Abstract
Background
Delayed hypersensitivity plays a large role in the pathogenesis of tuberculous pleural effusion (TPE). Macrophages infected with live Mycobacterium tuberculosis (MTB) increase the levels of adenosine deaminase2 (ADA2) in the pleural fluid of TPE patients. However, it is as yet unclear whether ADA2 can be produced by macrophages when challenged with MTB antigens alone. This study therefore evaluated the levels of ADA2 mRNA expression, using monocyte-derived macrophages (MDMs) stimulated with MTB antigens.
Methods
Purified monocytes from the peripheral blood mononuclear cells of healthy volunteers were differentiated into macrophages using granulocyte-macrophage colony-stimulating factor (GM-CSF) or macrophage colony-stimulating factor (M-CSF). The MDMs were stimulated with early secretory antigenic target protein 6 (ESAT6) and culture filtrate protein 10 (CFP10). The mRNA expression levels for the cat eye syndrome chromosome region, candidate 1 (CECR1) gene encoding ADA2 were then measured.
Results
CECR1 mRNA expression levels were significantly higher in MDMs stimulated with ESAT6 and CFP10, than in the unstimulated MDMs. When stimulated with ESAT6, M-CSF-treated MDMs showed more pronounced CECR1 mRNA expression than GM-CSF-treated MDMs. Interferon-γ decreased the ESAT6- and CFP10-induced CECR1 mRNA expression in MDMs. CECR1 mRNA expression levels were positively correlated with mRNA expression of tumor necrosis factor α and interleukin 10, respectively.
Conclusion
ADA2 mRNA expression increased when MDMs were stimulated with MTB antigens alone. This partly indicates that pleural fluid ADA levels could increase in patients with culture-negative TPE. Our results may be helpful in improving the understanding of TPE pathogenesis.
Keywords: Adenosine; Cat Eye Syndrome Chromosome Region, Candidate 1 Protein, Human; Mycobacterium tuberculosis Antigen; Macrophage
Introduction
It is well known that delayed hypersensitivity plays a large role in the pathogenesis of tuberculous pleural effusion (TPE)1,2. After acquiring adaptive immunity to Mycobacterium tuberculosis (MTB) infection, MTB proteins exposed to the pleural space initiates this delayed hypersensitivity reaction 3,4,5. This is supported by the observation that the positive MTB culture rate from the pleural fluid in patients with TPE is significantly lower than that from the sputum in those with pulmonary tuberculosis (TB).
This paucibacillary characteristic of TPE often makes diagnosis difficult. As an alternative tool for mycobacterial culture, adenosine deaminase (ADA) activity has been widely used to diagnose TPE in TB-prevalent areas2,6. Among the two different forms of ADA, ADA1, and ADA2, ADA2 is elevated in TPE. In addition, ADA2 is known to be produced by live MTB-infected macrophages7,8,9.
Given the classical delayed hypersensitivity hypothesis, it is difficult to explain the observation that pleural fluid ADA2 levels are elevated in most patients with MTB culture-negative TPE. Host immune responses to live MTB may differ from those to MTB antigens because they show different virulence as well as processing and presentation of antigens10,11. However, whether ADA2 can also be produced when MTB antigens alone, not live MTB, encounter the macrophages in the pleural space remains unknown. Therefore, we examined whether ADA2 expression can be induced by MTB-specific antigen-stimulated macrophages. We evaluated the mRNA expression levels of the cat eye syndrome chromosome region, candidate 1 (CECR1) gene encoding ADA2 in monocyte-derived macrophages (MDMs) stimulated with MTB-specific antigens (early secretory antigenic target protein 6 [ESAT6] and culture filtrate protein 10 [CFP10]).
Materials and Methods
The study protocols were reviewed and approved by the Institutional Review Board of the Kyungpook National University Hospital.
1. Isolation of human monocytes and differentiation into MDMs followed by MTB antigen stimulation
Peripheral blood mononuclear cells (PBMCs) were isolated from heparin-treated blood obtained from healthy volunteer donors using density gradient centrifugation (Ficoll-Paque; GE Healthcare Life Sciences, Marborough, MA, USA). PBMCs were suspended in complete RPMI 1640 medium (supplemented with 10% inactivated fetal bovine serum, 2 mM L-glutamine, 100 U/mL penicillin, and 100 g/mL streptomycin) at a concentration 2×106 cells/mL and 1 mL of the PBMC suspension was distributed into 24-well tissue culture plates (Costar, Cambridge, MA, USA). After incubation of 2 hours at 37℃ in a humidified 5% CO2 atmosphere, nonadherent cells were removed and the plates were washed five times with serum-free medium. Adherent monocytes were differentiated in the absence or presence of 50 ng/mL of macrophage colony-stimulating factor (M-CSF; R&D Systems, Minneapolis, MN, USA) or 50 ng/mL of granulocyte-macrophage colony-stimulating factor (GM-CSF; R&D Systems) for 7 days. The culture medium was replaced every third day. MDM were stimulated with ESAT6 (5 µg/mL) and CFP10 (5 µg/mL), which were purchased from MyBioSource (San Diego, CA, USA), for 12–18 hours. In cases treated with interferon-γ (IFN-γ; R&D Systems), 50 U/mL of IFN-γ was added at 16 hours before antigen stimulation.
2. THP-1 cell culture and MTB antigen stimulation
THP-1 cells, a human monocytic cell line, were cultured in complete RPMI-1640. The cells in complete medium were plated overnight at 1×106/well in a 12-well plate, and IFN-γ (50 U/mL) was treated to induce THP-1 activation. ESAT6 and CPF10 were added at 16 hours after IFN-γ stimulation.
3. Quantitative reverse transcriptase polymerase chain reaction
Total RNA was extracted from MDMs and THP-1 cells using an RNA extraction kit (Takara, Shiga, Japan) according to the manufacturer's protocol. Synthesis of first-strand complementary DNA was performed using the SuperScript III First-Strand Synthesis System (Invitrogen, Carlsbad, CA, USA). To evaluate mRNA expression levels, quantitative reverse transcriptase polymerase chain reaction was performed using SYBR Green Realtime PCR master Mix (Bio-Rad, Hercules, CA, USA) in a CFX96 Touch Real-Time PCR system (Bio-Rad). The following primers were used: human CECR1 gene: forward, 5′-CACACGCTGTCACTTCCTCT-3′; reverse, 5′-TCTCACAGGGTCGCTCCATA-3′); human interleukin 6 (IL-6) gene: forward, 5′-AAGCCAGAGCTGTGCAGATGAGTA-3′; reverse, 5′-TGTCCTGCAGCCACTGGTTC-3′; human tumor necrosis factor α (TNF-α) gene: forward, 5′-CCCAGGGACCTCTCTCTAATC-3′; reverse, 5′-ATGGGCTACAGGCTTGTCACT-3′; human interleukin 10 (IL-10) gene: forward, 5′-GAGATGCCTTCAGCAGAGTGAAGA-3′; reverse, 5′-AGTTCACATGCGCCTTGATGTC-3′; human β-actin gene: forward, 5′-GGACTTCGAGCAAGAGATGG-3′, reverse, 5′-AGCACTGTGTTGGCGTACAG-3′. Reactions were conducted in duplicate and data were analyzed using the 2-ΔΔcomparative threshold method. mRNA expression levels were normalized to the internal control β-actin and presented as fold-induction, normalized to unstimulated cells.
4. Statistical analysis
Statistical analyses were performed using GraphPad PRISM software (GraphPad Software Inc., San Diego, CA, USA). The Wilcoxon-paired signed rank test was used to compare data between the different simulations in each individual and Spearman correlation (ρ) analysis was used to examine the correlation between multiple cytokines. A p-value <0.05 was considered significant.
Results
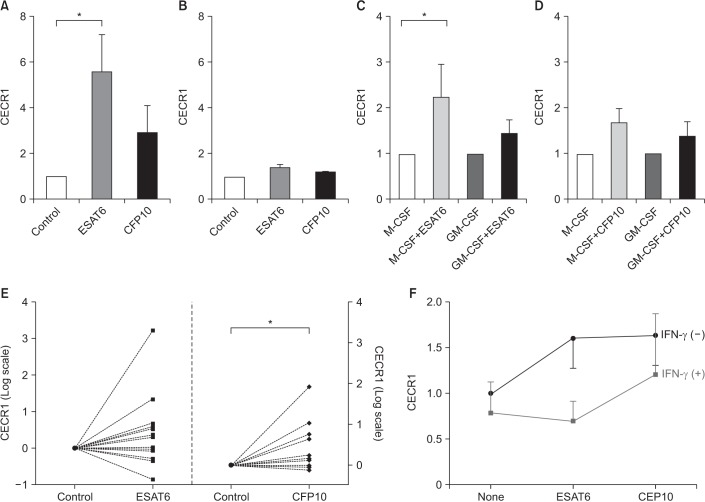
A total of 19 healthy volunteers with no known health problems were recruited and included 11 males and eight females. Their median age was 38 years. All of them had no symptoms and none of them was on medication. Figure 1 shows the relative levels of CECR1 mRNA expression in response to ESAT6 and CFP10 compared to controls. Pilot experiments were conducted using cytokine-untreated MDM and THP-1 cells (Figure 1A, B). CECR1 mRNA expression levels were significantly higher in ESAT6-stimulated MDMs than in unstimulated MDMs (Figure 1A). CECR1 was also more abundant in CFP10-stimulated MDMs than in unstimulated MDMs, but the difference was not significant. In THP-1 cells stimulated with ESAT6 and CFP10, CECR1 expression was slightly higher than in unstimulated THP-1 cells (Figure 1B).
Figure 1. ESAT6 and CFP10 stimulation induces increased CECR1 mRNA expression in MDMs. (A) Monocytes isolated from human peripheral blood mononuclear cells were differentiated into MDMs in the absence of cytokines for 7 days, and then cultured with medium alone (control), ESAT6 (5 µg/mL) and CFP10 (5 µg/mL) for 12–18 hours (n=3). CECR1 mRNA expression levels were measured using real-time reverse transcription-polymerase chain reaction and normalized to the internal control human β-actin gene. (B) THP-1 cells were incubated with medium alone, ESAT6 (5 µg/mL) and CFP10 (5 µg/mL). Data was expressed for three independent experiments. (C, D) Monocytes were differentiated into MDMs in the presence of M-CSF (50 ng/mL) or GM-CSF (50 ng/mL) for 7 days, following which they were incubated with medium alone (control), ESAT6 (5 µg/mL), and CFP10 (5 µg/mL) for 12–18 hours (n=6). (E) M-CSF–treated MDMs from 13 different individuals were stimulated with either ESAT6 (5 µg/mL) or CFP10 (5 µg/mL) for 12–18 hours. (F) CECR1 mRNA expression levels in response to ESAT6 or CFP10 stimulation were compared in the presence and absence of IFN-γ (50 U/mL) (n=4). Each column and bar represent the mean and standard error values in Figure 1A–D. Differences were analyzed by Wilcoxon-paired signed rank test, *p<0.05. ESAT6: early secretory antigenic target protein 6; CFP10: culture filtrate protein 10; CECR1: cat eye syndrome chromosome region, candidate 1; MDM: monocyte-derived macrophage; M-CSF: macrophage colony-stimulating factor; GM-CSF: granulocyte-macrophage colony-stimulating factor; IFN-γ: interferon γ.
Next, the effect of different MDM phenotypes on CECR1 expression was examined (Figure 1C, D). In M-CSF–treated MDMs, ESAT6 stimulation resulted in significantly higher CECR1 mRNA expression compared to in controls (p<0.05) (Figure 1C). In GM-CSF–treated MDMs, CECR1 mRNA expression levels following ESAT6 stimulation were slightly higher than in controls. For CFP10 stimulation, CECR1 expression was higher than control in M-CSF–treated MDMs and GM-CSF–treated MDMs, respectively, but the difference was not significant (Figure 1D).
To verify the increased CECR1 expression in response to ESAT6 and CFP10 stimulation, further experiments were conducted in 13 different individuals using M-CSF–treated MDMs. CECR1 mRNA expression levels were significantly increased when MDMs were stimulated with CFP10 (p<0.05) (Figure 1E). However, we observed inter-individual variability in terms of CECR1 expression in response to ESAT6.
Next, we evaluated whether IFN-γ affects CECR1 expression in MDMs stimulated with MTB-specific antigens because IFN-γ, a main cytokine involved in the immune response against TB infection, is also elevated in the pleural fluid of patients with TPE12. CECR1 mRNA expression levels in response to both ESAT6 and CFP10 stimulation were decreased by IFN-γ addition (Figure 1F).
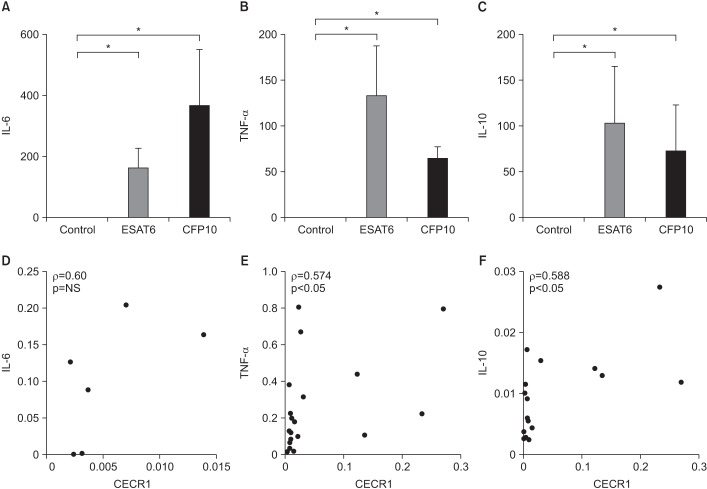
Figure 2A–C shows the mRNA expression levels of IL-6, TNF-α, and IL-10 in ESAT6- and CFP10-stimulated MDMs. All were significantly elevated in response to ESAT6 and CFP10 stimulation compared to in controls (p<0.05). Figure 2D–F shows the correlation between mRNA expression levels of each cytokine and CECR1 expression. There were positive correlations between CECR1 and IL-6, CECR1 and TNF-α, and CECR1 and IL-10 mRNA expression levels, and statistically significant correlation was detected between CECR1 and TNF-α as well as between CECR1 and IL-10.
Figure 2. Cytokine expression levels in MDMs in response to ESAT6 and CFP10 stimulation (A and D, n=3; B, C, E, and F, n=8). (A–C) IL-6, TNF-α, and IL-10 mRNA expression levels were significantly higher in MDMs stimulated with ESAT6 and CFP10 than in controls. The human β-actin gene served as an internal control. (D–F) There were significantly positive correlations between CECR1 and TNF-α, as well as CECR1 and IL-10 mRNA expression levels. Each column and bar represent the mean and standard error values in Figure 2A–C. Differences were analyzed by Wilcoxon-paired signed rank test in Figure 2A–C and by Spearman correlation (ρ) analysis in Figure 2D–F, *p<0.05. NS: not significant; MDM: monocyte-derived macrophage; ESAT6: early secretory antigenic target protein 6; CFP10: culture filtrate protein 10; IL-6: interleukin 6; TNF-α: tumor necrosis factor α; IL-10: interleukin 10; CECR1: cat eye syndrome chromosome region, candidate 1.
Discussion
The main findings of this study are as follows: (1) CECR1 mRNA expression levels were significantly higher in MDMs stimulated with ESAT6 and CFP10 than in unstimulated MDMs, although there was considerable inter-individual variability. (2) For ESAT6 stimulation, M-CSF–treated MDMs showed more pronounced CECR1 mRNA expression compared to GM-CSF–treated MDMs. (3) IFN-γ decreased EAST6- and CFP10-induced CECR1 mRNA expression by MDMs. (4) CECR1 mRNA expression levels were significantly positively correlated with TNF-α and IL-10 mRNA expression, respectively.
ADA2 levels are very low under physiological conditions, but are elevated when monocyte-macrophages are infected with intracellular microorganisms such as human immunodeficiency virus or MTB13,14. ADA2 is thought to regulate the concentration of 2′-deoxyadenosine, which is part of an innate response against offending intracellular microorganisms15. Few in-vitro studies have detected elevated ADA production in THP-1 and MDM cells infected with live MTB8,9. This is the first study to show that ADA2 is produced when the macrophages are exposed to MTB antigens, which is similar to the results following exposure to live MTB. This experimental finding can help explain why pleural fluid ADA2 levels are elevated even in culture-negative TPE patients, similarly to in culture-positive TPE patients.
Increased ADA2 expression in response to at least one of two MTB antigens, ESAT6 and CFP10, was identified from 11 of 13 donors (85%) in the present study (Figure 1E). However, substantial inter-individual variations in ADA2 expression under the same stimulation were observed. Some donors showed strong ADA2 expression (more than a 100-fold increase) compared to controls, while others revealed slightly elevated ADA2 expression. This agrees with the clinical findings that ADA levels vary among TPE cases, some of whom show ADA levels lower than the usual diagnostic threshold value for TPE. Factors associated with ADA levels in the pleural fluid of TPE patients remains unclear although a few previous studies have examined this9,16,17. Genetic factors may influence ADA2 levels in different individuals. Meanwhile, two out of 13 donors showed decreased ADA2 expression in response to both ESAT6 and CFP10 stimulation. It is difficult to explain whether this result was affected by experimental process or by other host factors. It is contemplated that ESAT6 and CFP10 used in this study cannot reflect antigenic diversity of MTB present in TPE patients. Thus, other MTB antigens unused in this study might induce increased ADA2 expression in them.
We found that IFN-γ treatment attenuated ADA2 expression by MDMs stimulated with EAST6 and CFP10. Our result agrees with that of Zavialov et al.18 who showed that ADA2 secretion during differentiation of monocytes into macrophages was decreased by IFN-γ addition. They suggested that IFN-γ may act as a feedback mechanism for the regulation of ADA2 expression during monocyte differentiatioin18, as other cytokines and growth factors. Given that IFN-γ plays a crucial role in TB infection and is highly elevated in the pleural fluid of TPE patients, these findings suggest that the level of ADA2 secretion by MTB-infected or MTB antigens-stimulated macrophages is regulated by different IFN-γ concentrations at the disease site. Further studies are required to examine the feedback mechanism of IFN-γ in the regulation of pleural fluid ADA2 levels in TPE patients.
How different macrophage phenotypes contribute to the immune response to MTB infection remains poorly understood. However, several studies indicate that each phenotype plays an opposing role in cell-mediated immunity to MTB infection and predominates at different stages in the immune response19,20. Our finding that M-CSF–treated MDMs expressed higher levels of ADA2 than GM-CSF–treated MDMs suggests that ADA2 levels are associated with the pleural microenvironment in TPE patients. In addition, ADA2 mRNA expression levels were positively correlated with IL-6, TNF-α, and IL-10, which are key pro-inflammatory or anti-inflammatory cytokines involved in the innate immune response to MTB infection and are highly expressed at the site of infection. Although the function of ADA2 and its regulatory mechanism in MTB infection remains poorly characterized, ADA2 is likely to be involved in innate immune responses against MTB infection.
The present study has several limitations. First, MDMs derived from the peripheral blood of healthy donor may not completely reflect the characteristics of pleural macrophages. Further studies should use macrophages from human pleural fluid. Second, we did not evaluate the differences between in-vitro and in-vivo microenvironments or cytokine cross-talk between T-cells and macrophages, which may determine final ADA levels in patients with TPE. Thus, these findings cannot be fully extrapolated to patients with TPE.
In conclusion, the present study showed that ADA2 mRNA expression was increased when MDMs were stimulated with MTB antigens alone. This finding partly explains the observation that pleural fluid ADA levels are increased in patients with culture-negative TPE. These results may be helpful in improving our understanding of TPE pathogenesis.
Acknowledgments
This study was supported by a 2015-Grant from the Korean Academy of Tuberculosis and Respiratory Diseases and by the National Research Foundation of Korea (NRF) grant funded by the Korea government (2015R1C1A2A01052502).
Footnotes
Conflicts of Interest: No potential conflict of interest relevant to this article was reported.
References
- 1.Light RW. Update on tuberculous pleural effusion. Respirology. 2010;15:451–458. doi: 10.1111/j.1440-1843.2010.01723.x. [DOI] [PubMed] [Google Scholar]
- 2.Jeon D. Tuberculous pleurisy: an update. Tuberc Respir Dis. 2014;76:153–159. doi: 10.4046/trd.2014.76.4.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Allen JC, Apicella MA. Experimental pleural effusion as a manifestation of delayed hypersensitivity to tuberculin PPD. J Immunol. 1968;101:481–487. [PubMed] [Google Scholar]
- 4.Leibowitz S, Kennedy L, Lessof MH. The tuberculin reaction in the pleural cavity and its suppression by antilymphocyte serum. Br J Exp Pathol. 1973;54:152–162. [PMC free article] [PubMed] [Google Scholar]
- 5.Yamamoto S, Dunn CJ, Willoughby DA. Studies on delayed hypersensitivity pleural exudates in guinea-pigs. II. The interrelationship of monocytic and lymphocytic cells with respect to migration activity. Immunology. 1976;30:513–519. [PMC free article] [PubMed] [Google Scholar]
- 6.Lee JY. Diagnosis and treatment of extrapulmonary tuberculosis. Tuberc Respir Dis. 2015;78:47–55. doi: 10.4046/trd.2015.78.2.47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gakis C. Adenosine deaminase (ADA) isoenzymes ADA1 and ADA2: diagnostic and biological role. Eur Respir J. 1996;9:632–633. doi: 10.1183/09031936.96.09040632. [DOI] [PubMed] [Google Scholar]
- 8.Kashyap RS, Deshpande PS, Nayak AR, Purohit HJ, Taori GM, Daginawala HF. Adenosine deaminase activity in the supernatant of monocytes infected with Mycobacterium tuberculosis. Int J Integr Biol. 2007;1:61–64. [Google Scholar]
- 9.Kim CH, Lee J, Lee J, Cliff JM, Toulza F, Smith S, et al. Mycobacterial load affects adenosine deaminase 2 levels of tuberculous pleural effusion. J Infect. 2015;71:488–491. doi: 10.1016/j.jinf.2015.05.015. [DOI] [PubMed] [Google Scholar]
- 10.Hasan Z, Jamil B, Ashraf M, Islam M, Dojki M, Irfan M, et al. Differential live Mycobacterium tuberculosis-, M. bovis BCG-, recombinant ESAT6-, and culture filtrate protein 10-induced immunity in tuberculosis. Clin Vaccine Immunol. 2009;16:991–998. doi: 10.1128/CVI.00091-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hasan Z, Schlax C, Kuhn L, Lefkovits I, Young D, Thole J, et al. Isolation and characterization of the mycobacterial phagosome: segregation from the endosomal/lysosomal pathway. Mol Microbiol. 1997;24:545–553. doi: 10.1046/j.1365-2958.1997.3591731.x. [DOI] [PubMed] [Google Scholar]
- 12.Wongtim S, Silachamroon U, Ruxrungtham K, Udompanich V, Limthongkul S, Charoenlap P, et al. Interferon gamma for diagnosing tuberculous pleural effusions. Thorax. 1999;54:921–924. doi: 10.1136/thx.54.10.921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Piras MA, Gakis C, Budroni M, Andreoni G. Adenosine deaminase activity in pleural effusions: an aid to differential diagnosis. Br Med J. 1978;2:1751–1752. doi: 10.1136/bmj.2.6154.1751-a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gakis C, Calia G, Naitana A, Pirino D, Serru G. Serum adenosine deaminase activity in HIV positive subjects: a hypothesis on the significance of ADA2. Panminerva Med. 1989;31:107–113. [PubMed] [Google Scholar]
- 15.Gakis C, Cappio-Borlino A, Pulina G. Enzymes (isoenzyme system) as homeostatic mechanisms the isoenzyme (ADA2) of adenosine deaminase of human monocytes-macrophages as a regulator of the 2’deoxyadenosine. Biochem Mol Biol Int. 1998;46:487–494. doi: 10.1080/15216549800204012. [DOI] [PubMed] [Google Scholar]
- 16.Tay TR, Tee A. Factors affecting pleural fluid adenosine deaminase level and the implication on the diagnosis of tuberculous pleural effusion: a retrospective cohort study. BMC Infect Dis. 2013;13:546. doi: 10.1186/1471-2334-13-546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lee SJ, Kim HS, Lee SH, Lee TW, Lee HR, Cho YJ, et al. Factors influencing pleural adenosine deaminase level in patients with tuberculous pleurisy. Am J Med Sci. 2014;348:362–365. doi: 10.1097/MAJ.0000000000000260. [DOI] [PubMed] [Google Scholar]
- 18.Zavialov AV, Gracia E, Glaichenhaus N, Franco R, Lauvau G. Human adenosine deaminase 2 induces differentiation of monocytes into macrophages and stimulates proliferation of T helper cells and macrophages. J Leukoc Biol. 2010;88:279–290. doi: 10.1189/jlb.1109764. [DOI] [PubMed] [Google Scholar]
- 19.Lugo-Villarino G, Verollet C, Maridonneau-Parini I, Neyrolles O. Macrophage polarization: convergence point targeted by Mycobacterium tuberculosis and HIV. Front Immunol. 2011;2:43. doi: 10.3389/fimmu.2011.00043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Verreck FA, de Boer T, Langenberg DM, Hoeve MA, Kramer M, Vaisberg E, et al. Human IL-23-producing type 1 macrophages promote but IL-10-producing type 2 macrophages subvert immunity to (myco)bacteria. Proc Natl Acad Sci U S A. 2004;101:4560–4565. doi: 10.1073/pnas.0400983101. [DOI] [PMC free article] [PubMed] [Google Scholar]