Abstract
The purpose of our study was to investigate the protective effects of a natural product—‘curcumin’— in Alzheimer's disease (AD)-like neurons. Although much research has been done in AD, very little has been reported on the effects of curcumin on mitochondrial biogenesis, dynamics, function and synaptic activities. Therefore, the present study investigated the protective effects against amyloid β (Aβ) induced mitochondrial and synaptic toxicities. Using human neuroblastoma (SHSY5Y) cells, curcumin and Aβ, we studied the protective effects of curcumin against Aβ. Further, we also studied preventive (curcumin+Aβ) and intervention (Aβ+curcumin) effects of curcumin against Aβ in SHSY5Y cells. Using real time RT-PCR, immunoblotting and immunofluorescence analysis, we measured mRNA and protein levels of mitochondrial dynamics, mitochondrial biogenesis and synaptic genes. We also assessed mitochondrial function by measuring hydrogen peroxide, lipid peroxidation, cytochrome oxidase activity and mitochondrial ATP. Cell viability was studied using the MTT assay. Aβ was found to impair mitochondrial dynamics, reduce mitochondrial biogenesis and decrease synaptic activity and mitochondrial function. In contrast, curcumin enhanced mitochondrial fusion activity and reduced fission machinery, and increased biogenesis and synaptic proteins. Mitochondrial function and cell viability were elevated in curcumin treated cells. Interestingly, curcumin pre- and post-treated cells incubated with Aβ showed reduced mitochondrial dysfunction, and maintained cell viability and mitochondrial dynamics, mitochondrial biogenesis and synaptic activity. Further, the protective effects of curcumin were stronger in pretreated SHSY5Y cells than in post-treated cells, indicating that curcumin works better in prevention than treatment in AD-like neurons. Our findings suggest that curcumin is a promising drug molecule to treat AD patients.
Keywords: Aging, Alzheimer Disease, Antioxidants
Introduction
Alzheimer's disease (AD) is the most common form of dementia in elderly individuals and is the sixth leading cause of death in the USA. AD is an age dependent and progressive neurodegenerative disease, characterized by the loss of memory, cognitive functions, and changes in behavior and personality.1–3 According to the 2015 World Alzheimer Report, it was estimated that 47.5 million people have dementia worldwide, and the numbers are estimated to increase to 75.6 million by 2030 and to 131.5 million by 2050. Dementia has a huge economic impact, and the 2015 total estimated healthcare cost is about US$818 billion and estimated to increase to US$2 trillion by 2015.4
Causal factors are known for AD for a small proportion (1–2%) of total AD patients, and causal factors are still unknown for the vast majority of AD cases. Several risk factors have been identified, the major one being ApoE4 genotype and polymorphisms in several genetic loci, including sortilin related receptor 1, clusterin, complement component receptor 1, CD2AP, CD33, and EPHA1, and MS4A4/MS4A6E genes are other contributing risk factors.5 In addition, type 2 diabetes, traumatic brain injury, stroke and diet, and environmental factors are other contributing factors. Above all, ageing is the number one risk factor.
Several years of research revealed that AD is associated with multiple cellular changes, including mitochondrial damage, loss of synapses, amyloid β (Aβ) formation and accumulation, activation of microglia and astrocytes, phosphorylation of tau and neurofibrillary tangles formation and loss of neurons.1–3 Therapeutic strategies have been developed based on these cellular changes and currently being tested in preclinical (animal models) and human clinical trials. However, we do not have drugs/agents that can delay and/or prevent disease progression of AD. Further, we still do not have early detectable biomarkers that can identify cognitive decline and memory problems in elderly individuals.
Physical exercise and healthy diets have been reported to have implications in delaying disease progression of AD in elderly individuals and improved cognitive function in subjects with mild cognitive impairment and early AD patients.6 Natural products are the major source of diets that have multiple neuroprotective effects, including anti-inflammatory, antioxidant, anti-arthritis and memory cognitive functions.7–9 There are a large number of natural products and herbs currently available, including curcumin, green tea and vitamin C, vitamin E, β carotene, Gingko biloba, ginseng, rosemary, sage and many others.6–9 As the main theme of this special topic is natural products, in the current study, we studied the protective effects of curcumin against Aβ induced toxicities in the pathogenesis of AD.
Curcumin is the major constituent of the Asian spice, turmeric, isolated from the rhizome of Curcuma longa.10 11 Curcumin was isolated in 1815 as a yellow coloring matter from the rhizomes of Curcuma longa (turmeric)12 and named curcumin. Curcumin has been used historically in Ayurvedic medicine (curcumin is popularly referred to as Haldi in India and its chemical name is diferuloylmethane; molecular formula is C21H20O6). It molecular mass is 368.37 g/mol. Curcumin is extensively used for medicinal purposes in Asia and other parts of the world. Curcumin is used in foods because of its color and flavor. It is also used as a cosmetic product, particularly for skin.
The chemical structure of curcumin is comprised of two aryl rings with ortho-methoxy OH groups linked to β-diketone moiety.13 Several years of research revealed that curcumin has several protective and therapeutic properties, including anti-inflammatory,14–17 antioxidant,14 15 18 19 antiproliferative, anti-atherosclerosis14 and anti-arthritis.14 Curcumin is a strong healing agent.20 A recent study reported that curcumin enhanced the levels of glutathione and antioxidant enzymes, superoxide dismutase and catalase in the brains of lead poisoned rats and significantly reduced lead induced damage.21
Several lines of evidence suggest that curcumin has anti-amyloid properties in AD.
Findings from an in vitro study revealed that curcumin inhibits Aβ aggregation as well as disaggregates to form fibrillar Aβ40.22
Several in vivo studies revealed that curcumin promotes disaggregation of existing amyloid deposits and prevents aggregation of new amyloid deposits, and even reduced the size of remaining deposits.22 23
Curcumin and its derivatives are reported to inhibit the fibrillar Aβ formation from Aβ monomer and also destabilize preformed fibrillar Aβ in vitro, indicating that curcumin is protective against Aβ toxicity.24
Levels of Aβ (40%) and Aβ deposits (43%) were reduced in the brains of amyloid precursor protein (APP) mice treated with low doses of curcumin relative to untreated APP mice. At higher concentrations, curcumin binds to Aβ and blocks its self assembly.22
A recent study reported that curcumin destabilizes Aβ40 and Aβ42.25
Further, curcumin derived isoxazoles and pyrazoles bind to Aβ and inhibit APP metabolism.26
Curcumin protects PC12 cells and normal human umbilical endothelial cells from Aβ induced oxidative stress.27
Curcumin reduced levels of oxidized proteins and interleukin 1B in the brains of APP mice.28
Curcumin enhances Aβ uptake by macrophages in AD patients; bone marrow derived dendritic cells may correct immune defects in AD patients and provide an immunotherapy approach to AD patients.29
Curcumin inhibits peroxidase and modulates the cytopathologies in AD patients.30
Curcumin binds to redox active metals (iron and copper); it suppresses inflammatory damage by preventing metal induction of nuclear factor κB.31
Curcumin crosses the blood–brain barrier because of its lipophilic properties and binds to amyloid deposits.22 Adverse effects have not been reported thus far. Therefore, curcumin can be used in clinical trials. Although much research has been done in AD, very little has been reported on the effects of curcumin on mitochondrial biogenesis, dynamics, function and synaptic activities. Therefore, the present study investigated the protective effects against Aβ induced mitochondrial and synaptic toxicities. In the current study, we used human neuroblastoma cells (SHSY5Y), Aβ25–35 peptide and curcumin.
Materials and methods
Chemicals and reagents
Aβ25–35 peptide was purchased from Anaspec (Fremont, California, USA). Dulbecco's modified eagle's medium, minimum essential medium, penicillin/streptomycin, trypsin-EDTA and fetal bovine serum were purchased from Gibco (Gaithersberg, Maryland, USA). Curcumin was purchased from Sigma-Aldrich (St Louis, Missouri, USA).
SHSY5Y cells
SHSY5Y cells were purchased from American Tissue Type Collection (ATCC), Virginia, USA. Figure 1 illustrates the experimental strategy of our cell culture work, including treatments. Cells were grown in a medium (1:1 Dulbecco's modified eagle's medium and minimum essential medium, 5% fetal bovine serum, 1× penicillin and streptomycin) at 37°C in a humidified incubator with a 5% CO2 environment. After seeding the cells were allowed to grow for 24–48 hours or until 80% confluence in six well plates and then used for experiments. We used five different groups of cells: (1) untreated SHSY5Y cells; (2) SHSY5Y cells incubated with Aβ peptide 25–35 (30 µM final concentration) for 4 hours; (3) SHSY5Y cells treated with curcumin (66.3 µM final concentration) for 24 hours; (4) SHSY5Y cells treated Aβ for 4 hours+curcumin for 24 hours and (5) SHSY5Y cells treated curcumin for 24 hours and Aβ for 4 hours. Half a million SHSY5Y cells were suspended per well into six well plates. We used Aβ peptide 25–35 and curcumin 66.3 nM in our experiments. (From here on, SHSY5Y cells will be referred to as cells). After the treatments, we harvested the cells, conducted experiments to measure levels of mRNA using a Sybr-Green based real time RT-PCR, protein levels using immunoblotting and immunofluorescence analysis and cell viability using MTT assay.
Figure 1.
Experimental strategy of amyloid β (Aβ), curcumin, Aβ+curcumin and curcumin+Aβ treatments in human neuroblastoma (SHSY5Y) cells.
Data were compared in two ways: (1) untreated cells versus cells treated with Aβ, curcumin, curcumin+Aβ and Aβ+curcumin and (2) cells treated with Aβ versus curcumin+Aβ and Aβ+curcumin.
Quantification of mRNA expression of mitochondrial dynamics, mitochondrial biogenesis and synaptic genes using real time RT-PCR
Using the reagent TriZol (Invitrogen), we isolated total RNA from control and experimental groups (figure 1). Using primer express software (Applied Biosystems), we designed the oligonucleotide primers for the housekeeping genes β actin; GAPDH; mitochondrial biogenesis genes PGC1α, Nrf1, Nrf2 and TFAM; mitochondrial structural genes; fission (Drp1 and Fis1); fusion genes (MFN1, MFN2, Opa1); mitochondrial matrix protein (CypD); and synaptic genes (synaptophysin and PSD95). The primer sequences and amplicon sizes are listed in table 1. Using Sybr-Green chemistry based quantitative real time RT-PCR, we measured mRNA expression of the genes mentioned above, as described by Manczak et al.32
Table 1.
Summary of quantitative real time RT-PCR oligonucleotide primers used in measuring mRNA expression in mitochondrial dynamics, mitochondrial biogenesis and synaptic genes
| Gene | DNA Sequence (5′-3′) | PCR product size |
|---|---|---|
| Mitochondrial dynamics genes | ||
| Drp1 | Forward primer ATGCCAGCAAGTCCACAGAA | 86 |
| Reverse primer TGTTCTCGGGCAGACAGTTT | ||
| Fis1 | Forward primer CAAAGAGGAACAGCGGGACT | 95 |
| Reverse primer ACAGCCCTCGCACATACTTT | ||
| MFN1 | Forward primer GCAGACAGCACATGGAGAGA | 83 |
| Reverse primer GATCCGATTCCGAGCTTCCG | ||
| MFN2 | Forward primer TGCACCGCCATATAGAGGAAG | 78 |
| Reverse primer TCTGCAGTGAACTGGCAATG | ||
| Cyclophilin D | Forward primer AGATGTCAAATTGGCAGGGGG | 91 |
| Reverse primer TGCGCTTTTCGGTATAGTGCT | ||
| Opa1 | Forward primer ACCTTGCCAGTTTAGCTCCC | 82 |
| Reverse primer TTGGGACCTGCAGTGAAGAA | ||
| Mitochondrial biogenesis genes | ||
| PGC1α | Forward primer GCAGTCGCAACATGCTCAAG | 83 |
| Reverse primer GGGAACCCTTGGGGTCATTT | ||
| Nrf1 | Forward primer AGAAACGGAAACGGCCTCAT | 96 |
| Reverse primer CATCCAACGTGGCTCTGAGT | ||
| Nrf2 | Forward primer ATGGAGCAAGTTTGGCAGGA | 96 |
| Reverse primer GCTGGGAACAGCGGTAGTAT | ||
| TFAM | Forward primer TCCACAGAACAGCTACCCAA | 84 |
| Reverse primer CCACAGGGCTGCAATTTTCC | ||
| Reverse primer AGACGGTTGTTGATTAGGCGT | ||
| Synaptic genes | ||
| Synaptophysin | Forward primer CTGCGTTAAAGGGGGCACTA | 81 |
| Reverse primer ACAGCCACGGTGACAAAGAA | ||
| PSD95 | Forward primer CTTCATCCTTGCTGGGGGTC | 90 |
| Reverse primer TTGCGGAGGTCAACACCATT | ||
| Housekeeping genes | ||
| β Actin | Forward primer AGAAGCTGTGCTATGTTGCTCTA | 91 |
| Reverse primer TCAGGCAGCTCATAGCTCTTC | ||
| GAPDH | Forward primer TTCCCGTTCAGCTCTGGG | 59 |
| Reverse primer CCCTGCATCCACTGGTGC | ||
Immunoblotting analysis
To determine whether curcumin or Aβ alters protein levels of mitochondrial and synaptic genes that showed altered mRNA expression in our real time RT-PCR, we performed immunoblotting analyses of protein lysates from cells from the control and experimental treatments in independent cell treatments (n=3), as described by Manczak et al.33 Details of proteins and dilutions of antibodies used for immunoblotting analysis are listed in table 2.
Table 2.
Summary of antibody dilutions and conditions used in the immunoblotting analysis of mitochondrial dynamics, mitochondrial biogenesis and synaptic proteins
| Marker | Primary antibody—species and dilution | Purchased from (company, location) | Secondary antibody, dilution | Purchased from (company, location) |
|---|---|---|---|---|
| Drp1 | Rabbit polyclonal 1:500 | Novus Biological, Littleton, Colorado, USA | Donkey anti-rabbit HRP 1:10,000 | GE Healthcare Amersham, Piscataway, New Jersey, USA |
| Fis1 | Rabbit polyclonal 1:500 | MBL International Corporation-life. Woburn, Massachusetts, USA | Donkey anti-rabbit HRP 1:10,000 | GE Healthcare Amersham, Piscataway, New Jersey, USA |
| Mfn1 | Rabbit polyclonal 1:400 | Novus Biological, Littleton, Colorado, USA | Donkey anti-rabbit HRP 1:10,000 | GE Healthcare Amersham, Piscataway, New Jersey, USA |
| Mfn2 | Rabbit polyclonal 1:400 | Abcam, Cambridge, Massachusetts, USA | Donkey anti-rabbit HRP 1:10,000 | GE Healthcare Amersham, Piscataway, New Jersey, USA |
| Opa1 | Rabbit polyclonal 1:500 | Novus Biological, Littleton, Colorado, USA | Donkey anti-rabbit HRP 1:10,000 | GE Healthcare Amersham, Piscataway, New Jersey, USA |
| CypD | Mouse monoclonal 1:500 | EMD, Calobiochem Chemicals INC, Gibbstown, New Jersey, USA | Sheep anti-mouse HRP 1:10,000 | GE Healthcare Amersham, Piscataway, New Jersey, USA |
| SYN | Rabbit polyclonal 1:400 | Abcam, Cambridge, Massachusetts, USA | Donkey anti-rabbit HRP 1:10,000 | GE Healthcare Amersham, Piscataway, New Jersey, USA |
| PGC1α | Rabbit polyclonal 0.5 µg/mL | Novus Biological, Littleton, Colorado, USA | Donkey anti-rabbit HRP 1:10,000 | GE Healthcare Amersham, Piscataway, New Jersey, USA |
| Nrf1 | Rabbit monoclonal 1:1000 | Abcam, Cambridge, Massachusetts, USA | Donkey anti-rabbit HRP 1:10,000 | GE Healthcare Amersham, Piscataway, New Jersey, USA |
| Nrf2 | Rabbit polyclonal 1:500 | Novus Biological, Littleton, Colorado, USA | Donkey anti-rabbit HRP 1:10,000 | GE Healthcare Amersham, Piscataway, New Jersey, USA |
| TFAM | Rabbit polyclonal 1:1000 | Abcam, Cambridge, Massachusetts, USA | Donkey anti-rabbit HRP 1:10,000 | GE Healthcare Amersham, Piscataway, New Jersey, USA |
| PSD95 | Rabbit monoclonal 1:300 | Abcam, Cambridge, Massachusetts, USA | Donkey anti-rabbit HRP 1:10,000 | GE Healthcare Amersham, Piscataway, New Jersey, USA |
| B-actin | Mouse monoclonal 1:500 | Sigma-Aldrich, St Louis, Missouri, USA | Sheep anti-mouse HRP 1:10,000 | GE Healthcare Amersham, Piscataway, New Jersey, USA |
Immunofluorescence analysis
To study the immunoreactivity of proteins of interest, cells were grown on coverslips using regular cell culture medium and immunofluorescence analysis was performed using the antibodies mentioned in the control untreated cells and in the cells from the multiple experimental treatments, as described Manzcak et al.33 Details of proteins and dilutions of antibodies used for the immunofluorescence analysis are given in table 3. Cells were washed with warm phosphate buffered saline (PBS), fixed in freshly prepared 4% paraformaldehyde in PBS for 10 min, and then washed with PBS and permeabilized with 0.1% Triton-X100 in PBS. They were blocked with 1% blocking solution (Invitrogen) for 1 hour at room temperature. All neurons were incubated overnight with primary antibodies. The neurons were incubated with appropriate secondary antibodies. The neurons were washed three times with PBS, and slides were mounted. Photographs were taken with a multiphoton laser scanning microscope system (ZeissMeta LSM510). To quantify the immunoreactivity of the proteins of interest, for each treatment 10–15 photographs were taken at ×20 magnification.
Table 3.
Summary of antibody dilutions and conditions used in the immunohistochemistry/immunofluorescence analysis of mitochondrial dynamics, mitochondrial biogenesis and synaptic proteins
| Marker | Primary antibody—species and dilution | Purchased from (company, location) | Secondary antibody, dilution, Alexa fluor dye | Purchased from (company, location) |
|---|---|---|---|---|
| Drp1 | Rabbit polyclonal 1:300 | Novus Biological, Littleton, Colorado, USA | Donkey anti-rabbit IgG Alexa Fluor 488 conjugate | Thermo Fisher Scientific, Waltham, Massachusetts, USA |
| Fis1 | Rabbit polyclonal 1:300 | Protein Tech Group, Inc, Chicago, Illinois, USA | Donkey anti-rabbit IgG Alexa Fluor 488 conjugate | Thermo Fisher Scientific, Waltham, Massachusetts, USA |
| Mfn1 | Rabbit polyclonal 1:300 | Protein Tech Group, Inc, Chicago, Illinois, USA | Donkey anti-rabbit IgG Alexa Fluor 488 conjugate | Thermo Fisher Scientific, Waltham, Massachusetts, USA |
| Mfn2 | Rabbit polyclonal 1:200 | Protein Tech Group, Inc, Chicago, Illinois, USA | Donkey anti-rabbit IgG Alexa Fluor 488 conjugate | Thermo Fisher Scientific, Waltham, Massachusetts, USA |
| OPA1 | Rabbit polyclonal 1:500 | Novus Biological, Littleton, Colorado, USA | Donkey anti-rabbit IgG Alexa Fluor 488 conjugate | Thermo Fisher Scientific, Waltham, Massachusetts, USA |
| SYN | Rabbit polyclonal 1:200 | Protein Tech Group, Inc, Chicago, Illinois, USA | Donkey anti-rabbit IgG Alexa Fluor 488 conjugate | Thermo Fisher Scientific, Waltham, Massachusetts, USA |
| PSD95 | Rabbit polyclonal 1:400 | Protein Tech Group, Inc, Chicago, Illinois, USA | Donkey anti-rabbit IgG Alexa Fluor 488 conjugate | Thermo Fisher Scientific, Waltham, Massachusetts, USA |
Hydrogen peroxide production
Using an Amplex Red H2O2 assay kit (Molecular Probes, Eugene, Oregon, USA), we measured the production of H2O2 in independent experiments (n=4) of SHSY5Y neurons treated (1) with and (2) without curcumin, curcumin treated and then incubated with Aβ, as described by Manczak et al.33
Cytochrome oxidase activity
Cytochrome oxidase activity was measured in mitochondria isolated from SHSY5Y cells of control and experimental treatments (n=4), as described by Manczak et al.33 Enzyme activity was assayed spectrophotometrically using a Sigma kit (Sigma-Aldrich) following the manufacturer's instructions.
ATP levels
ATP levels were measured in mitochondria isolated from SHSY5Y neurons of control and experimental treatments (n=4) using the ATP determination kit (Molecular Probes), as described by Manczak et al.33
Lipid peroxidation assay
Lipid peroxidates are unstable indicators of oxidative stress in neurons. 4-Hydroxy-2-nonenol (HNE) is the final product of lipid peroxidation and this was measured in the cell lysates from SHSY5Y cells of control and experimental treatments (n=4), using an HNE-His ELISA kit (Cell BioLabs, San Diego, California, USA), as described by Manczak et al.33
Cell viability test (MTT assay)
Mitochondrial respiration, an indicator of cell viability, was assessed in SHSY5Y cells from control and experimental treatments (n=4), using the mitochondrial dependent reduction of 3-(4,5-dimethyl-thiazol-2-yl)-2,5-diphenyl-tetrazolium bromide (MTT) to formazan, as described by Manczak et al.33
Statistical analyses
Statistical analyses were conducted in two ways: (1) untreated cells versus cells treated with Aβ, curcumin, curcumin+Aβ and Aβ+curcumin and (2) cells treated with Aβ versus curcumin+Aβ and Aβ+curcumin for mRNA and protein levels, cell viability and mitochondrial functional parameters H2O2, cytochrome oxidase activity, lipid peroxidation, ATP production and cell viability, using appropriate statistical analysis.
Results
mRNA expression of mitochondrial dynamics genes
Aβ
In cells treated with Aβ compared with untreated cells, mRNA expression levels significantly increased in Drp1, by 2.0 fold (p=0.004), and in Fis1, by 2.1 fold (p=0.002) (table 4). In contrast, levels of mRNA expression of mitochondrial fusion genes were significantly decreased: Mfn1 by 3.0 fold (p=0.001), Mfn2 by 1.8 fold (p=0.004) and Opa1 by 1.6 fold (p=0.01), indicating the presence of abnormal mitochondrial dynamics in cells treated with Aβ. Interestingly, mRNA expression of matrix gene CypD was significantly increased by 2.7-fold (p=0.002) in cells treated with Aβ.
Table 4.
mRNA fold changes in mitochondrial dynamics, mitochondrial biogenesis and synaptic genes in SHSY5Y cells treated with Aβ, curcumin, Aβ+curcumin and curcumin+Aβ relative to untreated SHSY5Y cells
| mRNA fold changes compared with untreated cells | mRNA fold changes compared with Aβ treated cells | |||||
|---|---|---|---|---|---|---|
| Genes | Aβ | Cur | Aβ+Cur | Cur+Aβ | Aβ+Cur | Cur+Aβ |
| Mitochondrial dynamics genes | ||||||
| Drp1 | 2.0* | −1.5† | −1.1 | 1.4 | −1.7† | −1.6† |
| Fis1 | 2.1* | −1.4† | 1.1 | 1.1 | −1.8* | −2.2† |
| Mfn1 | 3.0* | 1.5† | 1.1 | 1.2 | 2.8* | 3.8* |
| Mfn2 | −1.8* | 1.4† | 1.1 | 1.2 | 1.6† | 1.9† |
| OPA1 | −1.6† | 1.4† | −1.2 | 1.0 | 1.6† | 2.2* |
| Cyclophilin D | 2.7* | −1.6† | 1.4 | 1.2 | −2.0* | −2.2* |
| Mitochondrial biogenesis genes | ||||||
| PGC1α | −1.5† | −2.1† | 1.0 | 1.2 | 1.5† | 1.8† |
| Nrf1 | −1.9† | −2.1† | −1.2 | 1.1 | 1.6† | 2.2* |
| Nrf2 | −2.6* | −1.4† | −1.4 | 1.1 | 1.9† | 2.3* |
| TFAM | −1.7* | −1.8* | 1.0 | 1.2 | 1.8† | 1.8† |
| Synaptic genes | ||||||
| Synaptophysin | −2.3* | −1.6* | 1.4 | 1.2 | 3.2* | 2.6* |
| PSD-95 | −1.6† | −1.2 | 1.0 | 1.0 | 1.6† | 1.9† |
*p≤0.005.
†p≤0.05.
Aβ, amyloid β; Cur, curcumin.
Curcumin
mRNA levels were significantly decreased for Drp1 (1.5 fold decrease, p=0.02 in curcumin treated cells) and Fis1 (1.4 fold decrease, p=0.03) and increased for fusion genes (Mfn1 by 1.5 fold (p=0.04), Mfn2 by 1.4 fold (p=0.04) and Opa1 by 1.4 fold (p=0.04)). CypD was reduced by 1.6 fold (p=0.01) (table 4).
Aβ+curcumin
In cells incubated with Aβ and then treated with curcumin relative to untreated cells, mRNA levels were unchanged for fission genes Drp1 and Fis1, and fusion genes (Mfn1, Mfn2, Opa1 and CypD) (table 4).
Curcumin+Aβ
Similarly, cells pretreated with curcumin and incubated with Aβ relative to untreated cells, showed no significant mRNA changes for fission, fusion or matrix genes.
Mitochondrial biogenesis genes
Aβ
To determine the effects of Aβ and curcumin on mitochondrial biogenesis genes, mRNA expression levels of PGC1α, Nrf1, Nrf2 and TFAM genes were measured. Significantly decreased mRNA expression was found in all biogenesis genes (PGC1α by 1.5 fold (p=0.04), Nrf1 by 1.9 fold (p=0.02), Nrf2 by 2.6 fold (p=0.002) and TFAM by 1.7 fold (p=0.003)) in cells treated with Aβ relative to untreated cells.
Curcumin
mRNA levels were significantly increased for PGC1α (2.1 fold decrease, p=0.003), Nrf1 by 2.1 fold (p=0.003), Nrf2 by 1.4 fold (p=0.04) and TFAM by 1.8 fold (p=0.03) in curcumin treated cells relative to untreated cells (table 4). These observations indicate that curcumin increases mitochondrial biogenesis activity.
Aβ+curcumin
In cells incubated with Aβ first and then treated with curcumin relative to untreated cells, mRNA levels were unchanged for PGC1α and TFAM and slightly decreased for Nrf1 (by 1.2 fold) and Nrf2 (1.4 fold) (table 4).
Curcumin+Aβ
In cells pretreated with curcumin and incubated with Aβ relative to untreated cells, we found increased levels of mRNA for biogenesis genes (PGC1α by 1.2 fold, Nrf1 by 1.1 fold, Nrf2 by 1.2 and TFAM by 1.1). These results suggest that curcumin pretreatment prevented Aβ induced biogenesis toxicity.
Synaptic genes
Aβ
In cells treated with Aβ compared with untreated cells, mRNA expression levels were decreased for synaptophysin by 2.3 fold (p=0.01) and PSD95 by 1.6 fold (p=0.03), indicating that Aβ reduces synaptic activity (table 4).
Curcumin
mRNA levels were significantly increased for synaptophysin by 1.6 fold (p=0.01) and PSD95 by 1.2 fold in curcumin treated cells relative to untreated cells (table 4). These observations indicate that curcumin boosts synaptic activity in healthy cells.
Aβ+curcumin
In cells incubated with Aβ and then treated with curcumin relative to untreated cells, mRNA levels were increased for synaptophysin by 1.4 fold and unchanged for PSD95 (table 4). These observations indicate that curcumin rescued synaptic activity from Aβ induced toxicity.
Curcumin+Aβ
In cells pretreated with curcumin and incubated with Aβ relative to untreated cells, mRNA levels were increased for synaptophysin by 1.2 fold and 1.1 fold for PSD95, indicating that curcumin prevented Aβ induced synaptic activity.
Comparison with Aβ treated cells
As shown in table 4, mRNA levels of fission genes were reduced in cells treated with Aβ+curcumin (Drp1 by 1.7 fold, p=0.02; Fis1 by 1.8 fold, p=0.002) and curcumin+Aβ (Drp1 by 1.6 fold, p=0.02; Fis1 by 2.2 fold, p=0.003) relative to Aβ treated cells.
In contrast, fusion genes were increased in Aβ+curcumin (Mfn1 by 2.8 fold, p=0.003; Mfn2 by 1.6 fold, p=0.03; Opa1 by 1.6, p=0.03) and curcumin+Aβ (Mfn1 by 3.8 fold, p=0.002; Mfn2 by 1.9 fold, p=0.01; Opa1 by 2.2 fold, p=0.002) treated cells relative to Aβ treated cells.
Mitochondrial biogenesis genes were increased in Aβ+curcumin (PGC1α by 1.5 fold, p=0.04; Nrf1 by 1.6 fold, p=0.03; Nrf2 by 1.9 fold, p=0.01; and TFAM by 1.8 fold, p=0.03) and curcumin+Aβ (PGC1α by 1.8 fold, p=0.03; Nrf1 by 2.2 fold, p=0.002; Nrf2 by 2.3 fold, p=0.002; and TFAM by 1.8 fold, p=0.02) treated cells relative to Aβ treated cells.
Similar to biogenesis genes, synaptic genes were increased in Aβ+curcumin (synaptophysin by 3.2 fold, p=0.002; PSD95 by 1.6 fold, p=0.04) and curcumin+Aβ (synaptophysin 2.6 fold, p=0.004; PSD95 by 1.9 fold, p=0.03) treated cells relative to Aβ treated cells (table 4).
Immunoblotting analysis
To determine the effects of Aβ on mitochondrial proteins and the useful effects of curcumin at the protein level, we quantified mitochondrial proteins in three independent treatments of cells with Aβ, curcumin, Aβ+curcumin and curcumin+Aβ.
Comparison with untreated cells
Aβ
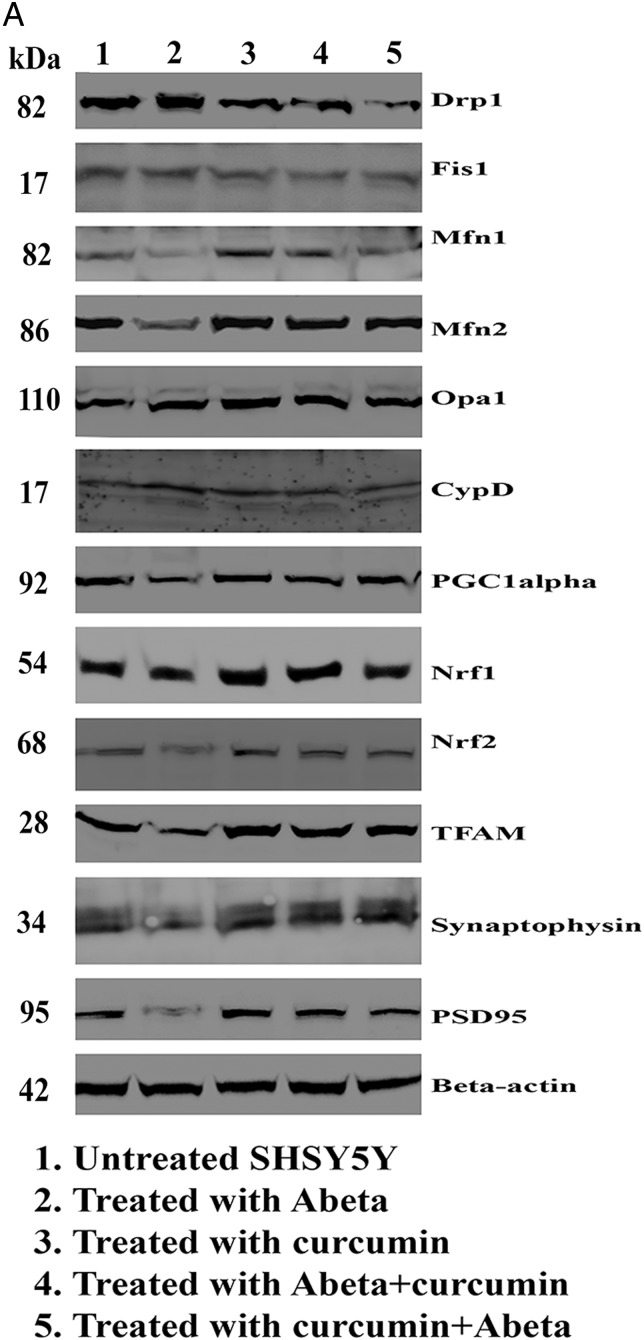
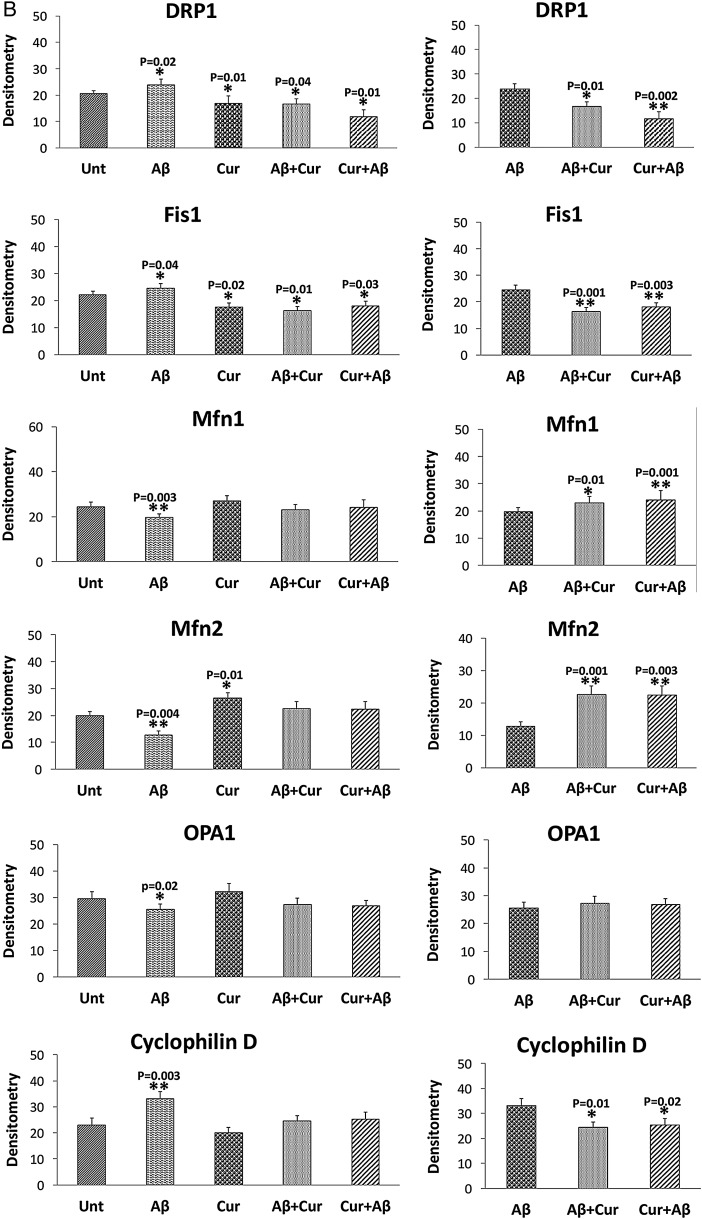
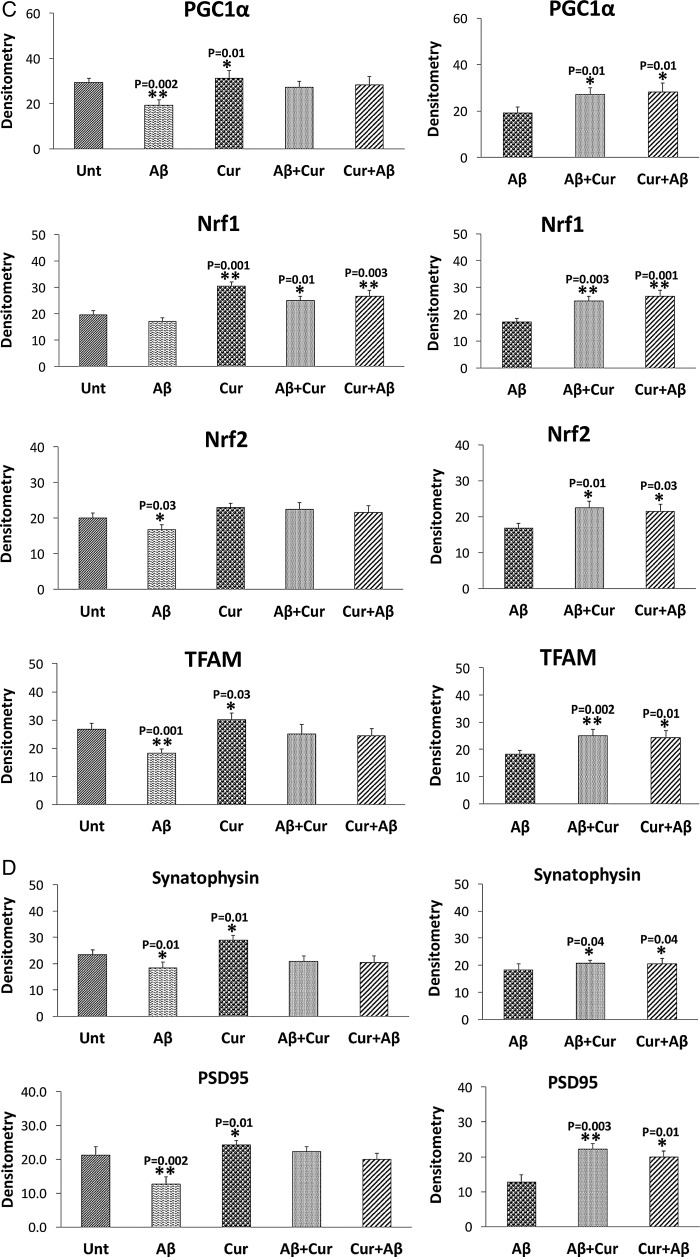
In SHSY5Y cells treated with Aβ compared with untreated SHSY5Y cells, significantly increased proteins levels were found for Drp1 (p=0.02) and Fis1 (p=0.04) (figure 2A, B). In contrast, decreased levels of mitochondrial fusion proteins, Mfn1 (p=0.003), Mfn2 (p=0.004) and Opa1 (p=0.02), were found in cells incubated with Aβ compared with untreated cells. CypD levels (p=0.003) were increased in Aβ treated cells, indicating that Aβ affects mitochondrial dynamic proteins and these observations concur with the mRNA findings. Mitochondrial biogenesis proteins (PGC1α, p=0.002; Nrf2, p=0.03; TFAM, p=0.001) were decreased in Aβ treated cells relative to untreated cells (figure 2A, C). Synaptic proteins, synaptophysin (p=0.01) and PSD95 (p=0.002) levels were significantly reduced in Aβ incubated cells relative to untreated cells (figure 2A, D).
Figure 2.
Immunoblotting analysis of human neuroblastoma (SHSY5Y) cells treated with amyloid β (Aβ), curcumin, Aβ+curcumin and curcumin+Aβ relative to untreated cells. (A) Representative immunoblot. (B–D) Quantitative densitometry analysis of mitochondrial dynamics, mitochondrial biogenesis and synaptic proteins. Cur, curcumin; Unt, untreated.
Curcumin
Mitochondrial fission proteins (Drp1, p=0.01; Fis1, p=0.02) were significantly reduced and fusion protein Mfn2 p=0.01 was significantly increased in cells (figure 2A, C). Interestingly, biogenesis proteins (PGC1α, p=0.01; Nrf1, p=0.001; TFAM, p=0.03) were significantly increased in curcumin treated cells relative to untreated cells. Synaptic proteins, synaptophysin (p=0.01) and PSD95 (p=0.01) levels significantly increased in Aβ incubated cells relative to untreated cells.
Aβ+curcumin
Mitochondrial fission proteins (Drp1, p=0.04; Fis1 p=0.01) were reduced in Aβ+curcumin treated cells relative to untreated cells (figure 2A, B).
Curcumin+Aβ
Similar results (Drp1, p=0.01; Fis1, p=0.03) were found in curcumin+Aβ treated cells relative to untreated cells (figure 2A, B). Overall, these findings suggest that curcumin reduces fission activity and enhances fusion and biogenesis activities in the presence of Aβ.
Comparison with Aβ treated cells
As shown in figure 2 (A, B), significantly reduced levels of fission proteins were found in cells treated with Aβ+curcumin (Drp1, p=0.01; Fis1, p=0.001) and curcumin+Aβ (Drp1, p=0.002; Fis1, p=0.003) relative to Aβ treated cells. In contrast, fusion proteins were increased in Aβ+curcumin (Mfn1, p=0.01; Mfn2, p=0.001) and curcumin+Aβ (Mfn1, p=0.001; Mfn2, p=0.003) treated cells relative to Aβ treated cells.
Mitochondrial biogenesis proteins were significantly increased in Aβ+curcumin (PGC1α, p=0.01; Nrf1, p=0.003; Nrf2, p=0.01; TFAM, p=0.002) and curcumin+Aβ (PGC1α, p=0.01; Nrf1, p=0.001; Nrf2, p=0.03; TFAM p=0.01) cells relative to Aβ treated cells (figure 2A, C), indicating that curcumin enhances biogenesis in the presence of Aβ.
Synaptic proteins were increased in Aβ+curcumin (synaptophysin, p=0.04; PSD95, p=0.003) and curcumin+Aβ (synaptophysin, p=0.04; PSD95, p=0.01) treated cells relative to Aβ treated cells (figure 2A, D), indicating that curcumin enhances synaptic activity in the presence of Aβ in cells.
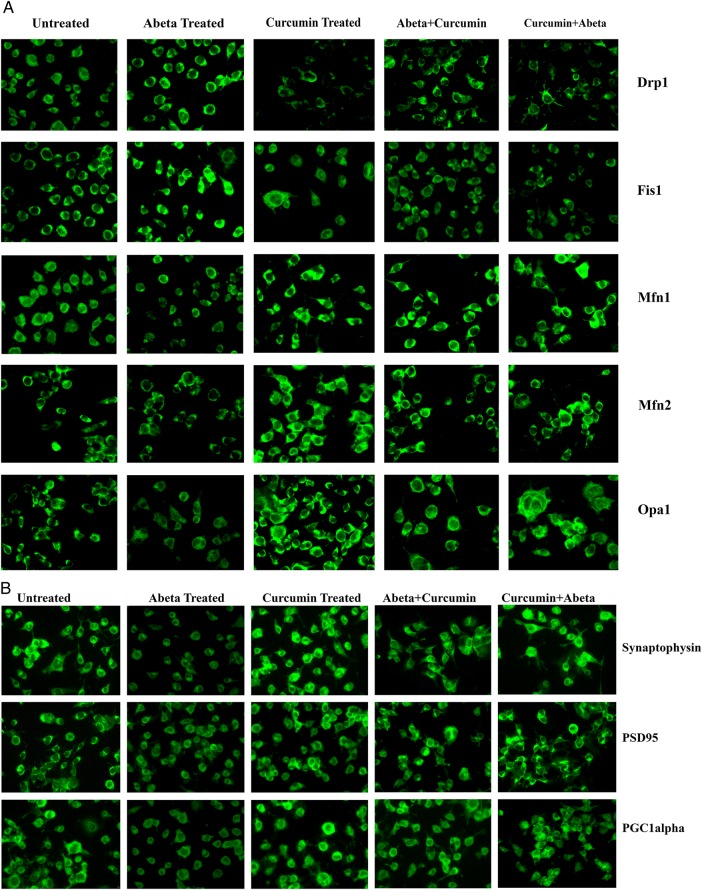
Immunofluorescence analysis
To determine the effect of Aβ and curcumin on mitochondrial dynamics (Drp1, Fis1—fission; Mfn1, Mfn2 and Opa1—fusion), mitochondrial biogenesis (PGC1α) and synaptic protein (synaptophysin and PSD95) levels and localizations, immunofluorescence analysis was performed in cells treated with Aβ and curcumin.
Comparison with untreated cells
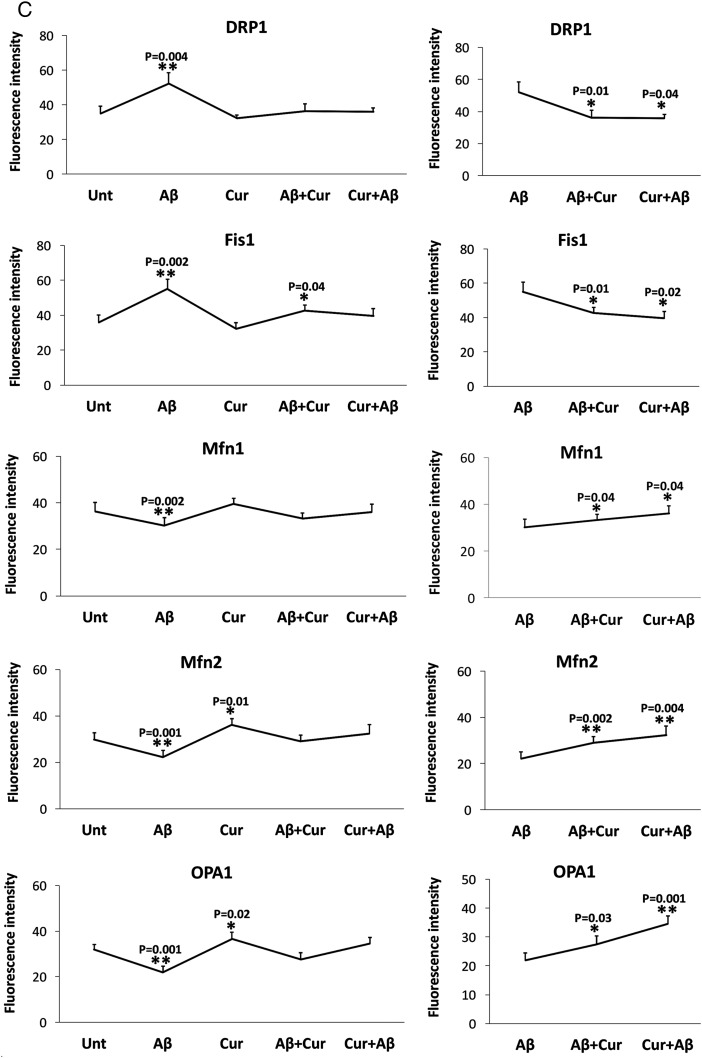
As shown in figure 3 (A, C), we found significantly increased levels of Drp1 (p=0.004) and Fis1 (p=0.02), and significantly decreased levels of Mfn1 (p=0.002), Mfn2 (p=0.001) and Opa1 (p=0.001) in Aβ treated cells relative to untreated cells, indicating that Aβ enhances fission activity and reduces fusion activity in cells. In contrast, decreased levels of Drp1 and Fis1 were found in curcumin treated cells relative to untreated cells, but this was not significant.
Figure 3.
Immunofluorescence analysis of human neuroblastoma (SHSY5Y) cells treated with amyloid β (Aβ), curcumin, Aβ+curcumin and curcumin+Aβ relative to untreated cells. (A) Representative immunofluorescence images of mitochondrial dynamic proteins. (B) Representative immunofluorescence analysis of mitochondrial biogenesis and synaptic proteins. (C) Quantitative immunofluorescence analysis of mitochondrial dynamics proteins. (D) Quantitative immunofluorescence analysis of mitochondrial biogenesis and synaptic proteins.
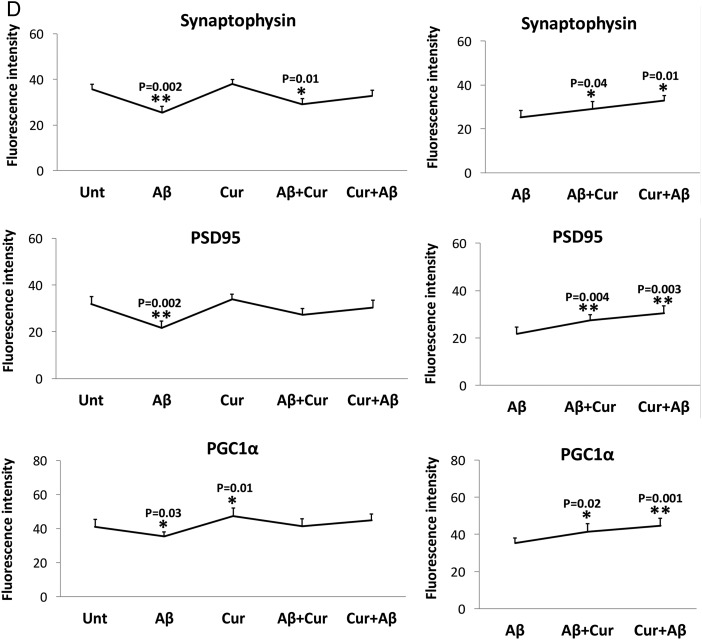
The synaptic proteins synaptophysin (p=0.002) and PSD95 (p=0.002), and PGC1α levels (p=0.03) were significantly reduced in Aβ treated cells relative to untreated cells (figure 3B, D). Overall, the immunofluorescence findings agreed with the immunoblotting results.
Comparison with Aβ treated cells
Significantly reduced levels of fission proteins were found in cells treated with Aβ+curcumin (Drp1, p=0.01; Fis1, p=0.04) and curcumin+Aβ (Drp1, p=0.04; Fis1, p=0.02) relative to Aβ treated cells (figure 3A, C). In contrast, fusion proteins were increased in Aβ+curcumin (Mfn1, p=0.04; Mfn2, p=0.002; Opa1, p=0.02) and curcumin+Aβ (Mfn1, p=0.04; Mfn2, p=0.004; Opa1 p=0.001) treated cells relative to Aβ treated cells (figure 3A, C).
Mitochondrial biogenesis protein PGC1α was significantly increased in Aβ+curcumin (PGC1α, p=0.02) and curcumin+Aβ (PGC1α, p=0.001) treated cells relative to Aβ treated cells (figure 3B, D), indicating that curcumin enhances mitochondrial biogenesis in the presence of Aβ.
Synaptic proteins were increased in Aβ+curcumin (synaptophysin, p=0.04; PSD95, p=0.004) and curcumin+Aβ (synaptophysin, p=0.01; PSD95, p=0.003) treated cells relative to Aβ treated cells (figure 3B, D), indicating that curcumin enhances synaptic activity in the presence of Aβ in cells.
Mitochondrial dysfunction
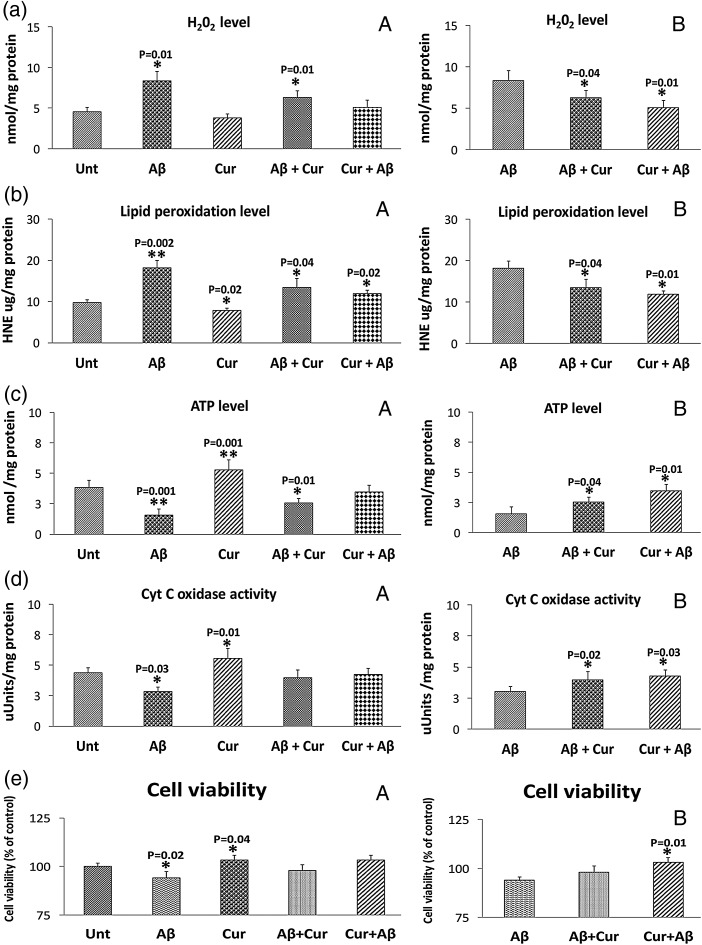
H2O2production
As shown in figure 4, significantly increased levels of hydrogen peroxide (H2O2) were found in mitochondria from cells incubated with Aβ (p=0.01). In measurements taken of H2O2 from mitochondria isolated from cells treated with curcumin, significantly decreased levels (p=0.01) of H2O2 were found relative to untreated cells. These findings suggest that Aβ increases free radicals and curcumin reduces H2O2 in the presence of Aβ.
Figure 4.
Mitochondrial functional parameters in control human neuroblastoma (SHSY5Y) cells, in amyloid β (Aβ) incubated SHSY5Y cells, in SHSY5Y cells treated with curcumin and in SHSY5Y cells incubated with Aβ and then treated with Aβ and in SHSY5Y cells treated with curcumin and then incubated with Aβ (n=4). We analyzed mitochondrial functional data in two ways: (1) the control SHSY5Y cells were compared with the SHSY5Y cells treated with Aβ, curcumin, Aβ+cucumin and curcumin+Aβ and (2) Aβ incubated SHSY5Y cells were compared with Aβ+curcumin SHSY5Y cells and curcumin+Aβ treated cells. We performed statistical analysis using ANOVA following the Dunnett correction, for: (a) H2O2 production, (b) lipid peroxidation, (c) ATP levels, (d) cytochrome oxidase activity and (e) cell viability.
When the data were compared between cells incubated with Aβ with Aβ+curcumin (p=0.04) and curcumin+Aβ (p=0.01), H2O2 levels were significantly reduced, indicating that curcumin reduces H2O2 levels in the presence of Aβ (figure 4).
Lipid peroxidation
To determine whether Aβ affects lipid peroxidation in cells that underwent Aβ incubation, we measured 4-HNE, an indicator of lipid peroxidation. Significantly increased levels of lipid peroxidation were found (p=0.002) in Aβ treated relative to untreated cells (figure 4). However, significantly decreased levels were found in the curcumin treated cells (p=0.02) relative to untreated cells. Significantly increased levels were found in cells incubated with Aβ+curcumin (p=0.04) and curcumin+Aβ (p=0.02) relative to untreated cells.
When the data were compared between cells incubated with Aβ with Aβ+curcumin (p=0.04) and curcumin+Aβ (p=0.01), lipid peroxidation levels were significantly reduced, indicating that curcumin reduces lipid peroxidation levels in the presence of Aβ (figure 4).
ATP production
As shown in figure 4, significantly decreased levels of ATP were found in cells that were incubated with Aβ (p=0.001) relative to untreated cells. Significantly increased levels of ATP were found in cells treated with curcumin (p=0.001) compared with untreated cells. Significantly increased levels were found in cells incubated with Aβ+curcumin (p=0.01) relative to untreated cells (figure 4).
Significantly increased ATP levels were found in Aβ+curcumin (p=0.01) and curcumin+Aβ treated (p=0.001) cells relative to Aβ incubated cells, indicating curcumin increases ATP levels in the presence of Aβ.
Cytochrome oxidase activity
Significantly decreased levels of cytochrome oxidase activity were found in cells that were incubated with Aβ (p=0.03) (figure 4). However, significantly increased levels of cytochrome oxidase activity were found in curcumin treated cells (p=0.01) relative to untreated cells. Cytochrome oxidase activity levels were unchanged in Aβ+curcumin and curcumin+Aβ cells relative to untreated cells.
Similar to ATP levels, cytochrome oxidase activity levels were increased in Aβ+curcumin (p=0.02) and curcumin+Aβ treated (p=0.03) cells relative to Aβ incubated cells (figure 4), indicating curcumin increases cytochrome oxidase activity levels in the presence of Aβ.
Cell viability
Significantly decreased levels of cell viability were found in Aβ treated cells (p=0.02) (figure 4) relative to untreated cells. Cell viability was also significantly increased in cells treated with curcumin (p=0.04) compared with untreated cells. Cell viability levels were unchanged in cells treated with Aβ+curcumin and curcumin+Aβ relative to untreated cells.
Significantly increased cell viability levels were found in cells treated with curcumin+Aβ (p=0.01) relative to Aβ incubated cells, suggesting that curcumin increases cell viability in the presence of Aβ.
Discussion
We investigated the protective effects of a natural product—curcumin—in healthy cells, and toxicity caused by Aβ. We studied preventive (curcumin+Aβ) and intervention (Aβ+curcumin) effects of curcumin against Aβ in cells. We measured mRNA and protein levels of mitochondrial dynamics, mitochondrial biogenesis, synaptic genes using real time RT-PCR, immunoblotting and immunofluorescence analysis. We also assessed mitochondrial function by measuring H2O2, lipid peroxidation, cytochrome oxidase activity and mitochondrial ATP. Further, we studied cell viability using the MTT assay. Aβ was found to impair mitochondrial dynamics (increased fission and decreased fusion), reduce biogenesis and decrease synaptic activity and mitochondrial function in healthy human neuroblastoma cells. On the other hand, curcumin enhanced fusion activity and reduced fission machinery and increased biogenesis and synaptic proteins. Mitochondrial function and cell viability were elevated in curcumin treated cells. Interestingly, curcumin pre- and post-treated cells incubated with Aβ showed reduced mitochondrial dysfunction, maintained cell viability and mitochondrial dynamics, mitochondrial biogenesis and synaptic activity. Further, the protective effects of curcumin were stronger in pretreated cells than in post-treated cells—in other words, curcumin works better in prevention than treatment in AD-like neurons.
mRNA and protein changes
Abnormal mitochondrial dynamics (increased fission and decreased fusion) was found in cells incubated with Aβ. Our findings concur with earlier studies.33–35 Increased expression of CypD was also found in cells incubated with Aβ, indicating that an increase in CypD damages mitochondria. Mitochondrial biogenesis genes were reduced in Aβ treated cells, indicating that biogenesis was affected by Aβ. Synaptic activity was reduced, marked by significantly decreased synaptic genes—synaptophysin and PSD95. Protein (immunoblotting and immunofluorescence) data confirmed mRNA changes, suggesting that Aβ affects both mRNA and proteins of mitochondria and synapses in AD neurons. In cells treated with curcumin, mitochondrial biogenesis and synaptic activity were enhanced, mitochondrial fission activity was reduced and fusion activity was enhanced. Our mRNA and protein data under preventive conditions of curcumin were positive, suggesting that curcumin prevents mitochondrial structural, biogenesis and synaptic genes from expressing abnormally. Overall, curcumin appears to protect the mitochondrial structure by regulating mitochondrial fission, fusion and matrix genes.
Mitochondrial function and cell viability
In Aβ treated cells, mitochondrial function was found to be defective. Further, cell viability was reduced in Aβ treated cells. Our observations agree with others on Aβ induced defective mitochondrial function and cell viability.33 36 37 Interestingly, curcumin treated cells showed enhanced mitochondrial function and increased cell viability (figure 4), implying that curcumin treated cells exhibited increased mitochondrial ATP, cytochrome oxidase activity and cell viability, and reduced free radicals and oxidative stress. These observations strongly suggest that curcumin reduces cellular toxicity and boosts mitochondrial function and promotes cell longevity.
Our major objective was to study the preventive and intervention effects of curcumin in AD neurons and we assessed these effects on mitochondrial function and cell viability using cells incubated with Aβ. Aβ induced defective mitochondrial function and cell viability were reversed in curcumin treated cells. The reversal effect was stronger in curcumin pretreated cells, indicating that curcumin is a strong preventive drug for AD. These findings are consistent with gene expression and protein data. In the presence of Aβ, curcumin reduced free radicals and lipid peroxidation, and increased mitochondrial ATP, cytochrome oxidase activity and cell viability. Thus all of our data point to curcumin protecting cells from Aβ toxicity.
Our observations need further support from AD mice studies—curcumin treatment in AD mice from early on (before mice develop toxic peptides and cognitive deficits). As mentioned in the introduction, several AD mice studies showed the beneficial effects of curcumin against Aβ induced toxicities but none has studied mitochondrial function or synaptic activity.
In summary, natural products such as curcumin have protective effects. Further, curcumin protects against Aβ induced mitochondrial and synaptic toxicities in AD neurons. Although research on curcumin has been done for several decades, more studies are needed in AD, mainly to determine the preventive effects against Aβ induced neuronal toxicities. It is also important to initiate clinical trials in early AD patients, subjects with mild cognitive impairment and perspective studies in elderly individuals.
Footnotes
Contributors: PHR designed and implemented the study, analyzed and interpreted the data, and wrote the manuscript and approved the study. MM, XY, MCG and AM performed the experiments and analyzed the data. RK and CSK assisted with manuscript preparation.
Funding: Work presented in this article is supported by NIH grants AG042178 and AG047812, and the Garrison Family Foundation.
Competing interests: None declared.
Provenance and peer review: Commissioned; externally peer reviewed.
References
- 1.Selkoe DJ. Alzheimer's disease: genes, proteins, and therapy. Physiol Rev 2001;81:741–66. [DOI] [PubMed] [Google Scholar]
- 2.Mattson MP. Pathways towards and away from Alzheimer's disease. Nature 2004;430:631–9. 10.1038/nature02621 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.LaFerla FM, Green KN, Oddo S. Intracellular amyloid-beta in Alzheimer's disease. Nat Rev Neurosci 2007;8:499–509. 10.1038/nrn2168 [DOI] [PubMed] [Google Scholar]
- 4.Prince M, Wimo A, Guerchet M, et al. . World Alzheimer Report 2015. The global impact of dementia. An analysis of prevalence, incidence, cost and trends. London: Alzheimer's Disease International (ADI), 2015 10.1016/j.bbadis.2011.08.005 [DOI] [Google Scholar]
- 5.Mao P, Reddy PH. Aging and amyloid beta-induced oxidative DNA damage and mitochondrial dysfunction in Alzheimer's disease: implications for early intervention and therapeutics. Biochim Biophys Acta 2011;1812:1359–70. 10.1016/j.bbadis.2011.08.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.van de Rest O, Berendsen AA, Haveman-Nies A, et al. . Dietary patterns, cognitive decline, and dementia: a systematic review. Adv Nutr 2015;6:154–68. 10.3945/an.114.007617 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mohd Sairazi NS, Sirajudeen KN, Asari MA, et al. . Kainic acid-induced excitotoxicity experimental model: protective merits of natural products and plant extracts. Evid Based Complement Alternat Med 2015;2015:972623 10.1155/2015/972623 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Venkatesan R, Ji E, Kim SY. Phytochemicals that regulate neurodegenerative disease by targeting neurotrophins: a comprehensive review. Biomed Res Int 2015;2015:814068 10.1155/2015/814068 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Solanki I, Parihar P, Mansuri ML, et al. . Flavonoid-based therapies in the early management of neurodegenerative diseases. Adv Nutr 2015;6:64–72. 10.3945/an.114.007500 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ammon HP, Wahl MA. Pharmacology of Curcuma longa. Planta Med 1991;57:1–7. 10.1055/s-2006-960004 [DOI] [PubMed] [Google Scholar]
- 11.Maheshwari RK, Singh AK, Gaddipati J, et al. . Multiple biological activities of curcumin: a short review . Life Sci 2006;78:2081–7. 10.1016/j.lfs.2005.12.007 [DOI] [PubMed] [Google Scholar]
- 12.Vogel H, Pelletier J. Curcumin-biological and medicinal properties. J Pharm 1815;I:289. [Google Scholar]
- 13.Lee WH, Loo CY, Bebawy M, et al. . Curcumin and its derivatives: their application in neuropharmacology and neuroscience in the 21st century. Curr Neuropharmacol 2013;11:338–78. 10.2174/1570159X11311040002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ramsewak RS, DeWitt DL, Nair MG. Cytotoxicity, antioxidant and anti-inflammatory activities of curcumins I-III from Curcuma longa. Phytomedicine 2000;7:303–8. 10.1016/S0944-7113(00)80048-3 [DOI] [PubMed] [Google Scholar]
- 15.Ringman JM, Frautschy SA, Cole GM, et al. . A potential role of the curry spice curcumin in Alzheimer's disease. Curr Alzheimer Res 2005;2:131–6. 10.2174/1567205053585882 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Baghdasaryan A, Claudel T, Kosters A, et al. . Curcumin improves sclerosing cholangitis in Mdr2-/- mice by inhibition of cholangiocyte inflammatory response and portal myofibroblast proliferation. Gut 2010;59:521–30. 10.1136/gut.2009.186528 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Moon DO, Kim MO, Choi YH, et al. . Curcumin attenuates inflammatory response in IL-1beta-induced human synovial fibroblasts and collagen-induced arthritis in mouse model. Int Immunopharmacol 2010;10:605–10. 10.1016/j.intimp.2010.02.011 [DOI] [PubMed] [Google Scholar]
- 18.Sharma OP. Antioxidant activity of curcumin and related compounds. Biochem Pharmacol 1976;25:1811–12. 10.1016/0006-2952(76)90421-4 [DOI] [PubMed] [Google Scholar]
- 19.Gilgun-Sherki Y, Melamed E, Offen D. Antioxidant treatment in Alzheimer's disease: current state. J Mol Neurosci 2003;21:1–11. 10.1385/JMN:21:1:1 [DOI] [PubMed] [Google Scholar]
- 20.Akbik D, Ghadiri M, Chrzanowski W, et al. . Curcumin as a wound healing agent. Life Sci 2014;116:1–7. 10.1016/j.lfs.2014.08.016 [DOI] [PubMed] [Google Scholar]
- 21.Shukla PK, Khanna VK, Khan MY, et al. . Protective effect of curcumin against lead neurotoxicity in rat. Hum Exp Toxicol 2003;22:653–8. 10.1191/0960327103ht411oa [DOI] [PubMed] [Google Scholar]
- 22.Yang F, Lim GP, Begum AN, et al. . Curcumin inhibits formation of amyloid beta oligomers and fibrils, binds plaques, and reduces amyloid in vivo. J Biol Chem 2005;280:5892–901. 10.1074/jbc.M404751200 [DOI] [PubMed] [Google Scholar]
- 23.Garcia-Alloza M, Borrelli LA, Rozkalne A, et al. . Curcumin labels amyloid pathology in vivo, disrupts existing plaques, and partially restores distorted neurites in an Alzheimer mouse model. J Neurochem 2007;102:1095–104. 10.1111/j.1471-4159.2007.04613.x [DOI] [PubMed] [Google Scholar]
- 24.Kim DS, Park SY. Discovery of natural products from curcuma longa that protect cells from beta-amyloid insult: a drug discovery effort against Alzheimer's disease. J Nat Prod 2002;65:1227–31. [DOI] [PubMed] [Google Scholar]
- 25.Ono K, Hasegawa K, Naiki H, et al. . Curcumin has potent anti-amyloidogenic effects for Alzheimer's beta-amyloid fibrils in vitro. J Neurosci Res 2004;75:742–50. 10.1002/jnr.20025 [DOI] [PubMed] [Google Scholar]
- 26.Narlawar R, Pickhardt M, Leuchtenberger S, et al. . Curcumin-derived pyrazoles and isoxazoles: Swiss army knives or blunt tools for Alzheimer's disease? Chem Med Chem 2008;3:165–72. 10.1002/cmdc.200700218 [DOI] [PubMed] [Google Scholar]
- 27.Kim DS, Park SY, Kim JK. Curcuminoids from Curcuma longa L. (Zingiberaceae) that protect PC12 rat pheochromocytoma and normal human umbilical vein endothelial cells from betaA(1-42) insult. Neurosci Lett 2001;303:57–61. [DOI] [PubMed] [Google Scholar]
- 28.Lim GP, Chu T, Yang F, et al. . The curry spice curcumin reduces oxidative damage and amyloid pathology in an Alzheimer transgenic mouse. J Neurosci 2001;21:8370–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Fiala M, Liu PT, Espinosa-Jeffrey A, et al. . Innate immunity and transcription of MGAT-III and Toll-like receptors in Alzheimer's disease patients are improved by bisdemethoxycurcumin. Proc Natl Acad Sci USA 2007;104:12849–54. 10.1073/pnas.0701267104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Atamna H, Boyle K. Amyloid-beta peptide binds with heme to form a peroxidase: relationship to the cytopathologies of Alzheimer's disease. Proc Natl Acad Sci USA 2006;103:3381–6. 10.1073/pnas.0600134103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Baum L, Ng A. Curcumin interaction with copper and iron suggests one possible mechanism of action in Alzheimer's disease animal models. J Alzheimers Dis 2004;6:367–77; discussion 443–9. [DOI] [PubMed] [Google Scholar]
- 32.Manczak M, Calkins MJ, Reddy PH. Impaired mitochondrial dynamics and abnormal interaction of amyloid beta with mitochondrial protein Drp1 in neurons from patients with Alzheimer's disease: implications for neuronal damage. Hum Mol Genet 2011;20:2495–509. 10.1093/hmg/ddr139 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Manczak M, Mao P, Calkins MJ, et al. . Mitochondria-targeted antioxidants protect against amyloid-beta toxicity in Alzheimer's disease neurons. J Alzheimers Dis 2010;20(Suppl 2):S60631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wang X, Su B, Siedlak SL, et al. . Amyloid-beta overproduction causes abnormal mitochondrial dynamics via differential modulation of mitochondrial fission/fusion proteins. Proc Natl Acad Sci USA 2008;105:19318–23. 10.1073/pnas.0804871105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Silva DF, Selfridge JE, Lu J, et al. . Bioenergetic flux, mitochondrial mass and mitochondrial morphology dynamics in AD and MCI cybrid cell lines. Hum Mol Genet 2013;22:3931–46. 10.1093/hmg/ddt247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Casley CS, Canevari L, Land JM, et al. . Beta-amyloid inhibits integrated mitochondrial respiration and key enzyme activities. J Neurochem 2002;80:91–100. 10.1046/j.0022-3042.2001.00681.x [DOI] [PubMed] [Google Scholar]
- 37.Aleardi AM, Benard G, Augereau O, et al. . Gradual alteration of mitochondrial structure and function by beta-amyloids: importance of membrane viscosity changes, energy deprivation, reactive oxygen species production, and cytochrome c release. J Bioenerg Biomembr 2005;37:207–25. 10.1007/s10863-005-6631-3 [DOI] [PubMed] [Google Scholar]