Abstract
The in vitro activity of the prodrug amodiaquine and its metabolite monodesethyl-amodiaquine has been studied for three strains of Plasmodium falciparum: LS-2, LS-3, and LS-1. Both compounds showed significant activity against all three strains; the activity of amodiaquine was slightly higher than that of the metabolite. By use of a checkerboard design, interaction studies with both compounds yielded evidence of significant synergism; means of the sums of the fractional inhibitory concentrations were 0.0392 to 0.0746 for strain LS-2, 0.1567 to 0.3102 for strain LS-3, and 0.025 to 0.3369 for strain LS-1. In further investigations, the interaction of amodiaquine with monodesethyl-amodiaquine was tested at clinically relevant concentrations of both compounds. In these studies, involving amodiaquine at picomolar and femtomolar concentrations, the compound was found to exert high potentiating activity on monodesethyl-amodiaquine. This interaction produced mean ratios of observed to expected activity of 0.0505 to 0.0642 for strain LS-2, 0.0882 to 0.3820 for strain LS-3, and 0.0752 to 0.2924 for strain LS-1. The synergistic activity was most marked at monodesethyl-amodiaquine/amodiaquine ratios up to 100,000:1 but was still evident at higher ratios.
The widespread increase of resistance to chloroquine in Plasmodium falciparum has resulted in renewed interest in amodiaquine (AQ) as a replacement for chloroquine in the treatment of malaria, especially in sub-Saharan Africa. Although AQ belongs to the same chemical class of compounds as chloroquine, the 4-aminoquinolines, this drug often shows adequate clinical-parasitological efficacy in chloroquine-resistant infections. Furthermore, adverse reactions reported previously, such as agranulocytosis (14) and hepatitis (20), were mainly associated with the prophylactic use of AQ, and more recent reports show that therapeutic regimens are well tolerated (22). Therefore, the World Health Organization has again listed AQ as a drug for treatment of chloroquine-resistant falciparum malaria (40).
During and after absorption, orally administered AQ is rapidly metabolized to desethylamodiaquine (DAQ) and other derivatives of lesser antimalarial significance (9). Clinically, the antimalarial activity of AQ is exerted mainly through DAQ (37). Earlier in vitro studies indicated that DAQ and AQ have equal activity against P. falciparum (8). Other observations suggest that the two compounds possess independent modes of action (7, 27). However, recent comparative in vitro studies with fresh isolates of P. falciparum showed a significant correlation between the activities of the two compounds that indicates similarities in the way they act against the parasite (12).
Chloroquine-resistant falciparum malaria on the African continent was first detected in Kenya and Tanzania. During the past 2 decades it has extended to practically all parts of tropical Africa (32, 39). AQ has been shown to maintain high efficacy in the areas of malaria endemicity in the continent where chloroquine resistance is still mostly at the R1 level of in vivo resistance (6, 23). Nevertheless, low levels of AQ resistance have been observed, usually linked to high grades of chloroquine resistance, possibly due to a codon 86Tyr mutation in the P. falciparum multidrug resistance 1 gene in association with mutations in the P. falciparum chloroquine resistance transporter gene (33). In some settings, low-grade resistance to AQ was also associated with reduced susceptibility to quinine (2, 4, 5). Several in vitro studies have shown a positive activity correlation between AQ and chloroquine. Cross-resistance between DAQ and chloroquine has also been observed in vitro in P. falciparum isolates from various regions of Africa (1a, 26), Southeast Asia (7, 31), and South America (28). However, in the presence of low-grade resistance to chloroquine, the activity correlation with AQ may be considered a phenomenon that might anticipate overt cross-resistance in the future but is not necessarily a sign of manifest clinical-parasitological AQ resistance (11).
As mentioned above, DAQ, the main metabolite of AQ, plays the major role in the antimalarial activity of the drug. This is due to the higher concentration-time profile and the longer half-life of DAQ compared to those of the parent compound (16). Nevertheless, the activities of AQ and DAQ might complement one another after oral administration of AQ, following which both compounds will be present. As a result of the ban of AQ in the 1980s and 1990s, discouraging further work on the compound, there are no substantial data on the effective concentrations of AQ or DAQ in the body or on whether AQ plays an auxiliary role in the antimalarial activity of DAQ. It has been suggested that the activities of AQ and DAQ might complement each other and that trace concentrations of AQ will enhance the effect of DAQ during the first 3 days of treatment and beyond (16, 19, 38). The extent to which low AQ concentrations exert pharmacological effects on the AQ metabolites is not known. Pharmacokinetic data for AQ indicate that the peak concentration (∼25 nM) is reached within 30 min after oral administration. AQ concentrations drop below the threshold of detection (∼10 nM) at 8 h after drug administration. The apparent half-life has been estimated at 5.2 h (36). With DAQ the peak concentration (∼500 nM) is reached 2.3 h after oral administration of AQ. The terminal half-life in plasma ranges from 9 to 18 days (25). While in the first hour after oral administration of AQ, concentrations of AQ are usually higher than those of DAQ, the ratio reverses from then on, in favor of DAQ. It is likely that the ratio of DAQ to AQ reaches equilibrium and stabilizes after DAQ has passed the tα1 and tα2 phases, a process that is usually completed within 24 and 48 h, respectively (25). From the pharmacokinetic concentration-time profiles of DAQ and AQ, it can be expected that the DAQ/AQ ratio at equilibrium will range from 500:1 to 5,000:1.
The reintroduction of AQ as a first line antimalarial agent in Africa needs further support by extensive in vivo studies as well as observations in suitable in vitro systems. The latter should be developed with the inclusion of both drugs (AQ and DAQ) in order to appropriately reflect the in vivo situation. Such a system has been established in this study, the primary objective of which was to assess the in vitro efficacy of the AQ and DAQ mono- compounds and their interaction in culture-adapted isolates of P. falciparum.
MATERIALS AND METHODS
Drugs.
The two compounds, AQ (base) and DAQ hydrochloride, were obtained from Y. Bergquist, Dalarna University College, Borlange, Sweden. AQ was dissolved in 95% (vol/vol) ethanol, and DAQ was dissolved in distilled water to prepare stock solutions of 10−3 M. Both compounds were serially diluted in water (to 10−4 to 10−12 M concentrations), and the dilutions were kept in plastic containers in order to avoid loss through adsorption onto glass. For precoating, the dilutions were dispensed into 96-well microtiter plates, and ethanol and water were added to the control wells. Following the dosing, the plates were kept in the incubator at 37°C for 1 day in order to evaporate the solvents. After drying, the plates were kept sterile, wrapped in aluminum foil, and stored at +4°C.
Culture medium and parasite cultures.
Three laboratory-adapted P. falciparum strains, two of African origin (LS-2 from Tanzania and LS-3 from The Gambia) and one from Southeast Asia (LS-1 from Thailand), were used for all in vitro experiments. The parasites were continuously cultured by using the modified candle-jar method of Trager and Jensen (29). RPMI 1640 malaria culture medium (Life Technologies AB, Stockholm, Sweden) was supplemented with 25 mM HEPES buffer, 2 mg of sodium bicarbonate/ml, 0.5 μg of gentamicin/ml, and 10% human AB+ serum. Uninfected human O+ erythrocytes were used after two washes with Tris Hanks' buffer (SBL Vaccin AB, Solna, Sweden).
Baseline drug sensitivity testing.
For the preparation of starting culture suspensions, stock culture material was diluted with the medium to produce an initial parasitemia of 0.2 to 0.5% at a hematocrit of 5%. The medium-cell suspension was dispensed into the wells of the predosed AQ and DAQ plates. Following incubation of the plates at 37°C for 48 h in a candle jar, thin blood films were prepared from each well. The slides were stained with Giemsa stain and read under a light microscope for the presence of parasites and the determination of their density. Each single concentration was tested in duplicate wells, and the results were read twice. In parallel to the drug series, standard controls (undosed wells) were run with each strain. Baseline experiments with single drugs showed that the three strains had different sensitivity patterns, as initially proposed according to the 50% effective concentration (EC50, defined as the drug concentration that produces 50% inhibition of parasite growth relative to that in the control). Only one strain (LS-1) was reported to be partially resistant; the other two (LS-2 and LS-3) were considered susceptible to AQ and DAQ. For our studies, serial drug dilutions were prepared on the basis of the estimated EC50s.
Checkerboard design for combination studies.
A checkerboard design, including single columns for the different test drugs alone and duplicate columns for drug-free controls, was used for the interaction experiments. Two experimental designs were used, corresponding to two different methods of demonstrating the interactive capacity between the prodrug (AQ) and its metabolite (DAQ). The widely used standard method is based on evaluation of the interaction as reflected by EC50, EC90, and EC99; the interaction is visualized by isobolograms reflecting the fractional inhibitory concentrations of the partner drugs (3) (Fig. 1). The other method takes the major concentration difference between AQ and DAQ into consideration and compares the observed inhibition with that expected from the mono- compounds if one assumes fully additive activity (13). The observed/expected (O/E) ratios at the various DAQ/AQ proportions also lend themselves to graphic presentation (Fig. 2).
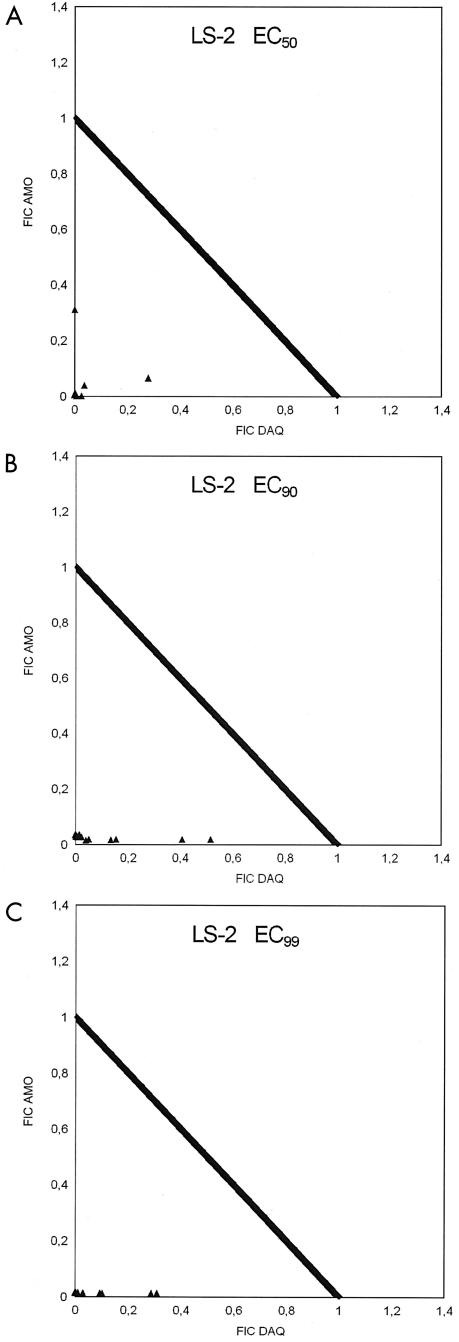
FIG. 1.
Fractional inhibitory concentrations (FIC) of AQ and DAQ in P. falciparum strain LS-2 at EC50, EC90, and EC99.
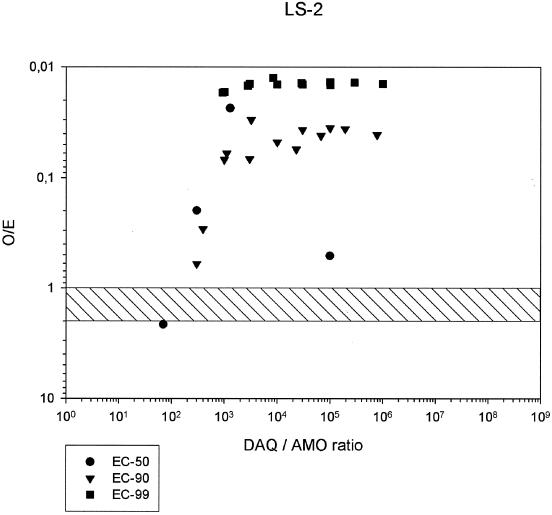
FIG. 2.
O/E ratios in combinations of AQ and DAQ at EC50, EC90, and EC99, and DAQ/AQ ratios, for P. falciparum strain LS-2.
AQ and DAQ were combined in various concentration ratios. The concentration range of AQ was 0.001 to 30 nM for the isobologram experiments, and that of DAQ was 0.1 to 30 nM. For assessment of the O/E ratios, the AQ concentration range was 0.000001 to 0.03 nM, and that of DAQ was 0.03 to 100 nM.
The drug solutions were introduced into 96-well flat-bottom microtiter plates and were also dried at 37°C prior to the in vitro culture experiments. The inoculated plates were incubated at 37°C for 48 h (29). Both parasite growth and the degree of inhibition were assessed by determination of parasite counts on Giemsa-stained thin blood films by light microscopy. All three strains were tested as described above.
Molecular genetic analysis.
For precise determination of the pfcrt and pfmdr1 genotypes of strains LS-1, LS-2, and LS-3, DNA was extracted from parasite smears of Giemsa-stained slides by using a Chelex extraction-based protocol (30). A standard PCR method was performed on the DNA extracts to analyze the pfcrt K76T and mdr1 N86Y single-nucleotide polymorphisms. pfcrt genotyping results were also confirmed by sequencing the DNA of the PCR products.
Data analyses for the mono- compounds and their interactions.
EC50, EC90, and EC99 are the parameters used for measuring the activities of the single drugs and combinations in vitro. This permits the determination of the fractional inhibitory concentrations and their sum (ΣFIC) (3), as well as the calculation of the O/E ratios.
The response of P. falciparum to AQ and DAQ is log-concentration normal. Therefore, the statistical analysis is based on log-normal regressions and on the log-concentration/response probit method of Litchfield and Wilcoxon (17). To carry out the analysis, the concentrations were transformed into logarithms and the percent inhibition values were transformed into probits. The transformed data were processed in least-square regressions (9a). In using the method of Litchfield and Wilcoxon (17), the various inhibitory concentrations were calculated according to the formula probit y = a + b logx, followed by taking the antilogx. Growth inhibition at a given drug concentration was calculated according to the same formula, by entering a given logx and converting the resulting y from probit to percent inhibition, by use of a probit table (1).
Interpretation in relation to clinically relevant concentration ratios.
The pharmacokinetic parameters Cmax (maximum concentration attained after drug administration), Tmax (time after drug administration at which the maximum concentration has been attained), and AUC (area under the curve) were used to relate the in vitro data to the in vivo situation.
The clinically relevant drug ratios were derived from the pharmacokinetic concentration-time profiles, assuming oral administration of AQ at normal therapeutic doses (16, 36). The time profile of the AQ concentrations has been calculated on the basis of a continuation of the logarithmic elimination curve after the AQ concentrations have dropped below the limit of detection by high-performance liquid chromatography.
RESULTS
Baseline sensitivity of mono- compounds.
The major response parameters—EC50, EC90, and EC99—of P. falciparum strains LS-2, LS-3, and LS-1 to AQ and DAQ are listed in Table 1. Strain LS-2 is the most sensitive of the three strains, and the activities of AQ and DAQ are quite similar. Strain LS-1 is considerably less sensitive than LS-2. Strain LS-1 is also more sensitive to AQ than to DAQ, especially at EC50 and EC90. Strain LS-3 is the least sensitive, both to AQ and to DAQ, among the three strains. The response pattern showed the most obvious difference between the activities of AQ and DAQ at the EC90.
TABLE 1.
EC50s, EC90s, and EC99s of AQ and DAQ for P. falciparum strains LS-2, LS-3, and LS-1
| Compound and strain | EC (nM)
|
Cmax (nM)a | ||
|---|---|---|---|---|
| 50% | 90% | 99% | ||
| AQ | ||||
| LS-2 | 0.52 | 3.47 | 86.48 | 31.4 |
| LS-3 | 0.23 | 7.01 | 343.85 | |
| LS-1 | 0.36 | 13.43 | 260.14 | |
| DAQ | ||||
| LS-2 | 0.56 | 9.62 | 97.93 | 2,507 |
| LS-3 | 0.77 | 29.58 | 546.98 | |
| LS-1 | 0.77 | 111.22 | 340.03 | |
Derived from the work of Minzi et al. (19).
Interaction of AQ and DAQ.
The interactions between AQ and DAQ have been determined by means of a checkerboard design. Evaluation end points were the ΣFIC for AQ and DAQ and the ratios between observed and expected concentrations (O/E ratios) of AQ plus DAQ at EC50, EC90, and EC99. In both systems, O/E ratios of ≥4 indicate marked antagonism, ratios of <4 and ≥2 indicate slight antagonism, ratios of ≤2 and ≥1 indicate additive activity, and ratios of <1 indicate synergism (3, 13). O/E ratios of <0.5 are usually considered to reflect strong synergism.
In general, the checkerboard regressions showed satisfactory correlation coefficients, with a geometric mean r of 0.9919 (range, 0.9536 to 1.0000) for strain LS-2, a geometric mean r of 0.9850 (range, 0.9500 to 1.0000) for strain LS-3, and a geometric mean r of 0.9850 (range, 0.9072 to 1.0000) for strain LS-1.
Strain LS-2.
All ΣFIC at the EC50, EC90, and EC99 were <1, indicating synergism between AQ and DAQ (Table 2). The degree of synergism was highest at the EC50 and only slightly lower at the EC99. Among the three strains, LS-2 showed the highest degree of synergism between AQ and DAQ (see the isobolograms in Fig. 1). The O/E ratios (Table 3) confirm this picture, indicating relatively high variability at the EC50 but a high degree of synergism, with O/E ratios consistently below 0.02 over a wide range of DAQ/AQ ratios, at the EC99 (Fig. 2).
TABLE 2.
ΣFIC for AQ and DAQ at EC50, EC90, and EC99 for P. falciparum strains LS-2, LS-3, and LS-1
| Strain and EC level | ΣFIC
|
||||
|---|---|---|---|---|---|
| No. of checkerboard lines with values of:
|
Range | Geometric mean | |||
| ≥2 | <2 and ≥1 | <1 | |||
| LS-2 | |||||
| EC50 | —a | — | 7 | 0.0042-0.3096 | 0.0392 |
| EC90 | — | — | 13 | 0.0303-0.5349 | 0.0746 |
| EC99 | — | — | 12 | 0.0143-0.3227 | 0.0451 |
| LS-3 | |||||
| EC50 | — | — | 8 | 0.0300-0.5166 | 0.1567 |
| EC90 | — | — | 10 | 0.0046-0.5861 | 0.3160 |
| EC99 | — | — | 10 | 0.0152-0.5495 | 0.3102 |
| LS-1 | |||||
| EC50 | — | — | 13 | 0.0094-0.4883 | 0.0825 |
| EC90 | — | — | 21 | 0.0617-0.5415 | 0.1580 |
| EC99 | — | — | 21 | 0.1101-0.7889 | 0.3369 |
—, none.
TABLE 3.
O/E ratios in combinations of AQ and DAQ at EC50, EC90, and EC99 levels in P. falciparum strains LS-2, LS-3, and LS-1
| Strain and EC level | No. of lines with O/E ratios of:
|
O/E ratios
|
Range of DAQ/AQ ratios | |||
|---|---|---|---|---|---|---|
| ≥2 | <2 and ≥1 | <1 | Range | Geometric mean | ||
| LS-2 | ||||||
| EC50 | 1 | —a | 6 | 0.0051-2.1489 | 0.0505 | 10-29,978 |
| EC90 | — | — | 13 | 0.0300-0.6047 | 0.0642 | 300-773,106 |
| EC99 | — | — | 12 | 0.0125-0.0171 | 0.0145 | 927-1,002,502 |
| LS-3 | ||||||
| EC50 | — | — | 8 | 0.0031-0.5316 | 0.0882 | 56-11,588 |
| EC90 | — | 1 | 9 | 0.0034-1.3768 | 0.3820 | 338-831,241 |
| EC99 | — | — | 10 | 0.0147-0.4572 | 0.3018 | 332-956,024 |
| LS-1 | ||||||
| EC50 | — | — | 13 | 0.0065-0.3906 | 0.0752 | 40-999,999 |
| EC90 | — | — | 21 | 0.0347-0.5416 | 0.1055 | 15-1.82 × 108 |
| EC99 | — | 1 | 20 | 0.0453-1.0708 | 0.2924 | 32-9.22 × 108 |
—, none.
Strain LS-3.
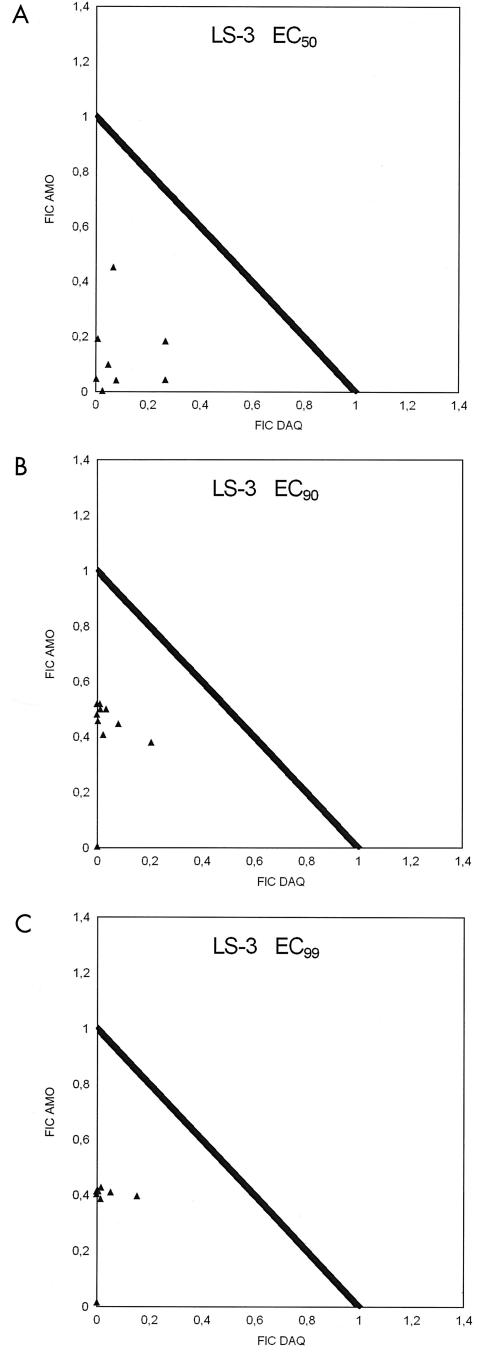
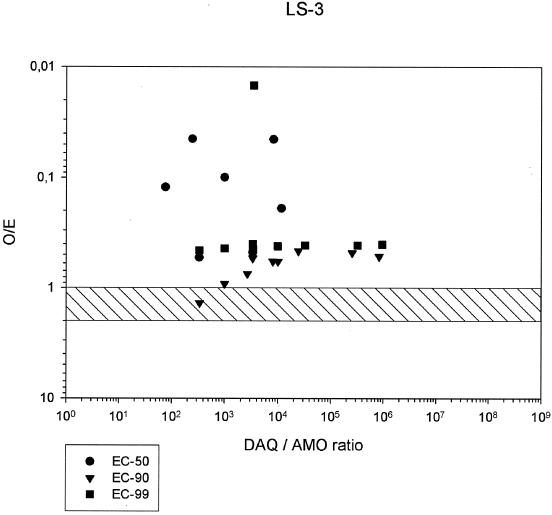
All ΣFIC at the EC50, EC90, and EC99 were below 1, showing the lowest mean but also the highest variation at the EC99 (Table 2 and isobolograms in Fig. 3). Nevertheless, substantial synergism was evident at all EC levels. The O/E ratios indicate the highest degree of synergism at the EC50 and the highest variability at the EC90. At this level, one of the readings was in the additive range. There was higher synergism and lower variability again at the EC99 (Table 3 and Fig. 4). At the EC99, the degree of synergism seemed to show little dependence on the DAQ/AQ ratios. Despite the evidence of marked synergism at all EC levels, the degree of synergism is apparently lower than that for strains LS-2 and LS-1, possibly due to the lower sensitivity of strain LS-3 to AQ and DAQ.
FIG. 3.
Fractional inhibitory concentrations (FIC) of AQ and DAQ for P. falciparum strain LS-3 at EC50, EC90, and EC99.
FIG. 4.
O/E ratios in combinations of AQ and DAQ at EC50, EC90, and EC99, and DAQ/AQ ratios, for P. falciparum strain LS-3.
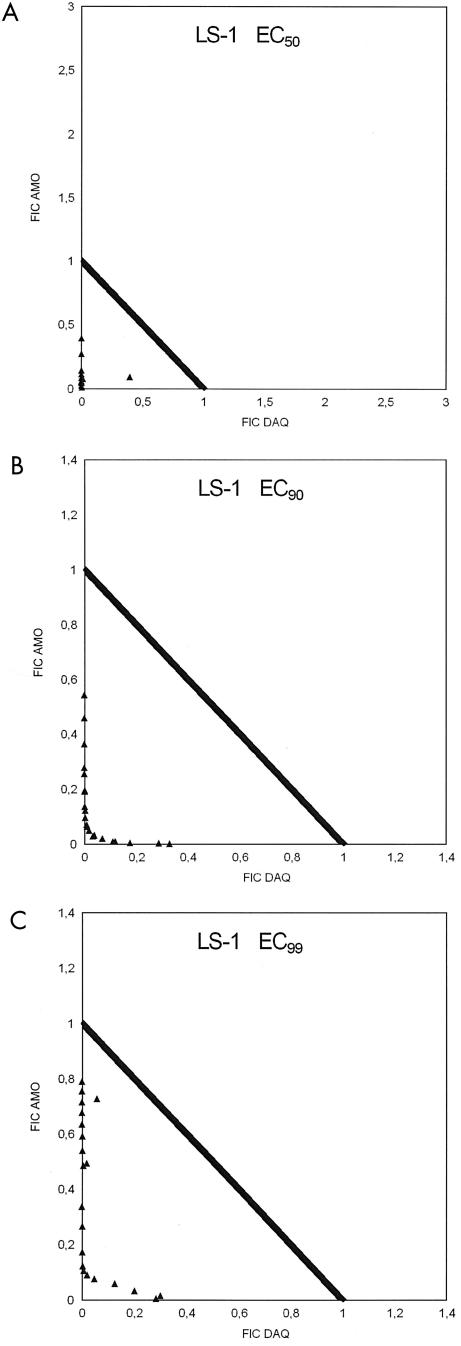
Strain LS-1.
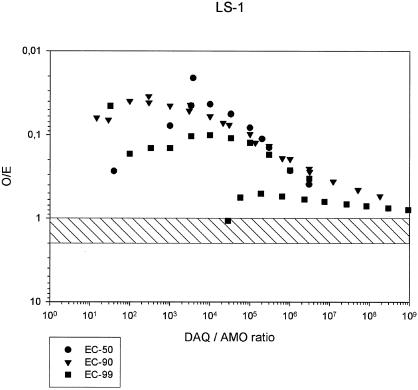
LS-1 had the original reputation of being the least sensitive of the three strains investigated in this study. All ΣFIC at the EC50, EC90, and EC99 were in the synergistic range (<1). The geometric mean ΣFIC showed an ascending pattern from the EC50 to the EC99 (Table 2 and isobolograms in Fig. 5), but nevertheless substantial synergism at the EC99. The O/E ratios followed practically the same synergistic pattern as the ΣFIC (Table 3 and Fig. 6), but 1 of the 21 lines showed an EC99 in the high additive range. The O/E ratios at the EC99 suggest some relation to the DAQ/AQ ratios inasmuch as O/E ratios of <0.2 are generally confined to DAQ/AQ ratios of <100,000.
FIG. 5.
Fractional inhibitory concentrations (FIC) of AQ and DAQ for P. falciparum strain LS-1 at EC50, EC90, and EC99.
FIG. 6.
O/E ratios in combinations of AQ and DAQ at EC50, EC90, and EC99, and DAQ/AQ ratios, for P. falciparum strain LS-1.
Molecular genotyping of pfcrt K76T and pfmdr1 N86Y.
Results from the pfcrt and pfmdr1 genotyping of strains LS-1, LS-2, and LS-3 are presented in Table 4.
TABLE 4.
Molecular genotyping results for LS-1, LS-2, and LS-3
| Strain | Amino acid residue at the following position:
|
|||
|---|---|---|---|---|
|
pfcrt
|
pfmdr1
|
|||
| K76T | A220S | N86Y | D1246Y | |
| LS-1 | K | A | N | D |
| LS-2 | T | S | Y | D |
| LS-3 | T | S | Y | D |
DISCUSSION
The studies confirmed the intrinsic antimalarial activities of AQ and its main metabolite, DAQ, toward P. falciparum strains LS-2, LS-3, and LS-1. All three strains investigated were sensitive to DAQ and AQ; among these, surprisingly and contrary to assumption, strain LS-3 turned out to be the least sensitive. AQ was slightly more active than DAQ against all the strains. Because AQ and DAQ behave similarly to chloroquine in vitro (18), exhibiting markedly selective uptake of the drug by erythrocytes and to an even greater degree by infected erythrocytes, the ECs of the monocompounds need to be considered in the light of this phenomenon. At a hematocrit of 5%, as used in this study, the threshold concentration of chloroquine, indicating full activity (MIC or EC99) in nonimmune individuals, is 100 nM in a blood-cell mixture. That of AQ would be 40 nM. Threshold concentrations for DAQ are likely to be similar to those of AQ, but there is a paucity of relevant data. The EC99 data for the three strains (AQ EC99, 86.48 to 343.85 nM; DAQ EC99, 97.93 to 546.98 nM) are therefore already clearly in the range of resistance. In the case of strain LS-1, originating from Thailand, this did not come as a surprise. However, for strains LS-2 and LS-3, it was unexpected. Here, long maintenance in in vitro culture may have produced the selection of 4-aminoquinoline-resistant parasites, since these are known to have a biological advantage over sensitive parasites.
After pharmacokinetic investigations in the late 1980s had produced evidence of the fast metabolic transformation of AQ to DAQ, attention became focused on DAQ, the more so because from 12 h after oral AQ administration on, it was the only active compound detectable. Despite the suggestion that the presence of AQ in low concentrations will enhance the effect of DAQ (16), these impressions were not followed up, probably due to the ban of the use of AQ that had discouraged work on this drug. The ban was only recently lifted.
This study also aimed to investigate the interaction of AQ and DAQ under conditions resembling the in vivo situation in the treated patient. Although AQ concentrations drop within 8 h from the oral uptake of AQ to a level below the limit of detection (∼10 nM), AQ concentrations in the picomolar and femtomolar range can be expected to last for several days. Our studies therefore concentrated on the interaction between DAQ at therapeutically observed concentrations and putative AQ concentrations as they are likely to occur between 12 and 168 h after the first intake of AQ.
The investigation produced evidence of marked interaction between AQ and DAQ in the sense that relatively low concentrations of AQ were able to potentiate the effect of DAQ. This was seen for all three P. falciparum strains, albeit to different degrees. The phenomenon was most marked in the least resistant strain (LS-2). However it was also substantial in the two other strains, spanning a wide range of DAQ/AQ ratios.
It follows from these observations that the clinical-parasitological activities of AQ and DAQ should not be considered in isolation, since both compounds are present after the oral administration of AQ. In view of the high degree of interaction and its strain specificity, it is highly questionable to advocate the use of DAQ instead of AQ for in vitro sensitivity testing of fresh isolates. The use of DAQ plus AQ at an appropriate ratio would reflect the real drug sensitivity much better. Considering the prevailing DAQ/AQ ratios during the 3 days of a treatment course of AQ, a ratio of 3,000:1 would be quite appropriate and would largely reflect conditions at ratios up to 100,000:1.
In relating the observations to the real-life situation in areas of malaria endemicity, there will be quite a difference between areas with low malaria transmission (i.e., low communal immunity) and those with intensive transmission (i.e., high naturally acquired immunity). In the presence of high levels of AQ resistance, treatment with AQ is likely to fail, since the degree of potentiation is likely to remain below the curative threshold. The situation will be different in areas with substantial immunity, since the critical threshold level for curative efficacy is situated close to the EC90. As seen from Table 1, the regression parameters of AQ and DAQ are peculiar in the sense that EC90 and EC99 generally differ by 1 order of magnitude or more, in contrast to the situation with chloroquine. Therefore, many patients will still clear the infection, since the DAQ concentration profiles do reach the EC90 level, even for fairly resistant parasites. Additional benefit will be derived from the enhancing effect of AQ.
Significant exposure of P. falciparum to chloroquine in the community has been instrumental in selecting chloroquine-resistant parasite populations and thus has compromised the efficacy of the drug in most areas of endemicity (26, 39). In view of the significant activity correlations between chloroquine and AQ as well as DAQ, it is plausible that resistance to chloroquine has also compromised the efficacy of AQ. However, as the example of the 4-quinolinemethanols mefloquine and quinine indicates, positive activity correlations do not automatically imply manifest cross-resistance. These factors, the antiplasmodial activity profiles of DAQ, the significant potentiating effect of AQ, and the supportive role of immunity may explain the maintained efficacy of AQ in areas of hyper- and holoendemicity where chloroquine resistance has necessitated the replacement of chloroquine in the treatment of uncomplicated falciparum malaria.
The results of this study seem to indicate a difference in the modes of action of AQ and DAQ, highlighting the crucial role of very low AQ concentrations in the potentiation of DAQ activity. This phenomenon is apparently not unique to AQ and DAQ; similar interactions have been observed recently between chloroquine with its metabolite (15), among diastereomers of the Cinchona alkaloids in vitro (10, 34), and between β-artemether and lumefantrine (21, 24).
Conclusions.
The in vitro studies with P. falciparum strains LS-2, LS-3, and LS-1 have consistently shown that AQ interacts positively with its major metabolite, DAQ, the detectable presence of which lasts for several weeks. This potentiating, synergistic activity requires only low AQ concentrations in the picomolar or femtomolar range. It is crucial for the therapeutic efficacy of the drug. In the course of a standard AQ treatment regimen, the putative residual concentrations of AQ are likely to provide sufficient synergistic activity to enhance the efficacy of DAQ for as long as 7 days, a time span usually sufficient to effect radical cure. AQ is a particularly interesting antimalarial drug inasmuch as it is almost completely transformed to the active metabolite, DAQ, but maintains minimal AQ concentrations which significantly potentiate the effect of the metabolite. Under this aspect, treatment with AQ is a combination therapy by itself and should be combined therapeutically with other drugs with different structural and chemical properties (35). The synergism between AQ and DAQ should also be considered in the methodology of in vitro tests with fresh isolates of P. falciparum.
Acknowledgments
This work has been financially supported by grants from the Swedish International Development Agency (Sida/SAREC).
We thank Marianne Laebbad and Berit Aydin Schmidt, Smiktskyddsinstitutet, Karolinska Institute, Stockholm, Sweden, and Margareta Hagstedt, Department of Immunology, Stockholm University, Stockholm, Sweden, for technical assistance.
REFERENCES
- 1.Alin, M. H., A. Bjorkman, and W. H. Wernsdorfer. 1999. Synergism of benflumetol and artemether in Plasmodium falciparum. Am. J. Trop. Med. Hyg. 61:439-445. [DOI] [PubMed] [Google Scholar]
- 1a.Basco, L. K., and P. Ringwald. 2003. In vitro activities of piperaquine and other 4-aminoquinolines against clinical isolates of Plasmodium falciparum in Cameroon. Antimicrob. Agents Chemother. 47:1391-1394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Benito, A., J. Roche, R. Molina, C. Amela, and J. Alvar. 1995. In vitro susceptibility of Plasmodium falciparum to chloroquine, amodiaquine, quinine, mefloquine, and sulphadoxine/pyrimethamine in Equatorial Guinea. Am. J. Trop. Med. Hyg. 53:526-531. [DOI] [PubMed] [Google Scholar]
- 3.Berenbaum, M. C. 1978. A method for testing for synergy with any number of agents. J. Infect. Dis. 137:122-130. [DOI] [PubMed] [Google Scholar]
- 4.Björkman, A., and P. A. Phillips-Howard. 1990. Drug resistant malaria: mechanisms of development and inferences for malaria control. Trans. R. Soc. Trop. Med. Hyg. 84:323-324. [DOI] [PubMed] [Google Scholar]
- 5.Brasseur, P., P. Agnamey, A. S. Ekobo, G. Samba, L. Favennec, and J. Kouamouo. 1995. Sensitivity of Plasmodium falciparum to amodiaquine and chloroquine in central Africa: a comparative study in vivo and in vitro. Trans. R. Soc. Trop. Med. Hyg. 89:528-530. [DOI] [PubMed] [Google Scholar]
- 6.Campbell, C. C., D. Payne, I. K. Schwartz, and O. J. Khatib. 1983. Evaluation of amodiaquine treatment of chloroquine-resistant Plasmodium falciparum malaria in Zanzibar, 1982. Am. J. Trop. Med. Hyg. 32:1216-1220. [DOI] [PubMed] [Google Scholar]
- 7.Childs, G. E., E. F. Boudreau, W. K. Milhous, T. Wimonwattratee, N. Pooyindee, L. Pang, and D. E. Davidson, Jr. 1989. A comparison of the in vitro activities of amodiaquine and desethylamodiaquine against isolates of Plasmodium falciparum. Am. J. Trop. Med. Hyg. 40:7-11. [DOI] [PubMed] [Google Scholar]
- 8.Churchill, F. C., L. C. Patchen, C. C. Campbell, I. K. Schwartz, P. Nguyen-Dinh, and C. M. Dickinson. 1985. Amodiaquine as a prodrug: importance of metabolite(s) in the antimalarial effect of amodiaquine in humans. Life Sci. 36:53-62. [DOI] [PubMed] [Google Scholar]
- 9.Churchill, F. C., D. L. Mount, L. C. Patchen, and A. Bjorkman. 1986. Isolation, characterization and standardization of a major metabolite of amodiaquine by chromatographic and spectroscopic methods. J. Chromatogr. 377:307-318. [DOI] [PubMed] [Google Scholar]
- 9a.Daniel, W. W. 1991. Biostatistics: a foundation for analysis in the health sciences, 5th ed. John Wiley & Sons, New York, N.Y.
- 10.Druilhe, P., O. Brandicourt, T. Chongsuphajaisiddhi, and J. Berthe. 1988. Activity of a combination of three cinchona bark alkaloids against Plasmodium falciparum in vitro. Antimicrob. Agents Chemother. 32:250-254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Elueze, E. I., S. L. Croft, and D. C. Warhurst. 1996. Activity of pyronaridine and mepacrine against twelve strains of Plasmodium falciparum in vitro. J. Antimicrob. Chemother. 37:511-518. [DOI] [PubMed] [Google Scholar]
- 12.Gerstner, U., S. Prajakwong, G. Wiedermann, J. Sirichaisinthop, G. Wernsdorfer, and W. H. Wernsdorfer. 2003. Comparison of the in vitro activity of amodiaquine and its main metabolite, monodesethyl-amodiaquine, in Plasmodium falciparum. Wien. Klin. Wochenschr. 115(Suppl. 3):33-38. [PubMed] [Google Scholar]
- 13.Gupta, S., M. Thapar, S. T. Mariga, W. H. Wernsdorfer, and A. Björkman. 2002. Plasmodium falciparum: in vitro interactions of artemisinin with amodiaquine, pyronaridine, and chloroquine. Exp. Parasitol. 100:28-35. [DOI] [PubMed] [Google Scholar]
- 14.Hatton, C. S., T. E. Peto, C. Bunch, G. Pasvol, S. J. Russell, C. R. Singer, G. Edwards, and P. Winstanley. 1986. Frequency of severe neutropenia associated with amodiaquine prophylaxis against malaria. Lancet i:411-414. [DOI] [PubMed] [Google Scholar]
- 15.Kalkanidis, M., N. Klonis, S. Tschan, L. W. Deady, and L. Tilley. 2004. Synergistic interaction of a chloroquine metabolite with chloroquine against drug-resistant malaria parasites. Biochem. Pharmacol. 67:1347-1353. [DOI] [PubMed] [Google Scholar]
- 16.Laurent, F., S. Saivin, P. Chretien, J. F. Magnaval, F. Peyron, A. Sqalli, A. E. Tufenkji, Y. Coulais, H. Baba, and G. Campistron. 1993. Pharmacokinetic and pharmacodynamic study of amodiaquine and its two metabolites after a single oral dose in human volunteers. Arzneimittelforschung 43:612-616. [PubMed] [Google Scholar]
- 17.Litchfield, J. T., and F. Wilcoxon. 1949. A simplified method of evaluating dose-effect experiments. J. Pharm. Exp. Ther. 96:99-113. [PubMed] [Google Scholar]
- 18.McChesney, E. W., Jr., W. F. Banks, and R. J. Fabian. 1967. Tissue distribution of chloroquine, hydroxychloroquine, and desethylchloroquine in the rat. Toxicol. Appl. Pharmacol. 10:501-513. [DOI] [PubMed] [Google Scholar]
- 19.Minzi, O. M., M. Rais, J. O. Svensson, L. L. Gustafsson, and O. Ericsson. 2003. High performance liquid chromatographic method for determination of amodiaquine, chloroquine and their monodesethyl metabolites in biological samples. J. Chromatogr. B 783:473-480. [DOI] [PubMed] [Google Scholar]
- 20.Neftel, K. A., W. Woodtly, M. Schmid, P. G. Frick, and J. Fehr. 1986. Amodiaquine induced agranulocytosis and liver damage. Br. Med. J. (Clin. Res. Ed.) 292:721-723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Novartis. 1997. Meeting the challenge: a novel antimalarial combination. Current Medical Literature Ltd. Basel, Switzerland.
- 22.Olliaro, P., C. Nevill, J. LeBras, P. Ringwald, P. Mussano, P. Garner, and P. Brasseur. 1996. Systematic review of amodiaquine treatment in uncomplicated malaria. Lancet 348:1196-1201. [DOI] [PubMed] [Google Scholar]
- 23.Pradines, B., C. Rogier, T. Fusai, A. Tall, J. F. Trape, and J. C. Doury. 1998. In vitro activity of artemether against African isolates (Senegal) of Plasmodium falciparum in comparison with standard antimalarial drugs. Am. J. Trop. Med. Hyg. 58:354-357. [DOI] [PubMed] [Google Scholar]
- 24.Pradines, B., A. Tall, T. Fusai, A. Spiegel, R. Hienne, C. Rogier, J. F. Trape, J. Le Bras, and D. Parzy. 1999. In vitro activities of benflumetol against 158 Senegalese isolates of Plasmodium falciparum in comparison with those of standard antimalarial drugs. Antimicrob. Agents Chemother. 43:418-420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pussard, E., F. Verdier, F. Faurisson, J. M. Scherrmann, J. LeBras, and M. Blayo. 1987. Disposition of monodesethylamodiaquine after a single oral dose of amodiaquine and three regimens for prophylaxis against Plasmodium falciparum malaria. Eur. J. Clin. Pharmacol. 33:409-414. [DOI] [PubMed] [Google Scholar]
- 26.Ringwald, P., J. Bickii, and L. K. Basco. 1998. Amodiaquine as the first-line treatment of malaria in Yaounde, Cameroon: presumptive evidence from activity in vitro and cross-resistance patterns. Trans. R. Soc. Trop. Med. Hyg. 92:212-213. [DOI] [PubMed] [Google Scholar]
- 27.Ringwald, P., A. Keundjian, A. Same Ekobo, and L. K. Basco. 2000. Chemoresistance of Plasmodium falciparum in the urban region of Yaounde, Cameroon. Part 2. Evaluation of the efficacy of amodiaquine and sulfadoxine-pyrimethamine combination in the treatment of uncomplicated Plasmodium falciparum malaria in Yaounde, Cameroon. Trop. Med. Int. Health 5:620-627. [DOI] [PubMed] [Google Scholar]
- 28.Segurado, A. A., S. M. di Santi, and M. Shiroma. 1997. In vivo and in vitro Plasmodium falciparum resistance to chloroquine, amodiaquine and quinine in the Brazilian Amazon. Rev. Inst. Med. Trop. Sao Paulo 39:85-90. [DOI] [PubMed] [Google Scholar]
- 29.Trager, W., and J. B. Jensen. 1976. Human malaria parasites in continuous culture. Science 193:673-675. [DOI] [PubMed] [Google Scholar]
- 30.Vince, A., M. Poljak, and K. Seme. 1998. DNA extraction from archival Giemsa-stained bone-marrow slides: comparison of six rapid methods. Br. J. Haematol. 101:349-351. [DOI] [PubMed] [Google Scholar]
- 31.Watt, G., G. W. Long, L. Padre, P. Alban, R. Sangalang, C. P Ranoa, and L. W. Laughlin. 1987. Amodiaquine less effective than chloroquine in the treatment of falciparum malaria in the Philippines. Am. J. Trop. Med. Hyg. 36:3-8. [DOI] [PubMed] [Google Scholar]
- 32.Wernsdorfer. W. H., and D. Payne. 1991. The dynamics of drug resistance in Plasmodium falciparum. Pharmacol. Ther. 50:95-121. [DOI] [PubMed] [Google Scholar]
- 33.Wernsdorfer, W. H., and H. Noedl. 2003. Molecular markers for drug resistance in malaria: use in treatment, diagnosis and epidemiology. Curr. Opin. Infect. Dis. 16:553-558. [DOI] [PubMed] [Google Scholar]
- 34.Wesche, D. L., and J. Black. 1990. A comparison of the antimalarial activity of the cinchona alkaloids against Plasmodium falciparum in vitro. J. Trop. Med. Hyg. 93:153-159. [PubMed] [Google Scholar]
- 35.White, N. 1999. Antimalarial drug resistance and combination chemotherapy. Phil. Trans. R. Soc. Lond. 354:739-749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Winstanley, P., G. Edwards, M. Orme, and A. Breckenridge. 1987. The disposition of amodiaquine in man after oral administration. Br. J. Clin. Pharmacol. 23:1-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Winstanley, P. A., G. Edwards, C. G. Curtis, M. L. Orme, G. M. Powell, and A. M. Breckenridge. 1988. Tissue distribution and excretion of amodiaquine in the rat. J. Pharm. Pharmacol. 40:343-349. [DOI] [PubMed] [Google Scholar]
- 38.Winstanley, P. A., O. Simooya, J. M. Kofi-Ekue, O. Walker, L. A. Salako, G. Edwards, M. L. Orme, and A. M. Breckenridge. 1990. The disposition of amodiaquine in Zambians and Nigerians with malaria. Br. J. Clin. Pharmacol. 29:695-701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Wongsrichanalai, C., A. L. Pickard, W. H. Wernsdorfer, and S. R. Meshnick. 2002. Epidemiology of drug-resistant malaria. Lancet Infect. Dis. 2:209-216. [DOI] [PubMed] [Google Scholar]
- 40.World Health Organization. 2001. The use of antimalarial drugs. Report of a WHO informal consultation. WHO document WHO/CDS/RBM/2001.33, p. 47-50. World Health Organization, Geneva, Switzerland.