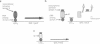Abstract
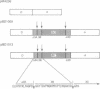
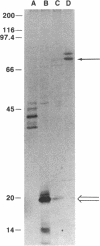
We describe a genetic system for monitoring the activity of a specific proteolytic enzyme by taking advantage of the properties of the yeast transcriptional activator GAL4. The GAL4 protein contains two separable and functionally essential domains: the amino-terminal DNA binding domain and the carboxyl-terminal transcriptional activating domain. We constructed two hybrid proteins by inserting between the DNA binding domain and the activation domain of GAL4 either (i) a self-cleaving protease (3C protease of a picornavirus, coxsackievirus B3) or (ii) a mutant form of the protease that is unable to cleave. We show that, although the hybrid protein containing the mutant protease activates transcription of GAL1-lacZ reporter gene, the hybrid protein bearing the wild-type protease is proteolytically cleaved and fails to activate transcription. Our approach to monitor the proteolytic activity could be used to develop simple genetic systems to study other proteases.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Billich A., Hammerschmid F., Winkler G. Purification, assay and kinetic features of HIV-1 proteinase. Biol Chem Hoppe Seyler. 1990 Mar;371(3):265–272. [PubMed] [Google Scholar]
- Dasmahapatra B., Rozhon E. J., Hart A. M., Cox S., Tracy S., Schwartz J. Cell-free expression of the coxsackievirus 3C protease using the translational initiation signal of an insect virus RNA and its characterization. Virus Res. 1991 Aug;20(3):237–249. doi: 10.1016/0168-1702(91)90078-a. [DOI] [PubMed] [Google Scholar]
- Geballe A. P., Spaete R. R., Mocarski E. S. A cis-acting element within the 5' leader of a cytomegalovirus beta transcript determines kinetic class. Cell. 1986 Sep 12;46(6):865–872. doi: 10.1016/0092-8674(86)90068-1. [DOI] [PubMed] [Google Scholar]
- Gill G., Ptashne M. Mutants of GAL4 protein altered in an activation function. Cell. 1987 Oct 9;51(1):121–126. doi: 10.1016/0092-8674(87)90016-x. [DOI] [PubMed] [Google Scholar]
- Gill G., Sadowski I., Ptashne M. Mutations that increase the activity of a transcriptional activator in yeast and mammalian cells. Proc Natl Acad Sci U S A. 1990 Mar;87(6):2127–2131. doi: 10.1073/pnas.87.6.2127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giniger E., Ptashne M. Transcription in yeast activated by a putative amphipathic alpha helix linked to a DNA binding unit. Nature. 1987 Dec 17;330(6149):670–672. doi: 10.1038/330670a0. [DOI] [PubMed] [Google Scholar]
- Hanecak R., Semler B. L., Ariga H., Anderson C. W., Wimmer E. Expression of a cloned gene segment of poliovirus in E. coli: evidence for autocatalytic production of the viral proteinase. Cell. 1984 Jul;37(3):1063–1073. doi: 10.1016/0092-8674(84)90441-0. [DOI] [PubMed] [Google Scholar]
- Ito H., Fukuda Y., Murata K., Kimura A. Transformation of intact yeast cells treated with alkali cations. J Bacteriol. 1983 Jan;153(1):163–168. doi: 10.1128/jb.153.1.163-168.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kakidani H., Ptashne M. GAL4 activates gene expression in mammalian cells. Cell. 1988 Jan 29;52(2):161–167. doi: 10.1016/0092-8674(88)90504-1. [DOI] [PubMed] [Google Scholar]
- Keegan L., Gill G., Ptashne M. Separation of DNA binding from the transcription-activating function of a eukaryotic regulatory protein. Science. 1986 Feb 14;231(4739):699–704. doi: 10.1126/science.3080805. [DOI] [PubMed] [Google Scholar]
- Kräusslich H. G., Wimmer E. Viral proteinases. Annu Rev Biochem. 1988;57:701–754. doi: 10.1146/annurev.bi.57.070188.003413. [DOI] [PubMed] [Google Scholar]
- Lindberg A. M., Stålhandske P. O., Pettersson U. Genome of coxsackievirus B3. Virology. 1987 Jan;156(1):50–63. doi: 10.1016/0042-6822(87)90435-1. [DOI] [PubMed] [Google Scholar]
- Loeb D. D., Swanstrom R., Everitt L., Manchester M., Stamper S. E., Hutchison C. A., 3rd Complete mutagenesis of the HIV-1 protease. Nature. 1989 Aug 3;340(6232):397–400. doi: 10.1038/340397a0. [DOI] [PubMed] [Google Scholar]
- Louis J. M., Wondrak E. M., Copeland T. D., Smith C. A., Mora P. T., Oroszlan S. Chemical synthesis and expression of the HIV-1 protease gene in E. coli. Biochem Biophys Res Commun. 1989 Feb 28;159(1):87–94. doi: 10.1016/0006-291x(89)92408-x. [DOI] [PubMed] [Google Scholar]
- Ma J., Ptashne M. Deletion analysis of GAL4 defines two transcriptional activating segments. Cell. 1987 Mar 13;48(5):847–853. doi: 10.1016/0092-8674(87)90081-x. [DOI] [PubMed] [Google Scholar]
- Mallon R., Borkowski J., Albin R., Pepitoni S., Schwartz J., Kieff E. The Epstein-Barr virus BZLF1 gene product activates the human immunodeficiency virus type 1 5' long terminal repeat. J Virol. 1990 Dec;64(12):6282–6285. doi: 10.1128/jvi.64.12.6282-6285.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matayoshi E. D., Wang G. T., Krafft G. A., Erickson J. Novel fluorogenic substrates for assaying retroviral proteases by resonance energy transfer. Science. 1990 Feb 23;247(4945):954–958. doi: 10.1126/science.2106161. [DOI] [PubMed] [Google Scholar]
- Neurath H. Evolution of proteolytic enzymes. Science. 1984 Apr 27;224(4647):350–357. doi: 10.1126/science.6369538. [DOI] [PubMed] [Google Scholar]
- Parks G. D., Duke G. M., Palmenberg A. C. Encephalomyocarditis virus 3C protease: efficient cell-free expression from clones which link viral 5' noncoding sequences to the P3 region. J Virol. 1986 Nov;60(2):376–384. doi: 10.1128/jvi.60.2.376-384.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pontremoli S., Melloni E. Extralysosomal protein degradation. Annu Rev Biochem. 1986;55:455–481. doi: 10.1146/annurev.bi.55.070186.002323. [DOI] [PubMed] [Google Scholar]
- Richards A. D., Phylip L. H., Farmerie W. G., Scarborough P. E., Alvarez A., Dunn B. M., Hirel P. H., Konvalinka J., Strop P., Pavlickova L. Sensitive, soluble chromogenic substrates for HIV-1 proteinase. J Biol Chem. 1990 May 15;265(14):7733–7736. [PubMed] [Google Scholar]
- Sadowski I., Niedbala D., Wood K., Ptashne M. GAL4 is phosphorylated as a consequence of transcriptional activation. Proc Natl Acad Sci U S A. 1991 Dec 1;88(23):10510–10514. doi: 10.1073/pnas.88.23.10510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith T. A., Kohorn B. D. Direct selection for sequences encoding proteases of known specificity. Proc Natl Acad Sci U S A. 1991 Jun 15;88(12):5159–5162. doi: 10.1073/pnas.88.12.5159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wellink J., van Kammen A. Proteases involved in the processing of viral polyproteins. Brief review. Arch Virol. 1988;98(1-2):1–26. doi: 10.1007/BF01321002. [DOI] [PubMed] [Google Scholar]
- Werner G., Rosenwirth B., Bauer E., Seifert J. M., Werner F. J., Besemer J. Molecular cloning and sequence determination of the genomic regions encoding protease and genome-linked protein of three picornaviruses. J Virol. 1986 Mar;57(3):1084–1093. doi: 10.1128/jvi.57.3.1084-1093.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Windheuser M. G., Dwyer S., Dasmahapatra B. Expression of functional beta-galactosidase containing the coxsackievirus 3C protease as an internal fusion. Biochem Biophys Res Commun. 1991 May 31;177(1):243–251. doi: 10.1016/0006-291x(91)91974-h. [DOI] [PubMed] [Google Scholar]