Abstract
Adoptive cell therapy is a potentially curative therapeutic approach for patients with cancer. In this treatment modality, antitumor T cells are exponentially expanded in vitro prior to infusion. Importantly, the results of recent clinical trials suggest that the quality of expanded T cells critically affects their therapeutic efficacy. Although anti-CD3 mAb-based stimulation is widely used to expand T cells in vitro, a protocol to generate T cell grafts for optimal adoptive therapy has yet to be established. In this study, we investigated the differences between T cell stimulation mediated by anti–CD3/CD28 mAb–coated beads and cell-based artificial antigen-presenting cells (aAPCs) expressing CD3/CD28 counter-receptors. We found that transient stimulation with cell-based aAPCs, but not prolonged stimulation with beads, resulted in the superior expansion of CD8+ T cells. Transiently stimulated CD8+ T cells maintained a stem cell–like memory phenotype and were capable of secreting multiple cytokines significantly more efficiently than chronically stimulated T cells. Importantly, the chimeric antigen receptor–engineered antitumor CD8+ T cells expanded via transient stimulation demonstrated superior persistence and antitumor responses in adoptive immunotherapy mouse models. These results suggest that restrained stimulation is critical for generating T cell grafts for optimal adoptive immunotherapy for cancer.
Transient stimulation with cell-based artificial antigen presenting cells produces superior antitumor T cells that are less exhausted and enriched in a stem cell-like memory T cell subset.
Introduction
Cancer immunotherapy, which exploits the patient’s own immune system to attack tumor cells, is a rapidly emerging treatment modality. Among different therapeutic strategies, adoptive cell therapy is one of the promising options (1, 2). In this therapy, tumor-reactive T cells are expanded from a patient’s tumor tissue or peripheral blood or generated via genetic engineering of T cells in vitro, and the cells are then infused back into the patient. Recent clinical trials have demonstrated the efficacy of this therapy in some types of malignancies that are refractory to conventional treatment (3–9). There is a growing consensus that the persistence of the transferred T cells is critically important for inducing durable clinical responses (5, 10–12). When cultivated in vitro, naive T cells gradually acquire the surface marker phenotypes of memory T cells following T cell receptor (TCR) stimulation. They are classified into stem cell–like memory (TSCM), central memory (TCM), and effector memory (TEM) T cells (13). Among these populations, TSCM cells possess superior persistence and antitumor effects in multiple cancer immunotherapy models (12–18). Since cultured memory T cells irreversibly differentiate in the order of TSCM, TCM, and TEM and lose their long-term survival potential, in vitro expansion of antitumor T cells needs to be optimized to maintain a TSCM phenotype while accomplishing efficient expansion.
Optimal T cell activation and proliferation requires multiple signals involving TCR stimulation (signal 1), a costimulatory signal (signal 2), and cytokine engagement (signal 3) (19). For in vitro polyclonal T cell stimulation, anti-CD3 antibodies are widely used for conferring signal 1; these antibodies can be immobilized on the surface of wells or cross-linked with Fc receptor–bearing accessory cells such as monocytes and B cells. Magnetic beads conjugated with anti-CD3 and anti-CD28 antibodies provide an alternative approach for T cell stimulation (20, 21). Although anti–CD3/CD28 beads have been used in clinical trials, this methodology preferentially expands CD4+ T cells and is not optimal for expanding CD8+ T cells (22, 23). We and others previously developed K562-based artificial antigen-presenting cells (aAPCs) for the in vitro expansion of T cells in both an antigen-specific and nonspecific manner (22, 24–28). In contrast to inert particles, stimulation with cell-based aAPCs expressing a membranous form of anti-CD3 antibody and the costimulatory molecules CD80 and CD83 (aAPC/mOKT3) resulted in the preferential expansion of polyclonal CD8+ T cells (28). However, the precise mechanism underlying the difference and the effects of aAPCs on the quality of the expanded T cells for adoptive transfer have yet to be determined.
In this study, we performed comparative analysis of polyclonal T cells stimulated with anti–CD3/CD28 beads versus cell-based aAPCs and found that prolonged stimulatory signals from beads constrict their proliferative potential. In contrast, transient bead stimulation achieved superior expansion of CD8+ T cells. Moreover, the expanded CD8+ T cells receiving transient stimulation better maintained a TSCM phenotype and possessed superior in vivo persistence and antitumor effects compared with the chronically stimulated T cells. These findings provide important insights into the strategy to generate T cell grafts in vitro for the purpose of adoptive immunotherapy.
Results
Transient stimulation with cell-based aAPCs induces superior expansion of CD8+ T cells.
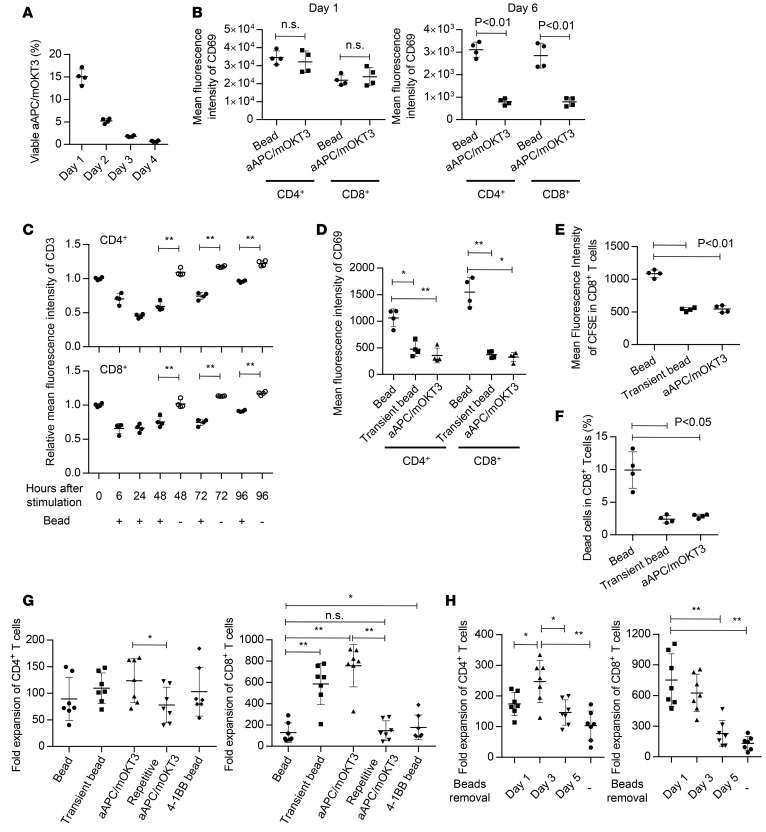
First, we analyzed the proliferation of T cells and their functional properties upon stimulation with anti–CD3/CD28 beads or cell-based aAPCs. Since the purpose of this study was to explore an optimal method to expand T cells for adoptive cancer immunotherapy, T cells were transduced with the anti-CD19 chimeric antigen receptor (CAR) gene during expansion throughout the experiments as a model of the protocol to generate antitumor T cell grafts (Figure 1A) (29). We confirmed that stimulation of polyclonal CD3+ T cells with aAPC/mOKT3 preferentially expands CD8+ T cells in the presence of IL-2 and IL-15, while anti–CD3/CD28 bead stimulation results in less proliferation of CD8+ T cells (Figure 1, B and C) (28). T cell stimulation with K562 cells expressing mOKT3 and CD80, but not CD83, similarly induced the preferential expansion of CD8+ T cells. There was not a significant difference between the 3 treatments in CD4+ T cell expansion. We also evaluated T cell expansion in the presence of IL-7 and IL-15, which was recently reported to promote more efficient proliferation of CD8+ T cells than IL-2 (14). While IL-7 and IL-15 supplementation improved the expansion of bead-stimulated T cells, aAPC/mOKT3–stimulated CD8+ T cells still showed an explicit proliferative advantage (Supplemental Figure 1; supplemental material available online with this article; doi:10.1172/jci.insight.89580DS1). When memory T cell phenotypes were analyzed, CD8+ T cells stimulated with the cell-based aAPCs maintained a CD45RA+CD62L+CCR7+ phenotype significantly better than those stimulated with the beads (Figure 1, D–F). These CD8+ T cells were also positive for CD27, CD28, and CD95 (Supplemental Figure 2), indicating that they had a recently described TSCM phenotype (13). Furthermore, the CD8+ T cells expanded with the cell-based aAPCs were more potent in secreting multiple cytokines including IL-2, IFN-γ, and TNF-α than the bead-stimulated T cells (Figure 1, G and H). These results indicate that cell-based aAPC treatment is more efficient than antibody-coated inert particles in generating CD8+ T cells with a TSCM phenotype that can secrete multiple cytokines. In contrast, the frequency of TSCM cells within CD4+ T cells was comparable between bead- and cell-based aAPC-stimulated cells (Supplemental Figure 3A). Secretion of IFN-γ, but not IL-2 or TNF-α, was significantly more robust in cell-based aAPC-stimulated CD4+ T cells than in bead-stimulated cells (Supplemental Figure 3B).
Figure 1. Stimulation with cell-based artificial antigen-presenting cells (aAPCs) provides superior expansion of CD8+ T cells with stem cell–like memory T cell features.
(A–C) Peripheral blood CD3+ T cells were stimulated with anti–CD3/CD28 beads, K562-based aAPCs that express a membrane-bound form of anti-CD3 mAb (clone OKT3) and the costimulatory molecules CD80 and CD83 (aAPC/mOKT3), or K562 cells with mOKT3 and CD80 (K562-mOKT3/CD80). Then, the cells were retrovirally transduced with the anti-CD19 chimeric antigen receptor (CAR) consisting of anti-CD19 scFv, CD28, and CD3z. The T cells were further cultured with 100 IU/ml IL-2 and 10 ng/ml IL-15 (A). Representative FACS plots showing the frequencies of CD4+ and CD8+ T cells (B) and the fold expansion of each T cell subset (C) 14 days following stimulation are shown (n = 7, paired ANOVA). (D–F) Surface expression of CD45RA, CD62L, and CCR7 in T cells expanded by the indicated stimulation method. Representative FACS plots (D) and the frequency (E) and fold expansion (F) of CD45RA+CD62L+CCR7+ cells within the CD8+ T cell population on day 14 are shown (n = 7, paired ANOVA). (G and H) Secretion of IL-2, IFN-γ, and TNF-α upon restimulation with aAPC/mOKT3 was evaluated by intracellular flow cytometry in CD8+ T cells cultured for 14 days. The frequency of individual cytokine-producing cells (G) and those secreting all 3 cytokines (H) is shown (n = 6, paired ANOVA).
Transient stimulation provides superior proliferative signals for CD8+ T cells.
While antibody-conjugated beads or inert particles can persist after encounters with T cells, cell-based aAPCs are irradiated and readily lysed upon ligation with T cells. In fact, the number of aAPC/mOKT3 cocultured with T cells progressively declined and reached levels as low as approximately 0.5% within 96 hours (Figure 2A). Although stimulation of T cells with anti–CD3/CD28 beads and aAPC/mOKT3 similarly upregulated the expression of the activation marker CD69 24 hours later, the expression declined more quickly in the aAPC/mOKT3–stimulated T cells, indicating that they were stimulated for a shorter time period than those stimulated with the beads (Figure 2B and Supplemental Figure 4A). We postulated that transient T cell stimulation provided signals optimal for in vitro CD8+ T cell proliferation, while continuous stimulation led to overstimulation, thereby depriving the T cells of proliferative potential in vitro. To test this, we stimulated CD3+ T cells with anti–CD3/CD28 beads and removed them the next day to recapitulate the transient stimulation provided by aAPC/mOKT3. T cell stimulation with anti–CD3/CD28 beads downregulated the surface expression of CD3, which quickly recovered after removal of the beads (Figure 2C and Supplemental Figure 4B). The T cells that were chronically stimulated by anti–CD3/CD28 beads still maintained the surface expression of CD3 to some extent, which appeared to make them undergo prolonged stimulatory signaling (Figure 2C). Consistent with this, the T cells with beads removed displayed decreased CD69 expression 6 days following stimulation, as was observed with stimulation with aAPC/mOKT3 (Figure 2D and Supplemental Figure 4C). The transiently stimulated T cells demonstrated more cellular division and better viability than the chronically stimulated ones, indicating that the superior expansion mediated by transient stimulation is probably due to both increased proliferation and reduced cell death (Figure 2, E and F, and Supplemental Figure 4D). The transient stimulation with anti–CD3/CD28 beads achieved CD8+ T cell expansion comparable to aAPC/mOKT3–mediated stimulation (Figure 2G). Conversely, CD8+ T cell proliferation was significantly impaired when these cells were repeatedly stimulated by aAPC/mOKT3. We also evaluated the T cell proliferation upon stimulation with anti–CD3/CD28/4-1BB beads. Although the addition of 4-1BB costimulation slightly improved the T cell expansion, transient stimulation resulted in better proliferation of CD8+ T cells. We further evaluated T cell proliferation upon variable durations of stimulation by removing the beads on day 1, 3, or 5 and found that longer periods of stimulation negatively affected CD8+ T cell proliferation (Figure 2H). Interestingly, CD4+ T cells showed the most robust expansion with a 3-day stimulation protocol, suggesting that CD4+ and CD8+ T cells require different stimulation durations for optimal proliferation.
Figure 2. Transient stimulation provides superior proliferative signals for CD8+ T cells.
(A) Frequency of live artificial antigen-presenting cells that express a membrane-bound form of anti-CD3 mAb (clone OKT3) and the costimulatory molecules CD80 and CD83 (aAPC/mOKT3) at 24, 48, 72, and 96 hours after coculture with T cells at an effector/target ratio of 5:1 (n = 4). (B) Mean fluorescence intensity (MFI) of CD69 in CD4+ and CD8+ T cells at the indicated time points following stimulation with anti–CD3/CD28 beads or aAPC/mOKT3 (n = 4, paired t test). (C) CD3+ T cells were stimulated with anti–CD3/CD28 beads. The beads were removed or kept in culture the following day. The MFI of CD3 normalized to the day-0 value at the indicated time points is shown (n = 4, paired t test). (D–F) CD3+ T cells were stimulated with anti–CD3/CD28 beads or aAPC/mOKT3. The beads were removed on day 1 or kept in the culture media. The MFI of CD69 on day 6 (D) and carboxyfluorescein succinimidyl ester (CFSE) on day 5 (E), and frequency of dead cells on day 5 (F) in CD8+ T cells were analyzed by flow cytometry (n = 4, paired ANOVA). (G) CD3+ T cells were stimulated as indicated in the graph. Fold expansion of CD4+ and CD8+ T cells was calculated 14 days following stimulation (n = 7, paired ANOVA). (H) CD3+ T cells were stimulated with anti–CD3/CD28 beads. The beads were removed on day 1, 3, or 5 or kept in culture, and the proliferation of CD4+ and CD8+ T cells was analyzed 14 days following stimulation (n = 7, paired ANOVA). *P < 0.05, **P < 0.01.
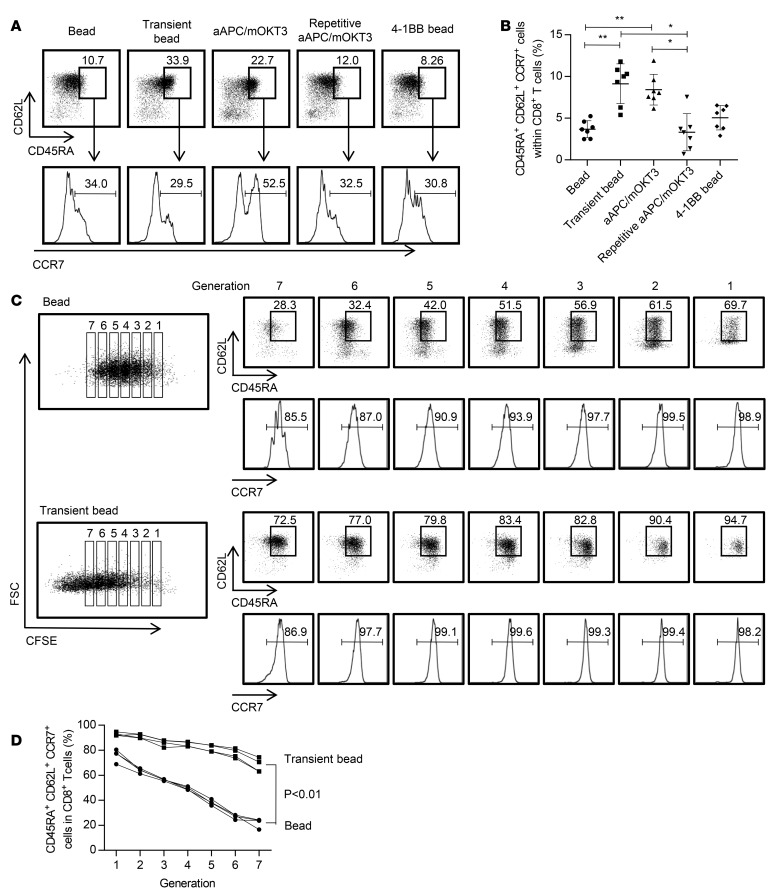
We then compared the properties of CD8+ T cells with chronic or transient stimulation. In addition to the proliferative advantage, T cells with transient bead stimulation possessed a higher frequency of TSCM cells within CD8+ T cells than did the chronically stimulated T cells (Figure 3, A and B). To investigate the effect of the duration of stimulation on T cell differentiation in more detail, CD45RA+CD62L+CCR7+ cells were isolated, labeled with carboxyfluorescein succinimidyl ester (CFSE), and stimulated with beads in a chronic or transient manner. The T cell differentiation profiles were analyzed at each CFSE dilution peak (Figure 3C). Notably, transiently stimulated T cells maintained the CD45RA+CD62L+CCR7+ phenotype significantly better than the chronically stimulated cells, indicating the superior self-renewal potential of T cells that underwent transient stimulation (Figure 3D). In addition to marker phenotypes, transiently stimulated CD8+ T cells secreted more IL-2, IFN-γ, and TNF-α and were superior in the ability to produce multiple cytokines upon restimulation with aAPC/mOKT3 (Figure 4, A and B). Likewise, transiently stimulated CAR-T cells secreted more cytokines than the continuously stimulated cells when stimulated with K562 cells expressing CD19 (Figure 4, C and D). These results demonstrated that lingering stimulation of antitumor CD8+ T cells in vitro impairs their functions in terms of proliferative capacity and cytokine secretion. The addition of 4-1BB costimulation significantly improved cytokine secretion, which is consistent with recent studies showing that the 4-1BB signal ameliorates the functional impairment of T cells (30).
Figure 3. Persistent stimulation induces T cell differentiation into a CD45RA– phenotype in CD8+ T cells.
(A and B) CD3+ T cells were stimulated as indicated, and surface expression profiles of CD45RA, CD62L, and CCR7 within the CD8+ T cell population were analyzed on day 14. Representative FACS plots (A) and the frequency (B) of CD45RA+CD62L+CCR7+ cells within the CD8+ T cell population are shown. *P < 0.05, **P < 0.01 (n = 7, paired ANOVA). (C and D) CD3+CD45RA+CD62L+CCR7+ T cells were labeled with carboxyfluorescein succinimidyl ester (CFSE) and chronically or transiently stimulated with anti–CD3/CD28 beads. T cell differentiation was compared at each CFSE dilution peak on day 7. Representative FACS plots (C) and the frequency of CD45RA+CD62L+CCR7+ cells in CD8+ T cells are shown (n = 4, paired t test).
Figure 4. Persistent stimulation impairs cytokine secretion by CD8+ T cells.
(A–D) CD3+ T cells were stimulated as indicated in the graph and transduced with the anti-CD19 chimeric antigen receptor (CAR) gene. After 14 days of expansion, the CAR-T cells were restimulated with artificial antigen-presenting cells that express a membrane-bound form of anti-CD3 mAb (clone OKT3) and the costimulatory molecules CD80 and CD83 (aAPC/mOKT3) (A and B) or K562 cells expressing CD19 (K562-CD19) (C and D), and secretion of IL-2, IFN-γ, and TNF-α by the CD8+ T cell population was analyzed by flow cytometry. The frequency of each type of cytokine-secreting cell (A and C) and those secreting all 3 cytokines (B and D) is shown (n = 5 or 6, paired ANOVA). *P < 0.05, **P < 0.01.
IL-2 receptor expression is induced by transient stimulation and maintained at sufficient levels by IL-2 exposure.
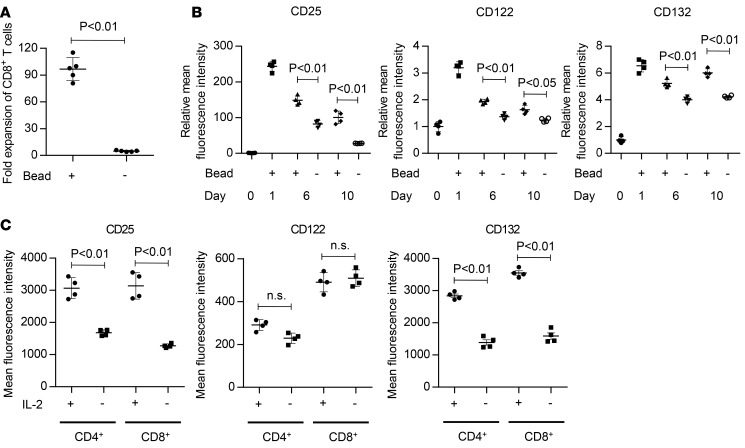
We then investigated how transiently stimulated T cells are able to maintain proliferation after bead removal. As optimal in vitro T cell expansion requires TCR stimulation and costimulation prior to cytokine supplementation, cytokine treatment alone cannot induce sufficient T cell proliferation (Figure 5A). T cell stimulation upregulates the expression of cytokine receptors, which is required for T cell proliferation. We investigated the expression kinetics of IL-2 receptors α, β, and γ following TCR/CD28 engagement. The expression of all the receptors in CD8+ T cells was rapidly upregulated upon bead stimulation and gradually downregulated along with continued culture (Figure 5B and Supplemental Figure 5). IL-2 receptor expression in bead-removed T cells was maintained at higher levels than those in the freshly isolated T cells for approximately 2 weeks. Similar results were obtained with CD4+ T cells (data not shown). Importantly, the removal of cytokines significantly downregulated the expression of IL-2 receptors α and γ, confirming the positive feedback mechanism between cytokines and their receptors (Figure 5C) (31). Collectively, transient stimulation and subsequent cytokine supplementation induce and maintain the expression of IL-2 receptors at levels sufficient for T cell proliferation.
Figure 5. IL-2 receptors are sufficiently upregulated by transient stimulation and maintained during the culture.
(A) CD3+ T cells were cultured in the presence of IL-2 and IL-15 with or without transient stimulation with anti–CD3/CD28 beads. Fold expansion of CD8+ T cells on day 7 is shown (n = 4, paired t test). (B) Surface expression of IL-2 receptors (CD25, CD122, and CD132) upon transient or prolonged stimulation with anti–CD3/CD28 beads was analyzed at the indicated time points. The mean fluorescence intensity normalized to the day-0 values is shown (n = 4 each, unpaired t test). (C) CD3+ T cells were transiently stimulated with anti–CD3/CD28 beads and cultured in the presence of IL-2. On day 7, the T cells were kept in culture or transferred to cytokine-free media, and the expression of IL-2 receptors was analyzed by flow cytometry on the next day (n = 4, paired t test).
Transiently stimulated CD8+ T cells exhibit better in vivo persistence and antitumor effects.
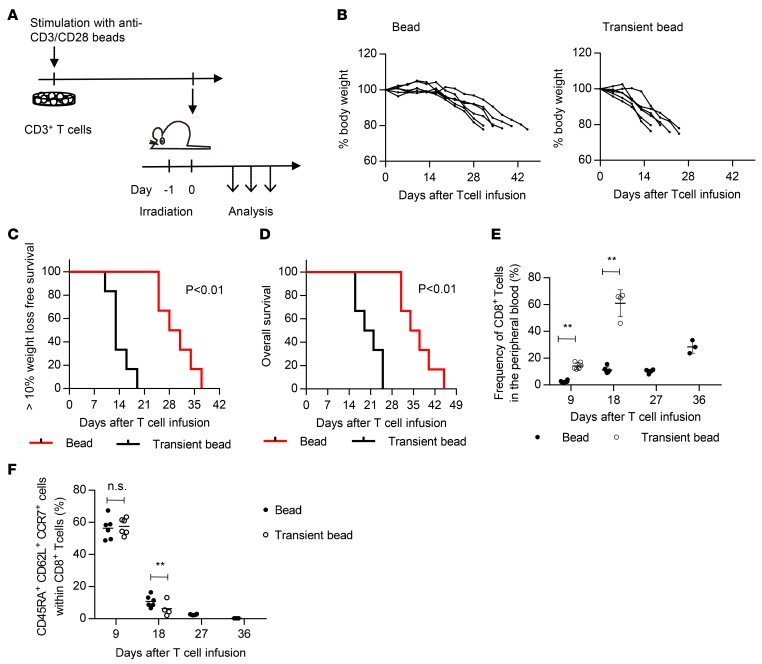
We then compared the in vivo persistence capacity of CD8+ T cells expanded with transient or prolonged stimulation. CD3+ T cells were stimulated with anti–CD3/CD28 beads in either a transient or prolonged manner, cultured in the presence of IL-2 and IL-15, and transplanted into irradiated NOD/SCID-IL2Rγnull (NSG) mice (Figure 6A). To make a fair comparison of their persistence capacity, T cells were expanded until a CD8+ T cell subset accomplished 100-fold expansion. Since CD8+ T cells with chronic bead stimulation proliferated more slowly, it took more days until they reached 100-fold expansion (Supplemental Figure 6A). As reported previously, the T cell–infused mice progressively lost weight and succumbed to lethality due to xenogeneic graft-versus-host disease (GVHD) (Figure 6, B–E) (13, 32). Importantly, transiently stimulated CD8+ T cells were observed at a significantly higher frequency in the peripheral blood at each time point and caused a more rapid development of GVHD than the chronically stimulated T cells. The transplanted T cells underwent proliferation and differentiation from a TSCM to a CD45RA– population when monitored in the peripheral blood (Figure 6F and Supplemental Figure 6B). These results indicate that the CD8+ T cells expanded with transient stimulation were superior in persistence and proliferative capacity when adoptively transferred in vivo.
Figure 6. T cells expanded via transient stimulation have superior persistence in vivo.
(A) CD3+ T cells were stimulated with anti–CD3/CD28 beads for 24 hours or persistently and cultured until CD8+ T cells underwent a 100-fold expansion. The T cells were then infused into irradiated NOD/SCID-IL2Rγnull (NSG) mice, and the mice were sequentially monitored for body weight and T cell persistence. (B) The sequential data of the body weight of the transplanted mice are shown (n = 6). (C and D) Kaplan-Meier analysis for more than 10% weight-loss-free survival (C) and overall survival (D) following T cell transplantation (n = 6, log-rank test). The data shown are representative of 2 experiments. (E) Human CD8+ T cell chimerism in the peripheral blood was analyzed at the indicated time points (unpaired t test). (F) The frequency of CD45RA+CD62L+CCR7+ cells within the CD8+ T cell population was analyzed at the indicated time points (unpaired t test). **P < 0.01.
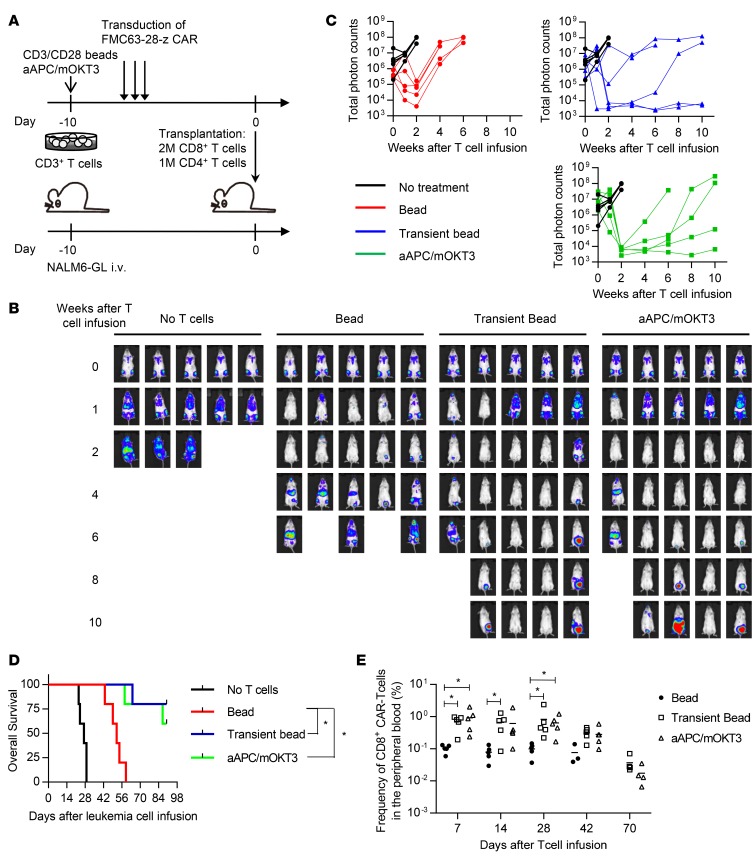
We next addressed the therapeutic effects of antitumor T cells expanded using the transient stimulation protocol. CD3+ T cells were stimulated transiently or chronically with anti–CD3/CD28 beads or aAPC/mOKT3 and were retrovirally transduced with the anti-CD19 CAR gene (Figure 7A). NSG mice were intravenously injected with the human CD19+ acute lymphoblastic leukemia cell line NALM-6 transduced with a firefly luciferase-EGFP fusion gene (NALM6-GL) 10 days prior to T cell infusion. Since different CD4+/CD8+ T cell ratios could influence antitumor activity independent of the quality of the CAR-T cells, we separated CD4+ and CD8+ T cells before transplantation and infused a defined number of each T cell subset (2 million CD8+ T cells and 1 million CD4+ T cells). The CAR-T cell grafts efficiently eradicated leukemia soon after T cell infusion regardless of the stimulation protocol. However, all of the mice treated with the chronically stimulated CAR-T cells relapsed thereafter (Figure 7, B and C). In contrast, transiently stimulated CAR-T cells controlled leukemia progression for a longer duration, which resulted in better overall survival (Figure 7D). Although the CAR-T cells progressively declined in frequency after transplantation, transiently stimulated CAR-T cells exhibited significantly better persistence in the peripheral blood at each time point, which appeared to correlate with the superior control of leukemia cells (Figure 7E and Supplemental Figure 7). Collectively, these findings demonstrate that controlled in vitro stimulation leads to the expansion of CAR-T cells equipped with superior persistence and antitumor activities.
Figure 7. T cells expanded with transient stimulation have better antitumor effects.
(A) CD3+ T cells were stimulated with anti–CD3/CD28 beads or artificial antigen-presenting cells that express a membrane-bound form of anti-CD3 mAb (clone OKT3) and the costimulatory molecules CD80 and CD83 (aAPC/mOKT3), and retrovirally transduced with the anti-CD19 chimeric antigen receptor (CAR). The CAR-T cells (1 million CD4+ T cells and 2 million CD8+ T cells) were transplanted into NOD/SCID-IL2Rγnull (NSG) mice that were intravenously infused with NALM-6 transduced with a firefly luciferase-EGFP fusion gene (NALM6-GL). (B and C) In vivo bioluminescence imaging of luciferase activity was analyzed following the infusion of T cells. Images of each mouse (B) and the total photon counts (C) at the indicated time points are shown. (D) Kaplan–Meier curve for the overall survival of the mice untreated or treated with CAR-transduced T cells (n = 5, log-rank test with multiple comparisons). Representative data from 2 experiments are shown. (E) Frequency of human CD45+CD8+ CAR-transduced T cells in the peripheral blood at the indicated time points (n = 5, nonparametric ANOVA with multiple comparison tests). *P < 0.05.
Discussion
In this study, we investigated qualitative differences in T cell stimulation conferred by inert particles and cell-based aAPCs and showed that a short period of stimulation with anti–CD3/CD28 antibodies provides superior proliferative signals for CD8+ T cells. CD8+ T cells expanded with transient stimulation were more enriched in the TSCM population and possessed superior cytokine-producing capacity upon restimulation. Importantly, transiently stimulated T cells exhibited long-lasting survival potential and antitumor immunity in vivo.
Ex vivo or in vitro expansion of antitumor T cells is an essential step to generate antitumor T cell grafts in adoptive cancer immunotherapy. Recent studies have revealed that a variety of cytokines and costimulatory signals can coordinately affect the quantity and quality of the expanded CD8+ T cells (14, 25, 27, 33–36). Although it has been reported that anti–CD3/CD28 beads can efficiently expand CD4+ T cells but not CD8+ T cells, the underlying mechanism has not been fully addressed. Our results indicate that CD8+ T cell expansion is markedly enhanced by shortening the duration of T cell stimulation. Furthermore, our data suggest that the duration of T cell stimulation for optimal T cell expansion was different between CD4+ and CD8+ T cells and that CD4+ T cells required more prolonged stimulation than CD8+ T cells. These observations are consistent with recent mouse studies; CD8+ T cells were able to proliferate and differentiate into effector T cells in response to transient antigen presentation, while CD4+ T cells required sustained antigen exposure for continued growth (37, 38).
In addition to the impaired proliferation, chronically stimulated CD8+ T cells were inferior in cytokine secretion compared with those that were transiently stimulated. The phenotypes of constitutively stimulated T cells were similar to those of exhausted T cells. T cell exhaustion is a state of T cell dysfunction observed in conditions such as chronic infection or the tumor microenvironment, where T cells are continuously exposed to antigens (39–41). Similar phenotypes can be induced in the in vitro setting by repeated stimulation, which attenuates the proliferative potential and cytokine-secreting capacity of cultured T cells (42–44). Recent studies have revealed that CD28-mediated costimulatory signaling is also associated with impairment of T cell functions. Several CAR constructs containing a CD28 intracellular signaling domain provoke antigen-independent T cell activation and reduce the antitumor efficacy of CAR-T cells by inducing exhaustive states prior to infusion (30). Kawalekar et al. reported that CD28 and 4-1BB signaling differentially affect T cell metabolism and proliferative potential during ex vivo expansion (45). While CD28-mediated costimulatory signals support T cell proliferation, they might accelerate the induction of exhaustive phenotypes in the context of persistent stimulation. Consistent with previous studies, 4-1BB costimulation enhanced the expansion of CD8+ T cells and ameliorated the functional impairment caused by chronic stimulation. Although T cell proliferation is generally coupled with differentiation, transient stimulation contributed to the superior maintenance of a TSCM phenotype and better proliferative potential. Consistent with their marker profiles, the transiently stimulated T cells exhibited improved persistence and antitumor activity when adoptively transferred in vivo, demonstrating that they possess superior functional properties compatible with their TSCM phenotype. These results indicate that the duration of stimulation can significantly affect the quality of antitumor T cells.
In summary, we demonstrated that short-term T cell stimulation significantly augments the functional properties and proliferative potential of cultured CD8+ T cells, which results in improved antitumor efficacy in vivo. These findings can be readily applied to expanding superior T cell grafts for various types of adoptive T cell therapy, such as tumor-infiltrating lymphocyte therapy, TCR gene therapy, and CAR therapy.
Methods
In vitro culture of human T cells.
Healthy donor–derived peripheral blood mononuclear cells were isolated by Ficoll-Paque PLUS density gradient centrifugation (GE Healthcare). CD3+ T cells were purified through negative magnetic selection using a Pan T Cell Isolation Kit (Miltenyi Biotec). To evaluate memory T cell differentiation, CD45RA+CD62L+CCR7+ cells were isolated with a FACS Aria flow cytometer (BD Biosciences). Purified T cells were stimulated with aAPC/mOKT3 irradiated with 200 Gy at an effector/target (E:T) ratio of 5:1, anti–CD3/CD28 Dynabeads or anti–CD3/CD28/CD137 Dynabeads (Thermo Fisher Scientific) at a bead/T cell ratio of 1:1 according to the manufacturer’s instructions. For repetitive stimulation by aAPC/mOKT3, the irradiated aAPC/mOKT3 corresponding to 85% of the cells cocultured on day 1 was added from day 2 through day 7. On day one, 100 IU/ml IL-2 and 10 ng/ml IL-15 (Peprotech) were added to the cultures. IL-7 (Peprotech) was used at 10 ng/ml. The culture media were replenished every 3 days. The CD4/CD8 ratio of each CD3+ T cell sample was determined by flow cytometry for calculating the fold expansion of CD4+ and CD8+ T cells.
Cell lines.
The aAPC/mOKT3 cells were derived from the human erythroleukemic cell line K562 (American Type Culture Collection) and established as previously described (28). The human CD19+ NALM-6 leukemia cell line was obtained from DSMZ and transduced with pMX-EGFP-firefly luciferase (NALM6-GL) for in vivo imaging studies. All the cells were routinely checked for the presence of mycoplasma contamination using PCR-based technology.
Flow cytometry.
The following antibodies were used for flow cytometry: APC-Cy7-anti-CD4 (clone RPA-T4; BioLegend), PE-Cy7-anti-CD8 (clone SFCI21Thy2D3; Beckman Coulter), Pacific Blue-anti-CD8 (clone B9.11; Beckman Coulter), Pacific Blue-anti-CD3 (clone UCHT1; BioLegend), FITC-anti-CD45RA (clone MEM-56; Thermo Fisher Scientific), APC-Cy7-anti-CD45RA (clone HI100, BioLegend), PE-anti-CD62L (clone DREG-56; BioLegend), Pacific Blue-anti-CCR7 (clone G043H7; BioLegend), APC-Cy7-anti-CD27 (clone O323; BioLegend), APC-anti-CD28 (clone CD28.2; BioLegend), PerCP/Cy5.5-anti-CD95 (clone DX2, BD Biosciences), APC-anti-CD45 (clone HI30; BioLegend), FITC-anti-IL-2 (clone 5344.111; BD Biosciences), PE-anti-TNF-α (clone MAb11; BioLegend), PE-Cy7-anti-IFN-γ (clone 4S.B3; BioLegend), PE-anti-CD69 (clone FN50; BioLegend), PE-Cy7-anti-CD25 (clone B1.49.9; Beckman Coulter), PE-anti-CD122 (clone TU27; BioLegend), and APC-anti-CD132 (clone TUGh4; BioLegend). The stained cells were analyzed with a FACS Canto II (BD Biosciences). The data analysis was performed with FlowJo software (Tree Star). For intracellular flow cytometry analysis of cytokine production, T cells were stimulated with aAPC/mOKT3 or K562-CD19 at an E:T ratio of 1:1 and incubated for 6 hours. Brefeldin A (BioLegend) was added to the cultures 2 hours following the stimulation. The cells were then fixed and permeabilized using a Cytofix/Cytoperm kit (BD Biosciences). For CFSE dilution assays, T cells were labeled with 5 μM CFSE (Thermo Fisher Scientific) before culture. Dead cells were discriminated with the LIVE/DEAD Fixable Dead Cell Stain (Thermo Fisher Scientific). The CAR-transduced T cells were stained by biotin-labeled protein L (Genscript) followed by streptavidin-PE (Thermo Fisher Scientific).
Mouse experiments.
In murine experiments, 6- to 10-week-old male NSG mice (The Jackson Laboratory) were used. In the experiments for evaluating the in vivo persistence of T cells, CD3+ T cells were stimulated with anti–CD3/CD28 beads for 24 hours or chronically, transduced with the CAR-gene, and expanded until CD8+ T cells had more than 100-fold expansion. The number of T cells was counted every 2 days, and the CD4+/CD8+ T cell ratio was analyzed by flow cytometry. The mice were irradiated with 1.5 Gy prior to T cell infusion. The incidence of GVHD was monitored daily, and the mice were sacrificed when they exhibited one of the following symptoms: more than 20% loss of the initial body weight, pronounced lethargy, hunched posture, severe diarrhea, or severe dermatitis. In the leukemia treatment model, 5 million NALM6-GL cells were intravenously injected into NSG mice. CD3+ T cells were retrovirally transduced with a second-generation anti-CD19 CAR gene (FMC63 anti-CD19 scFv linked with the CD28 and CD3ζ intracellular domains) and transplanted into the mice 10 days following NALM6-GL infusion. Imaging of the tumor cell localization was performed with a Xenogen XPM-2 (Perkin Elmer). Leukemic mice were randomly assigned to treatment groups in each experiment.
Retroviral transduction of T cells.
PG13 packaging cells (American Type Culture Collection) stably transduced with the pMX retroviral plasmid encoding the CAR construct were used for the infection of T cells. The pMX retrovirus vector was provided by Toshio Kitamura (University of Tokyo, Tokyo, Japan). Transduction was performed 2 days following anti–CD3/CD28 bead stimulation for 3 consecutive days. Polybrene was added at a concentration of 1 μg/ml during the transduction.
Statistics.
The statistical significance of differences between 2 groups was assessed with a 2-tailed paired or unpaired t test. Comparisons of more than 2 groups were performed by ANOVA with multiple comparison tests. The frequency of CAR-T cells in the peripheral blood was analyzed by the Kruskal-Wallis test (nonparametric ANOVA) followed by Dunn’s multiple comparison analysis at each time point. Differences were considered statistically significant at a P value of less than 0.05. In mouse experiments, the weight-loss-free survival and overall survival of the mice were depicted by a Kaplan-Meier curve, and the survival difference between groups was compared with a log-rank test. When more than 2 groups were compared, the P value was adjusted by the number of comparisons. All statistical analysis was performed using GraphPad Prism 6. No statistical method was used to predetermine the sample size.
Study approval.
This study was performed in accordance with the Helsinki Declaration and approved by the Research Ethics Board of University Health Network, Toronto, Canada. Written informed consent was obtained from all healthy donors who provided peripheral blood samples. All animal experiments were approved by the Ontario Cancer Institute/Princess Margaret Cancer Centre Animal Care Committee at the University Health Network.
Author contributions
YK and NH designed the project. YK, MN, TO, YC, KS, TG, and MA performed the experiments. MOB provided critical samples. YK and NH analyzed the results and wrote the manuscript.
Supplementary Material
Acknowledgments
This work was supported by the following grants and fellowships: Japan Society for the Promotion of Science Postdoctoral Fellowship for Overseas Researchers (to YK); the Princess Margaret Cancer Foundation (to MOB and NH); NIH grant R01 CA148673 (to NH); and Ontario Institute for Cancer Research Clinical Investigator Award IA-039 (to NH).
Footnotes
Conflict of interest: The authors have declared that no conflict of interest exists.
Reference information:JCI Insight. 2017;2(2):e89580. doi:10.1172/jci.insight.89580.
Contributor Information
Yuki Kagoya, Email: ykagoya-tky@umin.ac.jp.
Munehide Nakatsugawa, Email: munakatsu@gmail.com.
Toshiki Ochi, Email: occhie_34@hotmail.com.
Yuchen Cen, Email: samcen2003@yahoo.ca.
Tingxi Guo, Email: tguo@uhnres.utoronto.ca.
Mark Anczurowski, Email: mark.anczurowski@mail.utoronto.ca.
Kayoko Saso, Email: ksaso@uhnresearch.ca.
Naoto Hirano, Email: naoto.hirano@utoronto.ca.
References
- 1.Restifo NP, Dudley ME, Rosenberg SA. Adoptive immunotherapy for cancer: harnessing the T cell response. Nat Rev Immunol. 2012;12(4):269–281. doi: 10.1038/nri3191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Maus MV, Fraietta JA, Levine BL, Kalos M, Zhao Y, June CH. Adoptive immunotherapy for cancer or viruses. Annu Rev Immunol. 2014;32:189–225. doi: 10.1146/annurev-immunol-032713-120136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Robbins PF, et al. Tumor regression in patients with metastatic synovial cell sarcoma and melanoma using genetically engineered lymphocytes reactive with NY-ESO-1. J Clin Oncol. 2011;29(7):917–924. doi: 10.1200/JCO.2010.32.2537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Besser MJ, et al. Adoptive transfer of tumor-infiltrating lymphocytes in patients with metastatic melanoma: intent-to-treat analysis and efficacy after failure to prior immunotherapies. Clin Cancer Res. 2013;19(17):4792–4800. doi: 10.1158/1078-0432.CCR-13-0380. [DOI] [PubMed] [Google Scholar]
- 5.Rosenberg SA, et al. Durable complete responses in heavily pretreated patients with metastatic melanoma using T-cell transfer immunotherapy. Clin Cancer Res. 2011;17(13):4550–4557. doi: 10.1158/1078-0432.CCR-11-0116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Brentjens RJ, et al. CD19-targeted T cells rapidly induce molecular remissions in adults with chemotherapy-refractory acute lymphoblastic leukemia. Sci Transl Med. 2013;5(177):177ra38. doi: 10.1126/scitranslmed.3005930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Maude SL, et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. N Engl J Med. 2014;371(16):1507–1517. doi: 10.1056/NEJMoa1407222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Porter DL, et al. Chimeric antigen receptor T cells persist and induce sustained remissions in relapsed refractory chronic lymphocytic leukemia. Sci Transl Med. 2015;7(303):303ra139. doi: 10.1126/scitranslmed.aac5415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Goff SL, et al. Randomized, prospective evaluation comparing intensity of lymphodepletion before adoptive transfer of tumor-infiltrating lymphocytes for patients with metastatic melanoma. J Clin Oncol. 2016;34(20):2389–2397. doi: 10.1200/JCO.2016.66.7220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Robbins PF, et al. Cutting edge: persistence of transferred lymphocyte clonotypes correlates with cancer regression in patients receiving cell transfer therapy. J Immunol. 2004;173(12):7125–7130. doi: 10.4049/jimmunol.173.12.7125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Louis CU, et al. Antitumor activity and long-term fate of chimeric antigen receptor-positive T cells in patients with neuroblastoma. Blood. 2011;118(23):6050–6056. doi: 10.1182/blood-2011-05-354449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Xu Y, et al. Closely related T-memory stem cells correlate with in vivo expansion of CAR.CD19-T cells and are preserved by IL-7 and IL-15. Blood. 2014;123(24):3750–3759. doi: 10.1182/blood-2014-01-552174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gattinoni L, et al. A human memory T cell subset with stem cell-like properties. Nat Med. 2011;17(10):1290–1297. doi: 10.1038/nm.2446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cieri N, et al. IL-7 and IL-15 instruct the generation of human memory stem T cells from naive precursors. Blood. 2013;121(4):573–584. doi: 10.1182/blood-2012-05-431718. [DOI] [PubMed] [Google Scholar]
- 15.Klebanoff CA, et al. Determinants of successful CD8+ T-cell adoptive immunotherapy for large established tumors in mice. Clin Cancer Res. 2011;17(16):5343–5352. doi: 10.1158/1078-0432.CCR-11-0503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gattinoni L, Klebanoff CA, Restifo NP. Paths to stemness: building the ultimate antitumour T cell. Nat Rev Cancer. 2012;12(10):671–684. doi: 10.1038/nrc3322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lugli E, et al. Superior T memory stem cell persistence supports long-lived T cell memory. J Clin Invest. 2013;123(2):594–599. doi: 10.1172/JCI66327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Klebanoff CA, et al. Memory T cell-driven differentiation of naive cells impairs adoptive immunotherapy. J Clin Invest. 2016;126(1):318–334. doi: 10.1172/JCI81217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kershaw MH, Westwood JA, Darcy PK. Gene-engineered T cells for cancer therapy. Nat Rev Cancer. 2013;13(8):525–541. doi: 10.1038/nrc3565. [DOI] [PubMed] [Google Scholar]
- 20.Levine BL, et al. Effects of CD28 costimulation on long-term proliferation of CD4+ T cells in the absence of exogenous feeder cells. J Immunol. 1997;159(12):5921–5930. [PubMed] [Google Scholar]
- 21.Laux I, et al. Response differences between human CD4(+) and CD8(+) T-cells during CD28 costimulation: implications for immune cell-based therapies and studies related to the expansion of double-positive T-cells during aging. Clin Immunol. 2000;96(3):187–197. doi: 10.1006/clim.2000.4902. [DOI] [PubMed] [Google Scholar]
- 22.Maus MV, et al. Ex vivo expansion of polyclonal and antigen-specific cytotoxic T lymphocytes by artificial APCs expressing ligands for the T-cell receptor, CD28 and 4-1BB. Nat Biotechnol. 2002;20(2):143–148. doi: 10.1038/nbt0202-143. [DOI] [PubMed] [Google Scholar]
- 23.Li Y, Kurlander RJ. Comparison of anti-CD3 and anti-CD28-coated beads with soluble anti-CD3 for expanding human T cells: differing impact on CD8 T cell phenotype and responsiveness to restimulation. J Transl Med. 2010;8:104. doi: 10.1186/1479-5876-8-104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Thomas AK, Maus MV, Shalaby WS, June CH, Riley JL. A cell-based artificial antigen-presenting cell coated with anti-CD3 and CD28 antibodies enables rapid expansion and long-term growth of CD4 T lymphocytes. Clin Immunol. 2002;105(3):259–272. doi: 10.1006/clim.2002.5277. [DOI] [PubMed] [Google Scholar]
- 25.Suhoski MM, et al. Engineering artificial antigen-presenting cells to express a diverse array of co-stimulatory molecules. Mol Ther. 2007;15(5):981–988. doi: 10.1038/mt.sj.6300134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Latouche JB, Sadelain M. Induction of human cytotoxic T lymphocytes by artificial antigen-presenting cells. Nat Biotechnol. 2000;18(4):405–409. doi: 10.1038/74455. [DOI] [PubMed] [Google Scholar]
- 27.Hirano N, et al. Engagement of CD83 ligand induces prolonged expansion of CD8+ T cells and preferential enrichment for antigen specificity. Blood. 2006;107(4):1528–1536. doi: 10.1182/blood-2005-05-2073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Butler MO, et al. Ex vivo expansion of human CD8+ T cells using autologous CD4+ T cell help. PLoS ONE. 2012;7(1):e30229. doi: 10.1371/journal.pone.0030229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Nicholson IC, et al. Construction and characterisation of a functional CD19 specific single chain Fv fragment for immunotherapy of B lineage leukaemia and lymphoma. Mol Immunol. 1997;34(16–17):1157–1165. doi: 10.1016/s0161-5890(97)00144-2. [DOI] [PubMed] [Google Scholar]
- 30.Long AH, et al. 4-1BB costimulation ameliorates T cell exhaustion induced by tonic signaling of chimeric antigen receptors. Nat Med. 2015;21(6):581–590. doi: 10.1038/nm.3838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Malek TR. The biology of interleukin-2. Annu Rev Immunol. 2008;26:453–479. doi: 10.1146/annurev.immunol.26.021607.090357. [DOI] [PubMed] [Google Scholar]
- 32.Kagoya Y, et al. BET bromodomain inhibition enhances T cell persistence and function in adoptive immunotherapy models. J Clin Invest. 2016;126(9):3479–3494. doi: 10.1172/JCI86437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hinrichs CS, et al. IL-2 and IL-21 confer opposing differentiation programs to CD8+ T cells for adoptive immunotherapy. Blood. 2008;111(11):5326–5333. doi: 10.1182/blood-2007-09-113050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zeng R, et al. Synergy of IL-21 and IL-15 in regulating CD8+ T cell expansion and function. J Exp Med. 2005;201(1):139–148. doi: 10.1084/jem.20041057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Klebanoff CA, et al. IL-15 enhances the in vivo antitumor activity of tumor-reactive CD8+ T cells. Proc Natl Acad Sci USA. 2004;101(7):1969–1974. doi: 10.1073/pnas.0307298101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhang H, et al. 4-1BB is superior to CD28 costimulation for generating CD8+ cytotoxic lymphocytes for adoptive immunotherapy. J Immunol. 2007;179(7):4910–4918. doi: 10.4049/jimmunol.179.7.4910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Rabenstein H, et al. Differential kinetics of antigen dependency of CD4+ and CD8+ T cells. J Immunol. 2014;192(8):3507–3517. doi: 10.4049/jimmunol.1302725. [DOI] [PubMed] [Google Scholar]
- 38.Foulds KE, Zenewicz LA, Shedlock DJ, Jiang J, Troy AE, Shen H. Cutting edge: CD4 and CD8 T cells are intrinsically different in their proliferative responses. J Immunol. 2002;168(4):1528–1532. doi: 10.4049/jimmunol.168.4.1528. [DOI] [PubMed] [Google Scholar]
- 39.Wherry EJ, Kurachi M. Molecular and cellular insights into T cell exhaustion. Nat Rev Immunol. 2015;15(8):486–499. doi: 10.1038/nri3862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wherry EJ. T cell exhaustion. Nat Immunol. 2011;12(6):492–499. doi: 10.1038/ni.2035. [DOI] [PubMed] [Google Scholar]
- 41.Baitsch L, et al. Exhaustion of tumor-specific CD8+ T cells in metastases from melanoma patients. J Clin Invest. 2011;121(6):2350–2360. doi: 10.1172/JCI46102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Jelley-Gibbs DM, Dibble JP, Filipson S, Haynes L, Kemp RA, Swain SL. Repeated stimulation of CD4 effector T cells can limit their protective function. J Exp Med. 2005;201(7):1101–1112. doi: 10.1084/jem.20041852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Janelle V, Carli C, Taillefer J, Orio J, Delisle JS. Defining novel parameters for the optimal priming and expansion of minor histocompatibility antigen-specific T cells in culture. J Transl Med. 2015;13:123. doi: 10.1186/s12967-015-0495-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ho WY, Nguyen HN, Wolfl M, Kuball J, Greenberg PD. In vitro methods for generating CD8+ T-cell clones for immunotherapy from the naïve repertoire. J Immunol Methods. 2006;310(1–2):40–52. doi: 10.1016/j.jim.2005.11.023. [DOI] [PubMed] [Google Scholar]
- 45.Kawalekar OU, et al. Distinct signaling of coreceptors regulates specific metabolism pathways and impacts memory development in CAR T cells. Immunity. 2016;44(2):380–390. doi: 10.1016/j.immuni.2016.01.021. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.