wave sleep and the risk of type 2 diabetes in humans. Proc Natl Acad Sci U S A 2008;105:1044–1049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yoder JC, Staisiunas PG, Meltzer DO, Knutson KL, Arora VM. Noise and sleep among adult medical inpatients: far from a quiet night. Arch Intern Med 2012;172:68–70 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.American Academy of Nursing. Don’t wake the patient for routine care unless the patient’s condition or care specifically requires it [article online]. Available from http://www.choosingwisely.org/clinician-lists/american-academy-nursing-avoid-waking-patients-for-routine-care/. Accessed 5 August 2015
- 27.Herzog N, Jauch-Chara K, Hyzy F, et al. Selective slow wave sleep but not rapid eye movement sleep suppression impairs morning glucose tolerance in healthy men. Psychoneuroendocrinology 2013;38:2075–2082 [DOI] [PubMed] [Google Scholar]
- 28.Stamatakis KA, Punjabi NM. Effects of sleep fragmentation on glucose metabolism in normal subjects. Chest 2010;137:95–101. DOI: 10.1378/chest.09-0791 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.